Can Optimum Solar Radiation Exposure or Supplemented Vitamin D Intake Reduce the Severity of COVID-19 Symptoms?
Abstract
1. Introduction
2. Methodology
3. The Symptomatic Background of COVID-19
4. Some Background to Vitamin D Studies
5. The Influence of Vitamin D in Reducing ARTI and ARDS
6. The Link between Vitamin D levels, Geographical Context, Ethnicity, and Age
6.1. Vitamin D and Geographical Context during the First Wave
6.2. Vitamin D and Ethnicity
6.3. Vitamin D and Old Age
7. Vitamin D and COVID-19
| Study Details/Objective(S) | Results/Conclusions | Reference(s) |
|---|---|---|
| Measured serum vitamin D levels of 42 COVID-19 patients with ARDS in a hospital in Italy. | It was found that the potential mortality of hypovitaminosis patients was 50% after 10 days of hospitalisation, whereas it was 5% for only those with vitamin D levels above 10 ng/mL | [21] |
| The study analysed the serum vitamin D levels of COVID-19 patients in a single hospital (small cohort). | The serum 25(OH)D levels of 20 confirmed COVID-19 patients found that 65% required ICU admissions. Among the ICU patients, 84.6% were vitamin D deficient than floor patients (57.1% only). Apart from this, 100% of the ICU patients less than 75 years of age were vitamin D deficient. Even though it was a small observational study, it highlighted a link between vitamin D deficiency and COVID-19 risk. | [22] |
| Probed the efficacy of vitamin D (calcifediol) in treating COVID-19 patients—specifically the link between ICU admission and mortality. | Seventy-five COVID-19 positive patients were randomly selected and divided into two groups: both received standard care, and one group received vitamin D (calcifediol) oral supplement as additional care. From the vitamin D treated group, only 2% went to ICU, whereas it was 50% from the untreated group. Treatment with vitamin D (calcifediol) significantly reduced the ICU admission and mortality. | [28] |
| A retrospective analysis in Switzerland investigated the association between vitamin D and COVID-19. | In this study, significantly lower serum 25(OH)D level obtained in COVID-19 positive (27 patients) (11.1 ng/mL) cohort compared to the negative patients (80 people) (22.2 ng/mL), which was comparable to that of the control group (n = 1377). | [95] |
| Influence of vitamin D/ B12 and Mg combination was investigated in older (above 50 years) COVID-19 patients. | The result has shown that the combination therapy reduced the need for oxygen therapy and/or ICU support. | [97] |
| Investigated the link between vitamin D and COVID-19 severity, including mortality. | The study revealed a link between severe vitamin D deficiency and COVID-19 severity and mortality. | [99] |
| Researchers compared the COVID-19 test results of 14,000 people with their previous vitamin D levels. | The mean vitamin D level was significantly lower in the cohort found positive for COVID-19, compared to those who tested negative. Low plasma 25(OH)D concentration was found to be an independent risk factor for COVID-19. | [100] |
| This study assessed the significance of vitamin D in older COVID-19 patients. | The study found that the COVID-19 positive group had a lower median serum 25(OH)D level (10.8 ng/mL) compared to the negative group (20.8 ng/mL). | [101] |
| In the absence of vaccines and proper treatment, probed the potentiality of chemoprophylaxis. | An in vitro study has shown that calcitriol (vitamin D) among various potential libraries was found to be effective against SARS-CoV-2 with 0.69 log10 inhibition in human nasal epithelial cells (in vitro). If the result is replicated in clinical trials, host-directed therapy receives consideration, and calcitriol can be used as ring prophylaxis of the contacts of COVID-19 patients. | [102] |
| Researchers have given vitamin D oral supplementation to four COVID-19 confirmed patients—cholecalciferol 1000 or ergocalciferol 50,000 IU/day/5 days. | Patients who received high dose supplementation achieved normal vitamin D levels, which was seen in their clinical recovery level (lower oxygen requirements, reduction in inflammatory marker status, and shorter length of stay). | [103] |
| Determined the efficacy of bolus vitamin D3 supplementation during or just before COVID-19 in elderly adults. | They observed that 82.5% of the intervention group survived COVID-19, whereas only 44.4% survived in the comparator group. | [104] |
| The vitamin D serum levels of two cohorts of COVID-19 patients (active and expired) were statistically analysed. | Univariate analysis results showed that vitamin D deficiency is associated with the odds of death. While controlling the age, sex, and comorbidity, vitamin D deficiency has shown a strong association with COVID-19 mortality. | [105] |
8. The Role of Vitamin D in Infection Control
9. Acquiring Sufficient Levels of Vitamin D
- Exposing the body sensibly to natural solar radiation;
- Consuming a vitamin D-rich diet consisting of: egg yolk, cod liver oil, vitamin D fortified dairy products/juice/or other foods, wild mushroom, oily fish such as tuna, mackerel, herring, sardine, and wild salmon;
- Regularly taking vitamin D supplements in tablet form (recommended under medical supervision to those who have any health risk).
10. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, T.; Wu, Q.; Zhang, Z. Probable pangolin origin of SARS-CoV-2 is associated with the COVID-19, outbreak. Curr. Biol. 2020, 30, 1346–1351. [Google Scholar] [CrossRef]
- Wu, Y.; Ho, W.; Huang, Y.; Jin, D.-Y.; Li, S.; Liu, S.-L.; Liu, X.; Qiu, J.; Sang, Y.; Wang, Q.; et al. SARS- CoV-2 is an appropriate name for the new coronavirus. Lancet 2020, 395, 949–950. [Google Scholar] [CrossRef]
- Johns Hopkins, Covid-19 Dashboard, Johns Hopkins University and Medicine. Available online: https://coronavirus.jhu.edu/map.html (accessed on 4 January 2021).
- Shereen, M.A.; Khan, S.; Kazmi, A.; Bashir, N.; Siddique, R. COVID-19 infection: Origin, transmission, and characteristics of human coronaviruses. J. Adv. Res. 2020, 24, 91–98. [Google Scholar] [CrossRef]
- Cascella, M.; Rajnik, M.; Cuomo, A.; Dulebohn, S.C.; Napoli, R.D. Features, Evaluation, and Treatment Coronavirus (COVID-19); Star Pearls Publishing: Treasure Island, FL, USA, 2020; Available online: https://www.ncbi.nlm.nih.gov/books/NBK554776/ (accessed on 15 October 2020).
