Susceptibility to COVID-19 after High Exposure to Perfluoroalkyl Substances from Contaminated Drinking Water: An Ecological Study from Ronneby, Sweden
Abstract
:1. Introduction
2. Materials and Methods
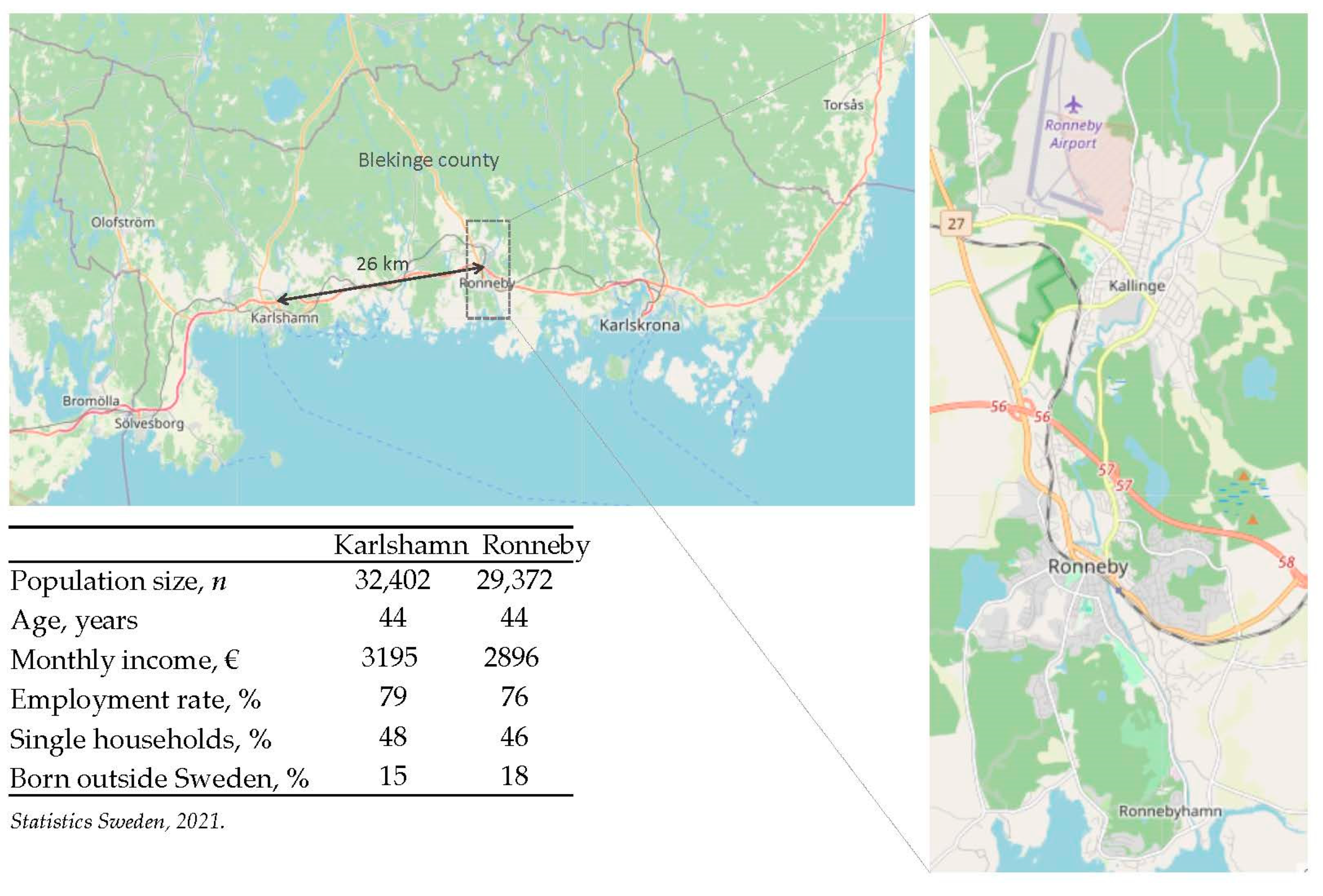
2.1. Setting
2.2. Variables and Data Sources
2.3. Patient and Public Involvement
2.4. Statistical Analysis
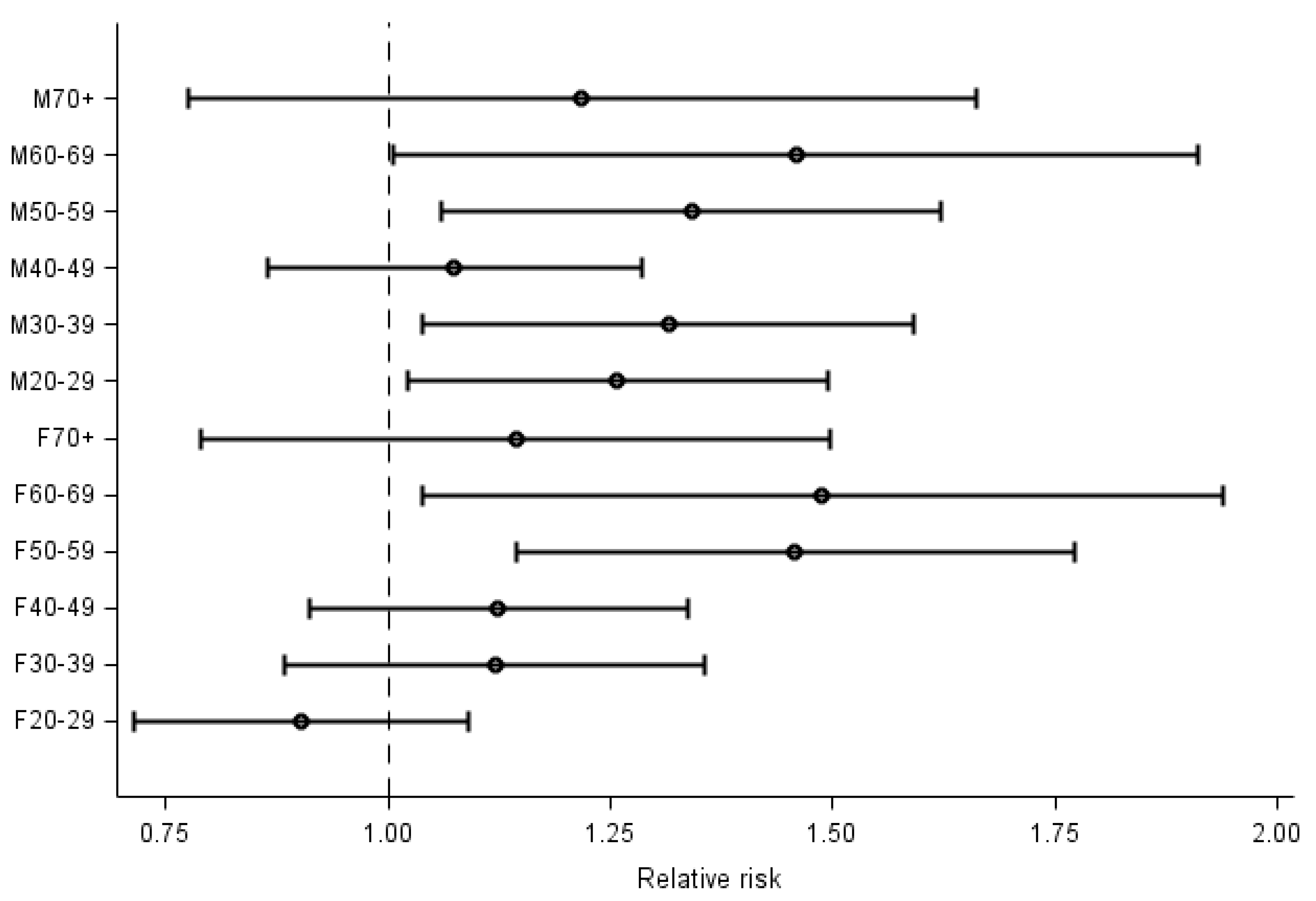
3. Results
4. Discussion
Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Quinete, N.; Hauser-Davis, R.A. Drinking water pollutants may affect the immune system: Concerns regarding COVID-19 health effects. Environ. Sci. Pollut. Res. 2021, 28, 1235–1246. [Google Scholar] [CrossRef] [PubMed]
- DeWitt, J.C.; Blossom, S.J.; Schaider, L.A. Exposure to per-fluoroalkyl and polyfluoroalkyl substances leads to immunotoxicity: Epidemiological and toxicological evidence. J. Expo. Sci. Environ. Epidemiol. 2019, 29, 148–156. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA) Panel on Contaminants in the Food Chain. Risk to human health related to the presence of perfluoroalkyl substances in food. EFSA J. 2020, 18, 6223. [Google Scholar]
- National Toxicology Program (NTP). Monograph on Immunotoxicity Associated with Exposure to Perfluorooctanoic Acid (PFOA) and Perfluorooctane Sulfonate (PFOS); National Toxicology Program: Durham, NC, USA, 2016. [Google Scholar]
- Agency for Toxic Substances and Disease Registry (ATSDR). Statement on Potential Intersection between PFAS Exposure and COVID-19. Available online: https://www.atsdr.cdc.gov/pfas/health-effects/index.html (accessed on 27 April 2021).
