Abstract
Per- and polyfluoroalkyl substances (PFAS) are highly persistent synthetic organic contaminants that can cause serious human health concerns such as obesity, liver damage, kidney cancer, hypertension, immunotoxicity and other human health issues. Integrated crop–livestock systems combine agricultural crop production with milk and/or meat production and processing. Key sources of PFAS in these systems include firefighting foams near military bases, wastewater sludge and industrial discharge. Per- and polyfluoroalkyl substances regularly move from soils to nearby surface water and/or groundwater because of their high mobility and persistence. Irrigating crops or managing livestock for milk and meat production using adjacent waters can be detrimental to human health. The presence of PFAS in both groundwater and milk have been reported in dairy production states (e.g., Wisconsin and New Mexico) across the United States. Although there is a limit of 70 parts per trillion of PFAS in drinking water by the U.S. EPA, there are not yet regional screening guidelines for conducting risk assessments of livestock watering as well as the soil and plant matrix. This systematic review includes (i) the sources, impacts and challenges of PFAS in integrated crop–livestock systems, (ii) safety measures and protocols for sampling soil, water and plants for determining PFAS concentration in exposed integrated crop–livestock systems and (iii) the assessment, measurement and evaluation of human health risks related to PFAS exposure.
1. Introduction
The burgeoning global population has increased the need for maximizing food production while simultaneously minimizing its ecological footprint. Climate change challenges demand resilient, sustainable and economically competitive agricultural practices such as integrated crop–livestock systems. Integrated crop–livestock systems (ICLS) have played an important role in enhancing practices to improve soil health, nutrient cycling, pathological management, weed infestation and optimizing the nutrient losses from the system. We define ICLS as a combined system including plant and livestock components that have synergistic impacts on agricultural, economic and environmental outcomes. Here, we discuss ICLS in the context of environmental exposure pathways for contaminant mobility from sources such as soil and groundwater, which eventually affect crop biomass, livestock and dairy products, as well as human health. Managing and regulating contaminants in ICLS are a major challenge. In developed countries such as the United States, federal regulations, such as the National Pollutant Discharge Elimination System (NPDES), and state regulatory orders, keep track of exposure pathways in ICLS and ensure they remain below the allowable screening limits for environmental and human exposure. These screening and regulatory limits are defined for commonly occurring contaminants such as nitrate and metal(loid)s; however, the risk and extent of impacts from emerging contaminants such as per- and polyfluoroalkyl substance (PFAS) compounds are unknown and still require extensive research. Research and decisions regarding PFAS currently focus on regulatory studies for drinking water and food products. This paper highlights PFAS exposure in the environment as a holistic pathway affecting soils, water, plants, livestock and dairy products.
Per- and polyfluoroalkyl substance compounds are among the most pervasive environmental contaminants which bio accumulate, move long distances in the environment and persist through the food chain [1,2,3]. They are a class of fluorinated synthetic compounds with unique multiple carbon–fluorine bonds, which are highly polar and display amphiphilic properties [4]. Approximately 4700 of these compounds were developed in the late 1940s and are used in various industrial and commercial processes. Because of their strong carbon chain, in addition to fluorine atoms, they do not degrade easily and remain in the environment for a long time. Therefore, it is difficult to estimate the environmental half-life of these compounds. They originate from regular use and disposal of consumer products such as non-stick cookware, clothes and carpets resistant to stains and the formulation of aqueous fire-fighting foams. Fire-fighting foams are retardants used for fires and a variety of other industries, including aerospace, automotive, construction, electronics and military. In agroecosystems, abiotic or biotic degradation of PFAS release various other impurities into the environment commonly containing perfluorooctane sulfonic acid (PFOS), perfluorooctanoic acid (PFOAs), perfluoro hexane sulfonic acid (PFHxS), perfluorononanoic acid (PFNA), Perfluorodecanoic acid (PFDeA), and many more [5,6].
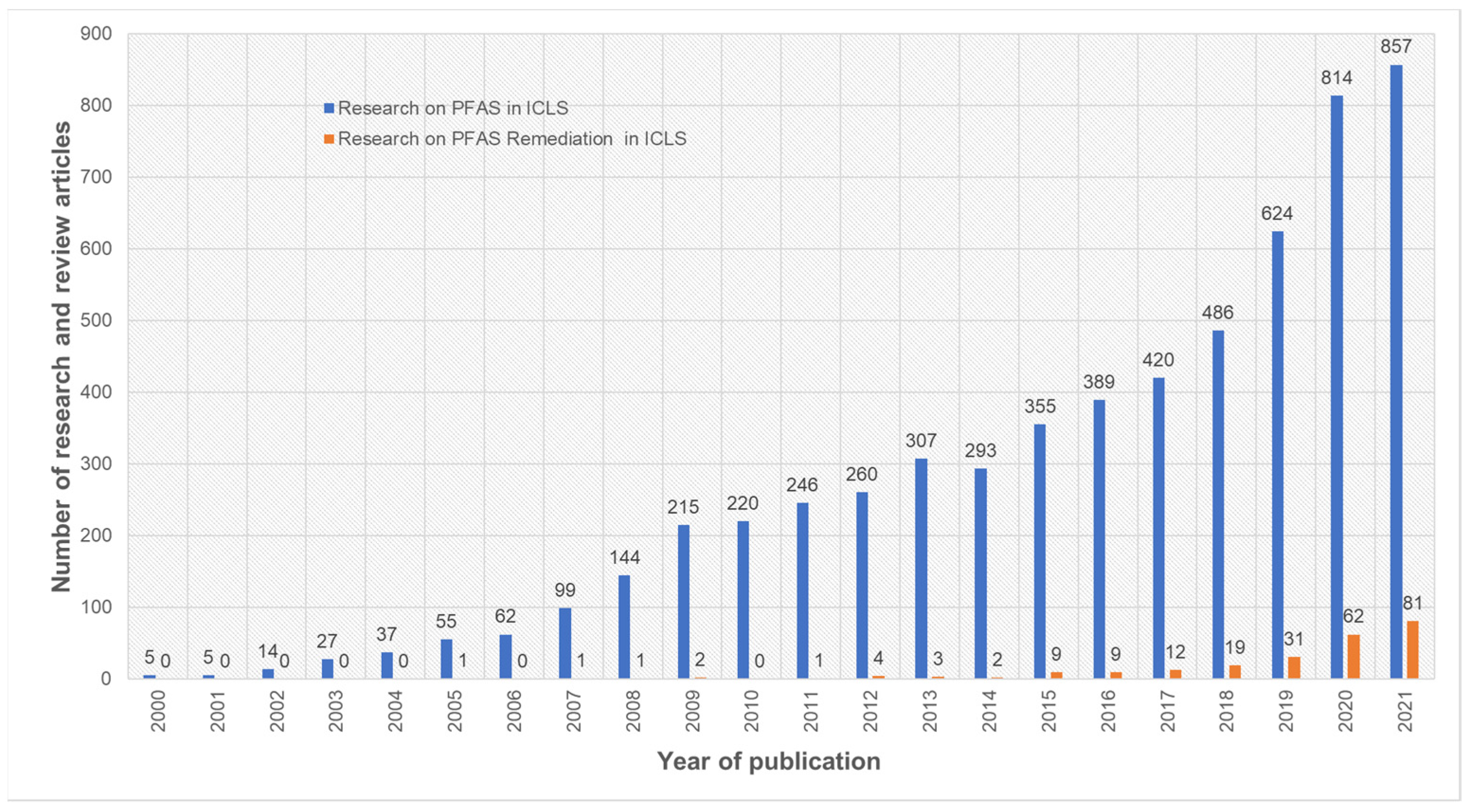
PFOS and PFOA are the contaminants commonly studied for their long-term persistence, and they are commonly detected in wastewater treatment, fresh water and ground water systems during environmental risk analysis [7,8,9,10,11,12]. Their sources of origin (Table 1) include industrial emissions, consumer products, contaminated drinking water, surface water and house dust [11,12]. We conducted a systematic analysis of published research and review articles over the last two decades (2000–2021) on 14 November 2021, using the Web of Science search engine. Our analysis indicated that the number of research articles published on PFAS in ICLS increased over the last two decades (Figure 1). More specifically, there was roughly a four-fold increase in PFAS research work in ICLS over the last decade. Moreover, we observed an increasing trend in published work on PFAS remediation in ICLS over the last nine years (2012–2021) based on our search criteria. However, the articles published on PFAS remediation in ICLS were limited in both spatial and temporal scales to laboratory experiments or other emerging techniques. Therefore, this emerging need for PFAS assessment and remediation in ICLS warrants an extensive review and discussion.

Table 1.
Summary of major point and nonpoint production and manufacturing sources of PFAS released to the environment.
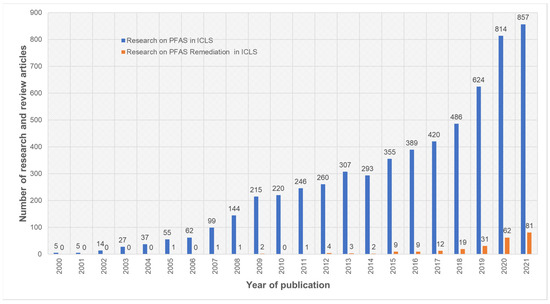
Figure 1.
Number of research and review articles published on PFAS in integrated crop–livestock agroecosystem (ICLS) research over last decades (between 2000 to 2021). The plot was generated using Web of Science search engine using keywords with Boolean operations as (PFAS OR PFOS OR PFOA OR PFBA OR PFBS OR PFPeA OR PFHxS OR PFHxA OR GenX OR PFHpA OR PFNA) AND (Environment OR Human exposure OR Livestock OR Soil OR Milk OR Crop OR Dairy) AND (Remediation).
2. “Forever Chemicals”: Persistence and Mobility
Anthropogenic activities are widely regarded as the major source of PFAS contamination. The sources of PFAS in the environment depend on production methods or application during the manufacturing of other products [26]. Primary manufacturing industries produce PFAS and secondary production facilities use PFAS to produce goods for industrial, commercial and consumer applications [27]. Point source facilities include industrial and military training sites as well as wastewater treatment plants [16]. Industries of the military, textiles, leather and paper products often require PFAS in either training programs (military) or in production processes. As a result, PFAS are often present in surface water and groundwater near industrial, military and wastewater treatment sites. Aqueous Film Forming Foam (AFFF) is a major compound in combating fires and its effectiveness as a fire retardant is often attributed to the presence of PFAS [28]. Other sources of PFAS in agriculture include sewage sludge, bio-solids and treated wastewater, which become inadvertent point sources for PFAS contamination even when judiciously used [29]. Ubiquitous in the environment, PFAS are found in air, water, soil, vegetation and livestock, and thus can be threat to human health if not properly regulated and managed.
Acquiring the epithet “forever chemicals” is not a coincidence. Per- and polyfluoroalkyl substances are tremendously resistant to biological and chemical degradation [30]. The persistence of PFAS in the environment is strongly associated with the length and strength of carbon fluorine chain structures where strength increases with increasing chain lengths [30]. Additionally, PFAS with five or more carbon atoms tend to have higher octanol-water partitioning coefficients than short chain PFAS compounds, which make short chain PFAS compounds more soluble than their long chain counterparts [23]. A negative consequence of the unique physical and chemical properties is the provision of several pathways of PFAS to have long-term stability in soils. Additionally, being biotically stable, they are impervious to both metabolic and bacterial breakdown [31]. Since PFAS are resistant to biotic degradation, once in water or soil, humans and wildlife can have direct or indirect contact through their diets from bioaccumulation in the food chain. In fact, PFAS were added to the Persistent Organic Pollutant (POPs) list under the Stockholm Convention in 2009 for their persistence and subsequent negative environmental and health impacts [32]. There is evidence of biological degradation of perfluorinated chains by breaking bonds one carbon at a time starting at the carboxyl end, with carbon dioxide and fluoride as end products [33]. There is research underway explaining the biological and enzymatic degradation of the “forever chemicals”. Destructive methods can also be based on photolysis, enzymatic reduction or supercritical water oxidation. The bacterial degradation of PFOA and PFOS compounds by Acidimicrobium sp. led to the buildup of fluoride, short-chain perfluorinated products and acetate in pure and enriched culture using ammonium or hydrogen as the electron donor [34]. In addition to microbial degradation, the carbon–fluorine bond cleavage is also documented due to reductive defluorination by an organohalide-respiring microbial community [35].
