Appropriateness of Care for Common Childhood Infections at Low-Level Private Health Facilities in a Rural District in Western Uganda
Abstract
1. Introduction
2. Material and Methods
2.1. Research Design
2.2. Study Setting
2.3. Sampling and Recruitment
Selection Criteria
2.4. Variables
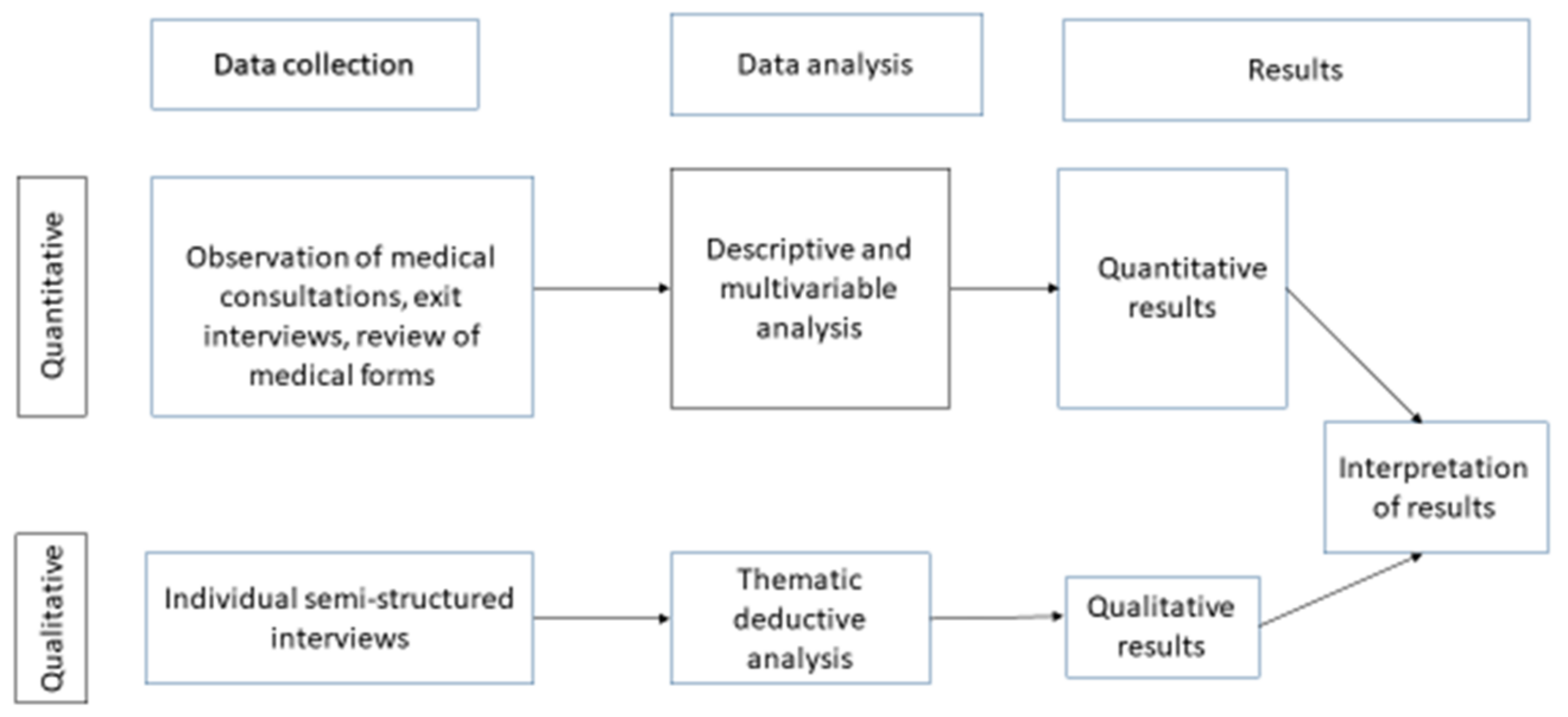
2.5. Quantitative Data Collection
Statistical Analysis
2.6. Qualitative Data Collection
Qualitative Data Analysis
3. Results
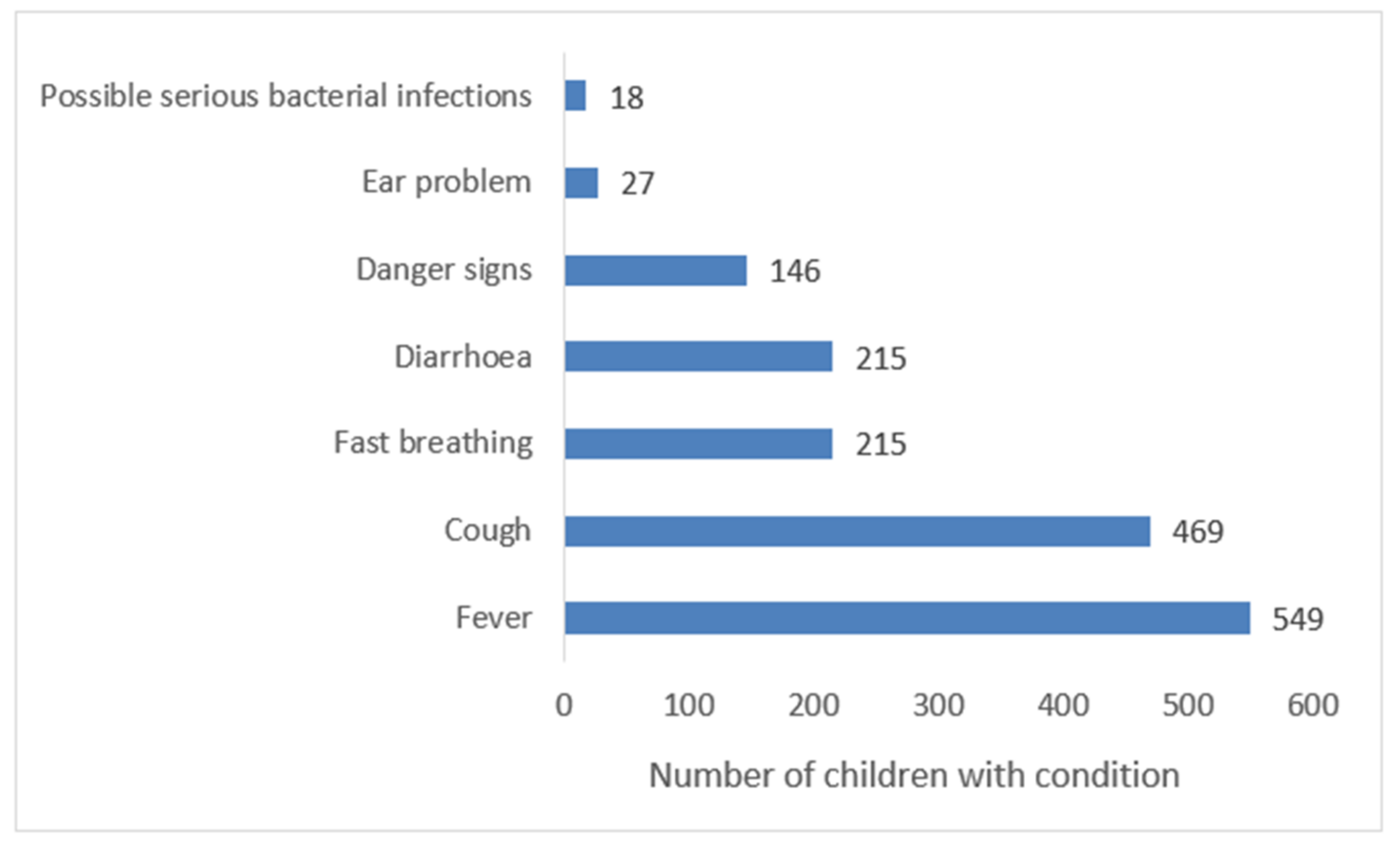
3.1. Management of the Sick Children by the Health Workers
3.2. Factors Associated with Child Receiving Appropriate Care
3.3. Perspectives of Policymakers and Health Workers on Quality of Care Offered by LLPHF
- (1)
- Referral of patients with severe conditions may not be timely
- (2)
- Inadequate infrastructure exacerbates inappropriate healthcare provision
- (3)
- The care is not patient centered but rather is driven by desire to make money or please the caretaker
- (4)
- Existing guidelines are not followed, leading to mismanagement patients
3.4. Referral of Patients with Severe Conditions May Not Be Timely
“Most of the low-level private clinics treat minor conditions, like simple coughs, malaria, pneumonias, they should not manage serious illnesses, as these should be referred to higher levels. The challenge is some facilities wait and only refer the child in very late stages.”(Male policymaker, DHO)
“Here we mostly handle outpatient clients and a few in patients especially when conditions don’t require us to refer. To give examples we handle simple malaria, we handle respiratory infections including pneumonia because our facility is categorized as health center III.”(Male nurse, rural clinic)
3.5. Inadequate Infrastructure Exacerbate Inappropriate Healthcare Provision
“It is expensive paying an enrolled nurse….so they prefer to hire nursing aids, who you can pay little money. You find expired reagents and untrained lab personnel simply picked because they are relatives to the owner of the clinic. The fridges are not functional, instead, they just put water in the bucket and put reagents and they refer to that as a fridge. So how do you expect to find a correct result….?”(Female policymaker from DHO)
