Post-COVID-19 Syndrome and the Potential Benefits of Exercise
Abstract
1. Introduction
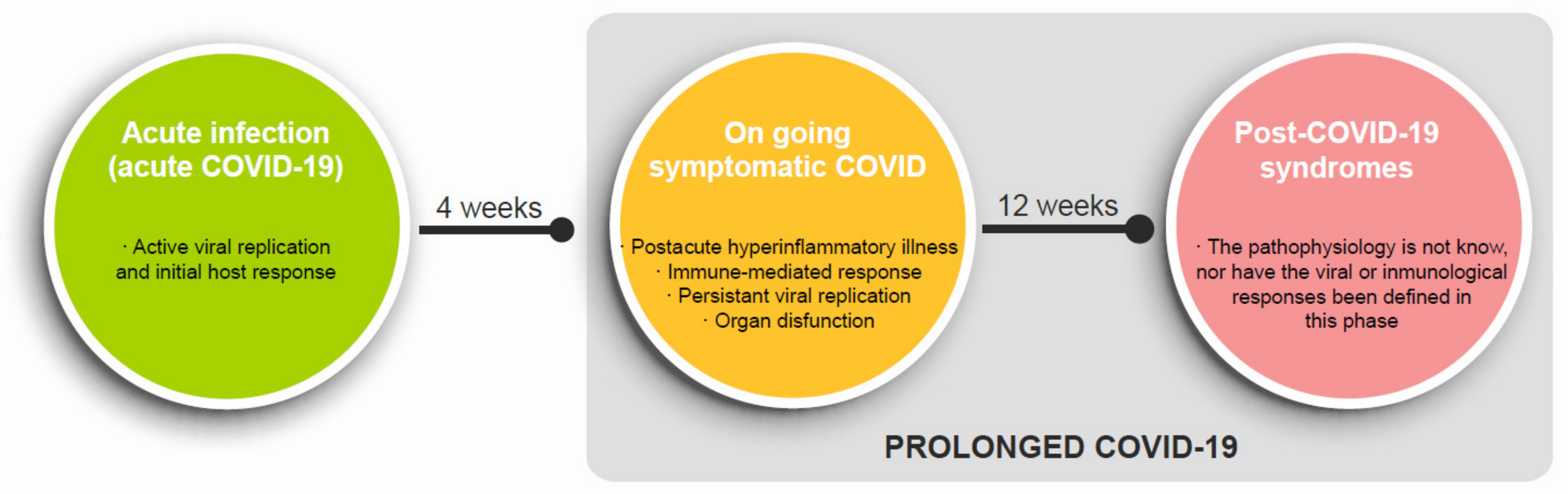
1.1. Definition of the Post-COVID-19 Syndrome
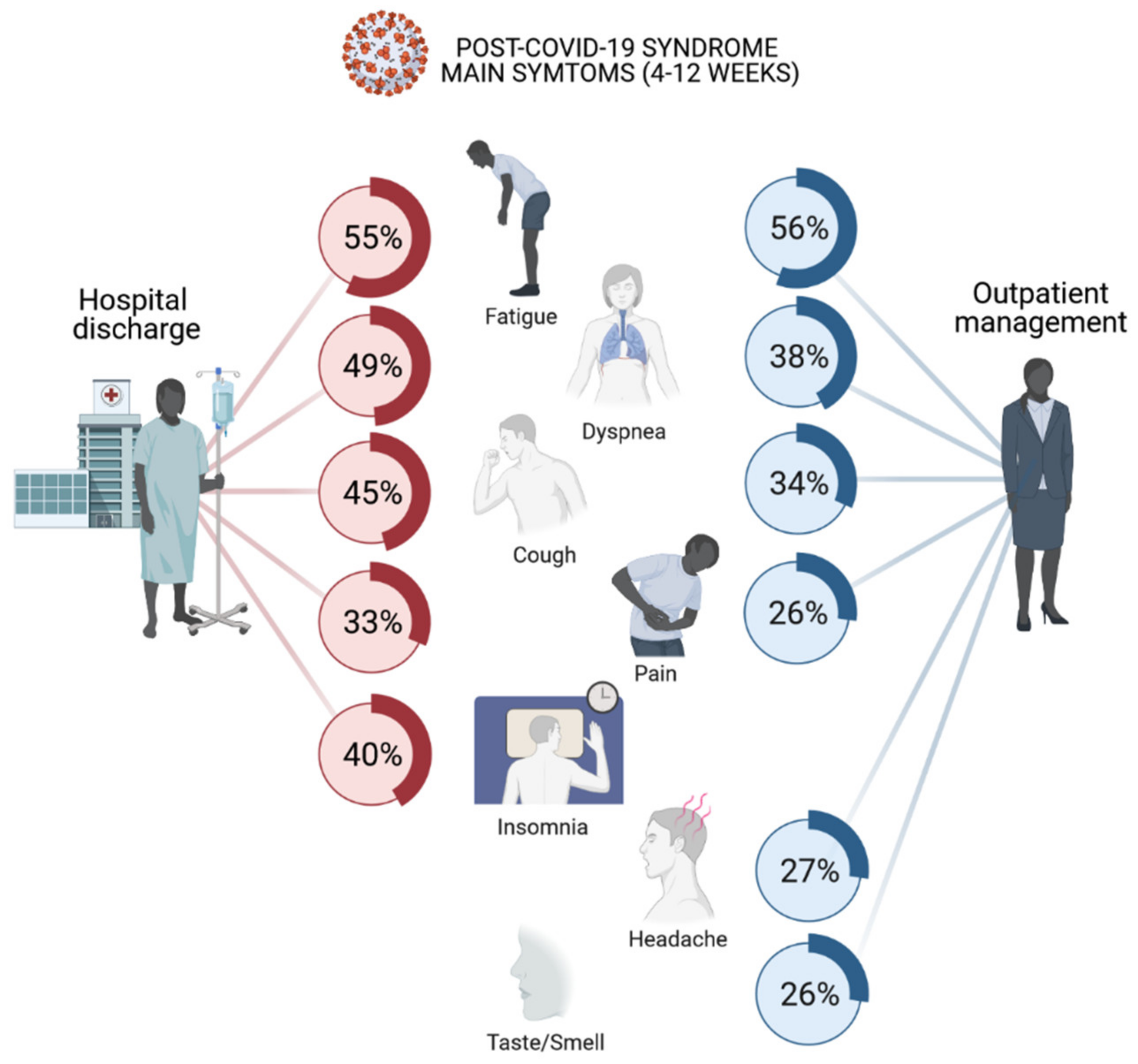
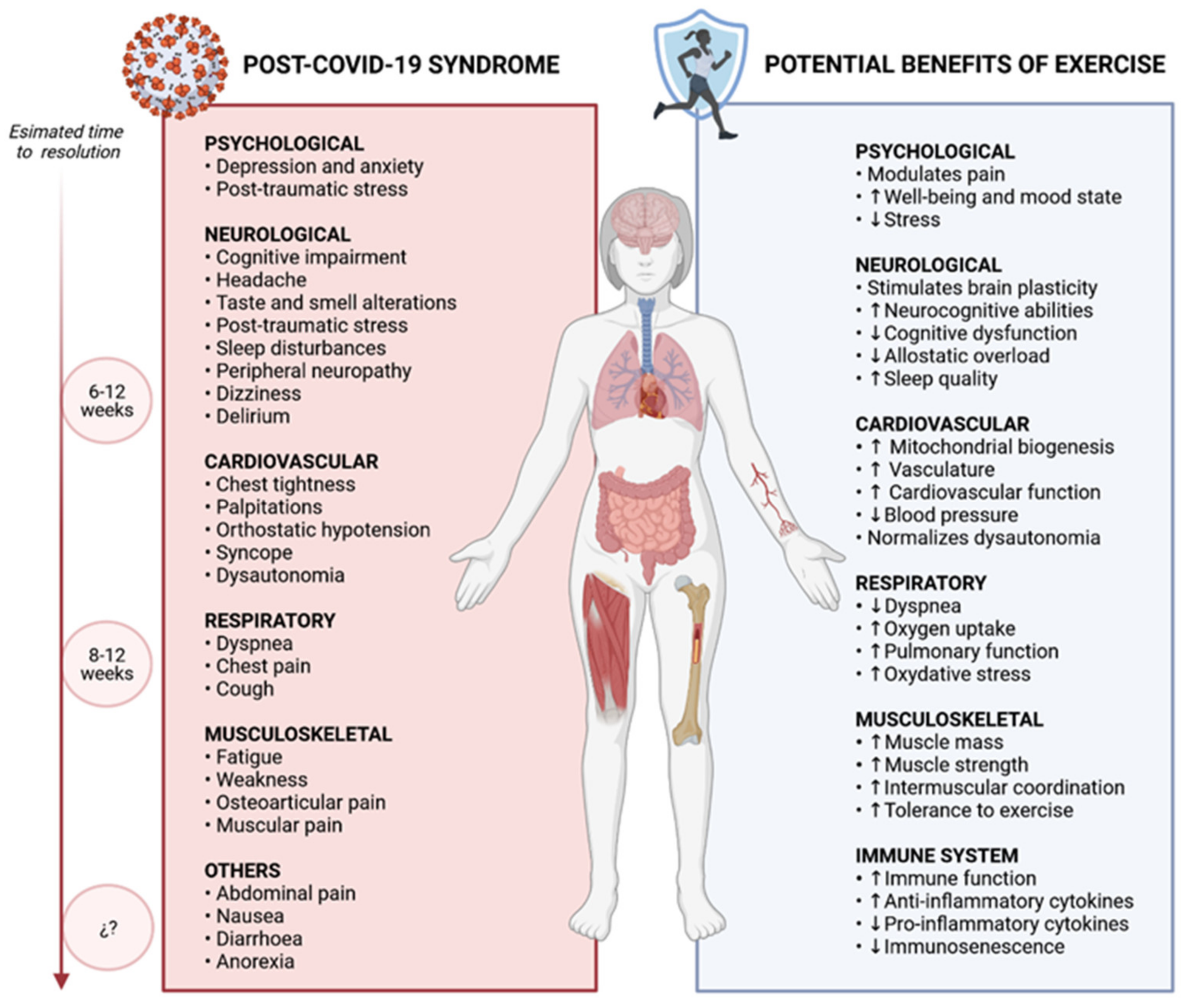
1.2. Clinical Manifestations of Post-COVID-19 Syndrome
- Women tend to be more affected than men [30,31,50,51,52]. The association between fatigue and myalgias is significantly more frequent in females than males. This phenomena has not been found in other symptoms cluster including low mood, anxiety, and sleep disturbance or in a cluster consisting of comprised memory impairment, attention deficit, and cognitive impairment [38].
- Persistent symptoms are more likely to appear with increasing age [31,52,53,58] and with severity of presentation [29,52,58]. After discharge, fatigue, dyspnoea and neuropsychological perturbances were significantly more frequent in ICU patients. Not all studies agree on a relationship with previous comorbidities.
- The symptoms are of a fluctuating nature and their appearance varies over time [59]. Patients’ perceptions were generally changeable both in time and form (relapsing-remitting). There is not a defined evolution pattern of the symptoms and patients usually respond to different stimuli (mainly physical or mental activity and stress).
- Recently, some studies has suggested that it is possible to identify the immunologic signatures of post-COVID-19 syndrome [60].
