Correlation between Polycyclic Aromatic Hydrocarbons in Wharf Roach (Ligia spp.) and Environmental Components of the Intertidal and Supralittoral Zone along the Japanese Coast
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Sampling
2.3. PAH Analysis
2.4. Quality Assurance and Quality Control
2.5. Statistical Analysis
3. Results and Discussion
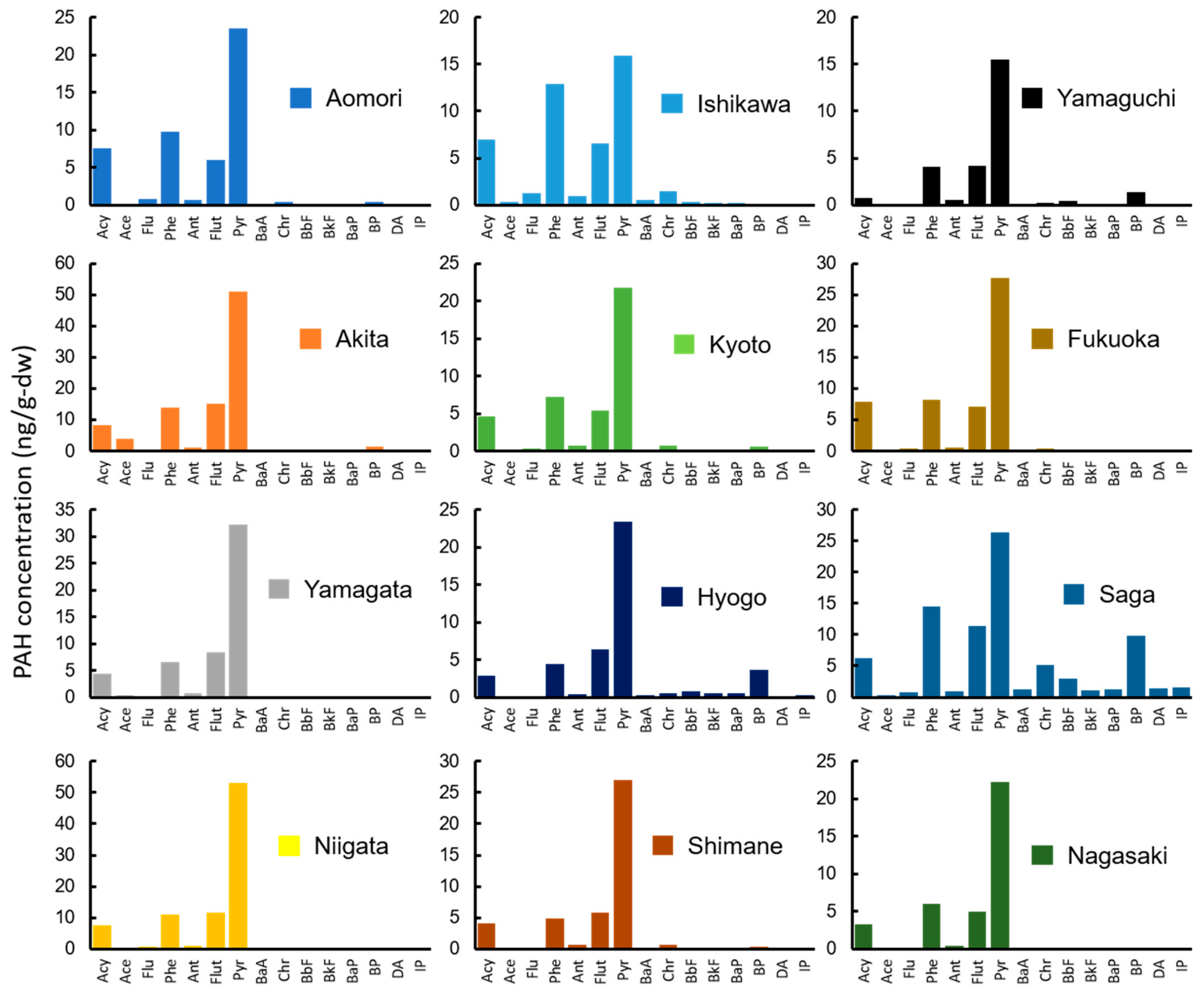
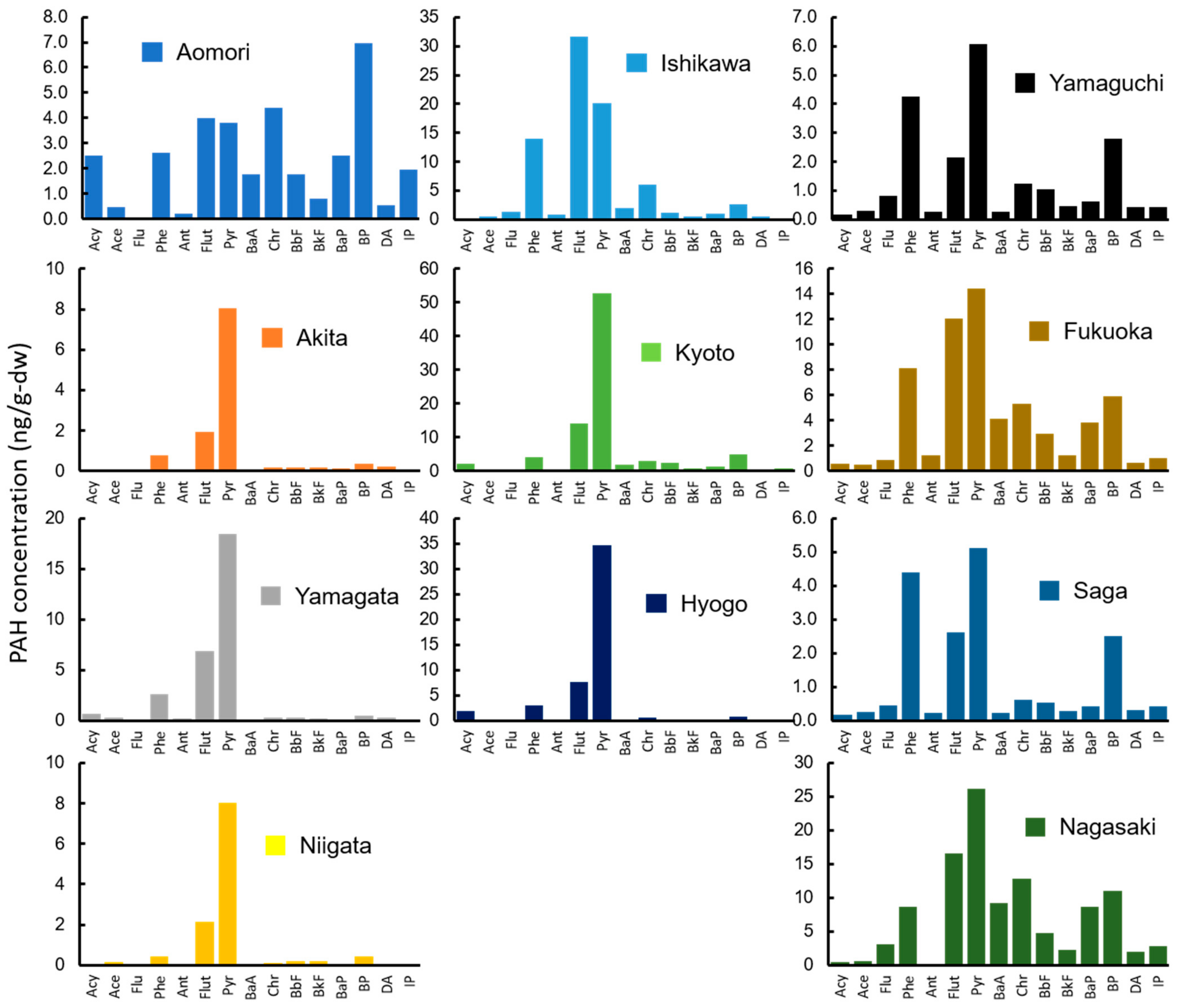
3.1. PAH Concentrations in Organisms
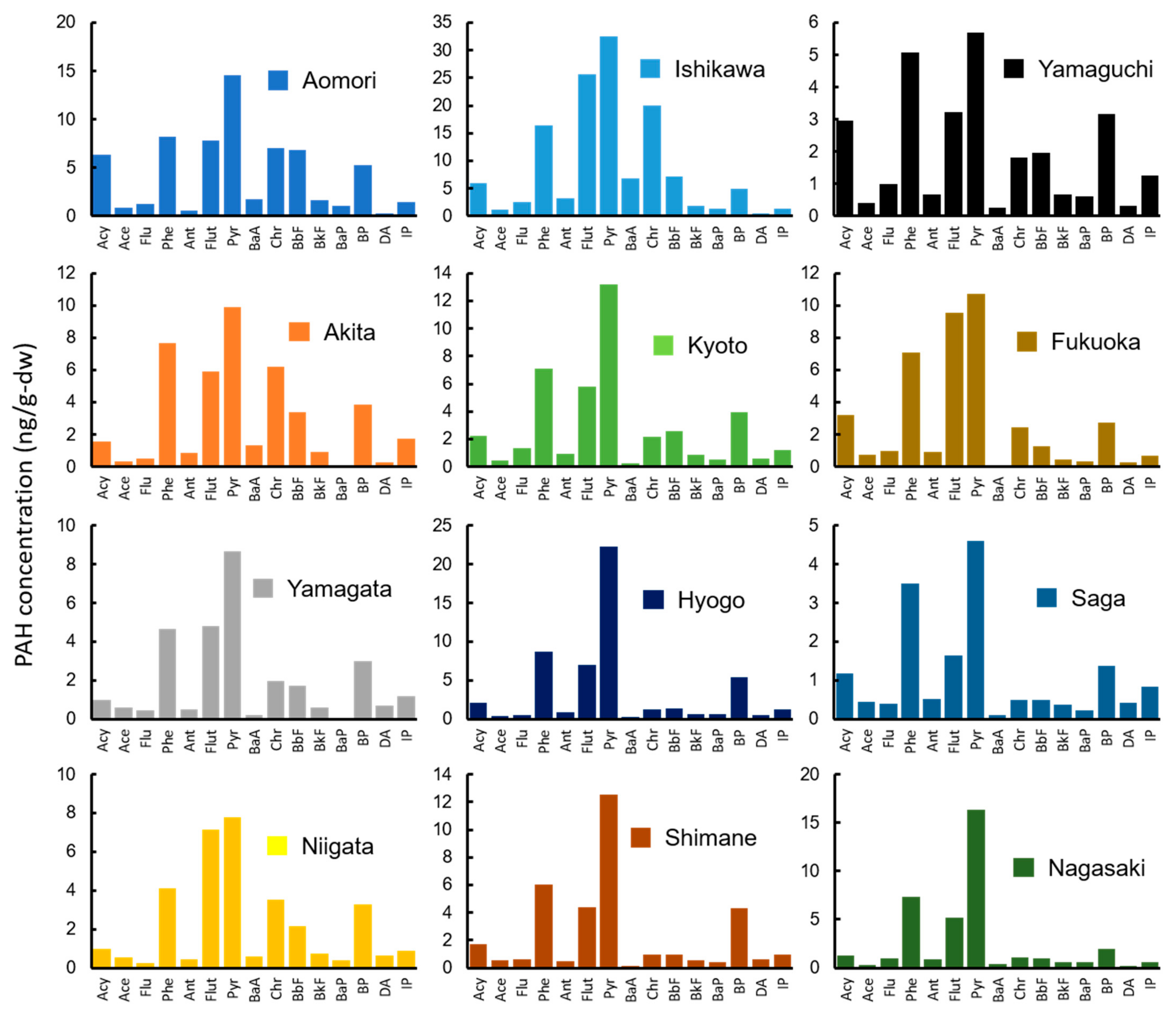
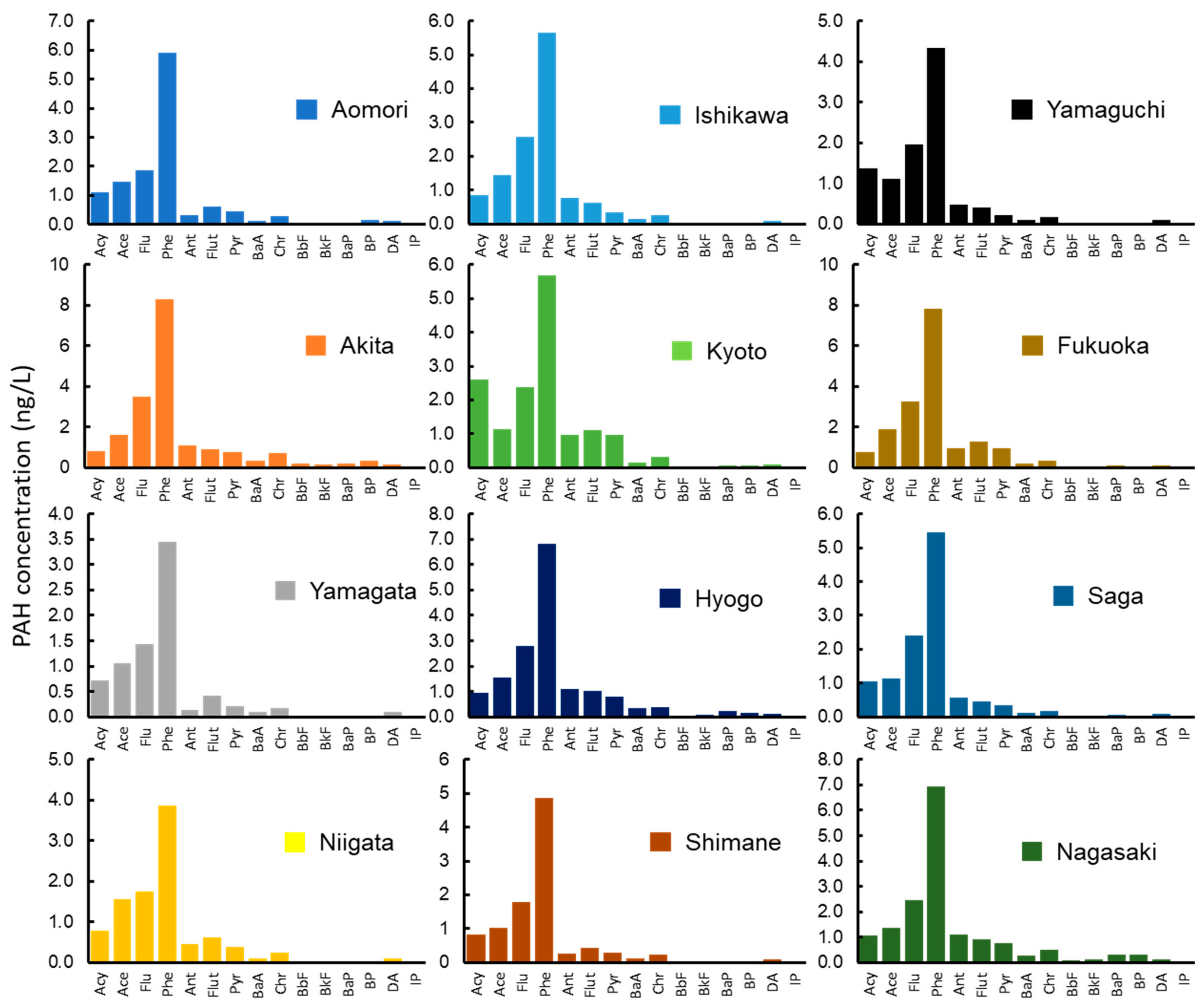
3.2. PAH Concentrations in Environmental Media
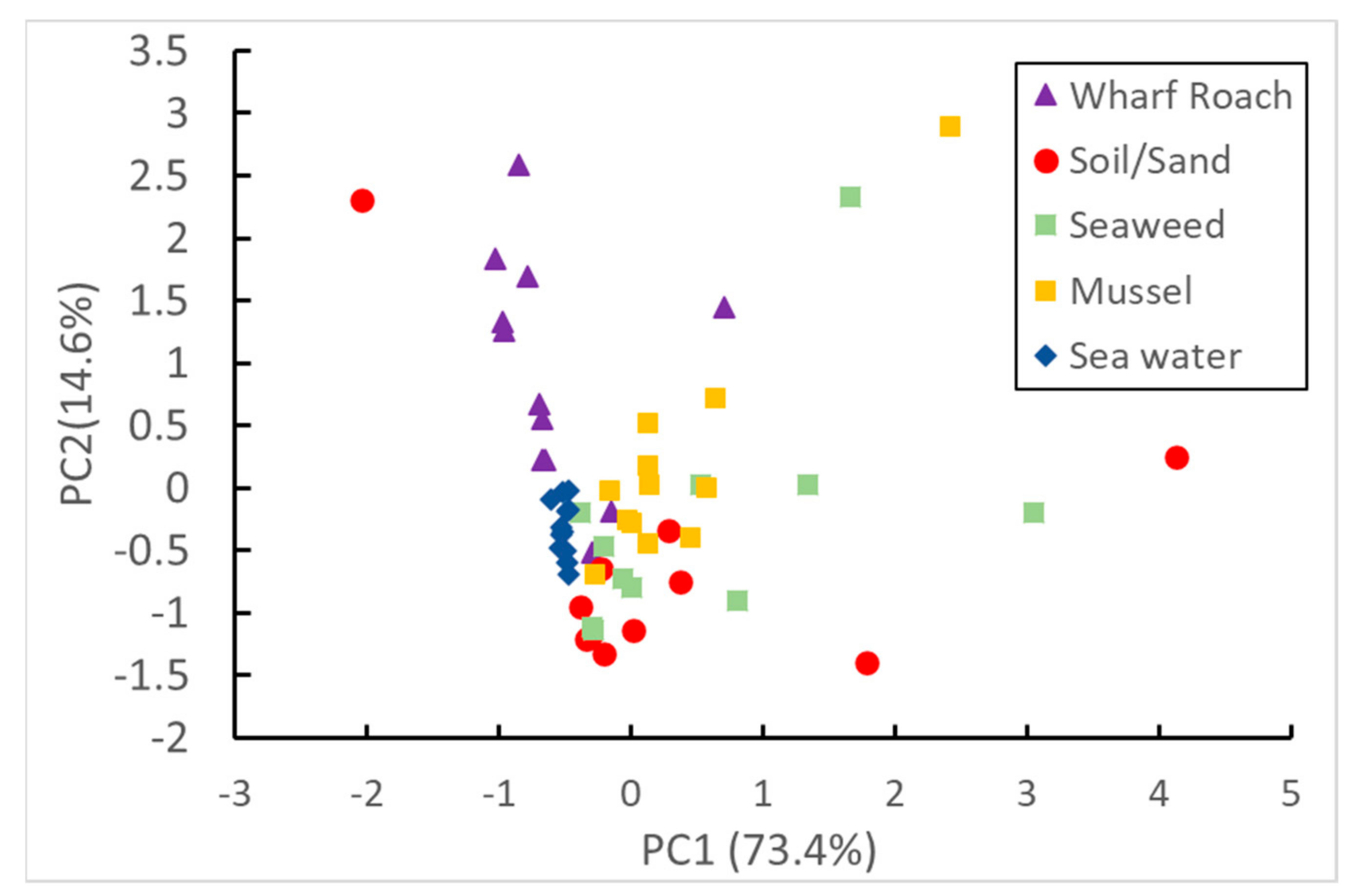
3.3. PCA
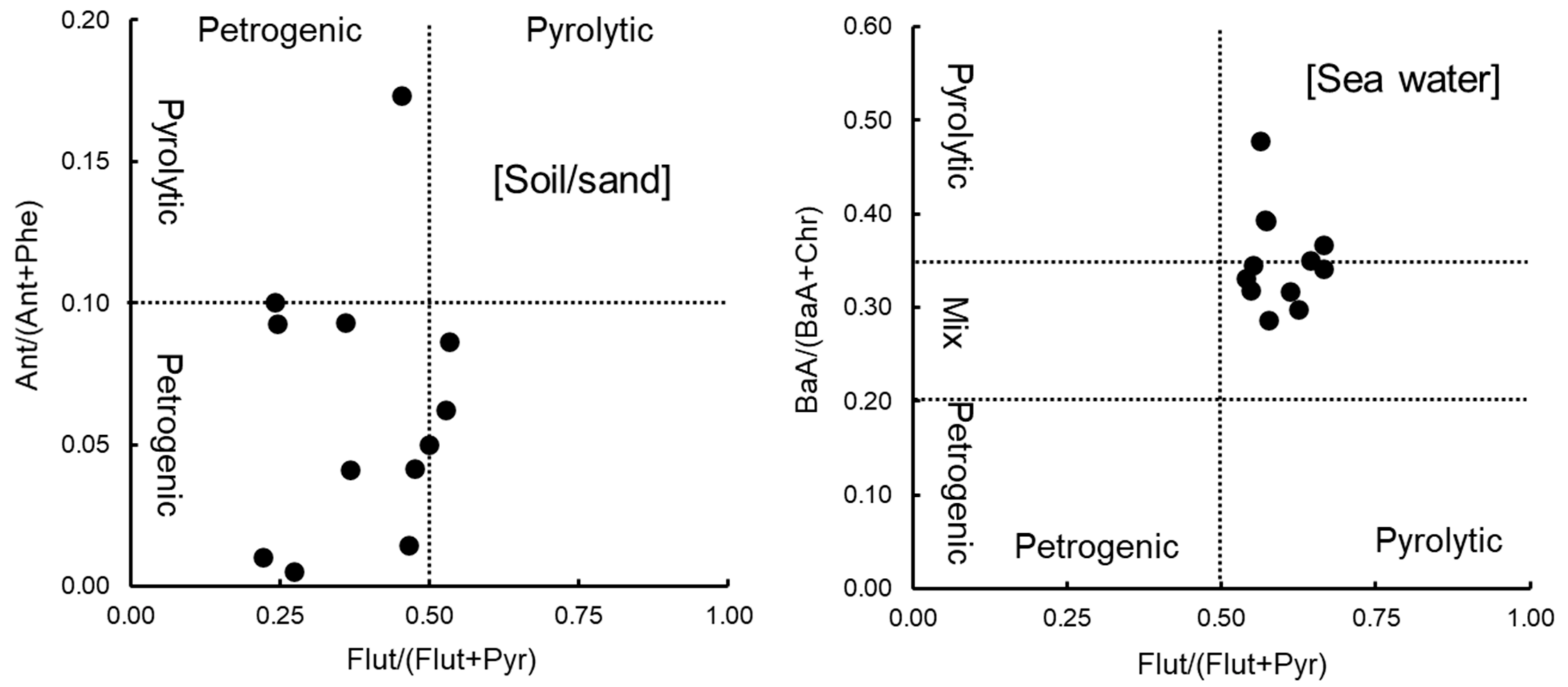
3.4. Identification of Pollution Sources
3.5. Correlation between PAH Concentrations in Wharf Roaches and Environmental Components
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Beyer, J.; Trannum, H.C.; Bakke, T.; Hodson, P.V.; Collier, T.K. Environmental effects of the Deepwater Horizon oil spill: A review. Mar. Pollut. Bull. 2016, 110, 28–51. [Google Scholar] [CrossRef] [PubMed]
- Koyama, J.; Uno, S.; Nagai, Y.; Anukorn, B. Early monitoring of spilled oil contamination in Rayong, Thailand. Jpn. J. Environ. Toxicol. 2016, 19, 25–33. [Google Scholar] [CrossRef]
- Uno, S.; Koyama, J.; Kokushi, E.; Monteclaro, H.; Santander, S.; Cheikyula, J.O.; Miki, S.; Añasco, N.; Pahila, I.G.; Taberna, H.S., Jr.; et al. Monitoring of PAHs and alkylated PAHs in aquatic organisms after 1 month from the Solar I oil spill off the coast of Guimaras Island, Philippines. Environ. Monit. Assess. 2010, 165, 501–515. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Shafy, H.I.; Mansour, M.S. A review on polycyclic aromatic hydrocarbons: Source, environmental impact, effect on human health and remediation. Egypt. J. Pet. 2016, 25, 107–123. [Google Scholar] [CrossRef]
- Mojiri, A.; Zhou, J.L.; Ohashi, A.; Ozaki, N.; Kindaichi, T. Comprehensive review of polycyclic aromatic hydrocarbons in water sources, their effects and treatments. Sci. Total Environ. 2019, 696, 133971. [Google Scholar] [CrossRef]
- Kim, E.J.; Oh, J.E.; Chang, Y.S. Effects of forest fire on the level and distribution of PCDD/Fs and PAHs in soil. Sci. Total Environ. 2003, 311, 177–189. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, S.; Lohmann, R.; Yu, N.; Zhang, C.; Gao, Y.; Zhao, J.; Ma, L. Source apportionment of gaseous and particulate PAHs from traffic emission using tunnel measurements in Shanghai, China. Atmos. Environ. 2015, 107, 129–136. [Google Scholar] [CrossRef]
