Effects of Exercise on the Skin Epithelial Barrier of Young Elite Athletes-Swimming Comparatively to Non-Water Sports Training Session
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Assessments
2.3. Statistical Analysis
2.4. Participants
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Font-Ribera, L.; Villanueva, C.M.; Gracia-Lavedan, E.; Borras-Santos, A.; Kogevinas, M.; Zock, J.P. Indoor swimming pool attendance and respiratory and dermal health in schoolchildren—HITEA Catalonia. Respir. Med. 2014, 108, 1056–1059. [Google Scholar] [CrossRef]
- Beggs, S.; Foong, Y.C.; Le, H.C.; Noor, D.; Wood-Baker, R.; Walters, J.A. Swimming training for asthma in children and adolescents aged 18 years and under. Cochrane Database Syst. Rev. 2013. [Google Scholar] [CrossRef] [PubMed]
- Couto, M.; Kurowski, M.; Moreira, A.; Bullens, D.M.A.; Carlsen, K.H.; Delgado, L.; Kowalski, M.L.; Seys, S.F. Mechanisms of exercise-induced bronchoconstriction in athletes: Current perspectives and future challenges. Allergy 2018, 73, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Carlsen, K.H.; Anderson, S.D.; Bjermer, L.; Bonini, S.; Brusasco, V.; Canonica, W.; Cummiskey, J.; Delgado, L.; Del Giacco, S.R.; Drobnic, F.; et al. Exercise-induced asthma, respiratory and allergic disorders in elite athletes: Epidemiology, mechanisms and diagnosis: Part I of the report from the Joint Task Force of the European Respiratory Society (ERS) and the European Academy of Allergy and Clinical Immunology (EAACI) in cooperation with GA2LEN. Allergy 2008, 63, 387–403. [Google Scholar] [CrossRef] [PubMed]
- Bernard, A.; Nickmilder, M.; Dumont, X. Chlorinated pool attendance, airway epithelium defects and the risks of allergic diseases in adolescents: Interrelationships revealed by circulating biomarkers. Environ. Res. 2015, 140, 119–126. [Google Scholar] [CrossRef]
- Couto, M.; Moreira, A. The athlete “out of breath”. Eur. Ann. Allergy Clin. Immunol. 2016, 48, 36–45. [Google Scholar]
- Fernandez-Luna, A.; Burillo, P.; Felipe, J.L.; del Corral, J.; Garcia-Unanue, J.; Gallardo, L. Perceived health problems in swimmers according to the chemical treatment of water in swimming pools. Eur. J. Sport Sci. 2016, 16, 256–265. [Google Scholar] [CrossRef]
- Lazarov, A.; Nevo, K.; Pardo, A.; Froom, P. Self-reported skin disease in hydrotherapists working in swimming pools. Contact Dermat. 2005, 53, 327–331. [Google Scholar] [CrossRef]
- Basler, R.S.W.; Basler, G.C.; Palmer, A.H.; Garcia, M.A. Special skin symptoms seen in swimmers. J. Am. Acad. Dermatol. 2000, 43, 299–305. [Google Scholar] [CrossRef]