- Zhou, Z.; Ren, L.; Zhang, L.; Zhong, J.; Xiao, Y.; Jia, Y.Z.; Guo, L.; Yang, J.; Wang, C.; Jiang, S.; et al. Heightened innate immune responses in the respiratory tract of COVID-19 patients. Cell Host Microbe 2020, 27, 883–890.e2. [Google Scholar] [CrossRef]
- Laird, E.; Rhodes, J.; Kenny, R.A. Vitamin D and inflammation: Potential implications for the severity of COVID-19. Ir. Med. J. 2020, 113, 81. [Google Scholar] [PubMed]
- Ebadi, M.; Bhanji, R.A.; Mazurak, V.C.; Lytvyak, E.; Mason, A.; Czaja, A.J.; Montano-Loza, A.J. Severe vitamin D deficiency is a prognostic biomarker in autoimmune hepatitis. Aliment. Pharmacol. Ther. 2019, 49, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Theodoratou, E.; Tzoulaki, I.; Zgaga, L.; Ioannidis, J.P. Vitamin D and multiple health outcomes: Umbrella review of systematic reviews and meta-analyses of observational studies and randomised trials. BMJ 2014, 348, g2035. [Google Scholar] [CrossRef] [PubMed]
- Dancer, R.C.; Parekh, D.; Lax, S.; D’Souza, V.; Zheng, S.; Bassford, C.R.; Park, D.; Bartis, D.G.; Mahida, R.; Turner, A.M.; et al. Vitamin D deficiency contributes directly to the acute respiratory distress syndrome (ARDS). Thorax 2015, 70, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef] [PubMed]
- Teymoori-Rad, M.; Shokri, F.; Salimi, V.; Marashi, S.M. The interplay between vitamin D and viral infections. Rev. Med. Virol. 2019, 29, e2032. [Google Scholar] [CrossRef]
- Zhang, Y.; Tan, H.; Tang, J.; Li, J.; Chong, W.; Hai, Y.; Feng, Y.; Lunsford, L.D.; Xu, P.; Jia, D.; et al. Effects of vitamin D supplementation on prevention of type 2 diabetes in patients with prediabetes: A systematic review and meta-analysis. Diabetes Care 2020, 43, 1650–1658. [Google Scholar] [CrossRef]
- Peterlik, M.; Cross, H.S. Vitamin D and calcium deficits predispose for multiple chronic diseases. Eur. J. Clin. Investig. 2005, 35, 290–304. [Google Scholar] [CrossRef] [PubMed]
- Dhaliwal, R.; Mikhail, M.; Feuerman, M.; Aloia, J. Vitamin D dose-response in obesity. Endocr. Pract. 2014, 20, 1258–1264. [Google Scholar] [CrossRef] [PubMed]
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet 2020, 395, 1033. [Google Scholar] [CrossRef]
- Kong, J.; Zhu, X.; Shi, Y.; Liu, T.; Chen, Y.; Bhan, I.; Zhao, Q.; Thadhani, R.; Li, Y.C. VDR attenuates acute lung injury by blocking the Ang 2-Tie-2 pathway and the renin-angiotensin system. Mol. Endocrinol. 2013, 27, 2116–2125. [Google Scholar] [CrossRef]
- Zhang, Y.; Leung, D.Y.; Richers, B.N.; Liu, Y.; Remigio, L.K.; Riches, D.W.; Goleva, E. Vitamin D inhibits monocyte/macrophage pro-inflammatory cytokine production by targeting MAPK phosphatase-1. J. Immunol. 2012, 1, 2127–2135. [Google Scholar] [CrossRef]
- Xu, J.; Yang, J.; Chen, J.; Luo, Q.; Zhang, Q.; Zhang, H. Vitamin D alleviates lipopolysaccharide-induced acute lung injury via regulation of the renin-angiotensin system. Mol. Med. Rep. 2017, 16, 7432–7438. [Google Scholar] [CrossRef]
- Zhou, Y.F.; Luo, B.A.; Qin, L.L. The association between vitamin D deficiency and community-acquired pneumonia: A meta-analysis of observational studies. Medicine 2018, 98, e17252. [Google Scholar] [CrossRef]
- Carpagnano, G.E.; Di Lecce, V.; Quaranta, V.N.; Zito, A.; Buonamico, E.; Palumbo, A.; Di Gioia, G.; Valerio, V.N.; Resta, O. Vitamin D deficiency as a predictor of poor prognosis in patients with acute respiratory failure due to COVID-19. J. Endocrinol. Investig. 2020. [Google Scholar] [CrossRef]
- Lau, F.H.; Majumder, R.; Torabi, R.; Saeg, F.; Hoffman, F.; Cirillo, J.D.; Greiffenstein, P. Vitamin D insufficiency is prevalent in severe COVID-19. MedRxiv 2020. [Google Scholar] [CrossRef]
- Alzaman, N.S.; Dawson-Hughes, B.; Nelson, J.; D’Alessio, D.; Pittas, A.G. Vitamin D status of black and white Americans and changes in vitamin D metabolites after varied doses of vitamin D supplementation A. J. Clin. Nutr. 2016, 104, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Nair, R.; Maseeh, A. Vitamin D: The “sunshine” vitamin. J. Pharm. Pharmacother. 2012, 3, 118. [Google Scholar]
- ICNARC. Intensive Care National Audit and Research Centre. A Report on COVID-19 in Critical Care. Available online: https://www.icnarc.org/About/LatestNews/2020/04/04/Report-On-2249-Patients (accessed on 28 October 2020).
- ONS. Office for National Statistics 2011 Census. Available online: www.ons.gov.uk (accessed on 12 October 2020).
- Rhodes, J.M.; Subramanian, S.; Laird, E.; Griffin, G.; Kenny, R.A. Perspective: Vitamin D deficiency and COVID-19 severity–plausibly linked by latitude, ethnicity, impacts on cytokines, ACE2 and thrombosis. J. Intern. Med. 2021, 289, 97–115. [Google Scholar] [CrossRef]
- Entrenas Castillo, M.; Entrenas Costa, L.M.; Vaquero Barrios, J.M.; Alcala Diaz, J.F.; Miranda, J.L.; Bouillon, R.; Quesada Gomez, J.M. Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID 19: A pilot randomized clinical study. J. Steroid Biochem. Mol. Biol. 2020, 203, 1–6. [Google Scholar] [CrossRef]
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.; Ou, C.; He, J.-X.; Liu, L.; Shan, H.; Lei, C.-l.; Hui, D.S.C.; et al. Clinical characteristics of coronavirus disease in 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Dargent, A.; Chatelain, E.; Kreitmann, L.; Quenot, J.P.; Cour, M.; Argaud, L.; The COVID-19 study group. Lung ultrasound score to monitor COVID-19 pneumonia progression in patients with ARDS. PLoS ONE 2020, 15, e0236312. [Google Scholar] [CrossRef]
- Thickett, D.R.; Moromizato, T.; Litonjua, A.A.; Amrein, K.; Quraishi, S.A.; Lee-Sarwar, K.A.; Mogensen, K.M.; Purtle, S.W.; Gibbons, F.K.; Camargo, C.A., Jr. Association between prehospital vitamin D status and incident acute respiratory failure in critically ill patients: A retrospective cohort study. BMJ Open Respir. Res. 2015, 2, e000074. [Google Scholar] [CrossRef]
- Zhou, J.; Du, J.; Huang, L.; Wang, Y.; Shi, Y.; Lin, H. Preventive effects of vitamin D on seasonal influenza A in infants: A multicenter, randomized, open, controlled clinical trial. Pediatric Infect. Dis. J. 2018, 37, 749–754. [Google Scholar] [CrossRef]
- Lu, D.; Zhang, J.; Ma, C.; Yue, Y.; Zou, Z.; Yu, C.; Yin, F. The link between community-acquired pneumonia and vitamin D levels in older patients. Gerontol. Geriatr. 2018, 51, 435–439. [Google Scholar] [CrossRef]
- Zhou, W.; Mao, S.; Wu, L.; Yu, J. Association Between Vitamin D Status and Sepsis. Clin. Lab. 2018, 64, 451–460. [Google Scholar] [CrossRef]
- Li, Y.; Ding, S. Serum 25-Hydroxyvitamin D and the risk of mortality in adult patients with Sepsis: A meta-analysis. BMC Infect. Dis. 2020, 20, 189. [Google Scholar] [CrossRef]
