Evaluation of Preventive Treatment Protocols for Patients under Antiresorptive Therapy Undergoing Tooth Extraction at a Swiss University Clinic
Abstract
:1. Introduction
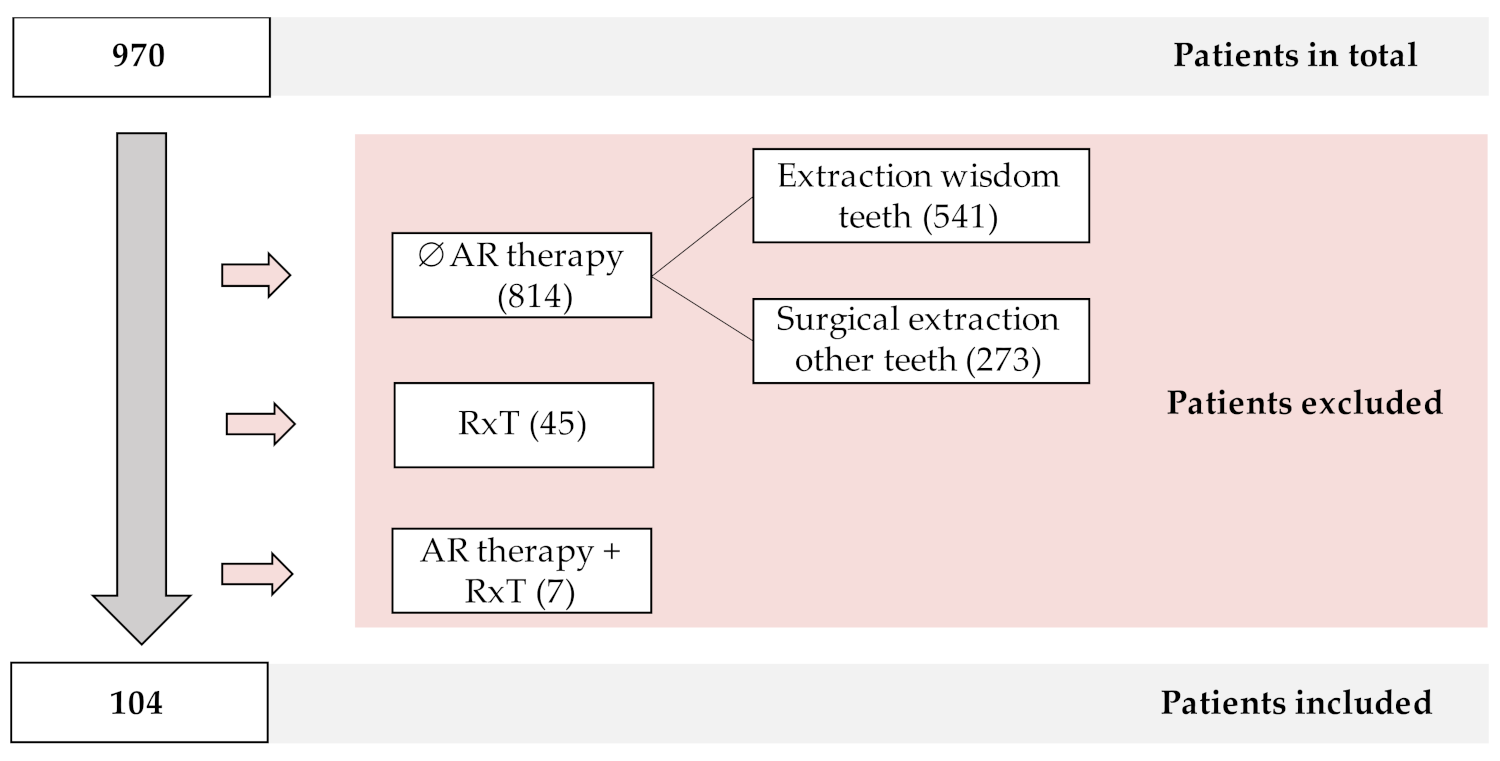
2. Materials and Methods
3. Results
3.1. Characteristics of Study Population and Antiresorptive Medication
3.2. Extraction Number and Site, Surgical Technique, Antibiotic Prophylaxis and Postoperative Inflammatory Complications
3.3. Occurrence of ARONJ
3.4. Characteristics of Patients Presenting with ARONJ
3.4.1. Total ARONJ Patients
3.4.2. Patient with ARONJ after Extraction, at the Same Location, without Previous Signs of ARONJ
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Di Fede, O.; Panzarella, V.; Mauceri, R.; Fusco, V.; Bedogni, A.; Muzio, L.L.; Board, S.O.; Campisi, G. The Dental Management of Patients at Risk of Medication-Related Osteonecrosis of the Jaw: New Paradigm of Primary Prevention. BioMed Res. Int. 2018, 2018, 2684924. [Google Scholar] [CrossRef]
- Ruggiero, S.L.; Dodson, T.B.; Fantasia, J.; Goodday, R.; Aghaloo, T.; Mehrotra, B.; O’Ryan, F. American Association of Oral and Maxillofacial Surgeons Position Paper on Medication-Related Osteonecrosis of the Jaw—2014 Update. J. Oral Maxillofac. Surg. 2014, 72, 1938–1956. [Google Scholar] [CrossRef] [PubMed]
- Otto, S.; Tröltzsch, M.; Jambrovic, V.; Panya, S.; Probst, F.; Ristow, O.; Ehrenfeld, M.; Pautke, C. Tooth extraction in patients receiving oral or intravenous bisphosphonate administration: A trigger for BRONJ development? J. Cranio-Maxillofac. Surg. 2015, 43, 847–854. [Google Scholar] [CrossRef] [PubMed]
- Bas, H. Schweizer Empfehlungen zur Osteoporosebehandlung. Rheumatol. Schmerz 2017, 11–12, 6–10. [Google Scholar]
- Lippuner, K. Epidemiologie und Stellenwert der Osteoporosein der Schweiz. Ther. Umsch. 2012, 69, 137–144. [Google Scholar] [CrossRef]
- Poxleitner, P.; Engelhardt, M.; Schmelzeisen, R.; Voss, P. The Prevention of Medication-related Osteonecrosis of the Jaw. Dtsch. Aerztebl. Int. 2017, 114, 63–69. [Google Scholar] [CrossRef] [Green Version]
- Spanou, A.; Nelson, K.; Ermer, M.A.; Steybe, D.; Poxleitner, P.; Voss, P.J. Primary wound closure and perioperative antibiotic therapy for prevention of bisphosphonate-related osteonecrosis of the jaw after tooth extraction. Quintessence Int. 2020, 51, 220–228. [Google Scholar] [PubMed]
- Mitsimponas, K.T.; Moebius, P.; Amann, K.; Stockmann, P.; Schlegel, K.-A.; Neukam, F.-W.; Wehrhan, F. Osteo-radio-necrosis (ORN) and bisphosphonate-related osteonecrosis of the jaws (BRONJ): The histopathological differences under the clinical similarities. Int. J. Clin. Exp. Pathol. 2014, 7, 496–508. [Google Scholar]
