Population Kidney Health. A New Paradigm for Chronic Kidney Disease Management
Abstract
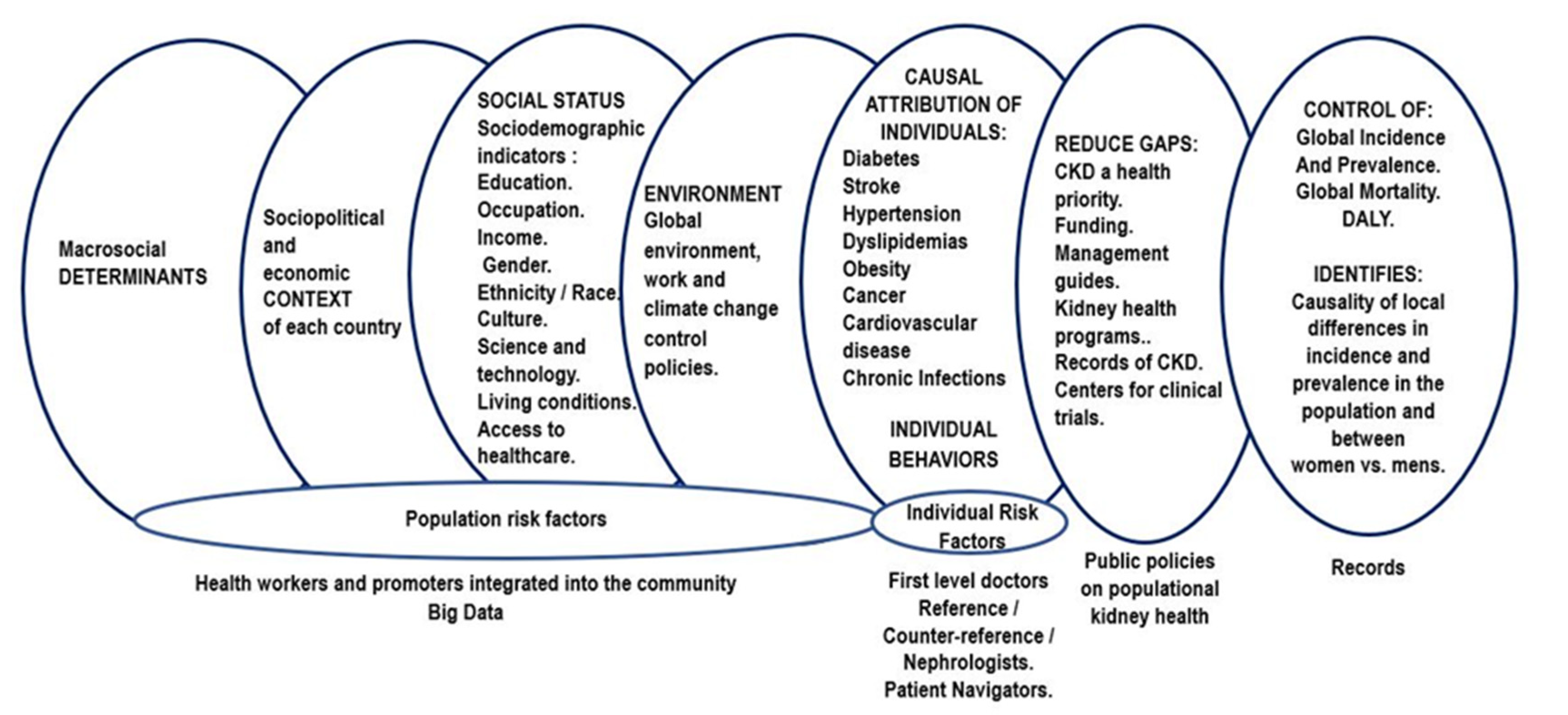
1. Introduction
- Adopting better governance for health and development;
- Promoting participation in policymaking and implementation;
- Further reorienting the health sector towards reducing health inequities;
- Strengthening global governance and collaboration;
- Monitoring progress and increasing accountability;
- Concreting a call for global action.