- Currently, there is no evidence supporting the notion that prolonged COVID-19 is equated to myalgic encephalomyelitis and/or chronic fatigue syndrome (ME/CFS). Both prolonged COVID-19, ME and CFS are heterogeneous and difficult to characterize, and while some patients with prolonged COVID-19 syndrome may meet diagnostic criteria for EM/CFS, there is a significant population with persistent fatigue that does not meet these criteria; therefore, further research is needed. Identification between the two syndromes could constitute a risk of avoiding other pathologies, complications or sequelae or erring in the management of the syndrome.
2. Physiopathology of the Post-COVID-19 Syndrome
2.1. Immune System
2.2. Cardiopulmonary Sequelae
2.3. Neurological and Biopsychosocial Disturbances
3. Treatment of the Post-COVID-19 Syndrome
4. The Potential Role of Exercise in Post-COVID-19 Syndrome
4.1. Exercise Is Beneficial for Immunological Health
4.2. Exercise Helps to Manage and Mitigate Physical Syndromes
4.3. Exercise Is an Effective Treatment for Pulmonary Complications
4.4. Exercise Improves Cardiovascular Health
4.5. Exercise Stimulates Brain Plasticity and Increases Psychological Well-Being
4.6. The role of Advanced Training Methods for Health
5. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- European Center For Disease Prevention and Control (ECDC). COVID-19 Situation Dashboard. Available online: https://qap.ecdc.europa.eu/public/extensions/COVID-19/COVID-19.html#global-overview-tab (accessed on 31 January 2021).