- Lancellotta, V.; Pagano, S.; Tagliaferri, L.; Piergentini, M.; Ricci, A.; Montecchiani, S.; Saldi, S.; Chierchini, S.; Cianetti, S.; Valentini, V.; et al. Individual 3-dimensional printed mold for treating hard palate carcinoma with brachytherapy: A clinical report. J. Prosthet. Dent. 2019, 121, 690–693. [Google Scholar] [CrossRef]
- Nicolatou-Galitis, O.; Papadopoulou, E.; Sarri, T.; Boziari, P.; Karayianni, A.; Kyrtsonis, M.-C.; Repousis, P.; Barbounis, V.; Migliorati, C.A. Osteonecrosis of the jaw in oncology patients treated with bisphosphonates: Prospective experience of a dental oncology referral center. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2011, 112, 195–202. [Google Scholar] [CrossRef]
- Schiodt, M.; Otto, S.; Fedele, S.; Bedogni, A.; Nicolatou-Galitis, O.; Guggenberger, R.; Herlofson, B.B.; Ristow, O.; Kofod, T. Workshop of European task force on medication-related osteonecrosis of the jaw—Current challenges. Oral Dis. 2019, 25, 1815–1821. [Google Scholar] [CrossRef] [Green Version]
- Migliorati, C.A.; Saunders, D.; Conlon, M.S.; Ingstad, H.K.; Vaagen, P.; Palazzolo, M.J.; Herlofson, B.B. Assessing the association between bisphosphonate exposure and delayed mucosal healing after tooth extraction. J. Am. Dent. Assoc. 2013, 144, 406–414. [Google Scholar] [CrossRef]
- Bermúdez-Bejarano, E.-B.; Serrera-Figallo, M.-Á.; Gutiérrez-Corrales, A.; Romero-Ruiz, M.-M.; Castillo-De-Oyagüe, R.; Gutiérrez-Pérez, J.-L.; Torres-Lagares, D. Prophylaxis and antibiotic therapy in management protocols of patients treated with oral and intravenous bisphosphonates. J. Clin. Exp. Dent. 2017, 9, e141–e149. [Google Scholar] [CrossRef]
- Matsumoto, A.; Sasaki, M.; Schmelzeisen, R.; Oyama, Y.; Mori, Y.; Voss, P.J. Primary wound closure after tooth extraction for prevention of medication-related osteonecrosis of the jaw in patients under denosumab. Clin. Oral Investig. 2017, 21, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Poxleitner, P.; Steybe, D.; Kroneberg, P.; Ermer, M.A.; Yalcin-Ülker, G.M.; Schmelzeisen, R.; Voss, P.J. Tooth extractions in patients under antiresorptive therapy for osteoporosis: Primary closure of the extraction socket with a mucoperiosteal flap versus application of platelet-rich fibrin for the prevention of antiresorptive agent-related osteonecrosis of the jaw. J. Cranio-Maxillofac. Surg. 2020, 48, 444–451. [Google Scholar] [CrossRef]
- Wolff, T.F.; Grötz, K.A. Antiresorptiva-assoziierte Kiefernekrosen. Zahnmed. up2date 2017, 11, 23–37. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; Available online: https://www.R-project.org/ (accessed on 28 July 2021).
- Wickham, H.; Averick, M.; Bryan, J.; Chang, W.; McGowan, L.D.; François, R.; Grolemund, G.; Hayes, A.; Henry, L.; Hester, J.; et al. Welcome to the Tidyverse. J. Open Source Softw. 2019, 4, 1686. [Google Scholar] [CrossRef]
