Factors Associated with the Detection of Inappropriate Prescriptions in Older People: A Prospective Cohort
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Subjects
2.2. Data Collected
2.3. Medication Review
2.4. Inappropriate Prescription (IP)
2.5. Sample Size
2.6. Statistical Methods
3. Results
3.1. Subject Baseline Data
3.2. Data of IPs
3.3. Univariate Analysis and Multivariate Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- European Commission Directorate-General for Economic and Financial Affairs. The 2015 Ageing Report: Economic and budgetary projections for the 28 EU Member States (2013–2060). Econ. Financ. Afbf. 2015, 399. [CrossRef]
- Global Health and Aging, National Institutes of Health, U.S. Department of Health and Human Services; World Health Organization: Bethesda, MD, USA, 2011.
- Medicines Optimisation: The Safe and Effective Use of Medicines to Enable the Best Possible Outcomes; NICE Guideline: London, UK, 2015.
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in Older Adults: Evidence for a Phenotype. J. Gerontol. Ser. A Biol. Sco. Med. Sci. 2001, 56, M146–M157. [Google Scholar] [CrossRef] [PubMed]
- Herr, M.; Sirven, N.; Grondin, H.; Pichetti, S.; Sermet, C. Frailty, polypharmacy, and potentially inappropriate medications in old people: Findings in a representative sample of the French population. Eur. J. Clin. Pharmacol. 2017, 73, 1165–1172. [Google Scholar] [CrossRef] [Green Version]
- Duerden, M.; Avery, T.; Payne, R. Polypharmacy and Medicines Optimisation. Making It Safe and Sound Authors; The King’s Fund: London, UK, 2013. [Google Scholar]
- Gnjidic, D.; Hilmer, S.; Blyth, F.M.; Naganathan, V.; Waite, L.; Seibel, M.; McLachlan, A.; Cumming, R.; Handelsman, D.J.; Le Couteur, D. Polypharmacy cutoff and outcomes: Five or more medicines were used to identify community-dwelling older men at risk of different adverse outcomes. J. Clin. Epidemiol. 2012, 65, 989–995. [Google Scholar] [CrossRef]
- Hilmer, S.; Gnjidic, D. Prescribing for frail older people. Aust. Prescr. 2017, 40, 174–178. [Google Scholar] [CrossRef] [Green Version]
- Spinewine, A.; Schmader, K.E.; Barber, N.; Hughes, C.; Lapane, K.L.; Swine, C.; Hanlon, J.T. Appropriate prescribing in elderly people: How well can it be measured and optimised? Lancet 2007, 370, 173–184. [Google Scholar] [CrossRef]
- Scott, I.A.; Hilmer, S.N.; Reeve, E.; Potter, K.; Le Couteur, D.; Rigby, D.; Gnjidic, D.; Del Mar, C.B.; Roughead, E.E.; Page, A.; et al. Reducing Inappropriate Polypharmacy. JAMA Intern. Med. 2015, 175, 827. [Google Scholar] [CrossRef] [Green Version]
- Fernández, A.; Gómez, F.; Curcio, C.L.; Pineda, E.; de Souza, J.F. Prevalence and impact of potentially inappropriate medication on community dwelling older adults. Biomedica 2020, 41, 1–30. [Google Scholar]
- Reallon, E.; Chavent, B.; Gervais, F.; Dauphinot, V.; Vernaudon, J.; Krolak-Salmon, P.; Mouchoux, C.; Novais, T. Medication exposure and frailty in older community-dwelling patients: A cross-sectional study. Int. J. Clin. Pharm. 2020, 42, 508–514. [Google Scholar] [CrossRef] [PubMed]
- Sevilla-Sanchez, D.; Molist-Brunet, N.; Amblàs-Novellas, J.; Roura-Poch, P.; Espaulella-Panicot, J.; Codina-Jané, C. Adverse drug events in patients with advanced chronic conditions who have a prognosis of limited life expectancy at hospital admission. Eur. J. Clin. Pharmacol. 2017, 73, 79–89. [Google Scholar] [CrossRef]
