Abstract
Climate change generates negative impacts on human health. However, little is known about specific impacts on eye diseases, especially in arid and semi-arid areas where increases in air temperatures are expected. Therefore, the main goals of this research are: (i) to highlight the association between common eye diseases and environmental factors; and (ii) to analyze, through the available literature, the health expenditure involved in combating these diseases and the savings from mitigating the environmental factors that aggravate them. Mixed methods were used to assess the cross-variables (environmental factors, eye diseases, health costs). Considering Southern Spain as an example, our results showed that areas with similar climatic conditions could increase eye diseases due to a sustained increase in temperatures and torrential rains, among other factors. We highlight that an increase in eye diseases in Southern Spain is conditioned by the effects of climate change by up to 36.5%; the economic burden of the main eye diseases, extrapolated to the rest of the country, would represent an annual burden of 0.7% of Spain’s Gross Domestic Product. In conclusion, the increase in eye diseases has a strong economic and social impact that could be reduced with proper management of the effects of climate change. We propose a new concept: disease sink, defined as any climate change mitigation action which reduces the incidence or morbidity of disease.
1. Introduction
The Intergovernmental Panel on Climate Change 2014 Report on Human Health established three basic prospects through which climate change will affect human health [1]: (i) direct impacts, related to changes in the frequency of extreme events, including heatwaves, droughts and heavy rain; (ii) effects on natural systems, such as changes in disease vectors, waterborne diseases and air pollution; and, (iii) strongly conditioned effects by mismanaged human systems, such as labor impacts, malnutrition and mental stress. The third objective of the 2030 UN’s Agenda for Sustainable Development (https://www.un.org/sustainabledevelopment/es/health/ accessed on 13 March 2021) seeks to ensure a healthy life and promote the well-being of all ages, recognizing that further efforts are needed to address and eradicate many health problems.
Concerning direct impacts, Global Warming is a progressive trend with variable intensity all over the world. Thus, heatwaves are strongly associated with mortality increases. Christidis et al. (2012) [2] pointed out that extreme heat events increased by fourfold in Europe between 1999 and 2008, causing in some countries about 15,000 deaths [3]. Several scholars have stated that Global Warming is also responsible for an increase in cardiovascular, respiratory and kidney diseases, besides an increase in forest fires which are associated with an increase in smoke-related morbidity and mortality [4,5]. In terms of eye health, there is evidence of the effect of heat on the inflammatory response of the corneal cells [6]. Furthermore, this has been related to the predisposition to infectious viral processes such as herpes and viral conjunctivitis, besides bacterial and fungal processes, in addition to allergies [7,8]. Moreover, van der Leun et al. (2008) [9] noted that an effective dose of ultraviolet radiation (UVR) could increase by 2% for every degree Celsius as the temperature rises. This phenomenon could also suppress cell immunity and increase sensibility to infections, facilitating the activation of latent viruses [10,11]. Additionally, increasing UVR, in conjunction with summer thermal maximum, is related to the increase of ocular tumors and cataracts [12,13,14,15], along with an increased risk of retinal detachment [16].
Rising temperatures recorded in arid and semi-arid areas such as Southern Spain are common throughout the Mediterranean region [17]. Moreover, some estimations claim that mean air temperatures and solar radiation values may continue rising [18,19], besides facing an increasing frequency of heavy rain and floods [20,21], elements which have well-documented effects on infectious diseases [22].
The alteration of natural systems favors an increase in the incidence of vector-borne diseases and even their appearance in areas where they did not exist before. Some diseases eradicated in Spain such as malaria or trachoma could reappear [23,24]. Some other diseases could appear or increase their currently scarce numbers: dengue and hemorrhagic fever with renal syndrome (increasing with temperatures, rainfall and humidity), tick-borne diseases (increasing with temperatures), and other pests such as Chikungunya, Japanese encephalitis, Rift Valley fever [25,26,27,28]), or West Nile Fever [29,30]. These all have important and well-known effects on eye structures [31,32,33,34,35].
Air quality is also a conditioning factor for human health, especially concerning other pollutants than carbon dioxide. Among these, tropospheric ozone must be highlighted as a common urban pollutant [36] that may be magnified by heatwaves [37,38]. Moreover, acute pollution events, which are related to high atmospheric stability, heatwaves and droughts, have been associated with high levels of aerodynamic particles (PM10, PM2.5) involving severe outbreaks of premature mortality [4], which also are being studied in relation to the COVID-19 outbreak [39]. Additionally, allergenic particles have increased due to warmer atmospheric conditions, promoting dermatitis, asthma attacks, allergic rhinitis and allergic conjunctivitis [40]. Finally, air quality has been related to several complications of the ocular surface, implying damages to the cornea and the conjunctiva, and several disorders such as cataracts, conjunctivitis, glaucoma, and dry eye, among others [41,42,43].
Furthermore, as an effect of climate change, mismanaged human systems lead to malnutrition, work-related health problems and mental disorders. Thus, malnutrition or undernourishment is a direct effect of changes in harvest because of temperature changes or rainfall regimes, especially in those areas where agriculture and livestock have naturally low productivity [44,45] and are sensitive to price fluctuations. This leads to the consumption of food without crucial active principles of nourishment [46]. Malnutrition is implied in xerophthalmia, a disease caused by a deficiency of Vitamin A, which provokes blindness in children under the age of five, with high mortality [47,48]. Malnutrition is also responsible for ocular problems in adulthood [49], causing diseases such as AMD (Age-related Macular Degeneration), cataracts, or glaucoma [50]. In addition, outdoor workers (farmers, building and clean-up workers) directly suffer heatwaves, reducing their productivity in order to avoid them [51,52]. Moreover, they also suffer vector-borne outbreaks, psycho-physical damage and secondary ocular diseases such as pterygium, glaucoma and retino-choroidal injuries. Farmers have eight times higher possibilities of suffering ocular damage than other professionals.
However, information on the long-term impact of climate change on eye diseases regarding their economic effects is lacking. Therefore, the main aims of this investigation are (i) to demonstrate the association between common eye diseases and environmental factors, taking as a reference two highly populated cities in Southern Spain (Málaga and Almería), which are considered highly vulnerable areas to climate change; and (ii) to estimate, using the available literature, the health expenditure involved in combating these diseases and the savings that would be made by mitigating the environmental factors that aggravate them. To achieve these goals, mixed methods to assess the cross-variables (environmental factors, eye diseases and health costs) were used. We hypothesize that current research could focus on the work of those interested in the role of climate change regarding ocular health and may be useful to policymakers in order to explore associated economic damages of a policy of non-intervention concerning climate change.
2. Methods
Mixed methods were used to highlight the cross-variables for southern Spain: environmental factors, eye diseases and health costs.
Thus, climate data from two cities in Southern Spain (Málaga and Almería) have been taken and analysed for comparison and extension with previous research. Temperature and rainfall datasets to assess the climate trends of Málaga and Almería urban areas were used (574,674 and 198,533 inhabitants, respectively) [53]. Data from, at least, the last 30 years have been used to analyze precipitation, average, minimum and maximum temperatures and humidity. The climate stations selected were Málaga and Almería Airports due to the available long-term data series, as well as the location near to the sea. Both are representative examples of semi-arid (Málaga) and arid (Almería) conditions in southern Spain. The dataset has been extracted and analysed from the REDIAM (Environmental Information Network of the Andalusian Regional Government (http://www.juntadeandalucia.es/medioambiente/site/rediam accessed on 9 January 2021) database. Finally, the results were compared to and contrasted with the IPCC reports to verify whether or not they are consistent with the internationally established outlook for the region [1,54,55,56].
Then data on the most common eye diseases in the region were reviewed and their attribution to environmental variables associated with climate change analyzed.
The most frequent ocular pathologies associated with environmental variables that are undergoing special modifications in arid and semi-arid areas such as the south of Spain [57,58,59] are summarized as follows.
2.1. Cornea, Sclera and Conjunctive
Several pathologies of these ocular structures are related to a wide range of environmental variables; they are divisible into three groups:
- (A).
- Increasing inflammation: Allergic Keratoconjunctivitis, Marginal Keratitis, Dry Keratitis, Chronic Episcleritis, Corneal Metaplasia, and Pterygium;
- (B).
- Increasing infections and superinfections: Corneal and Conjunctival Herpes simplex and Herpes Zoster, Viral Keratoconjunctivitis, Accidental Corneal Fungal Injuries, Infectious Corneal Injuries, non-fungal injury by Contact Lens;
- (C).
- Tumor processes in Cornea, Conjunctive and Ocular Annexes: Epidermoid Neoplasia of Ocular Surface, Basal Cell Carcinoma of Eyelid.
2.2. Glaucoma
This disease has been related to environmental variables, highlighting temperatures, UVR and pollutants. It is divisible as follows:
- (D).
- Acute Glaucoma;
- (E).
- Chronic open-angle Glaucoma.
2.3. Cataracts
This visual impairment is also related to environmental variables, especially solar radiation. It is divisible into two groups:
- (F).
- Early Cortical and Subcapsular Cataracts;
- (G).
- Pseudo-exfoliation syndrome.
2.4. Tumor Processes in the Choroid, Iris and Ciliary Body
- (H).
- Uveal Melanoma is the main disease of this group related to environmental factors. Several authors have related it to exposure to UVB radiation [12,60,61].
2.5. Uveitis (Intraocular Inflammatory Processes)
Four different uveitic processes associated with various pathologies have been related to a wide range of environmental factors, as can be seen in Table S1. These groups are:
- (I).
- Infectious uveitis: Toxoplasmosis, Tuberculosis, Campylobacter, Chlamydia, Ocular Herpes simplex (VHS) and Zoster (VHZ), West Nile Fever, Borreliosis and Rickettsiosis, Shigellosis, Salmonellosis; the World Health Organization offers information about several pathogens and their infectivity [62,63], including those which are engaged in ocular diseases; a higher frequency of infectious uveitis has been highlighted during the summer [64], plus a change in patterns, incidence and prevalence [65].
- (J).
- Non-infectious uveitis associated with systemic diseases: Rheumatoid arthritis (RA), Ankylosing spondylitis (AS), Sarcoidosis, Multiple Sclerosis (MS), Inflammatory bowel diseases (IBD—Crohn and ulcer colitis), Behçet’s disease, Giant cells arteritis (Horton’s disease), Necrotizing systemic vasculitis.
- (K).
- Connective tissue diseases: Systemic Lupus Erythematosus (SLE), Dermatomyositis.
- (L).
- Uveitis merely as ocular disease, without associated systemic pathologies: Fuchs’ heterochronic Uveitis, Posner-Schlossmann syndrome, Intermedia uveitis and pars planitis, Birdshot choroidopathy, Vogt-Koyanagi-Harada syndrome (VKH), White Dots Syndromes [66].
2.6. Retina
Finally, several retinal injuries have been related to environmental factors, with particular emphasis on sunlight and UVR, among others. These have been classified into four groups:
- (M).
- Tractional Retinal Detachment and Retinal Tears.
- (N).
- Posterior Vitreous Detachment.
- (O).
- Age Macular Degeneration (AMD).
- (P).
- Central Serous Choroidopathy.
Finally, information on the costs of these diseases was examined in the available literature, resulting in the attribution of the health costs of eye diseases to climate change. An economic assessment of the estimated costs of the most frequent ocular pathologies has been developed. Sun exposure excess, high temperatures, pouring rain, wind or pollution effects, among others, are approachable variables with regard to land management solutions. By looking at the most common ocular diseases and their high impact on health, it is possible to see the magnitude of the problem.
Thus, where possible, the average cost of treatments for each eye disease in Spain has been analysed. When this was not possible, references were taken from Europe or, failing that, from the United States.
The environmental factors that predispose or exacerbate the different ocular pathologies have been consulted, searching the available literature especially for the percentage of attribution of environmental factors related to climate change to these pathologies. From this, we deduced the increased costs of failing to control environmental factors, producing a final sum of the economic cost of climate change-related eye pathologies.
Likewise, climate change mitigation measures and the economic weight that carrying them out would imply have been analysed in order to assess the final economic balance, comparing the benefits of adaptation with those of mitigation; the former would imply seeking additional resources to better address diseases, reducing vulnerability and increasing resilience capacity; while the latter would imply a reduction in the danger and risk associated with environmental variables [67,68,69].
3. Results
3.1. Background of Climate Conditions in Almería and Málaga
As in other parts of the Mediterranean Region [70], the observed trends in these two cities can be synthesized as follows (for more information, see Supplementary Material Tables S1 and S2): a sustained increase in air temperatures of about 0.1–0.4 °C/decade (Figure 1a), which has changed the Köppen-Geiger classification for Almería Airport from BSk to BSh (warmer sub-desertic climate), and the average annual temperature for Málaga Airport from 17.96 °C (1961–1990) to 18.76 °C (1991–2020); the higher frequency of heatwaves (maximum daily temperatures > 36.6 °C) (Figure 1b); increase in tropical days (minimum daily temperatures > 20 °C), 3 to 6/decade (Figure 1c), and torrid days (minimum daily temperatures > 25 °C) (Figure 1b). Moreover, droughts point to a higher frequency because of lower relative humidity (Figure 1d) and lower annual volume of rainfall (i.e., Almería Airport, 1961–1990: 216 mm.; Málaga Airport: 583 mm.; Almería Airport, 1991–2020, 201 mm; Málaga Airport: 487 mm.); both values classify Málaga as a semiarid climate and Almería as an arid one. Floods and erosive processes from pouring rain also tend to be more frequent and hydric resources are decreasing [21,71,72]. Coastal phenomena and coastal losses are increasing [73,74]. Habitats and endangered species are increasingly suffering, with an increase of waterborne diseases predicted in the near future [75]. Finally, measuring anthropogenic gases allows their exhaustive control [76], which is vital to analyze the evolution of triggering factors for global warming. No significant trend has been identified regarding UVR, despite a slight trend towards decrease. These results agree with other estimations conducted for Andalusia by the Ministry of Ecological Transition (MITECO) [77], as well as with general estimates for the Mediterranean region by other official reports [78].
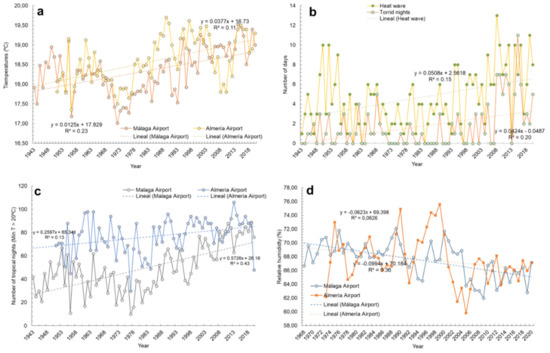
Figure 1.
Climate evolution and trends of Almería and Málaga airport meteorological stations. Mean annual air temperatures (a), number of heatwaves and torrid days (b), number of tropical days (c) and average relative humidity (d). Own elaboration from REDIAM data.