- Campisi, G.; Mauceri, R.; Bertoldo, F.; Bettini, G.; Biasotto, M.; Colella, G.; Consolo, U.; Di Fede, O.; Favia, G.; Fusco, V.; et al. Medication-Related Osteonecrosis of Jaws (MRONJ) Prevention and Diagnosis: Italian Consensus Update 2020. Int. J. Environ. Res. Public Health 2020, 17, 5998. [Google Scholar] [CrossRef]
- Kuster, I.; Valdec, S.; Rücker, M.; Bichsel, D. Plastische Deckung von Extraktionsalveolen mit einem xenogenen Bindegewebstransplantat. Swiss Dent. J. 2020, 130, 139–145. [Google Scholar]
- Pippi, R.; Giuliani, U.; Tenore, G.; Pietrantoni, A.; Romeo, U. What is the Risk of Developing Medication-Related Osteonecrosis in Patients With Extraction Sockets Left to Heal by Secondary Intention? A Retrospective Case Series Study. J. Oral Maxillofac. Surg. 2021, in press. [Google Scholar] [CrossRef] [PubMed]
- Heufelder, M.J.; Hendricks, J.; Remmerbach, T.; Frerich, B.; Hemprich, A.; Wilde, F. Principles of oral surgery for prevention of bisphosphonate-related osteonecrosis of the jaw. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2014, 117, e429–e435. [Google Scholar] [CrossRef]
- Ferlito, S.; Puzzo, S.; Liardo, C. Preventive Protocol for Tooth Extractions in Patients Treated With Zoledronate: A Case Series. J. Oral Maxillofac. Surg. 2011, 69, e1–e4. [Google Scholar] [CrossRef] [PubMed]
- Mozzati, M.; Arata, V.; Gallesio, G. Tooth extraction in patients on zoledronic acid therapy. Oral Oncol. 2012, 48, 817–821. [Google Scholar] [CrossRef]
- Madeira, M.; Rocha, A.C.; Moreira, C.A.; Aguiar, Á.M.M.; Maeda, S.S.; Cardoso, A.S.; Castro, C.H.D.M.; D’Alva, C.B.; Silva, B.C.C.; Ferraz-De-Souza, B.; et al. Prevention and treatment of oral adverse effects of antiresorptive medications for osteoporosis—A position paper of the Brazilian Society of Endocrinology and Metabolism (SBEM), Brazilian Society of Stomatology and Oral Pathology (Sobep), and Brazilian Association for Bone Evaluation and Osteometabolism (Abrasso). Arch. Endocrinol. Metab. 2020, 64, 664–672. [Google Scholar] [CrossRef]
- Napeñas, J.J.; Kujan, O.; Arduino, P.G.; Sukumar, S.; Galvin, S.; Baričević, M.; Costella, J.; Czerninski, R.; Peterson, D.E.; Lockhart, P.B. World Workshop on Oral Medicine VI: Controversies regarding dental management of medically complex patients: Assessment of current recommendations. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2015, 120, 207–226. [Google Scholar] [CrossRef] [PubMed]
- Meinen, A.; Reuss, A.; Willrich, N.; Feig, M.; Noll, I.; Eckmanns, T.; Al-Nawas, B.; Markwart, R. Antimicrobial Resistance and the Spectrum of Pathogens in Dental and Oral-Maxillofacial Infections in Hospitals and Dental Practices in Germany. Front. Microbiol. 2021, 12, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Llewelyn, M.J.; Fitzpatrick, J.M.; Darwin, E.; Tonkin-Crine, S.; Gorton, C.; Paul, J.; Peto, T.E.A.; Yardley, L.; Hopkins, S.; Walker, A.S. The antibiotic course has had its day. BMJ 2017, 358, j3418. [Google Scholar] [CrossRef] [Green Version]
