Temporal Geospatial Analysis of COVID-19 Pre-Infection Determinants of Risk in South Carolina
Abstract
:1. Introduction
2. Materials and Methods
2.1. Model Selection
2.2. Data Sources
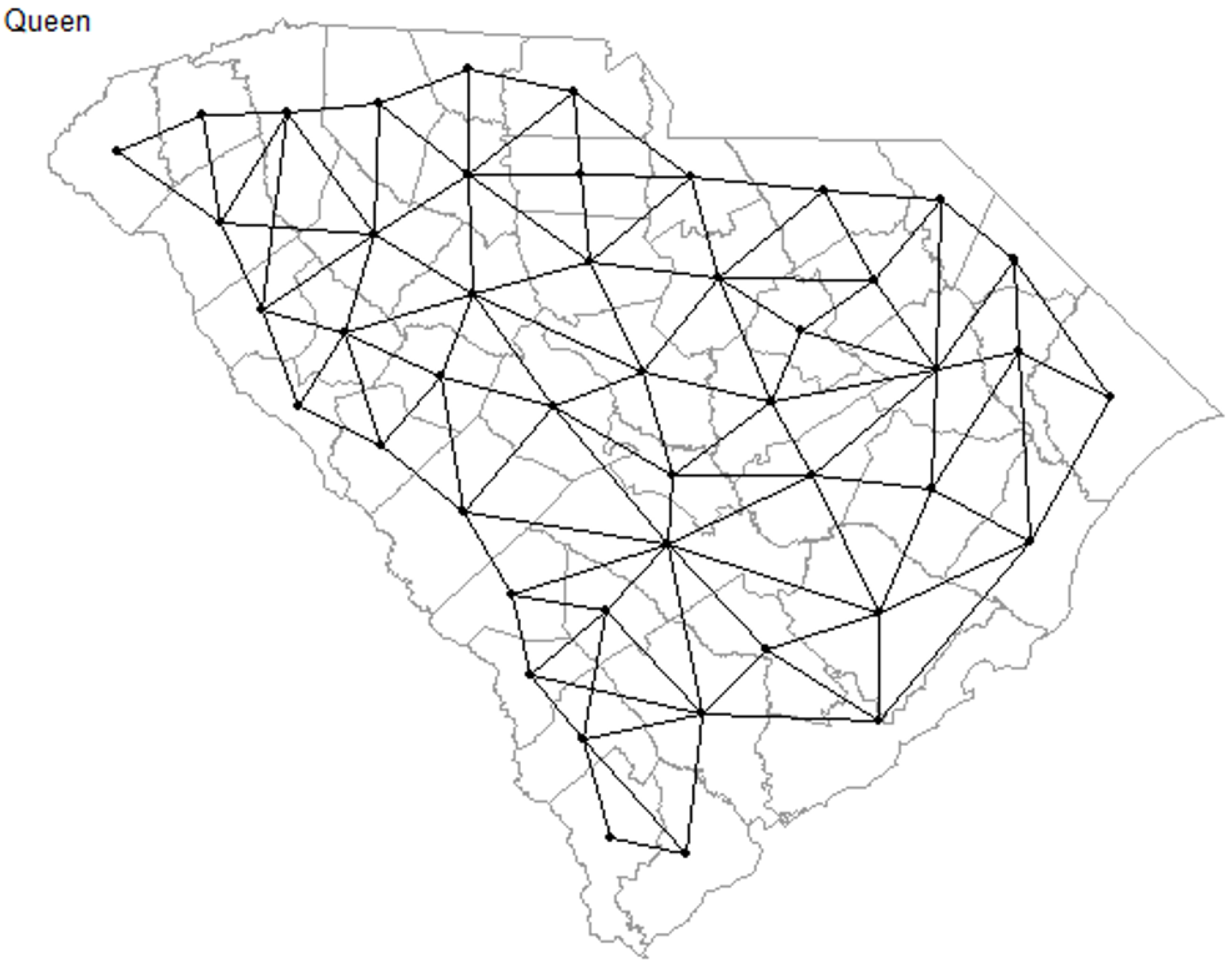
2.3. Spatial Regression Models
3. Results
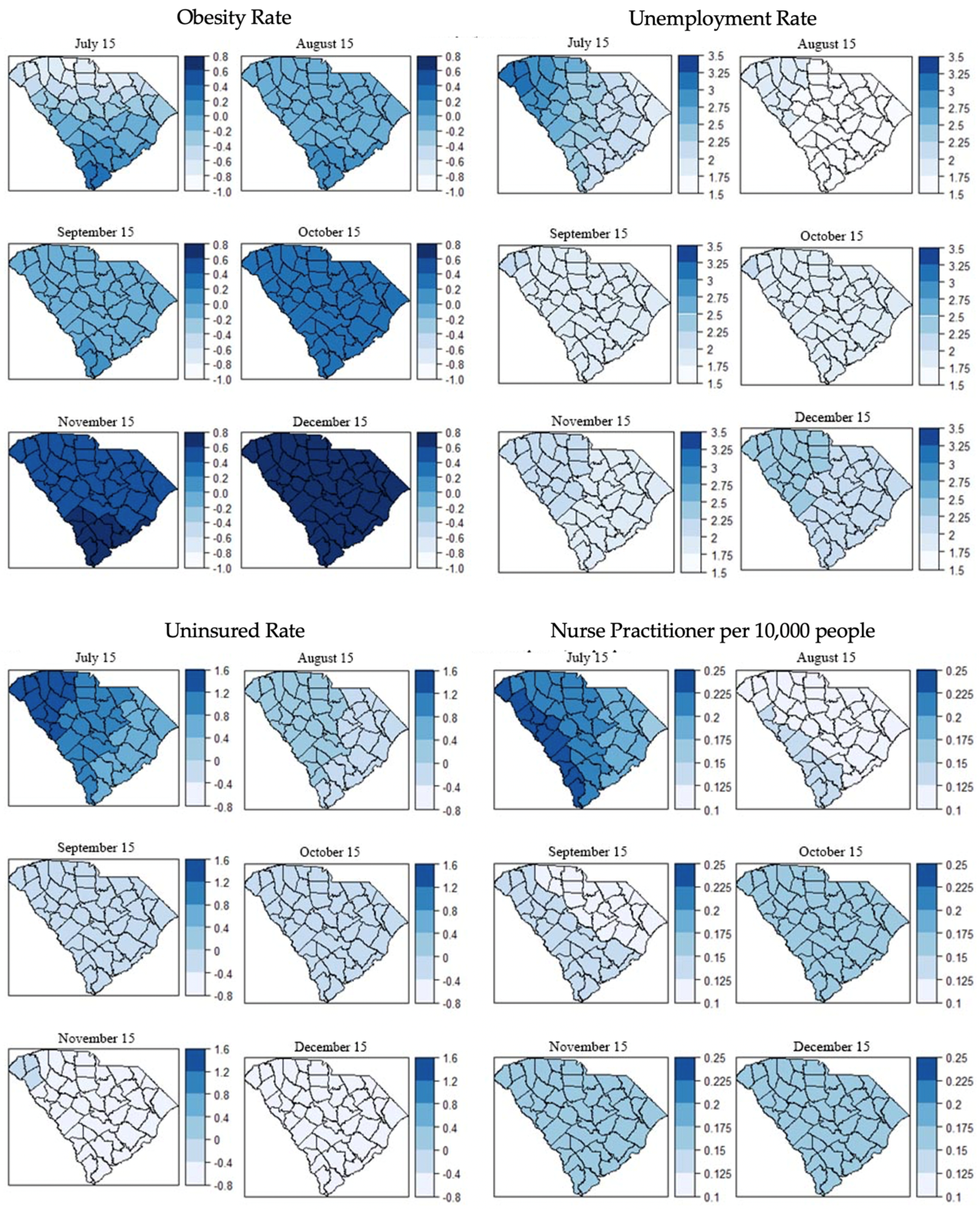
3.1. Distribution of COVID-19 Cases and Covariates
3.2. Global Models for Spatial Correlation
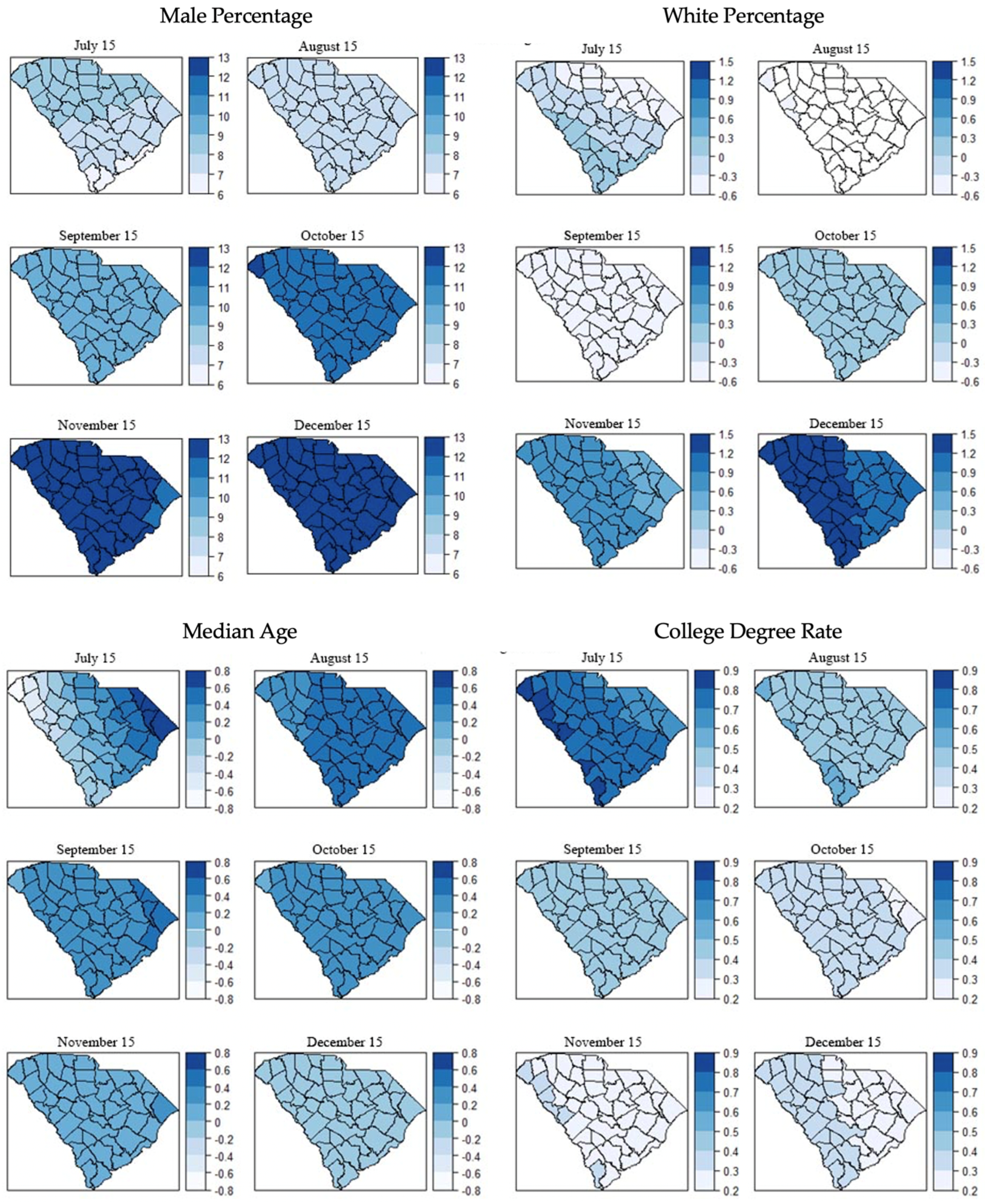
3.3. Local Models for Spatial Correlation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ahmad, F.B.; Cisewski, J.A.; Miniño, A.; Anderson, R.N.J.M. Provisional mortality data—United States, 2020. Morb. Mortal. Wkly. Rep. 2021, 70, 519. [Google Scholar] [CrossRef]
- USAFacts South Carolina Coronavirus Cases and Deaths. Available online: https://usafacts.org/visualizations/coronavirus-covid-19-spread-map/state/south-carolina (accessed on 29 January 2021).
