Human–Animal Interaction and Perinatal Mental Health: A Narrative Review of Selected Literature and Call for Research
Abstract
1. Introduction
Overview of Current Paper
2. Perinatal Mental Health
The Role of Psychoneuroimmunological Biomarkers in Maternal/Child Health
Mother–Child Dyad
3. Social Relationships, the Stress Response, and Perinatal Mental Health
Potential Benefits of HAI during the Perinatal Period
Biobehavioral Underpinnings of HAI
4. HAI and Potential Risks to Perinatal Mental Health
5. Discussion
5.1. Key Concepts from the Comprehensive Model of Mental Health during the Perinatal Period
5.2. Key Concepts from the HAI–HPA Transactional Model
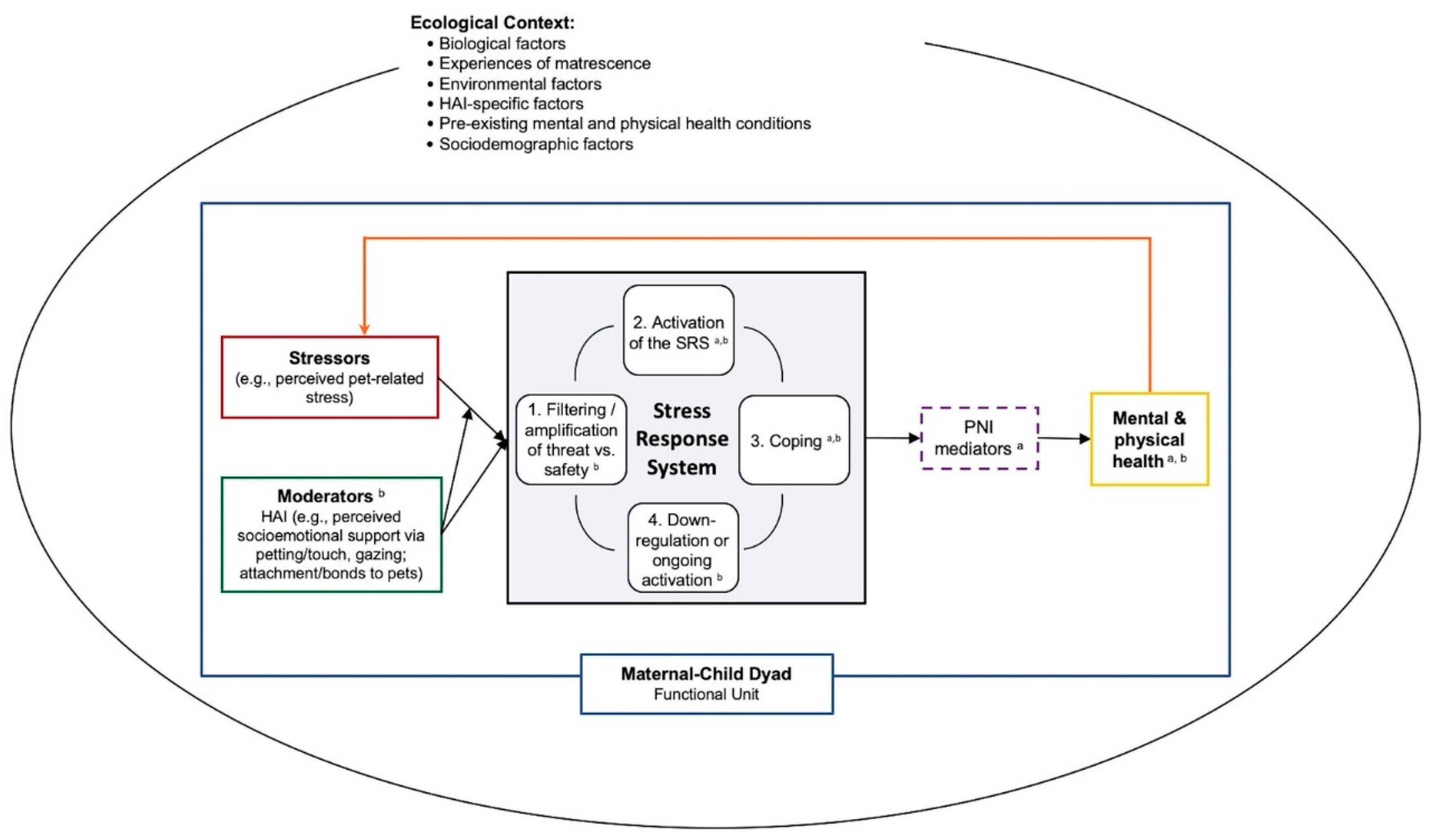
5.3. Integrating Models to Guide Research on the Intersection of HAI and Perinatal Health
5.3.1. HAI and the Stress Response System
5.3.2. HAI and Matrescence
5.3.3. HAI and the Mother–Child Dyad
HAI and Other Caregivers
5.3.4. HAI within the Ecological Context
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rodriguez, K.E.; Herzog, H.; Gee, N.R. Variability in Human-Animal Interaction Research. Front. Vet. Sci. 2021, 7, 619600. [Google Scholar] [CrossRef]
- Esposito, L.; McCune, S.; Griffin, J.A.; Maholmes, V. Directions in Human-Animal Interaction Research: Child Development, Health, and Therapeutic Interventions. Child Dev. Perspect. 2011, 5, 205–211. [Google Scholar] [CrossRef]
- Purewal, R.; Christley, R.; Kordas, K.; Joinson, C.; Meints, K.; Gee, N.; Westgarth, C. Companion Animals and Child/Adolescent Development: A Systematic Review of the Evidence. Int. J. Environ. Res. Public. Health 2017, 14, 234. [Google Scholar] [CrossRef]
- Tomlinson, C.A.; Matijczak, A.; McDonald, S.E.; Gee, N.R. The role of human-animal interaction in child and adolescent development. In The Encyclopedia of Child and Adolescent Health; Halpern-Fisher, B., Ed.; Elsevier: Amsterdam, The Netherlands, 2021. [Google Scholar]
- American Veterinary Medical Association Statement from the Committee on the Human-Animal Bond. J. Am. Vet. Med. Assoc. 1998, 212, 1675.
- Laing, M.; Maylea, C. “They Burn Brightly, But Only for a Short Time”: The Role of Social Workers in Companion Animal Grief and Loss. Anthrozoös 2018, 31, 221–232. [Google Scholar] [CrossRef]
- Risley-Curtiss, C. Social Work Practitioners and the Human-Companion Animal Bond: A National Study. Soc. Work 2010, 55, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Morley, C.; Fook, J. The Importance of Pet Loss and Some Implications for Services. Mortality 2005, 10, 127–143. [Google Scholar] [CrossRef]
- McCune, S.; McCardle, P.; Griffin, J.A.; Esposito, L.; Hurley, K.; Bures, R.; Kruger, K.A. Editorial: Human-Animal Interaction (HAI) Research: A Decade of Progress. Front. Vet. Sci. 2020, 7, 44. [Google Scholar] [CrossRef]
- Meints, K.; Brelsford, V.; Dimolareva, M.; Gee, N. Dog-Assisted Interventions with Children in Mainstream and Special Educational Needs Schools—What Works? In Proceedings of the International Society for Anthrozoology Annual Conference, Liverpool, UK, 5 September 2020.
- Rew, L. Friends and Pets as Companions: Strategies for Coping with Loneliness among Homeless Youth. J. Child Adolesc. Psychiatr. Nurs. 2000, 13, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Rhoades, H.; Winetrobe, H.; Rice, E. Pet Ownership among Homeless Youth: Associations with Mental Health, Service Utilization and Housing Status. Child Psychiatry Hum. Dev. 2015, 46, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Triebenbacher, S.L. The relationship between attachment to companion animals and self-esteem: A developmental perspective. In Companion Animals in Human Health; Wilson, C.C., Turner, D.C., Eds.; SAGE Publications, Inc.: Thousand Oaks, CA, USA, 1998; pp. 135–148. [Google Scholar]
- Mathers, M.; Canterford, L.; Olds, T.; Waters, E.; Wake, M. Pet Ownership and Adolescent Health: Cross-Sectional Population Study. J. Paediatr. Child Health 2010, 46, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Matijczak, A.; McDonald, S.E.; O’Connor, K.E.; George, N.; Tomlinson, C.A.; Murphy, J.L.; Ascione, F.R.; Williams, J.H. Do Animal Cruelty Exposure and Positive Engagement with Pets Moderate Associations between Children’s Exposure to Intimate Partner Violence and Externalizing Behavior Problems? Child Adolesc. Soc. Work J. 2020, 37, 601–613. [Google Scholar] [CrossRef]
- Hartwig, E.; Signal, T. Attachment to Companion Animals and Loneliness in Australian Adolescents. Aust. J. Psychol. 2020, 72, 337–346. [Google Scholar] [CrossRef]
- Paul, E.S.; Serpell, J.A. Obtaining a New Pet Dog: Effects on Middle Childhood Children and Their Families. Appl. Anim. Behav. Sci. 1996, 47, 17–29. [Google Scholar] [CrossRef]
- Crawford, K.M.; Zhu, Y.; Davis, K.A.; Ernst, S.; Jacobsson, K.; Nishimi, K.; Smith, A.D.A.C.; Dunn, E.C. The Mental Health Effects of Pet Death during Childhood: Is It Better to Have Loved and Lost than Never to Have Loved at All? Eur. Child Adolesc. Psychiatry 2020. [Google Scholar] [CrossRef]
- Barker, S.B.; Schubert, C.M.; Barker, R.T.; Kuo, S.I.C.; Kendler, K.S.; Dick, D.M. The Relationship between Pet Ownership, Social Support, and Internalizing Symptoms in Students from the First to Fourth Year of College. Appl. Dev. Sci. 2020, 24, 279–293. [Google Scholar] [CrossRef]
- Matijczak, A.; McDonald, S.E.; Tomlinson, C.A.; Murphy, J.L.; O’Connor, K. The Moderating Effect of Comfort from Companion Animals and Social Support on the Relationship between Microaggressions and Mental Health in LGBTQ+ Emerging Adults. Behav. Sci. 2021, 11, 1. [Google Scholar] [CrossRef]
- Carlisle, G.K.; Johnson, R.A.; Wang, Z.; Brosi, T.C.; Rife, E.M.; Hutchison, A. Exploring Human–Companion Animal Interaction in Families of Children with Autism. J. Autism Dev. Disord. 2020, 50, 2793–2805. [Google Scholar] [CrossRef]
- Fecteau, S.-M.; Boivin, L.; Trudel, M.; Corbett, B.A.; Harrell, F.E.; Viau, R.; Champagne, N.; Picard, F. Parenting Stress and Salivary Cortisol in Parents of Children with Autism Spectrum Disorder: Longitudinal Variations in the Context of a Service Dog’s Presence in the Family. Biol. Psychol. 2017, 123, 187–195. [Google Scholar] [CrossRef]
- Hall, S.S.; Wright, H.F.; Hames, A.; Mills, D.S.; Team, P. The Long-Term Benefits of Dog Ownership in Families with Children with Autism. J. Vet. Behav. 2016, 13, 46–54. [Google Scholar] [CrossRef]
- Hall, S.S.; Gee, N.R.; Mills, D.S. Reading to Dogs: A Systematic Review of the Literature. PLoS ONE 2016, 11, e0149759. [Google Scholar] [CrossRef]
- McCullough, A.; Ruehrdanz, A.; Jenkins, M.A.; Gilmer, M.J.; Olson, J.; Pawar, A.; Holley, L.; Sierra-Rivera, S.; Linder, D.E.; Pichette, D.; et al. Measuring the Effects of an Animal-Assisted Intervention for Pediatric Oncology Patients and Their Parents: A Multisite Randomized Controlled Trial. J. Pediatr. Oncol. Nurs. 2018, 35, 159–177. [Google Scholar] [CrossRef] [PubMed]
- Wright, H.F.; Hall, S.; Hames, A.; Hardiman, J.; Mills, R.; PAWS Team; Mills, D.S. Acquiring a Pet Dog Significantly Reduces Stress of Primary Carers for Children with Autism Spectrum Disorder: A Prospective Case Control Study. J. Autism Dev. Disord. 2015, 45, 2531–2540. [Google Scholar] [CrossRef] [PubMed]
- Applebaum, J.W.; MacLean, E.L.; McDonald, S.E. Love, Fear, and the Human-Animal Bond: On Adversity and Multispecies Relationships. Compr. Psychoneuroendocrinol. 2021, 7, 100071. [Google Scholar] [CrossRef] [PubMed]
- Applebaum, J.W.; Tomlinson, C.A.; Matijczak, A.; McDonald, S.E.; Zsembik, B.A. The Concerns, Difficulties, and Stressors of Caring for Pets during COVID-19: Results from a Large Survey of U.S. Pet Owners. Animals 2020, 10, 1882. [Google Scholar] [CrossRef] [PubMed]
- McDonald, S.E.; O’Connor, K.; Matijczak, A.; Murphy, J.; Applebaum, J.W.; Tomlinson, C.A.; Wike, T.L.; Kattari, S.K. Victimization and Psychological Wellbeing among Sexual and Gender Minority Emerging Adults: Testing the Moderating Role of Emotional Comfort from Companion Animals. J. Soc. Soc. Work Res. 2021, in press. [Google Scholar] [CrossRef]
