Allostatic Load and Exposure Histories of Disadvantage
Abstract
1. Introduction
Background
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Guthman, J.; Mansfield, B. The implications of environmental epigenetics: A new direction for geographic inquiry on health, space, and nature-society relations. Prog. Hum. Geogr. 2012, 37, 486–504. [Google Scholar] [CrossRef]
- Krieger, N. Epidemiology and the web of causation: Has anyone seen the spider? Soc. Sci. Med. 1994, 39, 887–903. [Google Scholar] [CrossRef]
- Prior, L.; Manley, D.; Sabel, C.E. Biosocial health geography: New “exposomic” geographies of health and place. Prog. Hum. Geogr. 2018, 43, 531–552. [Google Scholar] [CrossRef]
- Taylor, S.E.; Repetti, R.L.; Seeman, T. Health psychology: What is an unhealthy environment and how does it get under the skin? Annu. Rev. Psychol. 1997, 48, 411–447. [Google Scholar] [CrossRef]
- Delpierre, C.; Barbosa-Solis, C.; Torrisani, J.; Darnaudery, M.; Bartley, M.; Blane, D.; Kelly-Irving, M.; Getz, L.; Tomasdottir, M.O.; Roberston, T.; et al. Origins of heath inequalities: The case for Allostatic Load. Longit. Life Course Stud. 2016, 7, 79–103. [Google Scholar] [CrossRef]
- Boardman, J.D. Stress and physical health: The role of neighborhoods as mediating and moderating mechanisms. Soc. Sci. Med. 2004, 58, 2473–2483. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S. Allostasis and the epigenetics of brain and body health over the life course: The brain on stress. JAMA Psychiatry 2017, 74, 551–552. [Google Scholar] [CrossRef]
- Dulin-Keita, A.; Casazza, K.; Fernandez, J.R.; Goran, M.I.; Gower, B. Do neighbourhoods matter? Neighbourhood disorder and long-term trends in serum cortisol levels. J. Epidemiol. Community Health 2012, 66, 24–29. [Google Scholar] [CrossRef]
- Robinette, J.W.; Charles, S.T.; Gruenewald, T.L. Neighborhood cohesion, neighborhood disorder, and cardiometabolic risk. Soc. Sci. Med. 2018, 198, 70–76. [Google Scholar] [CrossRef]
- Ross, C.E.; Mirowsky, J. Neighbourhood disadvantage, disorder, and health. J. Health Soc. Behav. 2001, 42, 258–276. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S. Central effects of stress hormones in health and disease: Understanding the protective and damaging effects of stress and stress mediators. Eur. J. Pharmacol. 2008, 583, 174–185. [Google Scholar] [CrossRef]
- McEwen, B.S.; Seeman, T. Protective and damaging effects of mediators of stress: Elaborating and testing the concepts of allostasis and allostatic load. Ann. N. Y. Acad. Sci. 1999, 896, 30–47. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S.; Stellar, E. Stress and the individual: Mechanisms leading to disease. Arch. Intern. Med. 1993, 153, 2093–2101. [Google Scholar] [CrossRef] [PubMed]
- De Vries, S.; Van Dillen, S.M.E.; Groenewegen, P.P.; Spreeuwenberg, P. Streetscape greenery and health: Stress, social cohesion and physical activity as mediators. Soc. Sci. Med. 2013, 94, 26–33. [Google Scholar] [CrossRef]
- Finlay, J.; Franke, T.; McKay, H.; Sims-Gould, J. Therapeutic landscapes and wellbeing in later life: Impacts of blue and green spaces for older adults. Health Place 2015, 34, 97–106. [Google Scholar] [CrossRef]
- Hordyk, S.R.; Hanley, J.; Richard, É. “Nature is there; its free”: Urban greenspace and the social determinants of health of immigrant families. Health Place 2015, 34, 74–82. [Google Scholar] [CrossRef]
- Kawachi, I.; Berkman, L.F. Social ties and mental health. J. Urban Health 2001, 78, 458–467. [Google Scholar] [CrossRef]
- Uphoff, E.P.; Pickett, K.E.; Cabieses, B.; Small, N.; Wright, J. A systematic review of the relationships between social capital and socioeconomic inequalities in health: A contribution to understanding the psychosocial pathway of health inequalities. Int. J. Equity Health 2013, 12, 54. [Google Scholar] [CrossRef]
- Wild, C.P. The exposome: From concept to utility. Int. J. Epidemiol. 2012, 41, 24–32. [Google Scholar] [CrossRef]
- Wild, C.P. Complementing the genome with an “exposome”: The outstanding challenge of environmental exposure measurement in molecular epidemiology. Cancer Epidemiol. Biomark. Prev. 2005, 14, 1847–1850. [Google Scholar] [CrossRef] [PubMed]
- Jacquez, G.M.; Sabel, C.E.; Shi, C. Genetic GIScience: Toward a place-based synthesis of the genome, exposome, and behavome. Ann. Assoc. Am. Geogr. 2015, 105, 454–472. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Harford, J.; Schuch, H.S.; Watt, R.G.; Peres, M.A. Theoretical basis and explanation for the relationship between area-level social inequalities and population oral health outcomes—A scoping review. SSM Popul. Health 2016, 2, 451–462. [Google Scholar] [CrossRef][Green Version]
