Cost-Utility Analysis of Antibiotic Therapy versus Appendicectomy for Acute Uncomplicated Appendicitis
Abstract
:1. Introduction
1.1. Background
1.2. Motivation and Rationale
1.3. Study Objectives
1.4. Literature Review
2. Methods
2.1. Propensity Score Matching
2.2. Application to a UK Based Health System
2.3. Choice of Analysis
2.4. Choice of Perspective
2.5. Time Period
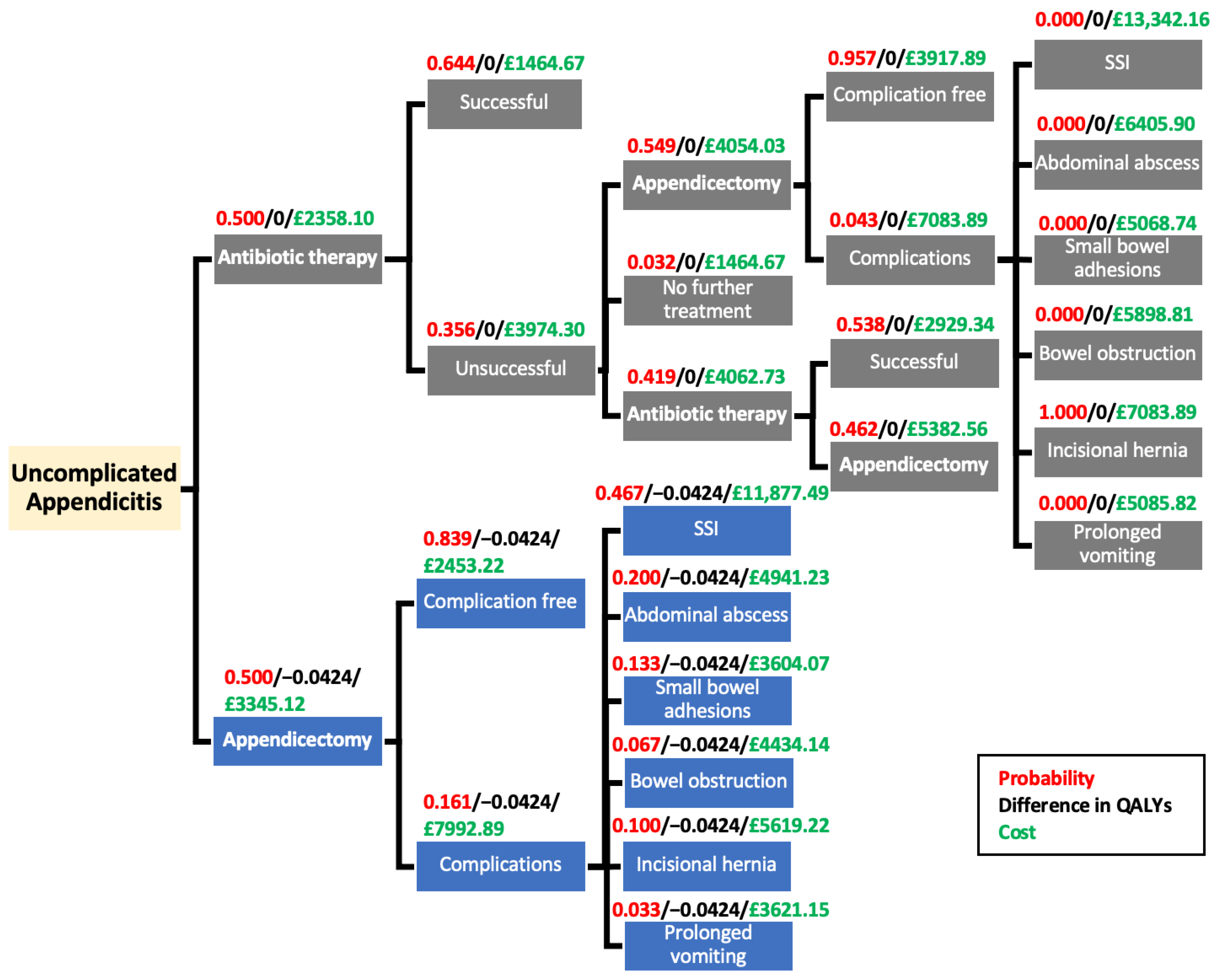
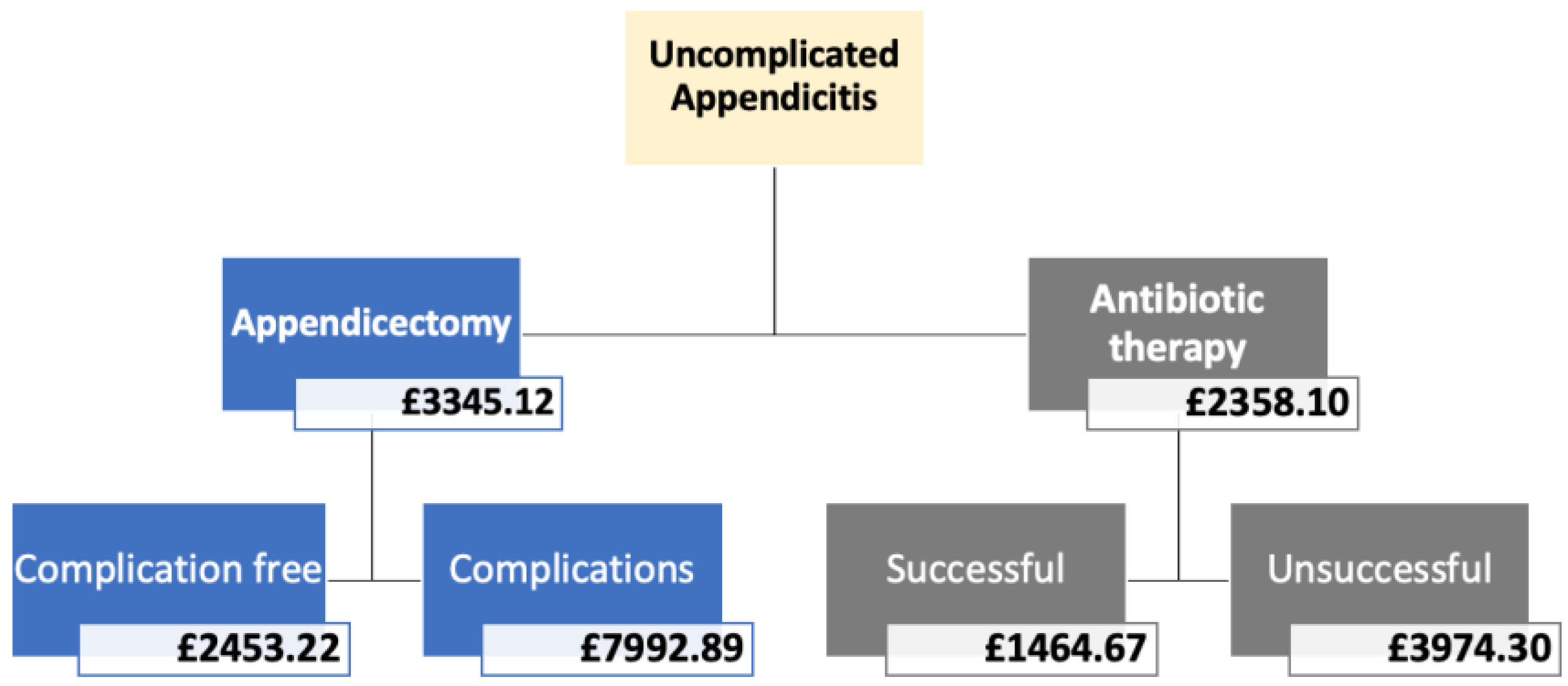
2.6. Modelling
2.7. Costs
2.7.1. Discount Rate
2.7.2. Appendicectomy
2.7.3. Antibiotic Therapy
2.8. Benefits
3. Results
3.1. Sensitivity Analysis
3.1.1. Sensitivity Analysis 1
3.1.2. Sensitivity Analysis 2
4. Discussion
4.1. Comparison of the Results with Literature
4.2. Limitations
4.3. Generalisability
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data availability statement
Conflicts of Interest
Appendix A
| Inclusion | Exclusion |
|---|---|
| Studies published in and after 2016 | Paediatric appendicitis |
| Randomised and non-randomised control trials | Non-English studies |
| Cohort studies | Studies investigating complicated appendicitis |
| Economic evaluations | Ideas, editorials, or opinions |
Appendix B
| Comparisons | Pre-Matching | Post-Matching | ||||
|---|---|---|---|---|---|---|
| Antibiotic | Appendicectomy | p Value | Antibiotic | Appendicectomy | p Value | |
| Patients: n (%) | 87 (27.4) | 231 (72.6) | - | 87 (50) | 87 (50) | - |
| Age: mean ± SD | 38.4 ± 18.4 | 40.0 ± 18.0 | 0.509 | 38.4 ± 18.4 | 38.4 ± 18.4 | 0.990 |