- Mueller, M.K.; King, E.K.; Callina, K.; Dowling-Guyer, S.; McCobb, E. Demographic and Contextual Factors as Moderators of the Relationship between Pet Ownership and Health. Health Psychol. Behav. Med. 2021, 9, 701–723. [Google Scholar] [CrossRef]
- Tomlinson, C.A.; Murphy, J.L.; Williams, J.M.; Hawkins, R.D.; Matijczak, A.; Applebaum, J.W.; McDonald, S.E. Testing the Moderating Role of Victimization and Microaggressions on the Relationship between Human-Animal Interaction and Psychological Adjustment among LGBTQ+ Emerging Adults. Hum. Anim. Interact. Bull. 2021, in press. [Google Scholar]
- Adane, A.A.; Bailey, H.D.; Morgan, V.A.; Galbally, M.; Farrant, B.M.; Marriott, R.; White, S.W.; Shepherd, C.C.J. The Impact of Maternal Prenatal Mental Health Disorders on Stillbirth and Infant Mortality: A Systematic Review and Meta-Analysis. Arch. Womens Ment. Health 2021, 24, 543–555. [Google Scholar] [CrossRef]
- Hazell Raine, K.; Nath, S.; Howard, L.M.; Cockshaw, W.; Boyce, P.; Sawyer, E.; Thorpe, K. Associations between Prenatal Maternal Mental Health Indices and Mother–Infant Relationship Quality 6 to 18 Months’ Postpartum: A Systematic Review. Infant Ment. Health J. 2020, 41, 24–39. [Google Scholar] [CrossRef]
- Kingston, D.; Tough, S.; Whitfield, H. Prenatal and Postpartum Maternal Psychological Distress and Infant Development: A Systematic Review. Child Psychiatry Hum. Dev. 2012, 43, 683–714. [Google Scholar] [CrossRef]
- Kinser, P.A.; Thacker, L.R.; Lapato, D.; Wagner, S.; Roberson-Nay, R.; Jobe-Shields, L.; Amstadter, A.; York, T.P. Depressive Symptom Prevalence and Predictors in the First Half of Pregnancy. J. Womens Health 2018, 27, 369–376. [Google Scholar] [CrossRef]
- Shonkoff, J.P.; Boyce, W.T.; Levitt, P.; Martinez, F.D.; McEwen, B. Leveraging the Biology of Adversity and Resilience to Transform Pediatric Practice. Pediatrics 2021, 147, e20193845. [Google Scholar] [CrossRef]
- Howard, L.M.; Khalifeh, H. Perinatal Mental Health: A Review of Progress and Challenges. World Psychiatry 2020, 19, 313–327. [Google Scholar] [CrossRef]
- Kinser, P.A.; Lyon, D.E. A Conceptual Framework of Stress Vulnerability, Depression, and Health Outcomes in Women: Potential Uses in Research on Complementary Therapies for Depression. Brain Behav. 2014, 4, 665–674. [Google Scholar] [CrossRef]
- Luca, D.L.; Margiotta, C.; Staatz, C.; Garlow, E.; Christensen, A.; Zivin, K. Financial Toll of Untreated Perinatal Mood and Anxiety Disorders Among 2017 Births in the United States. Am. J. Public Health 2020, 110, 888–896. [Google Scholar] [CrossRef]
- Leach, L.S.; Poyser, C.; Cooklin, A.R.; Giallo, R. Prevalence and Course of Anxiety Disorders (and Symptom Levels) in Men across the Perinatal Period: A Systematic Review. J. Affect. Disord. 2016, 190, 675–686. [Google Scholar] [CrossRef] [PubMed]
- Shorey, S.; Chan, V. Paternal Mental Health during the Perinatal Period: A Qualitative Systematic Review. J. Adv. Nurs. 2020, 76, 1307–1319. [Google Scholar] [CrossRef] [PubMed]
- McKee, K.; Admon, L.K.; Winkelman, T.N.A.; Muzik, M.; Hall, S.; Dalton, V.K.; Zivin, K. Perinatal Mood and Anxiety Disorders, Serious Mental Illness, and Delivery-Related Health Outcomes, United States, 2006. BMC Womens Health 2020, 20, 150. [Google Scholar] [CrossRef]
- Zeng, L.-N.; Chen, L.-G.; Yang, C.-M.; Zeng, L.-P.; Zhang, L.-Y.; Peng, T.-M. Mental Health Care for Pregnant Women in the COVID-19 Outbreak Is Urgently Needed. Women Birth 2021, 34, 210–211. [Google Scholar] [CrossRef] [PubMed]
- Kinser, P.A.; Jallo, N.; Amstadter, A.; Thacker, L.; Jones, E.; Moyer, S.; Rider, A.M.; Karjane, N.; Salisbury, A. Depression, Anxiety, Resilience, and Coping: The Experience of Pregnant and New Mothers during the First Few Months of the COVID-19 Pandemic. J. Womens Health 2021, 30, 654–664. [Google Scholar] [CrossRef]
- Lau, Y.; Wong, D.F.K.; Wang, Y.; Kwong, D.H.K.; Wang, Y. The Roles of Social Support in Helping Chinese Women with Antenatal Depressive and Anxiety Symptoms Cope With Perceived Stress. Arch. Psychiatr. Nurs. 2014, 28, 305–313. [Google Scholar] [CrossRef]
- Roos, A.; Faure, S.; Lochner, C.; Vythilingum, B.; Stein, D.J. Predictors of Distress and Anxiety during Pregnancy. Afr. J. Psychiatry 2013, 16, 118–122. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cox, T. Stress, Coping and Problem Solving. Work Stress 1987, 1, 5–14. [Google Scholar] [CrossRef]
- Fink, G. Stress, Definitions, Mechanisms, and Effects Outlined. In Stress: Concepts, Cognition, Emotion, and Behavior; Elsevier: Amsterdam, The Netherlands, 2016; pp. 3–11. [Google Scholar]
- Lazarus, R.S. Psychological Stress and the Coping Process; McGraw-Hill: New York, NY, USA, 1966. [Google Scholar]
- Lazarus, R.S.; Folkman, S. Stress, Appraisal, and Coping; Springer: New York, NY, USA, 1984. [Google Scholar]
- Chovatiya, R.; Medzhitov, R. Stress, Inflammation, and Defense of Homeostasis. Mol. Cell 2014, 54, 281–288. [Google Scholar] [CrossRef]
- Monaghan, P.; Spencer, K.A. Stress and Life History. Curr. Biol. 2014, 24, R408–R412. [Google Scholar] [CrossRef]
- Field, T.; Diego, M.; Hernandez-Reif, M.; Deeds, O.; Holder, V.; Schanberg, S.; Kuhn, C. Depressed Pregnant Black Women Have a Greater Incidence of Prematurity and Low Birthweight Outcomes. Infant Behav. Dev. 2009, 32, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Gavin, A.R.; Melville, J.L.; Rue, T.; Guo, Y.; Dina, K.T.; Katon, W.J. Racial Differences in the Prevalence of Antenatal Depression. Gen. Hosp. Psychiatry 2011, 33, 87–93. [Google Scholar] [CrossRef]
- Kim, H.G.; Kuendig, J.; Prasad, K.; Sexter, A. Exposure to Racism and Other Adverse Childhood Experiences Among Perinatal Women with Moderate to Severe Mental Illness. Community Ment. Health J. 2020, 56, 867–874. [Google Scholar] [CrossRef]
- Scott, K.A.; Britton, L.; McLemore, M.R. The Ethics of Perinatal Care for Black Women: Dismantling the Structural Racism in “Mother Blame” Narratives. J. Perinat. Neonatal Nurs. 2019, 33, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Miyake, Y.; Tanaka, K.; Sasaki, S.; Hirota, Y. Employment, Income, and Education and Risk of Postpartum Depression: The Osaka Maternal and Child Health Study. J. Affect. Disord. 2011, 130, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Tuten, M. Comparing Homeless and Domiciled Pregnant Substance Dependent Women on Psychosocial Characteristics and Treatment Outcomes. Drug Alcohol Depend. 2003, 69, 95–99. [Google Scholar] [CrossRef]
- Yamamoto, N.; Abe, Y.; Arima, K.; Nishimura, T.; Akahoshi, E.; Oishi, K.; Aoyagi, K. Mental Health Problems and Influencing Factors in Japanese Women 4 Months after Delivery. J. Physiol. Anthropol. 2014, 33, 32. [Google Scholar] [CrossRef] [PubMed]
- Herbell, K.; Zauszniewski, J.A. Stress Experiences and Mental Health of Pregnant Women: The Mediating Role of Social Support. Issues Ment. Health Nurs. 2019, 40, 613–620. [Google Scholar] [CrossRef]
- Aktan, N.M. Social Support and Anxiety in Pregnant and Postpartum Women: A Secondary Analysis. Clin. Nurs. Res. 2012, 21, 183–194. [Google Scholar] [CrossRef]
- Corrigan, C.P.; Kwasky, A.N.; Groh, C.J. Social Support, Postpartum Depression, and Professional Assistance: A Survey of Mothers in the Midwestern United States. J. Perinat. Educ. 2015, 24, 48–60. [Google Scholar] [CrossRef]
- Negron, R.; Martin, A.; Almog, M.; Balbierz, A.; Howell, E.A. Social Support During the Postpartum Period: Mothers’ Views on Needs, Expectations, and Mobilization of Support. Matern. Child Health J. 2013, 17, 616–623. [Google Scholar] [CrossRef]
- Islam, J.; Broidy, L.; Baird, K.; Mazerolle, P. Intimate Partner Violence around the Time of Pregnancy and Postpartum Depression: The Experience of Women of Bangladesh. PLoS ONE 2017, 12, e0176211. [Google Scholar] [CrossRef]
