Impact of Rehabilitation Intensity on 3-Year Mortality among Children with Moderate to Severe Cerebral Palsy: A Population-Based Cohort Study
Abstract
:1. Introduction
2. Methods
2.1. Data Source
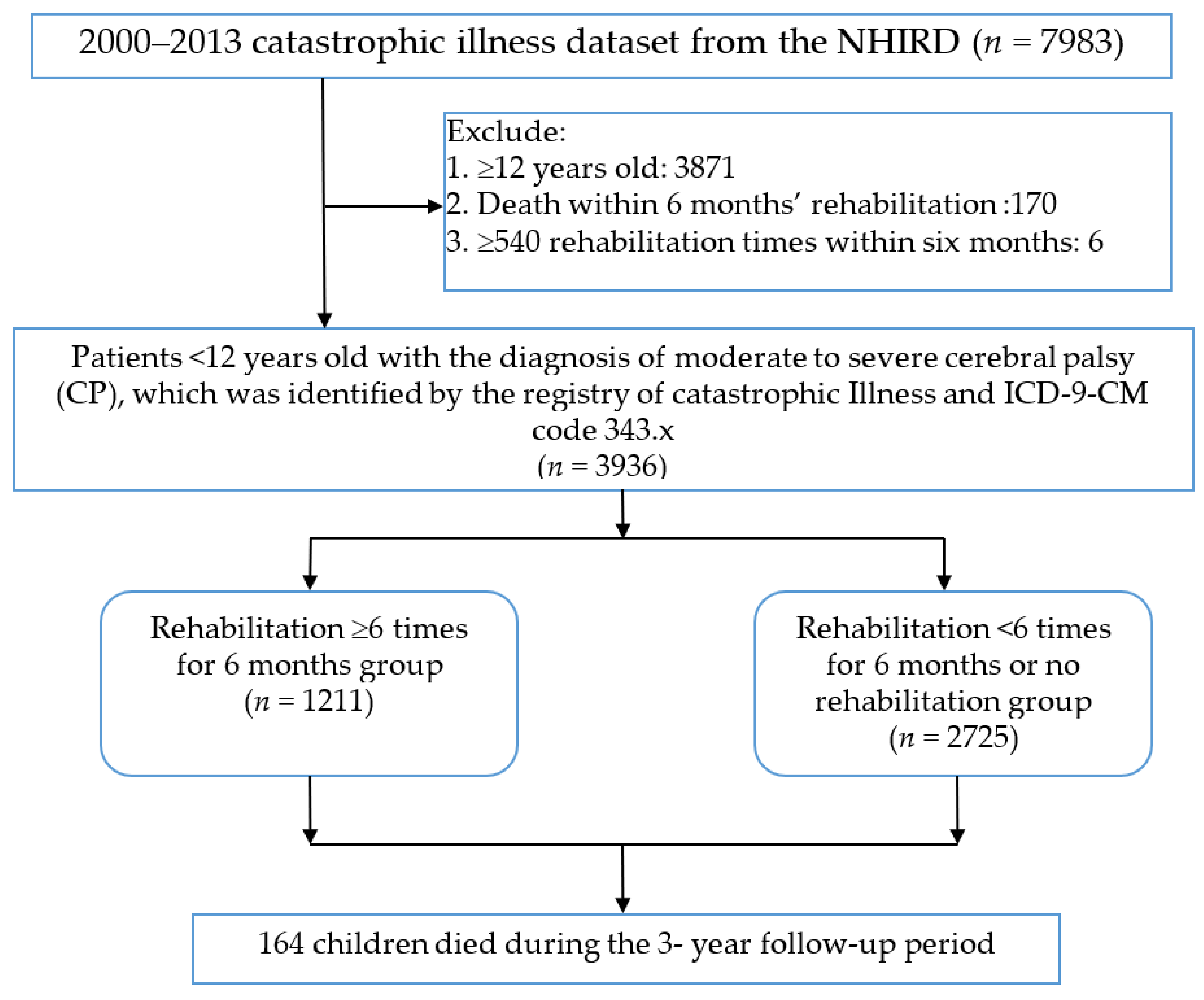
2.2. Study Population
2.3. Moderate to Severe CP Diagnosis
2.4. Variables
2.5. Outcome
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| NHIRD | National Health Insurance Research Database |
| RCIPD | Registry for Catastrophic Illnesses Patient Database |
| HR | Hazard ratio |
| aHR | adjusted hazard ratio |
| ICD-9-CM | International Classification of Disease, Ninth Revision, Clinical Modification |
| GERD | Gastroesophageal reflux disease |
References
- Strauss, D.; Brooks, J.; Rosenbloom, L.; Shavelle, R. Life expectancy in cerebral palsy: An update. Dev. Med. Child Neurol. 2008, 50, 487–493. [Google Scholar] [CrossRef]
- Blair, E.; Langdon, K.; McIntyre, S.; Lawrence, D.; Watson, L. Survival and mortality in cerebral palsy: Observations to the sixth decade from a data linkage study of a total population register and National Death Index. BMC Neurol. 2019, 19, 111. [Google Scholar] [CrossRef] [PubMed]
- Blair, E.; Watson, L.; Badawi, N.; Stanley, F.J. Life expectancy among people with cerebral palsy in Western Australia. Dev. Med. Child Neurol. 2001, 43, 508–515. [Google Scholar] [CrossRef] [PubMed]
- Reid, S.M.; Carlin, J.B.; Reddihough, D.S. Survival of individuals with cerebral palsy born in Victoria, Australia, between 1970 and 2004. Dev. Med. Child Neurol. 2012, 54, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Durufle-Tapin, A.; Colin, A.; Nicolas, B.; Lebreton, C.; Dauvergne, F.; Gallien, P. Analysis of the medical causes of death in cerebral palsy. Ann. Phys. Rehabil. Med. 2014, 57, 24–37. [Google Scholar] [CrossRef] [Green Version]
- Hutton, J.L.; Pharoah, P.O.D. Life expectancy in severe cerebral palsy. Arch. Dis. Child. 2006, 91, 254–258. [Google Scholar] [CrossRef] [Green Version]
- Morehead, R.S. Gastro-oesophageal reflux disease and non-asthma lung disease. Eur. Respir. Rev. 2009, 18, 233–243. [Google Scholar] [CrossRef]
- Strauss, D.J.; Shavelle, R.M.; Anderson, T.W. Life expectancy of children with cerebral palsy. Pediatr. Neurol. 1998, 18, 143–149. [Google Scholar] [CrossRef]
- Himmelmann, K.; Sundh, V. Survival with cerebral palsy over five decades in western Sweden. Dev. Med. Child Neurol. 2015, 57, 762–767. [Google Scholar] [CrossRef]
- Novak, I.; Morgan, C.; Adde, L.; Blackman, J.; Boyd, R.N.; Brunstrom-Hernandez, J.; Cioni, G.; Damiano, D.; Darrah, J.; Eliasson, A.-C.; et al. Early, Accurate Diagnosis and Early Intervention in Cerebral Palsy: Advances in Diagnosis and Treatment. JAMA Pediatr. 2017, 171, 897–907. [Google Scholar] [CrossRef]