- Greenhalgh, T.; Knight, M.; A’Court, C.; Buxton, M.; Husain, L. Management of post-acute COVID-19 in primary care. BMJ 2020, 370, m3026. [Google Scholar] [CrossRef]
- Baig, A.M. Deleterious Outcomes in Long-Hauler COVID-19: The Effects of SARS-CoV-2 on the CNS in Chronic COVID Syndrome. ACS Chem. Neurosci. 2020, 11, 4017–4020. [Google Scholar] [CrossRef]
- Pfefferbaum, B.; North, C.S. Mental Health and the Covid-19 Pandemic. N. Engl. J. Med. 2020, 383, 510–512. [Google Scholar] [CrossRef]
- Fuller, J.A.; Hakim, A.; Victory, K.R.; Date, K.; Lynch, M.; Dahl, B.; Henao, O. Mitigation Policies and COVID-19–Associated Mortality—37 European Countries, 23 January–30 June 2020. MMWR. Morb. Mortal. Wkly. Rep. 2021, 70, 58–62. [Google Scholar] [CrossRef]
- Dong, Y.; Dai, T.; Wei, Y.; Zhang, L.; Zheng, M.; Zhou, F. A systematic review of SARS-CoV-2 vaccine candidates. Signal Transduct. Target. Ther. 2020, 5, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Ruegsegger, G.N.; Booth, F.W. Health Benefits of Exercise. Cold Spring Harb. Perspect. Med. 2017, 8, a029694. [Google Scholar] [CrossRef]
- Talar, K.; Hernández-Belmonte, A.; Vetrovsky, T.; Steffl, M.; Kałamacka, E.; Courel-Ibáñez, J. Benefits of Resistance Training in Early and Late Stages of Frailty and Sarcopenia: A Systematic Review and Meta-Analysis of Randomized Controlled Studies. J. Clin. Med. 2021, 10, 1630. [Google Scholar] [CrossRef]
- Heiston, E.M.; Eichner, N.Z.; Gilbertson, N.M.; Malin, S.K. Exercise improves adiposopathy, insulin sensitivity and metabolic syndrome severity independent of intensity. Exp. Physiol. 2020, 105, 632–640. [Google Scholar] [CrossRef] [PubMed]
- Fiuza-Luces, C.; Garatachea, N.; Berger, N.A.; Lucia, A. Exercise is the Real Polypill. Physiology 2013, 28, 330–358. [Google Scholar] [CrossRef]
- Metsios, G.S.; Moe, R.H.; Kitas, G.D. Exercise and inflammation. Best Pr. Res. Clin. Rheumatol. 2020, 34, 101504. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, P.L.; Castillo-García, A.; Morales, J.S.; Izquierdo, M.; Serra-Rexach, J.A.; Santos-Lozano, A.; Lucia, A. Physical Exercise in the Oldest Old. Compr. Physiol. 2019, 9, 1281–1304. [Google Scholar] [CrossRef]
- Posadzki, P.; Pieper, D.; Bajpai, R.; Makaruk, H.; Könsgen, N.; Neuhaus, A.L.; Semwal, M. Exercise/physical activity and health outcomes: An overview of Cochrane systematic reviews. BMC Public Health 2020, 20, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Sallis, R.; Young, D.R.; Tartof, S.Y.; Sallis, J.F.; Sall, J.; Li, Q.; Smith, G.N.; Cohen, D.A. Physical inactivity is associated with a higher risk for severe COVID-19 outcomes: A study in 48 440 adult patients. Br. J. Sports Med. 2021. [Google Scholar] [CrossRef] [PubMed]
- Brawner, C.A.; Ehrman, J.K.; Bole, S.; Kerrigan, D.J.; Parikh, S.S.; Lewis, B.K.; Gindi, R.M.; Keteyian, C.; Abdul-Nour, K.; Keteyian, S.J. Inverse Relationship of Maximal Exercise Capacity to Hospitalization Secondary to Coronavirus Disease 2019. Mayo Clin. Proc. 2021, 96, 32–39. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Health and Research (NIHR). NIHR Themed Review: Living with Covid19—Second Review; NIHR: Manchester, UK, 2021. [Google Scholar]
- Faghy, M.A.; Arena, R.; Stoner, L.; Haraf, R.H.; Josephson, R.; Hills, A.P.; Dixit, S.; Popovic, D.; Smith, A.; Myers, J.; et al. The need for exercise sciences and an integrated response to COVID-19: A position statement from the international HL-PIVOT network. Prog. Cardiovasc. Dis. 2021. [Google Scholar] [CrossRef]
- National Insitute for Health and Care Excellence; SIGN. Royal College of General Practitioners COVID-19 Guideline Scope: Management of the Long-Term Effects of COVID-19; NICE: London, UK, 2020; pp. 1–7. [Google Scholar]
- UK Office for National Statistics. Prevalence of Ongoing Symptoms Following Coronavirus (COVID-19) Infection in the UK: 1 April 2021; ONS: London, UK, 2021.
- Mahase, E. Long covid could be four different syndromes, review suggests. BMJ 2020, 371, m3981. [Google Scholar] [CrossRef]
- Cevik, M.; Tate, M.; Lloyd, O.; Maraolo, A.E.; Schafers, J.; Ho, A. SARS-CoV-2, SARS-CoV, and MERS-CoV viral load dynamics, duration of viral shedding, and infectiousness: A systematic review and meta-analysis. Lancet Microbe 2021, 2, e13–e22. [Google Scholar] [CrossRef]
- Basile, K.; McPhie, K.; Carter, I.; Alderson, S.; Rahman, H.; Donovan, L.; Kumar, S.; Tran, T.; Ko, D.; Sivaruban, T.; et al. Cell-based culture of SARS-CoV-2 informs infectivity and safe de-isolation assessments during COVID-19. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Datta, S.D.; Talwar, A.; Lee, J.T. A Proposed Framework and Timeline of the Spectrum of Disease Due to SARS-CoV-2 Infection: Illness beyond Acute Infection and Public Health Implications. JAMA 2020, 324, 2251–2252. [Google Scholar] [CrossRef]
- Amenta, E.M.; Spallone, A.; Rodriguez-Barradas, M.C.; El Sahly, H.M.; Atmar, R.L.; Kulkarni, P.A. Postacute COVID-19: An Overview and Approach to Classification. Open Forum Infect. Dis. 2020, 7, ofaa509. [Google Scholar] [CrossRef]
- Raveendran, A. Long COVID-19: Challenges in the diagnosis and proposed diagnostic criteria. Diabetes Metab. Syndr. Clin. Res. Rev. 2021, 15, 145–146. [Google Scholar] [CrossRef] [PubMed]
- Tillett, R.L.; Sevinsky, J.R.; Hartley, P.D.; Kerwin, H.; Crawford, N.; Gorzalski, A.; Laverdure, C.; Verma, S.C.; Rossetto, C.C.; Jackson, D.; et al. Genomic evidence for reinfection with SARS-CoV-2: A case study. Lancet Infect. Dis. 2021, 21, 52–58. [Google Scholar] [CrossRef]
- Chapin-Bardales, J.; Gee, J.; Myers, T. Reactogenicity Following Receipt of mRNA-Based COVID-19 Vaccines. JAMA 2021. [Google Scholar] [CrossRef] [PubMed]
- WHO Director-General’s Opening Remarks at the Media Briefing on COVID-19—24 February 2020. Available online: https://www.who.int/director-general/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---24-february-2020 (accessed on 27 December 2020).