- Hayakawa, K.; Tang, N.; Nagato, E.G.; Toriba, A.; Sakai, S.; Kano, F.; Goto, S.; Endo, O.; Arashidani, K.I.; Kakimoto, H. Long term trends in atmospheric concentrations of polycyclic aromatic hydrocarbons and nitropolycyclic aromatic hydrocarbons: A study of Japanese cities from 1997 to 2014. Environ. Pollut. 2018, 233, 474–482. [Google Scholar] [CrossRef]
- Slezakova, K.; Pires, J.C.M.; Castro, D.; Alvim-Ferraz, M.C.M.; Delerue-Matos, C.; Morais, S.; Pereira, M.C. PAH air pollution at a Portuguese urban area: Carcinogenic risks and sources identification. Environ. Sci. Pollut. Res. 2013, 20, 3932–3945. [Google Scholar] [CrossRef]
- Offiong, N.A.O.; Inam, E.J.; Etuk, H.S.; Essien, J.P. Current status and challenges of remediating petroleum-derived PAHs in soils: Nigeria as a case study for developing countries. Remediation 2019, 30, 65–75. [Google Scholar] [CrossRef]
- Titaley, I.A.; Simonich, S.L.M.; Larsson, M. Recent Advances in the Study of the Remediation of Polycyclic Aromatic Compound (PAC)-Contaminated Soils: Transformation Products, Toxicity, and Bioavailability Analyses. Environ. Sci. Technol. Lett. 2020, 7, 873–882. [Google Scholar] [CrossRef]
- Miki, S.; Uno, S.; Ito, K.; Koyama, J.; Tanaka, H. Distributions of polycyclic aromatic hydrocarbons and alkylated polycyclic aromatic hydrocarbons in Osaka Bay, Japan. Mar. Pollut. Bull. 2014, 85, 558–565. [Google Scholar] [CrossRef] [PubMed]
- Sampei, Y.; Uraoka, S.I.; Ono, T.; Dettman, D.L. Polycyclic aromatic hydrocarbons (PAHs) in sediment cores from lakes Shinji and Nakaumi, SW Japan: A proxy of recent fire events in the watershed. Estuar. Coast. Shelf Sci. 2019, 226, 106269. [Google Scholar] [CrossRef]
- Wang, Q.; Peng, F.; Chen, Y.; Jin, L.; Lin, J.; Zhao, X.; Yin, J.; Li, J.Y. Heavy metals and PAHs in an open fishing area of the East China Sea: Multimedia distribution, source diagnosis, and dietary risk assessment. Environ. Sci. Pollut. Res. 2019, 26, 21140–21150. [Google Scholar] [CrossRef]
- Sarria-Villa, R.; Ocampo-Duque, W.; Páez, M.; Schuhmacher, M. Presence of PAHs in water and sediments of the Colombian Cauca River during heavy rain episodes, and implications for risk assessment. Sci. Total Environ. 2016, 540, 455–465. [Google Scholar] [CrossRef]
- Chizhova, T.; Koudryashova, Y.; Prokuda, N.; Tishchenko, P.; Hayakawa, K. Polycyclic Aromatic Hydrocarbons in the Estuaries of Two Rivers of the Sea of Japan. Int. J. Environ. Res. Public Health 2020, 17, 6019. [Google Scholar] [CrossRef]