- Green, D. Cerebral palsy in infancy: Targeted activity to optimize early growth and development. Br. J. Occup. Ther. 2014, 77, 634–636. [Google Scholar] [CrossRef]
- Williams, P.T.J.A.; Jiang, Y.-Q.; Martin, J.H. Motor system plasticity after unilateral injury in the developing brain. Dev. Med. Child Neurol. 2017, 59, 1224–1229. [Google Scholar] [CrossRef] [Green Version]
- Kulak, W.; Sobaniec, W.; Kuzia, J.S.; Bockowski, L. Neurophysiologic and neuroimaging studies of brain plasticity in children with spastic cerebral palsy. Exp. Neurol. 2006, 198, 4–11. [Google Scholar] [CrossRef]
- Trabacca, A.; Vespino, T.; Di Liddo, A.; Russo, L. Multidisciplinary rehabilitation for patients with cerebral palsy: Improving long-term care. J. Multidiscip. Healthc. 2016, 9, 455–462. [Google Scholar] [CrossRef] [Green Version]
- Araujo, Z.T.S.; Mendonça, K.M.P.P.; Souza, B.M.M.; Santos, T.Z.M.; Chaves, G.S.S.; Andriolo, B.N.G.; Nogueira, P.A.M.S. Pulmonary rehabilitation for people with chronic obstructive pulmonary disease: A protocol for an overview of Cochrane reviews. Medicine 2019, 98, e17129. [Google Scholar] [CrossRef]
- Goel, K.; Lennon, R.J.; Tilbury, R.T.; Squires, R.W.; Thomas, R.J. Impact of cardiac rehabilitation on mortality and cardiovascular events after percutaneous coronary intervention in the community. Circulation 2011, 123, 2344–2352. [Google Scholar] [CrossRef] [Green Version]
- Gibbs, J.C.; MacIntyre, N.J.; Ponzano, M.; Templeton, J.A.; Thabane, L.; Papaioannou, A.; Giangregorio, L.M. Exercise for improving outcomes after osteoporotic vertebral fracture. Cochrane Database Syst. Rev. 2019, 7, CD008618. [Google Scholar] [CrossRef]
- Hsing, A.W.; Ioannidis, J.P. Nationwide Population Science: Lessons from the Taiwan National Health Insurance Research Database. JAMA Int. Med. 2015, 175, 1527–1529. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, C.Y.; Su, C.C.; Shao, S.C.; Sung, S.F.; Lin, S.J.; Kao Yang, Y.H.; Lai, E.C. Taiwan’s National Health Insurance Research Database: Past and future. Clin. Epidemiol. 2019, 11, 349–358. [Google Scholar] [CrossRef] [Green Version]
- Administration, N.H.I. Patients with Catastrophic Illnesses or Rare Diseases. Available online: https://www.nhi.gov.tw/english/Content_List.aspx?n=F5B8E49CB4548C60&to%20pn=1D1ECC54F86E9050 (accessed on 19 January 2016).