- Coronavirus Disease 2019 (COVID-19): Clinical Features—UpToDate. Available online: https://www.uptodate.com/contents/coronavirus-disease-2019-covid-19-clinical-features?search=undefined&source=covid19_landing&usage_type=main_section#H4100426989 (accessed on 27 December 2020).
- SEMG. Encuesta COVID-19 Persistente. Presentación de Resultados. 11 de Noviembre de 2020. Available online: https://www.semg.es/images/2020/Noticias/20201111_Resultados_Encuesta_COVID_Persistente.pdf (accessed on 30 December 2020).
- Davis, H.E.; Assaf, G.S.; McCorkell, L.; Wei, H.; Low, R.J.; Re’Em, Y.; Redfield, S.; Austin, J.P.; Akrami, A. Characterizing Long COVID in an International Cohort: 7 Months of Symptoms and Their Impact. medRxiv 2020. [Google Scholar] [CrossRef]
- Lopez-Leon, S.; Wegman-Ostrosky, T.; Perelman, C.; Sepulveda, R.; Rebolledo, P.A.; Cuapio, A.; Villapol, S. More than 50 Long-term effects of COVID-19: A systematic review and meta-analysis. medRxiv 2021. [Google Scholar] [CrossRef]
- Michelen, M.; Manoharan, L.; Elkheir, N.; Cheng, V.; Dagens, D.; Hastie, C.; O’Hara, M.; Suett, J.C.; Burls, A.; Foote, C.; et al. Characterising long-term covid-19: A rapid living systematic review. medRxiv 2020. [Google Scholar] [CrossRef]
- Hoshijima, H.; Mihara, T.; Seki, H.; Hyuga, S.; Kuratani, N.; Shiga, T. Incidence of Long-term Post-acute Sequelae of SARS-CoV-2 Infection Related to Pain and Other Symptoms: A Living Systematic Review and Meta-analysis. medRxiv 2021. [Google Scholar] [CrossRef]
- Garrigues, E.; Janvier, P.; Kherabi, Y.; Le Bot, A.; Hamon, A.; Gouze, H.; Doucet, L.; Berkani, S.; Oliosi, E.; Mallart, E.; et al. Post-discharge persistent symptoms and health-related quality of life after hospitalization for COVID-19. J. Infect. 2020, 81, e4–e6. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Huang, L.; Wang, Y.; Li, X.; Ren, L.; Gu, X.; Kang, L.; Guo, L.; Liu, M.; Zhou, X.; et al. 6-month consequences of COVID-19 in patients discharged from hospital: A cohort study. Lancet 2021, 397, 220–232. [Google Scholar] [CrossRef]
- Bellan, M.; Soddu, D.; Balbo, P.E.; Baricich, A.; Zeppegno, P.; Avanzi, G.C.; Baldon, G.; Bartolomei, G.; Battaglia, M.; Battistini, S.; et al. Respiratory and Psychophysical Sequelae Among Patients With COVID-19 Four Months After Hospital Discharge. JAMA Netw. Open 2021, 4, e2036142. [Google Scholar] [CrossRef]
- Sykes, D.L.; Holdsworth, L.; Jawad, N.; Gunasekera, P.; Morice, A.H.; Crooks, M.G. Post-COVID-19 Symptom Burden: What is Long-COVID and How Should We Manage It? Lung 2021, 199, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Group, P.-C.C.; Evans, R.A.; McAuley, H.; Harrison, E.M.; Shikotra, A.; Singapuri, A.; Sereno, M.; Elneima, O.; Docherty, A.B.; Lone, N.I.; et al. Physical, cognitive and mental health impacts of COVID-19 following hospitalisation—A multi-centre prospective cohort study. medRxiv 2021. [Google Scholar] [CrossRef]
- Landi, F.; Carfì, A.; Benvenuto, F.; Brandi, V.; Ciciarello, F.; Monaco, M.R.L.; Martone, A.M.; Napolitano, C.; Pagano, F.; Paglionico, A.; et al. Predictive Factors for a New Positive Nasopharyngeal Swab Among Patients Recovered From COVID-19. Am. J. Prev. Med. 2021, 60, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Arnold, D.T.; Hamilton, F.; Milne, A.; Morley, A.J.; Viner, J.; Attwood, M.; Noel, A.; Gunning, S.; Hatrick, J.; Hamilton, S.; et al. Patient outcomes after hospitalisation with COVID-19 and implications for follow-up: Results from a prospective UK cohort. Thorax 2021, 76, 399–401. [Google Scholar] [CrossRef]
- Mazza, M.G.; De Lorenzo, R.; Conte, C.; Poletti, S.; Vai, B.; Bollettini, I.; Melloni, E.M.T.; Furlan, R.; Ciceri, F.; Rovere-Querini, P.; et al. Anxiety and depression in COVID-19 survivors: Role of inflammatory and clinical predictors. Brain, Behav. Immun. 2020, 89, 594–600. [Google Scholar] [CrossRef]
- D’Cruz, R.F.; Waller, M.D.; Perrin, F.; Periselneris, J.; Norton, S.; Smith, L.-J.; Patrick, T.; Walder, D.; Heitmann, A.; Lee, K.; et al. Chest radiography is a poor predictor of respiratory symptoms and functional impairment in survivors of severe COVID-19 pneumonia. ERJ Open Res. 2021, 7. [Google Scholar] [CrossRef]