| Sex M:F (M%:F%) | 38:49 (43.7 vs. 56.3) | 134:97 (58.0 vs. 42.0) | 0.024 | 38:49 (43.7 vs. 56.3) | 38:49 (43.7 vs. 56.3) | 0.990 |
| Appendicitis inflammatory response (AIR) score: mean ± SD | 5.7 ± 2.0 | 6.9 ± 2.1 | <0.00001 | 5.7 ± 2.0 | 6.9 ± 2.2 | 0.00016 |
| Length of primary hospital stay (days): mean ± SD | 3.1 ± 0.9 | 3.4 ± 1.9 | 0.158 | 3.1 ± 0.9 | 3.4 ± 1.5 | 0.111 |
| SF-12 score at 1-year follow-up (β (95% CI)) | Reference | −2.67 (−4.95, −0.38) | 0.02 | Reference | −4.24 (−6.34, −2.15) | <0.01 |
Appendix C
| Description | Year | Unadjusted Cost | Inflated (3.5% pa) | Reference | Comment |
|---|---|---|---|---|---|
| Mean Days in hospital = 3.1 days | 2018 | GBP 346 × 3.1 = GBP 1072.60 | GBP 1189.21 | ACTUAA 2021; [13] NHS 2017/18 Tariffs [30] | |
| Blood tests | 2019 | GBP 39.18 | GBP 41.97 | Clement et al. 2020 [28] | |
| Ultrasound | 2019 | GBP 52 | GBP 55.70 | Clement et al. 2020 [28] | |
| CT abdo/pelvis | 2019 | GBP 101 | GBP 108.19 | Clement et al. 2020 [28] | |
| Piperacillin-tazobactam 4.5 g every 8 h | 2021 | GBP 113.82 | GBP 113.83 | BNF 2021 [32] | Price per day = GBP 36.72 GBP 36.72 × 3.1 = GBP 113.82 |
| Ceftriaxone and metronidazole 1–2 g every 12 h-ceftriaxone 500 mg every 8 h-metronidazole | 2021 | GBP 88.20 | GBP 88.20 | BNF 2021 [33,34] | Price per day = GBP 28.45 GBP 28.45 × 3.1 = GBP 88.20 |
| Ceftazidime 1–2 g every 8 h | 2021 | GBP 26.35 | GBP 26.35 | BNF 2021 [35] | Price per day = GBP 8.50 GBP 8.50 × 3.1 = GBP 26.35 |