- Wilkinson, R.G.; Pickett, K.E. The problems of relative deprivation: Why some societies do better than others. Soc. Sci. Med. 2007, 65, 1965–1978. [Google Scholar] [CrossRef]
- Barrington, W.E.; Stafford, M.; Hamer, M.; Beresford, S.A.A.; Koepsell, T.; Steptoe, A. Neighborhood socioeconomic deprivation, perceived neighborhood factors, and cortisol responses to induced stress among healthy adults. Health Place 2014, 27, 120–126. [Google Scholar] [CrossRef]
- Hajat, A.; Moore, K.; Phuong Do, D.; Stein Merkin, S.; Punjabi, N.M.; Sáñchez, B.N.; Seeman, T.; Diez-Roux, A.V. Examining the cross-sectional and longitudinal association between diurnal cortisol and neighborhood characteristics: Evidence from the multi-ethnic study of atherosclerosis. Health Place 2015, 34, 199–206. [Google Scholar] [CrossRef]
- Rudolph, K.E.; Wand, G.S.; Stuart, E.A.; Glass, T.A.; Marques, A.H.; Duncko, R.; Merikangas, K.R. The association between cortisol and neighborhood disadvantage in a U.S. population-based sample of adolescents. Health Place 2014, 25, 68–77. [Google Scholar] [CrossRef][Green Version]
- Dowd, J.B.; Simanek, A.M.; Aiello, A.E. Socio-economic status, cortisol and allostatic load: A review of the literature. Int. J. Epidemiol. 2009, 38, 1297–1309. [Google Scholar] [CrossRef] [PubMed]
- Geronimus, A.T. The weathering hypothesis and the health of African-American women and infants: Evidence and speculations. Ethn. Dis. 1992, 2, 207–221. [Google Scholar] [PubMed]
- Geronimus, A.T.; Pearson, J.A.; Linnenbringer, E.; Schulz, A.J.; Reyes, A.G.; Epel, E.S.; Lin, J.; Blackburn, E.H. Race-ethnicity, poverty, urban stressors, and telomere length in a Detroit community-based sample. J. Health Soc. Behav. 2015, 56, 199–224. [Google Scholar] [CrossRef]
- Geronimus, A.T.; Hicken, M.T.; Pearson, J.A.; Seashols, S.J.; Brown, K.L.; Cruz, T.D. Do US black women experience stress-related accelerated biological aging? A novel theory and first population-based test of black-white differences in telomere length. Hum. Nat. 2010, 21, 19–38. [Google Scholar] [CrossRef] [PubMed]
- Juster, R.-P.; McEwen, B.S.; Lupien, S.J. Allostatic load biomarkers of chronic stress and impact on health and cognition. Neurosci. Biobehav. Rev. 2010, 35, 2–16. [Google Scholar] [CrossRef]
- Mattei, J.; Demissie, S.; Falcon, L.M.; Ordovas, J.M.; Tucker, K. Allostatic load is associated with chronic conditions in the Boston Puerto Rican Health Study. Soc. Sci. Med. 2010, 70, 1988–1996. [Google Scholar] [CrossRef]
- Seeman, T.E.; McEwen, B.S.; Rowe, J.W.; Singer, B.H. Allostatic load as a marker of cumulative biological risk: MacArthur studies of successful aging. Proc. Natl. Acad. Sci. USA 2001, 98, 4770–4775. [Google Scholar] [CrossRef]
- Seeman, T.E.; Singer, B.H.; Rowe, J.W.; Horwitz, R.I.; McEwen, B.S. Price of adaptation—Allostatic load and it’s health consequences: MacArthur Studies of Successful Aging. Arch. Intern. Med. 1997, 157, 2259–2268. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.C.; Cavallaro, F.L.; Leon, D.A. A systematic review of allostatic load in relation to socioeconomic position: Poor fidelity and major inconsistencies in biomarkers employed. Soc. Sci. Med. 2017, 192, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Van Ham, M.; Manley, D.; Bailey, N.; Simpson, L.; Maclennan, D. Neighborhood Effects Research: New Perspectives; Springer: Dordrecht, The Netherlands, 2012; Volume 91, ISBN 9789400723085. [Google Scholar]
- Bellatorre, A.; Finch, B.K.; Phuong Do, D.; Bird, C.E.; Beck, A.N. Contextual predictors of cumulative biological risk: Segregation and allostatic load. Soc. Sci. Quaterly 2011, 92, 1338–1362. [Google Scholar] [CrossRef]
- Bird, C.E.; Seeman, T.; Escarce, J.J.; Basurto-Dávila, R.; Finch, B.K.; Dubowitz, T.; Heron, M.; Hale, L.; Stein Merkin, S.; Weden, M.; et al. Neighbourhood socioeconomic status and biological “wear and tear” in a nationally representative sample of US adults. J. Epidemiol. Community Health 2010, 64, 860–865. [Google Scholar] [CrossRef] [PubMed]
- Mair, C.A.; Cutchin, M.P.; Kristen Peek, M. Allostatic load in an environmental riskscape: The role of stressors and gender. Health Place 2011, 17, 978–987. [Google Scholar] [CrossRef] [PubMed]