- Tseng, S.-H.; Lee, J.-Y.; Chou, Y.-L.; Sheu, M.-L.; Lee, Y.-W. Association between socioeconomic status and cerebral palsy. PLoS ONE 2018, 13, e0191724. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Houtrow, A.; Murphy, N.; Council on Children with, D. Prescribing Physical, Occupational, and Speech Therapy Services for Children with Disabilities. Pediatrics 2019, 143, e20190285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Foley, N.; Teasell, R.; Salter, K.; Kruger, E.; Martino, R. Dysphagia treatment post stroke: A systematic review of randomised controlled trials. Age Ageing 2008, 37, 258–264. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bower, E.; Michell, D.; Burnett, M.; Campbell, M.J.; McLellan, D.L. Randomized controlled trial of physiotherapy in 56 children with cerebral palsy followed for 18 months. Dev. Med. Child Neurol. 2001, 43, 4–15. [Google Scholar] [CrossRef] [Green Version]
- Chang, Y.C.; Lin, J.D.; Tung, H.J.; Chiang, P.H.; Hsu, S.W. Outpatient physical therapy utilization for children and adolescents with intellectual disabilities in Taiwan: A population-based nationwide study. Res. Dev. Disabil. 2014, 35, 498–505. [Google Scholar] [CrossRef]
- Jonnalagadda, S.; Rodríguez, O.; Estrella, B.; Sabin, L.L.; Sempértegui, F.; Hamer, D.H. Etiology of severe pneumonia in Ecuadorian children. PLoS ONE 2017, 12, e0171687. [Google Scholar] [CrossRef]
- Blackmore, A.M.; Bear, N.; Blair, E.; Langdon, K.; Moshovis, L.; Steer, K.; Wilson, A.C. Predicting respiratory hospital admissions in young people with cerebral palsy. Arch. Dis. Child. 2018, 103, 1119–1124. [Google Scholar] [CrossRef] [Green Version]
- Kenyon, C.C.; Melvin, P.R.; Chiang, V.W.; Elliott, M.N.; Schuster, M.A.; Berry, J.G. Rehospitalization for childhood asthma: Timing, variation, and opportunities for intervention. J. Pediatr. 2014, 164, 300–305. [Google Scholar] [CrossRef]
- McAllister, D.A.; Liu, L.; Shi, T.; Chu, Y.; Reed, C.; Burrows, J.; Adeloye, D.; Rudan, I.; Black, R.E.; Campbell, H.; et al. Global, regional, and national estimates of pneumonia morbidity and mortality in children younger than 5 years between 2000 and 2015: A systematic analysis. Lancet Glob. Health 2019, 7, e47–e57. [Google Scholar] [CrossRef] [Green Version]
- Boel, L.; Pernet, K.; Toussaint, M.; Ides, K.; Leemans, G.; Haan, J.; Van Hoorenbeeck, K.; Verhulst, S. Respiratory morbidity in children with cerebral palsy: An overview. Dev. Med. Child Neurol. 2019, 61, 646–653. [Google Scholar] [CrossRef] [PubMed]
- Lagos-Guimaraes, H.N.; Teive, H.A.; Celli, A.; Santos, R.S.; Abdulmassih, E.M.; Hirata, G.C.; Gallinea, L.F. Aspiration Pneumonia in Children with Cerebral Palsy after Videofluoroscopic Swallowing Study. Int. Arch. Otorhinolaryngol. 2016, 20, 132–137. [Google Scholar] [PubMed] [Green Version]