| Amoxicillin-clavulanic acid (co-amoxiclav) 1–2 g every 8 h | 2021 | GBP 32.86 | GBP 32.86 | BNF 2021 [36] | Price per day = GBP 10.60 GBP 10.60 × 3.1 = GBP 32.86 |
| Ciprofloxacin and metronidazole 400 mg every 12 h-ciprofloxacin 500 mg every 8 h-metronidazole | 2021 | GBP 136.56 | GBP 136.56 | BNF 2021 [37] | Price per day = GBP 44.05 GBP 44.05 × 3.1 = GBP 136.56 |
| Ertapenem 1 g every 24 h | 2021 | GBP 98.12 | GBP 98.12 | BNF 2021 [38] | Price per day = GBP 31.65 GBP 31.65 × 3.1 = GBP 98.12 |
| Weighted average calculation | [(113.83 × 27.6) + (88.20 × 11.5) + (26.35 × 16.1) + (32.86 × 35.6) + (136.56 × 8) + (98.12 × 1.2)]/100 = GBP 69.60 | ||||
| Antibiotics Total | GBP 1464.67 | ||||
| Description | Year | Unadjusted Cost | Inflated (3.5% pa) | Reference | Comment |
|---|---|---|---|---|---|
| Appendicectomy Total | 2019 | GBP 2290.11 | GBP 2453.22 | Clement et al. 2020 [28] | Costs for adults |
| Complications | |||||
| 3 days in hospital for complications = (72 h) | 2018 | GBP 346 × 3 = GBP 1038 | GBP 1150.85 | ACTUAA 2021; [13] NHS 2017/18 Tariffs [30] | 3.4 + 2 (1.5) = 6.4 days using ‘range rule of thumb’ 6.4 − 3.4 days = 3 days |
| Infected surgical wound | 2016 | GBP 6966 | GBP 8273.42 | Guest et al. 2018 [41] | Costs incurred for an infected surgical wound over 12 months |
| Surgical site infection (SSI) total | GBP 9424.27 | ||||
| Percutaneous Drainage | 2020 | GBP 730 | GBP 755.55 | NHS 2019/20 Tariffs; [31] BMJ Best Practice 2021 [45] | |
| Meropenem | 2021 | GBP 185.65 | N/A | BNF 2021 [46] | 1 g every 8 h: 9 doses |
| Metronidazole | 2021 | GBP 20.12 | N/A | BNF 2021 [34] | 500 mg every 12 h: 6 doses |