- Schulz, A.J.; Mentz, G.; Lachance, L.; Zenk, S.N.; Johnson, J.; Stokes, C.; Mandell, R. Do observed or perceived characteristics of the neighborhood environment mediate associations between neighborhood poverty and cumulative biological risk? Health Place 2013, 24, 147–156. [Google Scholar] [CrossRef]
- Schulz, A.J.; Mentz, G.; Lachance, L.; Johnson, J.; Gaines, C.; Israel, B.A. Associations between socioeconomic status and allostatic load: Effects of neighborhood poverty and tests of mediating pathways. Am. J. Public Health 2012, 102, 1706–1714. [Google Scholar] [CrossRef] [PubMed]
- Seeman, M.; Stein Merkin, S.; Karlamangla, A.; Koretz, B.; Seeman, T. Social status and biological dysregulation: The “status syndrome” and allostatic load. Soc. Sci. Med. 2014, 118, 143–151. [Google Scholar] [CrossRef]
- Stein Merkin, S.; Basurto-Dávila, R.; Karlamangla, A.; Bird, C.E.; Lurie, N.; Escarce, J.; Seeman, T. Neighborhoods and cumulative biological risk profiles by Race/Ethnicity in a national sample of U.S. adults: NHANES III. Ann. Epidemiol. 2009, 19, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Theall, K.P.; Drury, S.S.; Shirtcliff, E.A. Cumulative neighborhood risk of psychosocial stress and allostatic load in adolescents. Am. J. Epidemiol. 2012, 176 (Suppl. 7), S164–S174. [Google Scholar] [CrossRef]
- Prior, L.; Manley, D.; Jones, K. Stressed out? An investigation of whether allostatic load mediates associations between neighbourhood deprivation and health. Health Place 2018, 52, 25–33. [Google Scholar] [CrossRef]
- Ribeiro, A.I.; Amaro, J.; Lisi, C.; Fraga, S. Neighborhood socioeconomic deprivation and allostatic load: A scoping review. Int. J. Environ. Res. Public Health 2018, 15, 1092. [Google Scholar] [CrossRef]
- Xu, H. Multilevel socioeconomic differentials in allostatic load among Chinese adults. Health Place 2018, 53, 182–192. [Google Scholar] [CrossRef]
- Jiménez, M.P.; Osypuk, T.L.; Arevalo, S.; Tucker, K.L.; Falcon, L.M. Neighborhood socioeconomic context and change in allostatic load among older Puerto Ricans: The Boston Puerto Rican health study. Health Place 2015, 33, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Barker, D.J.P. Fetal origins of coronary heart disease. Br. Med. J. 1995, 311, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Barker, D.J.P.; Eriksson, J.G.; Forsen, T.; Osmond, C. Fetal origins of adult disease: Strength of effects and biological basis. Int. J. Epidemiol. 2002, 31, 1235–1239. [Google Scholar] [CrossRef]
- Barboza Solís, C.; Kelly-Irving, M.; Fantin, R.; Darnaudéry, M.; Torrisani, J.; Lang, T.; Delpierre, C. Adverse childhood experiences and physiological wear-and-tear in midlife: Findings from the 1958 British birth cohort. Proc. Natl. Acad. Sci. USA 2015, 112, E738–E746. [Google Scholar] [CrossRef] [PubMed]
- Barboza Solís, C.; Fantin, R.; Castagné, R.; Lang, T.; Delpierre, C.; Kelly-Irving, M. Mediating pathways between parental socio-economic position and allostatic load in mid-life: Findings from the 1958 British birth cohort. Soc. Sci. Med. 2016, 165, 19–27. [Google Scholar] [CrossRef]
- Friedman, E.M.; Karlamangla, A.S.; Gruenewald, T.L.; Koretz, B.; Seeman, T.E. Early life adversity and adult biological risk profiles. Psychosom. Med. 2015, 77, 176–185. [Google Scholar] [CrossRef]
- Cerin, E. Ways of unraveling how and why physical activity influences mental health through statistical mediation analyses. Ment. Health Phys. Act. 2010, 3, 51–60. [Google Scholar] [CrossRef]
- Walsemann, K.M.; Goosby, B.J.; Farr, D. Life course SES and cardiovascular risk: Heterogeneity across race/ethnicity and gender. Soc. Sci. Med. 2016, 152, 147–155. [Google Scholar] [CrossRef]
- Yang, Y.C.; Gerken, K.; Schorpp, K.; Boen, C.; Harris, K.M. Early-life socioeconomic status and adult physiological functioning: A life course examination of biosocial mechanisms. Biodemography Soc. Biol. 2017, 63, 87–103. [Google Scholar] [CrossRef]
- Gustafsson, P.E.; Janlert, U.; Theorell, T.; Westerlund, H.; Hammarström, A. Social and material adversity from adolescence to adulthood and allostatic load in middle-aged women and men: Results from the Northern Swedish Cohort. Ann. Behav. Med. 2012, 43, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Lemelin, E.T.; Diez Roux, A.V.; Franklin, T.G.; Carnethon, M.; Lutsey, P.L.; Ni, H.; O’Meara, E.; Shrager, S. Life-course socioeconomic positions and subclinical atherosclerosis in the multi-ethnic study of atherosclerosis. Soc. Sci. Med. 2009, 68, 444–451. [Google Scholar] [CrossRef]