2. Materials and Methods
3. Results
3.1. Results
3.2. Discussion
3.3. Research Limitations
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Saadi, G.; El Nahid, M.S. Renal Disease Burden in Sample Countries from Five Continents. Int. J. Nephrol. Kidney Failure 2020, 6. [Google Scholar] [CrossRef]
- Bikbov, B.; Purcell, C.A.; Levey, A.S.; Smith, M.; Abdoli, A.; Abebe, M.; Adebayo, O.M.; Afarideh, M.; Agarwal, S.K.; Agudelo-Botero, M.; et al. Global, regional, and national burden of chronic kidney disease, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2020, 395, 709–773. [Google Scholar] [CrossRef]
- Robles-Osorio, M.; Sabatha, E. Disparidad social, factores de riesgo y enfermedad renal crónica. Nefrología 2016, 36, 465–582. [Google Scholar] [CrossRef]
- Crews, D.; Kuczmarski, M.; Miller, E.; Zonderman, A.; Evans, M.; Powe, N. Dietary Habits, Poverty, and Chronic Kidney Disease in an Urban Population. J. Ren. Nutr. 2015, 25, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Pugsley, D.; Norris, K.; Garcia-Garcia, G.; Agodoa, L. Global approaches for understanding the disproportionate burden of chronic kidney disease. Ethn. Dis. 2009, 19 (Suppl. 1), S1-1–S1-2. [Google Scholar]
- Hoy, W.; Ordunez, P. Epidemic of Chronic Kidney Disease in Agricultural Communities in Central America. Case Definitions, Methodological Basis and Approaches for Public Health Surveillance; Pan American Health Organization: Washington, DC, USA, 2017. [Google Scholar]
- Depine, S.A.; Aroca Martínez, G. Desafiando a la Inequidad de Latinoamérica. Estrategias Facilitadoras de “Control” de la Enfermedad Renal Crónica; Mejoras: Barranquilla, Colombia, 2018. [Google Scholar]
- Norton, J.M.; Moxey-Mims, M.M.; Eggers, P.W.; Narva, A.S.; Star, R.A.; Kimmel, P.L.; Rodgers, G.P. Social Determinants of Racial Disparities in CKD. J. Am. Soc. Nephrol. Sep. 2016, 27, 2576–2595. [Google Scholar] [CrossRef]
- Marinovich, S.; Bisigniano, L.; Hansen Krogh, D.; Celia, E.; Tagliafichi, V.; Rosa Diez, G.; Fayad, A. Registro Argentino de Diálisis Crónica SAN-INCUCAI 2018; Sociedad Argentina de Nefrología-INCUCAI: Buenos Aires, Argentina, 2018.
- Cuenta de Alto Costo. 2018. Available online: https://cuentadealtocosto.org/site/publicaciones/situacion-de-la-enfermedad-renal-cronica-la-hipertension-arterial-y-la-diabetes-mellitus-en-colombia-2017/ (accessed on 12 March 2021).
- Crews, D.C.; Bello, A.K.; Saadi, G. Manejo del riesgo de enfermedad. Carga, acceso y disparidades en la enfermedad renal. Rev. Colomb. Nefrol. 2019, 6, 74–83. [Google Scholar] [CrossRef]
- Hill, N.R.; Fatoba, S.T.; Oke, J.L.; Hirst, J.A.; O’Callaghan, C.A.; Lasserson, D.S.; Hobbs, F.D.R. Global Prevalence of Chronic Kidney Disease—A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0158765. [Google Scholar] [CrossRef] [PubMed]
- Eriksen, B.O.; Ingebretsen, O.C. The progression of chronic kidney disease: A 10-year population-based study of the effects of gender and age. Kidney Int. 2006, 69, 375–382. [Google Scholar] [CrossRef]
- Carrero, J.J. Gender differences in chronic kidney disease: Underpinnings and therapeutic implications. Kidney Blood Press. Res. 2010, 33, 383–392. [Google Scholar] [CrossRef]
- Global Burden of Disease Study 2013 Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2015, 386, 743–800. [Google Scholar] [CrossRef]
- United Nations. World Population Projected to Reach 9.8 Billion in 2050, and 11.2 Billion in 2100. 21 June 2017. Available online: https://www.un.org/development/desa/es/news/population/world-population-prospects-2017.html (accessed on 20 January 2021).