“Most of us don’t carry out investigations so when the patients come with cough you give septrin (Cotrimoxazole), amoxicillin, plus Panadol (paracetamol) if the fever persists that is when the child may be taken for testing somewhere…..”(Female nurse, rural clinic)
“People want money; some may provide unnecessary treatment to make the treatment list very big and then get more money. I witnessed that behavior several times in one of the clinics I previously worked for.”(Male clinical officer, rural clinic)
“There is irrational use of antibiotics in private clinics partly because it is always difficult to explain to the mother that the child’s simple cough does not need an antibiotic…. Sometimes you give that higher antibiotic to impress the mother but of course knowing that you are not doing the right thing. Unfortunately the children somehow have been mismanaged, the facilities have learnt the fact that if you don’t inject you don’t get the money.”(Male doctor, urban clinic)
3.6. Existing Guidelines Are Not Followed, Leading to Mismanagement of Patients
“We sometimes attend conferences where they elaborate the new clinical guidelines that come up. Some of us follow the guidelines but many others do not. When they see someone has a fever, has a complication they start off by pumping the drugs.”(Male doctor, urban clinic)
“When we are assessing these children we use the knowledge we acquired from school but we have not been given any guidelines to use; that is the challenge.”(Male clinical officer, rural clinic)
“The services are fair….if the child is taken to the facility and is not well managed in the next one day will be moved to the next health facility for better services….we are in town and they can easily get to a number of facilities around.”(Male doctor, urban clinic).