- Guo, W.; He, M.; Yang, Z.; Lin, C.; Quan, X.; Men, B. Distribution, partitioning and sources of polycyclic aromatic hydrocarbons in Daliao River water system in dry season, China. J. Hazard. Mater. 2009, 164, 1379–1385. [Google Scholar] [CrossRef]
- Hamilton, P.B.; Cowx, I.G.; Oleksiak, M.F.; Griffiths, A.M.; Grahn, M.; Stevens, J.R.; Carvalho, G.R.; Nicol, E.; Tyler, C.R. Population-level consequences for wild fish exposed to sublethal concentrations of chemicals–a critical review. Fish Fish. 2016, 17, 545–566. [Google Scholar] [CrossRef]
- Ito, K.; Uno, S.; Tanaka, H.; Miki, S.; Kokushi, E.; Yamamoto, M.; Koyama, J.; Añasco, N.C. Distribution of parent and alkylated PAHs in bivalves collected from Osaka bay, Japan. Jpn. J. Environ. Toxicol. 2015, 18, 11–24. [Google Scholar] [CrossRef]
- Kannan, K.; Perrotta, E. Polycyclic aromatic hydrocarbons (PAHs) in livers of California sea otters. Chemosphere 2008, 71, 649–655. [Google Scholar] [CrossRef]
- Nakata, H.; Uehara, K.; Goto, Y.; Fukumura, M.; Shimasaki, H.; Takikawa, K.; Miyawaki, T. Polycyclic aromatic hydrocarbons in oysters and sediments from the Yatsushiro Sea, Japan: Comparison of potential risks among PAHs, dioxins and dioxin-like compounds in benthic organisms. Ecotoxicol. Environ. Saf. 2014, 99, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Senthilkumar, K.; Alomirah, H.; Moon, H.B.; Minh, T.B.; Mohd, M.A.; Nakata, H.; Kannan, K. Concentrations and profiles of urinary polycyclic aromatic hydrocarbon metabolites (OH-PAHs) in several Asian countries. Environ. Sci. Technol. 2013, 47, 2932–2938. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.B.; Lee, D.H.; Lee, Y.S.; Kannan, K. Occurrence and accumulation patterns of polycyclic aromatic hydrocarbons and synthetic musk compounds in adipose tissues of Korean females. Chemosphere 2012, 86, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Lawson, M.C.; Cullen, J.A.; Nunnally, C.C.; Rowe, G.T.; Hala, D.N. PAH and PCB body-burdens in epibenthic deep-sea invertebrates from the northern Gulf of Mexico. Mar. Pollut. Bull. 2020, 111825. [Google Scholar] [CrossRef]
- Hylland, K. Polycyclic aromatic hydrocarbon (PAH) ecotoxicology in marine ecosystems. J. Toxicol. Environ. Health Part A 2006, 69, 109–123. [Google Scholar] [CrossRef]
- Noh, J.; Kim, H.; Lee, C.; Yoon, S.J.; Chu, S.; Kwon, B.O.; Ryu, J.; Kim, J.J.; Lee, H.; Yim, U.H.; et al. Bioaccumulation of polycyclic aromatic hydrocarbons (PAHs) by the marine clam, Mactra veneriformis, chronically exposed to oil-suspended particulate matter aggregates. Environ. Sci. Technol. 2018, 52, 7910–7920. [Google Scholar] [CrossRef]