- Font-Ribera, L.; Kogevinas, M.; Zock, J.P.; Nieuwenhuijsen, M.J.; Heederik, D.; Villanueva, C.M. Swimming pool attendance and risk of asthma and allergic symptoms in children. Eur. Respir. J. 2009, 34, 1304–1310. [Google Scholar] [CrossRef]
- Fantuzzi, G.; Righi, E.; Predieri, G.; Giacobazzi, P.; Mastroianni, K.; Aggazzotti, G. Prevalence of Ocular, Respiratory and Cutaneous Symptoms in Indoor Swimming Pool Workers and Exposure to Disinfection By-Products (DBPs). Int. J. Environ. Res. Public Health 2010, 7, 1379–1391. [Google Scholar] [CrossRef] [PubMed]
- Pardo, A.; Nevo, K.; Vigiser, D.; Lazarov, A. The effect of physical and chemical properties of swimming pool water and its close environment on the development of contact dermatitis in hydrotherapists. Am. J. Ind. Med. 2007, 50, 122–126. [Google Scholar] [CrossRef] [PubMed]
- Kelleher, M.; Dunn-Galvin, A.; Hourihane, J.O.; Murray, D.; Campbell, L.E.; McLean, W.H.; Irvine, A.D. Skin barrier dysfunction measured by transepidermal water loss at 2 days and 2 months predates and predicts atopic dermatitis at 1 year. J. Allergy Clin. Immunol. 2015, 135, 930–935.e931. [Google Scholar] [CrossRef] [PubMed]
- Flohr, C.; England, K.; Radulovic, S.; McLean, W.H.; Campbel, L.E.; Barker, J.; Perkin, M.; Lack, G. Filaggrin loss-of-function mutations are associated with early-onset eczema, eczema severity and transepidermal water loss at 3 months of age. Br. J. Dermatol. 2010, 163, 1333–1336. [Google Scholar] [CrossRef]
- Horimukai, K.; Morita, K.; Narita, M.; Kondo, M.; Kabashima, S.; Inoue, E.; Sasaki, T.; Niizeki, H.; Saito, H.; Matsumoto, K.; et al. Transepidermal water loss measurement during infancy can predict the subsequent development of atopic dermatitis regardless of filaggrin mutations. Allergol. Int. 2016, 65, 103–108. [Google Scholar] [CrossRef]
- Boralevi, F.; Hubiche, T.; Leaute-Labreze, C.; Saubusse, E.; Fayon, M.; Roul, S.; Maurice-Tison, S.; Taieb, A. Epicutaneous aeroallergen sensitization in atopic dermatitis infants—Determining the role of epidermal barrier impairment. Allergy 2008, 63, 205–210. [Google Scholar] [CrossRef]
- Mattila, P.; Joenvaara, S.; Renkonen, J.; Toppila-Salmi, S.; Renkonen, R. Allergy as an epithelial barrier disease. Clin. Transl. Allergy 2011, 1, 5. [Google Scholar] [CrossRef]
- Cunha, M.; Mendes, F.; Paciência, I.; Rodolfo, A.; Carneiro-Leão, L.; Rama, T.; Rufo, J.; Delgado, L.; Moreira, A. The effect of inspiratory muscle training on swimming performance, inspiratory muscle strength, lung function, and perceived breathlessness in elite swimmers: A randomized controlled trial. Porto Biomed. J. 2019, 4, e49. [Google Scholar] [CrossRef]