3.2. Ocular Pathologies Related to Climate Variables
All the pathologies described in chapter 2 are described in detail in Table S1, where each pathology is related to the climatic and environmental variables that affect them. Furthermore, in order to address this problem concisely, environment variables and potentially affected eye structures have been crossed in Table 1. Both the environmental values of Southern Spain and the most frequent ocular pathologies in that region have been considered.

Table 1.
Relationship between environmental variables and potential eye damage.
3.3. Estimation of the Health Costs of the Most Frequent Ocular Pathologies Related to Climate Change in Southern Spain
Concerning Tractional Retinal Detachment (TRD), this is one of the most serious and costly pathologies, varying, depending on surgery type, from 1647 euros/patient for laser prophylaxis to 6690 euros/patient for vitrectomy [79]. The annual incidence of TRD in Spain is 1/10,000 [80], meaning 4700 cases/year.
Each heatwave increases the TRD risk by 2.47 times for people under age 75 years. In Spain, in 2020, the number of days per year in which heatwave thresholds are exceeded is twice as high as during the mid-1980s [81]. AEMET (Spanish State Meteorological Agency; http://www.aemet.es accessed on 9 January 2021) climate projections for the 21st century show an increase of about 20% in the number of warm days by the middle of the century; the frequency of extremely hot nights has increased tenfold from 1984 in the more populated Spanish cities [81]. Thus, for example, the threshold to consider a heatwave in Malaga city (Southern Spain) is 36.6 °C [82], which has been exceeded on 11 days during the summer of 2019 and seven during the summer of 2020. This implies an increase in TRD risk for people under age 75 years calculated at 17.3% only during that one summer. The trend identified from REDIAM data is a doubling of heatwave days from 1992 to 2020 (from 4 to 8 days) (Figure 1B).
Concerning Age-Related Macular Degeneration (AMD), people exposed to summer sun >5 h/day during adolescence and 30 years of age, at the initial examination, were at greater risk of developing increased macular pigment and early AMD (OR 2.14; CI = 95%, 0.99–4.61; p = 0.05) 10 years earlier than people sun-exposed <2 h/day during the same periods. Nevertheless, in patients reporting high summer sun exposure during their teenage years and 30 years old, the use of hats and sunglasses for half of those periods were associated with a lower risk of soft drusen (Relative Risk (RR) = 0.55; CI = 95%, 0.33–0.9; p = 0.02) and epithelial depigmentation of retinal pigment (RR = 0.51; CI = 95%, 0.29–0.91; p = 0.02) [83].
The progressive recovery of the ozone layer from international action on chlorine-fluorocarbon gases has achieved the maintenance or even a slight reduction in the average UVR (UV index) [84,85]. However, the trend of decreasing relative humidity due to increasing average temperatures aggressively increases UVR due to the lower radiative forcing (higher penetration) of this wavelength because of less filtering from water aerosols; moreover, air pollution also enhances the UVR penetration [86,87].
In 2006, it was estimated that by 2015, 400,000 Spanish inhabitants would suffer AMD, and more than one million could be at risk [50]. This reached 700,000 people in 2016 (1.5% of the Spanish population), almost twice as many as expected. Other factors which increase the risk of AMD are malnutrition and undernourishment [88]. Adverse and extreme climate events all over Spain are linked to a subsequent increase in agricultural, fruit and vegetable prices, which are essential to prevent the development of pathologies such as AMD and Glaucoma [89,90]. The annual burden of AMD is 8300 euros/patient, and half of this (48%) must be borne by the patients and their families.
Regarding glaucoma, direct costs exceed 612 euros/patient/year in Spain, whilst costs due to productivity losses reach 1946 euros, making a total cost of 2558 euros/patient/year [58,91]. Between 2011 and 2016 glaucoma cases in Spain increased by a factor of 3.77 (from 206,806 to 779,221 euros) [92]. From 1990 to 2015, the number of disability-adjusted life years (DALY) and the age-standardized DALY rate for glaucoma increased by 122% and 15%, respectively; in addition, glaucoma was associated positively with national levels of UVR and PM2.5. Besides, lower socio-economic level, advanced age, female gender, higher UVR (increasing the incidence by 11% depending on the number of hours outdoors) and higher air pollution level was significantly associated with a higher frequency of glaucoma [93]. Thus, as in AMD, glaucoma is conditioned at its onset by oxidative stress, secondary to UVR (UVA-UVB) [94,95], and its worsening and progression are also enhanced by malnutrition and undernourishment [50].
Besides this, cataracts surgery is one of the most frequent surgical procedures in the world; it is estimated that >22 million people/year underwent surgery [96]. The association UVR–cataracts has long been studied [12,14,61,97], and an odds ratio has been estimated with a 1.1 to 2.5 times higher risk of cortical and sub-capsular cataracts with higher exposure to UVB radiation; however, the use of sun-glasses has an odds ratio of 0.62 for posterior sub-capsular cataracts, thus protecting against the development of this type of cataracts.
The average cost of a cataract operation in Spain ranges between 910 and 1541 euros [58]. Because the number of cataract operations increased by 50% between 2004 and 2013 in Spain (almost 307,000 procedures) [98], a period in which the Spanish population grew by only 10% and elderly people by only 2% compared to 2004 [99], and because local data show a greater increase recently (Figure 2), it is necessary to address predisposing factors, since health care spending tends to increase year after year.
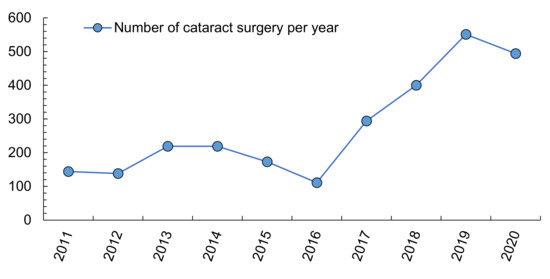
Figure 2.
Cataract surgeries in a local hospital of Southern Spain (Axarquía Hospital). Own elaboration from Servicio Andaluz de Salud, 2021 [100].
In the case of infectious diseases such as Herpes Zoster (VHZ), there is a 14% increased risk of outbreaks, especially in males, with overexposure to UVR, due to the immunosuppression it produces [8,101]. The average cost of treatment varies between 301.5 and 916.7 euros/outbreak, depending on its complication [102], with an estimated incidence of 60,000 cases/year [103].
Concerning Ocular Herpes Simplex (VHS), the risk of appearance and relapse increases, not so much because of the number of hours of outdoor activities, which could be a protection factor with a UV index < 4, but when these activities are carried out with a UV index > 4. Thus, a UV index > 4 increases the risk of a VHS outbreak by 33% [7]. In Southern Spain, the UV index > 4 is usually present from April to October [86]. The VHS incidence in Spain from 2000 to 2008 grew by 40% [104,105]. A global cost between 2033 and 2366 euros/outbreak/patient has been estimated, including costs for the medical procedure, sick leave, treatment and use of facilities. Between 2011 and 2017 116,000 cases/year were recorded, with a trend towards increase, only interrupted in 2017 [106].
Regarding auto-immune systemic diseases with ocular impact (non-infectious uveitis), it is necessary to highlight that environmental pollutants are one of the main triggering factors for its development, emphasizing especially:
- (a)
- In Rheumatoid Arthritis (RA), genetic factors explain <50% of the risk of developing this disease; however, a 31% increase in risk has been identified in people living within <50 m from the main road (with traffic and, therefore, intense pollution) [107]. In Spain alone, RA accounted for a €1.12 billion/year burden of necessary health care, indirect costs and associated sick leave [108]. Mitigating the pollutant factor could save the Spanish State 350 million (M) euros, as well as undoubtedly reducing the suffering caused by this type of disease.
- (b)
- In Ankylosing Spondylitis (AS), a >60% correlation with pollutant particles (PM2.5) has been found: a prolonged exposure leads to worsening in the control of the inflammatory outbreak of this disease [109]. An average cost to address AS in Spain of 11,462 euros/patient/year has been estimated, including direct costs and productivity losses [110]. In 2017, 1.9% of Spanish people suffered from this disease (900,000 people), implying an annual average cost of €10.3 billion of the Spanish Gross Domestic Product (GDP). Such a cost could be reduced by €6 billion just by controlling environmental factors.
- (c)
- Pollutants determine an association with inflammatory outbreaks of Multiple Sclerosis (MS). Thus, PM10 particles were 8% associated with an increase of relapse during the cold season, whilst ozone was 16% associated during the hot season (therefore, ozone is pathogenically triggered by high temperatures) [111,112]. Around 50,000 people in Spain suffer from this disease, whose average cost is 30,000 euros/patient/year; this implies a global burden of €1.4 billion/year [113], added to the severe disability which it involves. 112 to 224 million euros/year could be saved by controlling pollutants alone, without taking into account the benefit to be gained by controlling high temperatures and, thus, avoiding the exacerbation suffered by MS patients (Uhthoff phenomenon) [112,114].
- (d)
- Inflammatory Bowel Diseases (IBD) include Crohn’s disease (CD), Ulcerative Colitis (UC) and Non-classifiable IBD. Their estimated prevalence is 0.3% of the European population, showing an increasing incidence of 176,000 cases per year. It has been calculated that 2.5–3 million people in Europe suffer from IBD. This implies a very high total annual cost of health care. Based on an average of diagnosed patients in 2010 throughout 28 European medical centers during the first year after diagnosis, a total cost of 5942 euros/patient for CD, 2753 euros/patient for UC, and 2898 euros/patient for non-classifiable IBD was calculated. Furthermore, each day of heatwave increases by 4.6% the outbreak risk for IBD [115,116], and thus the derived visual impairment. For example, Málaga city (Southern Spain) suffered 11 heatwave days only in 2019; therefore, the outbreak risk of IBD was increased by 50.6%, adding to the high health care burden and personal suffering which this implies.
- (e)
- The prevalence of Systemic Lupus Erythematosus (SLE) in Spain is 9/10,000 inhabitants [117]. The patient/year cost ranges between 3604 and 5968 euros, according to the severity [118]. Relating to prevalence and cost per patient, the control of this disease involves a €201 million burden in Spain. Concerning environmental parameters, SLE outbreaks are twice as likely to occur in sun-exposed workers fin a year, and 7.9 times more likely to develop if they suffer sunburn from intense exposure to the sun [119].
- (f)
- Sarcoidosis is a disease with high prevalence in Spain (1/1000 inhabitants) [120]. Although there is no information about the cost of its treatment, data from the USA points out that insurance companies spend 19,714 dollars/patient/year among direct costs of medical attention and secondary costs because of sickness absences [121]. Extrapolating this data to Spain, the disease costs would be equivalent to almost €784 million/year. The relation between Sarcoidosis and environmental factors is essential, given that its pathogenesis is characterized by exposure to dust and both natural and urban pollution (PM10 and PM2.5), together with dryness and high temperatures [122]. Southern Spain records more than 20 events per year with Saharan dust advection from May to September [123]. There are no numerical studies on the evolution of this disease in Spain, but researches from the Midwest of the USA (where dust storms are frequent) reveal that the prevalence doubled between 1995 and 2010, without a relationship to an increase in the population [124]. Extrapolating these data to Spain, a higher frequency of dust storms related to higher dryness and advection from Africa could involve doubling of the health care costs related to Sarcoidosis.
The estimated costs of dealing with autoimmune diseases in the USA, where it has been studied more than in Europe, are over $100 billion, an underestimate since the eight most common autoimmune diseases alone account for between $51.8 and $70.6 billion/year [125]. Moreover, the chronic pain state and the disability to make changes in the mood of patients, sleeping disorders, and tiredness impact their social and working life [108].
The climate change-related eye diseases selected above represent an annual cost of between 22.63 and 23.31 billion euros, that is, between 1.9% and 1.95% of national Spanish GDP (Table 2). 36.5% of this figure can be attributed to climate change variables (0.7% GDP; between €8.26 and €8.51 billion). Mitigation measures centered on afforestation and greenhouse gases (GG) control involve a cost of €13.15 billion (1.1% GDP), but the expected benefit due to energy saving reaches between €16.5 and €25.7 billion (1.38–2.15% GDP). Thus, even with the least optimistic forecast, 0.28% GDP would be saved, adding the progressive reduction by 0.7% of GDP due to health care cost in ocular diseases partially attributable to climate change, and the intangible benefits of wellbeing and life-quality that reducing life-limiting illness implies (Supplementary Material Table S2). In any case, the cost of controlling GG would have benefits for any disease exacerbated by environmental variables related to climate change, and not just eye disease.

Table 2.
National Annual Costs of diseases involving eye structures, their attribution to climate change, expected evolution and mitigation costs.
- (1)
- Because of the sustained increase of CO2 concentration in Spain [126]
- (2)
- The trend in Andalucía has been practically stable since 2008, whilst the average of Spain reveals a clear decrease, showing values below the average of Andalucía [127]
- (3)
- In Andalucía during the last decade, the WHO criteria have been permanently broken (>100 µg/m3); there is no aggregated evolutionary data [128]
- (4)
- Considering just the costs of afforestation-reforestation and GG control, and ignoring the benefits of energy-saving
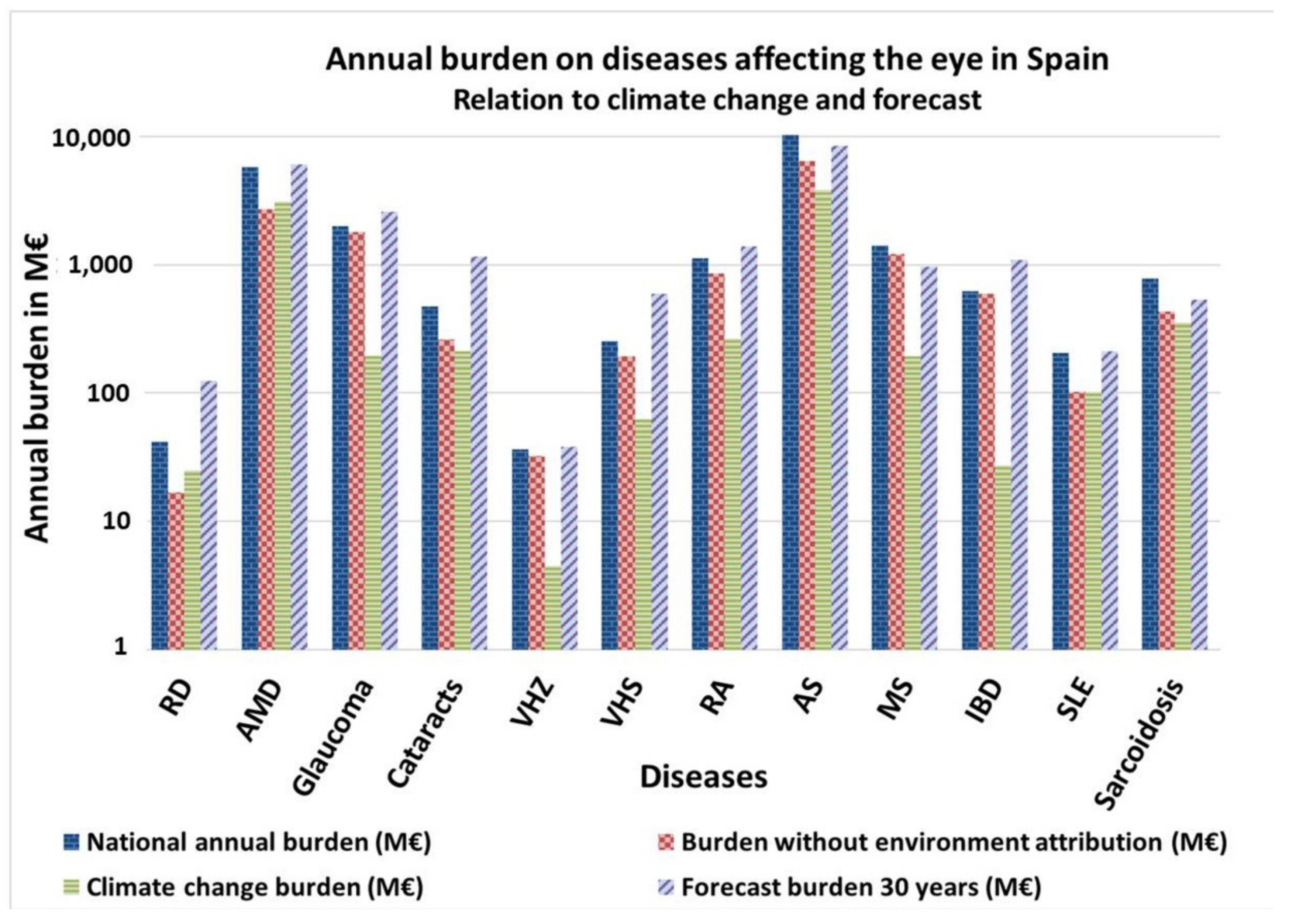
RD, AMD and SLE are the ocular impairments most affected by climate change, the cost of addressing them doubling due to environmental factors (Figure 3). Concerning the 30 years forecast, if the actual trend continues without intervention, diseases such as RD, AMD, Glaucoma, Cataracts, VHZ, VHS, RA, IBD and SLE will increase their incidence, and, subsequently, their associated burden.