- Manion, M.; Hullsiek, K.H.; Wilson, E.; Rhame, F.; Kojic, E.; Gibson, D.; Hammer, J.; Patel, P.; Brooks, J.T.; Baker, J.V.; et al. Vitamin D deficiency is associated with IL-6 levels and monocyte activation in HIV- infected persons. PLoS ONE 2017, 12, e0175517. [Google Scholar] [CrossRef] [PubMed]
- Dalvi, S.M.; Ramraje, N.N.; Patil, V.W.; Hegde, R.; Yeram, N. Study of IL-6 and vitamin D3 in patients of pulmonary tuberculosis. Indian J. Tuberc. 2019, 66, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Gao, Y.; Tian, L.; Zheng, L.; Wang, X.; Liu, W.; Zhang, Y.; Huang, G. Association of serum 25-hydroxyvitamin D3 with adipokines and inflammatory markers in persons with prediabetes mellitus. Clin. Chim. Acta 2017, 468, 152–158. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D: Physiology, Molecular Biology, and Clinical Applications; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2010. [Google Scholar]
- Lips, P.; Hosking, D.; Lippuner, K.; Norquist, J.M.; Wehren, L.; Maalouf, G.; Ragi-eis, S.; Chandler, J. The prevalence of vitamin D inadequacy amongst women with osteoporosis: An international epidemiological investigation. J. Intern. Med. 2006, 260, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Bilezikian, J.P.; Bikle, D.; Hewison, M.; Lazaretti-Castro, M.; Formenti, A.M.; Gupta, A.; Madhavan, M.V.; Nair, N.; Babalyan, B.; Hutchings, N.; et al. Mechanisms IN endocrinology:vitamin D and COVID-19. Eur. J. Endocrinol. 2020, 183, R133–R147. [Google Scholar] [CrossRef]
- Bergman, P. The link between vitamin D and Covid-19: Distinguishing facts from fiction. J. Intern. Med. 2020, 289, 131–133. [Google Scholar] [CrossRef]
- Quesada-Gomez, J.M.; Castillo, M.E.; Bouillon, R. Vitamin D Receptor stimulation to reduce Acute Respiratory Distress Syndrome (ARDS) in patients with Coronavirus SARS-CoV-2 infections: Revised Ms SBMB 2020_166. J. Steroid Biochem. Mol. Biol. 2020, 105719, 1–8. [Google Scholar]
- Liu, P.T.; Stenger, S.; Li, H.; Wenzel, L.; Tan, B.H.; Krutzik, S.R.; Ochoa, M.T.; Schauber, J.; Wu, K.; Meinken, C.; et al. Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response. Science 2006, 311, 1770–1773. [Google Scholar] [CrossRef]
- Herr, C.; Shaykhiev, R.; Bals, R. The role of cathelicidin and defensins in pulmonary inflammatory diseases. Expert Opin. Biol. Ther. 2007, 7, 1449–1461. [Google Scholar] [CrossRef]
- Adams, J.S.; Ren, S.; Liu, P.T.; Chun, R.F.; Lagishetty, V.; Gombart, A.F.; Borregaard, N.; Modlin, R.L.; Hewison, M. Vitamin D-directed rheostatic regulation of monocyte antibacterial responses. J. Immunol. 2009, 182, 4289–4295. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Ran, Z.; Jiang, Q.; Hu, N.; Yu, B.; Zhu, L.; Shen, L.; Zhang, S.; Chen, L.; Chen, H.; et al. Vitamin D Alleviates Rotavirus Infection through a Microrna-155-5p Mediated; Regulation of the TBK1/IRF3 Signaling Pathway In Vivo and In Vitro. Int. J. Mol. Sci. 2019, 20, 3562. [Google Scholar] [CrossRef] [PubMed]
- Lang, P.O.; Aspinall, R. Can we translate vitamin D immunomodulating effect in innate and adaptive immunity to vaccine response? Nutrients 2015, 7, 2044–2060. [Google Scholar] [CrossRef] [PubMed]
- Aranow, C. Vitamin D and the immune system. J. Investig. Med. 2011, 59, 881–886. [Google Scholar] [CrossRef]
- Prietl, B.; Treiber, G.; Pieber, T.R.; Amrein, K. Vitamin D and immune function. Nutrients 2013, 5, 2502–2521. [Google Scholar] [CrossRef]
- Hewison, M. Vitamin D and Innate and Adaptive Immunity. In Vitamins & Hormones; Academic Press: New York, NY, USA, 2011; pp. 23–62. [Google Scholar]
- Holick, M.F. Vitamin D status: Measurement, Interpretation, and Clinical Applications. Ann. Epidemiol. 2009, 19, 73–78. [Google Scholar] [CrossRef]
- Mithal, A.; Wahl, D.A.; Bonjour, J.P.; Burckhardt, P.; Dawson-Hughes, B.; Eisman, J.A.; El-Hajj Fuleihan, G.; Josse, R.G.; Lips, P.; Morales-Torres, J.; et al. Global vitamin D status and determinants of hypovitaminosis D. Osteoporos. Int. 2009, 20, 1807–1820. [Google Scholar] [CrossRef]
- Wei, J.; Zhu, A.; Ji, J.S. A comparison Study of Vitamin D Deficiency among Older Adults in China and the United States. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Jeng, L.; Yamshchikov, A.V.; Judd, S.E.; Blumberg, H.M.; Martin, G.S.; Ziegler, T.R.; Tangpricha, V. Alterations in vitamin D status and anti-microbial peptide levels in patients in the intensive care unit with sepsis. J. Transl. Med. 2009, 7, 28. [Google Scholar] [CrossRef]
- Clemens, T.L.; Henderson, S.L.; Adams, J.S.; Holick, M.F. Increased skin pigment reduces the capacity of the skin to synthesise vitamin D3. Lancet 1982, 319, 74–76. [Google Scholar] [CrossRef]
- Martínez-Moreno, J.; Hernandez, J.C.; Urcuqui-Inchima, S. Effect of high doses of vitamin D supplementation on dengue virus replication, Toll-like receptor expression, and cytokine profiles on dendritic cells. Mol. Cell. Biochem. 2020, 464, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Bergman, P.; Lindh, A.U.; Björkhem-Bergman, L.; Lindh, J.D. Vitamin D and respiratory tract infections: A systematic review and meta-analysis of randomized controlled trials. PLoS ONE. 2013, 8, e65835. [Google Scholar] [CrossRef] [PubMed]
- Leow, L.; Simpson, T.; Cursons, R.; Karalus, N.; Hancox, R.J. Vitamin D, innate immunity, and outcomes in community-acquired pneumonia. Respirology 2011, 16, 611–616. [Google Scholar] [CrossRef] [PubMed]
- Dancer, R.C.A.; Parekh, D.; Scott, A.; Perkins, G.D.; Thickett, D.R. T2 Vitamin D supplementation reduces perioperative systemic and alveolar inflammation in patients undergoing oesophagectomy: Results of the Vindaloo Trial. Thorax 2015, 70, A1. [Google Scholar] [CrossRef]
- Parekh, D.; Thickett, R.D.; Turner, A.M. Vitamin D deficiency and acute lung injury. Inflamm. Allergy Drug Targets 2013, 12, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Parekh, D.; Dancer, R.C.; Lax, S.; Cooper, M.S.; Martineau, A.R.; Fraser, W.D.; Tucker, O.; Alderson, D.; Perkins, G.D.; Gao-Smith, F.; et al. Vitamin D to prevent acute lung injury following oesophagectomy (VINDALOO): Study protocol for a randomised placebo controlled trial. Trials 2013, 14, 100. [Google Scholar] [CrossRef] [PubMed]
- Hansdottir, S.; Monick, M.M. Vitamin D effects on lung immunity and respiratory diseases. Vitam. Horm. 2011, 86, 217–237. [Google Scholar] [PubMed]
- Sabetta, J.R.; DePetrillo, P.; Cipriani, R.J.; Smardin, J.; Burns, L.A.; Landry, M.L. Serum 25-hydroxyvitamin d and the incidence of acute viral respiratory tract infections in healthy adults. PLoS ONE 2010, 5, e11088. [Google Scholar] [CrossRef] [PubMed]