PFAS contamination in soil has received much attention because of the role soil structure plays in PFAS chemical and physical transformation, as well as PFAS movement within or between environmental media. The protein component of soil organic matter (SOM) serves as an excellent sorption site for PFAS making SOM a significant PFAS reservoir [36]. PFAS persistence in soil is also attributed to its ability to partition in soils [37]. Brusseau et al. referred to soils as a long-term source of contamination because SOM serves as a sink to collect and store PFAS from industrial sources as well as a conduit to transfer PFAS to drinking water sources (rivers, lakes, surface water and groundwater) [38]. This leads to a cyclical exposure pathway of contaminations across soil, biota and the atmosphere [25]. Once in soils, PFAS compounds are soluble and can be transported into surface water or leach into groundwater, which leads to bioaccumulation in plants and animals, especially in ICLS. There is a high potential for PFAS uptake by forages and tubers [39]. Additionally, PFAS can be transferred from soil to plant roots via diffusion and sorption [40]. Based on a plethora of research data, it is evident that the occurrence of PFAS concentration in soils is a global issue as distribution of PFAS were more prominent in soils when compared with other media such as air, surface water, or groundwater [25]. Moreover, it was observed that PFAS not only occur at varying levels in both urban and rural areas, but also among diverse soil types over prolonged periods [41,42,43,44,45]. Total continental PFSAs concentration ranged between 29–14,300 and 7–3270 pg/g for PFCA and PFSA, respectively [46].
The physical and chemical behavior of PFAS in soils has been linked to specific PFAS manufacturing and production processes. One type of PFAS production process results in a molecular structure with significant side chains or branching, whereas another produces linear molecules with little or no branching. For example, Washington et al., in a study in Decatur, Alabama, USA, observed the persistence of PFAS in sludge-applied soils correlated to the length of the carbon–fluorine bonds—longer chained bonds resulted in less mobility, and vice-versa [47]. On contrary, polluted groundwater was associated with shorter chained (<8) PFAS carbon–fluorine bonds [31]. The production processes affect the surface-active behavior, and determine if PFAS compounds are either hydrophobic, oleophobic, lipophobic, or hydrophilic [48]. Uncertainty and complexity of PFAS behavior can be explained in part by the hydrophobic backbone, polar or ionic head structure and functional group of the head structure. Both the hydrophobic C-F backbone and the hydrophilic functional head can control sorption/solvation in environment [49].
3. Human Health Impacts and Exposure
In our exploration of PFAS impacts on ICLS, it is crucial to consider how PFAS exposure can adversely impact human health. The changing composition of PFAS during use, cumulative nature of bioaccumulation, potential for delayed and/or long-term health effects, as well as the modification of effects by coexisting environmental factors make it challenging to study the health effects of PFAS. However, several studies have shown an association with various adverse health issues in humans with exposure to PFAS. Effects on renal health, endocrine function, metabolism and bone health have been well studied and reported [50,51,52,53,54,55,56,57,58,59,60]. The health effects of intrauterine exposure to PFAS have also been investigated [50,61,62,63,64].
A growing body of literature suggests that PFAS exposure disrupts endocrine function and metabolism. Dysregulation of thyroid hormones have been studied most frequently. Exposure to PFAS has been positively associated with free thyroxine levels, though there has been no demonstrated association with Thyroid Stimulating Hormone levels [51]. The association varies with different PFAS, gender, iodine levels and smoking status [52]. Changes in glucose and lipid metabolism are also noted with PFAS exposure [53,54]. The prevalence of metabolic syndrome and its individual components have been found to have an association with PFAS exposure though the results are only consistent across studies for PFNA (perfluorononanoic acid) [53]. An effect on glucose metabolism may be particularly relevant in pregnancy, where PFOS exposure has a positive association with increased glucose levels, although overt gestational diabetes mellitus has not been seen [54].
Adverse kidney health has also been linked with exposure to different types of PFAS. Kidneys are the major route of elimination for PFAS, particularly short carbon chain, carboxylic acid functional group or branched isomer forms [55]. Three studies based on the National Health and Nutrition Examination Survey cohort found an association between direct PFOA and PFOS exposure, renal dysfunction (decreased Estimated Glomerular Filtration Rate) and prevalence of chronic kidney diseases among both adults and children [55]. Another cohort study noted an inverse relationship between serum perfluoro hexane sulfonic acid (PFHxS), perfluorononanoic acid (PFNA), perfluorodecanoic acid (PFDeA) levels and Estimated Glomerular Filtration Rate [56]. The effects of PFAS exposure in renal health may go beyond chronic kidney disease. An association has been postulated with renal and genitourinary cancers, though the data remain sparse and large-scale cohort studies with longer follow-up are required to further elucidate the role [55,57].
Toxicological studies have demonstrated several cellular and metabolic derangements associated with short- and long-term PFAS exposure, which provide insight into possible mechanisms. In animal models, exposure to PFOS in particular has been shown to cause cell death by inducing enzymes of cell apoptosis such as capsases and cytochrome c, and reduce antioxidant enzymes leading to oxidative stress [55,65]. This alteration in nuclear transcription may be mediated by the dysregulation of PPAR (peroxisome proliferators-activated receptors) pathways, which are involved modulation of gene expression in cells [55]. Enhanced endothelial permeability through actin filament remodeling has also been demonstrated with PFAS exposure, which could be a key mechanism of podocyte injury leading to chronic kidney disease [66].
Prenatal exposure to PFAS has been associated with reduced bone mass in young females and was attenuated when adjusted to body composition [50]. In addition, early childhood exposure has also been associated with lower bone mineral density (BMD) with a possible response related to the intensity and/or magnitude of exposure [58]. Repeated exposure to PFAS is also associated with worse bone health [59]. In a small sample of young male subjects, PFAS exposure was also associated with increase in osteoporosis risk and risk of fracture [60]. A mechanism suggested has been the affinity of PFAS to hydroxyapatite in bones [60]. Endocrine modifying effects have also been suggested to play a role [59]. The findings of the effects of PFAS on skeletal health in early life are particularly important as childhood and youth are important determinants of fracture risk later in life [58].
Different PFAS have been found in maternal blood and cord blood, indicating placental transmission [67,68,69]. Maternal exposure to PFAS is widespread, with detectable levels found in serum in 98% of studied pregnant women in one study [61]. Exposure to higher levels of PFOA and PFHpS (Perfluoroheptanesulfonic acid) have been found to be associated with higher odds of miscarriage [62]. Perfluorononanoic acid (PFNA) exposure has been linked to risk of preterm birth [61]. Prenatal PFAS exposure may also be associated with both maternal and fetal thyroid dysfunction though the effect seemed to vary with individual compounds [63]. A large epidemiological study in Italy also noted higher rates of severe small-for-gestational-age (SGA) births among populations living in PFAS-contaminated regions, though no link with SGA was found with maternal PFAS exposure in a small cohort of pregnant women in USA [61,64].
There is increasing recognition that human health does not exist in a void, but is interconnected with the health of the shared environment with soil, plants, water and livestock [70]. A ‘One Health’ approach involves combining the institutional knowledge of professions in these interconnected fields [70]. This is particularly relevant to PFAS, where a variety of human activities lead to PFAS generation, environmental contamination, exposure to ICLS and human exposure pathways simultaneously occurring in close geographical proximity.
4. Fate of PFAS Compounds in Integrated Crop–Livestock Systems
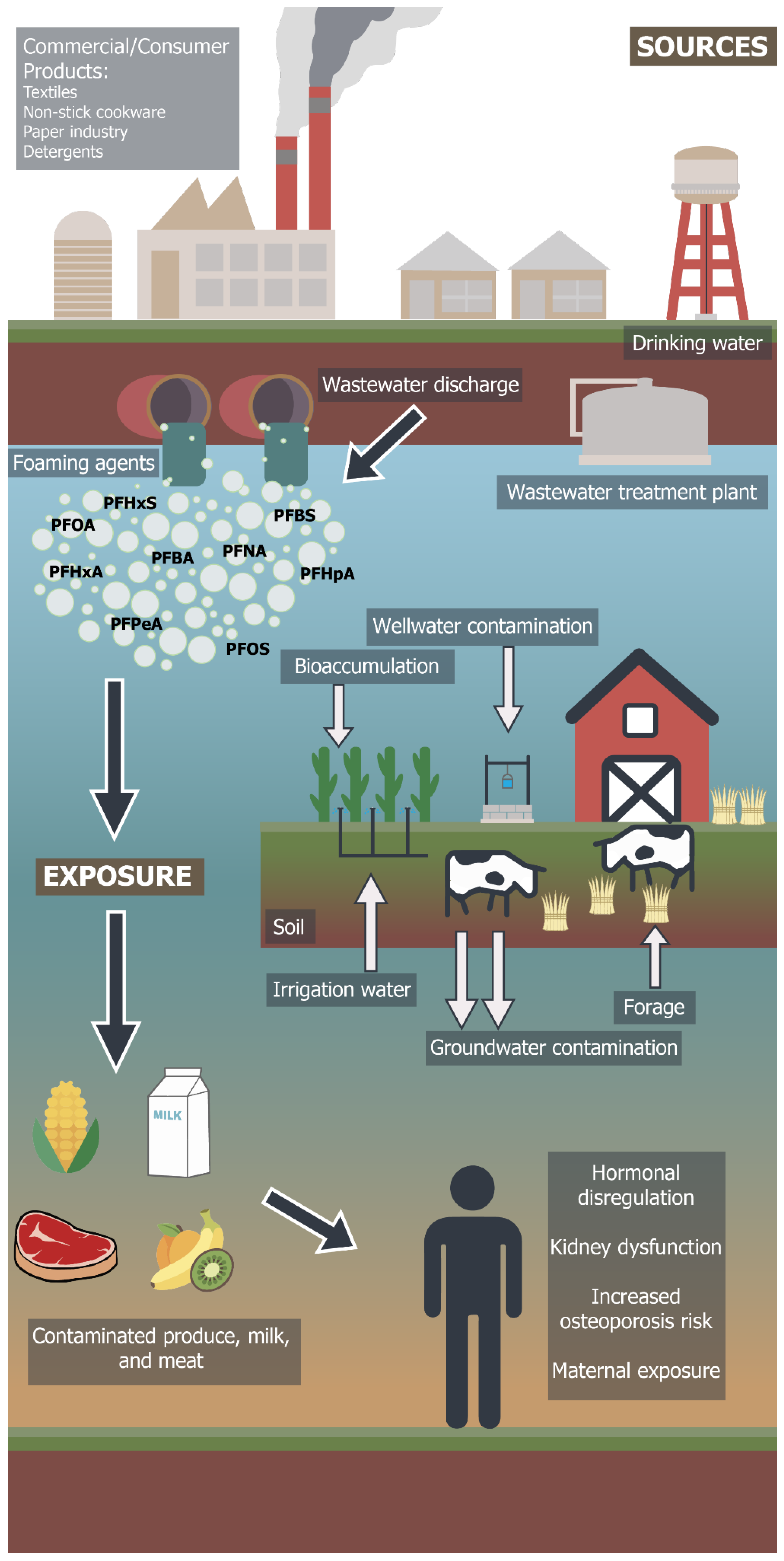
The PFAS group of chemicals cycle through water, soil, crops, dairy and meat products in ICLS and impact human health through various exposure pathways (Figure 2). Diet and drinking water are the main routes of human exposure to PFAS [71]. The presence of PFAS has been reported in the surface/subsurface water and groundwater of leading dairy producing countries such as India, the United States and China, including drinking water supplies [72,73]. According to research based on a recursive regression model of India’s ICLS, groundwater use accounts for 38% of the total value of milk output, whereas surface water use accounts for 15% of the same output [74]. However, water from the Ganges river of India has detectable concentrations of PFAS chemicals, mainly PFOA and PFOS [75,76]. The flux estimates of PFAS chemicals from Ganges River is reported to be in the range of several hundreds of kilograms per year [77]. Although these chemicals are reported below international standards, their persistence poses danger for biomagnification and accumulation at higher trophic levels in harmful concentrations, specifically for cattle and crop production.