- Asher, M.I.; Keil, U.; Anderson, H.R.; Beasley, R.; Crane, J.; Martinez, F.; Mitchell, E.A.; Pearce, N.; Sibbald, B.; Stewart, A.W.; et al. International Study of Asthma and Allergies in Childhood (Isaac)—Rationale and Methods. Eur. Respir. J. 1995, 8, 483–491. [Google Scholar] [CrossRef]
- Heinzerling, L.; Mari, A.; Bergmann, K.C.; Bresciani, M.; Burbach, G.; Darsow, U.; Durham, S.; Fokkens, W.; Gjomarkaj, M.; Haahtela, T.; et al. The skin prick test—European standards. Clin. Transl. Allergy 2013, 3, 3. [Google Scholar] [CrossRef]
- Miller, M.R.; Hankinson, J.; Brusasco, V.; Burgos, F.; Casaburi, R.; Coates, A.; Crapo, R.; Enright, P.; van der Grinten, C.P.; Gustafsson, P.; et al. Standardisation of spirometry. Eur. Respir. J. 2005, 26, 319–338. [Google Scholar] [CrossRef] [PubMed]
- Du Plessis, J.; Stefaniak, A.; Eloff, F.; John, S.; Agner, T.; Chou, T.-C.; Nixon, R.; Steiner, M.; Franken, A.; Kudla, I.; et al. International guidelines for the in vivo assessment of skin properties in non-clinical settings: Part 2. transepidermal water loss and skin hydration. Skin Res. Technol. 2013, 19, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Luebberding, S.; Kolbe, L.; Kerscher, M. Influence of sportive activity on skin barrier function: A quantitative evaluation of 60 athletes. Int. J. Dermatol. 2013, 52, 745–749. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, J.H.; Haskell, W.; Snell, P.; Van Camp, S.P. Task Force 8: Classification of sports. J. Am. Coll. Cardiol. 2005, 45, 1364–1367. [Google Scholar] [CrossRef]
- Sotoodian, B.; Maibach, H.I. Noninvasive test methods for epidermal barrier function. Clin. Dermatol. 2012, 30, 301–310. [Google Scholar] [CrossRef]
- Machado, M.; Hadgraft, J.; Lane, M.E. Assessment of the variation of skin barrier function with anatomic site, age, gender and ethnicity. Int. J. Cosmet. Sci. 2010, 32, 397–409. [Google Scholar] [CrossRef]
- Kottner, J.; Lichterfeld, A.; Blume-Peytavi, U. Transepidermal water loss in young and aged healthy humans: A systematic review and meta-analysis. Arch. Dermatol. Res. 2013, 305, 315–323. [Google Scholar] [CrossRef]
- Mayrovitz, H.N.; Bernal, M.; Brlit, F.; Desfor, R. Biophysical measures of skin tissue water: Variations within and among anatomical sites and correlations between measures. Skin Res. Technol. 2013, 19, 47–54. [Google Scholar] [CrossRef]