- Bornstein, M.; Filippi, A.; Heinzmann, J.L.; Kühl, S.; Stadlinger, B.; Weiss, P. Leitlinie Antibiotika in der Oralchirurgie. Swiss Dent. J. 2020, 130, 916–917. [Google Scholar] [PubMed]
- Blanck, J.; Gaßmann, G.; Antiresorptive Medikamente. ZWP Online, 8 February 2018. Available online: https://www.zwp-online.info/fachgebiete/prophylaxe/grundlagen/antiresorptive-medikamente-die-bedeutung-bei-parodontologischen-therapien (accessed on 18 July 2021).
- Ottesen, C.; Schiodt, M.; Gotfredsen, K. Efficacy of a high-dose antiresorptive drug holiday to reduce the risk of medication-related osteonecrosis of the jaw (MRONJ): A systematic review. Heliyon 2020, 6, e03795. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, T.; Ueda, N.; Yamada, S.; Kato, S.; Iwata, E.; Hayashida, S.; Kojima, Y.; Shinohara, M.; Tojo, I. Denosumab-related osteonecrosis of the jaw after tooth extraction and the effects of a short drug holiday in cancer patients: A multicenter retrospective study. Osteoporos. Int. 2021, in press. [Google Scholar] [CrossRef]
- Malan, J.; Ettinger, K.; Naumann, E.; Beirne, O.R. The relationship of denosumab pharmacology and osteonecrosis of the jaws. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2012, 114, 671–676. [Google Scholar] [CrossRef]
- Bensi, C.; Giovacchini, F.; Lomurno, G.; Eramo, S.; Barraco, G.; Tullio, A. Quality of life in patients affected by medication-related osteonecrosis of the jaws: A systematic review. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2021, 132, 182–189. [Google Scholar] [CrossRef]
- Campisi, G.; Fedele, S.; Fusco, V.; Pizzo, G.; Di Fede, O.; Bedogni, A. Epidemiology, clinical manifestations, risk reduction and treatment strategies of jaw osteonecrosis in cancer patients exposed to antiresorptive agents. Future Oncol. 2014, 10, 257–275. [Google Scholar] [CrossRef] [Green Version]
- Drudge-Coates, L.; Wyngaert, T.V.D.; Schiødt, M.; Van Muilekom, H.A.M.; Demonty, G.; Otto, S. Preventing, identifying, and managing medication-related osteonecrosis of the jaw: A practical guide for nurses and other allied healthcare professionals. Support. Care Cancer 2020, 28, 4019–4029. [Google Scholar] [CrossRef] [PubMed]
- Bruckmoser, E.; Palaoro, M.; Latzko, L.; Schnabl, D.; Neururer, S.; Laimer, J. Choosing the Right Partner for Medication Related Osteonecrosis of the Jaw: What Central European Dentists Know. Int. J. Environ. Res. Public Health 2021, 18, 4466. [Google Scholar] [CrossRef] [PubMed]
- Limones, A.; Sáez-Alcaide, L.; Díaz-Parreño, S.; Helm, A.; Bornstein, M.; Molinero-Mourelle, P. Medication-related osteonecrosis of the jaws (MRONJ) in cancer patients treated with denosumab VS. zoledronic acid: A systematic review and meta-analysis. Med. Oral Patol. Oral Cir. Bucal 2020, 25, e326–e336. [Google Scholar] [CrossRef]