- Boutayeb, A. The double burden of communicable and non-communicable diseases in developing countries. Trans. R. Soc. Trop. Med. Hyg. 2006, 100, 191–199. [Google Scholar] [CrossRef]
- Murray, C.; Frenk, J. A framework for assessing the performance of health systems. Bull. World Health Organ. 2000, 78, 717–731. [Google Scholar] [PubMed]
- Martinez-Marín, B.; Haro-Acosta, M.; Quiñones Montelongo, K.; Margarita González-Ramos, A.; Guerrero, F.J.N.; Román-Matus, A. Costes de invalidez por insuficiencia renal crónica en la Delegación Baja California del Instituto Mexicano del Seguro Social. Med. Segur. Trabajo 2017, 63, 120–130. [Google Scholar]
- PAHO. Plan of Action for the Prevention and Control of Noncommunicable Diseases in the Americas 2013–2019; PAHO: Washington, DC, USA, 2014. [Google Scholar]
- Bello, A.; Levin, A.; Tonelli, M.; Okpechi, I.G.; Feehally, J.; Harris, D.; Jindal, K.; Salako, B.L.; Rateb, A.; Osman, M.A.; et al. Assessment of Global Kidney Health Care Status. JAMA 2017, 317, 1864–1881. [Google Scholar] [CrossRef] [PubMed]
- Depine, S. The role of government and competing priorities in minority populations and developing nations. Ethn. Dis. 2009, 19, 73–79. [Google Scholar]
- Aroca-Martínez, G.; Cadena-Bonfanti, A.; Ardila-Cárdenas, M.E.; Gonzalez, H.; Luna, M. World kidney day 2019: Determination of risk factors for kidney disease in indigenous Colombian adults. Rev. Col. Nefrol. 2020, 8–17. [Google Scholar]
- Dover, D.C.; Belon, A.P. The health equity measurement framework: A comprehensive model to measure social inequities in health. Int. J. Equity Health 2019, 18, 1–12. [Google Scholar] [CrossRef]
- WHO. Rio Political Declaration on Social Determinants of Health. In Proceedings of the World Conference on Social Determinants of Health, Rio de Janeiro, Brazil, 19–21 October 2011; p. 2012. [Google Scholar]