- Gustafsson, P.E.; Sebastian, M.S.; Janlert, U.; Theorell, T.; Westerlund, H.; Hammarström, A. Life-course accumulation of neighborhood disadvantage and allostatic load: Empirical integration of three social determinants of health frameworks. Am. J. Public Health 2014, 104, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Stafford, M.; Gardner, M.; Kumari, M.; Kuh, D.; Ben-Shlomo, Y. Social isolation and diurnal cortisol patterns in an ageing cohort. Psychoneuroendocrinology 2013, 38, 2737–2745. [Google Scholar] [CrossRef][Green Version]
- Horan, J.M.; Widom, C.S. From childhood maltreatment to allostatic load in adulthood: The role of social support. Child Maltreat. 2015, 20, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Gruenewald, T.L.; Karlamangla, A.S.; Hu, P.; Stein Merkin, S.; Crandall, C.; Koretz, B.; Seeman, T.E. History of socioeconomic disadvantage and allostatic load in later life. Soc. Sci. Med. 2012, 74, 75–83. [Google Scholar] [CrossRef]
- Lin, Y.-H.; Jen, M.-H.; Chien, K.-L. Association between life-course socioeconomic position and inflammatory biomarkers in older age: A nationally representative cohort study in Taiwan. BMC Geriatr. 2017, 17, 1–11. [Google Scholar] [CrossRef] [PubMed]
- University of Essex; Institute for Social and Economic Research; NatCen Social Research; Kantar Public. Understanding Society: Waves 1–7, 2009–2016 and Harmonised BHPS: Waves 1–18, 1991–2009, [Data Collection], 10th ed.; UK Data Service. SN: 6614; 2018. [Google Scholar] [CrossRef]
- Benzeval, M.; Davillas, A.; Kumari, M.; Lynn, P. Understanding Society: The UK Household Longitudinal Study Biomarker User Guide and Glossary; University of Essex: Colchester, UK, 2014. [Google Scholar]
- McFall, S.; Petersen, J.; Kaminska, O.; Lynn, P. Understanding Society: The UK Household Longitudinal Study Waves 2 and 3 Nurse Health Assessment, 2010–2012 Guide to Nurse Health Assessment; University of Essex: Colchester, UK, 2014. [Google Scholar]
- University of Essex; Institute for Social and Economic Research; NatCen Social Research. Understanding Society: Waves 2 and 3 Nurse Health Assessment, 2010–2012, [data collection], 3rd ed.; UK Data Service. SN: 7251; 2014. [Google Scholar] [CrossRef]
- Townsend, P. Deprivation. J. Soc. Policy 1987, 16, 125–146. [Google Scholar] [CrossRef]
- Norman, P. The changing geography of deprivation in Britain: 1971 to 2011 and beyond. In Population Change in the United Kingdom; Champion, T., Falkingham, J., Eds.; Rowman & Littlefield: London, UK, 2016; pp. 193–214. [Google Scholar]
- Norman, P. Identifying change over time in small area socio-economic deprivation. Appl. Spat. Anal. Policy 2010, 3, 107–138. [Google Scholar] [CrossRef]
- Norman, P.; Darlington-Pollock, F. The changing geography of deprivation in Great Britain: Exploiting small area Census data, 1971 to 2011. In The Routledge Handbook of Census Resources, Methods and Applications: Unlocking the UK 2011 Census; Stillwell, J., Ed.; Routledge: London, UK, 2017; pp. 404–420. [Google Scholar]
- University of Essex; Institute for Social and Economic Research; NatCen Social Research; Kantar Public. Understanding Society: Waves 1–7, 2009–2016: Special Licence Access, Census 2011 Lower Layer Super Output Areas, [Data Collection], 7th ed.; UK Data Service. SN: 7248; 2018. [Google Scholar] [CrossRef]
- University of Essex; Institute for Social and Economic Research. British Household Panel Survey, Waves 1–18, 1991–2009: Special Licence Access, Lower Layer Super Output Areas and Scottish Data Zones, [Data Collection], 3rd ed.; UK Data Service. SN: 6136; 2014. [Google Scholar] [CrossRef]
- Office for National Statistics Lower Layer Super Output Area (2001) to Lower Layer Super Output Area (2011) to Local Authority District (2011) Lookup in England and Wales. Available online: http://geoportal.statistics.gov.uk/datasets/lower-layer-super-output-area-2001-to-lower-layer-super-output-area-2011-to-local-authority-district-2011-lookup-in-england-and-wales (accessed on 1 February 2019).
- Scottish Government Data Zone Centroids. 2001. Available online: https://data.gov.uk/dataset/d9e9b1c9-aa77-4fa6-b8a7-65621a463fa0/data-zone-centroids-2001 (accessed on 25 February 2019).
- Scottish Government Data Zone Boundaries. 2011. Available online: https://data.gov.uk/dataset/ab9f1f20-3b7f-4efa-9bd2-239acf63b540/data-zone-boundaries-2011 (accessed on 25 February 2019).
