Amino Acids and the Early Mammalian Embryo: Origin, Fate, Function and Life-Long Legacy
Abstract
1. Introduction
2. Origin of Amino Acids Available to Early Embryos
3. Amino Acid Composition of Oviduct Fluids
4. Proteins in Oviduct Fluid; Putative Functions and Regulation
5. Transport of Amino Acids into the Early Embryo
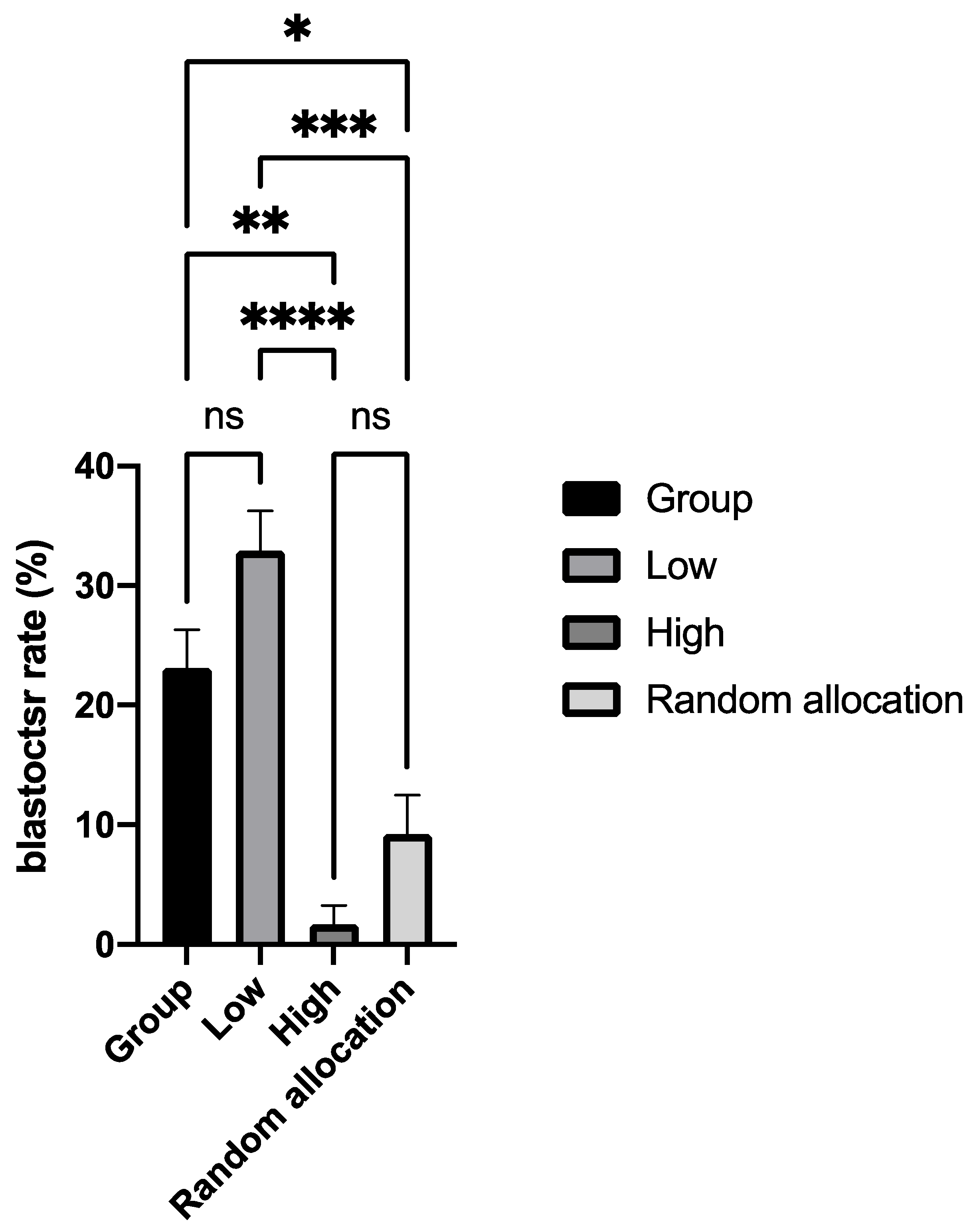
6. Functional Roles of Amino Acids in Preimplantation Development
6.1. Essential and Non-Essential Amino Acids
6.2. The Requirement for Amino Acids of Early Embryos in Culture
7. Metabolic Functions of Amino Acids in the Early Embryo
8. Protein Content of the Preimplantation Embryo
9. Ammonia Production
10. One-Carbon Metabolism and DNA Methylation
11. Functions of Specific Amino Acids in the Early Embryo
11.1. Arginine and Leucine
11.2. Glycine
11.3. Glycine, Taurine and Hypotaurine in Protection against Oxidative Stress
11.4. Methionine
11.5. Glutamine
11.6. The ‘Other’ Amino Acids
12. Amino Acids and the Early Mammalian Embryo: Life-Long Legacy
13. Amino Acid Depletion/Appearance (‘Turnover’); A Robust Marker of Individual Differences at the Cellular Level
14. Diagnostic Relevance of Amino Acid Metabolism
15. Amino Acid Depletion/Appearance; More Is Not Necessarily Better
16. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tarahomi, M.; Vaz, F.M.; Straalen, J.P.; Schrauwen, F.A.P.; Wely, M.V.; Hamer, G.; Repping, S.; Mastenbroek, S. The composition of human preimplantation embryo culture media and their stability during storage and culture. Hum. Reprod. 2019, 34, 1450–1461. [Google Scholar] [CrossRef]
- Harding, J.E. The nutritional basis of the fetal origins of adult disease. Int. J. Epidemiol. 2001, 30, 15–23. [Google Scholar] [CrossRef]
- Leese, H.J. The formation and function of oviduct fluid. J. Reprod. Fertil. 1988, 82, 843–856. [Google Scholar] [CrossRef]
- Leese, H.J. Studies on the movement of glucose, pyruvate and lactate into the ampulla and isthmus of the rabbit oviduct. Q. J. Exp. Physiol. 1983, 68, 89–96. [Google Scholar] [CrossRef]
- Harwalkerm, K.; Ford, M.; Teng, K.; Yamanaka, N.; Yang, B.; Burtscher, I.; LIckert, H.; Yamanaka, Y. Anatomical and cellular heterogeneity in the mouse oviduct—Its potential roles in reproduction and preimplantation development. Biol. Reprod. 2021, 104, 1249–1261. Available online: https://academic.oup.com/biolreprod/advance-article/doi/10.1093/biolre/ioab043/6163942?login=true (accessed on 19 July 2021). [CrossRef] [PubMed]