- Bejarano, A.C.; Michel, J. Oil spills and their impacts on sand beach invertebrate communities: A literature review. Environ. Pollut. 2016, 218, 709–722. [Google Scholar] [CrossRef]
- Balcıoğlu, E.B. Potential effects of polycyclic aromatic hydrocarbons (PAHs) in marine foods on human health: A critical review. Toxin Rev. 2016, 35, 98–105. [Google Scholar] [CrossRef]
- Moorthy, B.; Chu, C.; Carlin, D.J. Polycyclic aromatic hydrocarbons: From metabolism to lung cancer. Toxicol. Sci. 2015, 145, 5–15. [Google Scholar] [CrossRef]
- Lemieux, C.L.; Long, A.S.; Lambert, I.B.; Lundstedt, S.; Tysklind, M.; White, P.A. In vitro mammalian mutagenicity of complex polycyclic aromatic hydrocarbon mixtures in contaminated soils. Environ. Sci. Technol. 2015, 49, 1787–1796. [Google Scholar] [CrossRef]
- Yadetie, F.; Zhang, X.; Hanna, E.M.; Aranguren-Abadía, L.; Eide, M.; Blaser, N.; Brun, M.; Jonassen, I.; Goksøyr, A.; Karlsen, O.A. RNA-Seq analysis of transcriptome responses in Atlantic cod (Gadus morhua) precision-cut liver slices exposed to benzo[a]pyrene and 17-ethynylestradiol. Aquat. Toxicol. 2018, 201, 174–186. [Google Scholar] [CrossRef] [PubMed]
- Honda, M.; Suzuki, N. Toxicities of Polycyclic Aromatic Hydrocarbons for Aquatic Animals. Int. J. Environ. Res. Public Health 2020, 17, 1363. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Ogiso, S.; Yachiguchi, K.; Kawabe, K.; Makino, F.; Toriba, A.; Kiyomoto, M.; Sekiguchi, T.; Tabuchi, Y.; Kondo, T.; et al. Monohydroxylated polycyclic aromatic hydrocarbons influence spicule formation in the early development of sea urchins (Hemicentrotus pulcherrimus). Comp. Biochem. Physiol. Part C 2015, 171, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Azizi, G.; Akodad, M.; Baghour, M.; Layachi, M.; Moumen, A. The use of Mytilus spp. mussels as bioindicators of heavy metal pollution in the coastal environment. A review. J. Mater. Environ. Sci. 2018, 9, 1170–1181. [Google Scholar] [CrossRef]
- Hano, T.; Ito, K.; Ohkubo, N.; Sakaji, H.; Watanabe, A.; Takashima, K.; Sato, T.; Sugaya, T.; Matsuki, K.; Onduka, T.; et al. Occurrence of neonicotinoids and fipronil in estuaries and their potential risks to aquatic invertebrates. Environ. Pollut. 2019, 252, 205–215. [Google Scholar] [CrossRef]
- Loganathan, B.G.; Kumar, K.S.; Masunaga, S.; Sajwan, K.S. Polychlorinated dibenzo-p-dioxins, dibenzofurans, and dioxin-like polychlorinated biphenyls in sediment and mussel samples from Kentucky Lake, USA. Arch. Environ. Contam. Toxicol. 2008, 54, 20–30. [Google Scholar] [CrossRef]