- Jung, T.; Wickrama, K.A.S. An introduction to latent class growth analysis and growth mixture modeling. Soc. Personal. Psychol. Compass 2008, 2, 302–317. [Google Scholar] [CrossRef]
- Bakk, Z.; Vermunt, J.K. Robustness of stepwise latent class modeling with continuous distal outcomes. Struct. Equ. Model. A Multidiscip. J. 2016, 23, 20–31. [Google Scholar] [CrossRef]
- Dziak, J.J.; Bray, B.C.; Zhang, J.; Zhang, M.; Lanza, S.T. Comparing the performance of improved classify-analyze approaches for distal outcomes in latent profile analysis. Methodology 2016, 12, 107–116. [Google Scholar] [CrossRef]
- Vermunt, J.K. Latent class modeling with covariates: Two improved three-step approaches. Polit. Anal. 2010, 18, 450–469. [Google Scholar] [CrossRef]
- Bolck, A.; Croon, M.A.; Hagenaars, J.A. Estimating latent structure models with categorical variables: One-step versus three-step estimators. Polit. Anal. 2004, 12, 3–27. [Google Scholar] [CrossRef]
- Asparouhov, T.; Muthén, B. Auxiliary variables in mixture modeling: Using the BCH Method in Mplus to estimate a distal outcome model and an arbitrary secondary model. Mplus Web Notes 2018, 21, 1–23. [Google Scholar]
- StataCorp. Stata Statistical Software: Release 15; StataCorp LLC.: College Station, TX, USA, 2017. [Google Scholar]
- Muthén, L.K.; Muthén, B.O. Mplus User’s Guide, 7th ed.; Muthén & Muthén: Los Angeles, CA, USA, 2015. [Google Scholar]
- Lo, Y.; Mendell, N.R.; Rubin, D.B. Testing the number of components in a normal mixture. Biometrika 2001, 88, 767–778. [Google Scholar] [CrossRef]
- Norman, P. The Changing Geography of Deprivation in Britain: Exploiting Small Area Census Data 1971 to 2011. In Proceedings of the 23rd GIS Research UK Conference (GISRUK 2015), Leeds, UK, 5–17 April 2015; pp. 465–474. [Google Scholar]
- Blair, A.; Gariépy, G.; Schmitz, N. The longitudinal effects of neighbourhood social and material deprivation change on psychological distress in urban, community-dwelling Canadian adults. Public Health 2015, 129, 932–940. [Google Scholar] [CrossRef]
- Van Ham, M.; Hedman, L.; Manley, D.; Coulter, R.; Östh, J. Intergenerational transmission of neighbourhood poverty: An analysis of neighbourhood histories of individuals. Trans. Inst. Br. Geogr. 2014, 39, 402–417. [Google Scholar] [CrossRef] [PubMed]
- Kontopantelis, E.; Mamas, M.A.; Van Marwijk, H.; Ryan, A.M.; Buchan, I.E.; Ashcroft, D.M.; Doran, T. Geographical epidemiology of health and overall deprivation in England, its changes and persistence from 2004 to 2015: A longitudinal spatial population study. J. Epidemiol. Community Health 2018, 72, 140–147. [Google Scholar] [CrossRef]
- Meen, G.; Nygaard, C.; Meen, J. The causes of long-term neighbourhood change. In Understanding Neighbourhood Dynamics: New Insights for Neighbourhood Effects Research; Van Ham, M., Manley, D., Bailey, N., Simpson, L., Maclennan, D., Eds.; Springer: Dordrecht, The Netherlands, 2013; pp. 43–62. ISBN 9789400748538. [Google Scholar]
- Clark, W.A.V.; Van Ham, M.; Coulter, R. Spatial mobility and social outcomes. J. Hous. Built Environ. 2014, 29, 699–727. [Google Scholar] [CrossRef]
- Sharkey, P. Stuck in Place: Urban Neighborhoods and the End of Progress toward Racial Equality; University of Chicago Press: Chicago, IL, USA, 2013. [Google Scholar]
- Glass, T.A.; Bilal, U. Are neighborhoods causal? Complications arising from the ‘stickiness’ of ZNA. Soc. Sci. Med. 2016, 166, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Marmot, M. Fair Society, Healthy Lives: The Marmot Review Executive Summary; The Marmot Review: London, UK, 2010. [Google Scholar]
- Prior, L.; Manley, D. Poverty and health: Thirty years of progress? In Poverty and Social Exclusion in the UK: Volume 2—The Dimensions of Disadvantage; Bramley, G., Bailey, N., Eds.; Policy Press: Bristol, UK, 2018; pp. 203–223. [Google Scholar]
- Smith, K.E.; Hill, S.; Bambra, C. Health Inequalities: Critical Perspectives; Oxford University Press: Oxford, UK, 2016. [Google Scholar]
- The National Council for Voluntary Organisations UK Civil Society Almanac 2018: Volunteer Profiles. Available online: https://data.ncvo.org.uk/a/almanac18/volunteer-profiles-2015-16-2/ (accessed on 7 December 2018).