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- UNICEF; WHO; World Bank Group; United Nations. Levels and Trends in Child Mortality 2020; Estimates Developed by the UN Inter-Agency Group for Child Mortality Estimation; UNICEF: New York, NY, USA; World Health Organization: Geneva, Switzerland; World Bank: Washington, DC, USA; United Nations Population Division: New York, NY, USA, 2020. [Google Scholar]
- World Health Organization. World Health Statistics 2016: Monitoring Health for the SDGs Sustainable Development Goals; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- United Nations Children’s Fund. UNICEF Data: Monitoring the Situation of Children and Women; Trends in Under-Five Mortality Rate; UNICEF: New York, NY, USA, 2018. [Google Scholar]
- Bhutta, Z.A.; Black, R.E. Global Maternal, Newborn, and Child Health—So Near and Yet So Far. N. Engl. J. Med. 2013, 369, 2226–2235. [Google Scholar] [CrossRef]
- Abubakar, I.; Tillmann, T.; Banerjee, A. Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2015, 385, 117–171. [Google Scholar]
- Nambuusi, B.B.; Ssempiira, J.; Makumbi, F.E.; Kasasa, S.; Vounatsou, P. The effects and contribution of childhood diseases on the geographical distribution of all-cause under-five mortality in Uganda. Parasite Epidemiol. Control. 2019, 5, e00089. [Google Scholar] [CrossRef]
- UNICEF Uganda. Child and Maternal Health, Keeping Children Alive and Thriving; UNICEF Uganda: New York, NY, USA, 2020; Available online: https://www.unicef.org/uganda/what-we-do/child-survival-development (accessed on 12 April 2021).
- Boschi-Pinto, C.; Youn, M.; Black, R.E. The child health epidemiology reference group reviews of the effectiveness of interventions to reduce maternal, neonatal and child mortality. Int. J. Epidemiology 2010, 39 (Suppl. 1), i3–i6. [Google Scholar] [CrossRef]
- Lamberti, L.M.; Zakarija-Grković, I.; Fischer Walker, C.L.; Theodoratou, E.; Nair, H.; Campbell, H.; Black, R.E. Breastfeeding for reducing the risk of pneumonia morbidity and mortality in children under two: A systematic literature review and meta-analysis. BMC Public Health 2013, 13, S18. [Google Scholar] [CrossRef]
- Lamberti, L.M.; Fischer Walker, C.L.; Noiman, A.; Victora, C.; Black, R.E. Breastfeeding and the risk for diarrhea morbidity and mortality. BMC Public Health 2011, 11, S15. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation. Newborns: Improving Survival and Well-Being Geneva; World Health Organisation: Geneva, Switzerland, 2020; Available online: https://www.who.int/news-room/fact-sheets/detail/newborns-reducing-mortality (accessed on 20 July 2021).
- World Health Organization. Guidelines for the Treatment of Malaria; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Hategeka, C.; Tuyisenge, G.; Bayingana, C.; Tuyisenge, L. Effects of scaling up various community-level interventions on child mortality in Burundi, Kenya, Rwanda, Uganda and Tanzania: A modeling study. Glob. Health Res. Policy 2019, 4, 1–13. [Google Scholar] [CrossRef]
- Bradley, S.E.; Rosapep, L.; Shiras, T. Where Do Caregivers Take Their Sick Children for Care? An Analysis of Care Seeking and Equity in 24 USAID Priority Countries. Glob. Health Sci. Pract. 2020, 8, 518–533. [Google Scholar] [CrossRef] [PubMed]
- Morgan, R.; Ensor, T.; Waters, H. Performance of private sector health care: Implications for universal health coverage. Lancet 2016, 388, 606–612. [Google Scholar] [CrossRef]
- Basu, S.; Andrews, J.; Kishore, S.; Panjabi, R.; Stuckler, D. Comparative Performance of Private and Public Healthcare Systems in Low- and Middle-Income Countries: A Systematic Review. PLoS Med. 2012, 9, e1001244. [Google Scholar] [CrossRef]
- Leslie, H.H.; Spiegelman, D.; Zhou, X.; Kruk, M.E. Service readiness of health facilities in Bangladesh, Haiti, Kenya, Malawi, Namibia, Nepal, Rwanda, Senegal, Uganda and the United Republic of Tanzania. Bull. World Health Organ. 2017, 95, 738–748. [Google Scholar] [CrossRef]
- Kruk, M.E.; Chukwuma, A.; Mbaruku, G.; Leslie, H.H. Variation in quality of primary-care services in Kenya, Malawi, Namibia, Rwanda, Senegal, Uganda and the United Republic of Tanzania. Bull. World Health Organ. 2017, 95, 408. [Google Scholar] [CrossRef] [PubMed]
- Waiswa, P.; Akuze, J.; Peterson, S.; Kerber, K.; Tetui, M.; Forsberg, B.C.; Hanson, C. Differences in essential newborn care at birth between private and public health facilities in eastern Uganda. Glob. Health Action 2015, 8, 24251. [Google Scholar] [CrossRef]
- World Health Organization, Organisation for Economic Co-operation and Development; International Bank for Reconstruction and Development. Delivering Quality Health Services: A global Imperative for Universal Health Coverage; Licence: CC BY-NC-SA 3.0 IGO; World Health Organization, Organisation for Economic Co-operation and Development: Geneva, Switerland; International Bank for Reconstruction and Development: Washington, DC, USA, 2018. [Google Scholar]
- Berendes, S.; Heywood, P.; Oliver, S.; Garner, P. Quality of Private and Public Ambulatory Health Care in Low and Middle Income Countries: Systematic Review of Comparative Studies. PLoS Med. 2011, 8, e1000433. [Google Scholar] [CrossRef]
- Liow, E.; Kassam, R.; Sekiwunga, R. Investigating unlicensed retail drug vendors’ preparedness and knowledge about malaria: An exploratory study in rural Uganda. Acta Trop. 2017, 174, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health Uganda. Health Sector Development Plan 2015/16–2019/20; Ministry of Health: Kampala, Uganda, 2015.