- Meng, J.; Hong, S.; Wang, T.; Li, Q.; Yoon, S.J.; Lu, Y.; Giesy, J.P.; Khim, J.S. Traditional and new POPs in environments along the Bohai and Yellow Seas: An overview of China and South Korea. Chemosphere 2017, 169, 503–515. [Google Scholar] [CrossRef]
- Rodríguez-Navas, C.; Björklund, E.; Bak, S.A.; Hansen, M.; Krogh, K.A.; Maya, F.; Forteza, R.; Cerdà, V. Pollution pathways of pharmaceutical residues in the aquatic environment on the island of Mallorca, Spain. Arch. Environ. Contam. Toxicol. 2013, 65, 56–66. [Google Scholar] [CrossRef]
- Xue, X.; Xue, J.; Liu, W.; Adams, D.H.; Kannan, K. Trophic magnification of parabens and their metabolites in a subtropical marine food web. Environ. Sci. Technol. 2017, 51, 780–789. [Google Scholar] [CrossRef]
- Tanaka, H.; Onduka, T. Background levels of PAHs in the coastal waters of Japan based on residual concentrations of bivalves. J. Environ. Chem. 2010, 20, 137–148. [Google Scholar] [CrossRef][Green Version]
- Tsai, M.L.; Dai, C.F. Life history plasticity and reproductive strategy enabling the invasion of Ligia exotica (Crustacea: Isopoda) from the littoral zone to an inland creek. Mar. Ecol. Prog. Ser. 2001, 210, 175–184. [Google Scholar] [CrossRef]
- Hurtado, L.A.; Mateos, M.; Santamaria, C.A. Phylogeography of supralittoral rocky intertidal Ligia isopods in the Pacific region from central California to central Mexico. PLoS ONE 2010, 5, e11633. [Google Scholar] [CrossRef] [PubMed]
- Kang, C.; Choy, E.J.; Son, Y.; Lee, J.; Kim, J.K.; Kim, Y.; Lee, K. Food web structure of a restored macroalgal bed in the eastern Korean peninsula determined by C and N stable isotope analyses. Mar. Biol. 2008, 153, 1181–1198. [Google Scholar] [CrossRef]
- Undap, S.L.; Matsunaga, S.; Honda, M.; Sekiguchi, T.; Suzuki, N.; Khalil, F.; Qiu, X.; Shimasaki, Y.; Ando, H.; Sato-Okoshi, W.; et al. Accumulation of organotins in wharf roach (Ligia exotica Roux) and its ability to serve as a biomonitoring species for coastal pollution. Ecotoxicol. Environ. Saf. 2013, 96, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Qiu, X.; Undap, S.L.; Honda, M.; Sekiguchi, T.; Suzuki, N.; Shimasaki, Y.; Ando, H.; Sato-Okoshi, W.; Wada, T.; Sunobe, T.; et al. Pollution of radiocesium and radiosilver in wharf roach (Ligia sp.) by the Fukushima Dai-ichi Nuclear Power Plant accident. J. Radioanal. Nucl. Chem. 2016, 311, 121–126. [Google Scholar] [CrossRef]
- Honda, M.; Qiu, X.; Koyama, J.; Uno, S.; Undap, S.L.; Shimasaki, Y.; Oshima, Y. The wharf roach, Ligia sp., A novel indicator of polycyclic aromatic hydrocarbon contamination in coastal areas. Int. J. Environ. Res. 2018, 12, 1–11. [Google Scholar] [CrossRef]