- Giordano, G.N.; Lindstrom, M. The impact of changes in different aspects of social capital and material conditions on self-rated health over time: A longitudinal cohort study. Soc. Sci. Med. 2010, 70, 700–710. [Google Scholar] [CrossRef]
- Yip, W.; Subramanian, S.V.; Mitchell, A.D.; Lee, D.T.S.; Wang, J.; Kawachi, I. Does social capital enhance health and well-being? Evidence from rural China. Soc. Sci. Med. 2007, 64, 35–49. [Google Scholar] [CrossRef]
- Fujiwara, T.; Kawachi, I. A prospective study of individual-level social capital and major depression in the United States. J. Epidemiol. Community Health 2008, 62, 627–633. [Google Scholar] [CrossRef]
- Fone, D.; Dunstan, F.; Lloyd, K.; Williams, G.; Watkins, J.; Palmer, S. Does social cohesion modify the association between area income deprivation and mental health? A multilevel analysis. Int. J. Epidemiol. 2007, 36, 338–345. [Google Scholar] [CrossRef]
- Riumallo-Herl, C.J.; Kawachi, I.; Avendano, M. Social capital, mental health and biomarkers in Chile: Assessing the effects of social capital in a middle-income country. Soc. Sci. Med. 2014, 105, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Kwan, M.-P. From place-based to people-based exposure measures. Soc. Sci. Med. 2009, 69, 1311–1313. [Google Scholar] [CrossRef] [PubMed]
- Montello, D.R. Scale in geography. In International Encyclopedia of the Social and Behavioral Sciences; Smelser, N.J., Baltes, B., Eds.; Elsevier Science Ltd.: Oxford, UK, 2001; pp. 13501–13504. [Google Scholar]
- Perchoux, C.; Chaix, B.; Cummins, S.; Kestens, Y. Conceptualization and measurement of environmental exposure in epidemiology: Accounting for activity space related to daily mobility. Health Place 2013, 21, 86–93. [Google Scholar] [CrossRef] [PubMed]
| System | Biomarker | N | Mean (SD) | High Risk Cut-Off Values |
|---|---|---|---|---|
| Cardiovascular | Systolic Blood Pressure | 2628 | 126.44 (16.64) | ≥136.5 mmhg |
| Diastolic Blood Pressure | 2628 | 73.01 (10.84) | ≥80 mmhg | |
| Pulse Rate | 2628 | 68.79 (10.93) | ≥75.5 bpm | |
| Lipid Metabolism | HDL Cholesterol | 3138 | 1.53 (0.45) | <1.2 mmol/L |
| Total: HDL Cholesterol | 3137 | 3.75 (1.35) | ≥4.42 | |
| Triglycerides | 3144 | 1.79 (1.27) | ≥2.2 mmol/L | |
| BMI | 3112 | 28.02 (5.52) | ≥30.9 kg/m2 | |
| Waist Circumference | 3161 | 93.70 (14.52) | ≥103.1 cm | |
| Glucose Metabolism | HbA1c | 2969 | 37.30 (8.67) | ≥39 mmol/molhb |