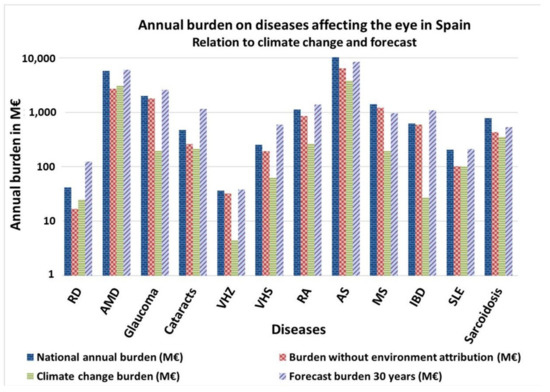
Figure 3.
Annual burden of diseases affecting ocular structures in Spain. Relation to climate change and 30 years forecast. Own elaboration. RD = Retinal detachment. AMD = Age-Associated Macular Degeneration. HZ = Herpes Zoster. HS = Herpes Simplex. RA= Rheumatoid Arthritis. AS = Ankylosing Spondylitis. MS = Multiple Sclerosis. IBD = Inflammatory Bowel diseases; SLE = Systemic Lupus Erythematosus.
4. Discussion. Challenges and Topics to Be Further Discussed
There is a wide and varied bibliography about the climate–ocular health relationship. Global warming behaves like a chain reaction that ominously affects eye health [129]. However, it is uncommon to find interdisciplinary studies where ophthalmologists and environment specialists work together. In the context of climate change, we start from the premise that if adequate management can mitigate the effects of enhanced climatic variables [130], it can consequently mitigate health problems. This study highlights the possibility of preventing an increase in eye diseases and their associated health costs through sustainability, taking into account that this aim can only be reached through environmental, social and economic balance [131], besides cultural factors.
The WHO [132] reports that >28% of the world’s population is visually impaired and almost half of this could be prevented. The same report highlights that “…while the median out-of-pocket spending on health represents <20% of total health spending in high-income countries, it accounts for >40% in low-income countries”. Additionally, rural areas of many countries have low coverage in ocular health care (childhood myopia, cataract) whilst urban lifestyle leads to an increase in myopia and diabetes complications (i.e., diabetic retinopathy).
Reducing global warming needs a reduction in GG emission, and other actions related to afforestation, reforestation and changes in crop management, given that the latter measures favor carbon sinks; furthermore, in urban environments, modulating temperature means controlling emissions of ozone, PM10 and PM2.5, as well as other pollutants; there is also a need to change the albedo of buildings and increase green spaces. The increase in radiation (especially UV) can be stopped if the increase in temperature is effectively controlled.
The benefit of the intervention in climate change variables could be appreciated via the reduction of related ocular pathologies. This would represent an important decrease in human morbidity and a significant reduction of the health care burden needed to address it. A carbon sink has been defined as any process or mechanism that removes a greenhouse gas, an aerosol or a precursor of greenhouse gas from the atmosphere. A given reservoir can be an atmospheric carbon sink if, during a given time interval, more carbon enters than leaves [133]. Similarly, any process or mechanism that eliminates or reduces a harmful effect or environmental health risk can be defined as a disease sink. In this way, this new concept of disease sink can also be understood as any action in favor of climate change mitigation. As was mentioned, the effects of climate change have direct impacts on the incidence or morbidity of diseases. Consequently, mitigation measures must involve a health gain, as a disease sink.
The real financial cost has two components: direct (health-related) and indirect (production losses, informal care and losses in wellbeing). It has been estimated that the total cost of health care for visual impairment in 2010 reached $1100 billion globally, excluding uncorrected refractive errors; the global financial cost reached $2954 billion. These costs include inpatients care, treatment, general eye services, and community and elderly people care, among others. In addition, associated costs of traumatism due to the visual impairment have been included [134].
Mitigation strategies look to reduce the net emissions to the atmosphere of long-term GG, which is the “meal” of anthropogenic climate change [54,135,136] and also look for an increase in the natural systems to eliminate these GGs. This reduction can be reached by:
- (1)
- Increasing the capacity of the carbon sink by means of afforestation and reforestation. From 1990 to 2020, the world lost 420 million (M) hectares (ha) of forest area; 80 M ha of these corresponded to primary woods [137]. Andalucía is one of the three Spanish communities which are developing an emission compensation system using forest projects [138]. The economic cost for one of these projects is 229,351.5 euros for 63.42 ha, and it is expected to capture 16,653 tCO2 in 30 years, that is, to assimilate 262.6 tCO2 per forestry hectare. According to the actual GG emission in Andalucía, where there was an annual production of 51 M tons CO2eq in 2015, it would be necessary to invest €702.27 M to offset through forestry works the GG emissions, and afforest or reforest 194,211.7 ha. Obviously, current efforts are insufficient.
- (2)
- Avoiding ocean acidification. In Andalucía, to avoid the acidification of the sea, and to control the waste of plastics, emissions of nitrogen fertilizers and untreated wastewater, it is essential to control overfishing and highly destructive fisheries to prevent the destruction of carbon sinks [55]. Furthermore, to stabilize emissions at around 450 ppm of CO2eq (recommended by IPCC), it would be necessary to reduce the annualized consumption growth rate by 0.06% per year during the 21st century [56]. While Global GDP (2019) is $87,698 billion [139], the efforts to achieve stabilization would be equivalent to $5.26 billion/year. It has been calculated that Spain would have to reduce its GDP by 1% by 2050 to comply with the limits set by the Kyoto Protocol, i.e., some 12.45 billion euros over 30 years [140].
- (3)
- Promoting the transition of the actual energy generation (electricity) to systems of low carbon emissions. In Spain, the Ministry for Ecological Transition has proposed that 70% of the electric system in 2030 came from renewable energies. Investments in this sector, energy savings and renewable energies (which are cheaper) will allow the GDP to grow by 1.8% by 2030 compared to a scenario without actions: between €16.5 and €25.7 billion [141]. This figure compensates for the reduction in GDP indicated in the previous item.
- (4)
- Increasing building insulation. Global warming makes predictable an increase in energy consumption for cooling buildings and homes. Current energy-efficient systems allow a building with solar protection, efficient ventilation and an insulated façade to save 38% in heating and 52% in cooling [142]. It is expected that the world energy consumption increase will average 57% between 2004 and 2030 [143]. Therefore, energy efficiency and near-zero energy homes will be essential because they have no additional costs [144].
- (5)
- Ensuring that new buildings use more natural air and sunlight. Accordingly, the efficient design of buildings through ventilation with heat recovery, building skin insulation without thermal bridges and dynamic solar control using a blind system (passive house model), would obtain a fairly constant temperature throughout the year, without exceeding 25 °C in summer, i.e., higher energy efficiency [142].
- (6)
- Holding back growth of energy demand: promoting private, public and collective transport, which would reduce around 25 Mt CO2 [145]; an energy culture based on saving and using more efficient and renewable energies; environmental tax to stimulate the change to non-wasteful production models [146].
- (7)
- Stop playing with Nature [147]. As mentioned above, environmental intervention due to inadequate land management, often because of lack of knowledge, produces irreversible ecological cascades which, increasingly common, affect human beings tragically [148]. One probable origin of Covid19 disease is the uncontrolled alteration of ecosystems (contact with wild animals such as the bat Rhinolophus affinis, or the Malaysian pangolin, Manis javanica) [149] and its triggering due to pollution [150]. Other unfortunate examples of human interventions were the myxomatosis virus [151] and HIV, possibly from apes [152].
All these measures could be included in the new concept, disease sink, because they contribute to decreasing the effects of climate change, and therefore reduce the incidence and morbidity of diseases.
The estimates made throughout this research are limited to diseases affecting the eyes; they are just one example, taken from Southern Spain, of the impact of climate change on human health and the associated expenditure. Ocular diseases depend on environmental factors related to climate change by up to 36.5%, which implies an annual burden of 0.7% of the Spanish Gross Domestic Product.
Returning to the goals of this research, the literature consulted amply demonstrates the links between environmental factors related to climate change and eye diseases. Applying this fact to an arid and semiarid area such as Southern Spain, the analysis of environmental variables shows a high vulnerability in the population to suffer increasingly from eye diseases due to the impact of climate change: higher temperatures, more frequent droughts, forest fires, floods and erosive processes from pouring rain, depletion and pollution of water resources, coastal losses and coastal phenomena, habitat alteration, water-borne diseases and other altered variables like ground-level ozone, pollutant precursors of ground-level ozone, greenhouse gases, acidifying agents, PM10, PM2.5, persistent organic pollutants, benzene, aromatic polycyclic hydrocarbons, dioxins, and furans. In addition, Andalusia’s gross domestic product accounts for 13.33% of Spain’s GDP. In contrast, the Andalusian population accounts for 18.1% of the total Spanish population. This implies lower levels of income and resources for Andalusia, which is the third Spanish autonomous community with the lowest income per capita (19,658€ compared to a national average of 26,438€), only ahead of the autonomous cities of Ceuta and Melilla [53]. Increased risk due to the above mentioned environmental factors and reduced availability of resources contribute to poorer eye health. Exactly to what extent they influence this region should be the subject of future research.
Finally, we have estimated the health expenditure for the fight against these diseases as between 22.63 and 23.31 billion euros per year, as can be seen in Table 2. We have estimated that, on average, 36.5% of this expenditure is attributable to the increase in eye diseases due to environmental factors related to climate change. There is no exact data at the regional level, but the higher incidence of climate change effects estimated for southern Spain indicates that a significant share of this figure corresponds to this region. In an ideal scenario of mitigating the effects of climate change, considering only the costs of afforestation-reforestation and greenhouse gas control, and ignoring the benefits of energy savings, an investment of 13.15 billion euros would be required, but this value is useful for any disease exacerbated by environmental variables related to climate change: this would be the estimated price of disease sinks for Spain.
Adaptation to climate change, therefore, would imply a permanent annual increase in expenditure on eye health to try to mitigate the effects of this process. Mitigation, on the other hand, would imply a reduction in expenditure, as well as an improvement in the health and quality of life of the population. The verification of this fact will depend on the success of global and local measures to mitigate the effects of climate change, but our data suggest that adaptation to climate change involves permanently increasing costs, while mitigation could reduce these if successful.
5. Conclusions and Implications
The increase in eye diseases will have a strong economic and social impact if no mitigation measures are taken against climate change, as much evidence points to a link between weather and climate variables enhanced by climate change and eye-related diseases. Therefore, if proper management can mitigate the effects of climate change, consequently, this can reduce health problems. We propose a new concept: disease sink. By analogy with carbon sinks, a disease sink can be understood as any action to mitigate environmental factors exacerbated by climate change that reduces the incidence or morbidity of disease and consequently generates a health benefit. Producing disease sinks can also bring social and economic benefits resulting from improved environmental quality and savings in climate change adaptation measures. Our data and the scientific literature analysed indicate that southern Spain is highly vulnerable to the effects of climate change. Higher temperatures, more frequent torrential rainfall and droughts, fires, floods, coastal phenomena, alteration of water resources, alteration of habitats and poor pollution control are all elements that directly or indirectly exacerbate numerous eye diseases. At the national level, the estimated health expenditure for the fight against these ocular diseases alone is between 22.63 and 23.31 billion euros per year, with 36.5% of this figure attributed to environmental variables exacerbated by climate change. There is no exact data at the regional level, but the higher incidence of climate change effects estimated for Andalusia indicates that a significant part of this figure is for this region, one of the lowest per capita income regions in Spain.
It would be advisable for other medical specialities to conduct similar studies to emphasize the need to mitigate climate change, as the health care costs of non-intervention in climate change will probably be unaffordable. In addition, more frequent contacts between climate and environmental scientists and medical specialists for joint research would also be desirable. This fact is especially remarkable in those areas where the effects of climate change have the greatest impact: arid and semiarid zones.
In conclusion, medium-term mitigation and long-term reversal of the effects of climate change are essential for our health; adaptation is not an option, as it requires more funds, which not all countries or regions can afford. We hope that this research will guide researchers interested in the role of climate change concerning health in general and eye health in particular, and will help policymakers to redress the economic damage associated with a policy of non-intervention regarding climate change.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/ijerph18137197/s1, Table S1: Pathologies with ocular involvement related to environmental variables, Table S2: Trends of environmental variables in Southern Spain. Table S1 and S2: https://drive.google.com/file/d/1sgHTDZxsekH8O7v8sL6gJVE9GtpWa6k2/view?usp=sharing (accessed on 4 July 2021). References [153,154,155,156,157,158,159,160,161,162,163,164,165,166,167,168,169,170,171,172,173,174,175,176,177,178,179,180,181,182,183,184,185,186,187,188,189,190,191,192,193,194,195,196,197,198,199,200,201,202,203,204,205,206,207,208,209,210,211,212,213,214,215,216,217,218,219,220,221,222,223,224,225,226,227,228,229,230,231,232,233,234,235,236,237,238,239,240,241,242,243,244,245,246,247,248,249,250,251,252,253,254,255,256,257,258] are cited in the Supplementary Materials.