- Dahlgren, G.; Whitehead, M. European Strategies for Tackling Social Inequities in Health: Levelling up Part 2; WHO European Office for Investment for Health and Development: Copenhague, Denmark, 2007. [Google Scholar]
- Palomino Moral, P.A.; Grande Gascón, M.A.; Linares Abad, M. La salud y sus determinantes sociales. Desigualdades y exclusión en la sociedad del siglo XXI. Rev. Int. Sociol. 2014, 72, 71–91. [Google Scholar] [CrossRef]
- Harris, D.C.H.; Davies, S.J.; Finkelstein, F.O.; Jha, V.; Bello, A.K.; Brown, M.; Caskey, F.J.; Donner, J.; Liew, A.; Muller, E.; et al. Strategic plan for integrated care of patients with kidney failure. Kidney Int. 2020, 98, S117–S134. [Google Scholar] [CrossRef] [PubMed]
- Vivante, A.; Hildebrandt, F. Exploring the genetic basis of early-onset chronic kidney disease. Nat. Rev. Nephrol. 2016, 12, 136–146. [Google Scholar] [CrossRef] [PubMed]
- Wyatt, C.M.; Schlondorff, D. Precision medicine comes of age in nephrology: Identification of novel biomarkers and therapeutic targets for chronic kidney disease. Kidney Int. 2016, 89, 732–737. [Google Scholar] [CrossRef][Green Version]
- Precision medicine in nephrology. Nat. Rev. Nephrol. 2020, 16, 615. [CrossRef] [PubMed]
- Streja, E.; Sreja, D.A.; Soohoo, M.; Kleine, C.-E.; Hsiung, J.-T.; Park, C.; Moradi, H. Precision Medicine and Personalized Management of Lipoprotein and Lipid Disorders in Chronic and End-Stage Kidney Disease. Semin. Nephrol. 2018, 38, 369–382. [Google Scholar] [CrossRef] [PubMed]
- Shaibi, G.Q.; Kullo, I.J.; Singh, D.P.; Hernandez, V.; Sharp, R.R.; Cuellar, I.; de Filippis, E.; Levey, S.; Breitkopf, C.R.; Mandarino, L.J.; et al. Returning genomic results in a Federally Qualified Health Center: The intersection of precision medicine and social determinants of health. Genet. Med. 2020, 22, 1552–1559. [Google Scholar] [CrossRef] [PubMed]
- Freund, K.M. Patient Navigation: The Promise to Reduce Health Disparities. J. Gen. Intern. Med. 2011, 26, 110–112. [Google Scholar] [CrossRef]