- Khajehei, M.; Doherty, M. Women’s Experience of Their Sexual Function during Pregnancy and after Childbirth: A Qualitative Survey. Br. J. Midwifery 2018, 26, 318–328. [Google Scholar] [CrossRef]
- Zhang, Y.; Zou, S.; Cao, Y.; Zhang, Y. Relationship between Domestic Violence and Postnatal Depression among Pregnant Chinese Women. Int. J. Gynecol. Obstet. 2012, 116, 26–30. [Google Scholar] [CrossRef]
- Huschke, S.; Murphy-Tighe, S.; Barry, M. Perinatal Mental Health in Ireland: A Scoping Review. Midwifery 2020, 89, 102763. [Google Scholar] [CrossRef] [PubMed]
- Johnstone, S.J.; Boyce, P.M.; Hickey, A.R.; Morris-Yates, A.D.; Harris, M.G. Obstetric Risk Factors for Postnatal Depression in Urban and Rural Community Samples. Aust. N. Z. J. Psychiatry 2001, 35, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Mezey, G.; Bacchus, L.; Bewley, S.; White, S. Domestic Violence, Lifetime Trauma and Psychological Health of Childbearing Women. BJOG Int. J. Obstet. Gynaecol. 2005, 112, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Bauer, G.R. Incorporating Intersectionality Theory into Population Health Research Methodology: Challenges and the Potential to Advance Health Equity. Soc. Sci. Med. 2014, 110, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Norhayati, M.N.; Nik Hazlina, N.H.; Asrenee, A.R.; Wan Emilin, W.M.A. Magnitude and Risk Factors for Postpartum Symptoms: A Literature Review. J. Affect. Disord. 2015, 175, 34–52. [Google Scholar] [CrossRef]
- Howell, E.A.; Mora, P.; Leventhal, H. Correlates of Early Postpartum Depressive Symptoms. Matern. Child Health J. 2006, 10, 149–157. [Google Scholar] [CrossRef]
- Pooler, J.; Perry, D.F.; Ghandour, R.M. Prevalence and Risk Factors for Postpartum Depressive Symptoms Among Women Enrolled in WIC. Matern. Child Health J. 2013, 17, 1969–1980. [Google Scholar] [CrossRef]
- Lara-Cinisomo, S.; Clark, C.T.; Wood, J. Increasing Diagnosis and Treatment of Perinatal Depression in Latinas and African American Women: Addressing Stigma Is Not Enough. Womens Health Issues 2018, 28, 201–204. [Google Scholar] [CrossRef]
- Bodnar-Deren, S.; Benn, E.K.T.; Balbierz, A.; Howell, E.A. Stigma and Postpartum Depression Treatment Acceptability Among Black and White Women in the First Six-Months Postpartum. Matern. Child Health J. 2017, 21, 1457–1468. [Google Scholar] [CrossRef]
- Corwin, E.J.; Ferranti, E.P. Integration of Biomarkers to Advance Precision Nursing Interventions for Family Research across the Life Span. Nurs. Outlook 2016, 64, 292–298. [Google Scholar] [CrossRef]
- Berlin, L.J.; Martoccio, T.L.; Bryce, C.I.; Jones Harden, B. Improving Infants’ Stress-Induced Cortisol Regulation through Attachment-Based Intervention: A Randomized Controlled Trial. Psychoneuroendocrinology 2019, 103, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Hagenaars, M.A.; Oitzl, M.; Roelofs, K. Updating Freeze: Aligning Animal and Human Research. Neurosci. Biobehav. Rev. 2014, 47, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Corwin, E.J.; Guo, Y.; Pajer, K.; Lowe, N.; McCarthy, D.; Schmiege, S.; Weber, M.; Pace, T.; Stafford, B. Immune Dysregulation and Glucocorticoid Resistance in Minority and Low Income Pregnant Women. Psychoneuroendocrinology 2013, 38, 1786–1796. [Google Scholar] [CrossRef]
- Corwin, E.J.; Pajer, K.; Paul, S.; Lowe, N.; Weber, M.; McCarthy, D.O. Bidirectional Psychoneuroimmune Interactions in the Early Postpartum Period Influence Risk of Postpartum Depression. Brain Behav. Immun. 2015, 49, 86–93. [Google Scholar] [CrossRef]
- Gunnar, M.R.; Talge, N.M.; Herrera, A. Stressor Paradigms in Developmental Studies: What Does and Does Not Work to Produce Mean Increases in Salivary Cortisol. Psychoneuroendocrinology 2009, 34, 953–967. [Google Scholar] [CrossRef]
- Porges, S.W. The Polyvagal Theory: Neurophysiological Foundations of Emotions, Attachment, Communication and Self-Regulation; Norton Series on Interpersonal Neurobiology; Norton: New York, NY, USA, 2011. [Google Scholar]
- Seth, S.; Lewis, A.J.; Galbally, M. Perinatal Maternal Depression and Cortisol Function in Pregnancy and the Postpartum Period: A Systematic Literature Review. BMC Pregnancy Childbirth 2016, 16, 124. [Google Scholar] [CrossRef] [PubMed]
- Cox, E.Q.; Stuebe, A.; Pearson, B.; Grewen, K.; Rubinow, D.; Meltzer-Brody, S. Oxytocin and HPA Stress Axis Reactivity in Postpartum Women. Psychoneuroendocrinology 2015, 55, 164–172. [Google Scholar] [CrossRef]
- Leff-Gelman, P.; Mancilla-Herrera, I.; Flores-Ramos, M.; Cruz-Fuentes, C.; Reyes-Grajeda, J.P.; del Pilar García-Cuétara, M.; Bugnot-Pérez, M.D.; Pulido-Ascencio, D.E. The Immune System and the Role of Inflammation in Perinatal Depression. Neurosci. Bull. 2016, 32, 398–420. [Google Scholar] [CrossRef]
- Stuebe, A.M.; Grewen, K.; Meltzer-Brody, S. Association Between Maternal Mood and Oxytocin Response to Breastfeeding. J. Womens Health 2013, 22, 352–361. [Google Scholar] [CrossRef]
- Eapen, V.; Dadds, M.; Barnett, B.; Kohlhoff, J.; Khan, F.; Radom, N.; Silove, D.M. Separation Anxiety, Attachment and Inter-Personal Representations: Disentangling the Role of Oxytocin in the Perinatal Period. PLoS ONE 2014, 9, e107745. [Google Scholar] [CrossRef]
- Kohlhoff, J.; Eapen, V.; Dadds, M.; Khan, F.; Silove, D.; Barnett, B. Oxytocin in the Postnatal Period: Associations with Attachment and Maternal Caregiving. Compr. Psychiatry 2017, 76, 56–68. [Google Scholar] [CrossRef] [PubMed]
- Levine, A.; Zagoory-Sharon, O.; Feldman, R.; Weller, A. Oxytocin during Pregnancy and Early Postpartum: Individual Patterns and Maternal–Fetal Attachment. Peptides 2007, 28, 1162–1169. [Google Scholar] [CrossRef]
- Uvnäs Moberg, K.; Ekström-Bergström, A.; Buckley, S.; Massarotti, C.; Pajalic, Z.; Luegmair, K.; Kotlowska, A.; Lengler, L.; Olza, I.; Grylka-Baeschlin, S.; et al. Maternal Plasma Levels of Oxytocin during Breastfeeding—A Systematic Review. PLoS ONE 2020, 15, e0235806. [Google Scholar] [CrossRef]
- Hantsoo, L.; Jašarević, E.; Criniti, S.; McGeehan, B.; Tanes, C.; Sammel, M.D.; Elovitz, M.A.; Compher, C.; Wu, G.; Epperson, C.N. Childhood Adversity Impact on Gut Microbiota and Inflammatory Response to Stress during Pregnancy. Brain Behav. Immun. 2019, 75, 240–250. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, A.M.; Christian, L.M. Repetitive Negative Thinking, Meaning in Life, and Serum Cytokine Levels in Pregnant Women: Varying Associations by Socioeconomic Status. J. Behav. Med. 2019, 42, 960–972. [Google Scholar] [CrossRef] [PubMed]
- Leff Gelman, P.; Mancilla-Herrera, I.; Flores-Ramos, M.; Saravia Takashima, M.F.; Cruz Coronel, F.M.; Cruz Fuentes, C.; Pérez Molina, A.; Hernández-Ruiz, J.; Silva-Aguilera, F.S.; Farfan-Labonne, B.; et al. The Cytokine Profile of Women with Severe Anxiety and Depression during Pregnancy. BMC Psychiatry 2019, 19, 104. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, L.; Nousiainen, N.; Scheinin, N.M.; Maksimow, M.; Salmi, M.; Lehto, S.M.; Tolvanen, M.; Lukkarinen, H.; Karlsson, H. Cytokine Profile and Maternal Depression and Anxiety Symptoms in Mid-Pregnancy—the FinnBrain Birth Cohort Study. Arch. Womens Ment. Health 2017, 20, 39–48. [Google Scholar] [CrossRef]
- Osborne, L.M.; Yenokyan, G.; Fei, K.; Kraus, T.; Moran, T.; Monk, C.; Sperling, R. Innate Immune Activation and Depressive and Anxious Symptoms across the Peripartum: An Exploratory Study. Psychoneuroendocrinology 2019, 99, 80–86. [Google Scholar] [CrossRef]
- Del Giudice, M.; Ellis, B.J.; Shirtcliff, E.A. The Adaptive Calibration Model of Stress Responsivity. Neurosci. Biobehav. Rev. 2011, 35, 1562–1592. [Google Scholar] [CrossRef]
- Davis, E.P.; Hankin, B.L.; Swales, D.A.; Hoffman, M.C. An Experimental Test of the Fetal Programming Hypothesis: Can We Reduce Child Ontogenetic Vulnerability to Psychopathology by Decreasing Maternal Depression? Dev. Psychopathol. 2018, 30, 787–806. [Google Scholar] [CrossRef]
- Kapoor, A.; Petropoulos, S.; Matthews, S.G. Fetal Programming of Hypothalamic–Pituitary–Adrenal (HPA) Axis Function and Behavior by Synthetic Glucocorticoids. Brain Res. Rev. 2008, 57, 586–595. [Google Scholar] [CrossRef]
