Increased Prevalence of Atopic Dermatitis in Children Aged 0–3 Years Highly Exposed to Parabens
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Outcome Assessment
2.3. Exposure Assessment
2.4. Ethics Statement
2.5. Statistical Analysis
3. Results
3.1. Comparisons of Characteristics
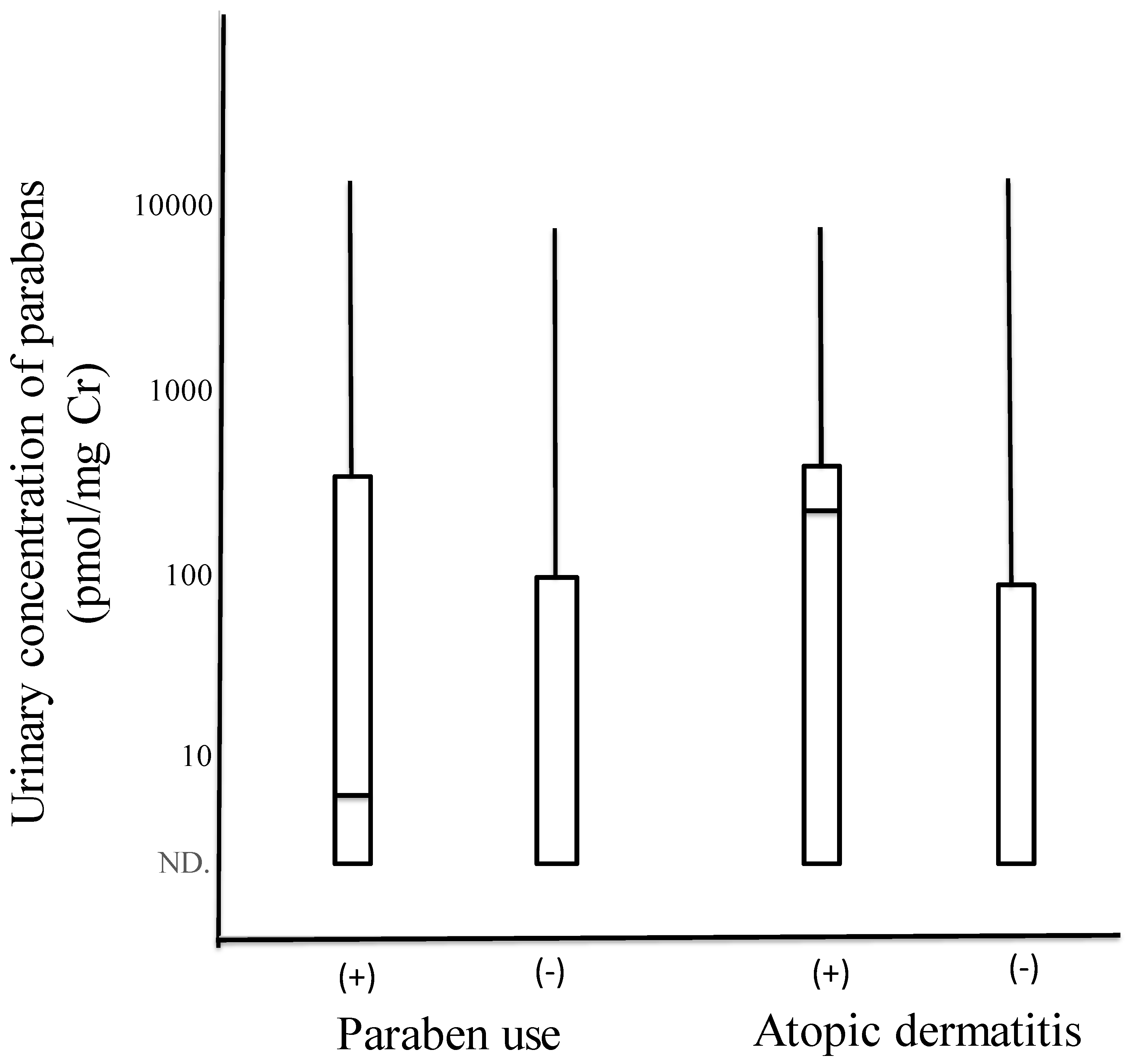
3.2. Comparisons of Distribution of Urinary Concentrations of Parabens
3.3. Comparison of the Prevalence of Allergic Diseases among Age Groups
3.4. Comparisons of the Prevalence of Allergic Diseases between High and Low Exposure to Parabens
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bonamonte, D.; Filoni, A.; Vestita, M.; Romita, P.; Foti, C.; Angelini, G. The Role of the Environmental Risk Factors in the Pathogenesis and Clinical Outcome of Atopic Dermatitis. Biomed. Res. Int. 2019, 2019, 2450605. [Google Scholar] [CrossRef]
- Kuo, C.H.; Kuo, H.U.; Huang, C.H.; Yang, S.N.; Lee, M.S.; Hung, C.H. Early life exposure to antibiotics and the risk of childhood allergic diseases: An update from the perspective of the hygiene hypothesis. J. Microbiol. Immunol. Infect. 2013, 46, 320–329. [Google Scholar] [CrossRef] [Green Version]
- Prokopakis, E.; Vardouniotis, A.; Kawauchi, H.; Scadding, G.; Georgalas, C.; Hellings, P.; Velegrakis, G.; Kalogjera, L. The pathophysiology of the hygiene hypothesis. Int. J. Pediatr. Otorhinolaryngol. 2013, 77, 1065–1071. [Google Scholar] [CrossRef]
- Clayton, E.M.; Todd, M.; Dowd, J.B.; Aiello, A.E. The impact of bisphenol A and triclosan on immune parameters in the U.S. population, NHANES 2003–2006. Environ. Health Perspect. 2011, 119, 390–396. [Google Scholar] [CrossRef]
- Bertelsen, R.J.; Longnecker, M.P.; Løvik, M.; Calafat, A.M.; Carlsen, K.H.; London, S.J.; Carlsen, K.C.L. Triclosan exposure and allergic sensitization in Norwegian children. Allergy 2013, 68, 84–91. [Google Scholar] [CrossRef]
- Spanier, A.J.; Fausnight, T.; Camacho, T.F.; Braun, J.M. The associations of triclosan and paraben exposure with allergen sensitization and wheeze in children. Allergy Asthma Proc. 2014, 35, 475–481. [Google Scholar] [CrossRef] [Green Version]
- Ruszkiewicz, J.A.; Li, S.; Rodriguez, M.B.; Aschner, M. Is Triclosan a neurotoxic agent? J. Toxicol. Environ. Health B Crit. Rev. 2017, 20, 104–117. [Google Scholar] [CrossRef]
- Bledzka, D.; Gromadzinska, J.; Wasowicz, W. Parabens. From environmental studies to human health. Environ. Int. 2014, 67, 27–42. [Google Scholar] [CrossRef]
- Mitsui-Iwama, M.; Yamamoto-Hanada, K.; Fukutomi, Y.; Hirota, R.; Muto, G.; Nakamura, T.; Yoshikawa, T.; Nakamura, H.; Mikami, M.; Morioka, I.; et al. Exposure to paraben and triclosan and allergic diseases in Tokyo: A pilot cross-sectional study. Asia Pac. Allergy 2019, 9, e5. [Google Scholar] [CrossRef]