- Hernández-Rodríguez, M.Á.; Sempere-Verdú, E.; Vicens, C.; González-Rubio, F.; Miguel-García, F.; Palop-Larrea, V.; Orueta-Sánchez, R.; Esteban-Jiménez, Ó.; Sempere-Manuel, M.; Arroyo-Aniés, M.P.; et al. Evolution of polypharmacy in a spanish population (2005–2015): A database study. Pharmacoepidemiol. Drug Saf. 2020, 29, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Yap, A.F.; Thirumoorthy, T.; Kwan, Y.H. Systematic review of the barriers affecting medication adherence in older adults. Geriatr. Gerontol. Int. 2015, 16, 1093–1101. [Google Scholar] [CrossRef]
- Woodford, H.J.; Fisher, J. New horizons in deprescribing for older people. Age Ageing 2019, 48, 768–775. [Google Scholar] [CrossRef]
- Hanlon, J.T.; Artz, M.B.; Pieper, C.F.; Lindblad, C.I.; Sloane, R.J.; Ruby, C.M.; Schmader, K.E. Inappropriate Medication Use Among Frail Elderly Inpatients. Ann. Pharmacother. 2004, 38, 9–14. [Google Scholar] [CrossRef]
- Poudel, A.; Peel, N.; Mitchell, C.; Nissen, L.; Hubbard, R. A systematic review of prescribing criteria to evaluate appropriateness of medications in frail older people. Rev. Clin. Gerontol. 2014, 24, 304–318. [Google Scholar] [CrossRef]
- Espaulella-Panicot, J.; Molist-Brunet, N.; Sevilla-Sánchez, D.; González-Bueno, J.; Amblàs-Novellas, J.; Solà-Bonada, N.; Codina, J. Patient-centred prescription model to improve adequate prescription and therapeutic adherence in patients with multiple disorders. Rev. Esp. Geriatr. Gerontol. 2017, 52, 278–281. [Google Scholar] [CrossRef]
- Boyd, K.; A Murray, S. Recognising and managing key transitions in end of life care. BMJ 2010, 341, c4863. [Google Scholar] [CrossRef] [PubMed]
- Granger, C.V.; Albretch, G.L.H. Outcome of comprehensive medical rehabilitation: Measurement by PULSES profile and the Barthel index. Arch. Phys. Med. Rehabil. 1979, 60, 145–154. [Google Scholar] [PubMed]
- Salisbury, C.; Johnson, L.; Purdy, S.; Valderas, J.M.; Montgomery, A. Epidemiology and impact of multimorbidity in primary care: A retrospective cohort study. Br. J. Gen. Pr. 2011, 61, e12–e21. [Google Scholar] [CrossRef] [PubMed]
- Charlson, M.E.; Pompei, P.; Ales, K.L.; Mackenzie, R. A New Method of Classifying Prognostic in Longitudinal Studies: Development. J. Chronic Dis. 1987, 40, 373–383. [Google Scholar] [CrossRef]
- Reisberg, B.; Ferris, S.; de Leon, M.; Crook, T. The Global Deterioration Scale for assessment of primary degenerative dementia. Am. J. Psychiatry 1982, 139, 1136–1139. [Google Scholar] [CrossRef] [Green Version]
- George, J.; Phun, Y.-T.; Bailey, M.J.; Kong, D.C.; Stewart, K. Development and Validation of the Medication Regimen Complexity Index. Ann. Pharmacother. 2004, 38, 1369–1376. [Google Scholar] [CrossRef] [PubMed]
- Hilmer, S.N.; Mager, D.E.; Simonsick, E.M.; Cao, Y.; Ling, S.M.; Windham, B.G.; Harris, T.B.; Hanlon, J.T.; Rubin, S.M.; Shorr, R.I.; et al. A Drug Burden Index to Define the Functional Burden of Medications in Older People. Arch. Intern. Med. 2007, 167, 781–787. [Google Scholar] [CrossRef] [Green Version]