- Urashima, M.; Segawa, T.; Okazaki, M.; Kurihara, M.; Wada, Y.; Ida, H. Randomized trial of vitamin D supplementation to prevent seasonal influenza A in schoolchildren. Am. J. Clin. Nutr. 2010, 91, 1255–1260. [Google Scholar] [CrossRef] [PubMed]
- Arihiro, S.; Nakashima, A.; Matsuoka, M.; Suto, S.; Uchiyama, K.; Kato, T.; Mitobe, J.; Komoike, N.; Itagaki, M.; Miyakawa, Y. Randomized trial of vitamin D supplementation to prevent seasonal influenza and upper respiratory infection in patients with inflammatory bowel disease. Inflamm. Bowel Dis. 2019, 25, 1088–1095. [Google Scholar] [CrossRef] [PubMed]
- Ginde, A.A.; Mansbach, J.M.; Camargo, C.A. Association between serum 25-hydroxyvitamin D level and upper respiratory tract infection in the Third National Health and Nutrition Examination Survey. Arch. Intern. Med. 2009, 23, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Jolliffe, D.A.; Griffiths, C.J.; Martineau, A.R. Vitamin D in the prevention of acute respiratory infection: A systematic review of clinical studies. J. Steroid Biochem. Mol. Biol. 2013, 136, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Charan, J.; Goyal, J.P.; Saxena, D.; Yadav, P. Vitamin D for prevention of respiratory tract analysis: A systematic review and meta-analysis. J. Pharm. Pharmacother. 2012, 3, 300–303. [Google Scholar] [CrossRef] [PubMed]
- Han, J.E.; Jones, J.L.; Tangpricha, V.; Brown, M.A.; Hao, L.; Hebbar, G.; Lee, M.J.; Liu, S.; Brown, L.A.S.; Ziegler, T.R.; et al. High dose vitamin D administration in ventilated intensive care unit patients: A pilot double-blind randomized controlled trial. J. Clin. Transl. Endocrinol. 2016, 4, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Panarese, A.; Shahini, E. Covid-19, and vitamin D. Aliment. Pharm. Ther. 2020, 51, 993. [Google Scholar] [CrossRef] [PubMed]
- Bajwa, E.K.; Bhan, I.; Quraishi, S.; Matthay, M.; Thompson, B.T. Low vitamin D status occurs in 90% of patients with ARDS and is associated with a longer duration of mechanical ventilation; In A53. Respiratory Failure: Risk factors and outcomes in ARDS. Am. Thorac. Soc. 2016, 193, A1846. [Google Scholar]
- Zheng, S.; Yang, J.; Hu, X.; Li, M.; Wang, Q.; Dancer, R.C.; Parekh, D.; Gao-Smith, F.; Thickett, D.R.; Jin, S. Vitamin D attenuates lung injury via stimulating epithelial repair, reducing epithelial cell apoptosis and inhibits TGF-β induced epithelial to mesenchymal transition. Biochem. Pharmacol. 2020, 3, 113955. [Google Scholar] [CrossRef]
- Greiller, C.L.; Martineau, A.R. Modulation of the immune response to respiratory viruses by vitamin D. Nutrients 2015, 7, 4240–4270. [Google Scholar] [CrossRef]
- Liu, X.; Baylin, A.; Levy, P.D. Vitamin D deficiency and insufficiency among US adults: Prevalence, predictors, and clinical implications. Br. J. Nutr. 2018, 119, 928–936. [Google Scholar] [CrossRef]
- Parva, N.R.; Tadepalli, S.; Singh, P.; Qian, A.; Joshi, R.; Kandala, H.; Nookala, V.K.; Cheriyath, P. Prevalence of vitamin D deficiency and associated risk factors in the US population (2011–2012). Cureus 2018, 10, e2741. [Google Scholar] [CrossRef]
- Ilie, P.C.; Stefanescu, S.; Smith, L. The role of vitamin D in the prevention of coronavirus disease 2019 infection and mortality. Aging Clin. Exp. Res. 2020, 32, 1195–1198. [Google Scholar] [CrossRef] [PubMed]
- Isaia, G.; Giorgino, R.; Rini, G.B.; Bevilacqua, M.; Maugeri, D.; Adami, S. Prevalence of hypovitaminosis D in elderly women in Italy: Clinical consequences and risk factors. Osteoporos. Int. 2003, 14, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Brehm, J.M.; Celedón, J.C.; Soto-Quiros, M.E.; Avila, L.; Hanninghake, G.M.; Forno, E.; Laskey, D.; Sylvia, J.S.; Hollis, B.W.; Weiss, S.T.; et al. Serum vitamin D levels and markers of severity of childhood asthma in Costa Rica. Am. J. Respir. Criti. Care Med. 2009, 179, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Lips, P.T.A.M. Worldwide status of vitamin D nutrition. J. Steroid Biochem. Mol. Biol. 2010, 121, 297–300. [Google Scholar] [CrossRef] [PubMed]
- Shoenfeld, N.; Amital, H.; Shoenfeld, Y. The effect of melanism and vitamin D synthesis on the incidence o autoimmune disease. Nat. Clin. Pract. Rheumatol. 2009, 5, 99–105. [Google Scholar] [CrossRef]
- Kumar, J.; Muntner, P.; Kaskel, F.J.; Hailpern, S.M.; Melamed, M.L. Prevalence and associations of 25- hydroxyvitamin D deficiency in US children: NHANES 2001–2004. Pediatrics 2009, 124, e362–e370. [Google Scholar] [CrossRef]
- Liu, Y.; Brook, R.D.; Liu, X.; Byrd, J.B. Geographic Latitude, Cholesterol, and Blood Pressure. BioRxiv 2018, 308726. [Google Scholar] [CrossRef]
- Yetley, E.A. Assessing the vitamin D status of the US population. Am. J. Clin. Nutr. 2008, 88, 558S–564S. [Google Scholar] [CrossRef]
- Nowson, C.A.; Margerison, C. Vitamin D intake and vitamin D status of Australians. Med. J. Aust. 2002, 177, 149–152. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D and bone health. J. Nutr. 1996, 126, 1159S–1164S. [Google Scholar] [CrossRef]
- Lagunova, Z.; Porojnicu, A.C.; Lindberg, F.; Hexeberg, S.; Moan, J. The dependency of vitamin D status on body mass index, gender, age, and season. Anticancer Res. 2009, 29, 3713–3720. [Google Scholar] [CrossRef] [PubMed]
- Van der Wielen, R.P.; De Groot, L.C.P.G.M.; Van Staveren, W.A.; Löwik, M.R.H.; Van den Berg, H.; Haller, J.; Moreiras, O. Serum vitamin D concentrations among elderly people in Europe. Lancet 1995, 346, 207–210. [Google Scholar] [CrossRef]
- Chen, T.C.; Lu, Z.; Holick, M.F. Photobiology of vitamin D. In Vitamin D; Holick, M.F., Ed.; Humana Press: Totowa, NJ, USA, 2010; pp. 35–60. [Google Scholar]
- Atli, T.; Gullu, S.; Uysal, A.R.; Erdogan, G. The prevalence of vitamin D deficiency and the effects of ultraviolet light on vitamin D levels in the elderly Turkish population. Arch. Gerontol. Geriatr. 2005, 40, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Bischoff-Ferrari, H.A.; Borchers, M.; Gudat, F.; Dürmüller, U.; Stähelin, H.B.; Dick, W. Vitamin D receptor expression in human muscle tissue decreases with age. J. Bone Miner. Res. 2004, 19, 265–269. [Google Scholar] [CrossRef]
- Lee, D.M.; Tajar, A.; O’Neill, T.W.; OÇonnor, D.B.; Bartfai, G.; Boonen, S.; Bouillon, R.; Casanueva, F.F.; Finn, J.D.; Forti, G. Lower vitamin D levels are associated with depression among community-dwelling European men. J. Psychopharmacol. 2011, 25, 1320–1328. [Google Scholar] [CrossRef]
- Laird, E.; Kenney, R.A. Vitamin-D Deficiency in Ireland-Implications for COVID-19-Results from the Irish Longitudinal Study on Aging (TILDA). 2020. Available online: https://tilda.tcd.ie/publications/reports/pdf/Report_Covid19VitaminD.pdf (accessed on 20 December 2020).