- Strauss, D.; Shavelle, R.; Reynolds, R.; Rosenbloom, L.; Day, S. Survival in cerebral palsy in the last 20 years: Signs of improvement? Dev. Med. Child Neurol. 2007, 49, 86–92. [Google Scholar] [CrossRef]
- Liptak, G.S.; O’Donnell, M.; Conaway, M.; Chumlea, W.C.; Wolrey, G.; Henderson, R.C.; Fung, E.; Stallings, V.A.; Samson-Fang, L.; Calvert, R.; et al. Health status of children with moderate to severe cerebral palsy. Dev. Med. Child Neurol. 2001, 43, 364–370. [Google Scholar] [CrossRef]
- Meehan, E.; Reid, S.M.; Williams, K.; Freed, G.L.; Sewell, J.R.; Vidmar, S.; Donath, S.; Reddihough, D.S. Hospital admissions in children with cerebral palsy: A data linkage study. Dev. Med. Child Neurol. 2017, 59, 512–519. [Google Scholar] [CrossRef] [PubMed]
- Neligan, A.; Bell, G.S.; Johnson, A.L.; Goodridge, D.M.; Shorvon, S.D.; Sander, J.W. The long-term risk of premature mortality in people with epilepsy. Brain 2011, 134 Pt 2, 388–395. [Google Scholar] [CrossRef]
- Singhi, P.; Jagirdar, S.; Khandelwal, N.; Malhi, P. Epilepsy in children with cerebral palsy. J. Child Neurol. 2003, 18, 174–179. [Google Scholar] [CrossRef] [PubMed]


| Variable | Total | Rehabilitation ≥ 6 Times within 6 Months | Rehabilitation < 6 Times within 6 Months | p-Value |
|---|---|---|---|---|
| n = 3936 | n = 1211 (30.8%) | n = 2725 (69.2%) | ||
| Age, years (Mean ± SD) | 3.52 ± 2.7 | 3.17 ± 2.2 | 3.68 ± 2.9 | <0.001 |
| Gender-Male | 2342 (60%) | 746 (62%) | 1596 (59%) | 0.074 |
| Mortality rate (3-year follow-up) | 164 (4.2%) | 127 (5%) | 37 (2.6%) | <0.001 |
| Hospital characteristics | ||||
| Medical center | 2424 (62%) | 796 (66%) | 1628 (60%) | |
| Regional | 1281 (32%) | 276 (23%) | 1005 (37%) | |
| Others | 231 (6%) | 139 (11%) | 92 (3%) | |
| Region | <0.001 | |||
| North | 2055 (52%) | 578 (48%) | 1477 (54%) | |
| Middle | 851 (22%) | 298 (24%) | 553 (20%) | |
| South/Others | 1030 (26%) | 335 (28%) | 695 (26%) | |
| Urbanization | 0.203 | |||
| Urban | 2252 (57%) | 680 (56%) | 1572 (57%) | |
| Suburban | 1392 (35%) | 450 (37%) | 942 (35%) | |
| Rural | 292 (8%) | 81 (7%) | 211 (8%) | |
| Comorbidity | ||||
| Inpatient care before diagnosis (≥2 times within 1 year) | 1095 (28%) | 343 (28%) | 752 (28%) | 0.638 |
| Pneumonia | 1437 (37%) | 489 (40%) | 948 (35%) | 0.001 |
| Epilepsy | 1679 (43%) | 580 (48%) | 1099 (40%) | <0.001 |
| Congenital heart disease | 96 (2%) | 37 (2%) | 59 (2%) | 0.581 |
| Perinatal complications | 2160 (55%) | 739 (61%) | 1421 (51%) | <0.001 |
| Scoliosis | 89 (2%) | 35 (3%) | 54 (2%) | 0.077 |
| GERD | 206 (5%) | 89 (7%) | 117 (4%) | 0.001 |
| Dysphagia | 1880 (48%) | 618 (51%) | 1262 (46%) | 0.006 |
| Intellectual disability | 569 (15%) | 221 (18%) | 348 (13%) | <0.001 |
| Variables | Univariate | Multivariate | ||