- Amblàs-Novellas, J.; Martori, J.C.; Espaulella, J.; Oller, R.; Molist-Brunet, N.; Inzitari, M.; Romero-Ortuno, R. Frail-VIG index: A concise frailty evaluation tool for rapid geriatric assessment. BMC Geriatr. 2018, 18, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amblàs-Novellas, J.; Martori, J.C.; Molist-Brunet, N.; Oller, R.; Gómez-Batiste, X. Frail-VIG index: Design and evaluation of a new frailty index based on the Comprehensive Geriatric Assessment. Rev. Esp. Geriatr. Gerontol. 2016, 52, 119–127. [Google Scholar] [CrossRef]
- Gómez-Batiste, X.; Martínez-Muñoz, M.; Blay, C.; Amblàs, J.; Vila, L.; Costa, X.; Espaulella, J.; Espinosa, J.; Constante, C.; Mitchell, G.K. Prevalence and characteristics of patients with advanced chronic conditions in need of palliative care in the general population: A cross-sectional study. Palliat. Med. 2014, 28, 302–311. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Kim, C.M. Individualizing Prevention for Older Adults. J. Am. Geriatr. Soc. 2018, 66, 229–234. [Google Scholar] [CrossRef]
- O’Mahony, D.; O’Connor, M.N. Pharmacotherapy at the end-of-life. Age Ageing 2011, 40, 419–422. [Google Scholar] [CrossRef] [Green Version]
- Curtin, D.; Gallagher, P.; O’Mahony, D. Deprescribing in older people approaching end-of-life: Development and validation of STOPPFrail version. Age Ageing 2021, 50, 465–471. [Google Scholar] [CrossRef]
- American Diabetes Association. 12-Older Adults: Standards of Medical Care in Diabetes-2021. Am. Diabetes Assoc. Diabetes Care 2021, 44, S168–S179. [Google Scholar] [CrossRef]
- Gómez-Huelgas, R.; Peralta, F.G.; Mañas, L.R.; Formiga, F.; Domingo, M.P.; Bravo, J.M.; Miranda, C.; Ena, J. Treatment of type 2 diabetes mellitus in elderly patients. Rev. Clín. Española 2018, 218, 74–88. [Google Scholar] [CrossRef]
- Hambling, C.E.; Khunti, K.; Cos, X.; Wens, J.; Martinez, L.; Topsever, P.; Del Prato, S.; Sinclair, A.; Schernthaner, G.; Rutten, G.; et al. Factors influencing safe glucose-lowering in older adults with type 2 diabetes: A PeRsOn-centred ApproaCh To IndiVidualisEd (PROACTIVE) Glycemic Goals for older people. Prim. Care Diabetes 2019, 13, 330–352. [Google Scholar] [CrossRef]
- O’Mahony, D.; O’Sullivan, D.; Byrne, S.; O’Connor, M.N.; Ryan, C.; Gallagher, P. STOPP/START criteria for potentially inappropriate prescribing in older people: Version. Age Ageing 2014, 44, 213–218. [Google Scholar] [CrossRef] [Green Version]
- Gomez-Peralta, F.; Abreu, C.; Lecube, A.; Bellido, D.; Soto, A.; Morales, C.; Brito-Sanfiel, M.; Umpierrez, G. Practical Approach to Initiating SGLT2 Inhibitors in Type 2 Diabetes. Diabetes Ther. 2017, 8, 953–962. [Google Scholar] [CrossRef]
- National Institute for Health and Carae Excellence (NICE). Hypertension in Adults: Diagnosis and Management; NICE: London, UK, 2016. [Google Scholar]
- Morley, J.E. Inappropriate Drug Prescribing and Polypharmacy Are Major Causes of Poor Outcomes in Long-Term Care. J. Am. Med. Dir. Assoc. 2014, 15, 780–782. [Google Scholar] [CrossRef]
- Boockvar, K.S.; Song, W.; Lee, S.; Intrator, O. Hypertension Treatment in US Long-Term Nursing Home Residents with and Without Dementia. J. Am. Geriatr. Soc. 2019, 67, 2058–2064. [Google Scholar] [CrossRef] [PubMed]