- Tawfiq, E.; Alawi, S.A.S.; Natiq, K. Effects of Training Health Workers in Integrated Management of Childhood Illness on Quality of Care for Under-5 Children in Primary Healthcare Facilities in Afghanistan. Int. J. Health Policy Manag. 2020, 9, 17–26. [Google Scholar] [CrossRef]
- Gera, T.; Shah, D.; Garner, P.; Richardson, M.; Sachdev, H.S. Integrated management of childhood illness (IMCI) strategy for children under five. Cochrane Database Syst. Rev. 2016, 22, CD010123. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Towards a Grand Convergence for Child Survival and Health: A Strategic Review of Options for the Future Building on Lessons Learnt from IMNCI; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Fick, C. Twenty years of IMCI implementation in South Africa: Accelerating impact for the next decade. S. Afr. Health Rev. 2017, 2017, 207–214. [Google Scholar]
- Kalyango, J.N.; Alfven, T.; Peterson, S.; Mugenyi, K.; Karamagi, C.; Rutebemberwa, E. Integrated community case management of malaria and pneumonia increases prompt and appropriate treatment for pneumonia symptoms in children under five years in Eastern Uganda. Malar. J. 2013, 12, 340. [Google Scholar] [CrossRef] [PubMed]
- Nsungwa-Sabiiti, J.; Burnham, G.; Pariyo, G. Implementation of a national integrated management of childhood illness (IMCI) program in Uganda. J. Health Popul. Dev. Ctries. 2004. [Google Scholar] [CrossRef]
- World Health Organisation. IMCI Chart Booklet; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- Avedis, D. The quality of care: How can it be assesses? JAMA 1988, 260, 1743. [Google Scholar]
- Creswell, J.W.; Clark, V.L.P. Designing and Conducting Mixed Methods Research; Sage Publications: New York, NY, USA, 2017. [Google Scholar]
- Creswell, J.W. Mixed-method research: Introduction and application. In Handbook of Educational Policy; Elsevier: Amsterdam, The Netherlands, 1999; pp. 455–472. [Google Scholar]
- MOH. National Health Facility Master List 2018, A complete list of All Health Facilities in Uganda, 2018 ed.; Ministry of Health: Kampala, Uganda, 2018; p. 164.
- Ministry of Health Uganda. National Policy on Public Private Partnerships in Health; Ministry of Health: Kampala, Uganda, 2015.
- Ministry of Health Uganda. National Support Supervision Guidelines for Health Services, 3rd ed.; Ministry of Health: Kampala, Uganda, 2020; p. 123.