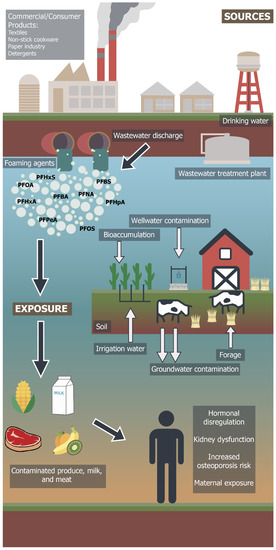
Figure 2.
Potential human exposure pathway of PFAS contaminants from commercial/industrial sources through crop livestock agroecosystems (Artwork by Dawson Diaz).
As of January 2021 in the United States, 2337 locations from 49 states were known to have PFAS contamination in water samples from military sites [78]. Most humans in the United States are exposed to PFAS, with blood, serum and urine containing PFOA and PFOS compounds [79]. In 2017, a well water district was shut down in Maine after PFAS were identified in the water samples, which also lead to the shutdown of a nearby ICLS with detectable levels of PFAS in soils, hay, milk and human blood samples [80]. The United States Food and Drug Administration has reported levels of PFAS in milk samples analyzed from one of the New Mexico dairies possessing potential human health concerns [81]. The PFAS compounds found in the milk samples from this dairy were PFOA (47-169 parts per trillion) and PFOS (881–5680 parts per trillion) [81]. Approximately 5000 cattle consumed PFAS-contaminated groundwater in this dairy [81]. The contamination emerged from a foaming agent used in the nearby air force base [81]. The Food Safety and Inspection Service of the United States Department of Agriculture ceased dairy production and shipment of cattle from the contaminated NM dairy until a baseline analysis and further research data on PFAS depletion kinetics are produced [82]. Well samples monitored in Madison, Wisconsin (the second largest dairy producing state in the United States), reported the presence of 18 different types of PFAS compounds across domestic, municipal and agricultural wells, ranging from 2.5 to 47 parts per trillion [83].
The PFAS contamination emerges from industrial wastes, contaminated sludges or foaming agents and infiltrates into the soil matrix, surface water and groundwater. Groundwater that is used for irrigating forage crops in ICLS can integrate PFAS into the leaf tissue through bioaccumulation [84]. Feeding hay to cattle contaminates dairy products that are consumed by humans [85]. PFAS can be associated with microplastics in lake environments and PFAS sorption onto microplastics can be enhanced in the presence of organic matter [86]. Once incorporated into food webs, PFAS compounds move across different trophic levels of food webs accumulating in different concentration ranges. Table 2 provides the examples of studies that reported different levels of PFAS compounds (mainly PFOS and PFOA) in different components of ICLS. Bioaccumulation of PFAS is related to the concentration and type of PFAS in cattle feed or forage. Long chain PFAS (10–20 fluorinated carbons) compounds ingested through livestock feed or watering have lower potential for removal from urine or milk and higher potential for accumulation and biomagnification in beef/tissue of cattle than short chain (nine fluorinated carbons) compounds [87,88,89]. The estimated half-life of PFOS in dairy cows is approximately 56 days based on a pharmacokinetic model describing the uptake of contamination by cows through contaminated forage vs. its elimination from milk [89]. However, this conclusion was based on assumption of complete elimination of consumed contamination through milk samples [90]. More research on accumulation and biomagnification factors in meat is required for developing regulatory policies for different groups of PFAS compounds based on its half-life, retention and assimilation in cattle.

Table 2.
Examples of reported levels of PFOS and PFAS in individual components of ICLS.
5. Limited Global Regulations and Standards to Address Environmental Concerns
Toxicological impacts of PFAS are site-specific, and regulations depend on socioeconomic and political factors [25]. The U.S. EPA has developed sampling methods for potable and non-potable water. However, there are no published protocols for analyzing bioaccumulated PFAS in forage crops or soil samples. There are currently no residential or regional screening levels available through federal guidelines for PFAS compounds in soils in the United States for agricultural or industrial limits. Agricultural, industrial and commercial soil screening levels for different PFAS compounds have been identified and defined by Health Canada [98]. For example; soil screening values for PFOA and PFOS in agricultural soils are 0.70 and 2.1, respectively [98]. In 2009, the United Nations Environment Programme’s Stockholm Convention listed PFOA (salts and related compounds) and PFOS in Annex A and C [99,100,101]. Annex A aims at eliminating the production and use whereas Annex C aims at restricting the production and use of chemicals [99,100,101]. The current health advisory levels of PFOA and PFOS from drinking water developed by the U.S. EPA have been established at a lifetime exposure of 70 parts per trillion [102]. However, it is important to develop limits, regulatory guidelines and screening levels for livestock watering to reduce the accumulation and transport of PFAS compounds in milk, meat and other dairy products to eliminate pathways to human exposure.
Investigations of PFOS, PFOA and PFBS in the United States take place under the Comprehensive, Environmental Response, Compensation and Liability Act, Toxic Substances Control Act, the Safe Drinking Water Act and the Defense Environmental Restoration Program. The Compensation and Liability Act follows investigation of PFOA and PFOS under EPA’s residential screening levels for tap water (0.4 ppb at hazard quotient = 1) and soil (1.3 ppm at hazard quotient = 1). The research underway by the U.S. EPA has validated test methods for 29 PFAS compounds, facilitated the clean-up of contaminated groundwater sites, developed the significant new use rule to regulate manufacture and import of PFOA and PFOS and provided assistance to more than 30 states for developing new tools for research and communications [103]. Research findings to date have focused widely on the need for regulation standards in drinking water; however, it is important to expand monitoring and develop regulatory standards for milk, meat and agricultural commodities. The sampling methods that are currently used for soil, potable and non-potable water, milk and human serum are listed in Table 3. Various agencies have developed unique sampling methods (Table 3) from simple liquid chromatography to isotope dilution to carefully separate out contaminants. These technologies are continually advancing to achieve higher efficiency.

Table 3.
Sampling methodology and storage techniques for PFAS contaminants in soil, water, milk, meat and human serum.
Several cases of milk contaminants have been reported in the US states such as Maine, New Mexico, Wisconsin, Alabama, but to date, no guideline values have been developed by regulatory agencies. In 2017, the Maine Center for Disease Control developed an action level for PFOS in cow’s milk of 210 ppb after contaminated samples were found in hay, monitoring well samples, milk and groundwater used for drinking and livestock watering [104]. Maine CDC developed this action level based on the U.S. EPA’s recommendations of a reference dose of 20 ng/kg of body weight per day for PFOS in drinking water [105]. Maine Department of Environmental Protection has also developed remedial action guidelines for soil in crop-based dairy systems considering a soil-hay-cow-milk exposure pathway model of 1,700,000 ng/kg dry weight [105]. The remedial action guidelines are based on an EPA reference dose for drinking water that uses an incidental soil ingestion exposure pathway for children [105]. The current discrepancies and knowledge gaps in formulating the action and guideline values require more research on residual toxicity in environment and biomagnification on different trophic levels.
The European Commission severely restricted the use of PFOS in Europe, except for the space industry. However, in 2020 the EU banned the production and marketing of PFOA salts and precursors for all industrial uses including space exploration [106]. The PFAS contaminants (PFOA, PFOS, PFNA and PFHxS) are now restricted under regulatory framework on food contaminants by the European Union [107]. The German Ministry of Health proposed regulatory guidelines of maximum 300 parts per trillion of combined PFOA and PFOS for human health exposure.
6. Remediation and Preventive Strategies
Soil and water are precursors to plant and animal food chains. The use of PFAS-contaminated soil and water can lead to entry of PFAS compounds into leaf tissues (forage, pasture or vegetables/fruits), edible produce, meat and dairy products. These chemicals cycle through water, soil, crop, dairy and meat products in an integrated livestock system and impact the human health. Assessing the risks and levels of PFAS in soils and water could help prevent the bioaccumulation in crops and livestock used for human consumption. Therefore, most of the remediation and preventive techniques presented here are more focused on soil and water. These measures are intended to prevent the migration of PFAS compounds into crops and livestock products by keeping the levels of these contaminants below the guideline values/residential screening levels.
In addition to conventional remediation techniques, new techniques are constantly being developed for PFAS remediation in soils and water. Most of the previous techniques, such as incineration and adsorption using activated carbon, have known challenges such as cost, energy consumption, difficulty with disposal and extreme operating conditions [108,109]. Many passive approaches have had partial success such as adsorption, filtration, reverse osmosis, enhanced photolysis, electrochemical oxidation and sonochemical destruction.
Conventional soil PFAS remediation methods include soil washing, excavation and thermal oxidation, chemical oxidation, ball milling, electron beams, immobilization methods, excavation and landfilling (Table 3). Some of the most effective PFAS remediation strategies known as ‘treatment train processes’ have been in use for in situ remediation which uses multiple synergistic technologies [110]. Some partially effective treatment methods were anion exchange, granular activated carbon (GAC) and reverse osmosis [111].
The manipulation of materials and structures at nanoscale dimensions, i.e., nanotechnology, has recently been used as one of the most innovative and promising technologies for PFAS remediation. As an example, engineered nanomaterials use a large specific surface area and the quantum nature of energy states to have more accessible adsorption sites and higher surface reactivities [112,113]. This enables effective remediation of contaminated water by adsorbing the PFAS compounds in the exposed surfaces of the nanomaterials [112,113,114]. Another technique with great potential for replication to remove PFAS is the use of modified nanosized iron oxides with high adsorption capacity and magnetic properties which serve as ideal sorbents for PFAS under multiple conditions [115]. Similarly, nano-photocatalysts under UV irradiation can be used to decompose PFOA in the presence of TiO2-based, Ga2O3-based, or In2O3 catalysts [115]. Research on remediation of PFAS in water is underway and evolving every day. Techniques for PFAS remediation include physical, chemical and biological approaches integrated within ICLS. These remediation techniques can be classified into three major categories: (a) soil and sediment remediation, (b) water remediation and (c) biological remediation.
6.1. Soil and Sediment Remediation Methods
Soil remediation techniques are based on adsorption/separation behavior, soil washing and thermal treatment, soil liquefaction, excavation and placement in impermeable materials or offsite disposal in landfills, sonochemical destruction, advanced oxidation or reduction processes, ball milling or vapor generation methods [116,117]. Soil adsorption/separation methods commonly use soil stabilization [116], where the PFAS sorbents are added and mixed with soil to stabilize and immobilize PFAS compounds [117]. In general, the sorbent adsorbs PFAS and reduces the potential of PFAS leaching to the groundwater. However, this approach works best in soil compared with water because PFAS release tends to occur on the surface, and some PFAS chemicals have a strong affinity to soil [16]. This method has significant importance in managing PFAS contamination in military bases. Alternatively, powdered or granular activated carbon (e.g., carbon fibers, BioNuchar etc.) with a porous structure and strong heterogeneous surface is used to sorb and remove harmful compounds [118,119,120]. Soil washing involves the separation of contaminants from soil and sediments through the application of water, solvents or air bubbles on the contaminated soil or water [121]. Thermal treatment involves increasing the soil temperature to 500–600 °C to vaporize organic contaminants and break PFAS compounds in the gas stream at a temperature of 1200 °C [122,123]. Another useful technique named soil liquefaction uses foam fractionation techniques to detach PFAS from liquefied soil and water to the induced bubbles [124].