- Onder, G.; Vetrano, D.L.; Marengoni, A.; Bell, J.S.; Johnell, K.; Palmer, K. Accounting for frailty when treating chronic diseases. Eur. J. Intern. Med. 2018, 56, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Van der Steen, J.T.; Radbruch, L.; Hertogh, C.M.; de Boer, M.E.; Hughes, J.C.; Larkin, P.; Francke, A.L.; Jünger, S.; Gove, D.; Firth, P.; et al. White paper defining optimal palliative care in older people with dementia: A Delphi study and recommendations from the European Association for Palliative Care. Palliat Med. 2014, 28, 197–209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bjerre, L.M.; Farrell, B.; Hogel, M.; Graham, L.; Lemay, G.; McCarthy, L.; Raman-Wilms, L.; Rojas-Fernandez, C.; Sinha, S.; Thompson, W.; et al. Deprescribing antipsychotics for behavioural and psychological symptoms of dementia and insomnia: Evidence-based clinical practice guideline. Can. Fam Physician 2018, 64, 17–27. [Google Scholar] [PubMed]
- Fick, D.M.; Semla, T.P.; Steinman, M.; Beizer, J.; Brandt, N.; Dombrowski, R.; DuBeau, C.E.; Pezzullo, L.; Epplin, J.J.; Flanagan, N. American Geriatrics Society 2019 Updated AGS Beers Criteria® for Potentially Inappropriate Medication Use in Older Adults. J. Am. Geriatr Soc. 2019, 67, 674–694. [Google Scholar]
- Gokula, M.; Holmes, H.M. Tools to Reduce Polypharmacy. Clin. Geriatr. Med. 2012, 28, 323–341. [Google Scholar] [CrossRef]
- Scottish Intercollegiate Guidelines Network (SIGN). Management of Chronic Pain. A National Clinical Guideline; Healthcare Improvement Scotland: Scotland, UK, 2019. [Google Scholar]
- Récoché, I.; Lebaudy, C.; Cool, C.; Sourdet, S.; Piau, A.; Lapeyre-Mestre, M.; Vellas, B.; Cestac, P. Potentially inappropriate prescribing in a popu-lation of frail elderly people. Int. J. Clin. Pharm. 2017, 39, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Castell Alcalá, M.V.; Otero Puime, Á.; Sánchez Santos, M.T.; Garrido Barral, A.; González Montalvo, J.I.; Zunzunegui, M.V. Prevalencia de fragilidad en una población urbana de mayores de 65 años y su relación con comorbilidad y discapacidad. Aten Primaria. 2010, 42, 520–527. [Google Scholar] [CrossRef] [Green Version]
- Liew, T.M.; Lee, C.S.; Goh, S.K.L.; Chang, Z.Y. The prevalence and impact of potentially inappropriate prescribing among older persons in primary care settings: Multilevel meta-analysis. Age Ageing 2020, 49, 570–579. [Google Scholar] [CrossRef]
- Poudel, A.; Peel, N.M.; Nissen, L.M.; Mitchell, C.A.; Gray, L.C.; Hubbard, R.E. Adverse Outcomes in Relation to Polypharmacy in Robust and Frail Older Hospital Patients. J. Am. Med. Dir. Assoc. 2016, 17, 767.e9–767.e13. [Google Scholar] [CrossRef] [Green Version]
- Bonaga, B.; Sánchez-Jurado, P.M.; Martínez-Reig, M.; Ariza, G.; Rodríguez-Mañas, L.; Gnjidic, D.; Salvador, T.; Abizanda, P. Frailty, Polypharmacy, and Health Outcomes in Older Adults: The Frailty and Dependence in Albacete Study. J. Am. Med. Dir. Assoc. 2018, 19, 46–52. [Google Scholar] [CrossRef] [Green Version]
- Griese-Mammen, N.; Hersberger, K.E.; Messerli, M.; Leikola, S.; Horvat, N.; Van Mil, J.W.F.; Kos, M. PCNE definition of medication review: Reaching agreement. Int. J. Clin. Pharm. 2018, 40, 1199–1208. [Google Scholar] [CrossRef]