- Saint-Dizier, M.; Schoen, J.; Chen, S.; Banliat, C.; Mermillod, P. Composing the early embryonic microenvironment: Physiology and regulation of oviductal secretions. Int. J. Mol. Sci. 2019, 21, 223. [Google Scholar] [CrossRef] [PubMed]
- Leese, H.J.; Hugentobler, S.A.; Gray, S.M.; Morris, D.G.; Sturmey, R.; Whitear, S.-L.; Sreenan, J.M. Female reproductive tract fluids: Composition, mechanism of formation and potential role in the developmental origins of health and disease. Reprod. Fertil. Dev. 2008, 20, 1–8. [Google Scholar] [CrossRef]
- Barker, D. The developmental origins of chronic adult disease. Acta Paediatr. 2004, 93, 26–33. Available online: http://doi.wiley.com/10.1111/j.1651-2227.2004.tb00236.x (accessed on 19 July 2021). [CrossRef] [PubMed]
- Bazer, F.W.; Spencer, T.E.; Johnson, G.A.; Burghardt, R.C. Uterine receptivity to implantation of blastocysts in mammals. Front. Biosci. 2011, 3, 745–767. [Google Scholar] [CrossRef] [PubMed]
- Aplin, J.D.; Myers, J.E.; Timms, K.; Westwood, M. Tracking placental development in health and disease. Nat. Rev. Endocrinol. 2020, 16, 479–494. [Google Scholar] [CrossRef]
- Brison, D.R.; Sturmey, R.G.; Leese, H.J. Metabolic heterogeneity during preimplantation development: The missing link? Hum. Reprod. Update 2014, 20, 632–640. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, J.; Reyley, M. The uterine tubal fluid: Secretion, composition and biological effects. Anim. Reprod. 2018, 2, 91–105. [Google Scholar]
- Rodríguez-Alonso, B.; Maillo, V.; Acuña, O.S.; López-úbeda, R.; Torrecillas, A.; Simintiras, C.A.; Sturmey, R.; Avilés, M.; Lonergan, P.; Rizos, D. Spatial and pregnancy-related changes in the protein, amino acid, and carbohydrate composition of bovine oviduct fluid. Int. J. Mol. Sci. 2020, 21, 1681. [Google Scholar] [CrossRef]
- Hugentobler, S.; Humpherson, P.; Leese, H.; Sreenan, J.; Morris, D. Energy substrates in bovine oviduct and uterine fluid and blood plasma during the oestrous cycle. Mol. Reprod. Dev. 2007, 75, 496–503. [Google Scholar] [CrossRef]
- Frayn, K. (Ed.) Metabolic Regulation: A Human Perspective, 3rd ed.; John Wiley & Sons: Chichester, UK, 2010; p. 190. Available online: https://www.wiley.com/en-us/Metabolic+Regulation%3A+A+Human+Perspective%2C+3rd+Edition-p-9781118685334 (accessed on 19 July 2021).