- Mohammed, D.; Matts, P.J.; Hadgraft, J.; Lane, M.E. Variation of stratum corneum biophysical and molecular properties with anatomic site. AAPS J. 2012, 14, 806–812. [Google Scholar] [CrossRef]
- Altemus, M.; Rao, B.; Dhabhar, F.S.; Ding, W.; Granstein, R.D. Stress-Induced Changes in Skin Barrier Function in Healthy Women. J. Investig. Dermatol. 2001, 117, 309–317. [Google Scholar] [CrossRef]
- Rougier, A.; Lotte, C.; Corcuff, P.; Maibach, H. Relationship between skin permeability and corneocyte size according to anatomic site, age, and sex in man. J. Soc. Cosmet. Chem. 1988, 39, 15–26. [Google Scholar]
- Felgueiras, F.; Mourão, Z.; Morais, C.; Santos, H.; Gabriel, M.F.; de Oliveira Fernandes, E. Comprehensive assessment of the indoor air quality in a chlorinated Olympic-size swimming pool. Environ. Int. 2020, 136, 105401. [Google Scholar] [CrossRef] [PubMed]
- Luebberding, S.; Krueger, N.; Kerscher, M. Age-related changes in skin barrier function—Quantitative evaluation of 150 female subjects. Int. J. Cosmet. Sci. 2013, 35, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Gardinier, S.; Guehenneux, S.; Latreille, J.; Guinot, C.; Tschachler, E. Variations of skin biophysical properties after recreational swimming. Skin Res. Technol. 2009, 15, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Garcia Bartels, N.; Rosler, S.; Martus, P.; Stroux, A.; Lonnfors, S.; Reisshauer, A.; Blume-Peytavi, U. Effect of baby swimming and baby lotion on the skin barrier of infants aged 3–6 months. J. Dtsch. Dermatol. Ges. 2011, 9, 1018–1025. [Google Scholar] [CrossRef]
- Seki, T.; Morimatsu, S.; Nagahori, H.; Morohashi, M. Free Residual Chlorine in Bathing Water Reduces the Water-Holding Capacity of the Stratum Corneum in Atopic Skin. J. Dermatol. 2003, 30, 196–202. [Google Scholar] [CrossRef]
- Mauro, T.; Holleran, W.M.; Grayson, S.; Gao, W.N.; Man, M.Q.; Kriehuber, E.; Behne, M.; Feingold, K.R.; Elias, P.M. Barrier recovery is impeded at neutral pH, independent of ionic effects: Implications for extracellular lipid processing (vol 290, pg 215, 1998). Arch. Dermatol. Res. 1998, 290, 405. [Google Scholar] [CrossRef]
| Characteristics | Swimmers | Football Players | p-Value |
|---|---|---|---|
| n (males) | 33 (10) | 25 (19) | 0.001 1 |
| Age (years), median (IQR) | 14 (13–16) | 15 (14–16) | 0.383 |
| BMI, kg/m2, median (IQR) | 20.8 (19.5–22.2) | 20.8 (19.4–22.5) | 0.588 |
| Asthma, n (%) | 1 (3.0) | 3 (12.0) | 0.609 |
| Rhinitis, n (%) | 10 (30.3) | 2 (8.0) | 0.018 1 |
| Atopic dermatitis, n (%) | 2 (6.1) | 1 (4.0) | 1.000 |
| Allergic sensitization, n (%) | 16 (48.5) | 9 (36.0) | 0.182 |