- Greiller, C.L.; Suri, R.; Jolliffe, D.A.; Kebadze, T.; Hirsman, A.G.; Griffiths, C.J.; Johnston, S.L.; Martineau, A.R. Vitamin D attenuates rhinovirus-induced expression of intercellular adhesion molecules-I (ICAM-I) and platelet-activating factor receptor (PAFR) in respiratory epithelial cells. J. Steroid Biochem. Mol. Biol. 2019, 187, 1359. [Google Scholar] [CrossRef]
- D’Avolio, A.; Avataneo, V.; Manca, A.; Cusato, J.; Nicolo, A.D.; Lucchini, R.; Keller, F.; Cantu, M. 25-Hydroxyvitamin D concentrations are lower in patients with positive PCR for SARS-CoV-2. Nutrients 2020, 12, 1359. [Google Scholar] [CrossRef]
- Fasano, A.; Cereda, E.; Barichella, M.; Cassani, E.; Ferri, V.; Zecchinelli, A.L.; Pezzoli, G. COVID-19 in Parkinson’s disease patients living in Lombardy, Italy. Mov. Disord. 2020, 35, 1089–1093. [Google Scholar] [CrossRef]
- Tan, C.W.; Ho, L.P.; Kalimuddin, S.; Cherng, B.P.Z.; Teh, Y.E.; Thien, S.Y.; Wong, H.M.; Tern, P.J.W.; Chandran, M.; Chay, J.W.M.; et al. A cohort study to evaluate the effect of combination Vitamin D, Magnesium, and Vitamin B12 on progression to severe outcome in older COVID-19 patients. Nutrition 2020, 111017. [Google Scholar] [CrossRef]
- Meltzer, D.O.; Best, T.J.; Zhang, H. Association of vitamin D status and other clinical characteristics with COVID-19 test results. JAMA 2020, 3, e2019722. [Google Scholar] [CrossRef]
- Radujkovic, A.; Hippchen, T.; Tiwari-Heckler, S.; Dreher, S.; Boxberger, M.; Merle, U. Vitamin D Deficiency and Outcome of COVID-19 Patients. Nutrients 2020, 12, 2757. [Google Scholar] [CrossRef] [PubMed]
- Merzon, E.; Tworowski, D.; Gorohovski, A.; Vinker, S.; Golan Cohen, A.; Green, I.; Frenkel, M. Morgenstern; Low plasma 25 (OH) vitamin D level is associated with increased risk of COVID-19 infection: An Israeli population-based study. FEBS J. 2020, 287, 3693–3702. [Google Scholar] [CrossRef] [PubMed]
- Baktash, V.; Hosack, T.; Patel, N.; Shah, S.; Kandiah, P.; Abbeele, K.V.D.; Mandal, A.K.; Missouris, C.G. Vitamin D status and outcomes for hospitalized older patients with Covid-19. BMJ 2020, 138712. [Google Scholar] [CrossRef]
- Mok, C.K.; Ng, Y.L.; Ahidjo, B.A.; Lee, R.C.H.; Loe, M.W.C.; Liu, J.; Tan, K.S.; Kaur, P.; Chng, W.J.; Eu- Li Wong, J.; et al. Calcitriol, the active form of vitamin D, is a promising candidate for COVID-19 prophylaxis. BioRxiv 2020, 162396. [Google Scholar] [CrossRef]
- Ohaegbulam, K.C.; Swalih, M.; Patel, P.; Smith, M.A.; Perrin, R. Vitamin D Supplementation in COVID-19 Patients: A Clinical Case Series. Am. J. Ther. 2020, 27, e485–e490. [Google Scholar]
- Annweiler, C.; Hanotte, B.; de l’Eprevier, C.G.; Sebatier, J.-M.; Lafaie, L.; Celarier, T. Vitamin D and survival in Covid-19 patients: A quasi experimental study. J. Steroid Biochem. Mol. Biol. 2020, 204, 105771. [Google Scholar] [CrossRef]
- Raharusun, P. Patterns of COVID-19 Mortality and Vitamin D: An Indonesian Study. SSRN Electron. J. 2020. [Google Scholar] [CrossRef]
- Zdrenghea, M.T.; Makrinioti, H.; Bagacean, C.; Bush, A.; Johnston, S.L.; Stanciu, L.A. Vitamin D modulation of innate immune responses to respiratory viral infections. Rev. Med. Virol. 2017, 27, e1909. [Google Scholar] [CrossRef]
- Beard, J.A.; Bearden, A.; Striker, R. Vitamin D, and the antiviral state. J. Clin. Virol. 2011, 1, 194–200. [Google Scholar] [CrossRef]
- Di Rosa, M.; Malaguarnera, M.; Nicoletti, F.; Malaguarnera, L. Vitamin D3: A helpful immuno-modulator. Immunology 2011, 1, 123–139. [Google Scholar] [CrossRef]
- Wei, R.; Christakos, S. Mechanisms underlying the regulation of innate and adaptive immunity by vitamin D. Nutrients 2015, 7, 8251–8260. [Google Scholar] [CrossRef] [PubMed]