- Niu, L.; Cai, H.; Van Gelder, P.H.; Luo, P.; Liu, F.; Yang, Q. Dynamics of polycyclic aromatic hydrocarbons (PAHs) in water column of Pearl River estuary (China): Seasonal pattern, environmental fate and source implication. Appl. Geochem. 2018, 90, 39–49. [Google Scholar] [CrossRef]
- Thomann, R.V.; Komlos, J. Model of biota-sediment accumulation factor for polycyclic aromatic hydrocarbons. Environ. Toxicol. Chem. Int. J. 1999, 18, 1060–1068. [Google Scholar] [CrossRef]
- Hayakawa, K.; Makino, F.; Yasuma, M.; Yoshida, S.; Chondo, Y.; Toriba, A.; Kameda, T.; Tang, N.; Kunugi, M.; Nakase, H.; et al. Polycyclic aromatic hydrocarbons in surface water of the Southeastern Japan Sea. Chem. Pharm. Bull. 2016, 64, 625–631. [Google Scholar] [CrossRef]
- Koudryashova, Y.; Chizhova, T.; Tishchenko, P.; Hayakawa, K. Seasonal Variability of Polycyclic Aromatic Hydrocarbons (PAHs) in a Coastal Marine Area in the Northwestern Region of the Sea of Japan/East Sea (Possiet Bay). Ocean Sci. J. 2019, 54, 635–655. [Google Scholar] [CrossRef]
- Zhang, H.; Sun, L.; Sun, T.; Li, H.; Luo, Q. Spatial distribution and seasonal variation of polycyclic aromatic hydrocarbons (PAHs) contaminations in surface water from the Hun River, Northeast China. Environ. Monit. Assess. 2013, 185, 1451–1462. [Google Scholar] [CrossRef]
- Xiao, R.; Bai, J.; Wang, J.; Lu, Q.; Zhao, Q.; Cui, B.; Liu, X. Polycyclic aromatic hydrocarbons (PAHs) in wetland soils under different land uses in a coastal estuary: Toxic levels, sources and relationships with soil organic matter and water-stable aggregates. Chemosphere 2014, 110, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Pavoni, B.; Caliceti, M.; Sperni, L.; Sfriso, A. Organic micropollutants (PAHs, PCBs, pesticides) in seaweeds of the lagoon of Venice. Oceanol. Acta 2003, 26, 585–596. [Google Scholar] [CrossRef]
- Jiang, Y.; Lin, T.; Wu, Z.; Li, Y.; Li, Z.; Guo, Z.; Yao, X. Seasonal atmospheric deposition and air–sea gas exchange of polycyclic aromatic hydrocarbons over the Yangtze River Estuary, East China Sea: Implications for source–sink processes. Atmos. Environ. 2018, 178, 31–40. [Google Scholar] [CrossRef]
- Inam, E.; Offiong, N.A.; Essien, J.; Kang, S.; Kang, S.Y.; Antia, B. Polycyclic aromatic hydrocarbons loads and potential risks in freshwater ecosystem of the Ikpa River Basin, Niger Delta—Nigeria. Environ. Monit. Assess. 2016, 188, 49. [Google Scholar] [CrossRef] [PubMed]