- Borm, J.M.; Moser, S.; Locher, M.; Damerau, G.; Stadlinger, B.; Grätz, K.W.; Jacobsen, C. [Risk assessment in patients undergoing osseous antiresorptive therapy in dentistry. An update]. Schweiz. Mon. Zahnmed. 2013, 123, 985–1001. [Google Scholar]
- Baumann, E.G.; Lambrecht, A.; Thomas, J.; Buser, D.; Grätz, K.W.; Obwegeser, J.; Imfeld, T.; Peltomäki, T.; Gnoinski, W.; Kuttenberger, M.; et al. Bornst, Bundesgesetz über die Kankenversicherung KVG (zahnärztliche Behandlungen), 4. Schweizerische Zahnärzte-Gesellschaft (SSO). 2021. Available online: http://sso-shop.ch/index.php?id=94&L=F72&tx_gishop_pi1%5Bpuid%5D=45 (accessed on 13 September 2021).


| Low-Risk 0–0.5% | Medium-Risk 1% | High-Risk 1–21% | ||||
|---|---|---|---|---|---|---|
| Indication | Primary osteoporosis | Therapy-induced Osteoporosis Prevention of SRE 2 Co-medication with immunomodulators 3 Additional risk factors 4 | Osseous metastasis Multiple myeloma | |||
| Medication/Route of administration | BP (p.o, i.v) 1 | DNO (s.c) 1 | BP (i.v) | BP (i.v) + immunomodulators | BP (i.v) | DNO (s.c) |
| Examples of dosage and frequency of administration every n month (M) or week (W) | Zoledronat 5 mg/12 M Ibandronat 3 mg/3 mL/3 M | 60 mg /6 M | Zoledronat 4 mg/6 M | Zoledronat 4 mg/4 W | 120 mg/4 W | |
| Variables Related to Number of Patients | |||
|---|---|---|---|
| Parameter | Category | Result | Percentage (%) |
| Age | Years (mean ± SD) | 71.54 ± 12.04 | |
| Gender | Male | 26 | 25.00 |
| Female | 78 | 75.00 | |
| Smoking | Yes | 27 | 25.96 |
| No | 77 | 74.04 | |
| Risk profile | Low | 66 | 63.46 |
| Medium | 18 | 17.30 | |
| High | 19 | 18.26 | |
| No information | 1 | 0.96 | |
| Antiresorptive drug/-- Route of administration | Prolia® (subcutaneous) | 38 | 36.53 |
| Xgeva® (subcutaneous) | 9 | 8.65 | |
| Zoledronat | 10 | 9.61 | |
| Intravenous | 9 | 90.00 | |
| Peroral | 0 | 0.00 | |
| No information | 1 | 10.00 | |
| Ibandronat | 28 | 26.92 | |
| Intravenous | 23 | 82.14 | |
| Peroral | 3 | 10.71 | |
| No information | 2 | 7.14 | |
| Alendronat | 13 | 12.50 | |
| Intravenous | 1 | 7.69 | |
| Peroral | 10 | 76.92 | |
| No information | 2 | 15.38 | |
| No information | 6 | 5.76 | |
| Duration antiresorptive therapy | Years (mean ± SD) | 4.08 ± 3.30 | |
| Administration schedule antiresorptive Therapy | Times per Year | ||
| 1 | 8 | 7.69 | |
| 2 | 36 | 34.61 | |
| 3 | 1 | 0.96 | |
| 4 | 21 | 20.19 | |
| 12 | 11 | 10.57 | |
| 52 | 8 | 7.69 | |
| No information | 19 | 18.26 | |
| Indication antiresorptive therapy | Osteoporosis | 67 | 64.42 |
| Osseous metastasis | 14 | 13.46 | |
| Multiple myeloma | 5 | 4.80 | |
| Systemic co-factors | Diabetes | 5 | 4.80 |
| Anemia | 2 | 1.92 | |
| Anemia and diabetes | 1 | 0.96 | |
| Secondary osteoporosis | 3 | 2.88 | |
| Prevention SRE | 1 | 0.96 | |
| Immunomodulators | 6 | 5.76 | |