- Kenny, D.A.; Humpherson, P.G.; Leese, H.J.; Morris, D.G.; Tomos, A.D.; Diskin, M.G.; Sreenan, J.M. Effect of elevated systemic concentrations of ammonia and urea on the metabolite and ionic composition of oviductal fluid in cattle. Biol. Reprod. 2002, 66, 1797–1804. [Google Scholar] [CrossRef] [PubMed]
- Hugentobler, S.A.; Diskin, M.G.; Leese, H.J.; Humpherson, P.G.; Watson, T.; Sreenan, J.M.; Morris, D.G. Amino acids in oviduct and uterine fluid and blood plasma during the estrous cycle in the bovine. Mol. Reprod. Dev. Inc. Gamete Res. 2007, 74, 445–454. [Google Scholar] [CrossRef]
- Hill, J.L.; Wade, M.G.; Nancarrow, C.D.; Kelleher, D.L.; Boland, M.P. Influence of ovine oviducal amino acid concentrations and an ovine oestrus-associated glycoprotein on development and viability of bovine embryos. Mol. Reprod. Dev. 1997, 47, 164–169. [Google Scholar] [CrossRef]
- Li, R.; Whitworth, K.; Liangxue, L.; Wax, D.; Spate, L.D.; Murphy, C.; Rieke, A.; Isom, C.; Hao, Y.; Zhong, Z.; et al. Concentration and composition of free amino acids and osmolalities of porcine oviductal and uterine fluid and their effects on development of porcine IVF embryos. Mol. Reprod. Dev. 2007, 74, 1228–1235. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.; Schultz, G.A. Amino acid content of preimplantation rabbit embryos and fluids of the reproductive tract. Biol. Reprod. 1987, 36, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Harris, S.E.; Gopichandran, N.; Picton, H.M.; Leese, H.J.; Orsi, N. Nutrient concentrations in murine follicular fluid and the female reproductive tract. Theriogenology 2005, 64, 992–1006. [Google Scholar] [CrossRef] [PubMed]
- Devreker, F.; Van den Bergh, M.; Biramane, J.; Winston, R.L.; Englert, Y.; Hardy, K. Effects of taurine on human embryo development in vitro. Hum. Reprod. 1999, 14, 2350–2356. [Google Scholar] [CrossRef]
- Van Winkle, L.J.; Patel, M.; Wasserlauf, H.G.; Dickinson, H.R.; Campione, A.L. Osmotic regulation of taurine transport via system β and novel processes in mouse preimplantation conceptuses. Biochim. Biophys. Acta Biomembr. 1994, 1191, 244–255. [Google Scholar] [CrossRef]
- Guérin, P.; Guillaud, J.; Ménézo, Y. Andrology: Hypotaurine in spermatozoa and genital secretions and its production by oviduct epithelial cells in vitro. Hum. Reprod. 1995, 10, 866–872. [Google Scholar] [CrossRef] [PubMed]
- Casslén, B.G. Free amino acids in human uterine fluid. Possible role of high taurine concentration. J. Reprod. Med. 1987, 32, 181–184. [Google Scholar] [PubMed]
- Ménézo, Y.; Elder, K. Epigenetic remodeling of chromatin in human ART: Addressing deficiencies in culture media. J. Assist. Reprod. Genet. 2020, 37, 1781–1788. [Google Scholar] [CrossRef]
- Bianchi, E.; Sun, Y.; Almansa-Ordonez, A.; Woods, M.; Goulding, D.; Martinez-Martin, N.; Wright, G.J. Control of oviductal fluid flow by the G-protein coupled receptor Adgrd1 is essential for murine embryo transit. Nat. Commun. 2021, 12, 1251. [Google Scholar] [CrossRef]
- Ménézo, Y.; Guérin, P.; Elder, K. The oviduct: A neglected organ due for re-assessment in IVF. Reprod. Biomed. Online 2015, 30, 233–240. [Google Scholar] [CrossRef]
- Ghersevich, S.; Massa, E.; Zumoffen, C. Oviductal secretion and gamete interaction. Reproduction 2015, 149, R1–R14. [Google Scholar] [CrossRef]
- Almiñana, C.; Tsikis, G.; Labas, V.; Uzbekov, R.; da Silveira, J.C.; Bauersachs, S.; Mermillod, P. Deciphering the oviductal extracellular vesicles content across the estrous cycle: Implications for the gametes-oviduct interactions and the environment of the potential embryo. BMC Genom. 2018, 19, 622. [Google Scholar] [CrossRef]
- Alminana-Brines, C.; Corbin, E.; Tsikis, G.; Neto, A.S.D.A.; Labas, V.; Reynaud, K.; Galio, L.; Uzbekov, R.; Garanina, A.; Druart, X.; et al. Oviduct extracellular vesicles protein content and their role during oviduct-embryo cross-talk. Reproduction 2017, 154, 253–268. [Google Scholar] [CrossRef] [PubMed]
- Kölle, S.; Hughes, B.; Steele, H. Early embryo-maternal communication in the oviduct: A review. Mol. Reprod. Dev. 2020, 87, 650–662. [Google Scholar] [CrossRef] [PubMed]
- Moura, M.T.; Latoracca, L.B.; Paula-Lopes, F.F. Contextualising autophagy during gametogenesis and preimplantation embryonic development. Int. J. Mol. Sci. 2021, 22, 6313. [Google Scholar] [CrossRef]
- Van Winkle, L.J. Amino acid transport regulation and early embryo development. Biol. Reprod. 2001, 64, 1–12. [Google Scholar] [CrossRef]
- Martin, P.M.; Sutherland, A.E.; Van Winkle, L.J. Amino acid transport regulates blastocyst implantation. Biol. Reprod. 2003, 69, 1101–1108. [Google Scholar] [CrossRef]
- Van Winkle, L.J.; Tesch, J.K.; Shah, A.; Campione, A.L. System B0,+ amino acid transport regulates the penetration stage of blastocyst implantation with possible long-term developmental consequences through adulthood. Hum. Reprod. Update 2006, 12, 145–157. [Google Scholar] [CrossRef]
- Bazer, F.W.; Johnson, G.A.; Wu, G. Amino acids and conceptus development during the peri-implantation period of pregnancy. Cell Signal. Dur. Mamm. Early Embryo Dev. 2015, 843, 23–52. [Google Scholar] [CrossRef]