| Ampicillin | 2021 | GBP 375.84 | N/A | BNF 2021 [47] | 2 g every 6 h: 12 doses |
| Postoperative abdominal abscess total | GBP 2488.01 | ||||
| Small bowel adhesions syndrome total | GBP 1150.85 | BMJ Best Practice, 2021 [48] | No treatment recommended | ||
| Abdominal Procedure | 2020 | GBP 802 | GBP 830.07 | NHS 2019/20 Tariffs; [31] BMJ Best Practice 2021 [49] | Emergency therapeutic general abdominal procedure |
| Bowel obstruction total | GBP 1980.92 | ||||
| Incisional hernia procedure | 2020 | GBP 1947 | GBP 2015.15 | NHS 2019/20 Tariffs [31] | |
| Incisional hernia total | GBP 3166 | ||||
| Ondansetron (anti-emetic) | 2021 | GBP 17.08 | N/A | BNF 2021 [50] | 4 mg per dose. Minimum 3 doses over 3 days |
| Prolonged vomiting | GBP 1167.93 | ||||
Appendix D
| (1) | Probability of each post-operative complication occurring was assumed to be the same for the pre- and post-matching appendicectomy group. |
| (2) | Those with post-operative complications represented the maximum value of days stayed in hospital when calculating the range from standard deviation. |
| (3) | Treatment for post-operative complications was given over the 3 extra days in hospital. |
| (4) | Laparoscopic and open surgery experienced similar outcomes in this study. |
| (5) | Italian and UK populations exhibit similar characteristics when considering uncomplicated appendicitis. |
References
- NICE Appendicitis. 2020. Available online: https://cks.nice.org.uk/topics/appendicitis/ (accessed on 12 February 2021).