- Park, M.; Brain, U.; Grunau, R.E.; Diamond, A.; Oberlander, T.F. Maternal Depression Trajectories from Pregnancy to 3 Years Postpartum Are Associated with Children’s Behavior and Executive Functions at 3 and 6 Years. Arch. Womens Ment. Health 2018, 21, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Sandman, C.A.; Class, Q.A.; Glynn, L.M.; Davis, E.P. Neurobehavioral disorders and developmental origins of health and disease. In The Epigenome and Developmental Origins of Health and Disease; Elsevier: Amsterdam, The Netherlands, 2016; pp. 235–266. [Google Scholar]
- Field, T.; Diego, M.; Dieter, J.; Hernandez-Reif, M.; Schanberg, S.; Kuhn, C.; Yando, R.; Bendell, D. Prenatal Depression Effects on the Fetus and the Newborn. Infant Behav. Dev. 2004, 27, 216–229. [Google Scholar] [CrossRef]
- Gonzalez, M.Z.; Wroblewski, K.L.; Allen, J.P.; Coan, J.A.; Connelly, J.J. OXTR DNA Methylation Moderates the Developmental Calibration of Neural Reward Sensitivity. Dev. Psychobiol. 2021, 63, 114–124. [Google Scholar] [CrossRef]
- Unternaehrer, E.; Bolten, M.; Nast, I.; Staehli, S.; Meyer, A.H.; Dempster, E.; Hellhammer, D.H.; Lieb, R.; Meinlschmidt, G. Maternal Adversities during Pregnancy and Cord Blood Oxytocin Receptor (OXTR) DNA Methylation. Soc. Cogn. Affect. Neurosci. 2016, 11, 1460–1470. [Google Scholar] [CrossRef] [PubMed]
- Lawler, J.M.; Bocknek, E.L.; McGinnis, E.W.; Martinez-Torteya, C.; Rosenblum, K.L.; Muzik, M. Maternal Postpartum Depression Increases Vulnerability for Toddler Behavior Problems through Infant Cortisol Reactivity. Infancy 2019, 24, 249–274. [Google Scholar] [CrossRef]
- Howland, M.A.; Sandman, C.A.; Davis, E.P.; Stern, H.S.; Phelan, M.; Baram, T.Z.; Glynn, L.M. Prenatal Maternal Mood Entropy Is Associated with Child Neurodevelopment. Emotion 2021, 21, 489–498. [Google Scholar] [CrossRef]
- Goodman, S.H.; Rouse, M.H.; Connell, A.M.; Broth, M.R.; Hall, C.M.; Heyward, D. Maternal Depression and Child Psychopathology: A Meta-Analytic Review. Clin. Child Fam. Psychol. Rev. 2011, 14, 1–27. [Google Scholar] [CrossRef] [PubMed]
- La Marca-Ghaemmaghami, P.; La Marca, R.; Dainese, S.M.; Haller, M.; Zimmermann, R.; Ehlert, U. The Association between Perceived Emotional Support, Maternal Mood, Salivary Cortisol, Salivary Cortisone, and the Ratio between the Two Compounds in Response to Acute Stress in Second Trimester Pregnant Women. J. Psychosom. Res. 2013, 75, 314–320. [Google Scholar] [CrossRef]
- Pulopulos, M.M.; Baeken, C.; De Raedt, R. Cortisol Response to Stress: The Role of Expectancy and Anticipatory Stress Regulation. Horm. Behav. 2020, 117, 104587. [Google Scholar] [CrossRef]
- Iob, E.; Kirschbaum, C.; Steptoe, A. Positive and Negative Social Support and HPA-Axis Hyperactivity: Evidence from Glucocorticoids in Human Hair. Psychoneuroendocrinology 2018, 96, 100–108. [Google Scholar] [CrossRef]
- Ozbay, F.; Johnson, D.C.; Dimoulas, E.; Morgan, C.A.; Charney, D.; Southwick, S. Social Support and Resilience to Stress: From Neurobiology to Clinical Practice. Psychiatry Edgmont Pa Townsh. 2007, 4, 35–40. [Google Scholar]
- Staufenbiel, S.M.; Koenders, M.A.; Giltay, E.J.; Elzinga, B.M.; Manenschijn, L.; Hoencamp, E.; van Rossum, E.F.C.; Spijker, A.T. Recent Negative Life Events Increase Hair Cortisol Concentrations in Patients with Bipolar Disorder. Stress 2014, 17, 451–459. [Google Scholar] [CrossRef]
- Cruwys, T.; Dingle, G.A.; Haslam, C.; Haslam, S.A.; Jetten, J.; Morton, T.A. Social Group Memberships Protect against Future Depression, Alleviate Depression Symptoms and Prevent Depression Relapse. Soc. Sci. Med. 2013, 98, 179–186. [Google Scholar] [CrossRef]
- Surkan, P.J.; Peterson, K.E.; Hughes, M.D.; Gottlieb, B.R. The Role of Social Networks and Support in Postpartum Women’s Depression: A Multiethnic Urban Sample. Matern. Child Health J. 2006, 10, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Seymour-Smith, M.; Cruwys, T.; Haslam, S.A.; Brodribb, W. Loss of Group Memberships Predicts Depression in Postpartum Mothers. Soc. Psychiatry Psychiatr. Epidemiol. 2017, 52, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Elmer, T.; Stadtfeld, C. Depressive Symptoms Are Associated with Social Isolation in Face-to-Face Interaction Networks. Sci. Rep. 2020, 10, 1444. [Google Scholar] [CrossRef]
- Carter, C.S.; Porges, S.W. Neural mechanisms underlying human-animal interaction: An evolutionary perspective. In The Social Neuroscience of Human-Animal Interaction; Freund, L.S., McCune, S., Esposito, L., Gee, N.R., McCardle, P., Eds.; American Psychological Association: Washington, DC, USA, 2016; pp. 89–105. [Google Scholar]
- Applebaum, J.W.; Peek, C.W.; Zsembik, B.A. Examining US Pet Ownership Using the General Social Survey. Soc. Sci. J. 2020. [Google Scholar] [CrossRef]
- McConnell, A.R.; Paige Lloyd, E.; Humphrey, B.T. We Are Family: Viewing Pets as Family Members Improves Wellbeing. Anthrozoös 2019, 32, 459–470. [Google Scholar] [CrossRef]
- McDonald, S.E.; Collins, E.A.; Maternick, A.; Nicotera, N.; Graham-Bermann, S.; Ascione, F.R.; Williams, J.H. Intimate Partner Violence Survivors’ Reports of Their Children’s Exposure to Companion Animal Maltreatment: A Qualitative Study. J. Interpers. Violence 2019, 34, 2627–2652. [Google Scholar] [CrossRef]
- McDonald, S.E.; Matijczak, A.; Nicotera, N.; Applebaum, J.W.; Kremer, L.; Natoli, G.; O’Ryan, R.; Booth, L.J.; Murphy, J.L.; Tomlinson, C.A.; et al. “He Was like, My Ride or Die”: Sexual and Gender Minority Emerging Adults’ Perspectives on Living with Pets during the Transition to Adulthood. Emerg. Adulthood 2021. [Google Scholar] [CrossRef]
- McNicholas, J.; Collis, G.M. Children’s Representations of Pets in Their Social Networks. Child Care Health Dev. 2001, 27, 279–294. [Google Scholar] [CrossRef]
- Wood, L.; Martin, K.; Christian, H.; Nathan, A.; Lauritsen, C.; Houghton, S.; Kawachi, I.; McCune, S. The Pet Factor—Companion Animals as a Conduit for Getting to Know People, Friendship Formation and Social Support. PLoS ONE 2015, 10, e0122085. [Google Scholar] [CrossRef]
- Hawkins, R.D.; McDonald, S.E.; O’Connor, K.; Matijczak, A.; Ascione, F.R.; Williams, J.H. Exposure to Intimate Partner Violence and Internalizing Symptoms: The Moderating Effects of Positive Relationships with Pets and Animal Cruelty Exposure. Child Abuse Negl. 2019, 98, 10416. [Google Scholar] [CrossRef] [PubMed]
- Riggs, D.W.; Taylor, N.; Signal, T.; Fraser, H.; Donovan, C. People of Diverse Genders and/or Sexualities and Their Animal Companions: Experiences of Family Violence in a Binational Sample. J. Fam. Issues 2018, 39, 4226–4247. [Google Scholar] [CrossRef]
- Rosenberg, S.; Riggs, D.W.; Taylor, N.; Fraser, H. ‘Being Together Really Helped’: Australian Transgender and Non-Binary People and Their Animal Companions Living through Violence and Marginalisation. J. Sociol. 2020, 56, 571–590. [Google Scholar] [CrossRef]
- McDonald, S.E.; Murphy, J.L.; Tomlinson, C.A.; Matijczak, A.; Applebaum, J.W.; Wike, T.L.; Kattari, S.K. Relations Between Sexual and Gender Minority Stress, Personal Hardiness, and Psychological Stress in Emerging Adulthood: Examining Indirect Effects via Human-Animal Interaction. Youth Soc. 2021, in press. [Google Scholar] [CrossRef]
- McNicholas, J.; Gilbey, A.; Rennie, A.; Ahmedzai, S.; Dono, J.-A.; Ormerod, E. Pet Ownership and Human Health: A Brief Review of Evidence and Issues. BMJ 2005, 331, 1252–1254. [Google Scholar] [CrossRef]
- Wood, L.; Giles-Corti, B.; Bulsara, M. The Pet Connection: Pets as a Conduit for Social Capital? Soc. Sci. Med. 2005, 61, 1159–1173. [Google Scholar] [CrossRef]
- Gee, N.R.; Mueller, M.K.; Curl, A.L. Human–Animal Interaction and Older Adults: An Overview. Front. Psychol. 2017, 8, 1416. [Google Scholar] [CrossRef]
- Graham, T.M.; Glover, T.D. On the Fence: Dog Parks in the (Un)Leashing of Community and Social Capital. Leis. Sci. 2014, 36, 217–234. [Google Scholar] [CrossRef]