- Molist Brunet, N.; Espaulella Panicot, J.; Sevilla-Sánchez, D.; Amblàs Novellas, J.; Codina Jané, C.; Altimiras Roset, J.; et al. A patient-centered prescription model assessing the appropriateness of chronic drug therapy in older patients at the end of life. Eur. Geriatr. Med. 2015, 6, 565–569. [Google Scholar] [CrossRef]
- Budnitz, D.S.; Lovegrove, M.C.; Shehab, N.; Richards, C.L. Emergency Hospitalizations for Adverse Drug Events in Older Americans. N. Engl. J. Med. 2011, 365, 2002–2012. [Google Scholar] [CrossRef]


| Target | Patients | ||
|---|---|---|---|
| Healthy Older Adults * | Frail Older Adults † | Older Adults in a Probable EOL Situation ‡ | |
| Qualitative Glycaemic | Similar to those for diabetic young adults | Assess the decrease of therapeutic intensity | Quality of life preservation ** |
| Quantitative Hba1c¶ | ≤7–7.5% | ≤8.0% | Avoid reliance on A1C ** |
| Therapeutic Goal †† | Prolong survival | Maintain functionality | Symptomatic treatment |
| Baseline Data | Total N = 428 | |
|---|---|---|
| Demographic Data | ||
| Age, mean (SD) | 85.52 (7.67) | |
| Gender, N (%) | Men | 143 (33.4%) |
| Women | 285 (66.6%) | |
| Origin, N (%) | Home | 210 (49.1%) |
| Nursing Home | 218 (50.9%) | |
| Clinical, Functional and Cognitive Data | ||
| Medication self-management * | 58 (27.6%) | |
| Barthel Index (BI), mean (SD) | 49.93 (32.14) | |
| BI (degrees) | Independence (BI ≥ 95) | 51 (11.9%) |
| Mild dependence (BI 90–65) | 120 (28.0%) | |
| Moderate dependence (BI 60–25) | 129 (30.2%) | |
| Severe dependence (BI ≤ 20) | 128 (29.9%) | |
| Cognitive status | No dementia | 112 (26.2%) |
| Mild dementia | 62 (14.5%) | |
| Moderate dementia (GDS 5 to GDS 6B) | 112 (26.2%) | |
| Advanced dementia (from GS 6C) | 142 (33.1%) | |
| Geriatric Syndromes (GS), mean (SD) | 2.92 (1.52) | |
| Type of GS | Falls | 144 (33.6%) |
| Dysphagia | 84 (19.6%) | |
| Pain | 99 (23.1%) | |
| Depressive syndrome | 198 (46.3%) | |
| Insomnia | 229 (53.5%) | |
| Morbidities, mean (SD) | 4.91 (2.16) | |
| Morbidities (number) | 1–2 | 43 (10.0%) |
| 3–4 | 168 (39.3%) | |
| 5 or more | 217 (50.7%) | |
| Morbidities (type) | Hypertension | 290 (67.8%) |
| Chronic renal failure | 186 (43.5%) | |
| Type 2 Diabetes | 110 (25.7%) | |
| Heart Failure | 88 (20.6%) | |
| Charlson Index, mean (SD) | 3.26 (2.27) | |
| Frailty (FI), mean (SD) | 0.39 (0.13) | |
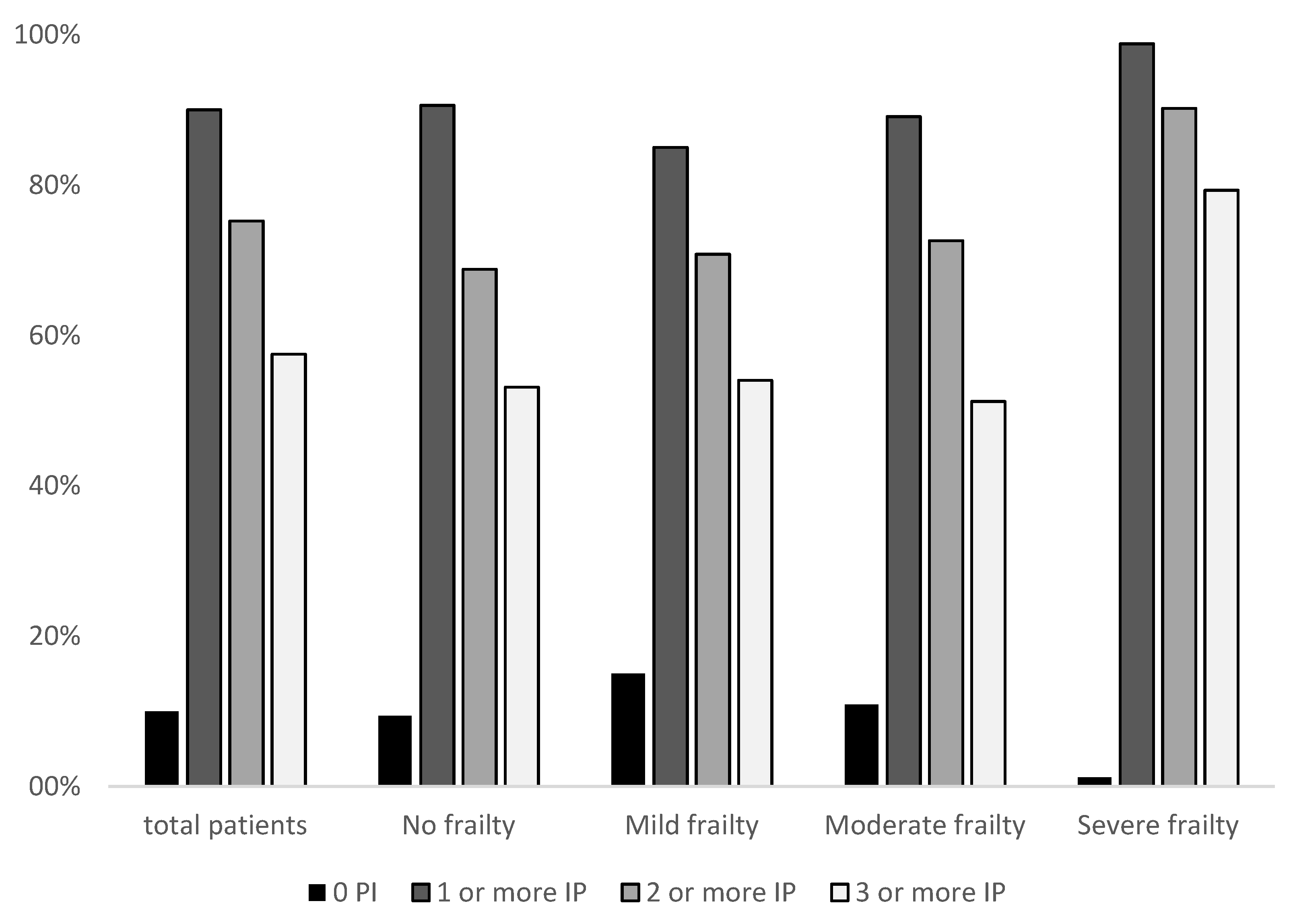
| FI (degrees) | No frailty (0–0.19) | 32 (7.5%) |
| Mild frailty (0.20–0.35) | 113 (26.4%) | |
| Moderate frailty (0.36–0.50) | 201 (47.0%) | |
| Severe frailty (0.51–1) | 82 (19.1%) | |
| End-of-life patients | 155 (36.2%) | |
| Therapeutic aim | Survival | 41 (9.6%) |
| Functionality | 223 (52.1%) | |
| Symptomatic | 164 (38.3%) | |
| Baseline Pharmacological Data | Total N = 428 | |
|---|---|---|
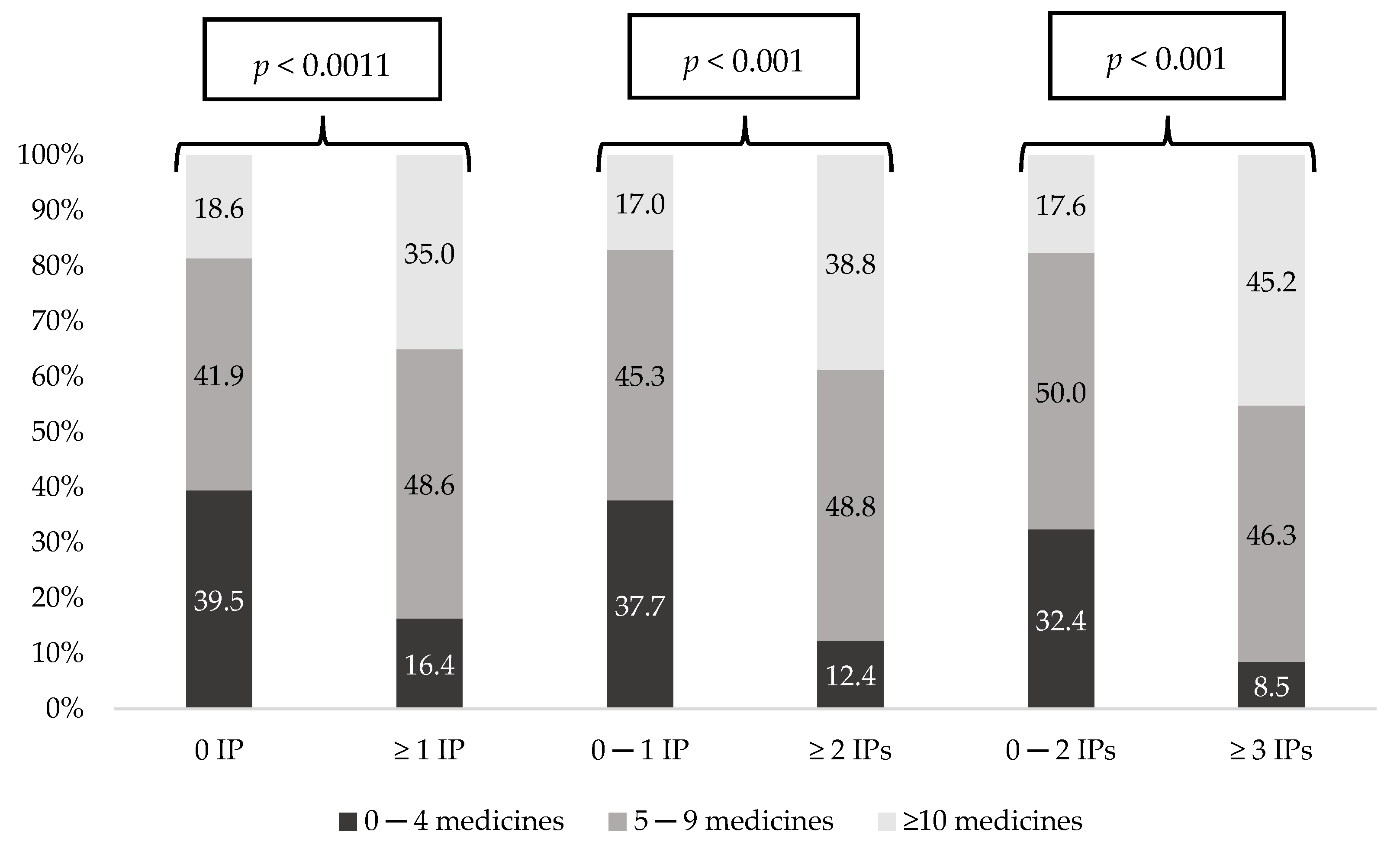
| Polypharmacy, mean (SD) | 8.13 (3.88) | |
| Polypharmacy (degree) | 0–4 medications | 80 (18.7%) |
| 5–9 medications | 205 (47.9%) | |
| 10 or more medications | 143 (33.4%) | |
| Medication Regimen Complexity Index (MRCI), mean (SD) | 30.74 (16.26) | |
| MRCI (degree) | Low complexity (0–19.99) | 109 (25.5%) |
| Moderate complexity (20–39.99) | 208 (48.6%) | |
| High complexity (40 or more) | 111 (25.9%) | |
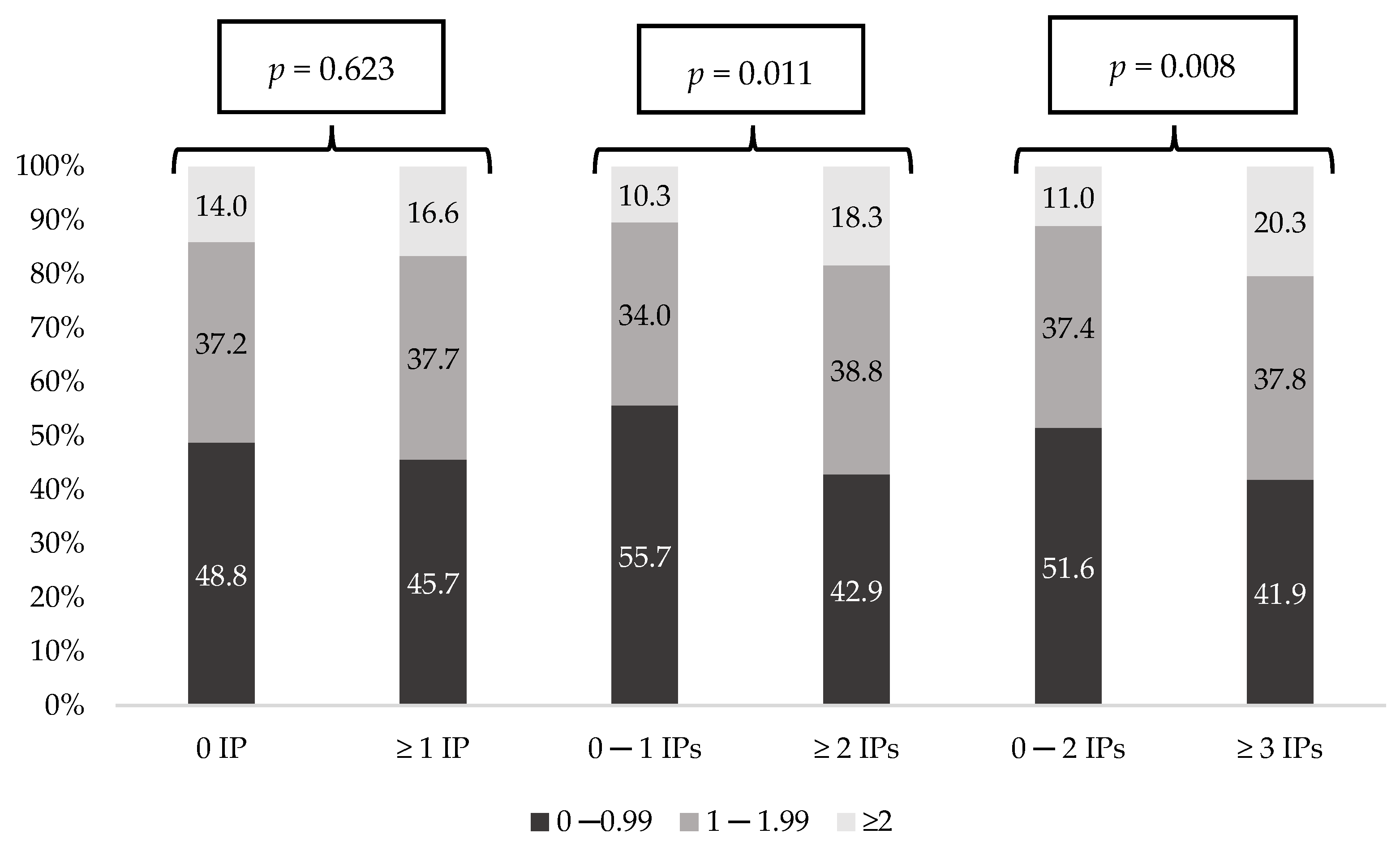
| Drug Burden Index (DBI), mean (SD) | 1.17 (0.84) | |
| DBI (degree) | Low DBI (0–0.99) | 70 (16.4%) |