- NHS Appendicitis. 2019. Available online: https://www.nhs.uk/conditions/appendicitis/ (accessed on 12 February 2021).
- Mariage, M.; Sabbagh, C.; Grelpois, G.; Prevot, F.; Darmon, I.; Regimbeau, J.M. Surgeon’s definition of complicated appendicitis: A Prospective video survey study. Euroasian J. Hepato-Gastroenterol. 2019, 9, 1. [Google Scholar] [CrossRef] [PubMed]
- NICE Scenario: Managing Suspected Appendicitis. 2020. Available online: https://cks.nice.org.uk/topics/appendicitis/management/managing-suspected-appendicitis/ (accessed on 9 February 2021).
- Di Saverio, S.; Podda, M.; De Simone, B.; Ceresoli, M.; Augustin, G.; Gori, A.; Boermeester, M.; Sartelli, M.; Coccolini, F.; Tarasconi, A.; et al. Diagnosis and treatment of acute appendicitis: 2020 update of the WSES Jesusalem guidelines. World J. Emerg. Surg. 2020, 15, 1–42. [Google Scholar] [CrossRef] [PubMed]
- BMJ Best Practice Acute Appendicitis in Adults: Treatment Algorithm. 2021. Available online: https://bestpractice.bmj.com/topics/en-gb/3000094/treatment-algorithm (accessed on 10 February 2021).
- Snyder, M.J.; Guthrie, M.; Cagle, S.D. Acute appendicitis: Efficient diagnosis and management. Am. Fam. Physician 2018, 98, 25–33. [Google Scholar]
- Harnoss, J.C.; Zelienka, I.; Probst, P.; Grummich, K.; Müller-Lantzsch, C.; Harnoss, J.M.; Ulrich, A.; Büchler, M.W.; Diener, M.K. Antibiotics versus surgical therapy for uncomplicated appendicitis: Systematic review and meta-analysis of controlled trials (PROSPERO 2015: CRD42015016882). Ann. Surg. 2017, 265, 889–900. [Google Scholar] [CrossRef] [PubMed]
- Paajanen, H.; Grönroos, J.M.; Rautio, T.; Nordström, P.; Aarnio, M.; Rantanen, T.; Hurme, S.; Dean, K.; Jartti, A.; Mecklin, J.-P.; et al. A prospective randomized controlled multicenter trial comparing antibiotic therapy with appendectomy in the treatment of uncomplicated acute appendicitis (APPAC trial). BMC Surg. 2013, 13, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- NIHR Dissemination Centre. Antibiotics May Be an Alternative First-Line Treatment for Uncomplicated Appendicitis; National Institute for Health Research: Southampton, UK, 2019. [CrossRef]
- BMJ Best Practice Acute Appendicitis in Adults: Investigation. 2021. Available online: https://bestpractice.bmj.com/topics/en-gb/3000094/investigations (accessed on 11 February 2021).