- Francioni, E.; Wagener, A.L.R.; Scofield, A.L.; Depledge, M.H.; Cavalier, B. Evaluation of the mussel Perna perna as a biomonitor of polycyclic aromatic hydrocarbon (PAH) exposure and effects. Mar. Pollut. Bull. 2007, 54, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Soclo, H.H.; Garrigues, P.; Ewald, M. Origin of polycyclic aromatic hydrocarbons (PAHs) in coastal marine sediments: Case studies in Cotonou (Benin) and Aquitaine (France) areas. Mar. Pollut. Bull. 2000, 40, 387–396. [Google Scholar] [CrossRef]
- Horiguchi, H.; Hironaka, M.; Meyer-Rochow, V.B.; Hariyama, T. Water uptake via two pairs of specialized legs in Ligia exotica (Crustacea, Isopoda). Biol. Bull. 2007, 213, 196–203. [Google Scholar] [CrossRef]
- Meador, J.P.; Casillas, E.; Sloan, C.A.; Varanasi, U. Comparative bioaccumulation of polycyclic aromatic hydrocarbons from sediment by two infaunal invertebrates. Mar. Ecol. Prog. Ser. 1995, 123, 107–124. [Google Scholar] [CrossRef]
- Hickey, C.W.; Roper, D.S.; Holland, P.T.; Trower, T.M. Accumulation of organic contaminants in two sediment-dwelling shellfish with contrasting feeding modes: Deposit-(Macomona liliana) and filter-feeding (Austrovenus stutchburyi). Arch. Environ. Contam. Toxicol. 1995, 29, 221–231. [Google Scholar] [CrossRef]
- Van Brummelen, T.C.; Verweij, R.A.; Wedzinga, S.A.; Van Gestel, C.A. Polycyclic aromatic hydrocarbons in earthworms and isopods from contaminated forest soils. Chemosphere 1996, 32, 315–341. [Google Scholar] [CrossRef]
- Van Gestel, C.A.; Loureiro, S. Terrestrial isopods as model organisms in soil ecotoxicology: A review. ZooKeys 2018, 801, 127–162. [Google Scholar] [CrossRef] [PubMed]
- Haritash, A.K.; Kaushik, C.P. Biodegradation aspects of polycyclic aromatic hydrocarbons (PAHs): A review. J. Hazard. Mater. 2009, 169, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Tse, M.F.; Young, L.; Wong, M.H. Distribution patterns of polycyclic aromatic hydrocarbons (PAHs) in the sediments and fish at Mai Po Marshes Nature Reserve, Hong Kong. Water Res. 2007, 41, 1303–1311. [Google Scholar] [CrossRef]
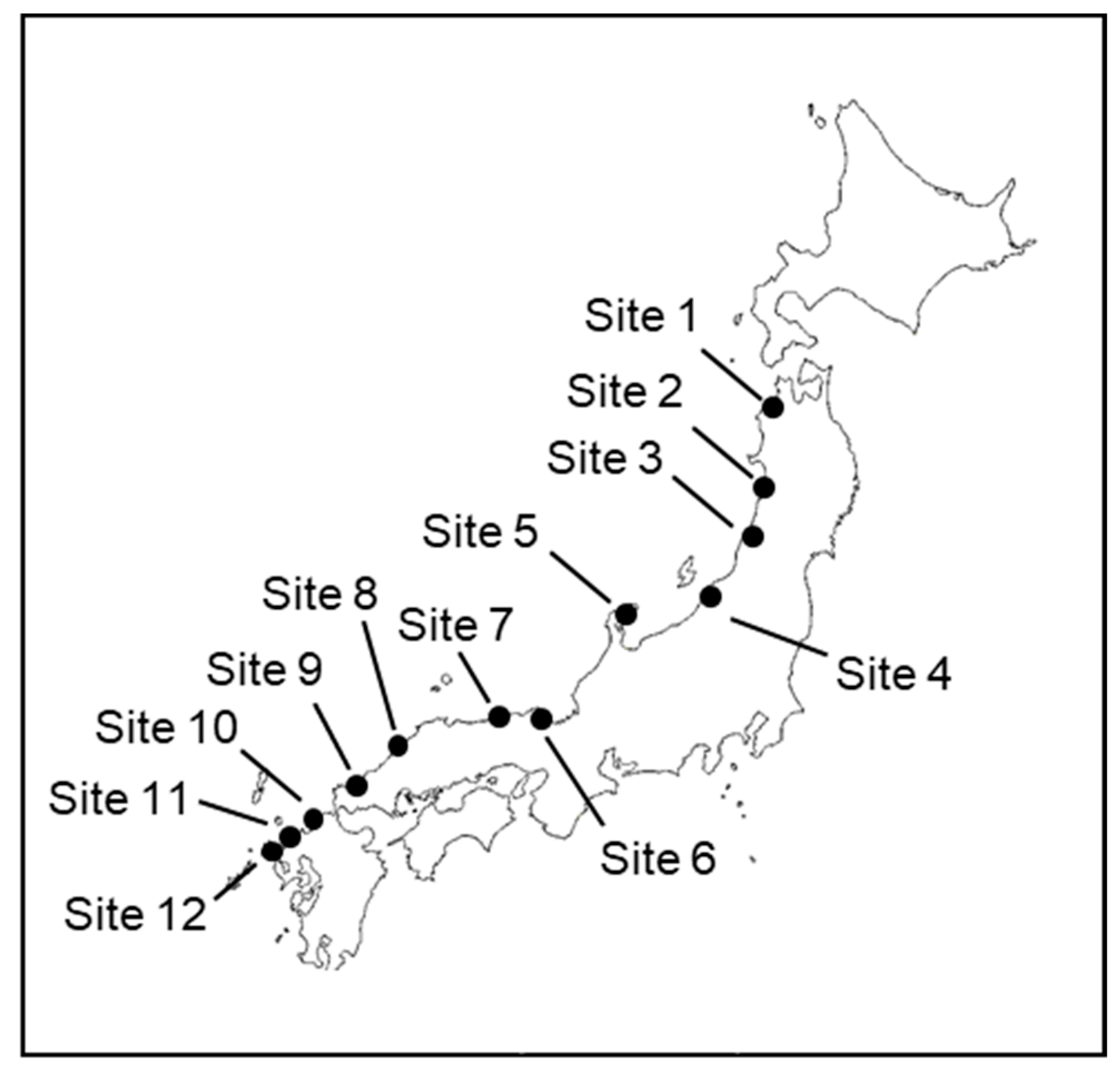

| Site No. | City, Prefecture | GPS Coordinate | Sampling Date |
|---|---|---|---|
| 1 | Ajigasawa, Aomori | 40.779312, 140.215660 | 12 September 2018 |
| 2 | Yurihonjo, Akita | 39.501981, 140.045146 | 13 September 2018 |
| 3 | Tsuruoka, Yamagata | 38.722078, 139.685163 | 13 September 2018 |
| 4 | Nagaoka, Niigata | 37.646270, 138.764767 | 14 September 2018 |
| 5 | Suzu, Ishikawa | 37.307115, 137.231492 | 28 September 2018 |
| 6 | Miyazu, Kyoto | 35.558870, 135.186588 | 15 September 2018 |
| 7 | Kami, Hyogo | 35.652180, 134.605145 | 16 September 2018 |