In recent years, other in situ and off-site ways to remediate PFAS have been developed. One such technique is excavation, which can be used in two different ways. First, excavating and placing soils in impermeable materials reduces infiltration, isolates impacted material and controls seepage by a proper drainage system. It is a short-term, uncertain solution. Often, contamination may still be present on-site, and any small physical disturbance can spread the contaminants. Thus, the impermeable materials require continuous monitoring and long-term management [125,126]. On the contrary, excavation and offsite disposal in landfills involves reduction in the volume and concentration of PFAS and immobilization before locating in an offsite landfill. However, this process needs additional steps to collect, destroy and monitor all PFAS separately, and should not be considered as a preferred remediation technique from a management standpoint [127,128].
Ex situ methods such as sorption, filtration and sonochemical destruction methods help lower toxicity by degradation of PFOS and PFOA in soil and water [129]. One such technique includes sonolysis, which uses sound waves at 20–1100 kHz frequencies to facilitate cavitation in water and create bubbles with large surface area enabling decomposition of PFAS [130,131]. In the advanced oxidation/reduction process, contaminants (phenols, endocrine-disrupting chemicals) are destroyed either by direct anodic oxidation or in solution using strong oxidants generated by cathodic electrochemical reactions [132,133]. In general, this process is versatile, has a long life span, is energy efficient, automated and cost-effective [134]. In addition, ozonation is a commonly used advanced oxidation process for water treatment plants in the United States [135]. In recent times, use of an electron beam is considered as a more advanced oxidation-reduction process, involving irradiating material with accelerated electrons to destroy PFAS. However, this electron beam approach has been used only on wastewater and aqueous solutions, thus warrants further testing for its applicability to ICLS [136,137]. Another common method similar to ball milling is a mechanochemical (MC) destruction method, which employs mechanical force such as shaking to allow reactions on the surface of ball mills to effectively destroy PFOS and PFOA [138]. In addition, vapor energy generators use 1100 °C steam to destroy PFAS from impacted soils in an ex situ treatment chamber [139].
6.2. Water Remediation Methods
The principal techniques in remediation of water from PFAS include adsorption and separation using foam fractionation, reverse osmosis or nanofiltration, resin membrane-based ion exchange, incineration, electrochemical oxidation (ozonation and ozofractionation), photolysis, enzymatic reduction and water oxidation. In adsorption and separation, PFAS adsorbent materials are introduced into the contaminated waters to immobilize PFAS using adsorption and separation techniques. This method can also use foam fractionation to detach PFAS from liquefied soil and water with induced bubbles [124]. The advanced oxidation process of PFAS contaminant removal can be destructive by direct anodic oxidation—contaminants (phenols, endocrine-disrupting chemicals) adsorb onto the anode surface and are destroyed by an electron transfer reaction or oxidation reaction through strong oxidants generated by cathodic electrochemical reactions [135]. It is an evolving destructive technology for many PFAS. Ozonation is a commonly used advanced oxidation process method for water treatment plants in the United States. This method treats liquid waste by chemically oxidizing organic contaminants and forming concentrated foam fractionates, which can be separated from the treated water [140,141].
There are some economically feasible and environmentally sustainable water remediation methods which have been developed in recent years. For example, photolysis in aqueous solutions involves producing products such as carbonates and bicarbonate radicals which react to produce PFAS molecules with shorter chains [142]. Another technique called supercritical water oxidation uses the super critical state of water (water at temperature 373.9 °C and pressure of 221.1 bar), where the chemical oxidation process breaks down strong carbon fluorine bonds and decomposes various hazardous organic pollutants into non-toxic steam waste [143,144,145].
6.3. Microbial and Phytoremediation
Phytoremediation could be an effective approach for controlling PFAS, as with many other contaminants of concern in the environment. Limited information on plant accumulation and analytical techniques for bioavailable fractions in plant roots have restricted these remediation methods. However, research and protocols for monitoring PFAS exposure pathways have initiated some concepts and principles to apply in remediating the contaminants from rhizosphere using degradation techniques. Some of these techniques include rhizodegradation, phytoextraction, phytotransfer, mycoremediation and biodegradation. Rhizodegradation is a plant-assisted bioremediation in which organisms residing in the root zone alter or degrade chemicals [146]. It is an aerobic process facilitated by enzyme catalyzed (oxidative) degradation [147]. Rhizodegradation is cost efficient, low maintenance and most sustainable for PFAS-contaminated sites; however, the process can be a slow and long-term approach [148]. Some phytoremediation candidate species include (a) the wetland species Juncus effuses, that has been demonstrated to accumulate 11.4% of seven PFAS compounds from PFAS-spiked soil [146], and (b) Betula pendula and Picea abies, which were reported to accumulate up to 97 and 94 ng g−1 of PFAS compounds at a firefighting training site near Stockholm, Sweden [147]. The PFAS contaminants can also be degraded by lignolytic fungi or via biodegradation by aerobic bacteria into non-toxic compounds such as carbon dioxide, water and ammonia [149,150]. The ability of Gram-positive and Gram-negative bacteria to break down PFAS depends on soil organic matter content, and involves electrostatic and hydrostatic connections in the bacterial sorption of compounds [151].
7. Exposure and Equity
Human exposure to PFAS has been a public health concern due to its high environmental persistence. Research on location or clusters of certain industries in low income, underserved communities and related adverse health impacts has expanded significantly over recent years, raising issues of environmental justice [152,153]. PFAS contamination is also an environmental justice problem. Accumulating scientific evidence has linked adverse outcomes in community health of low-income households and underserved communities inhabiting within five miles of PFAS sites [154,155]. A comparison of over 70 non-military sites found that 39,000 more low-income households and 295,000 more underserved people were within five miles of sites contaminated with PFAS [156]. In the US state of Michigan, environmental inequities are even more pronounced, with 36,170 more low-income households and 134,488 more underserved people living within five miles of a PFAS-contaminated site [156].
Public policy initiatives have been increasingly investigating environmental quality as an indicator of individual human health and well-being [157]. As environmental risks are not evenly distributed across socioeconomic groups, additional considerations for exposure assessment have included socioeconomic factors of affected populations [158]. Framed as environmental justice or the inequitable distribution of risks and exposure, numerous studies have reported detrimental environment effects that have disproportionately affected socially disadvantaged and underserved populations. Whereas a myriad of factors affect health outcomes, socioeconomic status and race have been found to highly correlate with health and environmental inequalities showing strong associations and translating inequalities to inequities [159,160]. Studies have also shown that socioeconomic factors influence contaminant exposure and specific health outcomes [158]. Disparities in human exposure to harmful chemicals such as PFAS have also been shown to exist among educational statuses. For example, underserved communities with lower levels of educational attainment and political capital often have less access to funding to conduct hazard identification, exposure analysis and health related effects [161]. Moreover, it was observed that poor access to health care information and healthcare means lower health promotion rates, lower risk avoidance, a less healthy diet and more adverse conditions that increase susceptibility to exposure [162]. Underserved communities are underrepresented in media and underserved by government entities [163]. Other factors include polluting industries or those with the greatest risk to produce harmful chemicals and pollute the environment are more likely to be located near low-income communities. These groups are often placed at a disproportionately higher risk for environmental chemical exposure [164]. Therefore, accounting for institutional differences and understanding actions by government entities in perpetuating resource injustice, is a prerequisite for advancing equitable solutions related to PFAS contamination and exposure [165].
In our systematic review, we did not find any studies examining the impact of PFAS exposure related to soil, plants, livestock, and water adjacent to underserved communities. Moreover, we did not find any studies examining the feasibility and any additional challenges for PFAS remediation in socially and economically disadvantaged agricultural communities. This is an important area for future interdisciplinary work in order to avoid further inequities from arising related to exposure, remediation and monitoring.
8. Conclusions
In integrated crop–livestock systems, PFAS compounds are impacting surface water and groundwater by infiltrating through soils from industrial sources of contamination. Once groundwater is contaminated, it can lead to exposure pathways of bioaccumulation in plants and cattle contaminating the entirety of farm produce and dairy products. Consumption of these contaminated products leads to severe human health issues. There is evidence of PFAS contamination in milk and meat samples from dairies in countries such as the United States and China. Therefore, more vigorous research and regulatory guidelines are critical and required for monitoring and developing screening guidelines not only for the dairy products but also for soil, groundwater, forage and crops. Lack of assessment of the extent of exposure pathways in underserved communities living near PFAS sites may be further advancing disparate adverse health impacts. However, more research is required to understand the combined agronomic and epidemiological impacts for developing diet regulatory guidelines.