- Fleming, T.P.; Kwong, W.Y.; Porter, R.; Ursell, E.; Fesenko, I.; Wilkins, A.; Miller, D.J.; Watkins, A.; Eckert, J.J. The Embryo and Its Future1. Biol. Reprod. 2004, 71, 1046–1054. [Google Scholar] [CrossRef] [PubMed]
- Sturmey, R.G.; Brison, D.R.; Leese, H.J. Assessing embryo viability by measurement of amino acid turnover. Reprod. Biomed. Online 2008, 17, 486–496. [Google Scholar] [CrossRef]
- Picton, H.M.; Elder, K.; Houghton, F.; Hawkhead, J.A.; Rutherford, A.J.; Hogg, J.E.; Leese, H.J.; Harris, S.E. Association between amino acid turnover and chromosome aneuploidy during human preimplantation embryo development in vitro. Mol. Hum. Reprod. 2010, 16, 557–569. [Google Scholar] [CrossRef]
- Epstein, C.J.; Smith, S.A. Amino acid uptake and protein synthesis in preimplantation mouse embryos. Dev. Biol. 1973, 33, 171–184. [Google Scholar] [CrossRef]
- Takahashi, Y.; First, N. In vitro development of bovine one-cell embryos: Influence of glucose, lactate, pyruvate, amino acids and vitamins. Theriogenology 1992, 37, 963–978. [Google Scholar] [CrossRef]
- Partridge, R.; Leese, H. Consumption of amino acids by bovine preimplantation embryos. Reprod. Fertil. Dev. 1996, 8, 945–950. [Google Scholar] [CrossRef]
- Baltz, J.M. Intracellular pH regulation in the early embryo. BioEssays 1993, 15, 523–530. [Google Scholar] [CrossRef]
- Baltz, J.M. Osmoregulation and cell volume regulation in the preimplantation embryo. Curr. Top. Dev. Biol. 2001, 52, 55–106. [Google Scholar] [CrossRef]
- Menezo, Y.J.; Silvestris, E.; Dale, B.; Elder, K. Oxidative stress and alterations in DNA methylation: Two sides of the same coin in reproduction. Reprod. Biomed. Online 2016, 33, 668–683. [Google Scholar] [CrossRef] [PubMed]
- Ho, Y.; Wigglesworth, K.; Eppig, J.J.; Schultz, R.M. Preimplantation development of mouse embryos in KSOM: Augmentation by amino acids and analysis of gene expression. Mol. Reprod. Dev. 1995, 41, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Summers, M.C.; Biggers, J.D. Chemically defined media and the culture of mammalian preimplantation embryos: Historical perspective and current issues. Hum. Reprod. Update 2003, 9, 557–582. [Google Scholar] [CrossRef]
- Rose, W.C. The nutritive significance of the amino acids. Physiol. Rev. 1938, 18, 109–136. [Google Scholar] [CrossRef]
- Eagle, H. Amino acid metabolism in mammalian cell cultures. Science 1959, 130, 432–437. [Google Scholar] [CrossRef]
- Maddy, K.H.; Elvehjem, C. Studies on growth of mice fed rations containing free amino acids. J. Biol. Chem. 1949, 177, 577–590. [Google Scholar] [CrossRef]
- Brinster, R.L. Studies on the development of mouse embyrosin vitro. III. The effect of fixed-nitrogen source. J. Exp. Zool. 1965, 158, 69–77. [Google Scholar] [CrossRef]
- Kane, M.; Foote, R.H. Culture of two- and four-cell rabbit embryos to the expanding blastocyst stage in synthetic media. Exp. Biol. Med. 1970, 133, 921–925. [Google Scholar] [CrossRef]
- Biggers, J.D. Pioneering mammalian embryo culture. In Mamm Preimplantation Embryo; Springer: Boston, MA, USA, 1987; pp. 1–22. Available online: http://link.springer.com/10.1007/978-1-4684-5332-4_1 (accessed on 19 July 2021).
- Kane, M.T. Culture of preimplantation rabbit embryos. In Comparative Embryo Culture; Humana: New York, NY, USA, 2019; pp. 63–91. Available online: http://link.springer.com/10.1007/978-1-4939-9566-0_5 (accessed on 19 July 2021).
- Kane, M.T. Historical note: Lessons learned from developing the first semi-defined medium for culture of one-cell rabbit embryos to expanding blastocysts. In Comparative Embryo Culture; Humana: New York, NY, USA, 2019; pp. 1–12. Available online: http://link.springer.com/10.1007/978-1-4939-9566-0_1 (accessed on 19 July 2021).
- Bavister, B.D.; Leibfried, M.L.; Lieberman, G. Development of preimplantation embryos of the golden hamster in a defined culture medium. Biol. Reprod. 1983, 28, 235–247. [Google Scholar] [CrossRef] [PubMed]
- Trounson, A.; Pushett, D.; MacLellan, L.; Lewis, I.; Gardner, D. Current status of IVM/IVF and embryo culture in humans and farm animals. Theriogenology 1994, 41, 57–66. [Google Scholar] [CrossRef]
- Thompson, J. In vitro culture and embryo metabolism of cattle and sheep embryos—A decade of achievement. Anim. Reprod. Sci. 2000, 60, 263–275. [Google Scholar] [CrossRef]
- Devreker, F.; Hardy, K.; Van den Bergh, M.; Vannin, A.S.; Emiliani, S.; Englert, Y. Amino acids promote human blastocyst development in vitro. Hum. Reprod. 2001, 16, 749–756. [Google Scholar] [CrossRef]
- Leese, H.J.; Conaghan, J.; Martin, K.L.; Hardy, K. Early human embryo metabolism. BioEssays 1993, 15, 259–264. [Google Scholar] [CrossRef]
- Leese, H.J.; Houghton, F.D.; Macmillan, D.A.; Donnay, I. Metabolism of the early embryo: Energy production and utilization. In ART and the Human Blastocyst; Springer: New York, NY, USA, 2001; pp. 61–68. Available online: http://link.springer.com/10.1007/978-1-4613-0149-3_6 (accessed on 19 July 2021).