- Pourhoseingholi, M.A.; Vahedi, M.; Rahimzadeh, M. Sample size calculation in medical studies. Gastroenterol. Hepatol. Bed Bench 2013, 6, 14–17. [Google Scholar] [PubMed]
- Arya, R.; Antonisamy, B.; Kumar, S. Sample Size Estimation in Prevalence Studies. Indian J. Pediatrics 2012, 79, 1482–1488. [Google Scholar] [CrossRef]
- Amouzou, A.; Banda, B.; Baya, B.; Black, R.; Bryce, J.; Degefie, T.; Guiella, G.; Hazel, E.; Heidkamp, R.; Hill, K.; et al. Data Associated with the Integrated Community Case Management of Childhood Illness (iCCM) Project, V8 ed.; Bryce, J., Ed.; Johns Hopkins University Data Archive: Baltimore, MD, USA, 2015. [Google Scholar]
- Creswell, J.W.; Poth, C.N. Qualitative Inquiry and Research Design: Choosing among Five Approaches; Sage Publications: New York, NY, USA, 2016. [Google Scholar]
- Morse, J.M. Determining Sample Size; Sage Publications: Thousand Oaks, CA, USA, 2000. [Google Scholar]
- Nowell, L.S.; Norris, J.M.; White, D.E.; Moules, N.J. Thematic analysis: Striving to meet the trustworthiness criteria. Int. J. Qual. Methods 2017, 16. [Google Scholar] [CrossRef]
- World Health Organisation. Standards for Improving the Quality of Care for Children and Young Adolescents in Health Facilities; World Health Organisation: Geneva, Switzerland, 2018. [Google Scholar]
- Mbonye, A.K.; Buregyeya, E.; Rutebemberwa, E.; Lal, S.; Clarke, S.E.; Hansen, K.S.; Magnussen, P.; LaRussa, P. Treatment of sick children seeking care in the private health sector in Uganda: A cluster randomized trial. Am. J. Trop. Med. Hyg. 2020, 102, 658–666. [Google Scholar] [CrossRef]
- Kjærgaard, J.; Anastasaki, M.; Stubbe Østergaard, M.; Isaeva, E.; Akylbekov, A.; Nguyen, N.Q.; Reventlow, S.; Lionis, C.; Sooronbaev, T.; Pham, L.A.; et al. Diagnosis and treatment of acute respiratory illness in children under five in primary care in low-, middle-, and high-income countries: A descriptive FRESH AIR study. PLoS ONE 2019, 14, e0221389. [Google Scholar] [CrossRef]
- O’Boyle, S.; Bruxvoort, K.J.; Ansah, E.K.; Burchett, H.E.; Chandler, C.I.; Clarke, S.E.; Goodman, C.; Mbacham, W.; Mbonye, A.K.; Onwujekwe, O.E.; et al. Patients with positive malaria tests not given artemisinin-based combination therapies: A research synthesis describing under-prescription of antimalarial medicines in Africa. BMC Med. 2020, 18, 17. [Google Scholar] [CrossRef]
- Kabaghe, A.N.; Phiri, M.D.; Phiri, K.S.; van Vugt, M. Challenges in implementing uncomplicated malaria treatment in children: A health facility survey in rural Malawi. Malar. J. 2017, 16, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Efunshile, A.M.; Ezeanosike, O.; Nwangwu, C.C.; König, B.; Jokelainen, P.; Robertson, L.J. Apparent overuse of antibiotics in the management of watery diarrhoea in children in Abakaliki, Nigeria. BMC Infect. Dis. 2019, 19, 275. [Google Scholar] [CrossRef]
- Getachew, T.; Abebe, S.M.; Yitayal, M.; Persson, L.Å.; Berhanu, D. Assessing the quality of care in sick child services at health facilities in Ethiopia. BMC Health Serv. Res. 2020, 20, 574. [Google Scholar] [CrossRef]
- Mbonye, A.K.; Awor, P.; Kayendeke, M.; Hansen, K.S.; Magnussen, P.; Clarke, S.E. Management of Childhood Infections in Poorly Planned Urban Settlements in Kampala and Wakiso Districts of Uganda. Am. J. Trop. Med. Hyg. 2020, 103, 1681–1690. [Google Scholar] [CrossRef]