Author Contributions
Conceptualization, G.J., V.K., D.S., B.D. and M.N.; methodology, G.J., V.K., E.M., D.S., B.D. and M.N.; resources, G.J., V.K., E.M, S.P., D.S., B.D. and M.N.; writing—original draft preparation, G.J., V.K., E.M. and S.P.; writing—review and editing, G.J., V.K., E.M., S.P., D.S., B.D., D.D. and M.N.; visualization, D.D.; supervision, B.D., D.S. and M.N.; project administration, G.J.; funding acquisition, M.N. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors acknowledge the technical and financial support to the UC Davis Open Access Fund (UCD-OAF) by UC Davis Library for promoting broad access of this collaborative project.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Haukås, M.; Berger, U.; Hop, H.; Gulliksen, B.; Gabrielsen, G.W. Bioaccumulation of per- and polyfluorinated alkyl substances (PFAS) in selected species from the Barents Sea food web. Environ. Pollut. 2007, 148, 360–371. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.W.; Smithwick, M.M.; Braune, B.M.; Hoekstra, P.F.; Muir, D.C.G.; Mabury, S.A. Identification of Long-Chain Perfluorinated Acids in Biota from the Canadian Arctic. Environ. Sci. Technol. 2004, 38, 373–380. [Google Scholar] [CrossRef]
- Taniyasu, S.; Kannan, K.; Horii, Y.; Hanari, N.; Yamashita, N. A Survey of Perfluorooctane Sulfonate and Related Perfluorinated Organic Compounds in Water, Fish, Birds, and Humans from Japan. Environ. Sci. Technol. 2003, 37, 2634–2639. [Google Scholar] [CrossRef]
- Buck, R.C.; Franklin, J.; Berger, U.; Conder, J.M.; Cousins, I.T.; de Voogt, P.; Jensen, A.A.; Kannan, K.; Mabury, S.A.; van Leeuwen, S.P. Perfluoroalkyl and polyfluoroalkyl substances in the environment: Terminology, classification, and origins. Integr. Environ. Assess. Manag. 2011, 7, 513–541. [Google Scholar] [CrossRef] [PubMed]
- Paul, A.G.; Jones, K.C.; Sweetman, A.J. A First Global Production, Emission, And Environmental Inventory For Perfluorooctane Sulfonate. Environ. Sci. Technol. 2009, 43, 386–392. [Google Scholar] [CrossRef]
- Prevedouros, K.; Cousins, I.T.; Buck, R.C.; Korzeniowski, S.H. Sources, Fate and Transport of Perfluorocarboxylates. Environ. Sci. Technol. 2006, 40, 32–44. [Google Scholar] [CrossRef]
- Boiteux, V.; Dauchy, X.; Bach, C.; Colin, A.; Hemard, J.; Sagres, V.; Rosin, C.; Munoz, J.-F. Concentrations and patterns of perfluoroalkyl and polyfluoroalkyl substances in a river and three drinking water treatment plants near and far from a major production source. Sci. Total Environ. 2017, 583, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Lam, N.H.; Cho, C.-R.; Kannan, K.; Cho, H.-S. A nationwide survey of perfluorinated alkyl substances in waters, sediment and biota collected from aquatic environment in Vietnam: Distributions and bioconcentration profiles. J. Hazard. Mater. 2017, 323, 116–127. [Google Scholar] [CrossRef]
- Loganathan, B.G.; Sajwan, K.S.; Sinclair, E.; Senthil Kumar, K.; Kannan, K. Perfluoroalkyl sulfonates and perfluorocarboxylates in two wastewater treatment facilities in Kentucky and Georgia. Water Res. 2007, 41, 4611–4620. [Google Scholar] [CrossRef]
- Wang, Y.; Chang, W.; Wang, L.; Zhang, Y.; Zhang, Y.; Wang, M.; Wang, Y.; Li, P. A review of sources, multimedia distribution and health risks of novel fluorinated alternatives. Ecotoxicol. Environ. Saf. 2019, 182, 109402. [Google Scholar] [CrossRef]
- Hoffman, K.; Webster, T.F.; Weisskopf, M.G.; Weinberg, J.; Vieira, V.M. Exposure to Polyfluoroalkyl Chemicals and Attention Deficit/Hyperactivity Disorder in U.S. Children 12–15 Years of Age. Environ. Health Perspect. 2010, 118, 1762–1767. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calafat, A.M.; Wong, L.-Y.; Kuklenyik, Z.; Reidy, J.A.; Needham, L.L. Polyfluoroalkyl Chemicals in the U.S. Population: Data from the National Health and Nutrition Examination Survey (NHANES) 2003–2004 and Comparisons with NHANES 1999–2000. Environ. Health Perspect. 2007, 115, 1596–1602. [Google Scholar] [CrossRef] [PubMed]
- Clara, M.; Scharf, S.; Weiss, S.; Gans, O.; Scheffknecht, C. Emissions of perfluorinated alkylated substances (PFAS) from point sources—identification of relevant branches. Water Sci. Technol. 2008, 58, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Lindstrom, A.B.; Strynar, M.J.; Libelo, E.L. Polyfluorinated Compounds: Past, Present, and Future. Environ. Sci. Technol. 2011, 45, 7954–7961. [Google Scholar] [CrossRef]
- Shahsavari, E.; Rouch, D.; Khudur, L.S.; Thomas, D.; Aburto-Medina, A.; Ball, A.S. Challenges and Current Status of the Biological Treatment of PFAS-Contaminated Soils. Front. Bioeng. Biotechnol. 2021, 8, 1493. [Google Scholar] [CrossRef]
- Hu, X.C.; Andrews, D.Q.; Lindstrom, A.B.; Bruton, T.A.; Schaider, L.A.; Grandjean, P.; Lohmann, R.; Carignan, C.C.; Blum, A.; Balan, S.A.; et al. Detection of Poly- and Perfluoroalkyl Substances (PFASs) in U.S. Drinking Water Linked to Industrial Sites, Military Fire Training Areas, and Wastewater Treatment Plants. Environ. Sci. Technol. Lett. 2016, 3, 344–350. [Google Scholar] [CrossRef]
- Cui, J.; Gao, P.; Deng, Y. Destruction of Per- and Polyfluoroalkyl Substances (PFAS) with Advanced Reduction Processes (ARPs): A Critical Review. Environ. Sci. Technol. 2020, 54, 3752–3766. [Google Scholar] [CrossRef] [PubMed]
- Hamid, H.; Li, L.Y.; Grace, J.R. Review of the fate and transformation of per- and polyfluoroalkyl substances (PFASs) in landfills. Environ. Pollut. 2018, 235, 74–84. [Google Scholar] [CrossRef]
- Hepburn, E.; Madden, C.; Szabo, D.; Coggan, T.L.; Clarke, B.; Currell, M. Contamination of groundwater with per- and polyfluoroalkyl substances (PFAS) from legacy landfills in an urban re-development precinct. Environ. Pollut. 2019, 248, 101–113. [Google Scholar] [CrossRef]
- Curtzwiler, G.W.; Silva, P.; Hall, A.; Ivey, A.; Vorst, K. Significance of Perfluoroalkyl Substances (PFAS) in Food Packaging. Integr. Environ. Assess. Manag. 2021, 17, 7–12. [Google Scholar] [CrossRef]
- Pontius, F. Regulation of Perfluorooctanoic Acid (PFOA) and Perfluorooctane Sulfonic Acid (PFOS) in Drinking Water: A Comprehensive Review. Water 2019, 11, 2003. [Google Scholar] [CrossRef] [Green Version]
- Hamid, H.; Li, L. Role of wastewater treatment plant in environmental cycling of poly- and perfluoroalkyl substances. Ecocycles 2016, 2, 43–53. [Google Scholar] [CrossRef]
- Ghisi, R.; Vamerali, T.; Manzetti, S. Accumulation of perfluorinated alkyl substances (PFAS) in agricultural plants: A review. Environ. Res. 2019, 169, 326–341. [Google Scholar] [CrossRef]
- Higgins, C.; Field, J.; Criddle, C.; Luthy, R. Quantitative Determination of Perfluorochemicals in Sediments and Domestic Sludge. Environ. Sci. Technol. 2005, 39, 3946–3956. [Google Scholar] [CrossRef]
- Abunada, Z.; Alazaiza, M.Y.D.; Bashir, M.J.K. An Overview of Per- and Polyfluoroalkyl Substances (PFAS) in the Environment: Source, Fate, Risk and Regulations. Water 2020, 12, 3590. [Google Scholar] [CrossRef]
- Langberg, H.A.; Arp, H.P.H.; Breedveld, G.D.; Slinde, G.A.; Høiseter, Å.; Grønning, H.M.; Jartun, M.; Rundberget, T.; Jenssen, B.M.; Hale, S.E. Paper product production identified as the main source of per- and polyfluoroalkyl substances (PFAS) in a Norwegian lake: Source and historic emission tracking. Environ. Pollut. 2021, 273, 116259. [Google Scholar] [CrossRef]
- Boone, J.S.; Vigo, C.; Boone, T.; Byrne, C.; Ferrario, J.; Benson, R.; Donohue, J.; Simmons, J.E.; Kolpin, D.W.; Furlong, E.T.; et al. Per- and polyfluoroalkyl substances in source and treated drinking waters of the United States. Sci. Total Environ. 2019, 653, 359–369. [Google Scholar] [CrossRef]
- Backe, W.J.; Day, T.C.; Field, J.A. Zwitterionic, Cationic, and Anionic Fluorinated Chemicals in Aqueous Film Forming Foam Formulations and Groundwater from U.S. Military Bases by Nonaqueous Large-Volume Injection HPLC-MS/MS. Environ. Sci. Technol. 2013, 47, 5226–5234. [Google Scholar] [CrossRef]
- Blaine, A.C.; Rich, C.D.; Sedlacko, E.M.; Hundal, L.S.; Kumar, K.; Lau, C.; Mills, M.A.; Harris, K.M.; Higgins, C.P. Perfluoroalkyl Acid Distribution in Various Plant Compartments of Edible Crops Grown in Biosolids-Amended soils. Environ. Sci. Technol. 2014, 48, 7858–7865. [Google Scholar] [CrossRef]
- Parsons, J.R.; Sáez, M.; Dolfing, J.; de Voogt, P. Biodegradation of Perfluorinated Compounds. In Reviews of Environmental Contamination and Toxicology; Whitacre, D.M., Ed.; Springer US: New York, NY, USA, 2008; pp. 53–71. ISBN 978-0-387-78444-1. [Google Scholar]
- Cousins, I.T.; DeWitt, J.C.; Glüge, J.; Goldenman, G.; Herzke, D.; Lohmann, R.; Ng, C.A.; Scheringer, M.; Wang, Z. The high persistence of PFAS is sufficient for their management as a chemical class. Environ. Sci. Process. Impacts 2020, 22, 2307–2312. [Google Scholar] [CrossRef]
- Martin, J.W.; Asher, B.J.; Beesoon, S.; Benskin, J.P.; Ross, M.S. PFOS or PreFOS? Are perfluorooctane sulfonate precursors (PreFOS) important determinants of human and environmental perfluorooctane sulfonate (PFOS) exposure? J. Environ. Monit. 2010, 12, 1979–2004. [Google Scholar] [CrossRef]
- Wackett, L.P.; Robinson, S.L. The ever-expanding limits of enzyme catalysis and biodegradation: Polyaromatic, polychlorinated, polyfluorinated, and polymeric compounds. Biochem. J. 2020, 477, 2875–2891. [Google Scholar] [CrossRef]
- Huang, S.; Jaffé, P.R. Defluorination of Perfluorooctanoic Acid (PFOA) and Perfluorooctane Sulfonate (PFOS) by Acidimicrobium sp. Strain A6. Environ. Sci. Technol. 2019, 53, 11410–11419. [Google Scholar] [CrossRef] [Green Version]
- Yu, Y.; Zhang, K.; Li, Z.; Ren, C.; Chen, J.; Lin, Y.-H.; Liu, J.; Men, Y. Microbial Cleavage of C–F Bonds in Two C 6 Per- and Polyfluorinated Compounds via Reductive Defluorination. Environ. Sci. Technol. 2020, 54, 14393–14402. [Google Scholar] [CrossRef]
- Wei, C.; Song, X.; Wang, Q.; Hu, Z. Sorption kinetics, isotherms and mechanisms of PFOS on soils with different physicochemical properties. Ecotoxicol. Environ. Saf. 2017, 142, 40–50. [Google Scholar] [CrossRef]
- Bolan, N.; Sarkar, B.; Yan, Y.; Li, Q.; Wijesekara, H.; Kannan, K.; Tsang, D.C.W.; Schauerte, M.; Bosch, J.; Noll, H.; et al. Remediation of poly- and perfluoroalkyl substances (PFAS) contaminated soils—To mobilize or to immobilize or to degrade? J. Hazard. Mater. 2021, 401, 123892. [Google Scholar] [CrossRef]
- Brusseau, M.L.; Anderson, R.H.; Guo, B. PFAS concentrations in soils: Background levels versus contaminated sites. Sci. Total Environ. 2020, 740, 140017. [Google Scholar] [CrossRef] [PubMed]
- Stahl, T.; Heyn, J.; Thiele, H.; Hüther, J.; Failing, K.; Georgii, S.; Brunn, H. Carryover of Perfluorooctanoic Acid (PFOA) and Perfluorooctane Sulfonate (PFOS) from Soil to Plants. Arch. Environ. Contam. Toxicol. 2009, 57, 289–298. [Google Scholar] [CrossRef]
- Lechner, M.; Knapp, H. Carryover of Perfluorooctanoic Acid (PFOA) and Perfluorooctane Sulfonate (PFOS) from Soil to Plant and Distribution to the Different Plant Compartments Studied in Cultures of Carrots (Daucus carota ssp. Sativus), Potatoes (Solanum tuberosum), and Cucumbers (Cucumis Sativus). J. Agric. Food Chem. 2011, 59, 11011–11018. [Google Scholar] [CrossRef] [PubMed]
- Meijer, S.N.; Ockenden, W.A.; Sweetman, A.; Breivik, K.; Grimalt, J.O.; Jones, K.C. Global Distribution and Budget of PCBs and HCB in Background Surface Soils: Implications for Sources and Environmental Processes. Environ. Sci. Technol. 2003, 37, 667–672. [Google Scholar] [CrossRef] [PubMed]
- Naile, J.E.; Khim, J.S.; Wang, T.; Chen, C.; Luo, W.; Kwon, B.-O.; Park, J.; Koh, C.-H.; Jones, P.D.; Lu, Y.; et al. Perfluorinated compounds in water, sediment, soil and biota from estuarine and coastal areas of Korea. Environ. Pollut. 2010, 158, 1237–1244. [Google Scholar] [CrossRef]
- Sepulvado, J.G.; Blaine, A.C.; Hundal, L.S.; Higgins, C.P. Occurrence and Fate of Perfluorochemicals in Soil Following the Land Application of Municipal Biosolids. Environ. Sci. Technol. 2011, 45, 8106–8112. [Google Scholar] [CrossRef] [PubMed]
- Washington, J.W.; Henderson, W.M.; Ellington, J.J.; Jenkins, T.M.; Evans, J.J. Analysis of perfluorinated carboxylic acids in soils II: Optimization of chromatography and extraction. J. Chromatogr. A 2008, 1181, 21–32. [Google Scholar] [CrossRef]
- Yoo, H.; Washington, J.W.; Ellington, J.J.; Jenkins, T.M.; Neill, M.P. Concentrations, Distribution, and Persistence of Fluorotelomer Alcohols in Sludge-Applied Soils near Decatur, Alabama, USA. Environ. Sci. Technol. 2010, 44, 8397–8402. [Google Scholar] [CrossRef]
- Rankin, K.; Mabury, S.A.; Jenkins, T.M.; Washington, J.W. A North American and global survey of perfluoroalkyl substances in surface soils: Distribution patterns and mode of occurrence. Chemosphere 2016, 161, 333–341. [Google Scholar] [CrossRef]
- Washington, J.W.; Yoo, H.; Ellington, J.J.; Jenkins, T.M.; Libelo, E.L. Concentrations, Distribution, and Persistence of Perfluoroalkylates in Sludge-Applied Soils near Decatur, Alabama, USA. Environ. Sci. Technol. 2010, 44, 8390–8396. [Google Scholar] [CrossRef]
- US EPA. Basic Information on PFAS. Available online: https://www.epa.gov/pfas/basic-information-pfas (accessed on 29 September 2021).