- Brinster, R.L. Protein content of the mouse embryo during the first five days of development. Reproduction 1967, 13, 413–420. [Google Scholar] [CrossRef]
- Sellens, M.H.; Stein, S.; Sherman, M.I. Protein and free amino acid content in preimplantation mouse embryos and in blastocysts under various culture conditions. Reproduction 1981, 61, 307–315. [Google Scholar] [CrossRef]
- Schiffner, J.; Spielmann, H. Fluorometric assay of the protein content of mouse and rat embryos during preimplantation development. Reproduction 1976, 47, 145–147. [Google Scholar] [CrossRef]
- Thompson, J.G.; Sherman, A.N.M.; Allen, N.W.; McGowan, L.T.; Tervit, H.R. Total protein content and protein synthesis within pre-elongation stage bovine embryos. Mol. Reprod. Dev. 1998, 50, 139–145. [Google Scholar] [CrossRef]
- Turner, K.; Goldstein, D.; Rogers, A. Variation in the dry mass of mouse embryos throughout the preimplantation period. Hum. Reprod. 1992, 7, 112–116. [Google Scholar] [CrossRef]
- Orsi, N.M.; Leese, H.J. Ammonium exposure and pyruvate affect the amino acid metabolism of bovine blastocysts in vitro. Reproduction 2004, 127, 131–140. [Google Scholar] [CrossRef]
- Donnay, I.; Leese, H. Embryo metabolism during the expansion of the bovine blastocyst. Mol. Reprod. Dev. 1999, 53, 171–178. [Google Scholar] [CrossRef]
- Biggers, J.D.; McGinnis, L.; A Lawitts, J. Enhanced effect of glycyl-L-glutamine on mouse preimplantation embryos in vitro. Reprod. Biomed. Online 2004, 9, 59–69. [Google Scholar] [CrossRef]
- Kim, S.J.; Koo, O.J.; Kwon, D.K.; Kang, J.-T.; Park, S.J.; Gomez, M.N.; Atikuzzaman, M.; Jang, G.; Lee, B.-C. Replacement of glutamine with the dipeptide derivative alanyl-glutamine enhances in vitro maturation of porcine oocytes and development of embryos. Zygote 2013, 22, 286–289. [Google Scholar] [CrossRef]
- Motiei, M.; Vaculikova, K.; Cela, A.; Tvrdonova, K.; Khalili, R.; Rumpik, D.; Rumpikova, T.; Glatz, Z.; Saha, T. Non-invasive human embryo metabolic assessment as a developmental criterion. J. Clin. Med. 2020, 9, 4094. [Google Scholar] [CrossRef] [PubMed]
- Lucas, E. Epigenetic effects on the embryo as a result of periconceptional environment and assisted reproduction technology. Reprod. Biomed. Online 2013, 27, 477–485. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Diehl, K.L.; Muir, T.W. Chromatin as a key consumer in the metabolite economy. Nat. Chem. Biol. 2020, 16, 620–629. [Google Scholar] [CrossRef]
- Fuks, F. DNA methylation and histone modifications: Teaming up to silence genes. Curr. Opin. Genet. Dev. 2005, 15, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Davuluri, R.V.; Grosse, I.; Zhang, M.Q. Computational identification of promoters and first exons in the human genome. Nat. Genet. 2001, 29, 412–417. [Google Scholar] [CrossRef]
- Takai, D.; Jones, P.A. Comprehensive analysis of CpG islands in human chromosomes 21 and 22. Proc. Natl. Acad. Sci. USA 2002, 99, 3740–3745. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.A. Functions of DNA methylation: Islands, start sites, gene bodies and beyond. Nat. Rev. Genet. 2012, 13, 484–492. [Google Scholar] [CrossRef]
- Guo, H.; Zhu, P.; Yan, L.; Li, R.; Hu, B.; Lian, Y.; Yan, J.; Ren, X.; Lin, S.; Li, J.; et al. The DNA methylation landscape of human early embryos. Nature 2014, 511, 606–610. [Google Scholar] [CrossRef] [PubMed]
- Barres, R.; Zierath, J.R. DNA methylation in metabolic disorders. Am. J. Clin. Nutr. 2011, 93, 897S–900S. [Google Scholar] [CrossRef] [PubMed]
- Milagro, F.; Gómez-Abellán, P.; Campion, J.; Martinez, J.A.; Ordovas, J.; Garaulet, M. CLOCK, PER2 and BMAL1 DNA methylation: Association with obesity and metabolic syndrome characteristics and monounsaturated fat intake. Chrono. Int. 2012, 29, 1180–1194. [Google Scholar] [CrossRef]
- Carless, M.A.; Kulkarni, H.; Kos, M.Z.; Charlesworth, J.; Peralta, J.M.; Göring, H.H.H.; Curran, J.E.; Almasy, L.; Dyer, T.D.; Comuzzie, A.G.; et al. Genetic effects on DNA methylation and its potential relevance for obesity in mexican Americans. PLoS ONE 2013, 8, e73950. [Google Scholar] [CrossRef]
- Kaelin, W.G.; McKnight, S.L. Influence of metabolism on epigenetics and disease. Cell 2013, 153, 56–69. [Google Scholar] [CrossRef]
- Clare, C.; Brassington, A.H.; Kwong, W.Y.; Sinclair, K.D. One-carbon metabolism: Linking nutritional biochemistry to epigenetic programming of long-term development. Annu. Rev. Anim. Biosci. 2019, 7, 263–287. [Google Scholar] [CrossRef]