- Krause-Parello, C.A.; Gulick, E.E.; Basin, B. Loneliness, Depression, and Physical Activity in Older Adults: The Therapeutic Role of Human–Animal Interactions. Anthrozoös 2019, 32, 239–254. [Google Scholar] [CrossRef]
- Wells, D.L. The State of Research on Human–Animal Relations: Implications for Human Health. Anthrozoös 2019, 32, 169–181. [Google Scholar] [CrossRef]
- Wood, L.; Martin, K.; Christian, H.; Houghton, S.; Kawachi, I.; Vallesi, S.; McCune, S. Social Capital and Pet Ownership—A Tale of Four Cities. SSM Popul. Health 2017, 3, 442–447. [Google Scholar] [CrossRef]
- Cohen, S.; McKay, G. Social support, stress and the buffering hypothesis: A theoretical analysis. In Handbook of Psychology and Health; Baum, A., Taylor, S.E., Singer, J.E., Eds.; Lawrence Erlbaum Associations, Inc.: Hillsdale, NJ, USA, 1984; Volume 4, pp. 253–267. [Google Scholar]
- Hagerty, B.M.; Williams, R.A.; Coyne, J.C.; Early, M.R. Sense of Belonging and Indicators of Social and Psychological Functioning. Arch. Psychiatr. Nurs. 1996, 10, 235–244. [Google Scholar] [CrossRef]
- Kruse, J.A.; Hagerty, B.M.; Byers, W.S.; Gatien, G.; Williams, R.A. Considering a Relational Model for Depression in Navy Recruits. Mil. Med. 2014, 179, 1293–1300. [Google Scholar] [CrossRef]
- Lee, S.; Chung, J.E.; Park, N. Network Environments and Well-Being: An Examination of Personal Network Structure, Social Capital, and Perceived Social Support. Health Commun. 2018, 33, 22–31. [Google Scholar] [CrossRef]
- Ditzen, B.; Neumann, I.D.; Bodenmann, G.; von Dawans, B.; Turner, R.A.; Ehlert, U.; Heinrichs, M. Effects of Different Kinds of Couple Interaction on Cortisol and Heart Rate Responses to Stress in Women. Psychoneuroendocrinology 2007, 32, 565–574. [Google Scholar] [CrossRef]
- Pendry, P.; Vandagriff, J.L. Salivary studies of the social neuroscience of human-animal interaction. In Salivary Bioscience; Granger, D.A., Taylor, M.K., Eds.; Springer: Cham, Switzerland, 2020; pp. 555–581. [Google Scholar]
- Beetz, A.; Uvnäs-Moberg, K.; Julius, H.; Kotrschal, K. Psychosocial and Psychophysiological Effects of Human-Animal Interactions: The Possible Role of Oxytocin. Front. Psychol. 2012, 3, 234. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, C.; Kingdon, D.; Ellenbogen, M.A. A Meta-Analytic Review of the Impact of Intranasal Oxytocin Administration on Cortisol Concentrations during Laboratory Tasks: Moderation by Method and Mental Health. Psychoneuroendocrinology 2014, 49, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.C.; Kennedy, C.C.; DeVoe, D.C.; Hickey, M.; Nelson, T.; Kogan, L. An Examination of Changes in Oxytocin Levels in Men and Women Before and After Interaction With a Bonded Dog. Anthrozoös 2009, 22, 31–42. [Google Scholar] [CrossRef]
- Nagasawa, M.; Kikusui, T.; Onaka, T.; Ohta, M. Dog’s Gaze at Its Owner Increases Owner’s Urinary Oxytocin during Social Interaction. Horm. Behav. 2009, 55, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Christian, H.E.; Westgarth, C.; Bauman, A.; Richards, E.A.; Rhodes, R.E.; Evenson, K.R.; Mayer, J.A.; Thorpe, R.J. Dog Ownership and Physical Activity: A Review of the Evidence. J. Phys. Act. Health 2013, 10, 750–759. [Google Scholar] [CrossRef] [PubMed]
- Coleman, K.J.; Rosenberg, D.E.; Conway, T.L.; Sallis, J.F.; Saelens, B.E.; Frank, L.D.; Cain, K. Physical Activity, Weight Status, and Neighborhood Characteristics of Dog Walkers. Prev. Med. 2008, 47, 309–312. [Google Scholar] [CrossRef] [PubMed]
- Dall, P.M.; Ellis, S.L.H.; Ellis, B.M.; Grant, P.M.; Colyer, A.; Gee, N.R.; Granat, M.H.; Mills, D.S. The Influence of Dog Ownership on Objective Measures of Free-Living Physical Activity and Sedentary Behaviour in Community-Dwelling Older Adults: A Longitudinal Case-Controlled Study. BMC Public Health 2017, 17, 496. [Google Scholar] [CrossRef]
- Levine, G.N.; Allen, K.; Braun, L.T.; Christian, H.E.; Friedmann, E.; Taubert, K.A.; Thomas, S.A.; Wells, D.L.; Lange, R.A. Pet Ownership and Cardiovascular Risk: A Scientific Statement From the American Heart Association. Circulation 2013, 127, 2353–2363. [Google Scholar] [CrossRef]
- McConnell, A.R.; Brown, C.M.; Shoda, T.M.; Stayton, L.E.; Martin, C.E. Friends with Benefits: On the Positive Consequences of Pet Ownership. J. Personal. Soc. Psychol. 2011, 101, 1239–1252. [Google Scholar] [CrossRef]
- Potter, K.; Teng, J.E.; Masteller, B.; Rajala, C.; Balzer, L.B. Examining How Dog ‘Acquisition’ Affects Physical Activity and Psychosocial Well-Being: Findings from the BuddyStudy Pilot Trial. Animals 2019, 9, 666. [Google Scholar] [CrossRef]
- Schulz, C.; König, H.-H.; Hajek, A. Differences in Self-Esteem between Cat Owners, Dog Owners, and Individuals without Pets. Front. Vet. Sci. 2020, 7, 552. [Google Scholar] [CrossRef]
- Van Houtte, B.A.; Jarvis, P.A. The Role of Pets in Preadolescent Psychosocial Development. J. Appl. Dev. Psychol. 1995, 16, 463–479. [Google Scholar] [CrossRef]
- Hinic, K.; Kowalski, M.O.; Holtzman, K.; Mobus, K. The Effect of a Pet Therapy and Comparison Intervention on Anxiety in Hospitalized Children. J. Pediatr. Nurs. 2019, 46, 55–61. [Google Scholar] [CrossRef]
- Lynch, C.E.; Magann, E.F.; Barringer, S.N.; Ounpraseuth, S.T.; Eastham, D.G.; Lewis, S.D.; Stowe, Z.N. Pet Therapy Program for Antepartum High-Risk Pregnancies: A Pilot Study. J. Perinatol. 2014, 34, 816–818. [Google Scholar] [CrossRef]
- Lem, M.; Coe, J.B.; Haley, D.B.; Stone, E.; O’Grady, W. The Protective Association between Pet Ownership and Depression among Street-Involved Youth: A Cross-Sectional Study. Anthrozoös 2016, 29, 123–136. [Google Scholar] [CrossRef]
- Liu, S.; Powell, L.; Chia, D.; Russ, T.C.; McGreevy, P.D.; Bauman, A.E.; Edwards, K.M.; Stamatakis, E. Is Dog Ownership Associated with Mental Health? A Population Study of 68,362 Adults Living in England. Anthrozoös 2019, 32, 729–739. [Google Scholar] [CrossRef]
- Souter, M.A.; Miller, M.D. Do Animal-Assisted Activities Effectively Treat Depression? A Meta-Analysis. Anthrozoös 2007, 20, 167–180. [Google Scholar] [CrossRef]
- Colombo, G.; Buono, M.D.; Smania, K.; Raviola, R.; De Leo, D. Pet Therapy and Institutionalized Elderly: A Study on 144 Cognitively Unimpaired Subjects. Arch. Gerontol. Geriatr. 2006, 42, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Jessen, J.; Cardiello, F.; Baun, M.M. Avian Companionship in Alleviation of Depression, Loneliness, and Low Morale of Older Adults in Skilled Rehabilitation Units. Psychol. Rep. 1996, 78, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Barker, S.B.; Pandurangi, A.K.; Best, A.M. Effects of Animal-Assisted Therapy on Patients’ Anxiety, Fear, and Depression before ECT. J. ECT 2003, 19, 38–44. [Google Scholar] [CrossRef]
- Braungart-Rieker, J.M.; Garwood, M.M.; Powers, B.P.; Wang, X. Parental Sensitivity, Infant Affect, and Affect Regulation: Predictors of Later Attachment. Child Dev. 2001, 72, 252–270. [Google Scholar] [CrossRef]
- Brown, S.M.; Schlueter, L.J.; Hurwich-Reiss, E.; Dmitrieva, J.; Miles, E.; Watamura, S.E. Parental Buffering in the Context of Poverty: Positive Parenting Behaviors Differentiate Young Children’s Stress Reactivity Profiles. Dev. Psychopathol. 2020, 32, 1778–1787. [Google Scholar] [CrossRef]
- Kok, R.; Thijssen, S.; Bakermans-Kranenburg, M.J.; Jaddoe, V.W.V.; Verhulst, F.C.; White, T.; van IJzendoorn, M.H.; Tiemeier, H. Normal Variation in Early Parental Sensitivity Predicts Child Structural Brain Development. J. Am. Acad. Child Adolesc. Psychiatry 2015, 54, 824–831.e1. [Google Scholar] [CrossRef]
- Dreschel, N.A.; Granger, D.A. Advancing the social neuroscience of human-animal interaction: The role of salivary bioscience. In The Social Neuroscience of Human-Animal Interaction; Freund, L.S., McCune, S., Esposito, L., Gee, N.R., McCardle, P., Eds.; American Psychological Association: Washington, DC, USA, 2016; pp. 195–216. [Google Scholar]