- The CODA Collaborative. A randomized trial comparing antibiotics with appendectomy for appendicitis. N. Engl. J. Med. 2020, 383, 1907–1919. [Google Scholar] [CrossRef] [PubMed]
- Podda, M.; The ACTUAA Study Collaborative Working Group; Poillucci, G.; Pacella, D.; Mortola, L.; Canfora, A.; Aresu, S.; Pisano, M.; Erdas, E.; Pisanu, A.; et al. Appendectomy versus conservative treatment with antibiotics for patients with uncomplicated acute appendicitis: A propensity score–matched analysis of patient-centered outcomes (the ACTUAA prospective multicenter trial). Int. J. Color. Dis. 2021, 36, 589–598. [Google Scholar] [CrossRef] [PubMed]
- Javanmard-Emamghissi, H.; Boyd-Carson, H.; Hollyman, M.; Doleman, B.; Adiamah, A.; Lund, J.N.; Clifford, R.; Dickerson, L.; Richards, S.; Pearce, L.; et al. The management of adult appendicitis during the COVID-19 pandemic: An interim analysis of a UK cohort study. Tech. Coloproctol. 2021, 25, 401–411. [Google Scholar] [CrossRef] [PubMed]
- Sippola, S.; Grönroos, J.; Tuominen, R.; Paajanen, H.; Rautio, T.; Nordström, P.; Aarnio, M.; Rantanen, T.; Hurme, S.; Salminen, P. Economic evaluation of antibiotic therapy versus appendicectomy for the treatment of uncomplicated acute appendicitis. Br. J. Surg. 2017, 104, 1355–1361. [Google Scholar] [CrossRef] [PubMed]
- Haijanen, J.; Sippola, S.; Tuominen, R.; Grönroos, J.; Paajanen, H.; Rautio, T.; Nordström, P.; Aarnio, M.; Rantanen, T.; Hurme, S.; et al. Cost analysis of antibiotic therapy versus appendectomy for treatment of uncomplicated acute appendicitis: 5-year results of the APPAC randomized clinical trial. PLoS ONE 2019, 14, e0220202. [Google Scholar] [CrossRef] [PubMed]
- Sceats, L.A.; Ku, S.; Coughran, A.; Barnes, B.; Grimm, E.; Muffly, M.; Spain, D.A.; Kin, C.; Owens, U.K.; Goldhaber-Fiebert, J.D. Operative Versus Nonoperative Management of Appendicitis: A Long-Term Cost Effectiveness Analysis. MDM Policy Pract. 2019, 4, 2381468319866448. [Google Scholar] [CrossRef] [Green Version]
- D’Agostino, R.B., Jr. Propensity score methods for bias reduction in the comparison of a treatment to a non-randomized control group. Stat. Med. 1998, 17, 2265–2281. [Google Scholar] [CrossRef]
- Apolone, G.; Lattuada, L. Health Coverage in Italy. J. Ambul. Care Manag. 2003, 26, 378–382. [Google Scholar] [CrossRef] [PubMed]
- Busse, R.; Jakubowski, E.; Chambers, G.R. Health Care Systems in the EU, a Comparative Study; Publications Office of the European Union: Luxembourg, 2020; Available online: https://op.europa.eu/en/publication-detail/-/publication/6125eac3-c1d3-4ea3-8f3d-9d9888d25e56 (accessed on 11 February 2021).
- Atella, V.; Belotti, F.; Bojke, C.; Castelli, A.; Grašič, K.; Kopinska, J.; Mortari, A.P.; Street, A. How health policy shapes healthcare sector productivity? Evidence from Italy and UK. Health Policy 2018, 123, 27–36. [Google Scholar] [CrossRef]
- Financial Times, Currencies: GBPEUR. 2021. Available online: https://markets.ft.com/data/currencies/tearsheet/summary?s=GBPEUR (accessed on 2 August 2021).
- Gafni, A. The quality of QALYs (quality-adjusted-life-years): Do QALYs measure what they at least intend to measure? Health Policy 1989, 13, 81–83. [Google Scholar] [CrossRef]
- Whitehead, S.J.; Ali, S. Health outcomes in economic evaluation: The QALY and utilities. Br. Med. Bull. 2010, 96, 5–21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- NICE Assessing Cost Effectiveness. 2012. Available online: https://www.nice.org.uk/process/pmg6/chapter/assessing-cost-effectiveness (accessed on 9 February 2021).