Author Contributions
Conceptualization, L.E.-L. and J.M.S.-G.; methodology, L.E.-L. and J.M.S.-G.; software, J.M.S.-G. and J.R.-C.; validation, L.E.-L., J.M.S.-G., M.E.M.-H. and J.R.-C.; formal analysis, L.E.-L. and J.M.S.-G.; investigation, L.E.-L., J.M.S.-G., M.E.M.-H. and J.R.-C.; resources, L.E.-L. and J.M.S.-G.; data curation, L.E.-L. and J.M.S.-G.; writing—original draft preparation, L.E.-L. and J.M.S.-G.; writing—review and editing, J.M.S.-G. and J.R.-C.; visualization: J.M.S.-G. and J.R.-C.; supervision, J.M.S.-G.; project administration, J.M.S.-G.; funding acquisition, J.M.S.-G. and J.R.-C. All authors have read and agreed to the published version of the manuscript.
Funding
There is no competing financial interest in this research. This research has not received external funding from public or private entities, nor do any of its authors have a personal financial interest in them or any family member or institutional affiliation. Furthermore, none of the authors has received grants or any forms of compensation (travel funding, consultancies, board position, patents, royalty arrangement, stock share or bonds). None of the authors has been nominated for relevant employment or received remuneration for being an expert witness or providing testimony about this ongoing research. The APC was funded by PAIDI Group RNM-279 “Geografía Física y Territorio”.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
No human or animal experiments have been performed in this research. No need for Informed Consents.
Data Availability Statements
Source data are cited in the article. Supplementary Materials Tables S1 and S2 are Excel spreadsheets with processed data from REDIAM (Supplementary Material Table S1) and the different cost calculations by pathology applied from the referenced literature and Andalusian Health Service data (Supplementary Material Table S2). https://drive.google.com/file/d/1LvqRyH_DV83waommEG5wdYI0ptbrQXWl/view?usp=sharing; https://drive.google.com/file/d/1q_eTEw5HAvXm5aWU_s42eS9-Kce63ojS/view?usp=sharing (accessed on 4 July 2021).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Smith, K.R.; Woodward, A. Human Health: Impacts, Adaptation, and Co-Benefits—IPCC. 2014. Available online: https://www.ipcc.ch/report/ar5/wg2/human-health-impacts-adaptation-and-co-benefits/ (accessed on 8 April 2020).
- Christidis, N.; Stott, P.A.; Jones, G.S.; Shiogama, H.; Nozawa, T.; Luterbacher, J. Human activity and anomalously warm seasons in Europe. Int. J. Clim. 2010, 32, 225–239. [Google Scholar] [CrossRef]
- Fouillet, A.; Rey, G.; Wagner, V.; Laaidi, K.; Empereur-Bissonnet, P.; Le Tertre, A.; Frayssinet, P.; Bessemoulin, P.; Laurent, F.; De Crouy-Chanel, P.; et al. Has the impact of heat waves on mortality changed in France since the European heat wave of summer 2003? A study of the 2006 heat wave. Int. J. Epidemiol. 2008, 37, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Johnston, F.H.; Henderson, S.B.; Chen, Y.; Randerson, J.T.; Marlier, M.; DeFries, R.S.; Kinney, P.; Bowman, D.M.; Brauer, M. Estimated Global Mortality Attributable to Smoke from Landscape Fires. Environ. Health Perspect. 2012, 120, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Rice, M.B.; Thurston, G.D.; Balmes, J.R.; Pinkerton, K.E. Climate Change. A Global Threat to Cardiopulmonary Health. Am. J. Respir. Crit. Care Med. 2014, 189, 512–519. [Google Scholar] [CrossRef]
- Tsai, M.-J.; Hsu, Y.-L.; Wu, K.-Y.; Yang, R.-C.; Chen, Y.-J.; Yu, H.-S.; Kuo, P.-L. Heat Effect Induces Production of Inflammatory Cytokines Through Heat Shock Protein 90 Pathway in Cornea Cells. Curr. Eye Res. 2013, 38, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Ludema, C.; Cole, S.R.; Poole, C.; Smith, J.S.; Schoenbach, V.J.; Wilhelmus, K.R. Association between unprotected ultraviolet radiation exposure and recurrence of ocular herpes simplex virus. Am. J. Epidemiol. 2013, 179, 208–215. [Google Scholar] [CrossRef]
- Kawai, K.; VoPham, T.; Drucker, A.; Curhan, S.G.; Curhan, G.C. Ultraviolet Radiation Exposure and the Risk of Herpes Zoster in Three Prospective Cohort Studies. Mayo Clin. Proc. 2020, 95, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Van der Leun, J.C.; Piacentini, R.D.; de Gruijl, F.R. Climate change and human skin cancer. Photochem. Photobiol. Sci. 2008, 7, 730–733. [Google Scholar] [CrossRef]
- Aragonés Cruz, B.; Alemañy Martorell, J. Relación de la radiación ultravioleta y el pterigión primario. Rev. Cuba. Oftalmol. 2009, 22. [Google Scholar]
- Caspi, R.R. A look at autoimmunity and inflammation in the eye. J. Clin. Investig. 2010, 120, 3073–3083. [Google Scholar] [CrossRef]
- Cullen, A.P. Ozone Depletion and Solar Ultraviolet Radiation: Ocular Effects, a United Nations Environment Programme Perspective. Eye Contact Lens Sci. Clin. Pract. 2011, 37, 185–190. [Google Scholar] [CrossRef]
- Consejería de Salud. Junta de Andalucia. Informe Inicial Sobre Adaptación al Cambio Climático en el Ámbito de Salud. 2012. Available online: http://www.juntadeandalucia.es/medioambiente/portal_web/web/temas_ambientales/clima/actuaciones_cambio_climatico/adaptacion/vulnerabilidad_impactos_medidas/isis/isi_salud.pdf (accessed on 24 May 2021).
- Yam, J.C.S.; Kwok, A.K.H. Ultraviolet light and ocular diseases. Int. Ophthalmol. 2014, 34, 383–400. [Google Scholar] [CrossRef]
- IARC Working Group on the Evaluation of Carcinogenic Risk to Humans. Solar and Ultraviolet Radiation. Available online: https://www.ncbi.nlm.nih.gov/books/NBK304366/ (accessed on 13 April 2020).
- Auger, N.; Rhéaume, M.-A.; Bilodeau-Bertrand, M.; Tang, T.; Kosatsky, T. Climate and the eye: Case-crossover analysis of retinal detachment after exposure to ambient heat. Environ. Res. 2017, 157, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Barcena-Martin, E.; Molina, J.; Ruiz-Sinoga, J.D. Issues and challenges in defining a heat wave: A Mediterranean case study. Int. J. Clim. 2019, 39, 331–342. [Google Scholar] [CrossRef]
- Medina Martín, F. Impactos, Vulnerabilidad y Adaptación al Cambio Climático en el Sector Agrario. Aproximación al Conocimiento y Prácticas de Gestión en España. 2016. Available online: https://www.miteco.gob.es/es/cambio-climatico/temas/impactos-vulnerabilidad-y-adaptacion/impactos_vulnerabilidad_adaptacion_cambio_climatico_sector_agrario__tcm30-178448.pdf (accessed on 24 May 2021).
- Bárcena-Martín, E.; Molina, J.; Hueso, P.; Ruiz-Sinoga, J.D. A Class of Indices and a Graphical Tool to Monitor Temperature Anomalies. Air Soil Water Res. 2020, 13, 1–11. [Google Scholar] [CrossRef]
- Sinoga, J.D.R.; Marin, R.G.; Murillo, J.F.M.; Galeote, M.A.G. Precipitation dynamics in southern Spain: Trends and cycles. Int. J. Clim. 2010, 31, 2281–2289. [Google Scholar] [CrossRef]
- Senciales-González, J.M.; Ruiz Sinoga, J. Análisis espacio-temporal de las lluvias torrenciales en la ciudad de Málaga. Bol. Asoc. Geógrafos Españoles 2013, 61, 7–24. [Google Scholar] [CrossRef]
- Polgreen, P.M.; Polgreen, E.L. Infectious Diseases, Weather, and Climate. Clin. Infect. Dis. 2017, 66, 815–817. [Google Scholar] [CrossRef] [PubMed]
- Burton, M.J. Trachoma: An overview. Br. Med. Bull. 2007, 84, 99–116. [Google Scholar] [CrossRef]
- Last, A.; Versteeg, B.; Abdurahman, O.S.; Robinson, A.; Dumessa, G.; Aga, M.A.; Bejiga, G.S.; Negussu, N.; Greenland, K.; Czerniewska, A.; et al. Detecting extra-ocular Chlamydia trachomatis in a trachoma-endemic community in Ethiopia: Identifying potential routes of transmission. PLoS Negl. Trop. Dis. 2020, 14, e0008120. [Google Scholar] [CrossRef]
- Angelini, P.; Macini, P.; Finarelli, A.C.; Pol, C.; Venturelli, C.; Bellini, R.; Dottori, M. Chikungunya epidemic outbreak in Emilia-Romagna (Italy) during summer 2007. Parasitologia 2008, 50, 97–98. [Google Scholar]
- Bai, L.; Morton, L.; Liu, Q. Climate change and mosquito-borne diseases in China: A review. Glob. Health 2013, 9, 10. [Google Scholar] [CrossRef] [PubMed]
- Caminade, C.; Ndione, J.-A.; Kebe, C.M.F.; Jones, A.E.; Danuor, S.K.; Tay, S.; Tourre, Y.M.; Lacaux, J.-P.; Vignolles, C.; Duchemin, J.B.; et al. Mapping Rift Valley fever and malaria risk over West Africa using climatic indicators. Atmos. Sci. Lett. 2010, 12, 96–103. [Google Scholar] [CrossRef]
- Anyamba, A.; Linthicum, K.J.; Small, J.L.; Collins, K.M.; Tucker, C.J.; Pak, E.W.; Britch, S.C.; Eastman, J.R.; Pinzon, J.E.; Russell, K.L. Climate Teleconnections and Recent Patterns of Human and Animal Disease Outbreaks. PLoS Negl. Trop. Dis. 2012, 6, e1465. [Google Scholar] [CrossRef] [PubMed]
- Ben Yahia, S.; Khairallah, M. Ocular Manifestations of West Nile Virus Infection. Int. J. Med. Sci. 2009, 6, 114–115. [Google Scholar] [CrossRef] [PubMed]
- ECDC. Weekly Updates: 2020 West Nile Virus Transmission Season. 2020. Available online: https://www.ecdc.europa.eu/en/west-nile-fever/surveillance-and-disease-data/disease-data-ecdc (accessed on 2 October 2020).
- Madsen, L.M. How Global Warming May Affect the Prevalence of Lyme Disease. 2019. Available online: https://www.vetfolio.com/learn/article/how-global-warming-may-affect-the-prevalence-of-lyme-disease (accessed on 13 April 2020).
- Yip, V.C.-H.; Sanjay, S.; Koh, Y.T. Ophthalmic Complications of Dengue Fever: A Systematic Review. Ophthalmol. Ther. 2012, 1, 1–19. [Google Scholar] [CrossRef]
- Mahendradas, P.; Avadhani, K.; Shetty, R. Chikungunya and the eye: A review. J. Ophthalmic Inflamm. Infect. 2013, 3, 35. [Google Scholar] [CrossRef]
- Ghosh, D.; Basu, A. Japanese Encephalitis—A Pathological and Clinical Perspective. PLoS Negl. Trop. Dis. 2009, 3, e437. [Google Scholar] [CrossRef]
- Al-Hazmi, A.; Al-Rajhi, A.A.; Abboud, E.B.; Ayoola, E.A.; Al-Hazmi, M.; Saadi, R.; Ahmed, N. Ocular complications of Rift Valley fever outbreak in Saudi Arabia. Ophthalmology 2005, 112, 313–318. [Google Scholar] [CrossRef]
- Bell, M.L.; Peng, R.D.; Dominici, F. The Exposure–Response Curve for Ozone and Risk of Mortality and the Adequacy of Current Ozone Regulations. Environ. Health Perspect. 2006, 114, 532–536. [Google Scholar] [CrossRef]
- Dear, K.; Ranmuthugala, G.; Kjellström, T.; Skinner, C.; Hanigan, I. Effects of Temperature and Ozone on Daily Mortality During the August 2003 Heat Wave in France. Arch. Environ. Occup. Health 2005, 60, 205–212. [Google Scholar] [CrossRef]
- Ren, C.; Williams, G.; Morawska, L.; Mengersen, K.; Tong, S. Ozone modifies associations between temperature and cardiovascular mortality: Analysis of the NMMAPS data. Occup. Environ. Med. 2008, 65, 255–260. [Google Scholar] [CrossRef]
- Rodrigo-Comino, J.; Senciales-González, J.M. A Regional Geography Approach to Understanding the Environmental Changes as a Consequence of the COVID-19 Lockdown in Highly Populated Spanish Cities. Appl. Sci. 2021, 11, 2912. [Google Scholar] [CrossRef]
- Beggs, P.J. Adaptation to Impacts of Climate Change on Aeroallergens and Allergic Respiratory Diseases. Int. J. Environ. Res. Public Health 2010, 7, 3006–3021. [Google Scholar] [CrossRef] [PubMed]
- Saxena, R.; Srivastava, S.; Trivedi, D.; Anand, E.; Joshi, S.; Gupta, S.K. Impact of environmental pollution on the eye. Acta Ophthalmol. Scand. 2003, 81, 491–494. [Google Scholar] [CrossRef]
- Shubhrica, D. Effect of Environment on Eyes: A Review. Indian J. Clin. Pract. 2013, 24, 381–384. [Google Scholar]
- Gupta, P.; Muthukumar, A. Minor to Chronic Eye Disorders Due to Environmental Pollution: A review. J. Ocul. Infect. Inflamm. 2018, 2, 2. [Google Scholar]
- Knox, J.; Hess, T.; Daccache, A.; Wheeler, T. Climate change impacts on crop productivity in Africa and South Asia. Environ. Res. Lett. 2012, 7. [Google Scholar] [CrossRef]
- Brevik, E.C.; Oliver, M.; Zhao, F.-J. Special section on soil and human health—An editorial. Eur. J. Soil Sci. 2019, 70, 859–861. [Google Scholar] [CrossRef]
- Müller, O.; Krawinkel, M. Malnutrition and health in developing countries. Can. Med. Assoc. J. 2005, 173, 279–286. [Google Scholar] [CrossRef]
- Kraemer, K.; Gilbert, C. Do vitamin A deficiency and undernutrition still matter? Community Eye Health 2013, 26, 61–63. [Google Scholar] [PubMed]
- Chandran, R.; Consultant, N.; Gedam, D.S. Ocular manifestations of childhood malnutrition- an overview. Int. J. Med. Res. Rev. 2017, 5, 925–926. [Google Scholar] [CrossRef]
- Dantas, A.P.; Brandt, C.T.; Leal, D.N.B. Manifestações oculares em pacientes que tiveram desnutrição nos primeiros seis meses de vida. Arq. Bras. Oftalmol. 2005, 68, 753–756. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Araque, A.; Aranda, A.G.; Pardo, C.L.; Aragüés, A.R. Los antioxidantes en el proceso de patologías oculares. Nutr. Hosp. 2017, 34, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Sahu, S.; Sett, M.; Kjellstrom, T. Heat Exposure, Cardiovascular Stress and Work Productivity in Rice Harvesters in India: Implications for a Climate Change Future. Ind. Health 2013, 51, 424–431. [Google Scholar] [CrossRef]
- Jovanovic, N.; Peek-Asa, C.; Swanton, A.; Young, T.; Alajbegovic-Halimic, J.; Cavaljuga, S.; Nisic, F. Prevalence and risk factors associated with work-related eye injuries in Bosnia and Herzegovina. Int. J. Occup. Environ. Health 2016, 22, 325–332. [Google Scholar] [CrossRef]
- INE. Available online: https://www.ine.es/jaxiT3/Datos.htm?t=2911 (accessed on 24 May 2021).