- Daher, A.; Balfanz, P.; Cornelissen, C.; Müller, A.; Bergs, I.; Marx, N.; Müller-Wieland, D.; Hartmann, B.; Dreher, M.; Müller, T. Follow up of patients with severe coronavirus disease 2019 (COVID-19): Pulmonary and extrapulmonary disease sequelae. Respir. Med. 2020, 174, 106197. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.; Yang, B.; Jiang, N.; Fu, W.; He, X.; Zhou, Y.; Ma, W.-L.; Wang, X. Three-Month Follow-Up Study of Survivors of Coronavirus Disease 2019 after Discharge. J. Korean Med. Sci. 2020, 35, e418. [Google Scholar] [CrossRef]
- Weerahandi, H.; Hochman, K.A.; Simon, E.; Blaum, C.; Chodosh, J.; Duan, E.; Garry, K.; Kahan, T.; Karmen-Tuohy, S.L.; Karpel, H.C.; et al. Post-Discharge Health Status and Symptoms in Patients with Severe COVID-19. medRxiv Prepr. Serv. Health Sci. 2020. [Google Scholar] [CrossRef]
- Xiong, Q.; Xu, M.; Li, J.; Liu, Y.; Zhang, J.; Xu, Y.; Dong, W. Clinical sequelae of COVID-19 survivors in Wuhan, China: A single-centre longitudinal study. Clin. Microbiol. Infect. 2021, 27, 89–95. [Google Scholar] [CrossRef]
- Mandal, S.; Barnett, J.; Brill, S.E.; Brown, J.S.; Denneny, E.K.; Hare, S.S.; Heightman, M.; Hillman, T.E.; Jacob, J.; Jarvis, H.C.; et al. ‘Long-COVID’: A cross-sectional study of persisting symptoms, biomarker and imaging abnormalities following hospitalisation for COVID-19. Thorax 2021, 76, 396–398. [Google Scholar] [CrossRef]
- Carfì, A.; Bernabei, R.; Landi, F. Persistent symptoms in patients after acute COVID-19. JAMA 2020, 324, 603–605. [Google Scholar] [CrossRef]
- Halpin, S.J.; McIvor, C.; Whyatt, G.; Adams, A.; Harvey, O.; McLean, L.; Walshaw, C.; Kemp, S.; Corrado, J.; Singh, R.; et al. Postdischarge symptoms and rehabilitation needs in survivors of COVID-19 infection: A cross-sectional evaluation. J. Med Virol. 2021, 93, 1013–1022. [Google Scholar] [CrossRef] [PubMed]
- Poyraz, B.C.; Poyraz, C.A.; Olğun, Y.; Gürel, O.; Alkan, S.; Özdemir, Y.E.; Balkan, I.I.; Karaali, R. Psychiatric morbidity and protracted symptoms in recovered COVID-19 patients. medRxiv 2020. [Google Scholar] [CrossRef]
- Goërtz, Y.M.; Van Herck, M.; Delbressine, J.M.; Vaes, A.W.; Meys, R.; Machado, F.V.; Houben-Wilke, S.; Burtin, C.; Posthuma, R.; Franssen, F.M.; et al. Persistent symptoms 3 months after a SARS-CoV-2 infection: The post-COVID-19 syndrome? ERJ Open Res. 2020, 6, 00542–02020. [Google Scholar] [CrossRef] [PubMed]
- Valiente-De Santis, L.; Pérez-Camacho, I.; Sobrino, B.; González, G.E.; Ruíz-Mesa, J.D.; Plata, A.; Márquez-Gómez, I.; Delgado-Fernández, M.; Castaño, M.; Oñate, F.; et al. Clinical and immunoserological status 12 weeks after infection with COVID-19. medRxiv 2020. [Google Scholar] [CrossRef]
- Tenforde, M.W.; Kim, S.S.; Lindsell, C.J.; Rose, E.B.; Shapiro, N.I.; Files, D.C.; Gibbs, K.W.; Erickson, H.L.; Steingrub, J.S.; Smithline, H.A.; et al. Symptom Duration and Risk Factors for Delayed Return to Usual Health Among Outpatients with COVID-19 in a Multistate Health Care Systems Network—United States, March–June 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 993–998. [Google Scholar] [CrossRef]
- Boscolo-Rizzo, P.; Borsetto, D.; Fabbris, C.; Spinato, G.; Frezza, D.; Menegaldo, A.; Mularoni, F.; Gaudioso, P.; Cazzador, D.; Marciani, S.; et al. Evolution of Altered Sense of Smell or Taste in Patients With Mildly Symptomatic COVID-19. JAMA Otolaryngol. Neck Surg. 2020, 146, 729. [Google Scholar] [CrossRef] [PubMed]
- Kamal, M.; Omirah, M.A.; Hussein, A.; Saeed, H. Assessment and characterisation of post-COVID-19 manifestations. Int. J. Clin. Pract. 2021, 75. [Google Scholar] [CrossRef]
- Dennis, A.; Mbbch, J.A.; Badley, A.D.; Anton, G.; Mbbch, D. Multi-organ impairment in low-risk individuals with long COVID. medRxiv 2020. [Google Scholar] [CrossRef]
- Carvalho-Schneider, C.; Laurent, E.; Lemaignen, A.; Beaufils, E.; Bourbao-Tournois, C.; Laribi, S.; Flament, T.; Ferreira-Maldent, N.; Bruyère, F.; Stefic, K.; et al. Follow-up of adults with noncritical COVID-19 two months after symptom onset. Clin. Microbiol. Infect. 2021, 27, 258–263. [Google Scholar] [CrossRef]
- Assaf, G.; Davis, H.; McCorkell, L.; Wei, H.; Brooke, N.; Akrami, A.; Low, R.; Mercier, J. What Does COVID-19 Recovery Actually Look Like? An Analysis of the Prolonged COVID-19 Symptoms Survey by Patient-Led Research Team. Available online: www.patientresearchcovid19.com (accessed on 31 January 2021).