- USAFacts US Coronavirus Cases and Deaths. Available online: https://usafacts.org/visualizations/coronavirus-covid-19-spread-map/ (accessed on 29 January 2021).
- BEA Gross Domestic Product, 4th Quarter and Year 2020 (Advance Estimate). Available online: https://www.bea.gov/news/2021/gross-domestic-product-4th-quarter-and-year-2020-advance-estimate (accessed on 30 January 2021).
- Walmsley, T.; Rose, A.; Wei, D. The Impacts of the Coronavirus on the Economy of the United States. Econ. Disasters Clim. Chang. 2021, 5, 1–52. [Google Scholar] [CrossRef]
- Dangermond, J.; De Vito, C.; Pesaresi, C.J.J. Using GIS in the Time of the COVID-19 Crisis, casting a glance at the future. A joint discussion. J-READING J. Res. Didat. Geogr. 2020, 1, 195–205. [Google Scholar]
- Chen, T.; Wu, D.; Chen, H.; Yan, W.; Yang, D.; Chen, G.; Ma, K.; Xu, D.; Yu, H.; Wang, H.; et al. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: Retrospective study. BMJ 2020, 368, m1091. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.; Xu, S.; Yu, M.; Wang, K.; Tao, Y.; Zhou, Y.; Shi, J.; Zhou, M.; Wu, B.; Yang, Z.; et al. Risk factors for severity and mortality in adult COVID-19 inpatients in Wuhan. J. Allergy Clin. Immunol. 2020, 146, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Williamson, E.J.; Walker, A.J.; Bhaskaran, K.; Bacon, S.; Bates, C.; Morton, C.E.; Curtis, H.J.; Mehrkar, A.; Evans, D.; Inglesby, P.; et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature 2020, 584, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; McGoogan, J.M. Characteristics of and Important Lessons from the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases from the Chinese Center for Disease Control and Prevention. JAMA 2020, 323, 1239–1242. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Price-Haywood, E.G.; Burton, J.; Fort, D.; Seoane, L. Hospitalization and Mortality among Black Patients and White Patients with Covid-19. N. Engl. J. Med. 2020, 382, 2534–2543. [Google Scholar] [CrossRef]
- Park, J.J.; Decloedt, E.H.; Rayner, C.R.; Cotton, M.; Mills, E.J. Clinical trials of disease stages in COVID 19: Complicated and often misinterpreted. Lancet Glob. Health 2020, 8, e1249–e1250. [Google Scholar] [CrossRef]
- Paul, A.; Englert, P.; Varga, M. Socio-economic disparities and COVID-19 in the USA. medRxiv 2020, 2. [Google Scholar] [CrossRef]
- de Lusignan, S.; Dorward, J.; Correa, A.; Jones, N.; Akinyemi, O.; Amirthalingam, G.; Andrews, N.; Byford, R.; Dabrera, G.; Elliot, A.; et al. Risk factors for SARS-CoV-2 among patients in the Oxford Royal College of General Practitioners Research and Surveillance Centre primary care network: A cross-sectional study. Lancet Infect. Dis. 2020, 20, 1034–1042. [Google Scholar] [CrossRef]
- Hawkins, R.B.; Charles, E.J.; Mehaffey, J.H. Socio-economic status and COVID-19–related cases and fatalities. Public Health 2020, 189, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Lewis, N.M.; Friedrichs, M.; Wagstaff, S.; Sage, K.; LaCross, N.; Bui, D.; McCaffrey, K.; Barbeau, B.; George, A.; Rose, C.J.M.; et al. Disparities in COVID-19 incidence, hospitalizations, and testing, by area-level deprivation—Utah, March 3–July 9 2020. Morb. Mortal. Wkly. Rep. 2020, 69, 1369. [Google Scholar] [CrossRef] [PubMed]
- Papageorge, N.W.; Zahn, M.V.; Belot, M.; van den Broek-Altenburg, E.; Choi, S.; Jamison, J.C.; Tripodi, E. Socio-demographic factors associated with self-protecting behavior during the Covid-19 pandemic. J. Popul. Econ. 2021, 34, 691–738. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Li, Q.; Du, X.; Zuo, D.; Ding, Y.; Tan, X.; Liu, Q. Health Behavior Toward COVID-19: The Role of Demographic Factors, Knowledge, and Attitude Among Chinese College Students During the Quarantine Period. Asia Pac. J. Public Health 2020, 32, 533–535. [Google Scholar] [CrossRef] [PubMed]
- NIH Research Opportunity Announcement OTA-21-015B: Post-Acute Sequelae of SARS-CoV-2 Infection Initiative: SARS-CoV-2 Recovery Cohort Studies. Available online: https://covid19.nih.gov/sites/default/files/2021-02/PASC-ROA-OTA-Recovery-Cohort-Studies.pdf (accessed on 15 July 2021).