- Savage, J.H.; Matsui, E.C.; Wood, R.A.; Keet, C.A. Urinary levels of triclosan and parabens are associated with aeroallergen and food sensitization. J. Allergy Clin. Immunol. 2012, 130, 453–460.e7. [Google Scholar] [CrossRef]
- Hafeez, F.; Maibach, H. An overview of parabens and allergic contact dermatitis. Skin Ther. Lett. 2013, 18, 5–7. [Google Scholar] [PubMed]
- Gimenez-Arnau, A.M.; Deza, G.; Bauer, A.; Johnston, G.A.; Mahler, V.; Schuttelaar, M.L.; Sanchez-Perez, J.; Silvestre, J.F.; Wilkinson, M.; Uter, W. Contact allergy to preservatives: ESSCA* results with the baseline series, 2009–2012. J. Eur. Acad. Dermatol. Venereol. 2017, 31, 664–671. [Google Scholar] [CrossRef] [PubMed]
- Saeki, H. Management of Atopic Dermatitis in Japan. J. Nippon Med. Sch. 2017, 84, 2–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shimizu, Y.; Kambayashi, Y.; Tsujiguchi, H.; Hara, A.; Hori, D.; Nguyen, T.T.T.; Suzuki, F.; Hamagishi, T.; Yamada, Y.; Nakamura, H.; et al. Relationship between the use of parabens and allergic diseases in Japanese adults—A cross sectional study. J.-Multidiscip. Sci. J. 2018, 1, 148–155. [Google Scholar] [CrossRef] [Green Version]
- Lee-Sarwar, K.; Hauser, R.; Calafat, A.M.; Ye, X.; O’Connor, G.T.; Sandel, M.; Bacharier, L.B.; Zeiger, R.S.; Laranjo, N.; Gold, D.R.; et al. Prenatal and early-life triclosan and paraben exposure and allergic outcomes. J. Allergy Clin. Immunol. 2018, 142, 269–278.e15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fisher, M.; MacPherson, S.; Braun, J.M.; Hauser, R.; Walker, M.; Feeley, M.; Mallick, R.; Bérubé, R.; Arbuckle, T.E. Paraben Concentrations in Maternal Urine and Breast Milk and Its Association with Personal Care Product Use. Environ. Sci. Technol. 2017, 51, 4009–4017. [Google Scholar] [CrossRef]
- Leppert, B.; Strunz, S.; Seiwert, B.; Schlittenbauer, L.; Schlichting, R.; Pfeiffer, C.; Röder, S.; Bauer, M.; Borte, M.; Stangl, G.I.; et al. Maternal paraben exposure triggers childhood overweight development. Nat. Commun. 2020, 11, 561. [Google Scholar] [CrossRef]
- Hajizadeh, Y.; Feizabadi, G.K.; Feizi, A.; Ebrahimpour, K. The association of personal care products uses and dietary habits with the urinary concentration of parabens in Iranian adults. Int. J. Environ. Health Res. 2020, 1–17. [Google Scholar] [CrossRef]
- Vindenes, H.K.; Svanes, C.; Lygre, S.H.L.; Real, F.G.; Ringel-Kulka, T.; Bertelsen, R.J. Exposure to environmental phenols and parabens, and relation to body mass index, eczema and respiratory outcomes in the Norwegian RHINESSA study. Environ. Health 2021, 20, 81. [Google Scholar] [CrossRef]
- Berger, K.; Eskenazi, B.; Balmes, J.; Holland, N.; Calafat, A.M.; Harley, K.G. Associations between prenatal maternal urinary concentrations of personal care product chemical biomarkers and childhood respiratory and allergic outcomes in the CHAMACOS study. Environ. Int. 2018, 121 Pt 1, 538–549. [Google Scholar] [CrossRef]