- Oppenheimer, M.; Campos, M.; Warren, R.; Birkmann, J.; Luber, G.; O’Neill, B.; Takahashi, K. Emergent risks and key vulnerabilities. In Climate Change 2014: Impacts, Adaptation, and Vulnerability. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Field, C.B., Barros, V.R., Dokken, D.J., Mach, K.J., Mastrandrea, M.D., Bilir, T.E., Chatterjee, M., Ebi, K.L., Estrada, Y.O., Genova, R.C., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2014; pp. 1039–1099. [Google Scholar]
- PCC. AR5 Climate Change 2013: The Physical Science Basis—IPCC. 2013. Available online: https://www.ipcc.ch/report/ar5/wg1/ (accessed on 16 April 2020).
- Pachauri, R.K.; Mayer, L. (Eds.) Climate Change 2014: Synthesis Report; Intergovernmental Panel on Climate Change: Geneva, Switzerland, 2015. [Google Scholar]
- Bowling, B. Oftalmología Clínica. Un Enfoque Sistemático; Elsevier Masson: Paris, France, 2016; Available online: https://www.iberlibro.com/KANSKI-OFTALMOLOG%C3%8DA-CL%C3%8DNICA-B-BOWLING-ELSEVIER/20642079458/bd (accessed on 29 April 2020).
- Gómez-Ulla de Irazazábal, F.; Ondategui Parra, S. Informe Sobre la Ceguera en España. 2009. Available online: https://www.seeof.es/archivos/articulos/adjunto_20_1.pdf (accessed on 23 January 2021).
- Díaz-Llopis, M.; Calonge, M.; Sáinz de la Maza, M.T.; Benítez del Castillo, J.M.; Gallego Pinazo, R.; Arévalo, F. Uveítis y escleritis. Diagnóstico y tratamiento. Available online: https://www.oftalmoseo.com/libros_seo/ponencia_seo/uveitis-y-escleritis-diagnostico-y-tratamiento/ (accessed on 18 May 2021).
- Gichuhi, S.; Macharia, E.; Kabiru, J.; Zindamoyen, A.M.; Rono, H.; Ollando, E.; Wachira, J.; Munene, R.; Maina, J.; Onyuma, T.; et al. Topical fluorouracil after surgery for ocular surface squamous neoplasia in Kenya: A randomised, double-blind, placebo-controlled trial. Lancet Glob. Health 2016, 4, e378–e385. [Google Scholar] [CrossRef]
- Estelle, I. Cambio Climático—Escenario Actual, Salud e Implicancias en la Población Chilena. 2020. Available online: https://www.academia.edu/42152825/CAMBIO_CLIM%C3%81TICO_-ESCENARIO_ACTUAL_SALUD_E_IMPLICANCIAS_EN_LA_POBLACI%C3%93N_CHILENA (accessed on 13 April 2020).
- WHO. Infectious Diseases. 2020. Available online: https://www.who.int/topics/infectious_diseases/en/ (accessed on 6 November 2020).
- WHO. Water Sanitation and Health. 2020. Available online: http://www.who.int/water_sanitation_health/en/ (accessed on 8 May 2020).
- Cassel, G.H.; Burrows, A.; Jeffers, J.B.; Fischer, D.H. Anterior nongranulomatous uveitis: A seasonal variation. Ann. Ophthalmol. 1984, 16, 1066–1068. [Google Scholar]
- Luca, C.; Raffaella, A.; Sylvia, M.; Valentina, M.; Fabiana, V.; Marco, C.; Annamaria, S.; Luisa, S.; Alessandro, D.F.; Lucia, B.; et al. Changes in patterns of uveitis at a tertiary referral center in Northern Italy: Analysis of 990 consecutive cases. Int. Ophthalmol. 2017, 38, 133–142. [Google Scholar] [CrossRef]
- Rea, W.J.; Patel, K.D. Reversibility of Chronic Disease and Hypersensitivity: The Effects of Environmental Pollutants on the Organ System; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- NASA. Global Climate Change. Available online: https://climate.nasa.gov/solutions/adaptation-mitigation/ (accessed on 3 June 2021).
- Zhao, C.; Yan, Y.; Wang, C.; Tang, M.; Wu, G.; Ding, D.; Song, Y. Adaptation and mitigation for combating climate change—From single to joint. Ecosyst. Health Sustain. 2018, 4, 85–94. [Google Scholar] [CrossRef]
- Ginbo, T.; Di Corato, L.; Hoffmann, R. Investing in climate change adaptation and mitigation: A methodological review of real-options studies. Ambio 2021, 50, 229–241. [Google Scholar] [CrossRef] [PubMed]
- European Commission. EU Reference Scenario Energy, Transport and GHG Emissions Trends to 2050. 2016. Available online: https://ec.europa.eu/energy/sites/ener/files/documents/20160713%20draft_publication_REF2016_v13.pdf (accessed on 24 May 2021).
- Sillero-Medina, J.; Pérez-González, M.; Martínez-Murillo, J.; Ruiz-Sinoga, J. Factors affecting eco-geomorphological dynamics in two contrasting Mediterranean environments. Geomorphology 2020, 352, 106996. [Google Scholar] [CrossRef]
- Rodrigo-Comino, J.; Senciales, J.M.; Sillero-Medina, J.A.; Gyasi-Agyei, Y.; Ruiz-Sinoga, J.D.; Ries, J.B. Analysis of Weather-Type-Induced Soil Erosion in Cultivated and Poorly Managed Abandoned Sloping Vineyards in the Axarquía Region (Málaga, Spain). Air Soil Water Res. 2019, 12, 1178622119839403. [Google Scholar] [CrossRef]
- Diputación de Málaga. Estudio Sobre la Cuenca del Río Guadalhorce y Cuatro Tramos Representativos. Montes y Caminos. Ingenieros Consultores. 2013. Available online: http://static.malaga.es/malaga/subidas/descargas/archivos/3/2/206223/estudio-cuenca-del-rio-guadalhorce.pdf (accessed on 16 January 2021).
- REDMAR (Red de Medidas del nivel del mar y agitación de Puertos del Estado). Puerto de Málaga. Resumen de Parámetros Relacionados con el Nivel del Mar y la Marea que Afectan a Las Condiciones de Diseño y Explotación Portuaria. 2019. Available online: http://www.puertos.es/es-es/oceanografia/Paginas/portus.aspx (accessed on 6 February 2021).
- Alonso, B.L.; Rosel, A.B.; Villaverde-Royo, M.; Alonso, I.L. Malaria por Plasmodium falciparum en residente en España sin antecedente de viaje reciente a zonas endémicas. Semergen 2016, 42, e71–e72. [Google Scholar] [CrossRef] [PubMed]
- Junta de Andalucía. Informe del Inventario de Emisiones a la Atmósfera de la Comunidad Autónoma de Andalucía; Serie 2003–2016. Edición 2019. 2019. Available online: http://www.juntadeandalucia.es/medioambiente/site/portalweb/menuitem.7e1cf46ddf59bb227a9ebe205510e1ca/?vgnextoid=139f3d9292df3610VgnVCM100000341de50aRCRD&vgnextchannel=a35b445a0b5f4310VgnVCM2000000624e50aRCRD&lr=lang_es (accessed on 21 April 2020).
- Sanz, M.J.; Galán, E. Impactos y Riesgos Derivados del Cambio Climático en España; Ministerio para la Transición Ecológica y el Reto Demográfico (MITECO): Madrid, Spain, 2020.
- Giorgi, F.; Lionello, P. Climate change projections for the Mediterranean region. Glob. Planet. Chang. 2008, 63, 90–104. [Google Scholar] [CrossRef]
- Chang, J.; Smiddy, W.E. Cost-Effectiveness of Retinal Detachment Repair. Ophthalmology 2014, 121, 946–951. [Google Scholar] [CrossRef] [PubMed]
- Araiz Iribarren, J.; Piñero Bustamante, A. Manejo de las Degeneraciones Periféricas de la Retina; SERV, Sociedad Española de Retina y Vítreo: Madrid, Spain, 2017. [Google Scholar]
- AEMET. Julio de 2020 Finaliza con Récords de Temperatura y Riesgo Extremo de Incendios. 2020. Available online: http://www.aemet.es/es/noticias/2020/07/Olacalor_incendios_jul2020 (accessed on 7 November 2020).
- AEMET. Olas de Calor en España desde Área de Climatología y Aplicaciones Operativas. 2020. Available online: http://www.aemet.es/documentos/es/conocermas/recursos_en_linea/publicaciones_y_estudios/estudios/Olas_calor/Olas_Calor_ActualizacionMarzo2020.pdf (accessed on 24 May 2021).
- Tomany, S.C.; Cruickshanks, K.J.; Klein, R.; Klein, B.E.K.; Knudtson, M.D. Sunlight and the 10-Year Incidence of Age-Related Maculopathy. Arch. Ophthalmol. 2004, 122, 750–757. [Google Scholar] [CrossRef]
- Junta de Andalucía. Ozono y Radiación Ultravioleta en Andalucía. 2014. Available online: http://www.juntadeandalucia.es/medioambiente/site/ima/menuitem.5893969315ab596f7bbe6c6f5510e1ca/?vgnextoid=7be208a2c7001510VgnVCM2000000624e50aRCRD&vgnextchannel=8ac91193c89e0510VgnVCM1000001325e50aRCRD&lr=lang_es) (accessed on 24 May 2021).
- Junta de Andalucía. Relación de Indicadores Ambientales de Andalucía 2019. Índice Ultravioleta. Available online: https://www.juntadeandalucia.es/medioambiente/portal/landing-page-indicador/-/asset_publisher/OGeXai6LtflU/content/espesor-de-la-capa-de-ozono/20151?categoryVal (accessed on 8 May 2021).
- Agencia Estatal de Meteorología—AEMET; Gobierno de España. Radiación y Ozono—Radiación Ultravioleta (UVI) y Ozono. 2020. Available online: http://www.aemet.es/es/serviciosclimaticos/vigilancia_clima/radiacion_ozono?w=1 (accessed on 8 November 2020).
- Xia, N.; Chen, L.; Chen, H.; Luo, X.; Deng, T. Influence of atmospheric relative humidity on ultraviolet flux and aerosol direct radiative forcing: Observation and simulation. Asia Pac. J. Atmos. Sci. 2016, 52, 341–352. [Google Scholar] [CrossRef]
- WHO. Climate Change and Health. 2018. Available online: https://www.who.int/news-room/fact-sheets/detail/climate-change-and-health (accessed on 7 November 2020).
- INE. Índices Nacionales de Subclases. Índices de Precios al Consumo. 2020. Available online: https://www.ine.es/jaxiT3/Datos.htm?t=22347#!tabs-grafico (accessed on 8 November 2020).
- EFEverde + Redacción. Desastres Naturales: Treinta y Cinco Muertos en España en EFEverde. 2020. Available online: https://www.efeverde.com/noticias/desastres-naturales-treinta-y-cinco-muertos-en-espana-en-2012/ (accessed on 8 November 2020).
- Glaucoma Research Fundation. Datos y Estadísticas Sobre el Glaucoma. 2020. Available online: https://www.glaucoma.org/es/datos-y-estadisticas-sobre-el-glaucoma.php (accessed on 8 November 2020).
- Statista. Glaucoma: Número de Casos 2011–2016. Statista. Available online: https://es.statista.com/estadisticas/991862/numero-de-casos-de-glaucoma-en-espana/ (accessed on 17 November 2020).
- Wang, W.; He, M.; Li, Z.; Huang, W. Epidemiological variations and trends in health burden of glaucoma worldwide. Acta Ophthalmol. 2019, 97, e349–e355. [Google Scholar] [CrossRef]
- Osborne, N.N.; Lascaratos, G.; Bron, A.J.; Chidlow, G.; Wood, J.P.M. A hypothesis to suggest that light is a risk factor in glaucoma and the mitochondrial optic neuropathies. Br. J. Ophthalmol. 2006, 90, 237–241. [Google Scholar] [CrossRef]
- Ivanov, I.V.; Mappes, T.; Schaupp, P.; Lappe, C.; Wahl, S. Ultraviolet radiation oxidative stress affects eye health. J. Biophotonics 2018, 11, e201700377. [Google Scholar] [CrossRef] [PubMed]
- Europa Press. La cirugía de Catarata es Uno de los Procedimientos Quirúrgicos Más Comunes. 2015. Available online: https://www.infosalus.com/asistencia/noticia-cirugia-catarata-procedimientos-quirurgicos-mas-comunes-20150518140228.html (accessed on 9 November 2015).
- Sasaki, K.; Hockwin, O. Progress in Lens and Cataract Research: In Honour of Professor Kazuyuki Sasaki; Karger Medical and Scientific Publishers: Basel, Switzerland, 2002. [Google Scholar]
- Ministerio de Sanidad, Servicios Sociales e Igualdad. Informe Anual del Sistema Nacional de Salud 2015. 2016. Available online: https://www.mscbs.gob.es/estadEstudios/estadisticas/sisInfSanSNS/tablasEstadisticas/Inf_Anual_SNS_2015.1.pdf (accessed on 15 September 2020).
- Vidal, M.J.; Labeaga, J.M.; Casado, P.; Madrigal, A.; López, J.; Montero, A.; Meil, G. Informe 2016: Las Personas Mayores en España. Datos estadísticos estatales y por Comunidades Autónomas; Ministerio de Sanidad, Servicios Sociales e Igualdad, IMSERSO: Madrid, Spain, 2017; p. 540.