| Moderate DBI (1–1.99) | 197 (46.0%) | |
| High DBI (2 or more) | 161 (37.6%) | |
| Inappropriate prescriptions (IP), mean (SD) | 3.14 (2.27) | |
| Number of IP | 0 IP | 43 (10.0%) |
| 1 or more IP | 385 (90.0%) | |
| 2 or more IP | 322 (75.2%) | |
| 3 or more IP | 246 (57.5%) | |
| Inappropriate Prescriptions | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| N = 428 | 0 | ≥1 | p | 0–1 | ≥2 | p | 0–2 | ≥3 | p | |
| Baseline Demographic Data | ||||||||||
| Age, mean (SD) | 85.5 (7.2) | 85.5 (7.7) | 0.990 | 85.20 (8.5) | 85.63 (7.4) | 0.618 | 86.0 (7.8) | 85.2 (7.6) | 0.273 | |
| Gender | Men | 11 (7.7%) | 132 (92.3%) | 0.251 | 29 (20.3%) | 114 (79.7%) | 0.128 | 57 (39.9%) | 86 (60.1%) | 0.430 |
| Women | 32 (11.2%) | 253 (88.8%) | 77 (27.0%) | 208 (73.0%) | 125 (43.9%) | 160 (56.1%) | ||||
| Origin | Home | 25 (11.9%) | 185 (88.1%) | 0.209 | 49 (23.3%) | 161 (76.7%) | 0.500 | 81 (38.6%) | 129 (61.4%) | 0.105 |
| NH | 18 (8.3%) | 200 (91.7%) | 57 (26.1%) | 161 (73.9%) | 101 (46.3%) | 117 (53.7%) | ||||
| Baseline Clinical, Functional and Cognitive Data | ||||||||||
| Medication management | 44 (9.3%) | 54 (14.0%) | 0.391 | 14 (13.2%) | 44 (13.7%) | 0.905 | 26 (14.3%) | 32 (13.0%) | 0.703 | |
| BI, mean (SD) | 60.7 (31.1) | 48.7 (32.0) | 0.020 * | 53.21 (32.4) | 48.85 (32.0) | 0.226 | 50.5 (31.9) | 49.5 (32.3) | 0.755 | |
| BI (degree) | IB ≥ 95 | 8 (15.7%) | 43 (84.3%) | 0.013 * | 15 (29.4%) | 36 (70.6%) | 0.192 | 22 (43.1%) | 29 (56.9%) | 0.695 |
| IB 90–65 | 16 (13.3%) | 104 (86.7%) | 32 (26.7%) | 88 (73.3%) | 52 (43.3%) | 68 (56.7%) | ||||
| IB 60–25 | 12 (9.3%) | 117 (90.7%) | 32 (24.8%) | 97 (75.2%) | 52 (40.6%) | 76 (59.4%) | ||||
| IB ≤ 20 | 7 (5.5%) | 121 (94.5%) | 27 (21.1%) | 101 (78.9%) | 7 (5.5%) | 121 (94.5%) | ||||
| Cognitive Status (dementia) | No dementia | 10 (8.9%) | 102 (91.1%) | 0.546 | 28 (25.0%) | 84 (75.0%) | 0.730 | 45 (40.2%) | 67 (59.8%) | 0.608 |
| Mild | 2 (3.2%) | 60 (96.8%) | 11 (17.7%) | 51 (82.3%) | 26 (41.9%) | 36 (58.1%) | ||||
| Moderate † | 19 (17.0%) | 93 (83.0%) | 32 (28.6%) | 80 (71.4%) | 50 (44.6%) | 62 (55.4%) | ||||
| Advanced ‡ | 12 (8.5%) | 130 (91.5%) | 35 (24.6%) | 107 (75.4%) | 61 (43.0%) | 81 (57.0%) | ||||
| GS, mean (SD) | 2.7 (1.5) | 2.9 (1.5) | 0.400 | 2,6 (1,4) | 3.0 (1.5) | 0.020 * | 2.7 (1.4) | 3.1 (1.5) | 0.004 * | |
| GS Type | Fall | 10 (23.3%) | 134 (34.8%) | 0.128 | 27 (25.5%) | 117 (36.3%) | 0.040 * | 54 (29.7%) | 90 (36.6%) | 0.134 |
| Dysphagia | 5 (11.6%) | 79 (20.5%) | 0.164 | 16 (15.1%) | 68 (21.1%) | 0.176 | 29 (15.9%) | 55 (22.4%) | 0.098 * | |
| Pain | 13 (30.2%) | 86 (22.3%) | 0.244 | 24 (22.6%) | 75 (23.3%) | 0.890 | 36 (19.8%) | 63 (25.6%) | 0.157 | |
| Depressive Syndrome | 16 (37.2%) | 182 (47.3%) | 0.209 | 41 (38.7%) | 157 (48.8%) | 0.071 | 68 (37.4%) | 130 (52.8%) | 0.001 * | |
| Insomnia | 24 (55.8%) | 205 (53.2%) | 0.749 | 51 (48.1%) | 178 (55.3%) | 0.199 | 89 (48.9%) | 140 (56.9%) | 0.101 | |
| Morbidities, mean (SD) | 4.1 (2.1) | 5.0 (2.1) | 0.014 * | 4.2 (1.9) | 5.1 (2.2) | <0.001 * | 4.4 (2.0) | 5.3 (2.2) | <0.001 * | |
| Morbidities (number) | 1–2 | 8 (18.6%) | 35 (81.4%) | 0.011 * | 19 (44.2%) | 24 (55.8%) | 0.001 * | 26 (60.5%) | 17 (39.5%) | <0.001 * |
| 3–4 | 20 (11.9%) | 148 (88.1%) | 47 (28.0%) | 121 (72.0%) | 83 (49.4%) | 85 (50.6%) | ||||