- Waltenburg, M.A.; Victoroff, T.; Rose, C.E.; Butterfield, M.; Jervis, R.H.; Fedak, K.M.; Gabel, J.A.; Feldpausch, A.; Dunne, E.M.; Austin, C.J.M.; et al. Update: COVID-19 among workers in meat and poultry processing facilities―United States, April–May 2020. Morb. Mortal. Wkly. Rep. 2020, 69, 887. [Google Scholar] [CrossRef] [PubMed]
- Al-Aly, Z.; Xie, Y.; Bowe, B. High-dimensional characterization of post-acute sequelae of COVID-19. Nature 2021, 594, 259–264. [Google Scholar] [CrossRef]
- Deer, R.R.; Rock, M.A.; Vasilevsky, N.; Carmody, L.; Rando, H.; Anzalone, A.J.; Callahan, T.J.; Bramante, C.T.; Chute, C.G.; Greene, C.S.; et al. Characterizing Long COVID: Deep Phenotype of a Complex Condition. medRxiv 2021. [Google Scholar] [CrossRef]
- Ostfeld, R.S.; Glass, G.E.; Keesing, F. Spatial epidemiology: An emerging (or re-emerging) discipline. Trends Ecol. Evol. 2005, 20, 328–336. [Google Scholar] [CrossRef]
- Baker, S.; Holt, K.E.; Clements, A.C.A.; Karkey, A.; Arjyal, A.; Boni, M.F.; Dongol, S.; Hammond, N.; Koirala, S.; Duy, P.T.; et al. Combined high-resolution genotyping and geospatial analysis reveals modes of endemic urban typhoid fever transmission. Open Biol. 2011, 1, 110008. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Linard, C.; Tatem, A.J. Large-scale spatial population databases in infectious disease research. Int. J. Health Geogr. 2012, 11, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tatem, A.J.; Adamo, S.; Bharti, N.; Burgert, C.R.; Castro, M.; Dorelien, A.; Fink, G.; Linard, C.; John, M.; Montana, L.; et al. Mapping populations at risk: Improving spatial demographic data for infectious disease modeling and metric derivation. Popul. Health Metr. 2012, 10, 8. [Google Scholar] [CrossRef] [PubMed]
- Biggs, E.N.; Maloney, P.M.; Rung, A.L.; Peters, E.S.; Robinson, W.T. The Relationship between Social Vulnerability and COVID-19 Incidence among Louisiana Census Tracts. Front. Public Health 2021, 8, 1048. [Google Scholar] [CrossRef]
- Fortaleza, C.M.C.B.; Guimarães, R.B.; de Almeida, G.B.; Pronunciate, M.; Ferreira, C.P. Taking the inner route: Spatial and demographic factors affecting vulnerability to COVID-19 among 604 cities from inner São Paulo State, Brazil. Epidemiol. Infect. 2020, 148, e118. [Google Scholar] [CrossRef] [PubMed]
- Turner, N.A.; Pan, W.; Martinez-Bianchi, V.S.; Panayotti, G.M.M.; Planey, A.M.; Woods, C.W.; Lantos, P.M. Racial, Ethnic, and Geographic Disparities in Novel Coronavirus (Severe Acute Respiratory Syndrome Coronavirus 2) Test Positivity in North Carolina. Open Forum Infect. Dis. 2020, 8, ofaa413. [Google Scholar] [CrossRef]
- Zhang, C.H.; Schwartz, G.G. Spatial Disparities in Coronavirus Incidence and Mortality in the United States: An Ecological Analysis as of May 2020. J. Rural. Health 2020, 36, 433–445. [Google Scholar] [CrossRef]
- Martins-Filho, P.R. Relationship between population density and COVID-19 incidence and mortality estimates: A county-level analysis. J. Infect. Public Health 2021, 14, 1087–1088. [Google Scholar] [CrossRef]
- Mollalo, A.; Vahedi, B.; Rivera, K.M. GIS-based spatial modeling of COVID-19 incidence rate in the continental United States. Sci. Total. Environ. 2020, 728, 138884. [Google Scholar] [CrossRef]
- Qiu, Y.; Chen, X.; Shi, W. Impacts of social and economic factors on the transmission of coronavirus disease 2019 (COVID-19) in China. J. Popul. Econ. 2020, 33, 1127–1172. [Google Scholar] [CrossRef]
- Sannigrahi, S.; Pilla, F.; Basu, B.; Basu, A.S.; Molter, A. Examining the association between socio-demographic composition and COVID-19 fatalities in the European region using spatial regression approach. Sustain. Cities Soc. 2020, 62, 102418. [Google Scholar] [CrossRef] [PubMed]
- Mintzer, A.; Mallory, L. FULL LIST: Face Mask Ordinances in Place across SC. WIS NEWS. 2020. Available online: https://www.wistv.com/2020/07/08/full-list-face-mask-ordinances-place-across-sc/ (accessed on 13 September 2021).
- CDC People at Increased Risk. Available online: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/index.html (accessed on 26 January 2021).
- Cutter, S.L. Vulnerability to environmental hazards. Prog. Hum. Geogr. 1996, 20, 529–539. [Google Scholar] [CrossRef]
- Snyder, B.F.; Parks, V. Spatial variation in socio-ecological vulnerability to Covid-19 in the contiguous United States. Health Place 2020, 66, 102471. [Google Scholar] [CrossRef] [PubMed]
- Wisner, B.; Blaikie, P.; Blaikie, P.M.; Cannon, T.; Davis, I. At Risk: Natural Hazards, People's Vulnerability and Disasters; Psychology Press: New York, NY, USA, 2004. [Google Scholar]
- USAFacts Detailed Methodology and Sources: COVID-19 Data. Available online: https://usafacts.org/articles/detailed-methodology-covid-19-data/ (accessed on 28 August 2021).
- Brunsdon, C.; Fotheringham, S.; Charlton, M. Geographically Weighted Regression. J. R. Stat. Soc. Ser. D 1998, 47, 431–443. [Google Scholar] [CrossRef]
- Bwire, G.M. Coronavirus: Why Men are More Vulnerable to Covid-19 than Women? SN Compr. Clin. Med. 2020, 2, 874–876. [Google Scholar] [CrossRef]
- Li, G.; He, X.; Zhang, L.; Ran, Q.; Wang, J.; Xiong, A.; Wu, D.; Chen, F.; Sun, J.; Chang, C. Assessing ACE2 expression patterns in lung tissues in the pathogenesis of COVID-19. J. Autoimmun. 2020, 112, 102463. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhao, Z.; Wang, Y.; Zhou, Y.; Ma, Y.; Zuo, W. Single-Cell RNA Expression Profiling of ACE2, the Receptor of SARS-CoV-2. Am. J. Respir. Crit. Care Med. 2020, 202, 756–759. [Google Scholar] [CrossRef]
- Ghazeeri, G.; Abdullah, L.; Abbas, O. Immunological differences in women compared with men: Overview and contributing factors. Am. J. Reprod. Immunol. 2011, 66, 163–169. [Google Scholar] [CrossRef]
- Klein, S.L.; Flanagan, K.L. Sex differences in immune responses. Nat. Rev. Immunol. 2016, 16, 626–638. [Google Scholar] [CrossRef]
- de la Vega, R.; Ruíz-Barquín, R.; Boros, S.; Szabo, A. Could attitudes toward COVID-19 in Spain render men more vulnerable than women? Glob. Public Health 2020, 15, 1278–1291. [Google Scholar] [CrossRef]
- Selden, T.M.; Berdahl, T.A. COVID-19 and Racial/Ethnic Disparities In Health Risk, Employment, And Household Composition. Health Aff. 2020, 39, 1624–1632. [Google Scholar] [CrossRef]
- Moehring, K.; Weiland, A.; Reifenscheid, M.; Naumann, E.; Wenz, A.; Rettig, T.; Krieger, U.; Fikel, M.; Cornesse, C.; Blom, A.G. Inequality in Employment Trajectories and Their Socio-Economic Consequences during the Early Phase of the COVID-19 Pandemic in Germany. SocArXiv 2021. Available online: https://osf.io/preprints/socarxiv/m95df/ (accessed on 13 September 2021).