- Servicio Andaluz de Salud. Tiempos de Respuesta Asistencial. Listas de Espera. 2020. Available online: https://www.sspa.juntadeandalucia.es/servicioandaluzdesalud/ciudadania/derechos-y-garantias/tiempos-de-respuesta-asistencial-listas-de-espera (accessed on 9 November 2020).
- Zak-Prelich, M.; Borkowski, J.L.; Alexander, F.; Norval, M. The role of solar ultraviolet irradiation in zoster. Epidemiol. Infect. 2002, 129, 593–597. [Google Scholar] [CrossRef] [PubMed]
- Salvador Velázquez, P. Estimación del Coste Económico de la Asistencia a los Pacientes Diagnosticados de Herpes-Zoster; Trabajo de Investigación; Departamento de Farmacología: Terapéutica y Toxicología, UAB (Universidad Autónoma de Barcelona), 2012; p. 25. Available online: https://ddd.uab.cat/pub/trerecpro/2012/hdl_2072_203218/TR-SalvadorVelazquez.pdf (accessed on 22 May 2021).
- Ojeda Ruiz, E.; Vila Cordero, B.; López-Perea, N.; Carmona Alférez, R. Informe Epidemiológico Sobre la Situación de Herpes Zóster en España, 1998–2018. 2020. Available online: https://www.isciii.es/QueHacemos/Servicios/VigilanciaSaludPublicaRENAVE/EnfermedadesTransmisibles/Documents/archivos%20A-Z/HERPES%20ZOSTER/Informe_HZ_Espa%C3%B1a_1998-2018.pdf (accessed on 24 May 2021).
- Centro Nacional de Epidemiologia. Infección Genital por el Virus Herpes Simple. Sistema de Información Microbiológica. España. Años 2000–2008. Boletín epidemiológico Semin. 2011, 19, 1–5. [Google Scholar]
- Villanueva Blandón, R. Estudio Retrospectivo para la Evaluación de la Eficacia del Aciclovir Oral en la Prevención de Recurrencias en Pacientes con Queratitis Herpética Recidivante. 2016. Available online: https://idus.us.es/bitstream/handle/11441/39293/Tesis%20Rafael%20Villanueva.pdf?sequence=1&isAllowed=y (accessed on 24 May 2021).
- Statista. Herpes Simple: Número de casos 2011–2017. 2018. Available online: https://es.statista.com/estadisticas/1038556/numero-de-casos-de-herpes-simple-en-espana/ (accessed on 17 November 2020).
- Hart, J.E.; Laden, F.; Puett, R.C.; Costenbader, K.H.; Karlson, E.W. Exposure to Traffic Pollution and Increased Risk of Rheumatoid Arthritis. Environ. Health Perspect. 2009, 117, 1065–1069. [Google Scholar] [CrossRef]
- Sociedad Española de Reumatología. El Retraso en el Diagnóstico de Pacientes con Artritis Reumatoide se ha Reducido a Menos de 8 Meses. 2018. Available online: https://www.ser.es/retraso-diagnostico-pacientes-artritis-reumatoide-se-ha-reducido-menos-8-meses/ (accessed on 9 November 2020).
- Soleimanifar, N.; Nicknam, M.H.; Bidad, K.; Jamshidi, A.R.; Mahmoudi, M.; Mostafaei, S.; Hosseini-Khah, Z.; Nikbin, B. Effect of food intake and ambient air pollution exposure on ankylosing spondylitis disease activity. Adv. Rheumatol. 2019, 59, 9. [Google Scholar] [CrossRef]
- Plazuelo Ramos, P. Atlas de Espondiloartritis Axial en España 2017: Radiografía de la Enfermedad; Instituto Max Weber: Madrid, Spain, 2017. [Google Scholar]
- Jeanjean, M.; Bind, M.-A.; Roux, J.; Ongagna, J.-C.; de Sèze, J.; Bard, D.; Leray, E. Ozone, NO2 and PM10 are associated with the occurrence of multiple sclerosis relapses. Evidence from seasonal multi-pollutant analyses. Environ. Res. 2018, 163, 43–52. [Google Scholar] [CrossRef]
- Gallego-Gallego, M.; Anillo-Lombana, V.E.; Gómez-Mayordomo, V.; García-Miguel, F.J. Uhthoff’s phenomenon in a patient with multiple sclerosis during the perioperative period for hip surgery. Case report. Colomb. J. Anesthesiol. 2018, 46, 345–348. [Google Scholar] [CrossRef]
- Gaceta Médica. Cada Año se Diagnostican Unos 2000 Nuevos Casos de Esclerosis Múltiple en España. 2020. Available online: https://gacetamedica.com/investigacion/cada-ano-se-diagnostican-unos-2-000-nuevos-casos-de-esclerosis-multiple-en-espana/ (accessed on 9 November 2020).
- Panginikkod, S.; Rayi, A.; Rocha Cabrero, F.; Rukmangadachar, L.A. Uhthoff Phenomenon; StatPearls Publishing: Petersburg, FL, USA, 2020. Available online: http://www.ncbi.nlm.nih.gov/books/NBK470244/ (accessed on 9 November 2020).
- Manser, C.N.; Paul, M.; Rogler, G.; Held, L.; Frei, T. Heat Waves, Incidence of Infectious Gastroenteritis, and Relapse Rates of Inflammatory Bowel Disease: A Retrospective Controlled Observational Study. Am. J. Gastroenterol. 2013, 108, 1480–1485. [Google Scholar] [CrossRef] [PubMed]
- Sambuelli, M.; Negreira, S.; Gil, A.; Goncalves, S.; Chavero, P.; Tirado, P.; Bellicoso, M.; Huernos, S. Manejo de la Enfermedad Inflamatoria Intestinal. Revisión y Algoritmos de Tratamiento. 2019. Available online: http://actagastro.org/manejo-de-la-enfermedad-inflamatoria-intestinal-revision-y-algoritmos-de-tratamientos/ (accessed on 9 November 2020).
- Rúa-Figueroa, I.; López-Longo, F.J.; Calvo-Alén, J.; Galindo, M.; Loza, E.; de Yebenes, M.J.G.; Pego-Reigosa, J.M. Registro nacional de pacientes con lupus eritematoso sistémico de la Sociedad Española de Reumatología: Objetivos y metodología. Reumatol. Clínica 2014, 10, 17–24. [Google Scholar] [CrossRef]
- Cervera, R.; Rúa-Figueroa, I.; Gil-Aguado, A.; Sabio, J.; Pallarés, L.; Hernández-Pastor, L.; Iglesias, M. Coste económico directo del control y el tratamiento del lupus eritematoso sistémico activo y sus brotes en España: Estudio LUCIE. Rev. Clínica Española 2013, 213, 127–137. [Google Scholar] [CrossRef]
- Cooper, G.S.; Wither, J.; Bernatsky, S.; Claudio, J.O.; Clarke, A.; Rioux, J.D.; Fortin, P.R. CaNIOS GenES Investigators Occupational and environmental exposures and risk of systemic lupus erythematosus: Silica, sunlight, solvents. Rheumatology 2010, 49, 2172–2180. [Google Scholar] [CrossRef] [PubMed]
- UNIMID. Asociación de Personas con Enfermedades Crónicas Inflamatorias Inmunomediadas. 2019. Available online: http://www.unimid.es/ (accessed on 9 November 2020).
- Rice, J.B.; White, A.; Lopez, A.; Conway, A.; Wagh, A.; Nelson, W.W.; Philbin, M.; Wan, G.J. Economic burden of sarcoidosis in a commercially-insured population in the United States. J. Med. Econ. 2017, 20, 1048–1055. [Google Scholar] [CrossRef]
- Schweitzer, M.D.; Calzadilla, A.S.; Salamo, O.; Sharifi, A.; Kumar, N.; Holt, G.; Campos, M.; Mirsaeidi, M. Lung health in era of climate change and dust storms. Environ. Res. 2018, 163, 36–42. [Google Scholar] [CrossRef] [PubMed]
- AEMET. Informe Annual, 2017. 2018. Available online: https://www.aemet.es/documentos/es/conocenos/a_que_nos_dedicamos/informes/InformeAnualAEMET_2017_web.pdf (accessed on 24 May 2021).
- Erdal, B.S.; Clymer, B.D.; Yildiz, V.O.; Julian, M.W.; Crouser, E.D. Unexpectedly high prevalence of sarcoidosis in a representative U.S. Metropolitan population. Respir. Med. 2012, 106, 893–899. [Google Scholar] [CrossRef]
- AARDA (American Autoimmune Related Diseases Association). The Cost Burden of Autoimmune Disease: The Latest Front in the War on Healthcare Spending. 2020. Available online: http://www.diabetesed.net/page/_files/autoimmune-diseases.pdf (accessed on 1 May 2021).
- El País. Los SUV y la Gasolina, Responsables del Incremento de las Emisiones en Españ. El Motor. 2020. Available online: https://motor.elpais.com/actualidad/los-suv-y-la-gasolina-responsables-del-incremento-de-las-emisiones-en-espana/ (accessed on 18 November 2020).
- INE. Población Urbana Expuesta a Contaminación Del Aire (Micropartículas PM10, PM2,5). 2020. Available online: https://www.ine.es/ss/Satellite?L=es_ES&c=INESeccion_C&cid=1259944618679&p=1254735110672&pagename=ProductosYServicios%2FPYSLayout¶m1=PYSDetalleFichaIndicador¶m3=1259947308577 (accessed on 18 November 2020).
- Ceballos, M.Á. La Contaminación por Ozono en el Estado Español. 2019. Available online: https://www.ecologistasenaccion.org/wp-content/uploads/2019/10/informe-ozono-2019.pdf (accessed on 24 May 2021).
- El Hamichi, S.; Gold, A.; Murray, T.G.; Graversen, V.K. Pandemics, climate change, and the eye. Graefe’s Arch. Clin. Exp. Ophthalmol. 2020, 258, 2597–2601. [Google Scholar] [CrossRef]
- Segnalini, M.; Bernabucci, U.; Vitali, A.; Nardone, A.; Lacetera, N. Temperature humidity index scenarios in the Mediterranean basin. Int. J. Biometeorol. 2012, 57, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Lozano, R. Envisioning sustainability three-dimensionally. J. Clean. Prod. 2008, 16, 1838–1846. [Google Scholar] [CrossRef]
- WHO. World Report on Vision. 2019. Available online: https://www.who.int/publications/i/item/world-report-on-vision (accessed on 15 September 2020).
- Killmann, W. Instruments related to the United Nations Framework Convention on Climate Change and Their Potential for Sustainable Forest Management in Africa. Forest and Climate Change Working Paper FAO. Available online: http://www.fao.org/3/ac836e/AC836E01.htm#P45_290 (accessed on 24 May 2021).
- Gordois, A.; Pezzullo, L.; Cutler, H. The Global Economic Cost of Visual Impairment. 2010. Available online: http://www.icoph.org/dynamic/attachments/resources/globalcostofvi_finalreport.pdf (accessed on 24 May 2021).
- MITECO. Mitigación del Cambio Climático. 2020. Available online: https://www.miteco.gob.es/es/cambio-climatico/temas/mitigacion-politicas-y-medidas/mitigacion.aspx (accessed on 10 November 2020).
- Fischer, G.; Tubiello, F.N.; van Velthuizen, H.; Wiberg, D. Climate change impacts on irrigation water requirements: Effects of mitigation, 1990–2080. Technol. Forecast. Soc. Chang. 2007, 74, 1083–1107. [Google Scholar] [CrossRef]
- FAO; UNEP. The State of the World’s Forests Forests, Biodiversity and People; FAO: Rome, Italy, 2020. [Google Scholar] [CrossRef]
- Xunta de Galicia. LIFE14 CCM/ES/001271 “LIFE FOREST CO2” “Assessment of Forest-Carbon Sinks and Promotion of Compensation Systems as Tools for Climate Change Mitigation.” 2016. Available online: http://lifeforestco2.eu/wp-content/uploads/2017/02/A.1.01_Estudio_diagnostico_iniciativas_compensacion.pdf (accessed on 24 May 2021).
- The World Bank. GDP (Current US $)—Data. 2020. Available online: https://data.worldbank.org/indicator/NY.GDP.MKTP.CD (accessed on 10 November 2020).
- González-Eguino, M. Los costes de mitigar el cambio climático: Un análisis dinámico de equilibrio general aplicado. Rev. Econ. Apl. 2011, XIX, 89–121. [Google Scholar]
- MITECO. Envío a Cortes del Proyecto de Ley de Cambio Climático y Transición Energética Para Alcanzar la Neutralidad de Emisiones a Más Tardar en 2050. 2020. Available online: https://www.miteco.gob.es/es/prensa/ultimas-noticias/el-gobierno-env%C3%ADa-a-las-cortes-el-primer-proyecto-de-ley-de-cambio-clim%C3%A1tico-y-transici%C3%B3n-energ%C3%A9tica-para-alcanzar-la-neutralidad-de-emisiones-a/tcm:30-509229 (accessed on 10 November 2020).
- Hidalgo-Betanzos, J.M.; Iríbar-Solaberrieta, E.; de Lorenzo Urién, A. Guía Básica para el Control Térmico en Edificación. 2014. Available online: https://www.euskadi.eus/contenidos/informacion/area_termica_public/es_def/adjuntos/GB_control_termico.pdf (accessed on 24 May 2021).
- Energía Eficiente. Situación Energética Mundial. 2009. Available online: https://energiaeficiente.wordpress.com/2009/10/22/situacion-energetica-mundial/ (accessed on 10 November 2020).
- García-Pando, C. Cómo afecta la eficiencia energética al valor de la vivienda? 2014. Available online: https://www.pisos.com/aldia/como-afecta-la-eficiencia-energetica-al-valor-de-la-vivienda/44088/ (accessed on 10 November 2020).
- Rose, J.B.; Daeschner, S.; Easterling, D.R.; Curriero, F.C.; Lele, S.; Patz, J.A. Climate and waterborne disease outbreaks. J. Am. Water Work. Assoc. 2000, 92, 77–87. [Google Scholar] [CrossRef]
- Ecologistas en Acción. Propuestas para Reducir la Demanda Energética. 2011. Available online: https://www.ecologistasenaccion.org/19899/propuestas-para-reducir-la-demanda-energetica/ (accessed on 18 November 2020).
- Mukhopadhyay, R.; Karisiddaiah, S.; Mukhopadhyay, J. Climate Change: Alternate Governance Policy for South Asia; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar]
- El País. El 70% de los Últimos Brotes Epidémicos Han Comenzado con la Deforestación. 2021. Available online: https://elpais.com/ciencia/2021-02-05/el-70-de-los-ultimos-brotes-epidemicos-han-comenzado-con-la-deforestacion.html (accessed on 19 May 2021).