| 8 | Hamada, Shimane | 34.904503, 132.060741 | 17 September 2018 |
| 9 | Hagi, Yamaguchi | 34.437040, 131.416821 | 17 September 2018 |
| 10 | Fukuoka, Fukuoka | 33.603976, 130.274923 | 18 September 2018 |
| 11 | Karatsu, Saga | 33.484003, 129.944348 | 19 September 2018 |
| 12 | Hirado, Nagasaki | 33.361646, 129.626395 | 19 September 2018 |
| Prefecture | Wharf Roach (ng/g-dw) | Mussel (ng/g-dw) | Soil/Sand (ng/g-dw) | Drifting Seaweed (ng/g-dw) | Sea Water (ng/L) |
|---|---|---|---|---|---|
| Aomori | 49.2 | 65.1 | 23.8 | 34.4 | 12.4 |
| Akita | 96.0 | 44.5 | 9.95 | 12.4 | 19.1 |
| Yamagata | 53.0 | 30.1 | 37.6 | 31.0 | 7.79 |
| Niigata | 85.2 | 33.4 | 20.2 | 11.8 | 9.90 |
| Ishikawa | 47.9 | 132 | 506 | 82.4 | 12.8 |
| Kyoto | 42.1 | 43.2 | 138 | 89.5 | 15.6 |
| Hyogo | 44.1 | 53.3 | 58.1 | 50.6 | 16.4 |
| Shimane | 44.1 | 35.4 | 47.5 | na | 10.0 |
| Yamaguchi | 26.9 | 29.2 | 2.06 | 21.3 | 10.2 |
| Fukuoka | 52.8 | 41.1 | 3.45 | 62.9 | 17.8 |
| Saga | 85.0 | 16.6 | 3.85 | 18.7 | 11.9 |
| Nagasaki | 37.7 | 38.6 | 11.1 | 109 | 16.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Honda, M.; Mukai, K.; Nagato, E.; Uno, S.; Oshima, Y. Correlation between Polycyclic Aromatic Hydrocarbons in Wharf Roach (Ligia spp.) and Environmental Components of the Intertidal and Supralittoral Zone along the Japanese Coast. Int. J. Environ. Res. Public Health 2021, 18, 630. https://doi.org/10.3390/ijerph18020630
Honda M, Mukai K, Nagato E, Uno S, Oshima Y. Correlation between Polycyclic Aromatic Hydrocarbons in Wharf Roach (Ligia spp.) and Environmental Components of the Intertidal and Supralittoral Zone along the Japanese Coast. International Journal of Environmental Research and Public Health. 2021; 18(2):630. https://doi.org/10.3390/ijerph18020630
Chicago/Turabian StyleHonda, Masato, Koki Mukai, Edward Nagato, Seiichi Uno, and Yuji Oshima. 2021. "Correlation between Polycyclic Aromatic Hydrocarbons in Wharf Roach (Ligia spp.) and Environmental Components of the Intertidal and Supralittoral Zone along the Japanese Coast" International Journal of Environmental Research and Public Health 18, no. 2: 630. https://doi.org/10.3390/ijerph18020630
APA StyleHonda, M., Mukai, K., Nagato, E., Uno, S., & Oshima, Y. (2021). Correlation between Polycyclic Aromatic Hydrocarbons in Wharf Roach (Ligia spp.) and Environmental Components of the Intertidal and Supralittoral Zone along the Japanese Coast. International Journal of Environmental Research and Public Health, 18(2), 630. https://doi.org/10.3390/ijerph18020630