- Johnson, D.P.; Ravi, N.; Braneon, C.V. Spatiotemporal Associations Between Social Vulnerability, Environmental Measurements, and COVID-19 in the Conterminous United States. GeoHealth 2021, 5, e2021GH000423. [Google Scholar] [CrossRef]
- USDA Rural-Urban Continuum Codes—Ag Data Commons. Available online: https://data.nal.usda.gov/dataset/rural-urban-continuum-codes (accessed on 17 July 2021).
- USCB American Community Survey (ACS). Available online: https://www.census.gov/programs-surveys/acs (accessed on 17 July 2021).
- Cunningham, G.B.; Wigfall, L.T. Race, explicit racial attitudes, implicit racial attitudes, and COVID-19 cases and deaths: An analysis of counties in the United States. PLoS ONE 2020, 15, e0242044. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, S.M.; Bryant, A.S.J.O.; Clinics, G. Racial and ethnic disparities in health and health care. medRxiv 2017, 44, 1–11. [Google Scholar] [CrossRef]
- CDC People at Increased Risk and Other People Who Need to Take Extra Precautions. Available online: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/index.html (accessed on 15 July 2021).
- Brandt, K.; Goel, V.; Keeler, C.; Bell, G.J.; Aiello, A.E.; Corbie-Smith, G.; Wilson, E.; Fleischauer, A.; Emch, M.; Boyce, R.M. SARS-CoV-2 testing in North Carolina: Racial, ethnic, and geographic disparities. Health Place 2021, 69, 102576. [Google Scholar] [CrossRef] [PubMed]
- Oates, G.R.; Juarez, L.D.; Horswell, R.; Chu, S.; Miele, L.; Fouad, M.N.; Curry, W.A.; Fort, D.; Hillegass, W.B.; Danos, D.M. The Association Between Neighborhood Social Vulnerability and COVID-19 Testing, Positivity, and Incidence in Alabama and Louisiana. J. Community Health 2021, 7, 1–9. [Google Scholar]
- Seto, E.; Min, E.; Ingram, C.; Cummings, B.; Farquhar, S.A. Community-Level Factors Associated with COVID-19 Cases and Testing Equity in King County, Washington. Int. J. Environ. Res. Public Health 2020, 17, 9516. [Google Scholar] [CrossRef] [PubMed]
- Kamel Boulos, M.N.; Geraghty, E.M. Geographical tracking and mapping of coronavirus disease COVID-19/severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) epidemic and associated events around the world: How 21st century GIS technologies are supporting the global fight against outbreaks and epidemics. Int. J. Health Geogr. 2020, 19, 8. [Google Scholar]
- CDC CDC/ATSDR Social Vulnerability Index. Available online: https://www.atsdr.cdc.gov/placeandhealth/svi/index.html (accessed on 15 July 2021).
- Karaye, I.M.; Horney, J.A. The Impact of Social Vulnerability on COVID-19 in the U.S.: An Analysis of Spatially Varying Relationships. Am. J. Prev. Med. 2020, 59, 317–325. [Google Scholar] [CrossRef]
- Neelon, B.; Mutiso, F.; Mueller, N.T.; Pearce, J.L.; Benjamin-Neelon, S.E.J.P. Spatial and temporal trends in social vulnerability and COVID-19 incidence and death rates in the United States. PLoS ONE 2021, 16, e0248702. [Google Scholar] [CrossRef]
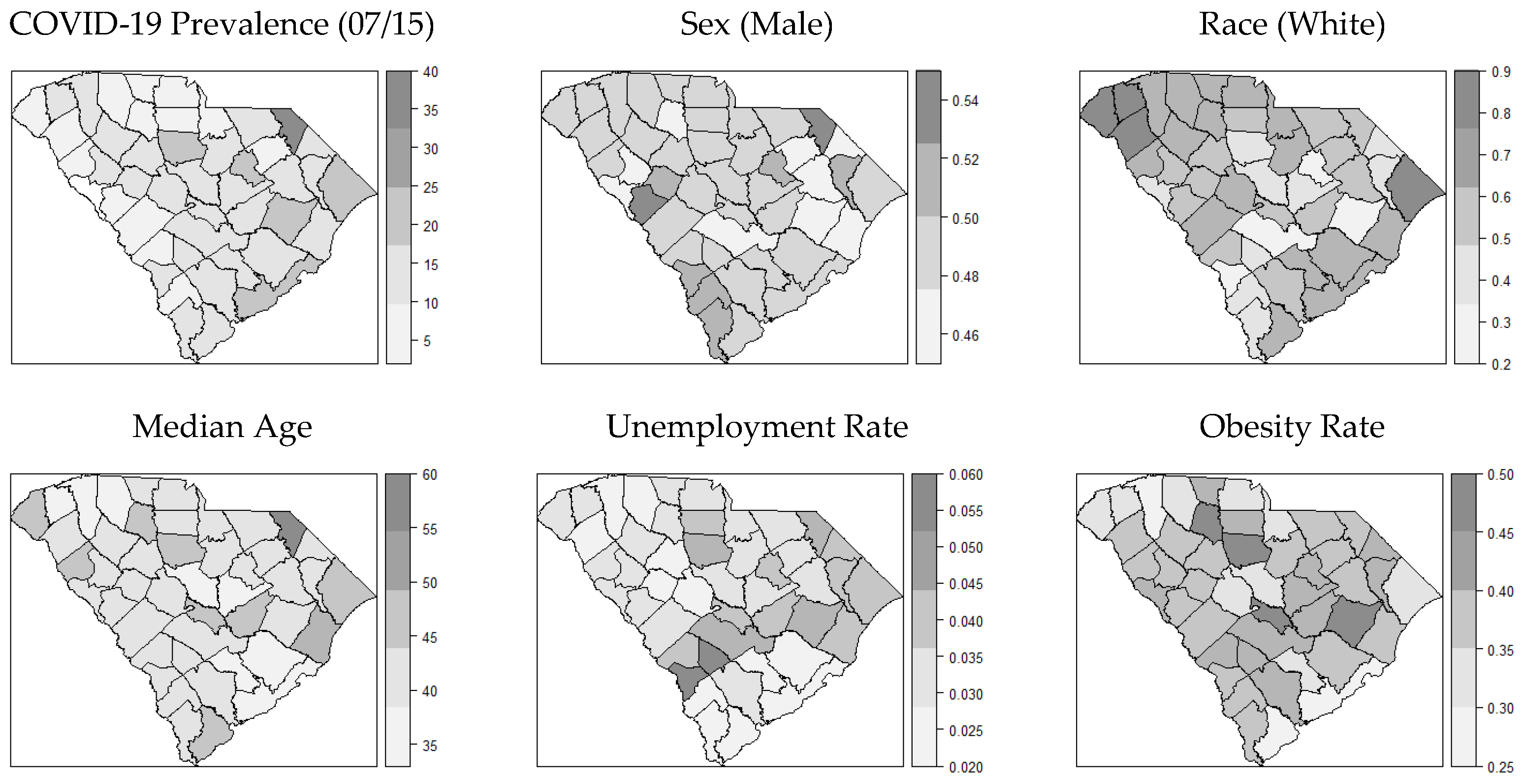
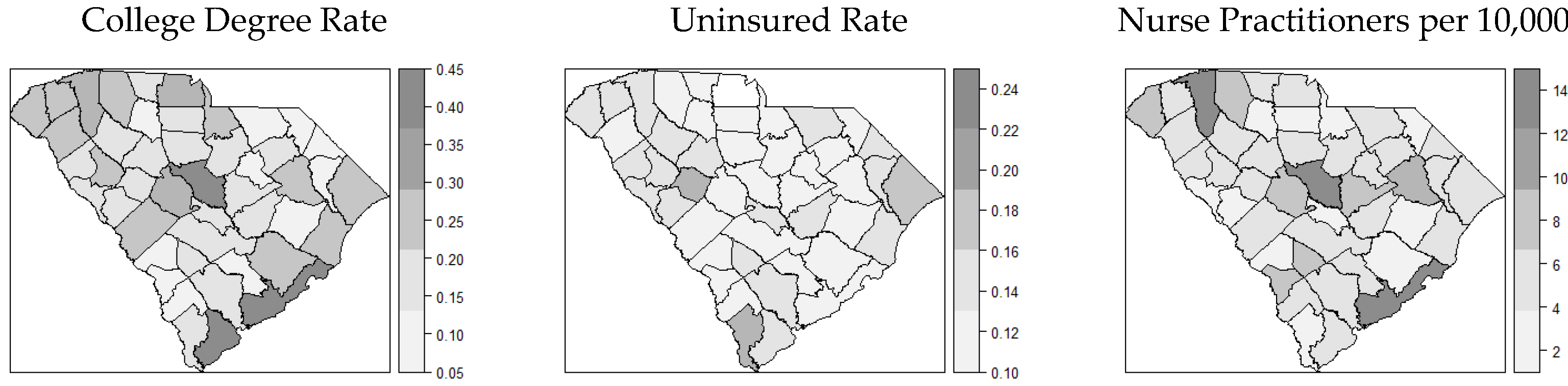
| Theme | Variable | Definition |
|---|---|---|
| Ecological | Age | Median age |
| Sex | Percentage of male population to the total population | |
| Population density | Population per square mile | |