| Variables related to number of extractions | |||
| Parameter | Category | Result | Percentage (%) |
| Extractions | Total | 203 | |
| Extraction site | Upper jaw | 95 | 46.80 |
| Molar | 43 | 45.26 | |
| Premolar | 31 | 32.63 | |
| Front tooth | 21 | 22.11 | |
| Lower jaw | 108 | 53.20 | |
| Molar | 52 | 48.15 | |
| Premolar | 34 | 31.48 | |
| Front tooth | 22 | 20.37 | |
| Current/previous antiresorptive therapy at time of extraction | Current Previous | 151 52 | 74.38 25.62 |
| Surgical technique wound closure | Mucoperiosteal flap | 190 | 93.59 |
| Xenogenic graft | 4 | 1.97 | |
| Mucosal flap | 2 | 0.98 | |
| No flap | 3 | 1.47 | |
| No information | 4 | 1.97 | |
| Antibiotic | Amoxicillin | 130 | 64.03 |
| Co-amoxicillin | 35 | 17.24 | |
| Clindamycin | 27 | 13.30 | |
| No informatio | 11 | 5.41 | |
| Duration antibiotic | Weeks (mean ± SD) | 2.20 ± 0.79 | |
| Inflammatory complications | Yes | 60 | 29.55 |
| Dehiscence and/or, | 14 | 23.33 | |
| With revision | 9 | 64.28 | |
| No ARONJ | 4 | 44.44 | |
| ARONJ | 5 | 55.56 | |
| Without revision | 5 | 35.71 | |
| No ARONJ | 3 | 60.00 | |
| ARONJ | 2 | 40.00 | |
| Pain and/or, | 27 | 45.00 | |
| Redness and/or, | 11 | 18.33 | |
| Hematoma and/or, | 21 | 35.00 | |
| Swelling | 10 | 16.67 | |
| No | 142 | 69.95 | |
| No information | 1 | 0.49 |
| Localization | Before Extraction | After Extraction | Before and After |
|---|---|---|---|
| Same | 8 | 3 | 3 |
| Different | 9 | 3 | 0 |
| Same and Different | 1 | 1 | 1 |
| Variables Related to Number of Patients | |||
|---|---|---|---|
| Parameter | Category | Result | Percentage |
| Age | Years (mean ± SD 1) | 73.48 ± 13.50 (56.00 ± 7.41 *) | |
| Gender | Male | 4 (2 *) | 19.05 (50.00 *) |
| Female | 17 (2 *) | 80.95 (50.00 *) | |
| Smoking | Yes | 7 (3 *) | 33.33 (75.00 *) |
| No | 14 (1 *) | 66.67 (25.00 *) | |
| Risk profile | Low | 12 (1 *) | 57.14 (25.00 *) |
| Medium | 3 (1 *) | 14.29 (25.00 *) | |
| High | 6 (2 *) | 28.57 (50.00 *) | |
| Antiresorptive drug/ Route of administration | Prolia® (subcutaneous) | 7 (1 *) | 33.33 (25.00 *) |
| Xgeva® (subcutaneous) | 4 (2 *) | 19.04 (50.00 *) | |
| Zoledronat | 2 (0 *) | 9.52 (0.00 *) | |
| Intravenous | 1 (0 *) | 50.00 (0.00 *) | |
| Peroral | 0 (0 *) | 00.00 (0.00 *) | |
| No information 2 | 1 (0 *) | 50.00 (0.00 *) | |
| Ibandronat | 4 (0 *) | 19.04 (0.00 *) | |
| Intravenous | 4 (0 *) | 100.00 (0.00 *) | |
| Peroral | 0 (0 *) | 0.00 (0.00 *) | |
| No information | 0 (0 *) | 0.00 (0.00 *) | |
| Alendronat | 4 (1 *) | 19.04 (25.00 *) | |
| Intravenous | 0 (0 *) | 00.00 (0.00 *) | |
| Peroral | 3 (1 *) | 75.00 (100.00 *) | |
| No information | 1 (0 *) | 25.00 (0.00 *) | |
| Duration antiresorptive therapy | Years (mean ± SD) | 4.85 ± 4.93 (2.00 ± 0.81 *) | |
| Indication antiresorptive therapy | Osteoporosis | 12 (1 *) | 57.14 (25.00 *) |