| 5 or more | 15 (6.9%) | 202 (93.1%) | 40 (18.4%) | 177 (81.6%) | 73 (33.6%) | 144 (66.4%) | ||||
| Morbidities (type) | Hypertension | 22 (51.1%) | 268 (69.6%) | 0.003 * | 61 (57.5%) | 229 (77.1%) | 0.002 * | 107 (58.8%) | 183 (74.3%) | <0.001 * |
| Chronic renal failure | 15 (34.9%) | 171 (44.4%) | 0.232 | 40 (37.7%) | 146 (45.3%) | 0.171 | 71 (39.0%) | 115 (46.7%) | 0.110 | |
| Type 2 Diabetes | 2 (4.7%) | 84 (21.8%) | 0.008 * | 15 (14.2%) | 95 (29.5%) | 0.009 * | 33 (18.1%) | 77 (31.3%) | 0.010 * | |
| Heart failure | 6 (14.0%) | 82 (21.3%) | 0.258 | 17 (16.0%) | 71 (22.0%) | 0.184 | 34 (18.7%) | 54 (22.0%) | 0.408 | |
| FI, mean (SD) | 0.34 (0.1) | 0.39 (0.1) | 0.023 * | 0.36 (0.12) | 0.40 (0.13) | 0.013 * | 0.36 (0.1) | 0.40 (0.1) | 0.007 * | |
| FI (degree) | No FI | 3 (9.4%) | 29 (90.6%) | 0.017 * | 10 (31.3%) | 22 (68.8%) | 0.004 * | 15 (46.9%) | 17 (53.1%) | 0.004 * |
| Mild FI | 17 (15.0%) | 96 (85.0%) | 33 (29.2%) | 80 (70.8%) | 52 (46.0%) | 61 (54.0%) | ||||
| Moderate FI | 22 (10.9%) | 179 (89.1%) | 55 (27.4%) | 146 (72.6%) | 98 (48.8%) | 103 (51.2%) | ||||
| Severe FI | 1 (1.2%) | 81 (98.8%) | 8 (9.8%) | 74 (90.2%) | 17 (20.7%) | 65 (79.3%) | ||||
| End-of-life patients | 11 (25.6%) | 144 (37.4%) | 0.126 | 34 (32.1%) | 121 (37.6%) | 0.307 | 64 (35.2%) | 91 (37.0%) | 0.697 | |
| Therapeutic Aim | Survival | 3 (7.3%) | 38 (92.7%) | 0.198 | 11 (26.8%) | 30 (73.2%) | 0.702 | 18 (43.9%) | 23 (56.1%) | 0.754 |
| Functionality | 28 (12.6%) | 195 (87.4%) | 58 (26.0%) | 165 (74.0%) | 98 (43.9%) | 125 (56.1%) | ||||
| Symptomatic | 12 (7.3%) | 152 (92.7%) | 37 (22.6%) | 127 (77.4%) | 66 (40.2%) | 98 (59.8%) | ||||
| ATC Group | Total |
|---|---|
| A–Alimentary tract and metabolism | 330 (24.7%) |
| B–Blood and blood-forming organs | 124 (9.3%) |
| C–Cardiovascular system | 412 (30.8%) |
| D–Dermatological | 0 |
| G–Genitourinary system and hormones | 26 (1.9%) |
| H–Systemic hormonal preparations (excluding sex hormones and insulin) | 7 (0.5%) |
| J–Anti-infective for systemic use | 1 (0.1%) |
| L–Antineoplastic and immunomodulation agents | 2 (0.2%) |
| M–Musculoskeletal system | 28 (2.3%) |
| N–Nervous system | 329 (24.6%) |
| R–Respiratory system | 69 (5.1%) |
| S–Sensory organs | 6 (0.4%) |
| V–Various | 0 |
| Patient Characteristics | Inappropriate Prescriptions | ||||||
|---|---|---|---|---|---|---|---|
| 0 vs. ≥1 | 0–1 vs. ≥2 | 0–2 vs. ≥3 | |||||
| OR | Univariate | Multivariate | Univariate | Multivariate | Univariate | Multivariate | |
| Barthel Index (BI), mean (SD) | 0.99 (0.98–0.99) | ||||||
| BI (degree) | Indep.: ≥95 | 1 | - | - | - | - | - |
| Mild: 90–65 | 1.20 (0.48–3.03) | ||||||
| Mod.: 60–25 | 1.81 (0.69–4.74) | ||||||
| Severe: ≤20 | 3.2 (1.10–9.40) | ||||||
| Geriatric Syndrome (GS), mean (SD) | 1.19 (1.03–1.39) | 1.21 (1.06–1.37) | |||||
| GS (degree) | 0 | - | - | 1 | - | 1 | - |
| 1–2 | 1.69 (0.6–4.6) | 0.99 (0.37–2.62) | |||||
| ≥3 | 2.24 (0.8–6.0) | 1.69 (0.65–4.41) | |||||
| Fall | Not | - | - | 1 | - | - | - |
| Yes | 1.67 (1.0–2.7) | ||||||
| Depressive Syndrome | Not | - | - | - | 1 | - | |
| Yes | 1.88 (1.27–2.78) | ||||||
| T2DM | Not | 1 | - | 1 | - | 1 | - |
| Yes | 5.7 (1.3–24.1) | 2.3 (1.2–4.5) | 1.9 (1.2–3.2) | ||||