| Social | Uninsured rate | Percentage of population under 65 years old without health insurance |
| High school degree rate | Percentage of population with a high school or higher degree | |
| College degree rate | Percentage of population with a college or higher degree | |
| Race | Percentage of white population to the total population | |
| Medical doctor abundance | The number of medical doctors per 10,000 people | |
| Nurse practitioner abundance | The number of nurse practitioners per 10,000 people | |
| Health | Obesity rate | Percentage of obese population (i.e., individuals whose BMI is 30 or higher) |
| Physical inactivity rate | Percentage of population not engaging in physical activity regularly | |
| Smoking prevalence | Percentage of population who smoke cigarettes regularly (i.e., have smoked at least 100 cigarettes in their life and currently smoke at least one cigarette a day) | |
| Economic | Income | Median household income |
| Poverty rate | Percentage of population in poverty | |
| Unemployment rate | Percentage of unemployment population |
| July 15 | August 15 | September 15 | ||||
|---|---|---|---|---|---|---|
| Coef. | p Value | Coef. | p Value | Coef. | p Value | |
| Male percentage | 9.26 | * 0.014 | 7.72 | * 0.013 | 9.65 | ** 0.002 |
| White percentage | 0.21 | 0.970 | −0.06 | 0.910 | −0.05 | 0.966 |
| Median age | −0.08 | 0.910 | 0.16 | 0.793 | 0.03 | 0.960 |
| College degree rate | 0.53 | 0.273 | 0.39 | 0.313 | 0.45 | 0.228 |
| Obesity rate | −2.17 | 0.198 | −0.76 | 0.589 | −0.25 | 0.846 |
| Unemployment rate | 2.88 | ** 0.003 | 2.07 | ** 0.009 | 2.19 | ** 0.004 |
| Uninsured rate | 1.91 | 0.052 | 0.49 | 0.539 | 0.12 | 0.875 |
| NP abundance | 0.08 | 0.575 | 0.04 | 0.737 | 0.07 | 0.490 |
| October 15 | November 15 | December 15 | ||||
| Coef. | p Value | Coef. | p Value | Coef. | p Value | |
| Male percentage | 11.30 | ** 0.000 | 11.92 | ** 0.000 | 11.84 | ** 0.000 |
| White percentage | 0.35 | 0.407 | 0.73 | 0.069 | 1.17 | ** 0.003 |
| Median age | 0.09 | 0.855 | −0.03 | 0.952 | −0.19 | 0.694 |
| College degree rate | 0.35 | 0.263 | 0.34 | 0.243 | 0.38 | 0.198 |
| Obesity rate | 0.32 | 0.765 | 0.58 | 0.568 | 0.68 | 0.467 |
| Unemployment rate | 2.05 | ** 0.001 | 2.10 | ** 0.000 | 2.28 | ** 0.000 |
| Uninsured rate | −0.25 | 0.689 | −0.46 | 0.438 | −0.67 | 0.260 |
| NP abundance | 0.12 | 0.160 | 0.13 | 0.111 | 0.13 | 0.106 |
| July 15 | August 15 | September 15 | ||||
|---|---|---|---|---|---|---|
| Coef. | p Value | Coef. | p Value | Coef. | p Value | |
| Male percentage | 10.70 | * 0.015 | 9.28 | ** 0.008 | 9.65 | ** 0.002 |
| White percentage | −0.07 | 0.911 | −0.48 | 0.348 | −0.02 | 0.966 |
| Median age | 0.04 | 0.962 | 0.41 | 0.537 | 0.03 | 0.960 |
| College degree rate | 0.44 | 0.372 | 0.22 | 0.583 | 0.45 | 0.228 |
| Obesity rate | −3.16 | 0.076 | −2.09 | 0.143 | −0.25 | 0.846 |
| Unemployment rate | 2.83 | ** 0.003 | 1.93 | * 0.011 | 2.19 | ** 0.004 |
| Uninsured rate | 1.75 | 0.081 | 0.64 | 0.419 | 0.12 | 0.875 |
| NP abundance | 0.07 | 0.640 | 0.03 | 0.801 | 0.07 | 0.490 |
| October 15 | November 15 | December 15 | ||||
| Coeff. | p Value | Coeff. | p Value | Coeff. | p Value | |
| Male percentage | 12.81 | ** 0.000 | 13.30 | ** 0.000 | 13.16 | ** 0.000 |