| Inflammatory | C-Reactive Protein | 3019 | 3.24 (6.60) | ≥3.2 mg/L |
| Fibrinogen | 3121 | 2.81 (0.62) | ≥3.2 g/L | |
| Albumin | 3139 | 46.62 (2.94) | <45 g/L | |
| HPA-axis | DHEAs | 3137 | 4.74 (3.36) | <2.2 mol/L |
| Factor | Mean (SD) | N | |
|---|---|---|---|
| Allostatic load | 3.07 (2.45) | 3210 | |
| Age | 51.53 (17.58) | 3210 | |
| % | |||
| Sex | Female * | 54.83 | 3210 |
| Male | 45.17 | ||
| Education level | Higher * | 31.29 | 3186 |
| Middle | 46.39 | ||
| Lower | 22.32 | ||
| Employment status | Employed * | 56.07 | 3210 |
| Retired | 29.16 | ||
| Unemployed/Inactive | 14.77 | ||
| Tenure | Owned * | 79.25 | 3206 |
| Privately rented | 8.86 | ||
| Socially rented | 11.79 | ||
| Marital status | Married * | 69.31 | 3210 |
| Single/SDW | 30.69 | ||
| Subjective financial situation | Comfortable/Alright * | 66.06 | 3209 |
| Just getting by | 25.62 | ||
| Finding it difficult | 8.32 |
| Classes | SSABIC | Smallest Class Size | Entropy | LMR-LRT | ||
|---|---|---|---|---|---|---|
| % | Count | |||||
| Townsend deprivation | 2 | 93,780.35 | 0.33 | 1019 | 0.907 | 0.000 |
| 3 | 88,670.63 | 0.14 | 425 | 0.892 | 0.000 | |
| 4 | 86,475.98 | 0.08 | 246 | 0.879 | 0.002 | |
| 5 | 85,530.94 | 0.05 | 147 | 0.844 | 0.276 | |
| 6 | 84,555.45 | 0.05 | 143 | 0.854 | 0.129 | |
| Social capital | 2 | 58,948.61 | 0.18 | 543 | 0.898 | 0.000 |
| 3 | 57,478.44 | 0.07 | 203 | 0.826 | 0.092 | |
| 4 | 56,809.16 | 0.02 | 48 | 0.808 | 0.021 | |
| 5 | 56,435.34 | 0.01 | 45 | 0.773 | 0.099 | |
| 6 | 56,189.13 | 0.01 | 46 | 0.761 | 0.362 | |
| Model 1: No Covariates | Model 2: Age and Sex | Model 3: Sociodemographics | |||||
|---|---|---|---|---|---|---|---|
| N | 3095 | 3095 | 3067 | ||||
| Allostatic load | Mean | S.E. | Mean | S.E. | Mean | S.E. | |
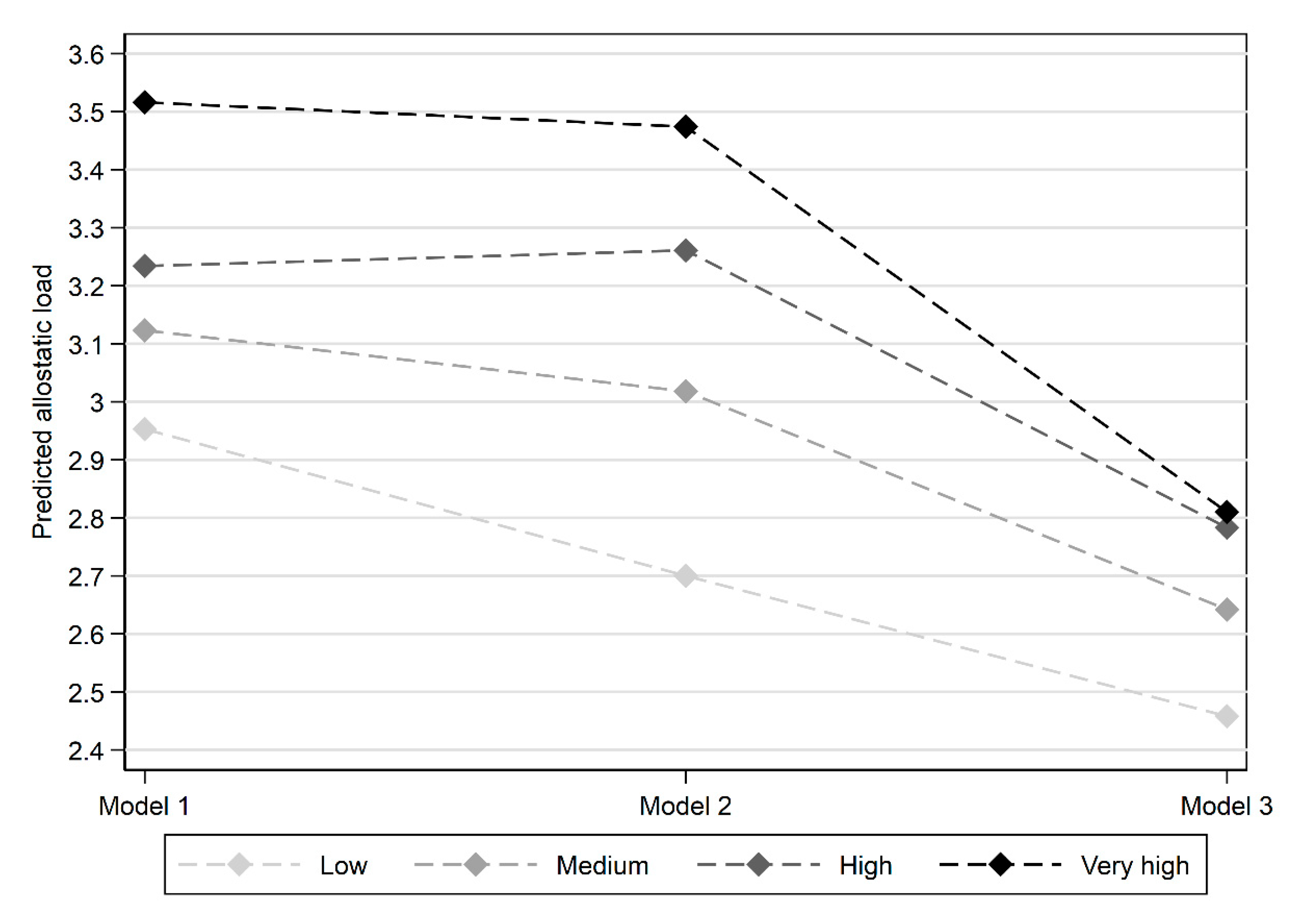
| Deprivation Exposure History | Low | 2.953 | 0.072 | 2.700 | 0.081 | 2.458 | 0.108 |
| Medium | 3.123 | 0.087 | 3.018 | 0.092 | 2.642 | 0.122 | |
| High | 3.234 | 0.109 | 3.261 | 0.108 | 2.783 | 0.140 | |
| Very high | 3.516 | 0.177 | 3.474 | 0.170 | 2.810 | 0.206 | |
| Overall test p-value | 0.015 | 0.000 | 0.050 | ||||
| Beta | S.E. | Beta | S.E. | Beta | S.E. | ||
| Age | 0.053 | 0.002 | 0.052 | 0.004 | |||
| Sex | Female * | ||||||
| Male | 0.292 | 0.079 | 0.302 | 0.080 | |||
| Education Level | Higher * | ||||||
| Middle | 0.238 | 0.096 | |||||
| Lower | 0.463 | 0.123 | |||||
| Employment Status | Employed * | ||||||
| Retired | −0.054 | 0.140 | |||||