- Patterson, B.K.; Guevara-Coto, J.; Yogendra, R.; Francisco, E.; Long, E.; Pise, A.; Rodrigues, H.; Parikh, P.; Mora, J.; Mora-Rodríguez, R.A. Immune-based prediction of COVID-19 severity and chronicity decoded using machine learning. bioRxiv 2020. [Google Scholar] [CrossRef]
- Chopra, V.; Flanders, S.A.; O’Malley, M.; Malani, A.N.; Prescott, H.C. Sixty-Day Outcomes Among Patients Hospitalized With COVID-19. Ann. Intern. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Richards, A.; Khalil, A.; Wogram, E.; Ma, H.; Jaenisch, R. SARS-CoV-2 RNA reverse-transcribed and integrated into the human genome 1. bioRxiv 2020. [Google Scholar] [CrossRef]
- Sokolowska, M.; Lukasik, Z.M.; Agache, I.; Akdis, C.A.; Akdis, D.; Akdis, M.; Barcik, W.; Brough, H.A.; Eiwegger, T.; Eljaszewicz, A.; et al. Immunology of COVID-19: Mechanisms, clinical outcome, diagnostics, and perspectives—A report of the European Academy of Allergy and Clinical Immunology (EAACI). Allergy 2020, 75, 2445–2476. [Google Scholar] [CrossRef]
- Wang, E.Y.; Mao, T.; Klein, J.; Dai, Y.; Huck, J.D.; Zheng, N.S.; Zhou, T.; Israelow, B.; Wong, P.; Lucas, C. Diverse Functional Autoantibodies in Patients with COVID-19. medRxiv 2020. [Google Scholar] [CrossRef]
- Jin, Y.; Ji, W.; Yang, H.; Chen, S.; Zhang, W.; Duan, G. Endothelial activation and dysfunction in COVID-19: From basic mechanisms to potential therapeutic approaches. Signal Transduct. Target. Ther. 2020, 5, 1–13. [Google Scholar] [CrossRef]
- Torres-Castro, R.; Vasconcello-Castillo, L.; Alsina-Restoy, X.; Solis-Navarro, L.; Burgos, F.; Puppo, H.; Vilaró, J. Respiratory function in patients post-infection by COVID-19: A systematic review and meta-analysis. Pulmonology 2020. [Google Scholar] [CrossRef]
- Xiao, S.; Luo, D.; Xiao, Y. Survivors of COVID-19 are at high risk of posttraumatic stress disorder. Glob. Health Res. Policy 2020, 5, 29. [Google Scholar] [CrossRef]
- Wang, Y.; Shi, L.; Que, J.; Lu, Q.; Liu, L.; Lu, Z.; Xu, Y.; Liu, J.; Sun, Y.; Meng, S.; et al. The impact of quarantine on mental health status among general population in China during the COVID-19 pandemic. Mol. Psychiatry 2021, 1–10. [Google Scholar] [CrossRef]
- Kumar, S.; Veldhuis, A.; Malhotra, T. Neuropsychiatric and Cognitive Sequelae of COVID-19. Front. Psychol. 2021, 12, 577529. [Google Scholar] [CrossRef]
- Dani, M.; Dirksen, A.; Taraborrelli, P.; Torocastro, M.; Panagopoulos, D.; Sutton, R.; Lim, P.B. Autonomic dysfunction in ‘long COVID’: Rationale, physiology and management strategies. Clin. Med. 2021, 21, e63–e67. [Google Scholar] [CrossRef] [PubMed]
- Search of: COVID-19—List Results—ClinicalTrials.gov. Available online: https://www.clinicaltrials.gov/ct2/results?cond=COVID-19 (accessed on 7 November 2020).
- U.S. Department of Health and Human Services. Physical Activity Guidelines for Americans, 2nd ed.; Department of Health and Human Services: Washington, DC, USA, 2018. [CrossRef]
- Campbell, J.P.; Turner, J.E. Debunking the Myth of Exercise-Induced Immune Suppression: Redefining the Impact of Exercise on Immunological Health Across the Lifespan. Front. Immunol. 2018, 9, 648. [Google Scholar] [CrossRef]
- Saçma, M.; Geiger, H. Exercise generates immune cells in bone. Nat. Cell Biol. 2021, 591, 371–372. [Google Scholar] [CrossRef]
- Scheffer, D.D.L.; Latini, A. Exercise-induced immune system response: Anti-inflammatory status on peripheral and central organs. BBA Mol. Basis Dis. 2020, 1866, 165823. [Google Scholar] [CrossRef]
- Casuso, R.A.; Huertas, J.R. Mitochondrial Functionality in Inflammatory Pathology-Modulatory Role of Physical Activity. Life 2021, 11, 61. [Google Scholar] [CrossRef]
- Padilha, C.S.; Figueiredo, C.; Minuzzi, L.G.; Chimin, P.; Deminice, R.; Krüger, K.; Rosa-Neto, J.C.; Lira, F.S. Immunometabolic responses according to physical fitness status and lifelong exercise during aging: New roads for exercise immunology. Ageing Res. Rev. 2021, 68, 101341. [Google Scholar] [CrossRef] [PubMed]
- Duggal, N.A.; Niemiro, G.; Harridge, S.D.R.; Simpson, R.J.; Lord, J.M. Can physical activity ameliorate immunosenescence and thereby reduce age-related multi-morbidity? Nat. Rev. Immunol. 2019, 19, 563–572. [Google Scholar] [CrossRef] [PubMed]
- Sharif, K.; Watad, A.; Bragazzi, N.L.; Lichtbroun, M.; Amital, H.; Shoenfeld, Y. Physical activity and autoimmune diseases: Get moving and manage the disease. Autoimmun. Rev. 2018, 17, 53–72. [Google Scholar] [CrossRef]
- Metcalfe, R.S.; Kemp, R.; Heffernan, S.M.; Churm, R.; Chen, Y.-C.; Ruffino, J.S.; Conway, G.E.; Tornillo, G.; Orange, S.T. Anti-carcinogenic effects of exercise-conditioned human serum: Evidence, relevance and opportunities. Graefe’s Arch. Clin. Exp. Ophthalmol. 2021, 1–18. [Google Scholar] [CrossRef]
- Valenzuela, P.L.; Simpson, R.J.; Castillo-García, A.; Lucia, A. Physical activity: A coadjuvant treatment to COVID-19 vaccination? Brain Behav. Immun. 2021, 94, 1–3. [Google Scholar] [CrossRef]
- Chastin, S.F.M.; Abaraogu, U.; Bourgois, J.G.; Dall, P.M.; Darnborough, J.; Duncan, E.; Dumortier, J.; Pavón, D.J.; McParland, J.; Roberts, N.J.; et al. Effects of Regular Physical Activity on the Immune System, Vaccination and Risk of Community-Acquired Infectious Disease in the General Population: Systematic Review and Meta-Analysis. Sports Med. 2021, 1–14. [Google Scholar] [CrossRef]