- Valdés, G.; Zúñiga, C. A small contribution to mitigate the collision of transmissible and chronic diseases, exemplified by the management of hypertension during the COVID-19 pandemic. J. Hum. Hypertens. 2020, 35, 387–388. [Google Scholar] [CrossRef]
- Freeman, H. Patient navigation: A community centered approach to reducing cancer mortality. J. Cancer Educ. 2006, 21, S11–S14. [Google Scholar] [CrossRef]
- Freund, K.; Battaglia, T.; Calhoun, E.; Dudley, D.J.; Fiscella, K.; Paskett, E.; Raich, P.C.; Roetzheim, R.G.; The Patient Navigation Research Program Group. National Cancer Institute Patient Navigation Research Program: Methods, protocol, and measures. Cancer 2008, 113, 3391–3399. [Google Scholar] [CrossRef] [PubMed]
- Wagner, E.; Bennett, S.; Austin, B.; Greene, S.; Schaefer, J.; Vonkorff, M. Finding common ground: Patient-centeredness and evidence-based chronic illness care. J. Altern. Complement. Med. 2005, 11 (Suppl. 1), S7–S15. [Google Scholar] [CrossRef]
- Wagner, E.; Austin, B.; Von Korff, M. Organizing Care for Patients with Chronic Illness. Milbank Q. 1996, 74, 511–544. [Google Scholar] [CrossRef]
- Whitehead, M.; Dahlgren, G. Concepts and principles for tackling social inequities in health: Levelling up Part 1. WHO Collab. Cent. Policy Res. Soc. Determ. Health 2006, 2, 460–474. [Google Scholar]
- Kindig, D.; Stoddart, G. What is population health? Am. J. Public Health 2003, 93, 380–383. [Google Scholar] [CrossRef] [PubMed]
- Colmenares, C.C.; Eslava-Schmalbach, D. PoPulation health from the social determinants of health PersPective. Rev. Salud Bosque 2015, 3, 75–80. [Google Scholar] [CrossRef]
- Mújica, O.J.; Moreno, C.M. From words to action: Measuring health inequalities to “leave no one behind”. Rev. Panam Salud Publica 2019, 43, e12. [Google Scholar]
- Pencheon, D. The Good Indicators Guide: Understanding How to Use and Choose Indicators; NHS: London, UK, 2008.
- Albert-Ballestar, S.; García-Altés, A. Measuring health inequalities: A systematic review of widely used indicators and topics. Int. J. Equity Health 2021, 20, 1–15. [Google Scholar] [CrossRef]