- Overgaard, L.E.; Main, K.M.; Frederiksen, H.; Stender, S.; Szecsi, P.B.; Williams, H.C.; Thyssen, J.P. Children with atopic dermatitis and frequent emollient use have increased urinary levels of low-molecular-weight phthalate metabolites and parabens. Allergy 2017, 2017, 13157. [Google Scholar] [CrossRef]
- Jackson-Browne, M.S.; Henderson, N.; Patti, M.; Spanier, A.; Braun, J.M. The Impact of Early-Life Exposure to Antimicrobials on Asthma and Eczema Risk in Children. Curr. Environ. Health Rep. 2019, 6, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Thürmann, L.; Herberth, G.; Seiwert, B.; Schlittenbauer, L.; Rolle-Kampczyk, U.; Röder, S.; Sack, U.; Borte, M.; Bergen, M.; Trump, S.; et al. Prenatal paraben exposure and atopic dermatitis-related outcomes among children. Allergy 2021. [Google Scholar] [CrossRef]
- Guo, J.; Wu, C.; Lu, D.; Jiang, S.; Liang, W.; Chang, X.; Xu, H.; Wang, G.; Zhou, Z. Urinary paraben concentrations and their associations with anthropometric measures of children aged 3 years. Environ. Pollut. 2017, 222, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Chevrier, J. Exposure to parabens and prevalence of obesity and metabolic syndrome: An analysis of the Canadian Health Measures Survey. Sci. Total Environ. 2020, 713, 135116. [Google Scholar] [CrossRef]
- Lee, Y.; Lee, E.; Yon, D.K.; Jee, H.M.; Baek, H.S.; Lee, S.W.; Cho, J.; Han, M.Y. The potential pathways underlying the association of propyl-paraben exposure with aeroallergen sensitization and EASI score using metabolomics analysis. Sci. Rep. 2021, 11, 3772. [Google Scholar] [CrossRef]
- Quiros-Alcala, L.; Hansel, N.N.; McCormack, M.C.; Matsui, E.C. Paraben exposures and asthma-related outcomes among children from the US general population. J. Allergy Clin. Immunol. 2019, 143, 948–956.e4. [Google Scholar] [CrossRef]
- Honda, M.; Robinson, M.; Kannan, K. Parabens in human urine from several Asian countries, Greece, and the United States. Chemosphere 2018, 201, 13–19. [Google Scholar] [CrossRef]
- Murawski, A.; Tschersich, C.; Rucic, E.; Schwedler, G.; Moos, R.K.; Kasper-Sonnenberg, M.; Brüning, T.; Koch, H.M.; Kolossa-Gehring, M. Parabens in urine of children and adolescents in Germany—Human biomonitoring results of the german environmental survey 2014–2017 (GerES V). Environ. Res. 2021, 194, 110502. [Google Scholar] [CrossRef]
- Hong, S.; Jeon, H.L.; Lee, J.; Kim, S.; Lee, C.; Lee, S.; Cho, Y.H.; Choi, K.; Yoo, J. Urinary parabens and their potential sources of exposure among Korean children and adolescents: Korean National Environmental Health Survey 2015–2017. Int. J. Hyg. Environ. Health 2021, 236, 113781. [Google Scholar] [CrossRef]
| Boy (N = 122) | Girl (N = 114) | |
|---|---|---|