- Gombart, A.F.; Pierre, A.; Maggini, S. A Review of Micronutrients and the Immune System–Working in Harmony to Reduce the Risk of Infection. Nutrients 2020, 12, 236. [Google Scholar] [CrossRef] [PubMed]
- Dhawan, P.; Wei, R.; Sun, C.; Gombart, A.F.; Koeffler, H.P.; Diamond, G.; Christakos, S. C/EBPα, and the vitamin D receptor cooperate in the regulation of cathelicidin in lung epithelial cells. J. Cell. Physiol. 2015, 230, 464–472. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.B.; Lahore, H.; McDonnell, S.L.; Baggerly, C.A.; French, C.B.; Aliano, J.L.; Bhattoa, H.P. Evidence that vitamin D supplementation could reduce the risk of influenza and COVID-19 infections and deaths. Nutrients 2020, 12, 988. [Google Scholar] [CrossRef]
- Jeffery, L.E.; Burke, F.; Mura, M. 1, 25-dihydroxy vitamin D3 and IL-2 combine to inhibit T cell production of inflammatory cytokines and promote the development of regulatory T cells expressing CTLA-4 and FoxP3. J. Immunol. 2009, 183, 5458–5467. [Google Scholar] [CrossRef]
- Li, Y.C.; Qiao, G.; Uskokovic, M.; Xiang, W.; Zheng, W.; Kong, J. Vitamin D: A negative endocrine regulator of the renin-angiotensin system and blood pressure. J. Steroid Biochem. Mol. Biol. 2004, 89, 387–392. [Google Scholar] [CrossRef]
- Shi, Y.Y.; Liu, T.J.; Fu, J.H.; Xu, W.; Wu, L.L.; Hou, A.N.; Xue, X.D. Vitamin D/VDR signalling attenuates lipopolysaccharide-induced acute lung injury by maintaining the integrity of the pulmonary epithelial barrier. Mol. Med. Rep. 2016, 13, 1186–1194. [Google Scholar] [CrossRef]
- Kong, J.; Zhang, Z.; Musch, M.W.; Ning, G.; Sun, J.; Hart, J.; Bissonnette, M.; Li, Y.C. Novel role of the vitamin D receptor in maintaining the integrity of the intestinal mucosal barrier. Am. J. Physiol. Gastrointest. Liver Physiol. 2008, 294, G208–G216. [Google Scholar] [CrossRef]
- Bombardini, T.; Picano, E. Angiotensin-converting enzyme 2 as the molecular bridge between epidemiologic and clinical features of COVID-19. Can. J. Cardiol. 2020, 36, 784-e1. [Google Scholar] [CrossRef]
- Rehan, V.K.; Torday, J.S.; Peleg, S.; Gennaro, L.; Vouros, P.; Rao, D.S.; Reddy, G.S. 1ἁ,25-dihydroxy-3-epi-vitamin D3, a natural metabolite of 1ἁ,25-dihydroxy vitamin D3: Production and biological activity studies in pulmonary alveolar type II cells. Mol. Genet. Metab. 2020, 76, 46–56. [Google Scholar] [CrossRef]
- Phokela, S.S.; Peleg, S.; Moya, F.R.; Alcorn, J.L. Regulation of human pulmonary surfactant protein gene expression by 1ἁ, 25-dihydroxyvitamin D3. Am. J. Physiol. Lung Cell Mol. Physiol. 2005, 289, 617–626. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.M.; Jones, J.L.; Han, J.E.; Alvarez, J.A.; Sloan, J.H.; Konrad, R.J.; Zughaier, S.M.; Martin, G.S.; Ziegler, T.R.; Tangpricha, V. High-Dose Vitamin D3 Administration Is Associated with Increases in Hemoglobin Concentrations in Mechanically Ventilated Critically Ill Adults: A Pilot Double-Blind, Randomized, Placebo-Controlled Trial. J. Parenter. Enter. Nutr. 2018, 42, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Telcian, A.G.; Zdrenghea, M.T.; Edwards, M.R.; Laza-Stanca, V.; Mallia, P.; Johnston, S.L.; Stanciu, L.A. Vitamin D increases the antiviral activity of bronchial epithelial cells in vitro. Antivir. Res. 2017, 137, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Christopher, K.B. Vitamin D and critical illness outcomes. Curr. Opin. Crit. Care 2016, 22, 332–338. [Google Scholar] [CrossRef]