|---|---|---|---|---|
| HR (95% CI) | p-Value | aHR (95% CI) | p-Value | |
| Age | 0.71 (0.64–0.79) | <0.001 | 0.84 (0.76–0.92) | <0.001 |
| Gender-Male | 1.12 (0.82–1.53) | 0.472 | ||
| Rehabilitation times | ||||
| Low (<6) vs. high (≥6) | 1.55 (1.24–1.95) | <0.001 | 1.96 (1.33–2.89) | <0.001 |
| Hospital characteristics Medical center | ||||
| Regional | 0.73 (0.52–1.04) | 0.079 | ||
| Others | 0.44 (0.18–1.07) | 0.070 | ||
| Region North | ||||
| Middle | 0.69 (0.46–1.06) | 0.089 | ||
| South and Others | 0.84 (0.58–1.21) | 0.345 | ||
| Residential (Rural vs. others) | 1.16 (0.67–2.00) | 0.604 | ||
| Inpatient care before diagnosis (≥2 times within 1 year) | 5.46 (3.94–7.56) | <0.001 | 2.88 (1.96–4.23) | <0.001 |
| Pneumonia | 2.34 (1.72–3.19) | <0.001 | 1.41 (1.00–1.96) | 0.047 |
| Epilepsy | 2.14 (1.56–2.94) | <0.001 | 1.41 (1.02–1.95) | 0.039 |
| Congenital heart disease | 2.06 (1.01–4.18) | 0.047 | ||
| Perinatal complications | 1.37 (0.99–1.88) | 0.052 | ||
| Scoliosis | 0.48 (0.00–4.24) | 0.184 | ||
| GERD | 1.64 (0.93–2.90) | 0.085 | ||
| Dysphagia | 3.21 (2.26–4.56) | <0.001 | 1.55 (1.06–2.26) | 0.024 |
| Intellectual disability | 0.58 (0.34–0.99) | 0.044 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hsu, C.-L.; Hung, C.-L.; Huang, S.-J.; Yin, C.-H.; Chu, C.-H.; Kuo, T.-J.; Hung, Y.-M. Impact of Rehabilitation Intensity on 3-Year Mortality among Children with Moderate to Severe Cerebral Palsy: A Population-Based Cohort Study. Int. J. Environ. Res. Public Health 2021, 18, 9932. https://doi.org/10.3390/ijerph18189932
Hsu C-L, Hung C-L, Huang S-J, Yin C-H, Chu C-H, Kuo T-J, Hung Y-M. Impact of Rehabilitation Intensity on 3-Year Mortality among Children with Moderate to Severe Cerebral Palsy: A Population-Based Cohort Study. International Journal of Environmental Research and Public Health. 2021; 18(18):9932. https://doi.org/10.3390/ijerph18189932
Chicago/Turabian StyleHsu, Chiao-Lin, Chia-Ling Hung, Shih-Ju Huang, Chun-Hao Yin, Chi-Hsiang Chu, Tsu-Jen Kuo, and Yao-Min Hung. 2021. "Impact of Rehabilitation Intensity on 3-Year Mortality among Children with Moderate to Severe Cerebral Palsy: A Population-Based Cohort Study" International Journal of Environmental Research and Public Health 18, no. 18: 9932. https://doi.org/10.3390/ijerph18189932
APA StyleHsu, C.-L., Hung, C.-L., Huang, S.-J., Yin, C.-H., Chu, C.-H., Kuo, T.-J., & Hung, Y.-M. (2021). Impact of Rehabilitation Intensity on 3-Year Mortality among Children with Moderate to Severe Cerebral Palsy: A Population-Based Cohort Study. International Journal of Environmental Research and Public Health, 18(18), 9932. https://doi.org/10.3390/ijerph18189932