- Watkins, A.J.; Ursell, E.; Panton, R.; Papenbrock, T.; Hollis, L.; Cunningham, C.; Wilkins, A.; Perry, V.H.; Sheth, B.; Kwong, W.Y.; et al. Adaptive responses by mouse early embryos to maternal diet protect fetal growth but predispose to adult onset disease. Biol. Reprod. 2008, 78, 299–306. Available online: http://www.ncbi.nlm.nih.gov/pubmed/17989357 (accessed on 19 July 2021). [CrossRef]
- Lillycrop, K.; Phillips, E.S.; Jackson, A.A.; Hanson, M.; Burdge, G.C. Dietary protein restriction of pregnant rats induces and folic acid supplementation prevents epigenetic modification of hepatic gene expression in the offspring. J. Nutr. 2005, 135, 1382–1386. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, K.D.; Allegrucci, C.; Singh, R.; Gardner, D.S.; Sebastian, S.; Bispham, J.; Thurston, A.; Huntley, J.F.; Rees, W.D.; Maloney, C.; et al. DNA methylation, insulin resistance, and blood pressure in offspring determined by maternal periconceptional B vitamin and methionine status. Proc. Natl. Acad. Sci. USA 2007, 104, 19351–19356. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, I.M.; Martin, P.M.; Burdsal, C.; Sloan, J.L.; Mager, S.; Harris, T.; Sutherland, A.E. Leucine and arginine regulate trophoblast motility through mTOR-dependent and independent pathways in the preimplantation mouse embryo. Dev. Biol. 2012, 361, 286–300. [Google Scholar] [CrossRef] [PubMed]
- Bröer, S.; Bröer, A. Amino acid homeostasis and signalling in mammalian cells and organisms. Biochem. J. 2017, 474, 1935–1963. [Google Scholar] [CrossRef]
- Milanesi, R.; Coccetti, P.; Tripodi, F. The regulatory role of key metabolites in the control of cell signaling. Biomolecules 2020, 10, 862. [Google Scholar] [CrossRef]
- Manser, R.; Leese, H.; Houghton, F. Effect of inhibiting nitric oxide production on mouse preimplantation embryo development and metabolism1. Biol. Reprod. 2004, 71, 528–533. [Google Scholar] [CrossRef]
- Leese, H.J. Quiet please, do not disturb: A hypothesis of embryo metabolism and viability. BioEssays 2002, 24, 845–849. [Google Scholar] [CrossRef]
- Leese, H.J.; Guerif, F.; Allgar, V.; Brison, D.R.; Lundin, K.; Sturmey, R.G. Biological optimization, the goldilocks principle, and how much is lagom in the preimplantation embryo. Mol. Reprod. Dev. 2016, 83, 748–754. [Google Scholar] [CrossRef]
- Hsu, C.N.; Tain, Y.L. The good, the bad, and the ugly of pregnancy nutrients and developmental programming of adult disease. Nutrients 2019, 11, 894. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, W.K.; Phillips, B.; Hill, I.; Greenhaff, P.; Lund, J.; Williams, J.P.; Rankin, D.; Wilkinson, D.J.; Smith, K.; Atherton, P.J. Human skeletal muscle is refractory to the anabolic effects of leucine during the postprandial muscle-full period in older men. Clin. Sci. 2017, 131, 2643–2653. [Google Scholar] [CrossRef]
- Ispoglou, T.; Deighton, K.; King, R.F.; White, H.; Lees, M. Novel essential amino acid supplements enriched with L-leucine facilitate increased protein and energy intakes in older women: A randomised controlled trial. Nutr. J. 2017, 16, 75. [Google Scholar] [CrossRef] [PubMed]
- Houghton, F.D.; Hawkhead, J.A.; Humpherson, P.G.; Hogg, J.E.; Balen, A.H.; Rutherford, A.J.; Leese, H.J. Non-invasive amino acid turnover predicts human embryo developmental capacity. Hum. Reprod. 2002, 17, 999–1005. [Google Scholar] [CrossRef] [PubMed]
- Eckert, J.J.; Houghton, F.D.; Hawkhead, J.A.; Balen, A.H.; Leese, H.J.; Picton, H.M.; Cameron, I.T.; Fleming, T.P. Human embryos developing in vitro are susceptible to impaired epithelial junction biogenesis correlating with abnormal metabolic activity. Hum. Reprod. 2007, 22, 2214–2224. [Google Scholar] [CrossRef][Green Version]
- Stokes, P.J.; Hawkhead, J.A.; Fawthrop, R.K.; Picton, H.M.; Sharma, V.; Leese, H.J.; Houghton, F.D. Metabolism of human embryos following cryopreservation: Implications for the safety and selection of embryos for transfer in clinical IVF. Hum. Reprod. 2006, 22, 829–835. [Google Scholar] [CrossRef]
- Martin, P.M.; Sutherland, A.E. Exogenous amino acids regulate trophectoderm differentiation in the mouse blastocyst through an mTOR-dependent pathway. Dev. Biol. 2001, 240, 182–193. [Google Scholar] [CrossRef]
- Baltz, J.M.; Zhou, C. Cell volume regulation in mammalian oocytes and preimplantation embryos. Mol. Reprod. Dev. 2012, 79, 821–831. [Google Scholar] [CrossRef]
- Gardner, D.K.; Phil, D.; Vella, P.; Lane, M.; Wagley, L.; Schlenker, T. Culture and transfer of human blastocysts increases implantation rates and reduces the need for multiple embryo transfers. Fertil. Steril. 1998, 69, 84–88. [Google Scholar] [CrossRef]