| Age (month (mean ± SD)) | 20.42 ± 16.66 | 25.72 ± 17.36 * |
| Height (cm (mean ± SD)) | 78.81 ± 14.44 | 82.08 ± 15.25 |
| Weight (kg (mean ± SD)) | 10.61 ± 3.67 | 11.36 ± 3.75 |
| Kaup’s index (kg/m2 (mean ± SD)) | 16.75 ± 1.68 | 16.32 ± 1.36 * |
| Number with passive smoking (percentage) | 41 (34.2%) | 34 (29.8%) |
| Food allergies (number (prevalence)) | 4 (3.3%) | 7 (6.1%) |
| Bronchial asthma (number (prevalence)) | 10 (8.2%) | 11 (9.6%) |
| Nasal allergies (number (prevalence)) | 1 (0.8%) | 3 (2.6%) |
| Atopic dermatitis (number (prevalence)) | 16 (13.1%) | 15 (13.2%) |
| Total allergies (number (prevalence)) | 27 (22.1%) | 31 (27.2%) |
| Number using parabens (percentage) | 36 (29.5%) | 46 (40.4%) |
| Number with high urinary concentrations of parabens (percentage) | 35 (28.7%) | 26 (22.8%) |
| Age Group (Number) | 4 Months (N = 77) | 18 Months (N = 60) | 36 Months (N = 99) | p Value |
|---|---|---|---|---|
| Number of boys (percentage) | 45 (58.4%) | 36 (60.0%) | 41 (41.4%) | 0.026 |
| Age (month (mean ± SD)) | 3.12 ± 0.32 | 17.25 ± 3.90 | 41.9 ± 2.48 | 0.000 |
| Height (cm (mean ± SD)) | 62.11 ± 2.37 | 79.08 ± 2.87 | 95.59 ± 3.87 | 0.000 |
| Weight (kg (mean ± SD)) | 6.66 ± 0.82 | 10.53 ± 0.99 | 14.58 ± 1.96 | 0.000 |
| Kaup’s index (kg/m2 (mean ± SD)) | 17.23 ± 1.57 | 16.9 ± 1.19 | 15.8 ± 1.40 | 0.000 |
| Number with passive smoking (percentage) | 22 (28.6%) | 18 (30.0%) | 35 (36.1%) | 0.534 |
| Food allergies (number (prevalence)) | 0 (0.0%) | 0 (0.0%) | 4 (4.0%) | - |
| Bronchial asthma (number (prevalence)) | 0 (0.0%) | 6 (10.0%) | 15 (15.2%) | 0.002 |
| Nasal allergies (number (prevalence)) | 0 (0.0%) | 0 (0.0%) | 4 (4.0%) | - |
| Atopic dermatitis (number (prevalence)) | 15 (19.5%) | 4 (6.7%) | 12 (12.1%) | 0.082 |
| Number using parabens (percentage) | 11 (14.3%) | 20 (33.3%) | 51 (51.5%) | 0.000 |
| Number with high urinary concentrations of parabens (percentage) | 23 (29.9%) | 17 (28.3%) | 21 (21.2%) | 0.380 |
| Paraben Use (Number) | (Paraben Use +) (N = 82) | (Paraben Use −) (N = 154) | p Value |
|---|---|---|---|
| Number of boys (percentage) | 36 (43.9%) | 86 (55.8%) | 0.082 |
| Age (month (mean ± SD)) | 30.44 ± 15.50 | 19.01 ± 16.73 | 0.000 |
| Kaup’s index (kg/m2 (mean ± SD)) | 16.24 ± 1.47 | 16.71 ± 1.57 | 0.029 |
| Number with passive smoking (percentage) | 23 (28.4%) | 52 (33.7%) | 0.385 |
| Food allergies (number (prevalence)) | 2 (2.4%) | 2 (1.3%) | - |
| Bronchial asthma (number (prevalence)) | 10 (12.2%) | 11 (7.1%) | 0.194 |
| Nasal allergies (number (prevalence)) | 1 (1.2%) | 3 (1.9%) | - |
| Atopic dermatitis (number (prevalence)) | 18 (22.0%) | 13 (8.4%) | 0.003 |
| Number with high urinary concentrations of parabens (percentage) | 27 (32.9%) | 34 (22.1%) | 0.082 |