- Zielinski, C. Infodemics and Infodemiology: A Short History, a Long Future; ScienceOpen, 12 May 2021; Available online: https://www.scienceopen.com/document?vid=1b982c1a-2e20-4ef5-b8e2-1d1bfda10dff (accessed on 3 June 2021).
- Bomback, A.; Derebail, V.; Shoham, D.; Anderson, C.A.; Steffen, L.M.; Rosamond, W.D.; Kshirsagar, A.V. Sugar-sweetened soda consumption, hyperuricemia, and kidney disease. Kidney Int. 2010, 77, 609–661. [Google Scholar] [CrossRef]
- Gutierrez, O.; Muntner, P.; Rizk, D.; McClellan, W.M.; Warnock, D.G.; Newby, P.K.; Judd, S.E. Dietary Patterns and Risk of Death and Progression to ESRD in Individuals With CKD: A Cohort Study. Am. J. Kidney Dis. 2014, 64, 204–213. [Google Scholar] [CrossRef]
- Crews, D.C.; Gutierrez, O.M.; Fedewa, S.A.; Luthi, J.C.; Shoham, D.; Judd, S.E.; Powe, N.R.; McClellan, W.M. Low income, community poverty and risk of end stage renal disease. BMC Nephrol. 2014, 15, 1–9. [Google Scholar] [CrossRef]
- Garrity, B.; Kramer, H.; Vellanki, K.; Leehey, D.; Brown, J.; Shoham, D.A. Time trends in the association of ESRD incidence with area-level poverty in the US population. Hemodial. Int. 2016, 20, 78–83. [Google Scholar] [CrossRef]
- Burgos Calderón, R.; Depine, S. Systematic approach for the management of chronic kidney disease: Moving beyond chronic kidney disease classification. Curr. Opin. Nephrol. Hypertens. 2010, 19, 208–213. [Google Scholar] [CrossRef]
- Vervaet, B.; Nast, C.; Jayasumana, C.; Schreurs, G.; Roels, F.; Herath, C.; Kojc, N.; Samaee, V.; Rodrigo, S.; Gowrishankar, S.; et al. Chronic interstitial nephritis in agricultural communities is a toxin-induced proximal tubular nephropathy. Kidney Int. 2020, 97, 350–369. [Google Scholar] [CrossRef]
- Lee, J.Y.; Park, J.T.; Joo, Y.S.; Lee, C.; Yun, H.-R.; Yoo, T.-H.; Kang, S.-W.; Choi, K.H.; Ahn, C.; Oh, K.-H.; et al. Association of Blood Pressure with the Progression of CKD: Findings From KNOW-CKD Study. Am. J. Kidney Dis. 2021. [Google Scholar] [CrossRef]