| Morbidities, mean (SD) | 1.24 (1.04–1.48) | 1.25 (1.11–1.41) | 1.24 (1.12–1.37) | ||||
| Morbidities (number) | 1–2 | 1 | - | 1 | - | 1 | - |
| 3–4 | 1.69 (0.68–4.15) | 2.04 (1.0–4.0) | 1.57 (0.79–3.10) | ||||
| ≥5 | 3.08 (1.21–7.80) | 3.5 (1.8–7.0) | 3.02 (1.54–5.91) | ||||
| Frailty Index (FI), mean (SD) | 15.09 (1.43–159.6) | 8.27 (1.5–44.5) | 7.63 (1.70–34.23) | ||||
| FI (degree) | None: 0–0.19 | 1 | - | 1 | 1 | 1 | 1 |
| Mild: 0.20–0.35 | 0.58 (0.16–2.13) | 1.10 (0.5–2.5) | 0.86 (0.34–2.12) | 1.04 (0.47–2.27) | 0.80 (0.54–1.89) | ||
| Mod.: 0.36–0.50 | 0.84 (0.53–2.99) | 1,21 (0.7–2.7) | 0.97 (0.41–2.3) | 0.93 (0.44–1.96) | 0.82 (0.62–1.84) | ||
| Severe: 0.51–1 | 8.38 (0.93–83.79) | 4.21 (1.5–11.9) | 2.62 (0.87–7.86) | 3.37 (1.41–8.10) | 2.13 (0.96–5.50) | ||
| Polypharmacy, mean (SD) | 1.21 (1.09–1.34) | - | 1.26 (1.17–1.36) | - | 1.26 (1.19–1.35) | - | |
| Medications (number) | 0–4 | 1 | 1 | 1 | 1 | 1 | 1 |
| 5–9 | 2.80 (1.36–5.77) | 2.80 (1.36–5.77) | 3.3 (1.9–5.6) | 2.9 (1.7–5.1) | 3.52 (1.99–6.22) | 3.17 (1.77–5.68) | |
| ≥10 | 4.55 (1.87–11.11) | 4.55 (1.87–11.11) | 6.9 (3.6–13.4) | 6.07 (3.1–11.8) | 9.75 (5.17–18.38) | 8.65 (4.53–16.51) | |
| MRCI, mean (SD) | 1.03 (1.01–1.06) | - | 1.05 (1.3–1.07) | - | 1.05 (1.04–1.07) | - | |
| MRCI (degree) | Low: 0–19,99 | 1 | - | 1 | - | 1 | - |
| Mod.: 20–39.99 | 3.6 (1.7–7.3) | 2.76 (1.68–4.54) | 2.80 (1.73–4.53) | ||||
| High: ≥40 | 3.6 (1.4–8.8) | 6.87 (3.31–14.24)) | 7.11 (3.87–13.06) | ||||
| DBI, mean (SD) | - | - | 1.442 (1.08–1.91) | - | 1.41 (1.11–1.79) | - | |
| DBI (degree) | Low: 0–0.99 | - | - | 1 | - | 1 | - |
| Mod.: 1–1.99 | 1.49 (0.92–2.40) | 1.25 (0.82–1.90) | |||||
| High: ≥2 | 2.29 (1.13–4.68) | 2.28 (1.27–4.11) | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molist-Brunet, N.; Sevilla-Sánchez, D.; Puigoriol-Juvanteny, E.; Espaulella-Ferrer, M.; Amblàs-Novellas, J.; Espaulella-Panicot, J. Factors Associated with the Detection of Inappropriate Prescriptions in Older People: A Prospective Cohort. Int. J. Environ. Res. Public Health 2021, 18, 11310. https://doi.org/10.3390/ijerph182111310
Molist-Brunet N, Sevilla-Sánchez D, Puigoriol-Juvanteny E, Espaulella-Ferrer M, Amblàs-Novellas J, Espaulella-Panicot J. Factors Associated with the Detection of Inappropriate Prescriptions in Older People: A Prospective Cohort. International Journal of Environmental Research and Public Health. 2021; 18(21):11310. https://doi.org/10.3390/ijerph182111310
Chicago/Turabian StyleMolist-Brunet, Núria, Daniel Sevilla-Sánchez, Emma Puigoriol-Juvanteny, Mariona Espaulella-Ferrer, Jordi Amblàs-Novellas, and Joan Espaulella-Panicot. 2021. "Factors Associated with the Detection of Inappropriate Prescriptions in Older People: A Prospective Cohort" International Journal of Environmental Research and Public Health 18, no. 21: 11310. https://doi.org/10.3390/ijerph182111310
APA StyleMolist-Brunet, N., Sevilla-Sánchez, D., Puigoriol-Juvanteny, E., Espaulella-Ferrer, M., Amblàs-Novellas, J., & Espaulella-Panicot, J. (2021). Factors Associated with the Detection of Inappropriate Prescriptions in Older People: A Prospective Cohort. International Journal of Environmental Research and Public Health, 18(21), 11310. https://doi.org/10.3390/ijerph182111310