- Greathouse, J.A.; Cygan, R.T.; Fredrich, J.T.; Jerauld, G.R. Adsorption of Aqueous Crude Oil Components on the Basal Surfaces of Clay Minerals: Molecular Simulations Including Salinity and Temperature Effects. J. Phys. Chem. C 2017, 121, 22773–22786. [Google Scholar] [CrossRef]
- Jeddy, Z.; Tobias, J.H.; Taylor, E.V.; Northstone, K.; Flanders, W.D.; Hartman, T.J. Prenatal concentrations of perfluoroalkyl substances and bone health in British girls at age 17. Arch. Osteoporos. 2018, 13, 1–7. [Google Scholar] [CrossRef]
- Coperchini, F.; Croce, L.; Ricci, G.; Magri, F.; Rotondi, M.; Imbriani, M.; Chiovato, L. Thyroid Disrupting Effects of Old and New Generation PFAS. Front. Endocrinol. 2020, 11, 612320. [Google Scholar] [CrossRef]
- van Gerwen, M.; Alpert, N.; Alsen, M.; Ziadkhanpour, K.; Taioli, E.; Genden, E. The Impact of Smoking on the Association between Perfluoroalkyl Acids (PFAS) and Thyroid Hormones: A National Health and Nutrition Examination Survey Analysis. Toxics 2020, 8, 116. [Google Scholar] [CrossRef]
- Christensen, K.Y.; Raymond, M.; Meiman, J. Perfluoroalkyl substances and metabolic syndrome. Int. J. Hyg. Environ. Health 2019, 222, 147–153. [Google Scholar] [CrossRef]
- Preston, E.V.; Rifas-Shiman, S.L.; Hivert, M.-F.; Zota, A.R.; Sagiv, S.K.; Calafat, A.M.; Oken, E.; James-Todd, T. Associations of Per- and Polyfluoroalkyl Substances (PFAS) With Glucose Tolerance During Pregnancy in Project Viva. J. Clin. Endocrinol. Metab. 2020, 105, e2864–e2876. [Google Scholar] [CrossRef]
- Stanifer, J.W.; Stapleton, H.M.; Souma, T.; Wittmer, A.; Zhao, X.; Boulware, L.E. Perfluorinated Chemicals as Emerging Environmental Threats to Kidney Health: A Scoping Review. Clin. J. Am. Soc. Nephrol. 2018, 13, 1479–1492. [Google Scholar] [CrossRef] [Green Version]
- Stern, J.E.; Son, S.; Biancardi, V.C.; Zheng, H.; Sharma, N.; Patel, K.P. Astrocytes Contribute to Angiotensin II Stimulation of Hypothalamic Neuronal Activity and Sympathetic Outflow. Hypertens. 2016, 68, 1483–1493. [Google Scholar] [CrossRef] [Green Version]
- Steenland, K.; Winquist, A. PFAS and cancer, a scoping review of the epidemiologic evidence. Environ. Res. 2021, 194, 110690. [Google Scholar] [CrossRef]
- Schmidt, C.W. Reduced Bone Mineral Density in Children: Another Potential Health Effect of PFAS. Environ. Health Perspect. 2020, 128, 044002. [Google Scholar] [CrossRef]
- Khalil, N.; Chen, A.; Lee, M.; Czerwinski, S.A.; Ebert, J.R.; DeWitt, J.C.; Kannan, K. Association of Perfluoroalkyl Substances, Bone Mineral Density, and Osteoporosis in the U.S. Population in NHANES 2009–2010. Environ. Health Perspect. 2016, 124, 81–87. [Google Scholar] [CrossRef] [Green Version]
- Di Nisio, A.; De Rocco Ponce, M.; Giadone, A.; Rocca, M.S.; Guidolin, D.; Foresta, C. Perfluoroalkyl substances and bone health in young men: A pilot study. Endocrine 2020, 67, 678–684. [Google Scholar] [CrossRef]
- Gardener, H.; Sun, Q.; Grandjean, P. PFAS concentration during pregnancy in relation to cardiometabolic health and birth outcomes. Environ. Res. 2021, 192, 110287. [Google Scholar] [CrossRef]
- Liew, Z.; Luo, J.; Nohr, E.A.; Bech, B.H.; Bossi, R.; Arah, O.A.; Olsen, J. Maternal Plasma Perfluoroalkyl Substances and Miscarriage: A Nested Case–Control Study in the Danish National Birth Cohort. Environ. Health Perspect. 2020, 128, 047007. [Google Scholar] [CrossRef] [Green Version]
- Preston, E.V.; Webster, T.F.; Claus Henn, B.; McClean, M.D.; Gennings, C.; Oken, E.; Rifas-Shiman, S.L.; Pearce, E.N.; Calafat, A.M.; Fleisch, A.F.; et al. Prenatal exposure to per- and polyfluoroalkyl substances and maternal and neonatal thyroid function in the Project Viva Cohort: A mixtures approach. Environ. Int. 2020, 139, 105728. [Google Scholar] [CrossRef]
- Manea, S.; Salmaso, L.; Lorenzoni, G.; Mazzucato, M.; Russo, F.; Mantoan, D.; Martuzzi, M.; Fletcher, T.; Facchin, P. Exposure to PFAS and small for gestational age new-borns: A birth records study in Veneto Region (Italy). Environ. Res. 2020, 184, 109282. [Google Scholar] [CrossRef]
- Wen, L.-L.; Lin, C.-Y.; Chou, H.-C.; Chang, C.-C.; Lo, H.-Y.; Juan, S.-H. Perfluorooctanesulfonate Mediates Renal Tubular Cell Apoptosis through PPARgamma Inactivation. PLoS ONE 2016, 11, e0155190. [Google Scholar] [CrossRef]
- Hu, W.; Jones, P.D.; Upham, B.L.; Trosko, J.E.; Lau, C.; Giesy, J.P. Inhibition of gap junctional intercellular communication by perfluorinated compounds in rat liver and dolphin kidney epithelial cell lines in vitro and Sprague-Dawley rats in vivo. Toxicol. Sci. 2002, 68, 429–436. [Google Scholar] [CrossRef]
- Aimuzi, R.; Luo, K.; Chen, Q.; Wang, H.; Feng, L.; Ouyang, F.; Zhang, J. Perfluoroalkyl and polyfluoroalkyl substances and fetal thyroid hormone levels in umbilical cord blood among newborns by prelabor caesarean delivery. Environ. Int. 2019, 130, 104929. [Google Scholar] [CrossRef]
- Li, J.; Cai, D.; Chu, C.; Li, Q.; Zhou, Y.; Hu, L.; Yang, B.; Dong, G.; Zeng, X.; Chen, D. Transplacental Transfer of Per- and Polyfluoroalkyl Substances (PFASs): Differences between Preterm and Full-Term Deliveries and Associations with Placental Transporter mRNA Expression. Environ. Sci. Technol. 2020, 54, 5062–5070. [Google Scholar] [CrossRef]
- Spratlen, M.J.; Perera, F.P.; Lederman, S.A.; Robinson, M.; Kannan, K.; Herbstman, J.; Trasande, L. The Association Between Perfluoroalkyl Substances and Lipids in Cord Blood. J. Clin. Endocrinol. Metab. 2020, 105, 43–54. [Google Scholar] [CrossRef]
- National Center for Emerging and Zoonotic Infectious Diseases (NCEZID). Centers for Disease Control and Prevention. Available online: https://www.cdc.gov/onehealth/basics/index.html (accessed on 15 November 2021).
- Domingo, J.L.; Nadal, M. Human exposure to per- and polyfluoroalkyl substances (PFAS) through drinking water: A review of the recent scientific literature. Environ. Res. 2019, 177, 108648. [Google Scholar] [CrossRef]
- Dairy Production and Products: Production. Available online: http://www.fao.org/dairy-production-products/production/en/ (accessed on 4 October 2021).
- Wang, Z. An Assessment Report on Issues of Concern: Chemicals and Waste Issues Posing Risks to Human Health and the Environment; United Nations Environment Programme: Nairobi, Kenya, 2020. [Google Scholar]
- Rajan, A.; Shah, T. Impact of Irrigation on India’s Dairy Economy. Agriculture 2020, 10, 53. [Google Scholar] [CrossRef] [Green Version]
- Sharma, B.M.; Bharat, G.K.; Tayal, S.; Larssen, T.; Bečanová, J.; Karásková, P.; Whitehead, P.G.; Futter, M.N.; Butterfield, D.; Nizzetto, L. Perfluoroalkyl substances (PFAS) in river and ground/drinking water of the Ganges River basin: Emissions and implications for human exposure. Environ. Pollut. 2016, 208, 704–713. [Google Scholar] [CrossRef]
- Yeung, L.W.Y.; Stadey, C.; Mabury, S.A. Simultaneous analysis of perfluoroalkyl and polyfluoroalkyl substances including ultrashort-chain C2 and C3 compounds in rain and river water samples by ultra performance convergence chromatography. J. Chromatogr. A 2017, 1522, 78–85. [Google Scholar] [CrossRef]
- Yeung, L.W.Y.; Yamashita, N.; Taniyasu, S.; Lam, P.K.S.; Sinha, R.K.; Borole, D.V.; Kannan, K. A survey of perfluorinated compounds in surface water and biota including dolphins from the Ganges River and in other waterbodies in India. Chemosphere 2009, 76, 55–62. [Google Scholar] [CrossRef] [PubMed]
- EWG Interactive Map. PFAS Contamination in the U.S. Available online: http://www.ewg.org/interactive-maps/pfas_contamination/map/ (accessed on 29 September 2021).
- Fourth National Report on Human Exposure to Environmental Chemicals Update; Centers for Disease Control and Prevention: Atlanta, Georgia, 2021.