- Konde-Lule, J.; Gitta, S.N.; Lindfors, A.; Okuonzi, S.; Onama, V.O.; Forsberg, B.C. Private and public health care in rural areas of Uganda. BMC Int. Health Hum. Rights 2010, 10, 29. [Google Scholar] [CrossRef]
- Buregyeya, E.; Rutebemberwa, E.; LaRussa, P.; Lal, S.; Clarke, S.E.; Hansen, K.S.; Magnussen, P.; Mbonye, A.K. Comparison of the capacity between public and private health facilities to manage under-five children with febrile illnesses in Uganda. Malar. J. 2017, 16, 183. [Google Scholar] [CrossRef][Green Version]
- Willcox, M.L.; Kumbakumba, E.; Diallo, D.; Mubangizi, V.; Kirabira, P.; Nakaggwa, F.; Mutahunga, B.; Diakité, C.; Dembélé, E.; Traoré, M.; et al. Circumstances of child deaths in Mali and Uganda: A community-based confidential enquiry. Lancet Glob. Health 2018, 6, e691–e702. [Google Scholar] [CrossRef]
- Di Giorgio, L.; Evans, D.K.; Lindelow, M.; Nguyen, S.N.; Svensson, J.; Wane, W.; Svensson, J.; Wane, W.; Tärneberg, A.W. An Analysis of Clinical Knowledge, Absenteeism, and Availability of Resources for Maternal and Child Health: A Cross-Sectional Quality of Care Study in 10 African Countries. BMJ Glob. Health 2020, 5, e003377. [Google Scholar] [CrossRef]
- Brunie, A.; Lenzi, R.; Lahiri, A.; Izadnegahdar, R. Leveraging the private sector for child health: A qualitative examination of caregiver and provider perspectives on private sector care for childhood pneumonia in Uttar Pradesh, India. BMC Health Serv. Res. 2017, 17, 159. [Google Scholar] [CrossRef]
- Farooqui, H.H.; Mehta, A.; Selvaraj, S. Outpatient antibiotic prescription rate and pattern in the private sector in India: Evidence from medical audit data. PLoS ONE 2019, 14, e0224848. [Google Scholar] [CrossRef]
- Maina, M.; Mwaniki, P.; Odira, E.; Kiko, N.; McKnight, J.; Schultsz, C.; English, M.; Tosas-Auguet, O. Antibiotic use in Kenyan public hospitals: Prevalence, appropriateness and link to guideline availability. Int. J. Infect. Dis. 2020, 99, 10–18. [Google Scholar] [CrossRef]
| Characteristic | Number |
|---|---|
| Population (inhabitants) | 470,000 |
| Population density/km | 99 |
| Administrative divisions | |
| Counties | 3 |
| Sub-counties | 16 |
| Parishes | 83 |
| Villages | 742 |
| Health facilities | |
| Hospitals | 7 |
| Health centers | 49 |
| Registered private health facilities (in 2017) | 124 |
| Characteristic | Number of Consultations n (%) |
|---|---|
| Health facilities | |
| Level of health facility (N = 777) | |
| Below HCII | 232 (30) |
| HCII | 337 (43) |
| HCIII | 208 (27) |
| Location of health facility (N = 777) | |
| Urban | 311 (40) |
| Rural | 466 (60) |
| Professional qualification of the health facility head (N = 776) | |
| Doctor | 79 (10) |
| Clinical officer | 310 (40) |
| Nurse/midwife | 292 (38) |
| Nursing assistant | 92 (12) |
| Laboratory technician | 3 (0.4) |
| Health facility licensing body (N = 776) | |
| Uganda Medical and Dental Practitioners Council | 92 (12) |
| Allied Health Professionals Council | 377 (49) |
| Uganda Nurses and Midwives Council | 108 (14) |
| Unregistered | 199 (26) |
| Healthcare workers, (N = 776) | |
| Males | 353 (45.5) |
| Females | 423 (54.5) |
| Working only in private health facilities | 704 (90.7) |
| Working in two or more health facilities | 229 (29.5) |
| Characteristics | Summary Statistics |
|---|---|
| Children | |
| Sex (N = 777) | |
| Male, n (%) | 376 (48.4) |
| Female, n (%) | 401 (51.6) |
| Mean age in months, (SD) | 24.0 (16.0) |