| Unemployed/Inactive | −0.005 | 0.126 | |||||
| Subjective Financial Situation | Comfortable/Alright * | ||||||
| Just getting by | 0.268 | 0.098 | |||||
| Finding it difficult | 0.478 | 0.170 | |||||
| Tenure | Owned * | ||||||
| Privately rented | 0.265 | 0.150 | |||||
| Socially rented | 0.699 | 0.160 | |||||
| Marital Status | Married * | ||||||
| Single/SDW | −0.163 | 0.090 | |||||
| Model 1: No Covariates | Model 2: Age and Sex | Model 3: Sociodemographics | |||||
|---|---|---|---|---|---|---|---|
| N | 3096 | 3096 | 3068 | ||||
| Allostatic load | Mean | S.E. | Mean | S.E. | Mean | S.E. | |
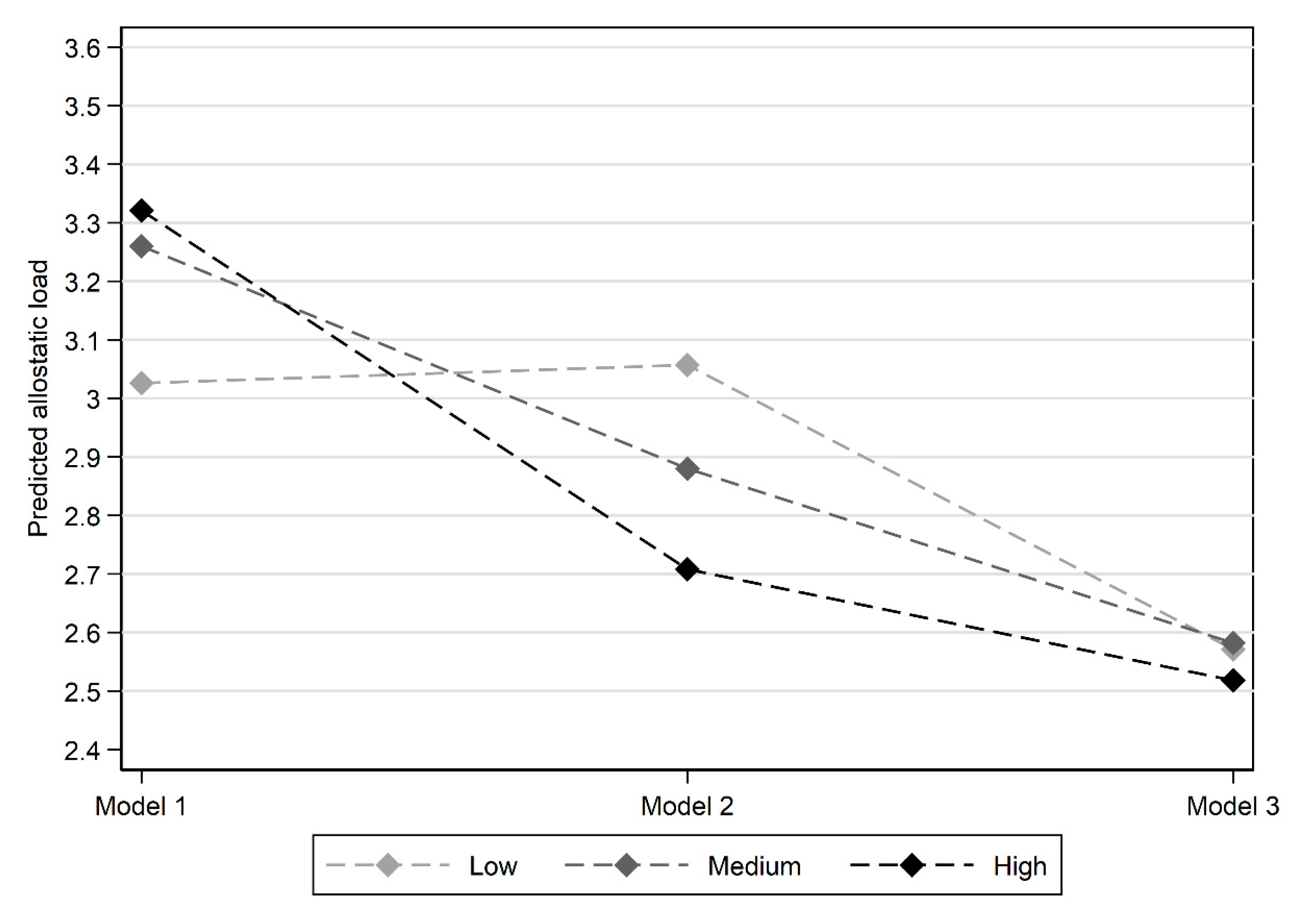
| Social Capital Class | Low | 3.026 | 0.060 | 3.057 | 0.066 | 2.571 | 0.114 |
| Medium | 3.260 | 0.105 | 2.880 | 0.108 | 2.582 | 0.121 | |
| High | 3.321 | 0.177 | 2.708 | 0.180 | 2.518 | 0.189 | |
| Overall test p-value | 0.072 | 0.087 | 0.950 | ||||
| Beta | S.E. | Beta | S.E. | Beta | S.E. | ||
| Age | 0.053 | 0.002 | 0.051 | 0.004 | |||
| Sex | Female * | ||||||
| Male | 0.277 | 0.079 | 0.300 | 0.080 | |||
| Education Level | Higher * | ||||||
| Middle | 0.244 | 0.100 | |||||
| Lower | 0.501 | 0.129 | |||||
| Employment Status | Employed * | ||||||
| Retired | −0.051 | 0.140 | |||||
| Unemployed/Inactive | −0.003 | 0.126 | |||||
| Subjective Financial Situation | Comfortable/Alright * | ||||||
| Just getting by | 0.293 | 0.098 | |||||
| Finding it difficult | 0.518 | 0.170 | |||||
| Tenure | Owned * | ||||||
| Privately rented | 0.280 | 0.149 | |||||
| Socially rented | 0.803 | 0.156 | |||||
| Marital Status | Married * | ||||||
| Single/SDW | −0.143 | 0.090 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prior, L. Allostatic Load and Exposure Histories of Disadvantage. Int. J. Environ. Res. Public Health 2021, 18, 7222. https://doi.org/10.3390/ijerph18147222
Prior L. Allostatic Load and Exposure Histories of Disadvantage. International Journal of Environmental Research and Public Health. 2021; 18(14):7222. https://doi.org/10.3390/ijerph18147222
Chicago/Turabian StylePrior, Lucy. 2021. "Allostatic Load and Exposure Histories of Disadvantage" International Journal of Environmental Research and Public Health 18, no. 14: 7222. https://doi.org/10.3390/ijerph18147222
APA StylePrior, L. (2021). Allostatic Load and Exposure Histories of Disadvantage. International Journal of Environmental Research and Public Health, 18(14), 7222. https://doi.org/10.3390/ijerph18147222