- Buttarazzi, D. Maine Dairy Farmer’s Blood Tests High for ‘Forever Chemicals’ from Toxic Sludge. Available online: https://pfasproject.com/2019/08/16/maine-dairy-farmers-blood-tests-high-for-forever-chemicals-from-toxic-sludge/ (accessed on 29 September 2021).
- FDA. Analytical Results of Testing Food for PFAS from Environmental Contamination. Available online: https://www.fda.gov/food/chemical-contaminants-food/analytical-results-testing-food-pfas-environmental-contamination (accessed on 4 October 2021).
- USDA ARS. Search for an ARS Project. Available online: https://www.ars.usda.gov/research/project/?accnNo=436179 (accessed on 29 September 2021).
- Madison Water Utility. Perfluorinated Compounds. Available online: https://www.cityofmadison.com/water/water-quality/water-quality-testing/perfluorinated-compounds (accessed on 29 September 2021).
- Li, J.; Sun, J.; Li, P. Exposure routes, bioaccumulation and toxic effects of per- and polyfluoroalkyl substances (PFASs) on plants: A critical review. Environ. Int. 2022, 158, 106891. [Google Scholar] [CrossRef] [PubMed]
- Ingelido, A.M.; Abballe, A.; Gemma, S.; Dellatte, E.; Iacovella, N.; De Angelis, G.; Marra, V.; Russo, F.; Vazzoler, M.; Testai, E.; et al. Serum concentrations of perfluorinated alkyl substances in farmers living in areas affected by water contamination in the Veneto Region (Northern Italy). Environ. Int. 2020, 136, 105435. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.W.; Gunderson, K.G.; Green, L.A.; Rediske, R.R.; Steinman, A.D. Perfluoroalkylated Substances (PFAS) Associated with Microplastics in a Lake Environment. Toxics 2021, 9, 106. [Google Scholar] [CrossRef]
- Death, C.; Bell, C.; Champness, D.; Milne, C.; Reichman, S.; Hagen, T. Per- and polyfluoroalkyl substances (PFAS) in livestock and game species: A review. Sci. Total Environ. 2021, 774, 144795. [Google Scholar] [CrossRef]
- Kowalczyk, J.; Ehlers, S.; Oberhausen, A.; Tischer, M.; Fürst, P.; Schafft, H.; Lahrssen-Wiederholt, M. Absorption, Distribution, and Milk Secretion of the Perfluoroalkyl Acids PFBS, PFHxS, PFOS, and PFOA by Dairy Cows Fed Naturally Contaminated Feed. J. Agric. Food Chem. 2013, 61, 2903–2912. [Google Scholar] [CrossRef]
- Vestergren, R.; Orata, F.; Berger, U.; Cousins, I.T. Bioaccumulation of perfluoroalkyl acids in dairy cows in a naturally contaminated environment. Environ. Sci. Pollut. Res. 2013, 20, 7959–7969. [Google Scholar] [CrossRef]
- van Asselt, E.D.; Kowalczyk, J.; van Eijkeren, J.C.H.; Zeilmaker, M.J.; Ehlers, S.; Fürst, P.; Lahrssen-Wiederholt, M.; van der Fels-Klerx, H.J. Transfer of perfluorooctane sulfonic acid (PFOS) from contaminated feed to dairy milk. Food Chem. 2013, 141, 1489–1495. [Google Scholar] [CrossRef]
- Liu, Z.; Lu, Y.; Shi, Y.; Wang, P.; Jones, K.; Sweetman, A.J.; Johnson, A.C.; Zhang, M.; Zhou, Y.; Lu, X.; et al. Crop bioaccumulation and human exposure of perfluoroalkyl acids through multi-media transport from a mega fluorochemical industrial park, China. Environ. Int. 2017, 106, 37–47. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.; Jin, Y.; Quan, X.; Sasaki, K.; Saito, N.; Nakayama, S.F.; Sato, I.; Tsuda, S. Perfluorosulfonates and perfluorocarboxylates in snow and rain in Dalian, China. Environ. Int. 2009, 35, 737–742. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Xu, D.; Lu, M.; Zhou, S.; Peng, T.; Yue, Z.; Zhou, Y. QuEChERs Combined with Online Interference Trapping LC-MS/MS Method for the Simultaneous Determination of 20 Polyfluoroalkane Substances in Dietary Milk. J. Agric. Food Chem. 2015, 63, 4087–4095. [Google Scholar] [CrossRef] [PubMed]
- Xing, Z.; Lu, J.; Liu, Z.; Li, S.; Wang, G.; Wang, X. Occurrence of Perfluorooctanoic Acid and Perfluorooctane Sulfonate in Milk and Yogurt and Their Risk Assessment. Int. J. Environ. Res. Public. Health 2016, 13, 1037. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Macheka, L.R.; Olowoyo, J.O.; Mugivhisa, L.L.; Abafe, O.A. Determination and assessment of human dietary intake of per and polyfluoroalkyl substances in retail dairy milk and infant formula from South Africa. Sci. Total Environ. 2021, 755, 142697. [Google Scholar] [CrossRef] [PubMed]
- Tittlemier, S.A.; Pepper, K.; Seymour, C.; Moisey, J.; Bronson, R.; Cao, X.-L.; Dabeka, R.W. Dietary Exposure of Canadians to Perfluorinated Carboxylates and Perfluorooctane Sulfonate via Consumption of Meat, Fish, Fast Foods, and Food Items Prepared in Their Packaging. J. Agric. Food Chem. 2007, 55, 3203–3210. [Google Scholar] [CrossRef]
- Guruge, K.S.; Manage, P.M.; Yamanaka, N.; Miyazaki, S.; Taniyasu, S.; Yamashita, N. Species-specific concentrations of perfluoroalkyl contaminants in farm and pet animals in Japan. Chemosphere 2008, 73, S210–S215. [Google Scholar] [CrossRef]
- Health Canada. HC (Health Canada) Updates to Health Canada Soil Screening Values for Perfluoroalkylated Substances (PFAS); Health Canada: Ottawa, Canada, 2019; pp. 1–2.
- UNEP. Fifteenth meeting of the Persistent Organic Pollutants Review Committee (POPRC.15). Available online: http://chm.pops.int/theconvention/popsreviewcommittee/meetings/poprc15/overview/tabid/8052/default.aspx (accessed on 5 October 2021).
- EEA relevance. Commission Regulation (EU) 2017/1000 of 13 June 2017 amending Annex XVII to Regulation (EC) No 1907/2006 of the European Parliament and of the Council concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) as regards perfluorooctanoic acid (PFOA), its salts and PFOA-related substances. Off. J. Eur. Union. 2017, 150, 14. [Google Scholar]
- EEA. Directive 2010/75/EU of the European Parliament and of the Council of 24 November 2010 on industrial emissions (integrated pollution prevention and control), Off. J. Eur. Union 2010, 337, 17.
- US EPA. Drinking Water Health Advisories for PFOA and PFOS. Available online: https://www.epa.gov/ground-water-and-drinking-water/drinking-water-health-advisories-pfoa-and-pfos (accessed on 11 October 2021).
- US EPA. PFAS Fact Sheets and Infographics. Available online: https://www.epa.gov/pfas/pfas-fact-sheets-and-infographics (accessed on 29 September 2021).
- Action Levels for PFOS in Cow’s Milk. Available online: https://www.maine.gov/dep/spills/topics/pfas/Derivation-of-Action-Levels-for-PFOS-in-Cows-Milk-03.28.17.pdf (accessed on 12 October 2021).
- Maine Department of Environmental Protection: Remedial Action Guidelines for Contaminated Sites (RAGs). Available online: https://www.maine.gov/dep/spills/publications/guidance/rags/Maine-Remedial-Action-Guidelines-2021-05-01.pdf (accessed on 12 October 2021).
- Bundesinstitut Für Risikobewertung. New Health-Based Guidance Values for the Industrial Chemicals PFOS and PFOA.; German Federal Institute for Risk Assessment: Berlin, Germany, 2019. [CrossRef]
- Country information—OECD Portal on Per and Poly Fluorinated Chemicals. Available online: https://www.oecd.org/chemicalsafety/portal-perfluorinated-chemicals/countryinformation/european-union.htm (accessed on 11 October 2021).
- Ross, I.; McDonough, J.; Miles, J.; Storch, P.; Kochunarayanan, P.T.; Kalve, E.; Hurst, J.; Dasgupta, S.S.; Burdick, J. A review of emerging technologies for remediation of PFASs. Remediat. J. 2018, 28, 101–126. [Google Scholar] [CrossRef]
- Kucharzyk, K.H.; Darlington, R.; Benotti, M.; Deeb, R.; Hawley, E. Novel treatment technologies for PFAS compounds: A critical review. J. Environ. Manag. 2017, 204, 757–764. [Google Scholar] [CrossRef]
- Lu, D.; Sha, S.; Luo, J.; Huang, Z.; Zhang Jackie, X. Treatment train approaches for the remediation of per- and polyfluoroalkyl substances (PFAS): A critical review. J. Hazard. Mater. 2020, 386, 121963. [Google Scholar] [CrossRef]
- Schröder, H.F.R.; José, H.J.; Gebhardt, W.; Moreira, R.F.P.M.; Pinnekamp, J. Biological wastewater treatment followed by physicochemical treatment for the removal of fluorinated surfactants. Water Sci. Technol. 2010, 61, 3208–3215. [Google Scholar] [CrossRef] [PubMed]
- Dobrovolskaia, M.A.; McNeil, S.E. Immunological properties of engineered nanomaterials. Nanosci. Technol. 2007, 2, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Piccinno, F.; Gottschalk, F.; Seeger, S.; Nowack, B. Industrial production quantities and uses of ten engineered nanomaterials in Europe and the world. J. Nanoparticle Res. 2012, 14, 1–11. [Google Scholar] [CrossRef] [Green Version]
- West, J.L.; Halas, N.J. Engineered nanomaterials for biophotonics applications: Improving sensing, imaging, and therapeutics. Annu. Rev. Biomed. Eng. 2003, 5, 285–292. [Google Scholar] [CrossRef] [Green Version]
- Zhang, W.; Zhang, D.; Liang, Y. Nanotechnology in remediation of water contaminated by poly- and perfluoroalkyl substances: A review. Environ. Pollut. 2019, 247, 266–276. [Google Scholar] [CrossRef]
- CRC CARE. Assessment, Management and Remediation for PFOS and PFOA Part 5: Management and Remediation of PFOS and PFOA.; CRC for Contamination Assessment and Remediation of the Environment: Newcastle, Australia, 2017. [Google Scholar]
- Darlington, R.; Barth, E.; McKernan, J. The Challenges of PFAS Remediation. Mil. Eng. 2018, 110, 58–60. [Google Scholar]
- Du, Z.; Deng, S.; Bei, Y.; Huang, Q.; Wang, B.; Huang, J.; Yu, G. Adsorption behavior and mechanism of perfluorinated compounds on various adsorbents—A review. J. Hazard. Mater. 2014, 274, 443–454. [Google Scholar] [CrossRef]
- Merino, N.; Qu, Y.; Deeb, R.A.; Hawley, E.L.; Hoffmann, M.R.; Mahendra, S. Degradation and Removal Methods for Perfluoroalkyl and Polyfluoroalkyl Substances in Water. Environ. Eng. Sci. 2016, 33, 615–649. [Google Scholar] [CrossRef] [Green Version]
- Chowdhury, Z.K. Activated Carbon; Marsh, H., Rodríguez-Reinoso, F., Eds.; Elsevier Science Ltd: Oxford, UK, 2006; ISBN 978-0-08-044463-5. [Google Scholar]
- De Bruecker, T. Status Report PFOS Remediation. DEC Environ. Solut. 2015, 12, 11–14. [Google Scholar]
- PFAS Remediation. Available online: https://www.ventia.com/capabilities/pfas-remediation (accessed on 5 October 2021).