| Urinary Concentrations of Parabens | High (Higher than 100 (pmol/Cr, N = 61)) | Low (Lower than 100 (pmol/Cr, N = 175)) | p Value |
|---|---|---|---|
| Number of boys (percentage) | 35 (57.4%) | 87 (49.7%) | 0.304 |
| Age (month (mean ± SD)) | 19.79 ± 17.45 | 24.09 ± 16.98 | 0.092 |
| Kaup’s index (kg/m2 (mean ± SD)) | 16.72 ± 1.61 | 16.48 ± 1.52 | 0.308 |
| Number with passive smoking (percentage) | 17 (28.3%) | 58 (33.3%) | 0.476 |
| Food allergies (number (prevalence)) | 0 (0.0%) | 4 (2.3%) | - |
| Bronchial asthma (number (prevalence)) | 6 (9.8%) | 15 (8.6%) | 0.765 |
| Nasal allergies (number (prevalence)) | 0 (0.0%) | 4 (2.3%) | - |
| Atopic dermatitis (number (prevalence)) | 18 (29.5%) | 13 (7.4%) | 0.000 |
| Number using parabens (percentage) | 27 (44.3%) | 55 (31.4%) | 0.082 |
| β (SE) | Exp (β) (95% Confidence Interval) | p Value | |
|---|---|---|---|
| Boy | −0.154 (0.411) | 0.857 (0.383–1.919) | 0.708 |
| Age | 0.007 (0.013) | 1.007 (0.981–1.033) | 0.602 |
| Kaup’s index | 0.013 (0.143) | 1.013 (0.765–1.341) | 0.928 |
| Passive smoking | −0.067 (0.445) | 0.935 (0.390–2.238) | 0.880 |
| Group with high urinary concentrations | 1.609 (0.407) | 4.995 (2.248–11.099) | 0.000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arafune, J.; Tsujiguchi, H.; Hara, A.; Shimizu, Y.; Hori, D.; Nguyen, T.T.T.; Suzuki, F.; Hamagishi, T.; Yamada, Y.; Nakamura, H.; et al. Increased Prevalence of Atopic Dermatitis in Children Aged 0–3 Years Highly Exposed to Parabens. Int. J. Environ. Res. Public Health 2021, 18, 11657. https://doi.org/10.3390/ijerph182111657
Arafune J, Tsujiguchi H, Hara A, Shimizu Y, Hori D, Nguyen TTT, Suzuki F, Hamagishi T, Yamada Y, Nakamura H, et al. Increased Prevalence of Atopic Dermatitis in Children Aged 0–3 Years Highly Exposed to Parabens. International Journal of Environmental Research and Public Health. 2021; 18(21):11657. https://doi.org/10.3390/ijerph182111657
Chicago/Turabian StyleArafune, Johichi, Hiromasa Tsujiguchi, Akinori Hara, Yukari Shimizu, Daisuke Hori, Thao Thi Thu Nguyen, Fumihiko Suzuki, Toshio Hamagishi, Yohei Yamada, Haruki Nakamura, and et al. 2021. "Increased Prevalence of Atopic Dermatitis in Children Aged 0–3 Years Highly Exposed to Parabens" International Journal of Environmental Research and Public Health 18, no. 21: 11657. https://doi.org/10.3390/ijerph182111657
APA StyleArafune, J., Tsujiguchi, H., Hara, A., Shimizu, Y., Hori, D., Nguyen, T. T. T., Suzuki, F., Hamagishi, T., Yamada, Y., Nakamura, H., Yoshikawa, T., Hayashi, K., Shibata, A., Fukutomi, Y., Ohya, Y., Yamamoto-Hanada, K., Muto, G., Hirota, R., Konoshita, T., ... Nakamura, H. (2021). Increased Prevalence of Atopic Dermatitis in Children Aged 0–3 Years Highly Exposed to Parabens. International Journal of Environmental Research and Public Health, 18(21), 11657. https://doi.org/10.3390/ijerph182111657