- Grandjean, P.; Timmermann, C.A.G.; Kruse, M.; Nielsen, F.; Vinholt, P.J.; Boding, L.; Heilmann, C.; Mølbak, K. Severity of COVID-19 at elevated exposure to perfluorinated alkylates. PLoS ONE 2021, 15, e0244815. [Google Scholar]
- Catelan, D.; Biggeri, A.; Russo, F.; Gregori, D.; Pitter, G.; Da Re, F.; Fletcher, T.; Canova, C. Exposure to perfluoroalkyl substances and mortality for COVID-19: A spatial ecological analysis in the Veneto region (Italy). Int. J. Environ. Res. Public Health 2021, 18, 2734. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.; Song, L.; Wang, J.; Yang, Z.; Yan, H.; Li, T.; Yu, L.; Jian, L.; Jiang, F.; Zheng, J.; et al. Association between urinary per- and poly-fluoroalkyl substances and COVID-19 susceptibility. Environ. Int. 2021, 153, 106524. [Google Scholar] [CrossRef] [PubMed]
- Cordner, A.; De La Rosa, V.Y.; Schaider, L.A.; Rudel, R.A.; Richter, L.; Brown, P. Guideline levels for PFOA and PFOS in drinking water: The role of scientific uncertainty, risk assessment decisions, and social factors. J. Expo. Sci. Environ. Epidemiol. 2019, 29, 157–171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Swedish Food Agency. Intagsberäkningar Som Underlag för Framtagande av Hälsobaserad Åtgärdsgräns för Perfluorerade Alkylsyror (PFAA) i Dricksvatten. Available online: https://www.livsmedelsverket.se/globalassets/livsmedel-innehall/oonskade-amnen/pfaa/intagsberakningar-for-atgardsgrans-for-pfaa-i-dricksvatten (accessed on 27 April 2021).
- Xu, Y.; Nielsen, C.; Li, Y.; Hammarstrand, S.; Andersson, E.M.; Li, H.; Olsson, D.S.; Engström, K.; Pineda, D.; Lindh, C.H.; et al. Serum perfluoroalkyl substances in residents following long-term drinking water contamination from firefighting foam in Ronneby, Sweden. Environ. Int. 2021, 147, 106333. [Google Scholar] [CrossRef] [PubMed]
- Busse, P.J.; Mathur, S.K. Age-related changes in immune function: Effect on airway inflammation. J. Allergy Clin. Immunol. 2010, 126, 690–699. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pijls, B.G.; Jolani, S.; Atherley, A.; Derckx, R.T.; Dijkstra, J.I.R.; Franssen, G.H.L.; Hendriks, S.; Richters, A.; Venemans-Jellema, A.; Zalpuri, S.; et al. Demographic risk factors for COVID-19 infection, severity, ICU admission and death: A meta-analysis of 59 studies. BMJ Open 2021, 11, e044640. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Peng, F.; Xu, B.; Zhao, J.; Liu, H.; Peng, J.; Li, Q.; Jiang, C.; Zhou, Y.; Liu, S.; et al. Risk factors of critical & mortal COVID-19 cases: A systematic literature review and meta-analysis. J. Infect. 2020, 81, e16–e25. [Google Scholar] [PubMed]
- Li, Y.; Fletcher, T.; Mucs, D.; Scott, K.; Lindh, C.H.; Tallving, P.; Jakobsson, K. Half-lives of PFOS, PFHxS and PFOA after end of exposure to contaminated drinking water. Occup. Environ. Med. 2018, 75, 46–51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Public Health Agency of Sweden. Statistik om SARS-CoV-2 Virusvarianter av Särskild Betydelse. Available online: https://www.folkhalsomyndigheten.se/smittskydd-beredskap/utbrott/aktuella-utbrott/COVID-19/statistik-och-analyser/sars-cov-2-virusvarianter-av-sarskild-betydelse/ (accessed on 3 March 2021).