- Powell, L.; Edwards, K.M.; Bauman, A.; Guastella, A.J.; Drayton, B.; Stamatakis, E.; McGreevy, P. Canine Endogenous Oxytocin Responses to Dog-Walking and Affiliative Human–Dog Interactions. Animals 2019, 9, 51. [Google Scholar] [CrossRef]
- Rodriguez, K.E.; Guérin, N.A.; Gabriels, R.L.; Serpell, J.A.; Schreiner, P.J.; O’Haire, M.E. The State of Assessment in Human-Animal Interaction Research. Hum. Anim. Interact. Bull. 2018, 6, 63–81. [Google Scholar]
- Beetz, A.; Bales, K. Affiliation in human-animal interaction. In The Social Neuroscience of Human-Animal Interaction; Freund, L.S., McCune, S., Esposito, L., Gee, N.R., McCardle, P., Eds.; American Psychological Association: Washington, DC, USA, 2016; pp. 107–125. [Google Scholar]
- MacLean, E.L.; Gesquiere, L.R.; Gee, N.R.; Levy, K.; Martin, W.L.; Carter, C.S. Effects of Affiliative Human–Animal Interaction on Dog Salivary and Plasma Oxytocin and Vasopressin. Front. Psychol. 2017, 8, 1606. [Google Scholar] [CrossRef] [PubMed]
- Demakis, G.J.; McAdams, D. Personality, Social Support and Well-Being among First Year College Students. Coll. Stud. J. 1994, 28, 235–243. [Google Scholar]
- Nagasawa, M.; Mitsui, S.; En, S.; Ohtani, N.; Ohta, M.; Sakuma, Y.; Onaka, T.; Mogi, K.; Kikusui, T. Oxytocin-Gaze Positive Loop and the Coevolution of Human-Dog Bonds. Science 2015, 348, 333–336. [Google Scholar] [CrossRef]
- Jenkins, J.L. Physiological Effects of Petting a Companion Animal. Psychol. Rep. 1986, 58, 21–22. [Google Scholar] [CrossRef] [PubMed]
- Vormbrock, J.K.; Grossberg, J.M. Cardiovascular Effects of Human-Pet Dog Interactions. J. Behav. Med. 1988, 11, 509–517. [Google Scholar] [CrossRef] [PubMed]
- Demello, L.R. The Effect of the Presence of a Companion-Animal on Physiological Changes Following the Termination of Cognitive Stressors. Psychol. Health 1999, 14, 859–868. [Google Scholar] [CrossRef]
- Handlin, L.; Hydbring-Sandberg, E.; Nilsson, A.; Ejdebäck, M.; Jansson, A.; Uvnäs-Moberg, K. Short-Term Interaction between Dogs and Their Owners: Effects on Oxytocin, Cortisol, Insulin and Heart Rate—An Exploratory Study. Anthrozoös 2011, 24, 301–315. [Google Scholar] [CrossRef]
- Charnetski, C.J.; Riggers, S.; Brennan, F.X. Effect of Petting a Dog on Immune System Function. Psychol. Rep. 2004, 95, 1087–1091. [Google Scholar] [CrossRef]
- Odendaal, J.S.J. Animal-Assisted Therapy—Magic or Medicine? J. Psychosom. Res. 2000, 49, 275–280. [Google Scholar] [CrossRef]
- Barker, S.B.; Knisely, J.S.; McCain, N.L.; Best, A.M. Measuring Stress and Immune Response in Healthcare Professionals Following Interaction with a Therapy Dog: A Pilot Study. Psychol. Rep. 2005, 96, 713–729. [Google Scholar] [CrossRef]
- Pendry, P.; Vandagriff, J.L. Animal Visitation Program (AVP) Reduces Cortisol Levels of University Students: A Randomized Controlled Trial. AERA Open 2019, 5, 233285841985259. [Google Scholar] [CrossRef]
- Kertes, D.A.; Liu, J.; Hall, N.J.; Hadad, N.A.; Wynne, C.D.L.; Bhatt, S.S. Effect of Pet Dogs on Children’s Perceived Stress and Cortisol Stress Response. Soc. Dev. 2017, 26, 382–401. [Google Scholar] [CrossRef]
- Polheber, J.P.; Matchock, R.L. The Presence of a Dog Attenuates Cortisol and Heart Rate in the Trier Social Stress Test Compared to Human Friends. J. Behav. Med. 2014, 37, 860–867. [Google Scholar] [CrossRef]
- Wells, D.L. The Facilitation of Social Interactions by Domestic Dogs. Anthrozoös 2004, 17, 340–352. [Google Scholar] [CrossRef]
- Prothmann, A.; Ettrich, C.; Prothmann, S. Preference for, and Responsiveness to, People, Dogs and Objects in Children with Autism. Anthrozoös 2009, 22, 161–171. [Google Scholar] [CrossRef]
- Cardoso, C.; Ellenbogen, M.A.; Orlando, M.A.; Bacon, S.L.; Joober, R. Intranasal Oxytocin Attenuates the Cortisol Response to Physical Stress: A Dose–Response Study. Psychoneuroendocrinology 2013, 38, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Cochran, D.M.; Fallon, D.; Hill, M.; Frazier, J.A. The Role of Oxytocin in Psychiatric Disorders: A Review of Biological and Therapeutic Research Findings. Harv. Rev. Psychiatry 2013, 21, 219–247. [Google Scholar] [CrossRef]
- Bertsch, K.; Gamer, M.; Schmidt, B.; Schmidinger, I.; Walther, S.; Kästel, T.; Schnell, K.; Büchel, C.; Domes, G.; Herpertz, S.C. Oxytocin and Reduction of Social Threat Hypersensitivity in Women With Borderline Personality Disorder. Am. J. Psychiatry 2013, 170, 1169–1177. [Google Scholar] [CrossRef]
- Norman, G.J.; Cacioppo, J.T.; Morris, J.S.; Malarkey, W.B.; Berntson, G.G.; DeVries, A.C. Oxytocin Increases Autonomic Cardiac Control: Moderation by Loneliness. Biol. Psychol. 2011, 86, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Marr, C.A.; French, L.; Thompson, D.; Drum, L.; Greening, G.; Mormon, J.; Henderson, I.; Hughes, C.W. Animal-Assisted Therapy in Psychiatric Rehabilitation. Anthrozoös 2000, 13, 43–47. [Google Scholar] [CrossRef]
- Yount, R.; Ritchie, E.C.; St. Laurent, M.; Chumley, P.; Olmert, M.D. The Role of Service Dog Training in the Treatment of Combat-Related PTSD. Psychiatr. Ann. 2013, 43, 292–295. [Google Scholar] [CrossRef]
- Kertes, D.A.; Hall, N.; Bhatt, S.S. Children’s Relationship With Their Pet Dogs and OXTR Genotype Predict Child–Pet Interaction in an Experimental Setting. Front. Psychol. 2018, 9, 1472. [Google Scholar] [CrossRef] [PubMed]
- Smearman, E.L.; Yu, T.; Brody, G.H. Variation in the Oxytocin Receptor Gene Moderates the Protective Effects of a Family-based Prevention Program on Telomere Length. Brain Behav. 2016, 6, e00423. [Google Scholar] [CrossRef]
- Kushner, S.C.; Herzhoff, K.; Vrshek-Schallhorn, S.; Tackett, J.L. Depression in Early Adolescence: Contributions from Relational Aggression and Variation in the Oxytocin Receptor Gene. Aggress. Behav. 2018, 44, 60–68. [Google Scholar] [CrossRef]
- Rodrigues, S.M.; Saslow, L.R.; Garcia, N.; John, O.P.; Keltner, D. Oxytocin Receptor Genetic Variation Relates to Empathy and Stress Reactivity in Humans. Proc. Natl. Acad. Sci. USA 2009, 106, 21437–21441. [Google Scholar] [CrossRef]
- Krueger, F.; Parasuraman, R.; Iyengar, V.; Thornburg, M.; Weel, J.; Lin, M.; Clarke, E.; McCabe, K.; Lipsky, R.H. Oxytocin Receptor Genetic Variation Promotes Human Trust Behavior. Front. Hum. Neurosci. 2012, 6, 4. [Google Scholar] [CrossRef]
- Saphire-Bernstein, S.; Way, B.M.; Kim, H.S.; Sherman, D.K.; Taylor, S.E. Oxytocin Receptor Gene (OXTR) Is Related to Psychological Resources. Proc. Natl. Acad. Sci. USA 2011, 108, 15118–15122. [Google Scholar] [CrossRef]
- Prothmann, A.; Albrecht, K.; Dietrich, S.; Hornfeck, U.; Stieber, S.; Ettrich, C. Analysis of Child—Dog Play Behavior in Child Psychiatry. Anthrozoös 2005, 18, 43–58. [Google Scholar] [CrossRef]
- O’Haire, M.E.; Guérin, N.A.; Kirkham, A.C. Animal-Assisted Intervention for Trauma: A Systematic Literature Review. Front. Psychol. 2015, 6, 1121. [Google Scholar] [CrossRef] [PubMed]
- O’Haire, M.E.; Tedeschi, P.; Jenkins, M.A.; Braden, S.R.; Rodriguez, K.E. The impact of human-animal interaction in trauma recovery. In Transforming Trauma: Resilience and Healing Through our Connections with Animals; New directions in the human-animal bond; Tedeschi, P., Jenkins, M.A., Eds.; Purdue University Press: West Lafayette, IN, USA, 2019; pp. 15–53. [Google Scholar]
- Janssens, M.; Janssens, E.; Eshuis, J.; Lataster, J.; Simons, M.; Reijnders, J.; Jacobs, N. Companion Animals as Buffer against the Impact of Stress on Affect: An Experience Sampling Study. Animals 2021, 11, 2171. [Google Scholar] [CrossRef] [PubMed]
- Chandler, C.K. Eight Domains of Pet-Owner Wellness: Implications for Counselors and Counselor Training. In Clinician’s Guide to Treating Companion Animal Issues; Elsevier: Amsterdam, The Netherlands, 2019; pp. 103–114. [Google Scholar]
- Beetz, A.; Schöfmann, I.; Girgensohn, R.; Braas, R.; Ernst, C. Positive Effects of a Short-Term Dog-Assisted Intervention for Soldiers with Post-Traumatic Stress Disorder—A Pilot Study. Front. Vet. Sci. 2019, 6, 170. [Google Scholar] [CrossRef] [PubMed]