| Mean weight in kg, (SD) | 11.1 (3.7) |
| Mean height/length in cm, (SD) | 75.7 (20.9) |
| Temperature ≥37.5 °C, n (%) | 390 (60.3) |
| Caretakers | |
| Relationship with the sick child, n (%) | |
| Biological parent | 695 (89.4) |
| Sibling | 2 (0.3) |
| Others (grandparents, aunts, uncles) | 80 (10.3) |
| Sex, n (%) | |
| Male | 130 (16.7) |
| Female | 647 (83.3) |
| Age, mean (SD) | 30 (8.0) |
| Residence, n (%) | |
| Rural | 481 (61.9) |
| Urban | 296 (38.1) |
| Level of education, n (%) | |
| None | 72 (9.3) |
| Primary | 366 (47.1) |
| Secondary | 230 (29.6) |
| Tertiary | 109 (14.0) |
| Marital status, n (%) | |
| Single/divorced/widowed | 160 (20.6) |
| Married/living with a partner | 617 (79.4) |
| Median monthly income in Uganda shillings, (IQR) | 100,000 (30,000, 300,000) |
| Condition, n (%) | ||||
|---|---|---|---|---|
| Pneumonia | Diarrhea | Malaria | Possible Serious Bacterial Infection | |
| Correct identification | ||||
| No | 62 (15) | 35 (16) | 142 (27) | 17 (94) |
| Yes | 360 (85) | 183 (84) | 390 (73) | 1 (5.6) |
| Correct classification | ||||
| No | 44 (10) | 57 (26) | 350 (66) | 17 (94) |
| Yes | 378 (90) | 161 (74) | 182 (34) | 1 (5.6) |
| Correct treatment | ||||
| No | 367 (87) | 123 (56) | 446 (84) | 12 (67) |
| Yes | 55 (13) | 95 (44) | 86 (16) | 6 (33) |
| Appropriately managed overall | ||||
| No | 393 (89) | 204 (92) | 457 (86) | 18 (100) |
| Yes | 49 (11) | 18 (8.1) | 75 (14) | 0 (0) |
| % Appropriate Care | Univariable Analysis | Multivariable Analysis | |||
|---|---|---|---|---|---|
| Characteristic | n/N (%) | cOR (95%CI) | p-Value | aOR (95%CI) | p-Value |
| Caretaker’s age | |||||
| <25 years | 7/154 (4.6) | Ref | Ref | ||
| ≥25 years | 71/614 (11.6) | 2.75 (1.24–6.10) | 0.013 | 1.95 (0.71–5.37) | 0.194 |
| County of H/F location | |||||
| Rwampara | 9/177 (5.1) | Ref | |||
| Kashari | 40/238 (16.8) | 3.77 (1.78–8.00) | 0.001 | ||
| Municipality | 29/326 (8.0) | 1.63 (0.75–3.51) | 0.216 | ||
| Location | |||||
| Urban | 22/311 (7.1) | Ref | Ref | ||
| Rural | 56/466 (12.0) | 1.79 (1.07–3.00) | 0.026 | 1.09 (0.62–1.93) | 0.756 |
| Time lag to nearest public H/Facility | |||||
| ≤15 min | 44/550 (8.0) | Ref | Ref | ||
| >15 min | 34/226 (15.0) | 2.04 (1.26–3.28) | 0.004 | 2.50 (1.23–5.10) | 0.012 |
| Residence of caretaker | |||||
| Rural | 58/481 (12.1) | Ref | Ref | ||
| Urban | 20/296 (6.8) | 0.53 (0.31–0.90) | 0.018 | 0.58 (0.23–1.45) | 0.247 |
| Temperature of child | |||||
| <37.5 °C | 17/257 (6.6) | Ref | |||
| ≥37.5 °C | 52/390 (13.3) | 2.17 (1.23–3.85) | 0.008 | ||
| Presence of fast breathing | |||||
| No | 38/562 (6.8) | Ref | Ref | ||
| Yes | 40/215 (18.6) | 3.15 (1.96–5.07) | <0.001 | 1.77 (0.95–3.12) | 0.072 |
| Presence of diarrhea | |||||
| No | 62/561 (11.1) | Ref | Ref | ||
| Yes | 16/216 (7.4) | 0.65 (0.36–1.15) | 0.137 | 0.29 (0.11–0.76) | 0.012 |
| Child able to drink | |||||
| No | 43/84 (51.2) | Ref | Ref | ||
| Yes | 35/693 (5.1) | 0.05 (0.03–0.09) | <0.001 | 0.07 (0.03–0.13) | <0.001 |
| Child vomiting everything | |||||
| No | 50/717 (7.0) | Ref | Ref | ||
| Yes | 28/58 (48.3) | 12.45 (6.90–22.46) | <0.001 | 10.1 (4.41–22.1) | <0.001 |
| History of convulsions | |||||
| No | 57/741 (7.7) | Ref | Ref | ||
| Yes | 21/34 (61.8) | 19.38 (9.22–40.73) | <0.001 | 7.54 (2.97–19.1) | <0.001 |
| Level of consciousness | |||||
| Awake | 69/763 (9.0) | Ref | Ref | ||