| Ronneby, All Districts | Contaminated District | Reference Town | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Sex | Age | Population (n) | Cases (n) | Cumulative Incidence | Population (n) | Cases (n) | Cumulative Incidence | Population (n) | Cases (n) | Cumulative Incidence |
| All | All | 13,141 | 898 | 68 | 3635 | 239 | 66 | 11,059 | 622 | 56 |
| Female | 20–29 | 853 | 88 | 103 | 221 | 22 | 100 | 813 | 93 | 114 |
| 30–39 | 922 | 86 | 93 | 277 | 23 | 83 | 756 | 63 | 83 | |
| 40–49 | 1000 | 107 | 107 | 286 | 30 | 105 | 693 | 66 | 95 | |
| 50–59 | 1045 | 83 | 79 | 305 | 25 | 82 | 899 | 49 | 55 | |
| 60–69 | 856 | 42 | 49 | 247 | 11 | 45 | 849 | 28 | 33 | |
| 70+ | 1800 | 40 | 22 | 447 | 9 | 20 | 1595 | 31 | 19 | |
| Male | 20–29 | 1041 | 109 | 105 | 240 | 31 | 129 | 1009 | 84 | 83 |
| 30–39 | 1127 | 87 | 77 | 300 | 19 | 63 | 869 | 51 | 59 | |
| 40–49 | 1079 | 100 | 93 | 285 | 27 | 95 | 707 | 61 | 86 | |
| 50–59 | 1149 | 87 | 76 | 357 | 20 | 56 | 903 | 51 | 56 | |
| 60–69 | 847 | 40 | 47 | 245 | 12 | 49 | 772 | 25 | 32 | |
| 70+ | 1422 | 29 | 20 | 425 | 10 | 24 | 1194 | 20 | 17 | |
| Expected (n) | Observed (n) | SIR (95% CI) | |
|---|---|---|---|
| Ronneby, all districts | 752 | 898 | 1.19 (1.12; 1.27) |
| Contaminated district | 206 | 239 | 1.16 (1.01; 1.30) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nielsen, C.; Jöud, A. Susceptibility to COVID-19 after High Exposure to Perfluoroalkyl Substances from Contaminated Drinking Water: An Ecological Study from Ronneby, Sweden. Int. J. Environ. Res. Public Health 2021, 18, 10702. https://doi.org/10.3390/ijerph182010702
Nielsen C, Jöud A. Susceptibility to COVID-19 after High Exposure to Perfluoroalkyl Substances from Contaminated Drinking Water: An Ecological Study from Ronneby, Sweden. International Journal of Environmental Research and Public Health. 2021; 18(20):10702. https://doi.org/10.3390/ijerph182010702
Chicago/Turabian StyleNielsen, Christel, and Anna Jöud. 2021. "Susceptibility to COVID-19 after High Exposure to Perfluoroalkyl Substances from Contaminated Drinking Water: An Ecological Study from Ronneby, Sweden" International Journal of Environmental Research and Public Health 18, no. 20: 10702. https://doi.org/10.3390/ijerph182010702
APA StyleNielsen, C., & Jöud, A. (2021). Susceptibility to COVID-19 after High Exposure to Perfluoroalkyl Substances from Contaminated Drinking Water: An Ecological Study from Ronneby, Sweden. International Journal of Environmental Research and Public Health, 18(20), 10702. https://doi.org/10.3390/ijerph182010702