- Crowley-Robinson, P.; Fenwick, D.C.; Blackshaw, J.K. A Long-Term Study of Elderly People in Nursing Homes with Visiting and Resident Dogs. Appl. Anim. Behav. Sci. 1996, 47, 137–148. [Google Scholar] [CrossRef]
- Connell, C.M.; Janevic, M.R.; Solway, E.; McLaughlin, S.J. Are Pets a Source of Support or Added Burden for Married Couples Facing Dementia? J. Appl. Gerontol. 2007, 26, 472–485. [Google Scholar] [CrossRef]
- Buller, K.; Ballantyne, K.C. Living with and Loving a Pet with Behavioral Problems: Pet Owners’ Experiences. J. Vet. Behav. 2020, 37, 41–47. [Google Scholar] [CrossRef]
- Jakeman, M.; Oxley, J.A.; Owczarczak-Garstecka, S.C.; Westgarth, C. Pet Dog Bites in Children: Management and Prevention. BMJ Paediatr. Open 2020, 4, e000726. [Google Scholar] [CrossRef] [PubMed]
- Lodge, C.J.; Lowe, A.J.; Gurrin, L.C.; Matheson, M.C.; Balloch, A.; Axelrad, C.; Hill, D.J.; Hosking, C.S.; Rodrigues, S.; Svanes, C.; et al. Pets at Birth Do Not Increase Allergic Disease in At-Risk Children. Clin. Exp. Allergy 2012, 42, 1377–1385. [Google Scholar] [CrossRef]
- Fretzayas, A.; Kotzia, D.; Moustaki, M. Controversial Role of Pets in the Development of Atopy in Children. World J. Pediatr. 2013, 9, 112–119. [Google Scholar] [CrossRef]
- Ahmed, M.; Sood, A.; Gupta, J. Toxoplasmosis in Pregnancy. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 255, 44–50. [Google Scholar] [CrossRef]
- Kravetz, J.D.; Federman, D.G. Toxoplasmosis in Pregnancy. Am. J. Med. 2005, 118, 212–216. [Google Scholar] [CrossRef]
- Shaevitz, M.H.; Tullius, J.A.; Callahan, R.T.; Fulkerson, C.M.; Spitznagel, M.B. Early Caregiver Burden in Owners of Pets with Suspected Cancer: Owner Psychosocial Outcomes, Communication Behavior, and Treatment Factors. J. Vet. Intern. Med. 2020, 34, 2636–2644. [Google Scholar] [CrossRef] [PubMed]
- Spitznagel, M.B.; Jacobson, D.M.; Cox, M.D.; Carlson, M.D. Predicting Caregiver Burden in General Veterinary Clients: Contribution of Companion Animal Clinical Signs and Problem Behaviors. Vet. J. 2018, 236, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Spitznagel, M.B.; Mueller, M.K.; Fraychak, T.; Hoffman, A.M.; Carlson, M.D. Validation of an Abbreviated Instrument to Assess Veterinary Client Caregiver Burden. J. Vet. Intern. Med. 2019, 33, 1251–1259. [Google Scholar] [CrossRef]
- Bussolari, C.J.; Habarth, J.; Katz, R.; Phillips, S.; Carmack, B.; Packman, W. The Euthanasia Decision-Making Process: A Qualitative Exploration of Bereaved Companion Animal Owners. Bereave. Care 2018, 37, 101–108. [Google Scholar] [CrossRef]
- Habarth, J.; Bussolari, C.; Gomez, R.; Carmack, B.J.; Ronen, R.; Field, N.P.; Packman, W. Continuing Bonds and Psychosocial Functioning in a Recently Bereaved Pet Loss Sample. Anthrozoös 2017, 30, 651–670. [Google Scholar] [CrossRef]
- Hunt, M.; Padilla, Y. Development of the Pet Bereavement Questionnaire. Anthrozoös 2006, 19, 308–324. [Google Scholar] [CrossRef]
- LaVallee, E.; Mueller, M.K.; McCobb, E. A Systematic Review of the Literature Addressing Veterinary Care for Underserved Communities. J. Appl. Anim. Welf. Sci. 2017, 20, 381–394. [Google Scholar] [CrossRef]
- Power, E.R. Renting with Pets: A Pathway to Housing Insecurity? Hous. Stud. 2017, 32, 336–360. [Google Scholar] [CrossRef]
- Rose, D.; McMillian, C.; Carter, O. Pet-Friendly Rental Housing: Racial and Spatial Inequalities. Space Cult. 2020. [Google Scholar] [CrossRef]
- Saadeh, F.B.; Clark, M.A.; Rogers, M.L.; Linkletter, C.D.; Phipps, M.G.; Padbury, J.F.; Vivier, P.M. Pregnant and Moving: Understanding Residential Mobility during Pregnancy and in the First Year of Life Using a Prospective Birth Cohort. Matern. Child Health J. 2013, 17, 330–343. [Google Scholar] [CrossRef]
- Applebaum, J.W.; Adams, B.L.; Eliasson, M.N.; Zsembik, B.A.; McDonald, S.E. How Pets Factor into Healthcare Decisions for COVID-19: A One Health Perspective. One Health 2020, 11, 100176. [Google Scholar] [CrossRef]
- Adams, B.L.; Applebaum, J.W.; Eliasson, M.N.; McDonald, S.E.; Zsembik, B.A. Child and Pet Care-Planning During COVID-19: Considerations for the Evolving Family Unit. Fam. Relat. 2021, 70, 705–716. [Google Scholar] [CrossRef] [PubMed]
- Parfitt, Y.; Ayers, S. Transition to Parenthood and Mental Health in First-Time Parents. Infant Ment. Health J. 2014, 35, 263–273. [Google Scholar] [CrossRef]
- Tham, E.K.H.; Tan, J.; Chong, Y.-S.; Kwek, K.; Saw, S.-M.; Teoh, O.-H.; Goh, D.Y.T.; Meaney, M.J.; Broekman, B.F.P. Associations between Poor Subjective Prenatal Sleep Quality and Postnatal Depression and Anxiety Symptoms. J. Affect. Disord. 2016, 202, 91–94. [Google Scholar] [CrossRef]
- Andre, C.J.; Lovallo, V.; Spencer, R.M.C. The Effects of Bed Sharing on Sleep: From Partners to Pets. Sleep Health 2021, 7, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Mojahed, A.; Alaidarous, N.; Kopp, M.; Pogarell, A.; Thiel, F.; Garthus-Niegel, S. Prevalence of Intimate Partner Violence among Intimate Partners during the Perinatal Period: A Narrative Literature Review. Front. Psychiatry 2021, 12, 601236. [Google Scholar] [CrossRef] [PubMed]
- McDonald, S.E.; Collins, E.A.; Nicotera, N.; Hageman, T.O.; Ascione, F.R.; Williams, J.H.; Graham-Bermann, S.A. Children’s Experiences of Companion Animal Maltreatment in Households Characterized by Intimate Partner Violence. Child Abuse Negl. 2015, 50, 116–127. [Google Scholar] [CrossRef]
- Collins, E.A.; Cody, A.M.; McDonald, S.E.; Nicotera, N.; Ascione, F.R.; Williams, J.H. A Template Analysis of Intimate Partner Violence Survivors’ Experiences of Animal Maltreatment: Implications for Safety Planning and Intervention. Violence Women 2018, 24, 452–476. [Google Scholar] [CrossRef]
- Monsalve, S.; Ferreira, F.; Garcia, R. The Connection between Animal Abuse and Interpersonal Violence: A Review from the Veterinary Perspective. Res. Vet. Sci. 2017, 114, 18–26. [Google Scholar] [CrossRef]
- Faver, C.A.; Strand, E.B. Domestic Violence and Animal Cruelty: Untangling the Web of Abuse. J. Soc. Work Educ. 2003, 39, 237–253. [Google Scholar] [CrossRef]
- Moyer, S.W.; Kinser, P.A. A Comprehensive Conceptual Framework to Guide Clinical Practice and Research About Mental Health during the Perinatal Period. J. Perinat. Neonatal Nurs. 2021, 35, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Highet, N.; Stevenson, A.L.; Purtell, C.; Coo, S. Qualitative Insights into Women’s Personal Experiences of Perinatal Depression and Anxiety. Women Birth 2014, 27, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Beetz, A.M. Theories and Possible Processes of Action in Animal Assisted Interventions. Appl. Dev. Sci. 2017, 21, 139–149. [Google Scholar] [CrossRef]
- Shoesmith, E.; Shahab, L.; Kale, D.; Mills, D.S.; Reeve, C.; Toner, P.; Santos de Assis, L.; Ratschen, E. The Influence of Human–Animal Interactions on Mental and Physical Health during the First COVID-19 Lockdown Phase in the U.K.: A Qualitative Exploration. Int. J. Environ. Res. Public. Health 2021, 18, 976. [Google Scholar] [CrossRef] [PubMed]
- Carter, C.S.; Kenkel, W.M.; MacLean, E.L.; Wilson, S.R.; Perkeybile, A.M.; Yee, J.R.; Ferris, C.F.; Nazarloo, H.P.; Porges, S.W.; Davis, J.M.; et al. Is Oxytocin “Nature’s Medicine”? Pharmacol. Rev. 2020, 72, 829–861. [Google Scholar] [CrossRef] [PubMed]
- Herman, J.P.; Cullinan, W.E. Neurocircuitry of Stress: Central Control of the Hypothalamo–Pituitary–Adrenocortical Axis. Trends Neurosci. 1997, 20, 78–84. [Google Scholar] [CrossRef]
- Milgrom, J.; Hirshler, Y.; Reece, J.; Holt, C.; Gemmill, A.W. Social Support—A Protective Factor for Depressed Perinatal Women? Int. J. Environ. Res. Public. Health 2019, 16, 1426. [Google Scholar] [CrossRef]
- Rallis, S.; Skouteris, H.; McCabe, M.; Milgrom, J. The Transition to Motherhood: Towards a Broader Understanding of Perinatal Distress. Women Birth 2014, 27, 68–71. [Google Scholar] [CrossRef]