- Cooper, D.M.; Radom-Aizik, S.; Schwindt, C.; Zaldivar, F. Dangerous exercise: Lessons learned from dysregulated inflammatory responses to physical activity. J. Appl. Physiol. 2007, 103, 700–709. [Google Scholar] [CrossRef] [PubMed]
- Liao, P.; He, Q.; Zhou, X.; Ma, K.; Wen, J.; Chen, H.; Li, Q.; Qin, D.; Wang, H. Repetitive Bouts of Exhaustive Exercise Induces a Systemic Inflammatory Response and Multi-Organ Damage in Rats. Front. Physiol. 2020, 11, 685. [Google Scholar] [CrossRef] [PubMed]
- Flockhart, M.; Nilsson, L.C.; Tais, S.; Ekblom, B.; Apró, W.; Larsen, F.J. Excessive exercise training causes mitochondrial functional impairment and decreases glucose tolerance in healthy volunteers. Cell Metab. 2021, 33, 957–970.e6. [Google Scholar] [CrossRef]
- Osthoff, A.K.R.; Niedermann, K.; Braun, J.; Adams, J.; Brodin, N.; Dagfinrud, H.; Duruoz, T.; Esbensen, B.A.; Günther, K.-P.; Hurkmans, E.; et al. 2018 EULAR recommendations for physical activity in people with inflammatory arthritis and osteoarthritis. Ann. Rheum. Dis. 2018, 77, 1251–1260. [Google Scholar] [CrossRef]
- Hilberdink, B.; Vlieland, T.V.; Van Der Giesen, F.; Van Gaalen, F.; Goekoop, R.; Peeters, A.; Fiocco, M.; Van Weely, S. Adequately dosed aerobic physical activity in people with axial spondyloarthritis: Associations with physical therapy. Rheumatol. Int. 2020, 40, 1519–1528. [Google Scholar] [CrossRef]
- Peçanha, T.; Bannell, D.; Sieczkowska, S.M.; Goodson, N.; Roschel, H.; Sprung, V.S.; Low, D.A. Effects of physical activity on vascular function in autoimmune rheumatic diseases: A systematic review and meta-analysis. Rheumatology 2021. [Google Scholar] [CrossRef]
- Bidonde, J.; Busch, A.J.; Schachter, C.L.; Webber, S.C.; Musselman, K.E.; Overend, T.J.; Góes, S.M.; Bello-Haas, V.D.; Boden, C. Mixed exercise training for adults with fibromyalgia. Cochrane Database Syst. Rev. 2019, 5, CD013340. [Google Scholar] [CrossRef]
- De Asteasu, M.L.S.; Martínez-Velilla, N.; Zambom-Ferraresi, F.; Ramírez-Vélez, R.; García-Hermoso, A.; Cadore, E.L.; Casas-Herrero, Á.; Galbete, A.; Izquierdo, M. Changes in muscle power after usual care or early structured exercise intervention in acutely hospitalized older adults. J. Cachex. Sarcopenia Muscle 2020, 11, 997–1006. [Google Scholar] [CrossRef]
- Cadore, E.L.; Pinto, R.S.; Bottaro, M.; Izquierdo, M. Strength and endurance training prescription in healthy and frail elderly. Aging Dis. 2014, 5, 183–195. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Velilla, N.; Casas-Herrero, A.; Zambom-Ferraresi, F.; De Asteasu, M.L.S.; Lucia, A.; Galbete, A.; García-Baztán, A.; Alonso-Renedo, J.; González-Glaría, B.; Gonzalo-Lázaro, M.; et al. Effect of Exercise Intervention on Functional Decline in Very Elderly Patients During Acute Hospitalization. JAMA Intern. Med. 2019, 179, 28–36. [Google Scholar] [CrossRef]
- Courel-Ibáñez, J.; Pallarés, J.G.; García-Conesa, S.; Buendía-Romero, Á.; Martínez-Cava, A.; Izquierdo, M. Supervised Exercise (Vivifrail) Protects Institutionalized Older Adults Against Severe Functional Decline After 14 Weeks of COVID Confinement. J. Am. Med Dir. Assoc. 2021, 22, 217–219.e2. [Google Scholar] [CrossRef] [PubMed]
- Maestroni, L.; Read, P.; Bishop, C.; Papadopoulos, K.; Suchomel, T.J.; Comfort, P.; Turner, A. The Benefits of Strength Training on Musculoskeletal System Health: Practical Applications for Interdisciplinary Care. Sports Med. 2020, 50, 1431–1450. [Google Scholar] [CrossRef]
- Vieira, A.F.; Umpierre, D.; Teodoro, J.L.; Lisboa, S.C.; Baroni, B.M.; Izquierdo, M.; Cadore, E.L. Effects of Resistance Training Performed to Failure or Not to Failure on Muscle Strength, Hypertrophy, and Power Output: A Systematic Review With Meta-Analysis. J. Strength Cond. Res. 2021, 35, 1165–1175. [Google Scholar] [CrossRef] [PubMed]
- Grgic, J.; Schoenfeld, B.J. Higher effort, rather than higher load, for resistance exercise-induced activation of muscle fibres. J. Physiol. 2019, 597, 4691–4692. [Google Scholar] [CrossRef]
- Schoenfeld, B.J.; Contreras, B.; Krieger, J.; Grgic, J.; Delcastillo, K.; Belliard, R.; Alto, A. Resistance Training Volume Enhances Muscle Hypertrophy but Not Strength in Trained Men. Med. Sci. Sports Exerc. 2019, 51, 94–103. [Google Scholar] [CrossRef]
- Bilberg, A.; Sveaas, S.H.; Dagfinrud, H.; Mannerkorpi, K. How Do Patients With Axial Spondyloarthritis Experience High-Intensity Exercise? ACR Open Rheumatol. 2020, 2, 207–213. [Google Scholar] [CrossRef]
- O’Brien, C.M.; Ntoumanis, N.; Duda, J.L.; Kitas, G.D.; van Zanten, J.J.C.S.V.; Metsios, G.S.; Fenton, S.A.M. Pain and fatigue are longitudinally and bi-directionally associated with more sedentary time and less standing time in rheumatoid arthritis. Rheumatology 2021, 1–10. [Google Scholar] [CrossRef]
- Van Zanten, J.J.V.; Fenton, S.A.; Rouse, P.C.; Ntoumanis, N.; Osailan, A.; Yu, C.-A.; Metsios, G.S.; Kitas, G.D.; Duda, J.L. Autonomous motivation, cardiorespiratory fitness, and exercise in rheumatoid arthritis: Randomised controlled trial. Psychol. Sport Exerc. 2021, 101904, 101904. [Google Scholar] [CrossRef]