- Andersen, K.G.; Rambaut, A.; Lipkin, W.I.; Holmes, E.C.; Garry, R.F. The proximal origin of SARS-CoV-2. Nat. Med. 2020, 26, 450–452. [Google Scholar] [CrossRef]
- SanJuan-Reyes, S.; Gómez-Oliván, L.M.; Islas-Flores, H. COVID-19 in the environment. Chemosphere 2021, 263, 127973. [Google Scholar] [CrossRef]
- OIE—World Organisation for Animal Health. Mixomatosis. 2020. Available online: https://www.oie.int/es/sanidad-animal-en-el-mundo/enfermedades-de-los-animales/mixomatosis (accessed on 18 November 2020).
- Hemelaar, J. The origin and diversity of the HIV-1 pandemic. Trends Mol. Med. 2012, 18, 182–192. [Google Scholar] [CrossRef] [PubMed]
- Byrne, M.P.; O’Gorman, P.A. Understanding Decreases in Land Relative Humidity with Global Warming: Conceptual Model and GCM Simulations. J. Clim. 2016, 29, 9045–9061. [Google Scholar] [CrossRef]
- MITECO. Cambio Climático. Available online: https://www.miteco.gob.es/es/cambio-climatico/temas/default.aspx (accessed on 14 April 2020).
- Gibson, J.H. UVB Radiation: Definition and Characteristics. 2020. Available online: http://uvb.nrel.colostate.edu/UVB/publications/uvb_primer.pdf (accessed on 24 May 2021).
- Manojkumar, N.; Srimuruganandam, B. Assessment of gaseous emissions and radiative forcing in Indian forest fires. Int. J. Environ. Stud. 2019, 76, 541–557. [Google Scholar] [CrossRef]
- Littell, J.S.; Peterson, D.L.; Riley, K.L.; Liu, Y.; Luce, C. A review of the relationships between drought and forest fire in the United States. Glob. Chang. Biol. 2016, 22, 2353–2369. [Google Scholar] [CrossRef] [PubMed]
- Álvarez, A.S.M.; De Huancayo, U.C.; Arredondo, R.E.; Yuli-Posadas, R.A. Determinación de la presencia de partículas (PM10) en Perú producidas por quema de biomasa con ayuda de modelos numéricos. Rev. Int. Contam. Ambient. 2017, 33, 99–108. [Google Scholar] [CrossRef]
- McMichael, A.J.; World Health Organization (Eds.) Climate Change and Human Health: Risks and Responses; World Health Organization: Geneva, Switzerland, 2003. [Google Scholar]
- Girardi, R.; Pinheiro, A.; Garbossa, L.H.P.; Torres, É. Water quality change of rivers during rainy events in a watershed with different land uses in Southern Brazil. Rev. Bras. Recur. Hídr. 2016, 21, 514–524. [Google Scholar] [CrossRef]
- García Aróstegui, J.L.; Cruz Sanjulián, J.J.; Hidalgo Estévez, M.C. Control de la intrusión marina y modelización del acuífero de vélez (Málaga, España). In Tecnología de la Intrusión de Agua de Mar en Acuíferos Costeros: Países Mediterráneos; IGME: Madrid, Spain, 2003; pp. 261–270. [Google Scholar]
- Marín Cots, P.; Sánchez Teba, E.M.; Molina Conde, I. Aproximación al Escenario de Cambio Climático en la Ciudad de Málaga. Composición y Estudio de Series Estadísticas. 2008. Available online: http://static.omau-malaga.com/omau/subidas/archivos/8/5/arc_858.pdf (accessed on 24 May 2021).
- McInnes, K.L.; Erwin, T.A.; Bathols, J.M. Global Climate Model projected changes in 10 m wind speed and direction due to anthropogenic climate change. Atmos. Sci. Lett. 2011, 12, 325–333. [Google Scholar] [CrossRef]
- Hallegatte, S.; Green, C.; Nicholls, R.J.; Corfee-Morlot, J. Future flood losses in major coastal cities. Nat. Clim. Chang. 2013, 3, 802–806. [Google Scholar] [CrossRef]
- Méndez Jiménez, M. Análisis Preliminar de la Vulnerabilidad de la Costa de Andalucía a la Potencial Subida del Nivel del Mar Asociada al Cambio Climático. 2011. Available online: https://www.juntadeandalucia.es/medioambiente/portal_web/web/temas_ambientales/clima/actuaciones_cambio_climatico/adaptacion/vulnerabilidad_impactos_medidas/vulnerabilidad_costas.pdf (accessed on 24 May 2021).
- Jiang, C.; Shaw, K.S.; Upperman, C.R.; Blythe, D.; Mitchell, C.; Murtugudde, R.; Sapkota, A.R.; Sapkota, A. Climate change, extreme events and increased risk of salmonellosis in Maryland, USA: Evidence for coastal vulnerability. Environ. Int. 2015, 83, 58–62. [Google Scholar] [CrossRef]
- Ceballos, G.; Ehrlich, P.R.; Barnosky, A.D.; García, A.; Pringle, R.M.; Palmer, T.M. Accelerated modern human–induced species losses: Entering the sixth mass extinction. Sci. Adv. 2015, 1, e1400253. [Google Scholar] [CrossRef]
- Karp, D.S.; Moeller, H.V.; Frishkoff, L.O. Nonrandom extinction patterns can modulate pest control service decline. Ecol. Appl. 2013, 23, 840–849. [Google Scholar] [CrossRef] [PubMed]
- Shaw, A. Habitat Destruction. National Geographic Kids. 2018. Available online: https://kids.nationalgeographic.com/explore/science/habitat-destruction/ (accessed on 14 April 2020).
- Capdevila-Argüelles, L.; Zilletti, B.; Suárez Álvarez, V.Á. Plataforma Nacional de Adaptación al Cambio Climático—Cambio Climático y Especies Exóticas Invasoras en España: Diagnóstico Preliminar y Bases de Conocimiento Sobre Impactos y Vulnerabilidad (2011). 2011. Available online: https://www.adaptecca.es/recursos/buscador/cambio-climatico-y-especies-exoticas-invasoras-en-espana-diagnostico-preliminar-y (accessed on 14 April 2020).
- El Zowalaty, M.E.; Järhult, J.D. From SARS to COVID-19: A previously unknown SARS- related coronavirus (SARS-CoV-2) of pandemic potential infecting humans—Call for a One Health approach. One Health 2020, 9, 100124. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.-J.; Lin, C.Y.; Wu, Y.-T.; Wu, P.-C.; Lung, S.-C.; Su, H.-J. Effects of Extreme Precipitation to the Distribution of Infectious Diseases in Taiwan, 1994–2008. PLoS ONE 2012, 7, e34651. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Hong, K.-H.; Hwandon, J.; Young-Jae, P.; Moojong, P.; Young, S. Effect of Precipitation on Air Pollutant Concentration in Seoul, Korea. Asian J. Atmos. Environ. 2014, 8, 202–211. [Google Scholar] [CrossRef]
- Hong, J.; Zhong, T.; Li, H.; Xu, J.; Ye, X.; Mu, Z.; Lu, Y.; Mashaghi, A.; Zhou, Y.; Tan, M.; et al. Ambient air pollution, weather changes and outpatient visits for allergic conjunctivitis: A retrospective registry study. Sci. Rep. 2016, 6, 23858. [Google Scholar] [CrossRef] [PubMed]
- Stach, A.; García-Mozo, H.; Prieto-Baena, J.C.; Czarnecka-Operacz, M.; Jenerowicz, D.; Silny, W.; Galán, C. Prevalence of Artemisia species pollinosis in western Poland: Impact of climate change on aerobiological trends, 1995–2004. J. Investig. Allergol. Clin. Immunol. 2007, 17, 39–47. [Google Scholar]
- Jung, S.J.; Mehta, J.S.; Tong, L. Effects of environment pollution on the ocular surface. Ocul. Surf. 2018, 16, 198–205. [Google Scholar] [CrossRef]
- Jaggernath, J.; Haslam, D.; Naidoo, K.S. Climate change: Impact of increased ultraviolet radiation and water changes on eye health. Health 2013, 05, 921–930. [Google Scholar] [CrossRef]
- Zhong, J.-Y.; Lee, Y.-C.; Hsieh, C.-J.; Tseng, C.-C.; Yiin, L.-M. Association between Dry Eye Disease, Air Pollution and Weather Changes in Taiwan. Int. J. Environ. Res. Public Health 2018, 15, 2269. [Google Scholar] [CrossRef]
- Copeland, R.; Afshari, N.A. Principles and Practice of Cornea, 1st ed.; The Eye Center and The Eye Foundation for Research in Ophthalmology. Jaypee Brothers Medical Publishers: New Delhi, India, 2013; Available online: https://www.jaypeedigital.com/book/9789350901724/chapter/ch46 (accessed on 29 April 2020).
- Miller, J.K.; Laycock, K.A.; Nash, M.M.; Pepose, J.S. Corneal Langerhans cell dynamics after herpes simplex virus reactivation. Investig. Ophthalmol. Vis. Sci. 1993, 34, 2282–2290. [Google Scholar]
- Wegman, D.H. Epidemic Keratoconjunctivitis. Am. J. Public Health Nations Health 1970, 60, 1230–1237. [Google Scholar] [CrossRef]
- Parra-Rodríguez, D.S.; García-Carmona, K.P.; Vázquez-Maya, L.; Bonifaz, A. Incidencia de úlceras corneales microbianas en el Servicio de Oftalmología del Hospital General de México Dr. Eduardo Liceaga. Rev. Mex. Oftalmol. 2016, 90, 209–214. [Google Scholar] [CrossRef]
- Hussein, A.S.; El Mofty, H.; Hassanien, M. Climate change and predicted trend of fungal keratitis in Egypt. East. Mediterr. Health J. 2011, 17, 468–473. [Google Scholar] [CrossRef]
- Chidambaram, J.D.; Prajna, N.V.; Srikanthi, P.; Lanjewar, S.; Shah, M.; Elakkiya, S.; Lalitha, P.; Burton, M.J. Epidemiology, risk factors, and clinical outcomes in severe microbial keratitis in South India. Ophthalmic Epidemiol. 2018, 25, 297–305. [Google Scholar] [CrossRef]
- Psoter, K.J.; De Roos, A.; Wakefield, J.; Mayer, J.; Rosenfeld, M.; Rolain, J.-M. Season is associated with Pseudomonas aeruginosa acquisition in young children with cystic fibrosis. Clin. Microbiol. Infect. 2013, 19, E483–E489. [Google Scholar] [CrossRef]
- Sanda, J. The influences of climate changes in acute glaucoma. Abstract. In Proceedings of the International Multidisciplinary Scientific GeoConference & EXPO SGEM2014, Albena, Bulgaria, 17–26 June 2014; Available online: https://www.sgem.org/SGEMLIB/spip.php?article5110 (accessed on 10 April 2021). [CrossRef]
- Stein, J.D.; Pasquale, L.R.; Talwar, N.; Kim, D.S.; Reed, D.; Nan, B.; Kang, J.H.; Wiggs, J.L.; Richards, J.E. Geographic and Climatic Factors Associated with Exfoliation Syndrome. Arch. Ophthalmol. 2011, 129, 1053–1060. [Google Scholar] [CrossRef]
- Meerburg, B.G.; Kijlstra, A. Changing climate—Changing pathogens: Toxoplasma gondii in North-Western Europe. Parasitol. Res. 2009, 105, 17–24. [Google Scholar] [CrossRef]
- Rothova, A. Ocular involvement in toxoplasmosis. Br. J. Ophthalmol. 1993, 77, 371–377. [Google Scholar] [CrossRef]
- Jones, B.A.; Grace, D.; Kock, R.; Alonso, S.; Rushton, J.; Said, M.; McKeever, D.; Mutua, F.; Young, J.; McDermott, J.; et al. Zoonosis emergence linked to agricultural intensification and environmental change. Proc. Natl. Acad. Sci. USA 2013, 110, 8399–8404. [Google Scholar] [CrossRef]
- Jones, N.P. The Manchester Uveitis Clinic: The First 3000 Patients—Epidemiology and Casemix. Ocul. Immunol. Inflamm. 2013, 23, 118–126. [Google Scholar] [CrossRef]
- Rathinam, S.R.; Namperumalsamy, P. Global variation and pattern changes in epidemiology of uveitis. Indian J. Ophthalmol. 2007, 55, 173–183. [Google Scholar] [CrossRef]
- Albert, D.M.; Raven, M.L. Ocular Tuberculosis. Microbiol. Spectr. 2016, 4. [Google Scholar] [CrossRef]
- Pope, J.E.; Krizova, A.; Garg, A.X.; Thiessen-Philbrook, H.; Ouimet, J.M. Campylobacter Reactive Arthritis: A Systematic Review. Semin. Arthritis Rheum. 2007, 37, 48–55. [Google Scholar] [CrossRef]
- Ntumba Kayembe, J.-M.; Ntumba Kayembe, H.-C. Pneumonia: A Challenging Health Concern with the Climate Change; IntechOpen: London, UK, 2017. [Google Scholar]
- Chee, S.-P.; Khairallah, M. Emerging Infectious Uveitis; Springer: Berlin/Heidelberg, Germany, 2017. [Google Scholar]
- Pleyer, U.; Chee, S.-P. Current aspects on the management of viral uveitis in immunocompetent individuals. Clin. Ophthalmol. 2015, 9, 1017–1028. [Google Scholar] [CrossRef]
- Gorodner, J.O.; Merino, D.E. Fiebre por virus del Nilo Occidental¿ Otra patología emergente relacionada con el cambio climático? Rev. Asoc. Médica Argent. 2012, 125, 9–12. [Google Scholar]
- Brownstein, J.S.; Holford, T.R.; Fish, D. Effect of Climate Change on Lyme Disease Risk in North America. EcoHealth 2005, 2, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Bednar, T.; What Will Climate Change Mean for Lyme Disease? Case Studies. Geology and Human Health. 2012. Available online: https://serc.carleton.edu/NAGTWorkshops/health/case_studies/lyme_disease.html (accessed on 30 April 2020).
- Duvall-Young, J. Emergency, Acute and Rapid Access Ophthalmology: Practical, Clinical and Managerial Aspects; Springer: Berlin/Heidelberg, Germany, 2018. [Google Scholar]
- Martinez-Berriotxoa, A.; Fonollosa, A.; Artaraz, J. Aproximación diagnóstica a las uveítis. Rev. Clínica Española 2012, 212, 442–452. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Rueda, A. Retinitis por Rickettsia conorii, una infección emergente en el sureste de la península ibérica. Arch. Sociedad Española Oftalmol. 2020, 95, 507–511. [Google Scholar] [CrossRef]
- Observatorio de Salud y Cambio Climático. Fiebre Botonosa Mediterránea. 2021. Available online: http://www.oscc.gob.es/es/general/salud_cambio_climatico/fiebre_botonosa_mediterranea_es.htm (accessed on 19 May 2021).