- Holick, M.F. The vitamin D deficiency pandemic: Approaches for diagnosis, treatment and prevention. Rev. Endocr. Metab. Disord. 2017, 1, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Van Der Pols, J.C.; Russell, A.; Bauer, U.; Neale, R.E.; Kimlin, M.G.; Green, A.C. Vitamin D status and skin cancer risk independent of time outdoors: An 11-year prospective study in an Australian community. J. Investig. Dermatol. 2013, 133, 637–641. [Google Scholar] [CrossRef]
- Zadshir, A.; Tareen, N.; Pan, D.; Norris, K.; Martins, D. The prevalence of hypovitaminosis D among US adults: Data from the NHANES III. Ethn. Dis. 2005, 15, S5. [Google Scholar]
- Serrano, M.A.; Cañada, J.; Moreno, J.C.; Gurrea, G. Solar ultraviolet doses and vitamin D in a northern mid-latitude. Sci. Total Environ. 2017, 574, 744–750. [Google Scholar] [CrossRef]
- Bleizgys, A. Vitamin D, and Covid-19: It is time to act. Int. J. Clin. Pract. 2020, e13748. [Google Scholar] [CrossRef]
- Gois, P.H.F.; Ferreira, D.; Olenski, S.; Seguro, A.C. Vitamin D and infectious diseases: Simple bystander or contributing factor? Nutrients 2017, 9, 651. [Google Scholar] [CrossRef]
- Vitamin D—Health Professional Fact Sheet. Available online: https://ods.od.nih.gov/factsheets/VitaminDHealthProfessional/ (accessed on 23 December 2020).
- EFSA. Dietary Deference Values for Vitamin D. Eur. Food Saf. Auth. J. 2016. Available online: https://efsa.onlinelibrary.wiley.com/doi/pdf/10.2903/j.efsa.2016.4547 (accessed on 23 December 2020).
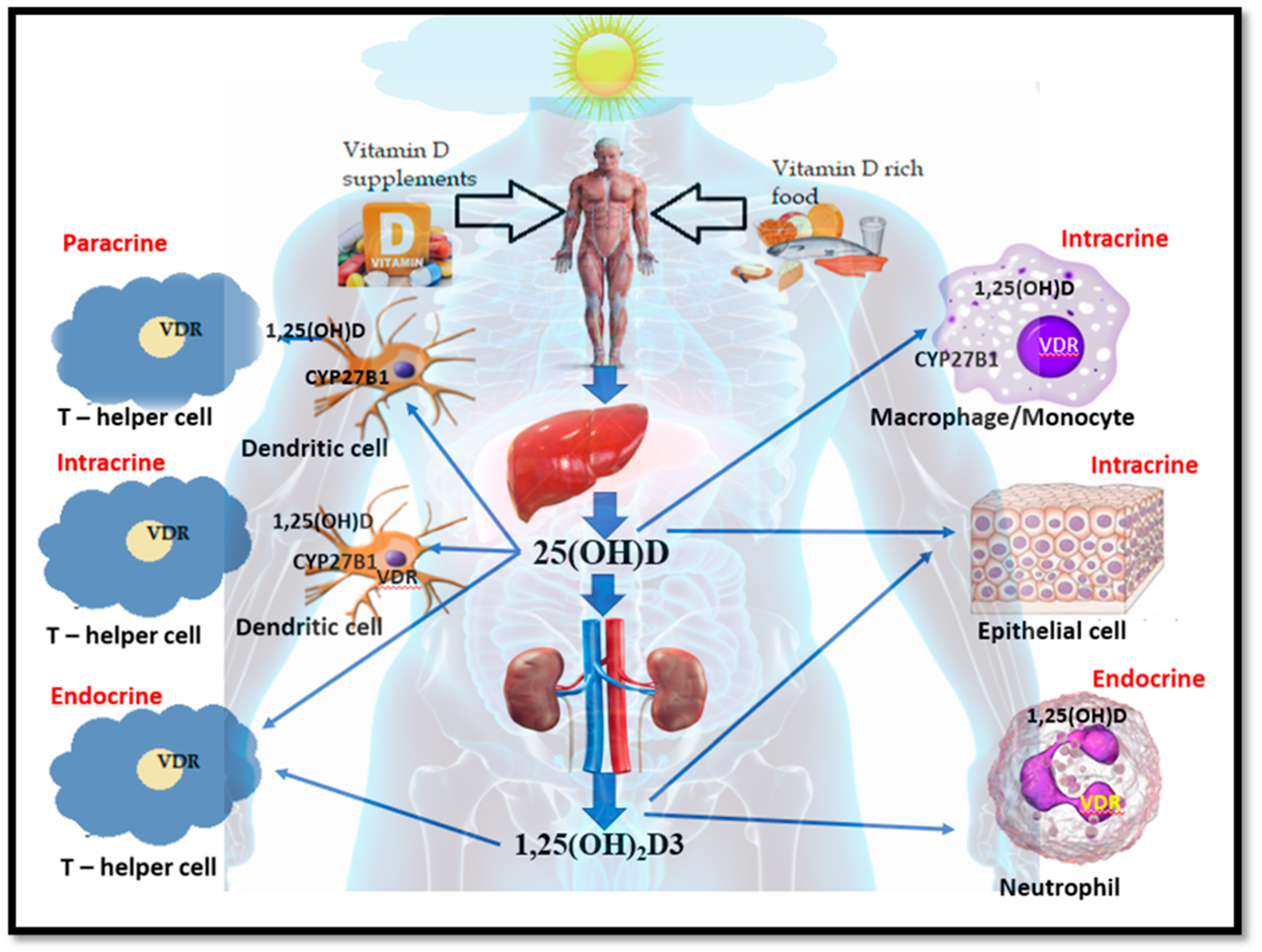
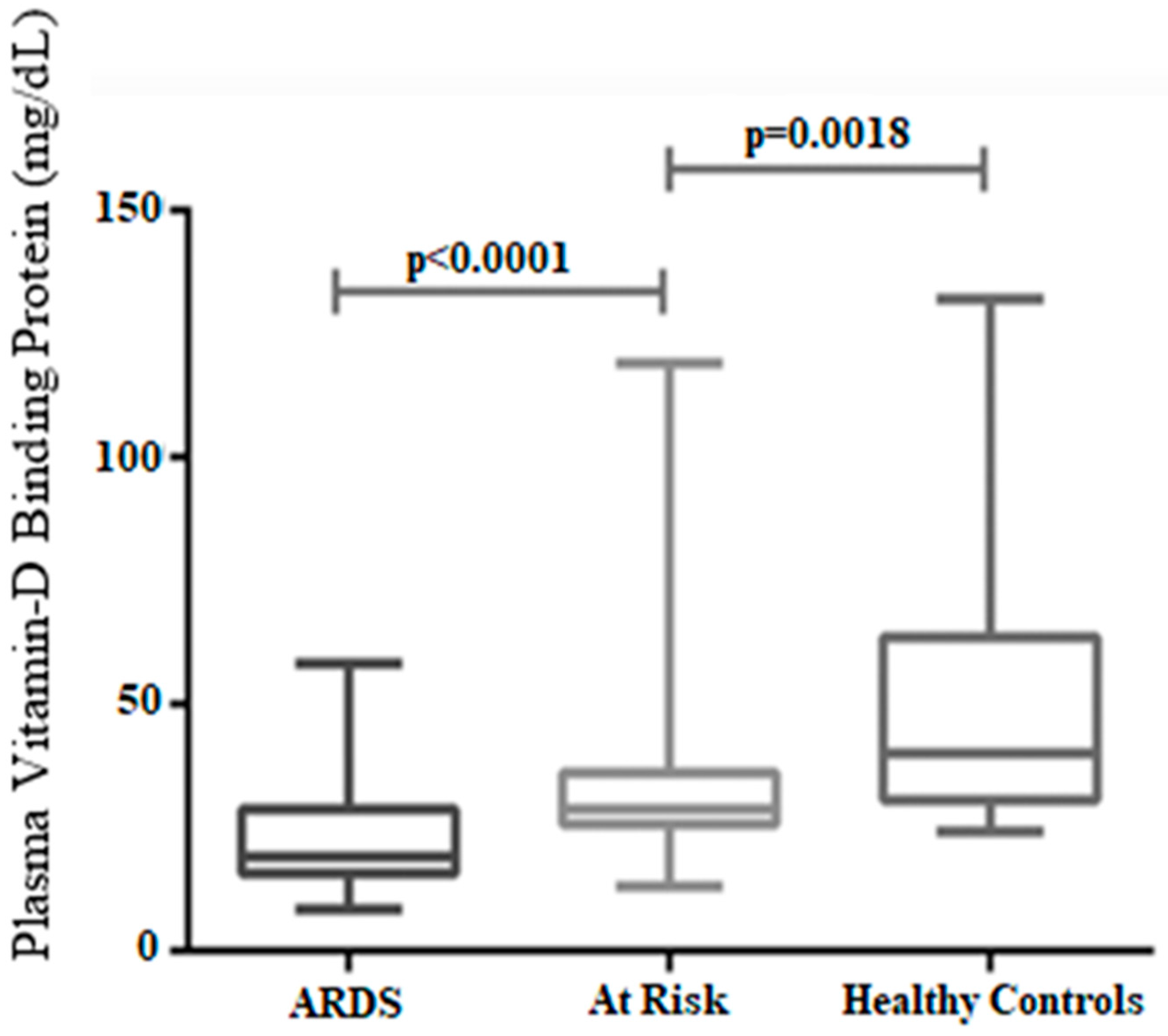
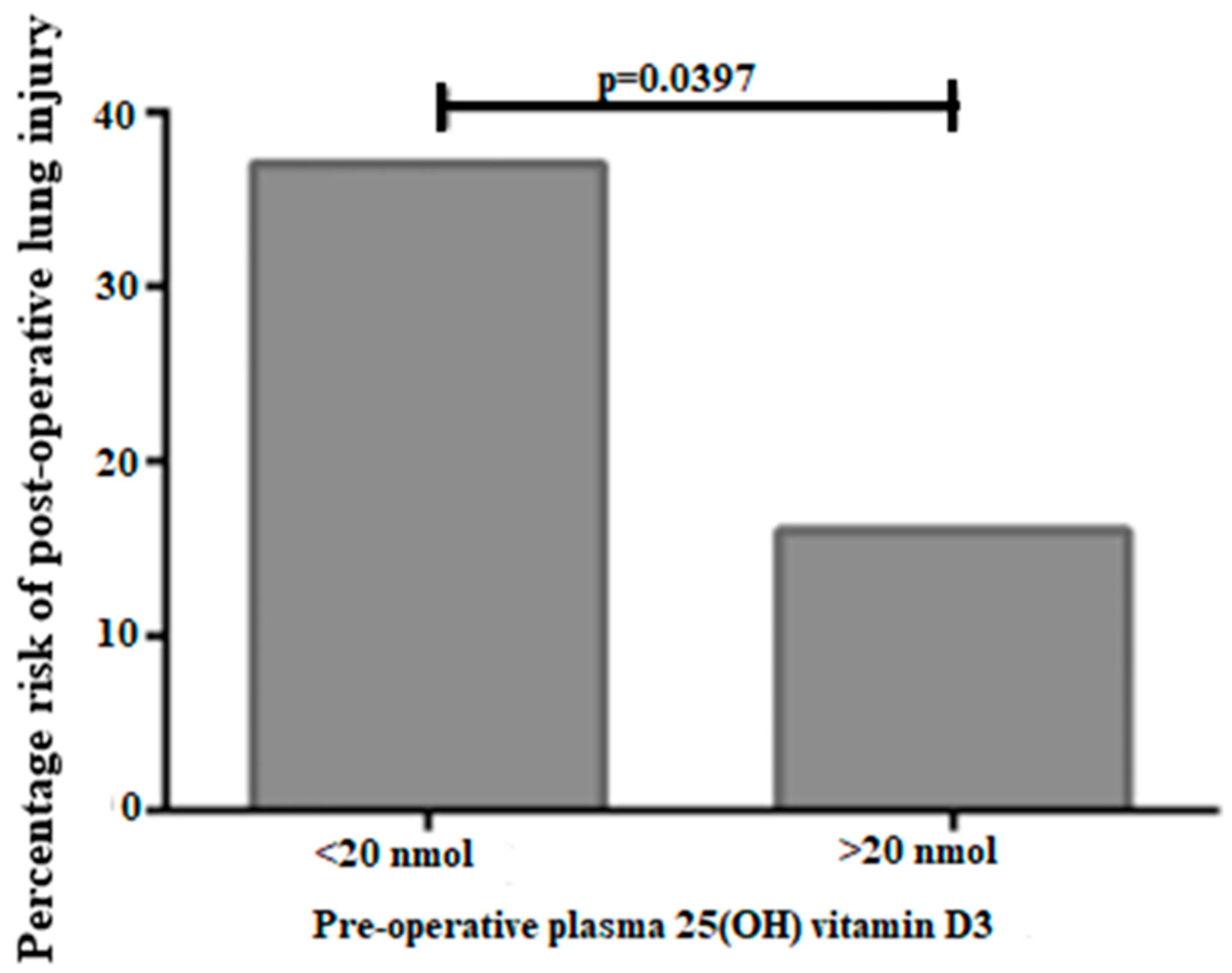
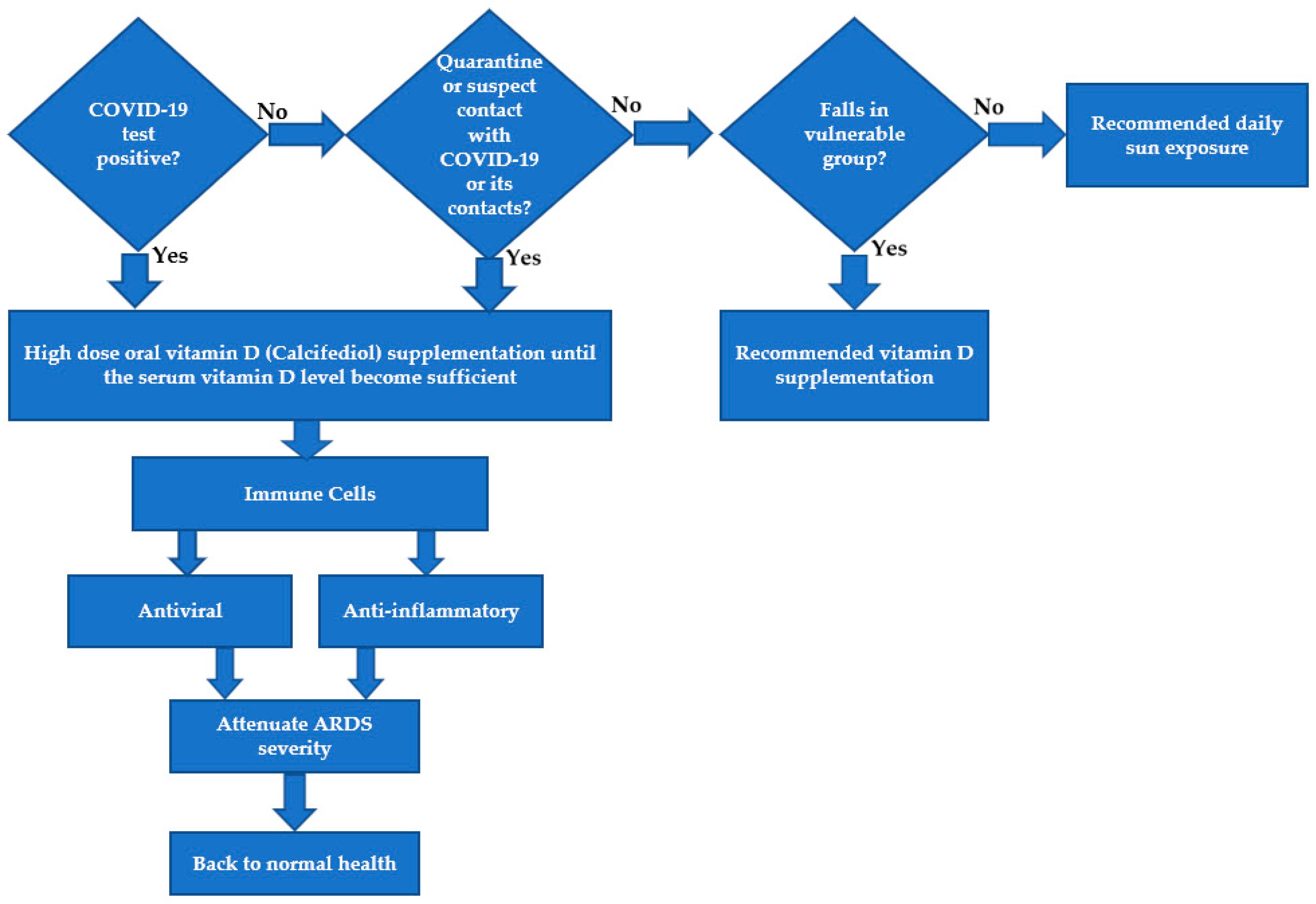
| Symptoms/Characteristics | Relation to Vitamin D | References |
|---|---|---|
| Acute respiratory distress syndrome (ARDS) risk | Inverse correlation | [10,31] |
| Severe pneumonia | Inverse correlation | [20,32,33] |
| Sepsis risk | inverse correlation | [34,35] |
| Pro-inflammatory | Inverse correlation cytokine production | [36,37] |
| C-Reactive Protein increase | Inverse correlation | [38] |
| Study Details | Results/Conclusion | Reference(s) |
|---|---|---|
| Determined the relationship between vitamin D and ARDS. | Vitamin D deficiency was ubiquitous with ARDS patients and was a risk factor in developing ARDS following the esophagectomy. It also found that the pharmacological repletion of vitamin D before esophagectomy reduced the alveolar–capillary damage compared to the vitamin D deficient patient. In a murine model, dietary-induced vitamin D deficiency generated alveolar inflammation, damage in epithelial cells, and hypoxemia. | [10] |
| Investigated the role of supplemented vitamin D in reducing ARTI in a meta-analysis. This study used 25 randomised controlled trial data from to 11,321 participants. | The study found that (i) vitamin D supplementation reduced the risk of ARTI in all patients (odds ratio-OR-0.88); (ii) protective effects were higher in those receiving a daily or weekly dose compared to the bolus dose (OR-0.81); and (iii) among the second group, protective effects were more potent in those who had vitamin D deficiency (<10 ng/mL). The overall conclusion is that vitamin D supplementation protected from ARTI. | [11] |
| Investigated whether vitamin D alleviates lipopolysaccharide (LPS)-induced acute lung injury (ALI). | Vitamin D exhibited positive effects to LPS-induced ALI in rats by modulating the expression of members of the renin–angiotensin system (RAS) such as angiotensin (Ang-1) converting enzyme (ACE and ACE2), renin and Ang-II. | [19] |