| Osseous metastasis | 4 (2 *) | 19.04 (50.00 *) | |
| Multiple myeloma | 2 (0 *) | 9.52 (0.00 *) | |
| Systemic co-factors | Diabetes | 1 (0 *) | 4.76 (0.00 *) |
| Anemia | 1 (0 *) | 4.76 (0.00 *) | |
| Anemia and diabetes | 0 (0 *) | 0.00 (0.00 *) | |
| Secondary osteoporosis | 0 (0 *) | 0.00 (0.00 *) | |
| Prevention SRE 3 | 0 (0 *) | 0.00 (0.00 *) | |
| Immunomodulators | 1 (1 *) | 4.76 (25.00 *) | |
| Variables related to number of extractions | |||
| Parameter | Category | Result | Percentage (%) |
| Extractions | Total | 29 (4 *) | 37.93 (75.00 *) |
| Extraction site | Upper jaw | 11 (3 *) | 27.27 (66.67 *) |
| Molar | 3 (2 *) | 45.45 (33.33 *) | |
| Premolar | 5 (1 *) | 27.27 (0.00 *) | |
| Front tooth | 3 (0 *) | 62.07 (25.00 *) | |
| Lower jaw | 18 (1 *) | 33.33 (100.00 *) | |
| Molar | 6 (1 *) | 50.00 (0.00 *) | |
| Premolar | 9 (0 *) | 16.67 (0.00 *) | |
| Front tooth | 3 (0 *) | 37.93 (75.00 *) | |
| Current/previous antiresorptive therapy at time of extraction | Current Previous | 19 (4 *) 10 (0 *) | 65.52 (100.00 *) 34.48 (0.00 *) |
| Inflammatory complications | Yes Dehiscence and/or, Pain and/or, Redness and/or, Hematoma and/or, Swelling No | 9 (4 *) 7 (4 *) 6 (3 *) 3 (1 *) 0 (0 *) 0 (0 *) 20 (0 *) | 31.03 (100.00 *) 77.78 (100.00 *) 66.67 (75.00 *) 33.33 (25.00 *) 0.00 (0.00 *) 0.00 (0.00 *) 68.97 (0.00 *) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pick, E.; Leuenberger, N.; Kuster, I.; Stutzmann, N.S.; Stadlinger, B.; Valdec, S. Evaluation of Preventive Treatment Protocols for Patients under Antiresorptive Therapy Undergoing Tooth Extraction at a Swiss University Clinic. Int. J. Environ. Res. Public Health 2021, 18, 9924. https://doi.org/10.3390/ijerph18189924
Pick E, Leuenberger N, Kuster I, Stutzmann NS, Stadlinger B, Valdec S. Evaluation of Preventive Treatment Protocols for Patients under Antiresorptive Therapy Undergoing Tooth Extraction at a Swiss University Clinic. International Journal of Environmental Research and Public Health. 2021; 18(18):9924. https://doi.org/10.3390/ijerph18189924
Chicago/Turabian StylePick, Ellen, Nicolas Leuenberger, Irina Kuster, Nicole Selina Stutzmann, Bernd Stadlinger, and Silvio Valdec. 2021. "Evaluation of Preventive Treatment Protocols for Patients under Antiresorptive Therapy Undergoing Tooth Extraction at a Swiss University Clinic" International Journal of Environmental Research and Public Health 18, no. 18: 9924. https://doi.org/10.3390/ijerph18189924
APA StylePick, E., Leuenberger, N., Kuster, I., Stutzmann, N. S., Stadlinger, B., & Valdec, S. (2021). Evaluation of Preventive Treatment Protocols for Patients under Antiresorptive Therapy Undergoing Tooth Extraction at a Swiss University Clinic. International Journal of Environmental Research and Public Health, 18(18), 9924. https://doi.org/10.3390/ijerph18189924