| White percentage | 0.18 | 0.646 | 0.66 | 0.077 | 1.21 | ** 0.001 |
| Median age | 0.26 | 0.604 | 0.11 | 0.815 | −0.10 | 0.840 |
| College degree rate | 0.15 | 0.608 | 0.14 | 0.611 | 0.19 | 0.522 |
| Obesity rate | −0.86 | 0.431 | −0.52 | 0.618 | −0.16 | 0.882 |
| Unemployment rate | 2.01 | ** 0.001 | 2.09 | ** 0.000 | 2.31 | ** 0.000 |
| Uninsured rate | 0.09 | 0.882 | −0.14 | 0.815 | −0.37 | 0.528 |
| NP abundance | 0.11 | 0.213 | 0.12 | 0.149 | 0.13 | 0.124 |
| July 15 | August 15 | September 15 | ||||
|---|---|---|---|---|---|---|
| Coef. | p Value | Coef. | p Value | Coef. | p Value | |
| Male percentage | 9.59 | * 0.012 | 8.10 | ** 0.009 | 9.97 | ** 0.001 |
| White percentage | 0.08 | 0.901 | −0.10 | 0.849 | 0.01 | 0.988 |
| Median age | −0.04 | 0.953 | 0.23 | 0.700 | 0.07 | 0.908 |
| College degree rate | 0.32 | 0.500 | 0.00 | 0.568 | 0.30 | 0.422 |
| Obesity rate | −3.13 | 0.065 | −1.61 | 0.242 | −1.09 | 0.402 |
| Unemployment rate | 2.82 | ** 0.002 | 1.99 | ** 0.008 | 2.17 | ** 0.003 |
| Uninsured rate | 2.16 | * 0.026 | 0.79 | 0.317 | 0.43 | 0.568 |
| NP abundance | 0.08 | 0.565 | 0.03 | 0.784 | 0.05 | 0.613 |
| October 15 | November 15 | December 15 | ||||
| Coeff. | p Value | Coeff. | p Value | Coeff. | p Value | |
| Male percentage | 11.39 | ** 0.000 | 12.01 | ** 0.000 | 11.97 | ** 0.000 |
| White percentage | 0.33 | 0.445 | 0.71 | 0.079 | 1.18 | ** 0.003 |
| Median age | 0.12 | 0.813 | 0.00 | 0.996 | −0.17 | 0.727 |
| College degree rate | 0.33 | 0.299 | 0.32 | 0.275 | 0.35 | 0.228 |
| Obesity rate | 0.24 | 0.821 | 0.53 | 0.598 | 0.66 | 0.512 |
| Unemployment rate | 2.02 | ** 0.001 | 2.07 | ** 0.000 | 2.27 | ** 0.000 |
| Uninsured rate | −0.22 | 0.728 | −0.44 | 0.461 | −0.64 | 0.279 |
| NP abundance | 0.13 | 0.146 | 0.13 | 0.096 | 0.14 | 0.091 |
| July 15th | |||||
|---|---|---|---|---|---|
| Covariates | Min. | Q1. | Median | Q3 | Max. |
| Male percentage | 6.73 | 7.68 | 8.11 | 8.50 | 8.98 |
| White percentage | −0.59 | −0.36 | −0.20 | −0.02 | 0.21 |
| Median age | −0.67 | −0.15 | 0.09 | 0.34 | 0.71 |
| College degree rate | 0.64 | 0.71 | 0.75 | 0.80 | 0.83 |
| Obesity rate | −0.81 | −0.49 | −0.32 | −0.10 | 0.22 |
| Unemployment rate | 1.82 | 2.15 | 2.36 | 2.66 | 3.18 |
| Uninsured rate | 0.48 | 0.76 | 0.93 | 1.20 | 1.58 |
| NP abundance | 0.17 | 0.20 | 0.21 | 0.23 | 0.24 |
| August 15th | |||||
| Covariates | Min. | Q1. | Median | Q3 | Max. |
| Male percentage | 7.11 | 7.33 | 7.44 | 7.60 | 7.85 |
| White percentage | −0.67 | −0.65 | −0.63 | −0.61 | −0.59 |
| Median age | 0.32 | 0.41 | 0.44 | 0.50 | 0.57 |
| College degree rate | 0.48 | 0.49 | 0.49 | 0.50 | 0.50 |
| Obesity rate | −0.06 | −0.05 | −0.03 | −0.01 | 0.02 |
| Unemployment rate | 1.57 | 1.64 | 1.69 | 1.75 | 1.85 |
| Uninsured rate | −0.05 | −0.01 | 0.01 | 0.05 | 0.10 |
| NP abundance | 0.12 | 0.12 | 0.12 | 0.12 | 0.13 |
| September 15th | |||||
| Covariates | Min. | Q1. | Median | Q3 | Max. |
| Male percentage | 9.28 | 9.44 | 9.55 | 9.69 | 9.90 |
| White percentage | −0.43 | −0.38 | −0.37 | −0.35 | −0.33 |
| Median age | 0.20 | 0.28 | 0.31 | 0.36 | 0.43 |
| College degree rate | 0.42 | 0.43 | 0.44 | 0.44 | 0.45 |
| Obesity rate | −0.07 | −0.05 | −0.04 | −0.02 | 0.01 |
| Unemployment rate | 1.77 | 1.83 | 1.87 | 1.93 | 2.02 |