- Yang, Z.; Sun, F.; Ai, S.; Wang, J.; Guan, W.; Liu, S. Meta-analysis of studies comparing conservative treatment with antibiotics and appendectomy for acute appendicitis in the adult. BMC Surg. 2019, 19, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- NHS Laparoscopy (Keyhole Surgery). 2018. Available online: https://www.nhs.uk/conditions/laparoscopy/ (accessed on 9 February 2021).
- Clement, K.D.; Emslie, K.; Maniam, P.; Wilson, M. What is the operative cost of managing acute appendicitis in the NHS: The impact of stump technique and perioperative imaging. World J. Surg. 2019, 44, 749–754. [Google Scholar] [CrossRef] [PubMed]
- Farnsworth, D.L.; Triola, M.F. Elementary statistics. Technometrics 1990, 32, 456. [Google Scholar] [CrossRef]
- NHS Reference Costs 2017/18: Highlights, Analysis and Introduction to the Data. 2018. Available online: https://improvement.nhs.uk/documents/1972/1_-_Reference_costs_201718.pdf (accessed on 12 February 2021).
- NHS Improvement National Tariff Payment System. 2019. Available online: https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&cad=rja&uact=8&ved=2ahUKEwjO3q-v-rfuAhViShUIHVr-CMkQFjABegQIARAC&url=https%3A%2F%2Fimprovement.nhs.uk%2Fdocuments%2F479%2FAnnex_DtA_National_tariff_workbook.xlsx&usg=AOvVaw0rr7lbjT54BHw8iMC3HQsj (accessed on 14 February 2020).
- BNF: British National Formulary, Nice Piperacillin with Tazobactam. 2021. Available online: https://bnf.nice.org.uk/medicinal-forms/piperacillin-with-tazobactam.html (accessed on 14 February 2021).
- BNF: British National Formulary, Nice Ceftriaxone. 2021. Available online: https://bnf.nice.org.uk/medicinal-forms/ceftriaxone.html (accessed on 14 February 2021).
- BNF: British National Formulary, Nice Metronidazole. 2021. Available online: https://bnf.nice.org.uk/medicinal-forms/metronidazole.html (accessed on 14 February 2021).
- BNF: British National Formulary, Nice Ceftazidime. 2021. Available online: https://bnf.nice.org.uk/medicinal-forms/ceftazidime.html (accessed on 14 February 2021).
- BNF: British National Formulary, Nice Co-Amoxiclav. 2021. Available online: https://bnf.nice.org.uk/medicinal-forms/co-amoxiclav.html (accessed on 14 February 2021).
- BNF: British National Formulary, Nice Ciprofloxacin. 2021. Available online: https://bnf.nice.org.uk/medicinal-forms/ciprofloxacin.html (accessed on 14 February 2021).
- BNF: British National Formulary, Nice Ertapenem. 2021. Available online: https://bnf.nice.org.uk/medicinal-forms/ertapenem.html (accessed on 14 February 2021).
- NICE. Guide to the Methods of Technology Appraisal. 2013. Available online: https://www.nice.org.uk/process/pmg9/resources/guide-to-the-methods-of-technology-appraisal-2013-pdf-2007975843781 (accessed on 2 June 2021).
- Faber, J.; Fonseca, L.M. How sample size influences research outcomes. Dent. Press J. Orthod. 2014, 19, 27–29. [Google Scholar] [CrossRef]
- Guest, J.F.; Fuller, G.W.; Vowden, P. Costs and outcomes in evaluating management of unhealed surgical wounds in the community in clinical practice in the UK: A cohort study. BMJ Open 2018, 8, e022591. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- BMJ Statements of Probability and Confidence Intervals. 1997. Available online: https://www.bmj.com/about-bmj/resources-readers/publications/statistics-square-one/4-statements-probability-and-confiden (accessed on 17 February 2021).