- Kearney, P.; Whelton, M.; Reynolds, K.; Muntner, P.; Whelton, P.; He, J. Global burden of hypertension: Analysis of worldwide data. Lancet 2005, 365, 217–223. [Google Scholar] [CrossRef]
- Mehta, R.L.; Cerdá, J.; Burdmann, E.A.; Tonelli, M.; García-García, G.; Jha, V.; Susantitaphong, P.; Rocco, M.; Vanholder, R.; Sever, M.S.; et al. International Society of Nephrology’s oby25 initiative for acute kidney injury (zero preventable deaths by 2025): A human rights case for nephrology. Lancet 2015, 385, 2616–2643. [Google Scholar] [CrossRef]
- Harris, D.; Dupuis, S.; Couser, W.; Feehally, J. Training nephrologists from developing countries: Does it have a positive impact? Kidney Int. 2012, 2, 275–278. [Google Scholar] [CrossRef]
- Challu, A.; Burgos Calderón, R.; Depine, S.; Feler, D.; Manzor, D. La Nefrología en Latinoamérica; SLANH: Buenos Aires, Argentina, 1999. [Google Scholar]
- Aroca Martínez, G. Propuesta de un Modelo de Gestión de Salud de la Nefritis Lúpica Basado en la Problemática Clínica y su Impacto Sociosanitario en la Región Caribe Colombiana; Mejoras: Barranquilla, Colombia, 2016. [Google Scholar]
- Aroca-Martínez, G.; Depine, S.; Consuegra-Machado, J.; González-Torres, H.; Árquez-Mendoza, M.; Estrada-García, E. Desarrollo y uso de una interfaz de programación de aplicaciones modificada de GoogleMaps© para la georreferenciación de pacientes con enfermedad glomerular. Nefrologia 2015, 35, 118–120. [Google Scholar] [PubMed]
- Burgos Calderón, R.; Depine, S. Sustainable and tenable renal health model: A Latin American proposal of classification, programming, and evaluation. Kidney Int. 2005, 68, S23–S30. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Iturbe, B. Progress in the prevention of chronic kidney disease in Latin America. Nat. Clin. Pract. Nephrol. 2008, 4, 233. [Google Scholar] [CrossRef][Green Version]