| Uninsured rate | −0.22 | −0.17 | −0.14 | −0.11 | −0.05 |
| NP abundance | 0.12 | 0.12 | 0.13 | 0.13 | 0.13 |
| October 15th | |||||
| Covariates | Min. | Q1. | Median | Q3 | Max. |
| Male percentage | 11.37 | 11.58 | 11.69 | 11.82 | 12.05 |
| White percentage | 0.07 | 0.11 | 0.13 | 015 | 0.17 |
| Median age | 0.20 | 0.24 | 0.27 | 0.29 | 0.35 |
| College degree rate | 0.30 | 0.31 | 0.32 | 0.33 | 0.33 |
| Obesity rate | 0.29 | 0.31 | 0.33 | 0.36 | 0.39 |
| Unemployment rate | 1.81 | 1.86 | 1.89 | 1.94 | 2.00 |
| Uninsured rate | −0.31 | −0.27 | −0.25 | −0.22 | −0.17 |
| NP abundance | 0.16 | 0.16 | 0.16 | 0.16 | 0.17 |
| November 15th | |||||
| Covariates | Min. | Q1. | Median | Q3 | Max. |
| Male percentage | 11.96 | 12.15 | 12.24 | 12.37 | 12.57 |
| White percentage | 0.57 | 0.60 | 0.63 | 0.65 | 0.68 |
| Median age | 0.03 | 0.08 | 0.11 | 0.14 | 0.20 |
| College degree rate | 0.27 | 0.28 | 0.29 | 0.30 | 0.30 |
| Obesity rate | 0.53 | 0.55 | 0.57 | 0.59 | 0.64 |
| Unemployment rate | 1.91 | 1.95 | 1.98 | 2.03 | 2.09 |
| Uninsured rate | −0.51 | −0.47 | −0.45 | −0.43 | −0.39 |
| NP abundance | 0.16 | 0.17 | 0.17 | 0.17 | 0.17 |
| December 15th | |||||
| Covariates | Min. | Q1. | Median | Q3 | Max. |
| Male percentage | 12.03 | 12.19 | 12.29 | 12.43 | 12.62 |
| White percentage | 1.14 | 1.18 | 1.21 | 1.23 | 1.27 |
| Median age | −0.18 | −0.13 | −0.10 | −0.07 | −0.02 |
| College degree rate | 0.28 | 0.29 | 0.30 | 0.31 | 0.32 |
| Obesity rate | 0.68 | 0.70 | 0.72 | 0.74 | 0.79 |
| Unemployment rate | 2.16 | 2.20 | 2.23 | 2.27 | 2.33 |
| Uninsured rate | −0.69 | −0.65 | −0.63 | −0.60 | −0.57 |
| NP abundance | 0.16 | 0.17 | 0.17 | 0.17 | 0.17 |
| SEM | SLM | CAR | GWR | |
|---|---|---|---|---|
| July 15 | 61.59 | 58.72 | 59.39 | 51.24 |
| August 15 | 41.47 | 36.64 | 39.81 | 34.49 |
| September 15 | 35.62 | 30.67 | 35.20 | 25.64 |
| October 15 | 17.00 | 11.96 | 17.42 | 5.31 |
| November 15 | 11.52 | 6.68 | 11.94 | −0.54 |
| December 15 | 10.74 | 8.31 | 11.13 | −1.48 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lyu, T.; Hair, N.; Yell, N.; Li, Z.; Qiao, S.; Liang, C.; Li, X. Temporal Geospatial Analysis of COVID-19 Pre-Infection Determinants of Risk in South Carolina. Int. J. Environ. Res. Public Health 2021, 18, 9673. https://doi.org/10.3390/ijerph18189673
Lyu T, Hair N, Yell N, Li Z, Qiao S, Liang C, Li X. Temporal Geospatial Analysis of COVID-19 Pre-Infection Determinants of Risk in South Carolina. International Journal of Environmental Research and Public Health. 2021; 18(18):9673. https://doi.org/10.3390/ijerph18189673
Chicago/Turabian StyleLyu, Tianchu, Nicole Hair, Nicholas Yell, Zhenlong Li, Shan Qiao, Chen Liang, and Xiaoming Li. 2021. "Temporal Geospatial Analysis of COVID-19 Pre-Infection Determinants of Risk in South Carolina" International Journal of Environmental Research and Public Health 18, no. 18: 9673. https://doi.org/10.3390/ijerph18189673
APA StyleLyu, T., Hair, N., Yell, N., Li, Z., Qiao, S., Liang, C., & Li, X. (2021). Temporal Geospatial Analysis of COVID-19 Pre-Infection Determinants of Risk in South Carolina. International Journal of Environmental Research and Public Health, 18(18), 9673. https://doi.org/10.3390/ijerph18189673