| Investigated the association between pre-hospital vitamin D level and acute respiratory failure. | The study found an association between pre-hospital vitamin D levels and the risk of incident acute respiratory failure in critically ill patients, including death. | [31] |
| Investigated the role of vitamin D supplementation in patients undergoing esophagectomy. | Vitamin D supplementation 3–14 days prior to the esophagectomy significantly reduced the potentiality of ARDS during the post-operative environment. | [60] |
| Investigated the association between vitamin D and ALI and ARDS through a review. | Reviewers found a profound role of vitamin D in modulating the immune response and a potential role in ALI. | [62] |
| Investigated the association between vitamin D and acute respiratory infection (ARI) through systematic review. | The review identified a consistent association between vitamin D deficiency and risk to ARI. | [68] |
| Vitamin D deficiency and ARDS. | Vitamin D deficiency was found to be prevalent (90%) among 476 ventilated patients with ARDS, also associated with a longer duration on mechanical ventilation | [72] |
| Investigated whether exogenous vitamin D attenuates lipopolysaccharide (LPS)-induced lung injury via modulating the epithelial cell proliferation. | It was found that vitamin D supplementation attenuates lung injury via (i) stimulating alveolar epithelial type II (ATII) cell proliferation and migration; (ii) reducing epithelial cell apoptosis; and (iii) inhibiting the transforming growth factor (TGF-β) induced epithelial–mesenchymal transition (EMT). The study suggested that vitamin D has therapeutic potential for the resolution of ARDS. | [73] |
| Investigated whether the vitamin D or VDR pathway ameliorates LPS-induced ALI (mice model). | It was found that vitamin D treatment alleviated the LPS-induced lung injury. | [74] |
| Location | Total Confirmed Cases | Total Mortality | Percentage of Mortality to Confirmed Cases | Confirmed Cases Per Million Population | Deaths Per Million Population | Total Population |
|---|---|---|---|---|---|---|
| North of 23.5° N Lat | 13,201,342 | 483,004 | 3.3 | 5378 | 159 | 3,655,796,736 |
| 23.5° N to 23.5° S | 12,320,973 | 364,084 | 2.3 | 3295 | 77 | 3,918,475,190 |
| South of 23.5° S Lat | 1,589,874 | 37,423 | 2.1 | 5811 | 139 | 197,607,413 |
| Age | EFSA (mcg/day) | (IU/day) | ANAM (mcg/day)/(IU/day) |
|---|---|---|---|
| 1–11 months | 10 | 400 | - |
| 1–18 years | 15 | 600 | - |
| >18 years | 15 | 600 | 20/800 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abraham, J.; Dowling, K.; Florentine, S. Can Optimum Solar Radiation Exposure or Supplemented Vitamin D Intake Reduce the Severity of COVID-19 Symptoms? Int. J. Environ. Res. Public Health 2021, 18, 740. https://doi.org/10.3390/ijerph18020740
Abraham J, Dowling K, Florentine S. Can Optimum Solar Radiation Exposure or Supplemented Vitamin D Intake Reduce the Severity of COVID-19 Symptoms? International Journal of Environmental Research and Public Health. 2021; 18(2):740. https://doi.org/10.3390/ijerph18020740
Chicago/Turabian StyleAbraham, Joji, Kim Dowling, and Singarayer Florentine. 2021. "Can Optimum Solar Radiation Exposure or Supplemented Vitamin D Intake Reduce the Severity of COVID-19 Symptoms?" International Journal of Environmental Research and Public Health 18, no. 2: 740. https://doi.org/10.3390/ijerph18020740
APA StyleAbraham, J., Dowling, K., & Florentine, S. (2021). Can Optimum Solar Radiation Exposure or Supplemented Vitamin D Intake Reduce the Severity of COVID-19 Symptoms? International Journal of Environmental Research and Public Health, 18(2), 740. https://doi.org/10.3390/ijerph18020740