| Swimmers (n = 33) | Football Players (n = 25) | Swimmers vs. Football Players | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Before (T0) | After (T1) | 30′ After (T2) | p-Value After | p-Value 30′ After | Before (T0) | After (T1) | 30′ After (T2) | p-Value After | p-Value 30′ After | p-Value After | p-Value 30′ After | |
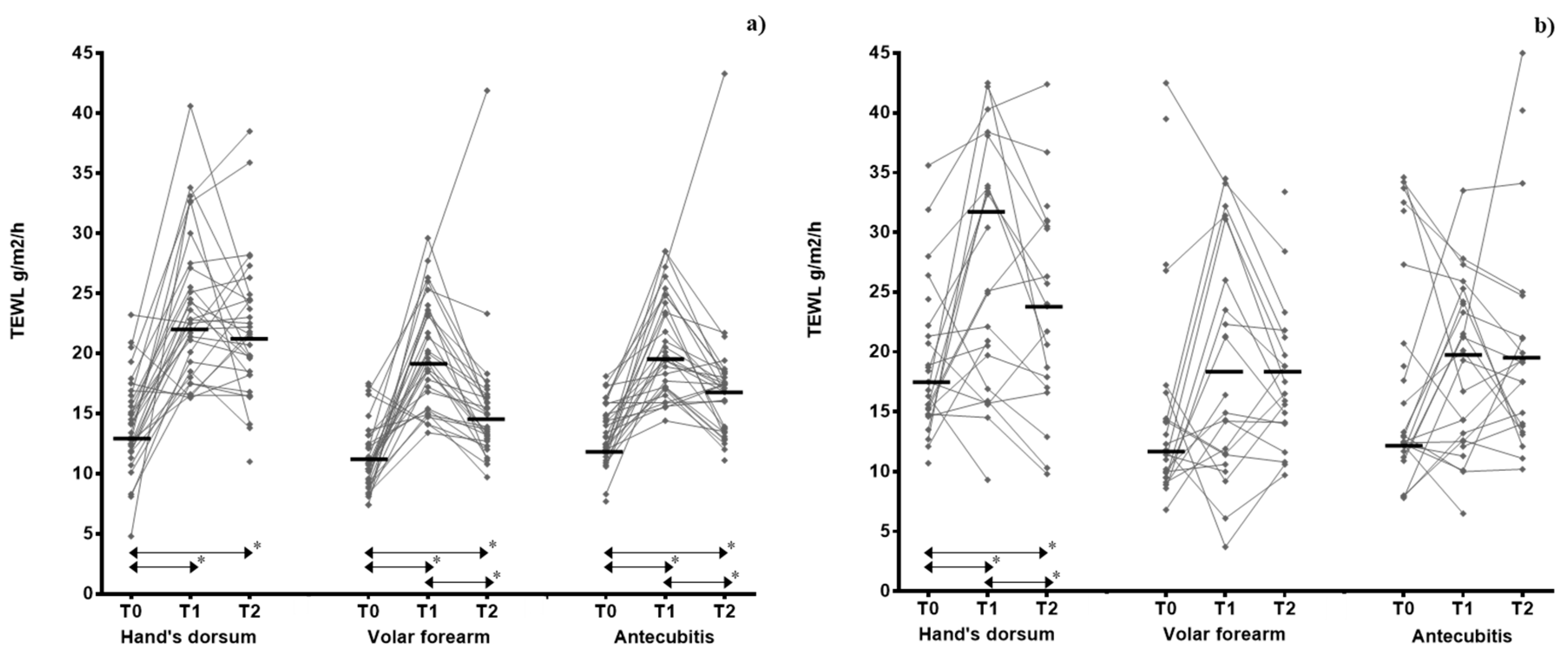
| Hand’s dorsum | 13.7 (11.8–16.3) | 22.7 (18.4–27.2) | 21.5 (18.2–24.5) | <0.001 | <0.001 | 17.1 (14.7–21.9) | 31.8 (17.6–39.8) | 24.0 (17.5–31.6) | 0.020 | 0.043 | 0.279 | 0.236 |
| Volar forearm | 10.7 (9.2–12.9) | 19.3 (15.4–23.5) | 14.4 (12.9–16.4) | <0.001 | <0.001 | 12.7 (9.9–17.0) | 18.8 (11.4–32.0) | 18.8 (14.0–25.9) | 0.086 | 0.085 | 0.002 | 0.199 |
| Antecubitis | 13.1 (11.8–15.6) | 19.6 (17.1–24.4) | 16.9 (13.6–18.2) | <0.001 | <0.001 | 13.2 (12.2–30.6) | 19.7 (12.5–24.9) | 19.3 (13.9–24.9) | 0.584 | 0.341 | 0.019 | 0.252 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paciência, I.; Rodolfo, A.; Leão, L.; Silva, D.; Cavaleiro Rufo, J.; Mendes, F.; Padrão, P.; Moreira, P.; Laerte Boechat, J.; Delgado, L.; et al. Effects of Exercise on the Skin Epithelial Barrier of Young Elite Athletes-Swimming Comparatively to Non-Water Sports Training Session. Int. J. Environ. Res. Public Health 2021, 18, 653. https://doi.org/10.3390/ijerph18020653
Paciência I, Rodolfo A, Leão L, Silva D, Cavaleiro Rufo J, Mendes F, Padrão P, Moreira P, Laerte Boechat J, Delgado L, et al. Effects of Exercise on the Skin Epithelial Barrier of Young Elite Athletes-Swimming Comparatively to Non-Water Sports Training Session. International Journal of Environmental Research and Public Health. 2021; 18(2):653. https://doi.org/10.3390/ijerph18020653
Chicago/Turabian StylePaciência, Inês, Ana Rodolfo, Leonor Leão, Diana Silva, João Cavaleiro Rufo, Francisca Mendes, Patrícia Padrão, Pedro Moreira, Jose Laerte Boechat, Luís Delgado, and et al. 2021. "Effects of Exercise on the Skin Epithelial Barrier of Young Elite Athletes-Swimming Comparatively to Non-Water Sports Training Session" International Journal of Environmental Research and Public Health 18, no. 2: 653. https://doi.org/10.3390/ijerph18020653
APA StylePaciência, I., Rodolfo, A., Leão, L., Silva, D., Cavaleiro Rufo, J., Mendes, F., Padrão, P., Moreira, P., Laerte Boechat, J., Delgado, L., & Moreira, A. (2021). Effects of Exercise on the Skin Epithelial Barrier of Young Elite Athletes-Swimming Comparatively to Non-Water Sports Training Session. International Journal of Environmental Research and Public Health, 18(2), 653. https://doi.org/10.3390/ijerph18020653