- Morbeck, D.E.; Krisher, R.L.; Herrick, J.; Baumann, N.A.; Matern, D.; Moyer, T. Composition of commercial media used for human embryo culture. Fertil. Steril. 2014, 102, 759–766.e9. [Google Scholar] [CrossRef]
- Huntriss, J.; Balen, A.; Sinclair, K.; Brison, D.R.; Picton, H.M. Epigenetics and reproductive medicine. BJOG Int. J. Obstet. Gynaecol. 2018, 125, e43–e54. [Google Scholar] [CrossRef] [PubMed]
- Canovas, S.; Ross, P.J.; Kelsey, G.; Coy, P. DNA methylation in embryo development: Epigenetic impact of ART (assisted reproductive technologies). BioEssays 2017, 39, 1700106. [Google Scholar] [CrossRef] [PubMed]
- Salilew-Wondim, D.; Saeed-Zidane, M.; Hoelker, M.; Gebremedhn, S.; Poirier, M.; Pandey, H.O.; Tholen, E.; Neuhoff, C.; Held, E.; Besenfelder, U.; et al. Genome-wide DNA methylation patterns of bovine blastocysts derived from in vivo embryos subjected to in vitro culture before, during or after embryonic genome activation. BMC Genom. 2018, 19, 424. [Google Scholar] [CrossRef]
- Sun, H.; Kang, J.; Su, J.; Zhang, J.; Zhang, L.; Liu, X.; Zhang, J.; Wang, F.; Lu, Z.; Xing, X.; et al. Methionine adenosyltransferase 2A regulates mouse zygotic genome activation and morula to blastocyst transition. Biol. Reprod. 2019, 100, 601–617. [Google Scholar] [CrossRef]
- Clare, C.; Pestinger, V.; Kwong, W.; Tutt, D.; Xu, J.; Byrne, H.; Barrett, D.; Emes, R.; Sinclair, K. Interspecific variation in one-carbon metabolism within the ovarian follicle, oocyte, and preimplantation embryo: Consequences for epigenetic programming of DNA methylation. Int. J. Mol. Sci. 2021, 22, 1838. [Google Scholar] [CrossRef]
- Devreker, F.; Winston, R.M.; Hardy, K. Glutamine improves human preimplantation development in vitro. Fertil. Steril. 1998, 69, 293–299. [Google Scholar] [CrossRef]
- Rieger, D. Relationships between energy metabolism and development of early mammalian embryos. Theriogenology 1992, 37, 75–93. [Google Scholar] [CrossRef]
- Alexiou, M.; Leese, H.J. Purine utilisation, de novo synthesis and degradation in mouse preimplantation embryos. Development 1992, 114, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Forsey, K.E.; Ellis, P.J.; Sargent, C.A.; Sturmey, R.G.; Leese, H.J. Expression and localization of creatine kinase in the preimplantation embryo. Mol. Reprod. Dev. 2013, 80, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Atkins, J.F.; Gesteland, R.F. The twenty-first amino acid. Nature 2000, 407, 463–464. [Google Scholar] [CrossRef]
- Tareq, K.M.A.; Akter, Q.S.; Khandoker, M.A.M.Y.; Tsujii, H. Selenium and vitamin E improve the in vitro maturation, fertilization and culture to blastocyst of porcine oocytes. J. Reprod. Dev. 2012, 58, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Coskun, P.; Wyrembak, J.; Schriner, S.E.; Chen, H.-W.; Marciniack, C.; LaFerla, F.; Wallace, D.C. A mitochondrial etiology of alzheimer and parkinson disease. Biochim. Biophys. Acta Gen. Subj. 2012, 1820, 553–564. [Google Scholar] [CrossRef]
- Young, M.J. Off-target effects of drugs that disrupt human mitochondrial DNA maintenance. Front. Mol. Biosci. 2017, 4, 74. [Google Scholar] [CrossRef]
- Rabiet, M.-J.; Huet, E.; Boulay, F. Human mitochondria-derivedN-formylated peptides are novel agonists equally active on FPR and FPRL1, whileListeria monocytogenes-derived peptides preferentially activate FPR. Eur. J. Immunol. 2005, 35, 2486–2495. [Google Scholar] [CrossRef]
- Barker, D.J.P. The effect of nutrition of the fetus and neonate on cardiovascular disease in adult life. Proc. Nutr. Soc. 1992, 51, 135–144. [Google Scholar] [CrossRef]
- Eckert, J.J.; Porter, R.; Watkins, A.J.; Burt, E.; Brooks, S.; Leese, H.J.; Humpherson, P.G.; Cameron, I.T.; Fleming, T.P. Metabolic induction and early responses of mouse blastocyst developmental programming following maternal low protein diet affecting life-long health. PLoS ONE 2012, 7, e52791. [Google Scholar]
- Velazquez, M.A.; Sheth, B.; Smith, S.J.; Eckert, J.J.; Osmond, C.; Fleming, T.P. Insulin and branched-chain amino acid depletion during mouse preimplantation embryo culture programmes body weight gain and raised blood pressure during early postnatal life. Biochim. Biophys. Acta Mol. Basis Dis. 2018, 1864, 590–600. [Google Scholar] [CrossRef] [PubMed]
- Fleming, T.P.; Watkins, A.J.; Velazquez, M.A.; Mathers, J.C.; Prentice, A.M.; Stephenson, J.; Barker, M.; Saffery, R.; Yajnik, C.S.; Eckert, J.J.; et al. Origins of lifetime health around the time of conception: Causes and consequences. Lancet 2018, 391, 1842–1852. [Google Scholar] [CrossRef]