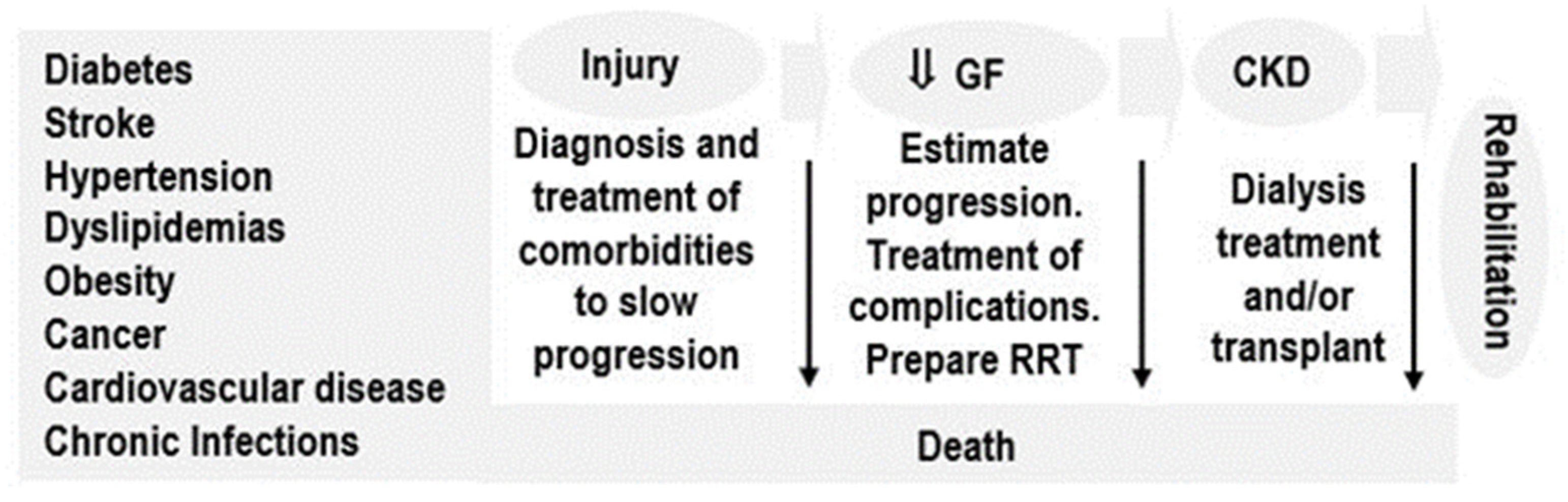
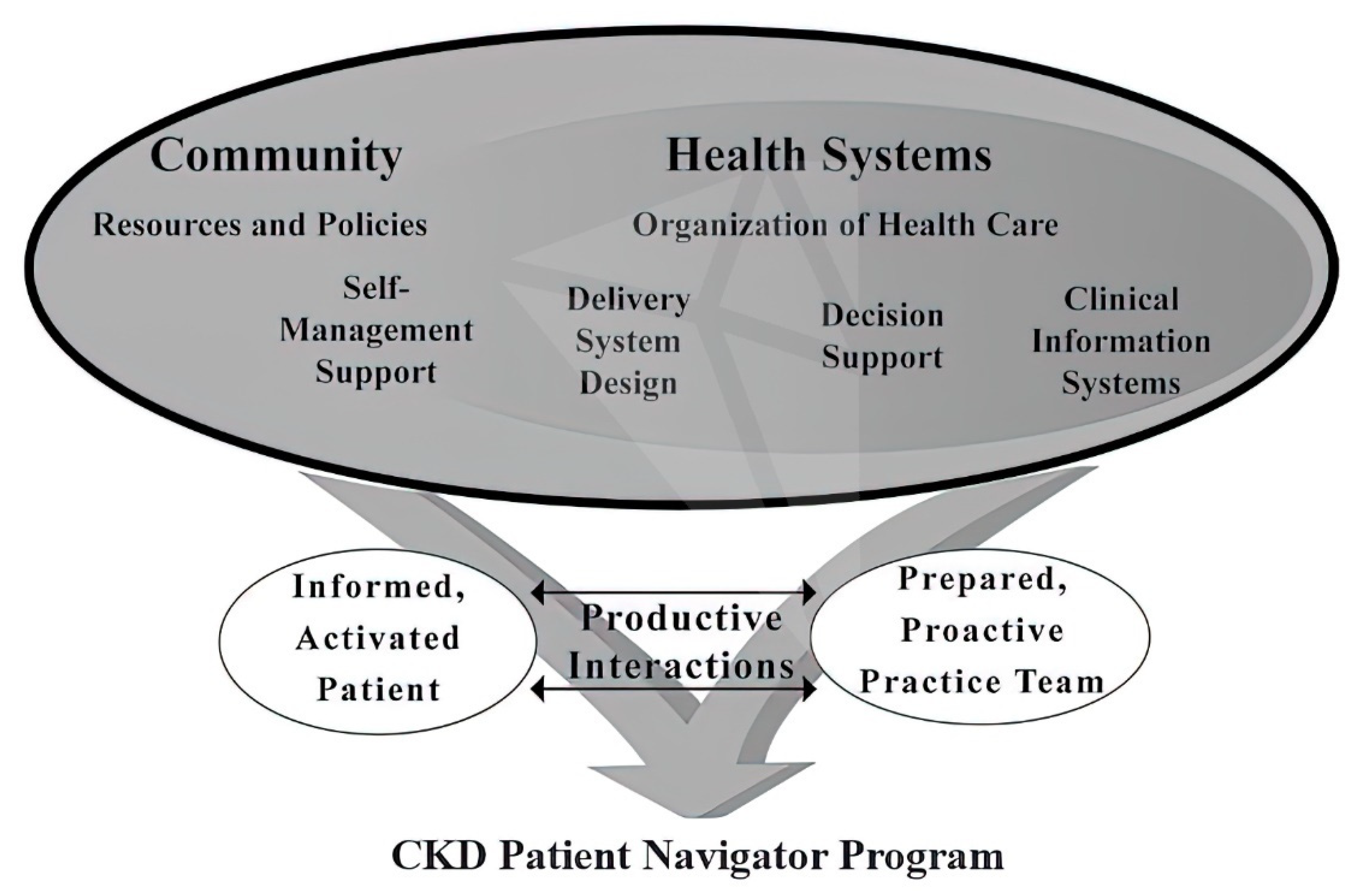
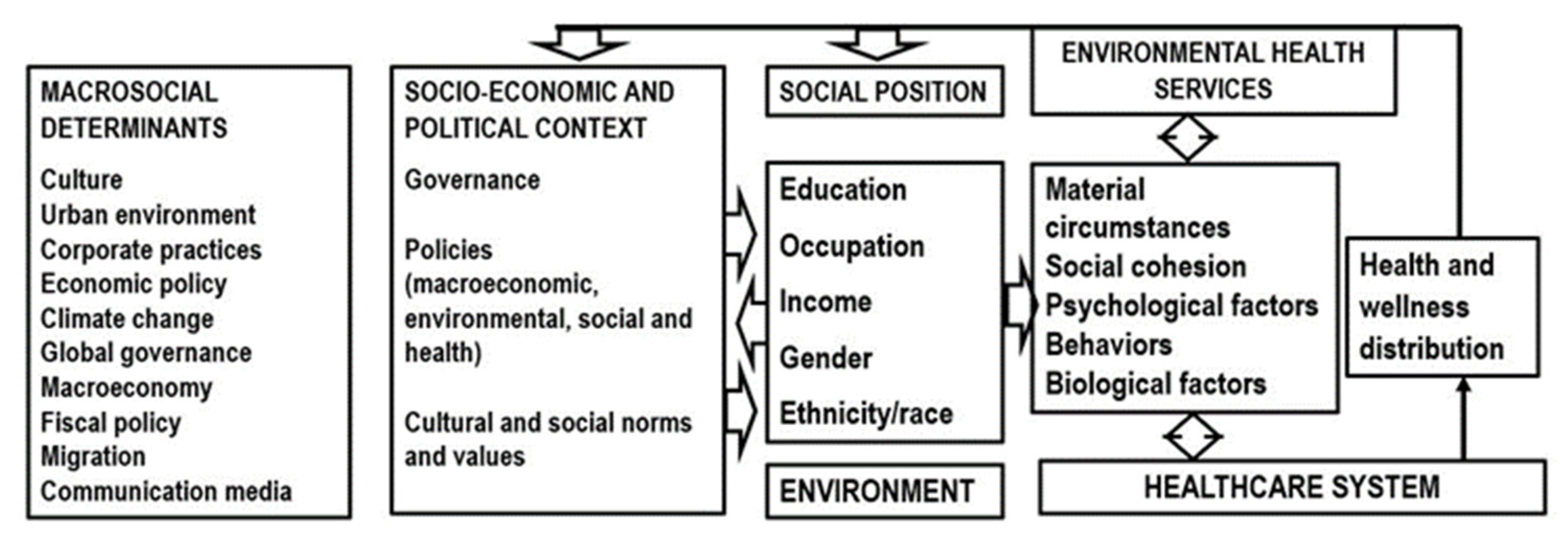
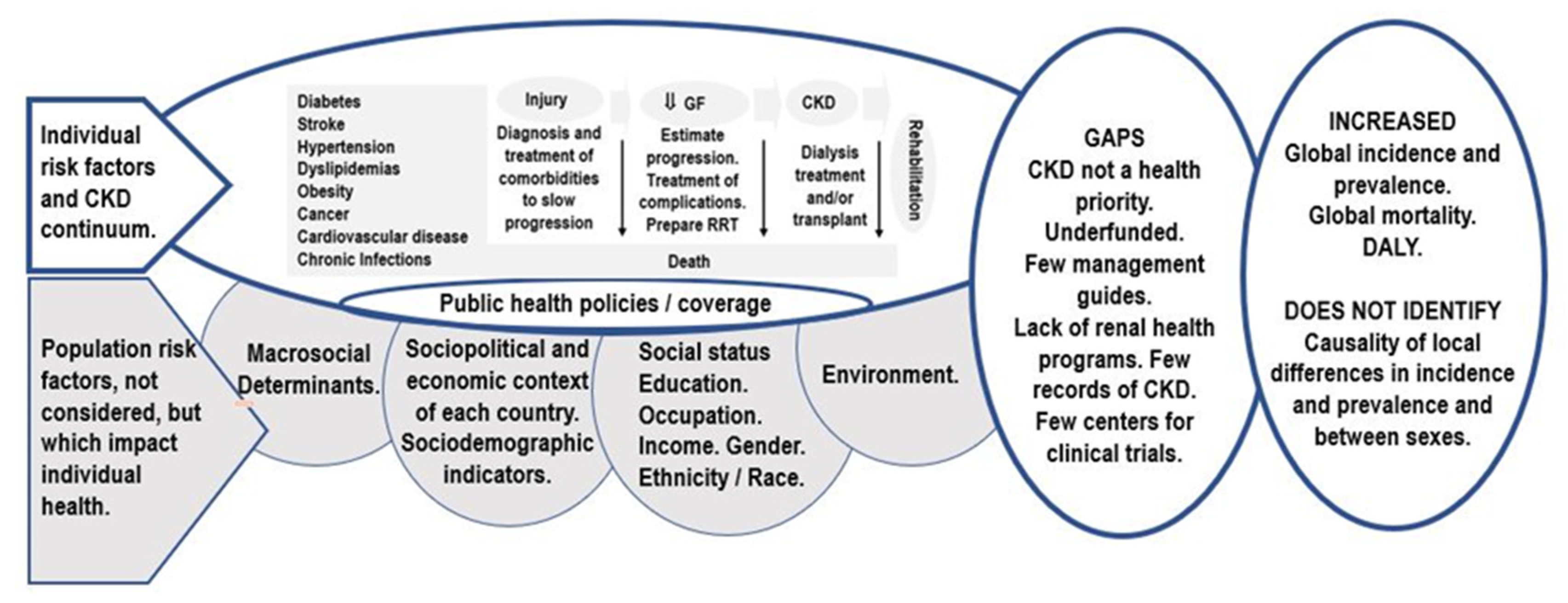
| CKD Care | Low-Income Countries (%) | Lower-Middle Income Countries (%) | Upper-Middle Income Countries (%) | High-Income Countries (%) |
|---|---|---|---|---|
| Government recognition of CKD as a health priority | 59 | 50 | 17 | 29 |
| Government provides funding for all aspects of CKD care | 13 | 21 | 40 | 53 |
| Availability of CKD management and reference guidelines (international, national or regional) | 46 | 73 | 83 | 97 |
| Current existence of CKD detection programs | 6 | 24 | 24 | 32 |
| Availability of dialysis records | 24 | 48 | 72 | 89 |
| Availability of academic centers for the management of renal clinical trials | 12 | 34 | 62 | 63 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burgos-Calderón, R.; Depine, S.Á.; Aroca-Martínez, G. Population Kidney Health. A New Paradigm for Chronic Kidney Disease Management. Int. J. Environ. Res. Public Health 2021, 18, 6786. https://doi.org/10.3390/ijerph18136786
Burgos-Calderón R, Depine SÁ, Aroca-Martínez G. Population Kidney Health. A New Paradigm for Chronic Kidney Disease Management. International Journal of Environmental Research and Public Health. 2021; 18(13):6786. https://doi.org/10.3390/ijerph18136786
Chicago/Turabian StyleBurgos-Calderón, Rafael, Santos Ángel Depine, and Gustavo Aroca-Martínez. 2021. "Population Kidney Health. A New Paradigm for Chronic Kidney Disease Management" International Journal of Environmental Research and Public Health 18, no. 13: 6786. https://doi.org/10.3390/ijerph18136786
APA StyleBurgos-Calderón, R., Depine, S. Á., & Aroca-Martínez, G. (2021). Population Kidney Health. A New Paradigm for Chronic Kidney Disease Management. International Journal of Environmental Research and Public Health, 18(13), 6786. https://doi.org/10.3390/ijerph18136786