- Sippola, S.; Haijanen, J.; Viinikainen, L.; Grönroos, J.; Paajanen, H.; Rautio, T.; Nordström, P.; Aarnio, M.; Rantanen, T.; Hurme, S.; et al. Quality of Life and Patient Satisfaction at 7-Year Follow-up of Antibiotic Therapy vs. Appendectomy for Uncomplicated Acute Appendicitis. JAMA Surg. 2020, 155, 283–289. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization Italy. Available online: https://www.euro.who.int/en/countries/italy (accessed on 8 February 2021).
- BMJ Best Practice Intra-Abdominal Abscess: Treatment Algorithm. 2021. Available online: https://bestpractice.bmj.com/topics/en-gb/996/treatment-algorithm (accessed on 16 February 2021).
- BNF: British National Formulary, Nice Meropenem. 2021. Available online: https://bnf.nice.org.uk/medicinal-forms/meropenem.html (accessed on 14 February 2021).
- BNF: British National Formulary, Nice Ampicillin. 2021. Available online: https://bnf.nice.org.uk/medicinal-forms/ampicillin.html (accessed on 14 February 2021).
- BMJ Best Practice Small Bowel Obstruction: Primary Prevention. 2021. Available online: https://bestpractice.bmj.com/topics/en-gb/3000119/prevention#referencePop10 (accessed on 14 February 2021).
- BMJ Best Practice Small Bowel Obstruction: Treatment Algorithm. 2021. Available online: https://bestpractice.bmj.com/topics/en-gb/3000119/treatment-algorithm (accessed on 11 February 2021).
- BNF: British National Formulary, Nice Ondansetron. 2021. Available online: https://bnf.nice.org.uk/medicinal-forms/ondansetron.html (accessed on 14 February 2021).

| SF-12 Score from Trial | Assigned Difference in QALY | |
|---|---|---|
| Antibiotic-first | Reference | 0 |
| Appendicectomy | −4.24 | −0.0424 |
| Group | Original Difference in QALY | Assigned Difference in QALY |
|---|---|---|
| Antibiotics-Successful | 0 (reference group) | 0 (reference group) |
| Antibiotics-Unsuccessful | 0 (reference group) | −0.0215 |
| Appendicectomy-Complications | −0.0424 | −0.0634 |
| Appendicectomy-No complications | −0.0424 | −0.0215 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ali, A.; Mobarak, Z.; Al-Jumaily, M.; Anwar, M.; Moti, Z.; Zaman, N.; Akbari, A.R.; de Preux, L. Cost-Utility Analysis of Antibiotic Therapy versus Appendicectomy for Acute Uncomplicated Appendicitis. Int. J. Environ. Res. Public Health 2021, 18, 8473. https://doi.org/10.3390/ijerph18168473
Ali A, Mobarak Z, Al-Jumaily M, Anwar M, Moti Z, Zaman N, Akbari AR, de Preux L. Cost-Utility Analysis of Antibiotic Therapy versus Appendicectomy for Acute Uncomplicated Appendicitis. International Journal of Environmental Research and Public Health. 2021; 18(16):8473. https://doi.org/10.3390/ijerph18168473
Chicago/Turabian StyleAli, Ayesha, Zina Mobarak, Mariam Al-Jumaily, Mehreen Anwar, Zaeem Moti, Nadia Zaman, Amir Reza Akbari, and Laure de Preux. 2021. "Cost-Utility Analysis of Antibiotic Therapy versus Appendicectomy for Acute Uncomplicated Appendicitis" International Journal of Environmental Research and Public Health 18, no. 16: 8473. https://doi.org/10.3390/ijerph18168473
APA StyleAli, A., Mobarak, Z., Al-Jumaily, M., Anwar, M., Moti, Z., Zaman, N., Akbari, A. R., & de Preux, L. (2021). Cost-Utility Analysis of Antibiotic Therapy versus Appendicectomy for Acute Uncomplicated Appendicitis. International Journal of Environmental Research and Public Health, 18(16), 8473. https://doi.org/10.3390/ijerph18168473