- Scorza, P.; Merz, E.C.; Spann, M.; Steinberg, E.; Feng, T.; Lee, S.; Werner, E.; Peterson, B.S.; Monk, C. Pregnancy-Specific Stress and Sensitive Caregiving during the Transition to Motherhood in Adolescents. BMC Pregnancy Childbirth 2021, 21, 458. [Google Scholar] [CrossRef]
- Louie, A.D.; Cromer, L.D.; Berry, J.O. Assessing Parenting Stress: Review of the Use and Interpretation of the Parental Stress Scale. Fam. J. 2017, 25, 359–367. [Google Scholar] [CrossRef]
- Nomaguchi, K.M.; Brown, S.L. Parental Strains and Rewards Among Mothers: The Role of Education. J. Marriage Fam. 2011, 73, 621–636. [Google Scholar] [CrossRef] [PubMed]
- Nomaguchi, K.M.; Milkie, M.A. Costs and Rewards of Children: The Effects of Becoming a Parent on Adults’ Lives. J. Marriage Fam. 2003, 65, 356–374. [Google Scholar] [CrossRef]
- Fewtrell, M.S. Randomised, Double Blind Trial of Oxytocin Nasal Spray in Mothers Expressing Breast Milk for Preterm Infants. Arch. Dis. Child. Fetal Neonatal Ed. 2005, 91, F169–F174. [Google Scholar] [CrossRef] [PubMed]
- Ruis, H.; Rolland, R.; Doesburg, W.; Broeders, G.; Corbey, R. Oxytocin Enhances Onset of Lactation among Mothers Delivering Prematurely. BMJ 1981, 283, 340–342. [Google Scholar] [CrossRef]
- Kingston, D.; Kehler, H.; Austin, M.-P.; Mughal, M.K.; Wajid, A.; Vermeyden, L.; Benzies, K.; Brown, S.; Stuart, S.; Giallo, R. Trajectories of Maternal Depressive Symptoms during Pregnancy and the First 12 Months Postpartum and Child Externalizing and Internalizing Behavior at Three Years. PLoS ONE 2018, 13, e0195365. [Google Scholar] [CrossRef]
- Mughal, M.K.; Giallo, R.; Arnold, P.; Benzies, K.; Kehler, H.; Bright, K.; Kingston, D. Trajectories of Maternal Stress and Anxiety from Pregnancy to Three Years and Child Development at 3 Years of Age: Findings from the All Our Families (AOF) Pregnancy Cohort. J. Affect. Disord. 2018, 234, 318–326. [Google Scholar] [CrossRef]
- Tien, J.; Lewis, G.D.; Liu, J. Prenatal Risk Factors for Internalizing and Externalizing Problems in Childhood. World J. Pediatr. 2020, 16, 341–355. [Google Scholar] [CrossRef]
- Davis, E.P.; Glynn, L.M.; Waffarn, F.; Sandman, C.A. Prenatal Maternal Stress Programs Infant Stress Regulation: Prenatal Cortisol and Infant Development. J. Child Psychol. Psychiatry 2011, 52, 119–129. [Google Scholar] [CrossRef]
- Werner, E.; Zhao, Y.; Evans, L.; Kinsella, M.; Kurzius, L.; Altincatal, A.; McDonough, L.; Monk, C. Higher Maternal Prenatal Cortisol and Younger Age Predict Greater Infant Reactivity to Novelty at 4 Months: An Observation-Based Study. Dev. Psychobiol. 2012, 55, 707–718. [Google Scholar] [CrossRef] [PubMed]
- Graham, A.M.; Rasmussen, J.M.; Rudolph, M.D.; Heim, C.M.; Gilmore, J.H.; Styner, M.; Potkin, S.G.; Entringer, S.; Wadhwa, P.D.; Fair, D.A.; et al. Maternal Systemic Interleukin-6 during Pregnancy Is Associated with Newborn Amygdala Phenotypes and Subsequent Behavior at 2 Years of Age. Biol. Psychiatry 2018, 83, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, J.M.; Graham, A.M.; Entringer, S.; Gilmore, J.H.; Styner, M.; Fair, D.A.; Wadhwa, P.D.; Buss, C. Maternal Interleukin-6 Concentration during Pregnancy Is Associated with Variation in Frontolimbic White Matter and Cognitive Development in Early Life. NeuroImage 2019, 185, 825–835. [Google Scholar] [CrossRef]
- Bernier, A.; Carlson, S.M.; Whipple, N. From External Regulation to Self-Regulation: Early Parenting Precursors of Young Children’s Executive Functioning. Child Dev. 2010, 81, 326–339. [Google Scholar] [CrossRef] [PubMed]
- Lunkenheimer, E.; Busuito, A.; Brown, K.M.; Skowron, E.A. Mother-Child Coregulation of Parasympathetic Processes Differs by Maltreatment Severity and Subtype. Child Maltreat. 2018, 23, 211–220. [Google Scholar] [CrossRef]
- Skowron, E.A.; Cipriano-Essel, E.; Benjamin, L.S.; Pincus, A.L.; Van Ryzin, M.J. Cardiac Vagal Tone and Quality of Parenting Show Concurrent and Time-Ordered Associations That Diverge in Abusive, Neglectful, and Non-Maltreating Mothers. Couple Fam. Psychol. Res. Pract. 2013, 2, 95–115. [Google Scholar] [CrossRef] [PubMed]
- Kalomiris, A.E.; Kiel, E.J. Mother-Toddler Cortisol Synchrony Moderates Risk of Early Internalizing Symptoms. Infancy 2018, 23, 232–251. [Google Scholar] [CrossRef]
- Chalmers, B. Family-Centred Perinatal Care: Improving Pregnancy, Birth and Postpartum Care; Cambridge University Press: Cambridge, UK, 2017. [Google Scholar]
- Chalmers, B. Family-centered, evidence-based, psycho-socially sensitive, and culturally respectful perinatal care: Still a futuristic dream! In Birthing Techno-Sapiens: Human-Technology Co-Evolution and the Future of Reproduction; Social science perspectives on childbirth and reproduction; Davis-Floyd, R., Ed.; Routledge: New York, NY, USA, 2021; pp. 222–235. [Google Scholar]
- Franck, L.S.; O’Brien, K. The Evolution of Family-centered Care: From Supporting Parent-delivered Interventions to a Model of Family Integrated Care. Birth Defects Res. 2019, 111, 1044–1059. [Google Scholar] [CrossRef]
- Paulson, J.F.; Bazemore, S.D. Prenatal and Postpartum Depression in Fathers and Its Association With Maternal Depression: A Meta-Analysis. JAMA 2010, 303, 1961. [Google Scholar] [CrossRef] [PubMed]
- Paulson, J.F.; Bazemore, S.D.; Goodman, J.H.; Leiferman, J.A. The Course and Interrelationship of Maternal and Paternal Perinatal Depression. Arch. Womens Ment. Health 2016, 19, 655–663. [Google Scholar] [CrossRef]
- Skjøthaug, T. Antecedents of Fathers’ Stress in Fatherhood. In Handbook of Fathers and Child Development; Fitzgerald, H.E., von Klitzing, K., Cabrera, N.J., Scarano de Mendonça, J., Skjøthaug, T., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 209–228. [Google Scholar]
- McCall, C.E.; Rodriguez, K.E.; Wadsworth, S.M.M.; Meis, L.A.; O’Haire, M.E. “A Part of Our Family”? Effects of Psychiatric Service Dogs on Quality of Life and Relationship Functioning in Military-Connected Couples. Mil. Behav. Health 2020, 8, 410–423. [Google Scholar] [CrossRef]
- Nieforth, L.O.; Craig, E.A.; Behmer, V.A.; MacDermid Wadsworth, S.; O’Haire, M.E. PTSD Service Dogs Foster Resilience among Veterans and Military Families. Curr. Psychol. 2021, in press. [Google Scholar] [CrossRef]
- Leahy-Warren, P.; McCarthy, G.; Corcoran, P. First-Time Mothers: Social Support, Maternal Parental Self-Efficacy and Postnatal Depression: First-Time Mothers. J. Clin. Nurs. 2012, 21, 388–397. [Google Scholar] [CrossRef]
- Carter, C.S. The Oxytocin–Vasopressin Pathway in the Context of Love and Fear. Front. Endocrinol. 2017, 8, 356. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
McDonald, S.E.; Tomlinson, C.A.; Applebaum, J.W.; Moyer, S.W.; Brown, S.M.; Carter, S.; Kinser, P.A. Human–Animal Interaction and Perinatal Mental Health: A Narrative Review of Selected Literature and Call for Research. Int. J. Environ. Res. Public Health 2021, 18, 10114. https://doi.org/10.3390/ijerph181910114
McDonald SE, Tomlinson CA, Applebaum JW, Moyer SW, Brown SM, Carter S, Kinser PA. Human–Animal Interaction and Perinatal Mental Health: A Narrative Review of Selected Literature and Call for Research. International Journal of Environmental Research and Public Health. 2021; 18(19):10114. https://doi.org/10.3390/ijerph181910114
Chicago/Turabian StyleMcDonald, Shelby E., Camie A. Tomlinson, Jennifer W. Applebaum, Sara W. Moyer, Samantha M. Brown, Sue Carter, and Patricia A. Kinser. 2021. "Human–Animal Interaction and Perinatal Mental Health: A Narrative Review of Selected Literature and Call for Research" International Journal of Environmental Research and Public Health 18, no. 19: 10114. https://doi.org/10.3390/ijerph181910114
APA StyleMcDonald, S. E., Tomlinson, C. A., Applebaum, J. W., Moyer, S. W., Brown, S. M., Carter, S., & Kinser, P. A. (2021). Human–Animal Interaction and Perinatal Mental Health: A Narrative Review of Selected Literature and Call for Research. International Journal of Environmental Research and Public Health, 18(19), 10114. https://doi.org/10.3390/ijerph181910114