- Sarsby, R.W. Environmental Geotechnics, 2nd ed.; Ice Publishing: London, UK, 2013; ISBN 978-0-7277-4187-5. [Google Scholar]
- Australian Research Council. Selection Report: PFAS Remediation Research Program (SR18) Round One. Available online: https://www.arc.gov.au/grants/grant-outcomes/selection-outcome-reports/selection-report-pfas-remediation-research-program-sr18-round-one (accessed on 5 October 2021).
- PFAS Management Area Plan—RAAF Base Williamtown. Available online: https://defence.gov.au/Environment/PFAS/docs/Williamtown/Reports/201907WilliamtownPMAP.pdf (accessed on 5 October 2021).
- PFAS Management Area Plan—Army Aviation Centre Oakey. Available online: https://defence.gov.au/Environment/PFAS/Docs/Oakey/Reports/AACOakeyPMAP.pdf (accessed on 5 October 2021).
- Sara, S. PFAS in the Baltic Sea Region. Available online: https://portal.helcom.fi/meetings/PRESSURE%207-2017-462/MeetingDocuments/7-7_Att%20Information%20on%20PFAS%20in%20the%20Baltc%20Sea%20Region.pdf (accessed on 5 October 2021).
- Environment Protection Authority Victoria. PFAS National Environmental Management Plan|Environment Protection Authority Victoria. Available online: https://www.epa.vic.gov.au/for-community/environmental-information/pfas/pfas-national-environmental-management-plan (accessed on 22 July 2021).
- Moriwaki, H.; Takagi, Y.; Tanaka, M.; Tsuruho, K.; Okitsu, K.; Maeda, Y. Sonochemical Decomposition of Perfluorooctane Sulfonate and Perfluorooctanoic Acid. Environ. Sci. Technol. 2005, 39, 3388–3392. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Vecitis, C.D.; Park, H.; Mader, B.T.; Hoffmann, M.R. Sonochemical Degradation of Perfluorooctane Sulfonate (PFOS) and Perfluorooctanoate (PFOA) in Groundwater: Kinetic Effects of Matrix Inorganics. Environ. Sci. Technol. 2010, 44, 445–450. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Drees, C.W. Sonochemical Degradation of Perfluorooctane Sulfonate (PFOS). Master’s Thesis, The Ohio State University, Columbus, OH, USA, 2005. [Google Scholar]
- Murugananthan, M.; Yoshihara, S.; Rakuma, T.; Uehara, N.; Shirakashi, T. Electrochemical degradation of 17β-estradiol (E2) at boron-doped diamond (Si/BDD) thin film electrode. Electrochim. Acta 2007, 52, 3242–3249. [Google Scholar] [CrossRef]
- Cañizares, P.; Lobato, J.; Paz, R.; Rodrigo, M.A.; Sáez, C. Electrochemical oxidation of phenolic wastes with boron-doped diamond anodes. Water Res. 2005, 39, 2687–2703. [Google Scholar] [CrossRef]
- Chen, X.; Chen, G.; Yue, P.L. Anodic oxidation of dyes at novel Ti/B-diamond electrodes. Chem. Eng. Sci. 2003, 58, 995–1001. [Google Scholar] [CrossRef]
- Crittenden, J.C.; Trussell, R.R.; Hand, D.W.; Howe, K.J.; Tchobanoglous, G. MWH’s Water Treatment: Principles and Design, 3rd ed.; Crittenden, J.C., Montgomery Watson Harza (Firm), Eds.; ETH Zurich: Zürich, Switzerland, 2012; ISBN 978-0-470-40539-0. [Google Scholar]
- Wang, L.; Batchelor, B.; Pillai, S.D.; Botlaguduru, V.S.V. Electron beam treatment for potable water reuse: Removal of bromate and perfluorooctanoic acid. Chem. Eng. J. 2016, 302, 58–68. [Google Scholar] [CrossRef]
- Ma, S.-H.; Wu, M.-H.; Tang, L.; Sun, R.; Zang, C.; Xiang, J.-J.; Yang, X.-X.; Li, X.; Xu, G. EB degradation of perfluorooctanoic acid and perfluorooctane sulfonate in aqueous solution. Nucl. Sci. Tech. 2017, 28, 1–8. [Google Scholar] [CrossRef]
- Zhang, K.; Huang, J.; Yu, G.; Zhang, Q.; Deng, S.; Wang, B. Destruction of Perfluorooctane Sulfonate (PFOS) and Perfluorooctanoic Acid (PFOA) by Ball Milling. Environ. Sci. Technol. 2013, 47, 6471–6477. [Google Scholar] [CrossRef]
- Javahrrian, M. Bench-Scale VEG Research & Development Study: Implementation Memorandum for Ex-Situ Thermal Desorption of Perfluoroalkyl Compounds (PFCs) in Soils; Endpoint Consulting Pty Ltd.: Sydney, Australia, 2016. [Google Scholar]
- Dickson, M. AusPat Patent Search Homepage. Available online: https://pericles.ipaustralia.gov.au/ols/auspat/applicationDetails.do?applicationNo=2012289835 (accessed on 5 October 2021).
- Dickson, M. Patent Application Publication. Available online: https://patentimages.sthttps://patentimages.storage.googleapis.com/a4/4f/92/6864036f22203d/US20140190896A1.pdf (accessed on 5 October 2021).
- Cao, M.H.; Wang, B.B.; Yu, H.S.; Wang, L.L.; Yuan, S.H.; Chen, J. Photochemical decomposition of perfluorooctanoic acid in aqueous periodate with VUV and UV light irradiation. J. Hazard. Mater. 2010, 179, 1143–1146. [Google Scholar] [CrossRef]
- Marrone, P.A.; Hodes, M.; Smith, K.A.; Tester, J.W. Salt precipitation and scale control in supercritical water oxidation—part B: Commercial/full-scale applications. J. Supercrit. Fluids 2004, 29, 289–312. [Google Scholar] [CrossRef]
- Mitton, D.B.; Eliaz, N.; Cline, J.A.; Latanision, R.M. An Overview of the Current Understanding of Corrosion in SCWO Systems for the Destruction of Hazardous Waste Products. Mater. Technol. 2001, 16, 44–53. [Google Scholar] [CrossRef]
- Jama, Y.; Lindholst, S.; Andreasen, R.R.; Andersen, H.R.; Kokkoli, A.; Svendsen, T.; Cai, Z.; Kragelund, C. PFAS removal from percolate by super critical water oxidation (SCWO). In Proceedings of the 14th DWF Water Research Conference-Abstracts, University of Copenhagen, Frederiksberg, Denmark, 30 January 2020; p. 48. [Google Scholar]
- Zhang, W.; Zhang, D.; Zagorevski, D.V.; Liang, Y. Exposure of Juncus effusus to seven perfluoroalkyl acids: Uptake, accumulation and phytotoxicity. Chemosphere 2019, 233, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Gobelius, L.; Lewis, J.; Ahrens, L. Plant Uptake of Per- and Polyfluoroalkyl Substances at a Contaminated Fire Training Facility to Evaluate the Phytoremediation Potential of Various Plant Species. Environ. Sci. Technol. 2017, 51, 12602–12610. [Google Scholar] [CrossRef]
- Huff, D.K.; Morris, L.A.; Sutter, L.; Costanza, J.; Pennell, K.D. Accumulation of six PFAS compounds by woody and herbaceous plants: Potential for phytoextraction. Int. J. Phytoremediation 2020, 22, 1538–1550. [Google Scholar] [CrossRef] [PubMed]
- Tseng, N.; Wang, N.; Szostek, B.; Mahendra, S. Biotransformation of 6:2 Fluorotelomer Alcohol (6:2 FTOH) by a Wood-Rotting Fungus. Environ. Sci. Technol. 2014, 48, 4012–4020. [Google Scholar] [CrossRef]
- OECD Glossary of Statistical Terms—Biodegradation Definition. Available online: https://stats.oecd.org/glossary/detail.asp?ID=%20203 (accessed on 5 October 2021).
- Butzen, M.L.; Wilkinson, J.T.; McGuinness, S.R.; Amezquita, S.; Peaslee, G.F.; Fein, J.B. Sorption and desorption behavior of PFOS and PFOA onto a Gram-positive and a Gram-negative bacterial species measured using particle-induced gamma-ray emission (PIGE) spectroscopy. Chem. Geol. 2020, 552, 119778. [Google Scholar] [CrossRef]
- Brown, P. Race, Class, and Environmental Health: A Review and Systematization of the Literature. Environ. Res. 1995, 69, 15–30. [Google Scholar] [CrossRef]
- Ringquist, E.J. Assessing evidence of environmental inequities: A meta-analysis. J. Policy Anal. Manag. 2005, 24, 223–247. [Google Scholar] [CrossRef]
- Nelson, J.W.; Scammell, M.K.; Hatch, E.E.; Webster, T.F. Social disparities in exposures to bisphenol A and polyfluoroalkyl chemicals: A cross-sectional study within NHANES 2003-2006. Environ. Health 2012, 11, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Park, S.K.; Peng, Q.; Ding, N.; Mukherjee, B.; Harlow, S.D. Determinants of per- and polyfluoroalkyl substances (PFAS) in midlife women: Evidence of racial/ethnic and geographic differences in PFAS exposure. Environ. Res. 2019, 175, 186–199. [Google Scholar] [CrossRef]
- The PFAS Project Lab. Available online: https://pfasproject.com/conference-presentations/ (accessed on 5 October 2021).
- Streimikiene, D. Environmental indicators for the assessment of quality of life. Intellect. Econ. 2015, 9, 67–79. [Google Scholar] [CrossRef] [Green Version]
- Buekers, J.; Colles, A.; Cornelis, C.; Morrens, B.; Govarts, E.; Schoeters, G. Socio-Economic Status and Health: Evaluation of Human Biomonitored Chemical Exposure to Per- and Polyfluorinated Substances across Status. Int. J. Environ. Res. Public Health 2018, 15, 2818. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ding, N.; Harlow, S.D.; Batterman, S.; Mukherjee, B.; Park, S.K. Longitudinal trends in perfluoroalkyl and polyfluoroalkyl substances among multiethnic midlife women from 1999 to 2011: The Study of Women′s Health Across the Nation. Environ. Int. 2020, 135, 105381. [Google Scholar] [CrossRef] [PubMed]
- Evans, G.W.; Kantrowitz, E. Socioeconomic Status and Health: The Potential Role of Environmental Risk Exposure. Annu. Rev. Public Health 2002, 23, 303–331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohai, P.; Pellow, D.; Roberts, J.T. Environmental Justice. Annu. Rev. Environ. Resour. 2009, 34, 405–430. [Google Scholar] [CrossRef]
- Gochfeld, M.; Burger, J. Disproportionate Exposures in Environmental Justice and Other Populations: The Importance of Outliers. Am. J. Public Health 2011, 101, S53–S63. [Google Scholar] [CrossRef]
- Fernandez-Bou, A.S.; Ortiz-Partida, J.P.; Dobbin, K.B.; Flores-Landeros, H.; Bernacchi, L.A.; Medellín-Azuara, J. Underrepresented, understudied, underserved: Gaps and opportunities for advancing justice in disadvantaged communities. Environ. Sci. Policy 2021, 122, 92–100. [Google Scholar] [CrossRef]
- Banzhaf, S.; Ma, L.; Timmins, C. Environmental Justice: The Economics of Race, Place, and Pollution. J. Econ. Perspect. 2019, 33, 185–208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dobbin, K.; Fencl, A. Institutional Diversity and Safe Drinking Water Provision in the United States. SSRN Electron. J. 2021. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).