- Red Nacional de Vigilancia Epidemiológica-ISCIII (Instituto de Salud Carlos III). Resultados de la Vigilancia Epidemiológica de las enfermedades transmisibles. Informe Anual. Años 2017–2018. Ministerio de Ciencia e Innovación. 2020. Available online: https://www.isciii.es/QueHacemos/Servicios/VigilanciaSaludPublicaRENAVE/EnfermedadesTransmisibles/Documents/INFORMES/INFORMES%20RENAVE/RENAVE_Informe_anual__2017-2018.pdf (accessed on 27 March 2021).
- Song, Y.-J.; Cheong, H.-K.; Ki, M.; Shin, J.-Y.; Hwang, S.-S.; Park, M.; Ki, M.; Lim, J. The Epidemiological Influence of Climatic Factors on Shigellosis Incidence Rates in Korea. Int. J. Environ. Res. Public Health 2018, 15, 2209. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Hoang, T.T.H.; Pham-Duc, P.; Lee, M.; Grace, D.; Hung, N.-V.; Thuc, V.M.; Nguyen-Viet, H. Seasonal and geographical distribution of bacillary dysentery (shigellosis) and associated climate risk factors in Kon Tum Province in Vietnam from 1999 to 2013. Infect. Dis. Poverty 2017, 6, 113. [Google Scholar] [CrossRef] [PubMed]
- Red Nacional de Vigilancia Epidemiológica-ISCIII (Instituto de Salud Carlos III). Resultados de la Vigilancia Epidemiológica de las enfermedades transmisibles. Informe Anual. Año 2016. Ministerio de Ciencia e Innovación. 2020. Available online: http://gesdoc.isciii.es/gesdoccontroller?action=download&id=25/01/2019-d8ee271b6f (accessed on 18 February 2021).
- Cheeti, A.; Chakraborty, R.K.; Ramphul, K. Reactive Arthritis (Reiter Syndrome); StatPearls Publishing: Petersburg, FL, USA, 2021. Available online: http://www.ncbi.nlm.nih.gov/books/NBK499831/ (accessed on 15 June 2020).
- Akil, L.; Ahmad, H.A.; Reddy, R.S. Effects of Climate Change on Salmonella Infections. Foodborne Pathog. Dis. 2014, 11, 974–980. [Google Scholar] [CrossRef]
- Karlson, E.W.; Deane, K. Environmental and Gene-Environment Interactions and Risk of Rheumatoid Arthritis. Rheum. Dis. Clin. N. Am. 2012, 38, 405–426. [Google Scholar] [CrossRef]
- Zlatanović, G.; Veselinović, D.; Cekic, S.; Zivković, M.; Đorđević-Jocić, J.; Zlatanović, M. Ocular manifestation of rheumatoid arthritis-different forms and frequency. Bosn. J. Basic Med. Sci. 2010, 10, 323–327. [Google Scholar] [CrossRef] [PubMed]
- Gupta, N.; Agarwal, A. Management of Uveitis in Spondyloarthropathy: Current Trends. Perm. J. 2018, 22. [Google Scholar] [CrossRef]
- Leli, I.; Salimbene, I.; Varone, F.; Fuso, L.; Valente, S. Husband and wife with sarcoidosis: Possible environmental factors involved. Multidiscip. Respir. Med. 2013, 8, 5. [Google Scholar] [CrossRef]
- Ungprasert, P.; Crowson, C.S.; Matteson, E.L. Seasonal variation in incidence of sarcoidosis: A population-based study, 1976–2013. Thorax 2016, 71, 1164–1166. [Google Scholar] [CrossRef]
- Ortiz-Perez, S.; Martínez-Lapiscina, E.H.; Gabilondo, I.; Fraga-Pumar, E.; Martínez-Heras, E.; Saiz, A.; Sanchez-Dalmau, B.F.; Villoslada, P. Retinal periphlebitis is associated with multiple sclerosis severity. Neurology 2013, 81, 877–881. [Google Scholar] [CrossRef]
- Kubaisi, B.; Kopplin, L.; Agarwal, A.; Rosenbaum, J.T.; Foster, C.S.; Nguyen, Q.D. Crohn’s Disease-Associated Uveitis. Uveitis Atlas 2019, 2020, 147–152. [Google Scholar] [CrossRef]
- Eglin, R. Detection of RNA complementary to herpes-simplex virus in mononuclear cells from patients with Behçet’s Syndrome and Recurrent Oral Ulcers. Lancet 1982, 320, 1356–1361. [Google Scholar] [CrossRef]
- Zouboulis, C.C.; May, T. Pathogenesis of Adamantiades-Behçet’s disease. Med. Microbiol. Immunol. 2003, 192, 149–155. [Google Scholar] [CrossRef]
- Watanabe, H.; Yashiro, M.; Asano, T.; Sato, S.; Takahashi, A.; Katakura, K.; Kobayashi, H.; Ohira, H. A case of Behçet’s disease and systemic sclerosis developing after an earthquake disaster. Fukushima J. Med. Sci. 2015, 61, 86–90. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ness, T.; Bley, T.A.; Schmidt, W.A.; Lamprecht, P. The Diagnosis and Treatment of Giant Cell Arteritis. Dtsch. Aerzteblatt Online 2013, 110, 376–386. [Google Scholar] [CrossRef] [PubMed]
- Nordborg, E. Giant cell arteritis: Epidemiological clues to its pathogenesis and an update on its treatment. Rheumatology 2003, 42, 413–421. [Google Scholar] [CrossRef]
- Lyons, H.S.; Quick, V.; Sinclair, A.J.; Nagaraju, S.; Mollan, S.P. A new era for giant cell arteritis. Eye 2019, 34, 1013–1026. [Google Scholar] [CrossRef] [PubMed]
- Vodopivec, I.; Rizzo, J.F. Ophthalmic manifestations of giant cell arteritis. Rheumatology 2018, 57, ii63–ii72. [Google Scholar] [CrossRef]
- Okazaki, T.; Shinagawa, S.; Mikage, H. Vasculitis syndrome-diagnosis and therapy. J. Gen. Fam. Med. 2017, 18, 72–78. [Google Scholar] [CrossRef]
- Abdgawad, M. History, Classification and Pathophysiology of Small Vessel Vasculitis. Updates Diagn. Treat. Vasc. 2013, 20. [Google Scholar] [CrossRef]
- Pagnoux, C.; Bienvenu, B.; Guillevin, L. The spectrum of granulomatous vasculitides. Futur. Rheumatol. 2006, 1, 729–750. [Google Scholar] [CrossRef]
- Thorne, J.E.; Jabs, D.A. Ocular Manifestations o Vasculitis. Rheum. Dis. Clin. N. Am. 2001, 27, 761–779. [Google Scholar] [CrossRef]
- Krause, I.; Shraga, I.; Molad, Y.; Guedj, D.; Weinberger, A. Seasons of the Year and Activity of SLE and Behçet’s Disease. Scand. J. Rheumatol. 1997, 26, 435–439. [Google Scholar] [CrossRef] [PubMed]
- Cansu, D.U.; Kasifoglu, T.; Korkmaz, C. Is there any relationship between season/weather and oral ulcer in Behçet’s disease? Eur. J. Rheumatol. 2014, 1, 89–91. [Google Scholar] [CrossRef] [PubMed]
- Kamen, D.L. Environmental Influences on Systemic Lupus Erythematosus Expression. Rheum. Dis. Clin. N. Am. 2014, 40, 401–412. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J. Ultraviolet B irradiation and cytomegalovirus infection synergize to induce the cell surface expression of 52-kD/Ro antigen. Clin. Exp. Immunol. 1996, 103, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Organización Nacional de Trastornos Raros (NORD). Dermatomyositis. 2018. Available online: https://rarediseases.org/rare-diseases/dermatomyositis/ (accessed on 1 May 2020).
- Orione, M.A.M.; Silva, C.A.; Sallum, A.M.E.; Campos, L.M.A.; Omori, C.H.; Braga, A.; Farhat, S.C.L. Risk Factors for Juvenile Dermatomyositis: Exposure to Tobacco and Air Pollutants During Pregnancy. Arthritis Rheum. 2014, 66, 1571–1575. [Google Scholar] [CrossRef] [PubMed]
- Pachman, L.M.; Khojah, A.M. Advances in Juvenile Dermatomyositis: Myositis Specific Antibodies Aid in Understanding Disease Heterogeneity. J. Pediatr. 2018, 195, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Calder, J.A.M.; Calder, D.N. Chikungunya Fever: A New Concern for the Western Hemisphere. Open Infect. Dis. J. 2015, 9, 13–19. [Google Scholar]
- Teoh, S.; Thean, L.; Koay, E. Cytomegalovirus in aetiology of Posner–Schlossman syndrome: Evidence from quantitative polymerase chain reaction. Eye 2004, 19, 1338–1340. [Google Scholar] [CrossRef] [PubMed]
- Willermain, F.; Van Laethem, Y.; Caspers, L. Global Variations and Changes in Patterns of Infectious Uveitis. Emerg. Infect. Uveitis 2017, 2020, 1–7. [Google Scholar] [CrossRef]
- Dion, M.; Hamelin, C. Enhanced reacivation of ultraviolet-irradiated human cytomegalovirus in normal host cells. Mutat. Res. Mol. Mech. Mutagen. 1987, 179, 49–53. [Google Scholar] [CrossRef]
- Moshirfar, M.; Murri, M.S.; Shah, T.J.; Skanchy, D.F.; Tuckfield, J.Q.; Ronquillo, Y.C.; Birdsong, O.C.; Hofstedt, D.; Hoopes, P.C. A Review of Corneal Endotheliitis and Endotheliopathy: Differential Diagnosis, Evaluation, and Treatment. Ophthalmol. Ther. 2019, 8, 195–213. [Google Scholar] [CrossRef]
- Donaldson, M.; Pulido, J.S.; Herman, D.C.; Diehl, N.; Hodge, D. Pars Planitis: A 20-Year Study of Incidence, Clinical Features, and Outcomes. Am. J. Ophthalmol. 2007, 144, 812–817.e2. [Google Scholar] [CrossRef] [PubMed]
- Ortega Madueño, I. Estudio Serológico del Herpesvirus Humano Tipo 6 en Pacientes con Esclerosis Múltiple. 2018. Available online: https://eprints.ucm.es/50311/1/T40714.pdf (accessed on 24 May 2021).
- Blanco Kelly, F. Esclerosis Múltiple: Estudio de la Asociación de la Genética con los Factores Ambientales, la Respuesta Inmune y el Tratamiento. Ph.D. Thesis, Departamento de Microbiología, Universidad Complutense de Madrid, Madrid, Spain, 2012; 220p. Available online: https://eprints.ucm.es/id/eprint/17158/1/T34047.pdf (accessed on 9 January 2021).
- Tang, P.H.; Chandramohan, A.; Do, D.V. Evolving Understanding and Treatment of Vogt-Koyanagi-Harada Syndrome. Retin. Physician 2019, 16, 40–45. Available online: https://www.retinalphysician.com/issues/2019/jan-feb-2019/evolving-understanding-and-treatment-of-vogt-koyan (accessed on 2 May 2020).
- Ng, J.Y.; Luk, F.O.; Lai, T.Y.; Pang, C.-P. Influence of molecular genetics in Vogt-Koyanagi-Harada disease. J. Ophthalmic Inflamm. Infect. 2014, 4, 20. [Google Scholar] [CrossRef] [PubMed]
- AlGahtani, H.; Alkhotani, A.; Shirah, B. Neurological Manifestations of Acute Posterior Multifocal Placoid Pigment Epitheliopathy. J. Clin. Neurol. 2016, 12, 460–467. [Google Scholar] [CrossRef]
- Goldhardt, R.; Patel, H.; Davis, J.L. Acute Posterior Multifocal Placoid Pigment Epitheliopathy Following Dengue Fever: A New Association for an Old Disease. Ocul. Immunol. Inflamm. 2016, 24, 610–614. [Google Scholar] [CrossRef] [PubMed]
- Rouse, S.; Jampol, L. Chapter 157—Acute posterior multifocal placoid pigment epitheliopathy, serpiginous choroiditis, and relentless placoid chorioretinitis. In Albert & Jakobiec’s Principles & Practice of Ophthalmology, 3rd ed.; Springer: Berlin/Heidelberg, Germany, 2008; Available online: https://doctorlib.info/ophthalmology/principles/36.html (accessed on 2 May 2020).
- Lin, H.-C.; Chen, C.-S.; Keller, J.J.; Ho, J.-D.; Lin, C.-C.; Hu, C.-C. Seasonality of Retinal Detachment Incidence and Its Associations with Climate: An 11-Year Nationwide Population-Based Study. Chronobiol. Int. 2011, 28, 942–948. [Google Scholar] [CrossRef]
- Thelen, U.; Gerding, H.; Clemens, S. Rhegmatogene Netzhautablösungen. Ophthalmologe 1997, 94, 638–641. [Google Scholar] [CrossRef]
- Bertelmann, T.; Cronauer, M.; Stoffelns, B.M.; Sekundo, W. Saisonale Variation des Auftretens rhegmatogener Netzhautablösungen zu Beginn des Jahrhunderts. Ophthalmologe 2011, 108, 1155–1163. [Google Scholar] [CrossRef]
- Prabhu, P.B.; Raju, K.V. Seasonal Variation in the Occurrence of Rhegmatogenous Retinal Detachment. Asia Pac. J. Ophthalmol. 2016, 5, 122–126. [Google Scholar] [CrossRef] [PubMed]
- Rahman, R.; Ikram, K.; Rosen, P.H.; Cortina-Borja, M.; Taylor, M.E. Do climatic variables influence the development of posterior vitreous detachment? Br. J. Ophthalmol. 2002, 86, 829. [Google Scholar] [CrossRef] [PubMed]
- Morris, B.; Imrie, F.; Armbrecht, A.-M.; Dhillon, B. Age-related macular degeneration and recent developments: New hope for old eyes? Postgrad. Med. J. 2007, 83, 301–307. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mauget-Faÿsse, M.; Kodjikian, L.; Quaranta, M.; Ben Ezra, D.; Trepsat, C.; Mion, F.; Mégraud, F. Helicobacter pylori in central serous chorioretinopathy and diffuse retinal epitheliopathy. Results of the first prospective pilot study. J. Français Ophtalmol. 2002, 25, 1021–1025. [Google Scholar]
- Roshani, M.; Davoodi, N.A.; Seyyedmajidi, M.; Zojaji, H.; Sherafat, S.J.; Hashemi, M.; Zali, M. Association of Helicobacter pylori with central serous chorioretinopathy in Iranian patients. Gastroenterol. Hepatol. Bed Bench 2014, 7, 63–67. [Google Scholar] [PubMed]
- Ahmed, N.; Tenguria, S.; Nandanwar, N. Helicobacter pylori—A seasoned pathogen by any other name. Gut Pathog. 2009, 1, 24. [Google Scholar] [CrossRef][Green Version]
- Aziz, R.K.; Khalifa, M.M.; Sharaf, R.R. Contaminated water as a source of Helicobacter pylori infection: A review. J. Adv. Res. 2015, 6, 539–547. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).