| Lethargic or unconscious | 9/14 (64.3) | 18.10 (5.90–55.53) | <0.001 | 4.42 (1.03–19.1) | 0.046 |
| Neck rigidity | |||||
| No | 76/773 (9.8) | Ref | |||
| Yes | 2/4 (50.0) | 9.17 (1.27–66.04) | 0.028 | ||
| Chest in drawing | |||||
| No | 55/664 (8.3) | Ref | |||
| Yes | 23/113 (20.4) | 2.82 (1.66–4.83) | <0.001 | ||
| Presence of stridor | |||||
| No | 72/753 (9.6) | Ref | |||
| Yes | 6/24 (25.0) | 3.15 (1.21–8.20) | 0.018 | ||
| Sunken eyes/signs of dehydration | |||||
| No | 66/712 (9.3) | Ref | Ref | ||
| Yes | 12/65 (18.5) | 2.21 (1.13–4.36) | 0.021 | 2.07 (0.67–6.33) | 0.204 |
| Age of Health worker | |||||
| <25 years | 13/202 (6.4) | Ref | |||
| 25–40 years | 57/502 (11.4) | 1.86 (1.00–3.48) | 0.052 | ||
| >40 years | 8/72 (11.1) | 1.82 (0.72–4.58) | 0.206 | ||
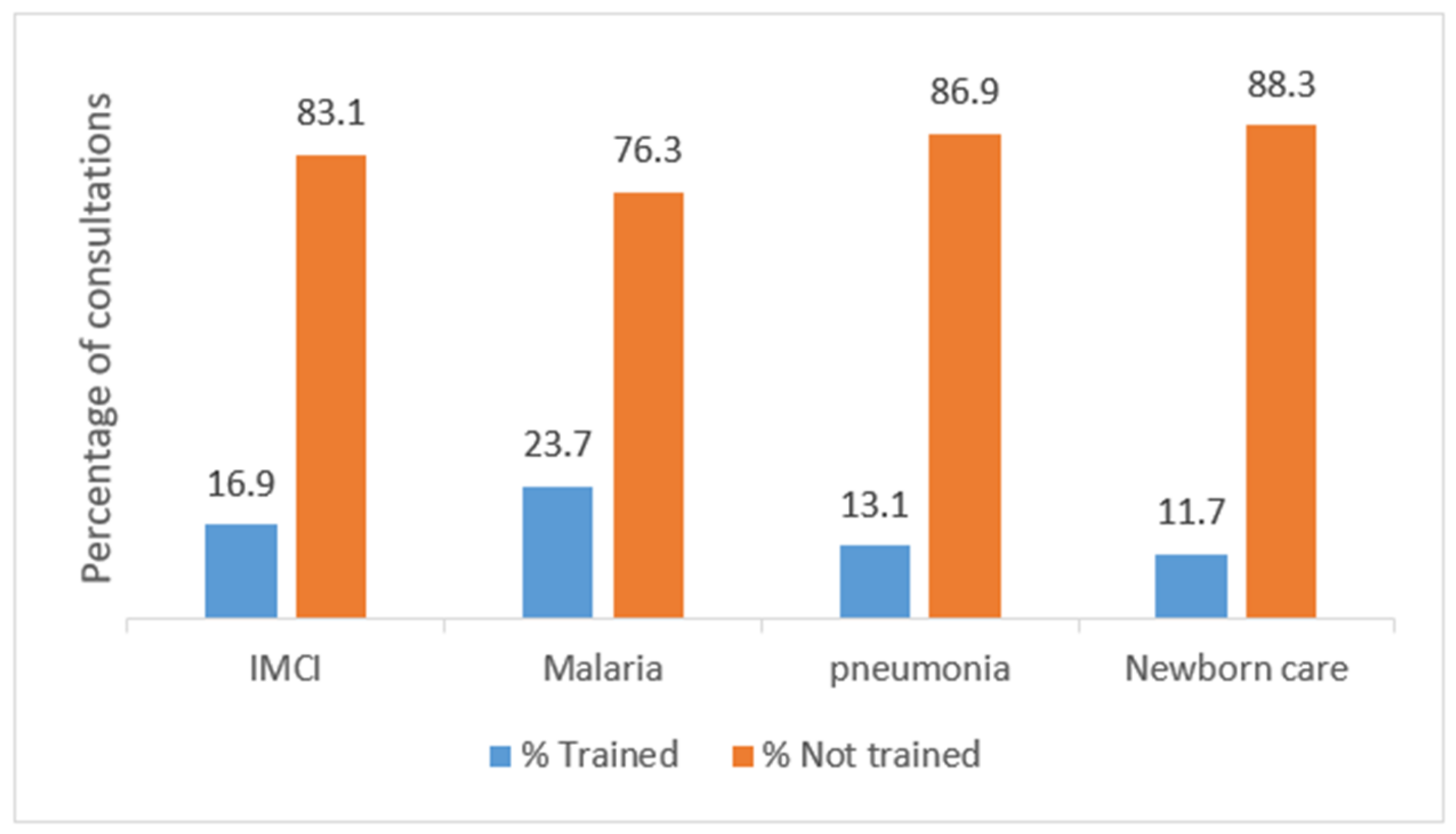
| Trained on management of malaria | |||||
| No | 55/592 (9.3) | Ref | |||
| Yes | 23/184 (12.5) | 1.39 (0.83–2.34) | 0.208 | ||
| Trained on management of pneumonia | |||||
| No | 62/675 (9.2) | Ref | |||
| Yes | 16/102 (15.7) | 1.84 (1.02–3.33) | 0.044 | ||
| Trained on management of bacterial infections | |||||
| No | 64/689 (9.3) | Ref | |||
| Yes | 14/88 (15.9) | 1.85 (0.99–3.46) | 0.055 | ||
| Trained on IMCI guidelines | |||||
| No | 64/646 (9.9) | Ref | |||
| Yes | 14/131 (10.7) | 1.09 (0.59–2.01) | 0.787 | ||
| Ever received refresher training | |||||
| No | 51/555 (9.2) | Ref | Ref | ||
| Yes | 27/222 (12.2) | 1.37 (0.83–2.24) | 0.149 | 1.50 (0.77–2.94) | 0.237 |
| Health worker professional qualification | |||||
| Nurse/midwife | 46/460 (10.0) | Ref | |||
| Doctor/clinical officer | 18/191 (4.2) | 0.94 (0.54–1.66) | 0.822 | ||
| Nursing assistant | 13/120 (16.7) | 1.09 (0.57–2.10) | 0.788 | ||
| Others | 1/6 (1.3) | 1.80 (0.21–15.7) | 0.595 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mwanga-Amumpaire, J.; Alfvén, T.; Obua, C.; Källander, K.; Migisha, R.; Stålsby Lundborg, C.; Ndeezi, G.; Kalyango, J.N. Appropriateness of Care for Common Childhood Infections at Low-Level Private Health Facilities in a Rural District in Western Uganda. Int. J. Environ. Res. Public Health 2021, 18, 7742. https://doi.org/10.3390/ijerph18157742
Mwanga-Amumpaire J, Alfvén T, Obua C, Källander K, Migisha R, Stålsby Lundborg C, Ndeezi G, Kalyango JN. Appropriateness of Care for Common Childhood Infections at Low-Level Private Health Facilities in a Rural District in Western Uganda. International Journal of Environmental Research and Public Health. 2021; 18(15):7742. https://doi.org/10.3390/ijerph18157742
Chicago/Turabian StyleMwanga-Amumpaire, Juliet, Tobias Alfvén, Celestino Obua, Karin Källander, Richard Migisha, Cecilia Stålsby Lundborg, Grace Ndeezi, and Joan Nakayaga Kalyango. 2021. "Appropriateness of Care for Common Childhood Infections at Low-Level Private Health Facilities in a Rural District in Western Uganda" International Journal of Environmental Research and Public Health 18, no. 15: 7742. https://doi.org/10.3390/ijerph18157742
APA StyleMwanga-Amumpaire, J., Alfvén, T., Obua, C., Källander, K., Migisha, R., Stålsby Lundborg, C., Ndeezi, G., & Kalyango, J. N. (2021). Appropriateness of Care for Common Childhood Infections at Low-Level Private Health Facilities in a Rural District in Western Uganda. International Journal of Environmental Research and Public Health, 18(15), 7742. https://doi.org/10.3390/ijerph18157742