- Chiumia, D.; Hankele, A.-K.; Drews, B.; Zehnder, T.; Berard, J.; Giesbertz, P.; Ulbrich, S.E.; Giller, K. Alpine and lowland grazing differentially alter the reproductive tract redox milieu and amino acid composition in cattle. Anim. Reprod. Sci. 2019, 213, 106268. [Google Scholar] [CrossRef]
- Sturmey, R.G.; Bermejo-Alvarez, P.; Gutierrez-Adan, A.; Rizos, D.; Leese, H.J.; Lonergan, P. Amino acid metabolism of bovine blastocysts: A biomarker of sex and viability. Mol. Reprod. Dev. 2010, 77, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Gardner, D.K.; Wale, P.L. Analysis of metabolism to select viable human embryos for transfer. Fertil. Steril. 2013, 99, 1062–1072. [Google Scholar] [CrossRef]
- Brison, D.; Houghton, F.; Falconer, D.; Roberts, S.; Hawkhead, J.; Humpherson, P.; Lieberman, B.; Leese, H. Identification of viable embryos in IVF by non-invasive measurement of amino acid turnover. Hum. Reprod. 2004, 19, 2319–2324. [Google Scholar] [CrossRef]
- Hemmings, K.E.; Leese, H.J.; Picton, H.M. Amino acid turnover by bovine oocytes provides an index of oocyte developmental competence in vitro1. Biol. Reprod. 2012, 86, 165. [Google Scholar] [CrossRef]
- Thompson, J.G.; Partridge, R.J.; Houghton, F.D.; Cox, C.I.; Leese, H.J. Oxygen uptake and carbohydrate metabolism by in vitro derived bovine embryos. Reproduction 1996, 106, 299–306. [Google Scholar] [CrossRef]
- Booth, P.J.; Humpherson, P.G.; Watson, T.J.; Leese, H.J. Amino acid depletion and appearance during porcine preimplantation embryo development in vitro. Reproduction 2005, 130, 655–668. [Google Scholar] [CrossRef] [PubMed]
- Humpherson, P.; Leese, H.; Sturmey, R. Amino acid metabolism of the porcine blastocyst. Theriogenology 2005, 64, 1852–1866. [Google Scholar] [CrossRef] [PubMed]
- Lamb, V.K.; Leese, H.J. Uptake of a mixture of amino acids by mouse blastocysts. Reproduction 1994, 102, 169–175. [Google Scholar] [CrossRef]
- Orsi, N.; Leese, H.J. Amino acid metabolism of preimplantation bovine embryos cultured with bovine serum albumin or polyvinyl alcohol. Theriogenology 2003, 61, 561–572. [Google Scholar] [CrossRef]
- Sunde, A.; Brison, D.; Dumoulin, J.; Harper, J.; Lundin, K.; Magli, M.C.; Van den Abbeel, E.; Veiga, A. Time to take human embryo culture seriously. Hum. Reprod. 2016, 31, 2174–2182. [Google Scholar] [CrossRef]
- Sturmey, R.G.; Hawkhead, J.A.; Barker, E.A.; Leese, H.J. DNA damage and metabolic activity in the preimplantation embryo. HumReprod. 2009, 24, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Leese, H.J.; Sathyapalan, T.; Allgar, V.; Brison, D.R.; Sturmey, R. Going to extremes: The Goldilocks/Lagom principle and data distribution. BMJ Open. 2019, 9, e027767. [Google Scholar] [CrossRef]
- Leary, C.; Smith, D.G.; Leese, H.J.; Sturmey, R.G. Amino acid turnover as a biomarker of embryo viability. In Practical Manual of In Vitro Fertilization; Springer: New York, NY, USA, 2012; pp. 431–438. Available online: http://link.springer.com/10.1007/978-1-4419-1780-5_47 (accessed on 19 July 2021).
- Leese, H.J.; Baumann, C.G.; Brison, D.R.; McEvoy, T.G.; Sturmey, R.G. Metabolism of the viable mammalian embryo: Quietness revisited. Mol. Hum. Reprod. 2008, 14, 667–672. [Google Scholar] [CrossRef]
- Stuart, T.; Satija, R. Integrative single-cell analysis. Nat. Rev. Genet. 2019, 20, 257–272. [Google Scholar] [CrossRef] [PubMed]
- Leese, H.J. Metabolism of the preimplantation embryo: 40 years on. Reproduction 2012, 143, 417–427. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leese, H.J.; McKeegan, P.J.; Sturmey, R.G. Amino Acids and the Early Mammalian Embryo: Origin, Fate, Function and Life-Long Legacy. Int. J. Environ. Res. Public Health 2021, 18, 9874. https://doi.org/10.3390/ijerph18189874
Leese HJ, McKeegan PJ, Sturmey RG. Amino Acids and the Early Mammalian Embryo: Origin, Fate, Function and Life-Long Legacy. International Journal of Environmental Research and Public Health. 2021; 18(18):9874. https://doi.org/10.3390/ijerph18189874
Chicago/Turabian StyleLeese, Henry J., Paul J. McKeegan, and Roger G. Sturmey. 2021. "Amino Acids and the Early Mammalian Embryo: Origin, Fate, Function and Life-Long Legacy" International Journal of Environmental Research and Public Health 18, no. 18: 9874. https://doi.org/10.3390/ijerph18189874
APA StyleLeese, H. J., McKeegan, P. J., & Sturmey, R. G. (2021). Amino Acids and the Early Mammalian Embryo: Origin, Fate, Function and Life-Long Legacy. International Journal of Environmental Research and Public Health, 18(18), 9874. https://doi.org/10.3390/ijerph18189874