- Dowman, L.; Hill, C.J.; May, A.; Holland, A.E. Pulmonary rehabilitation for interstitial lung disease. Cochrane Database Syst. Rev. 2021. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, B.; Casey, D.; DeVane, D.; Murphy, K.; Murphy, E.; Lacasse, Y. Pulmonary rehabilitation for chronic obstructive pulmonary disease. Cochrane Database Syst. Rev. 2015, 2, CD003793. [Google Scholar] [CrossRef] [PubMed]
- Lindenauer, P.K.; Stefan, M.S.; Pekow, P.S.; Mazor, K.M.; Priya, A.; Spitzer, K.A.; Lagu, T.C.; Pack, Q.R.; Pinto-Plata, V.M.; ZuWallack, R. Association between Initiation of Pulmonary Rehabilitation after Hospitalization for COPD and 1-Year Survival among Medicare Beneficiaries. JAMA 2020, 323, 1813–1823. [Google Scholar] [CrossRef]
- Sawyer, A.; Cavalheri, V.; Hill, K. Effects of high intensity interval training on exercise capacity in people with chronic pulmonary conditions: A narrative review. BMC Sports Sci. Med. Rehabil. 2020, 12, 1–10. [Google Scholar] [CrossRef]
- Greulich, T.; Kehr, K.; Nell, C.; Koepke, J.; Haid, D.; Koehler, U.; Koehler, K.; Filipovic, S.; Kenn, K.; Vogelmeier, C.; et al. A randomized clinical trial to assess the influence of a three months training program (Gym-based individualized vs. Calisthenics-based non-invidualized) in COPD-patients. Respir. Res. 2014, 15, 36. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nyberg, A.; Martin, M.; Saey, D.; Milad, N.; Patoine, D.; Morissette, M.C.; Auger, D.; Stål, P.; Maltais, F. Effects of Low-Load/High-Repetition Resistance Training on Exercise Capacity, Health Status, and Limb Muscle Adaptation in Patients With Severe COPD. Chest 2021, 159, 1821–1832. [Google Scholar] [CrossRef] [PubMed]
- Alcazar, J.; Losa-Reyna, J.; Rodriguez-Lopez, C.; Navarro-Cruz, R.; Alfaro-Acha, A.; Ara, I.; García-García, F.J.; Alegre, L.M.; Guadalupe-Grau, A. Effects of concurrent exercise training on muscle dysfunction and systemic oxidative stress in older people with COPD. Scand. J. Med. Sci. Sports 2019, 29, 1591–1603. [Google Scholar] [CrossRef] [PubMed]
- Guadalupe-Grau, A.; Aznar-Laín, S.; Mañas, A.; Castellanos, J.; Alcázar, J.; Ara, I.; Mata, E.; Daimiel, R.; García-García, F.J. Short- and Long-Term Effects of Concurrent Strength and HIIT Training in Octogenarians with COPD. J. Aging Phys. Act. 2017, 25, 105–115. [Google Scholar] [CrossRef]
- Liu, K.; Zhang, W.; Yang, Y.; Zhang, J.; Li, Y.; Chen, Y. Respiratory rehabilitation in elderly patients with COVID-19: A randomized controlled study. Complement. Ther. Clin. Pract. 2020, 39, 101166. [Google Scholar] [CrossRef]
- Desveaux, L.; Janaudis-Ferreira, T.; Goldstein, R.; Brooks, D. An International Comparison of Pulmonary Rehabilitation: A Systematic Review. COPD: J. Chronic Obstr. Pulm. Dis. 2014, 12, 144–153. [Google Scholar] [CrossRef]
- Pinckard, K.; Baskin, K.K.; Stanford, K.I. Effects of Exercise to Improve Cardiovascular Health. Front. Cardiovasc. Med. 2019, 6, 69. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R.J.; Balady, G.; Banka, G.; Beckie, T.M.; Chiu, J.; Gokak, S.; Ho, P.M.; Keteyian, S.J.; King, M.; Lui, K.; et al. 2018 ACC/AHA Clinical Performance and Quality Measures for Cardiac Rehabilitation: A Report of the American College of Cardiology/American Heart Association Task Force on Performance Measures. J. Am. Coll. Cardiol. 2018, 71, 1814–1837. [Google Scholar] [CrossRef] [PubMed]
- Candelaria, D.; Randall, S.; Ladak, L.; Gallagher, R. Health-related quality of life and exercise-based cardiac rehabilitation in contemporary acute coronary syndrome patients: A systematic review and meta-analysis. Qual. Life Res. 2020, 29, 579–592. [Google Scholar] [CrossRef] [PubMed]
- Spruit, M.A.; Rochester, C.L.; Pitta, F.; Kenn, K.; Schols, A.M.; Hart, N.; Wouters, E.F.; Nava, S.; Dreher, M.; Janssen, D.J.; et al. Pulmonary rehabilitation, physical activity, respiratory failure and palliative respiratory care. Thorax 2019, 74, 693–699. [Google Scholar] [CrossRef]
- Whelton, P.K.; Carey, R.M.; Aronow, W.S.; Casey, D.E.; Collins, K.J.; Himmelfarb, C.D.; DePalma, S.M.; Gidding, S.; Jamerson, K.A.; Jones, D.W.; et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults a report of the American College of Cardiology/American Heart Association Task Force on Clinical pr. Hypertension 2018, 71, E13–E115. [Google Scholar] [CrossRef] [PubMed]
- Pedralli, M.L.; Marschner, R.A.; Kollet, D.P.; Neto, S.G.; Eibel, B.; Tanaka, H.; Lehnen, A.M. Different exercise training modalities produce similar endothelial function improvements in individuals with prehypertension or hypertension: A randomized clinical trial. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Pelliccia, A.; Sharma, S.; Gati, S.; Bäck, M.; Börjesson, M.; Caselli, S. 2020 ESC Guidelines on sports cardiology and exercise in patients with cardiovascular disease. Eur. Heart J. 2020, 42, 17–96. [Google Scholar] [CrossRef]
- Vina, J.; Sanchis-Gomar, F.; Martinez-Bello, V.; Gomez-Cabrera, M.C. Exercise acts as a drug; the pharmacological benefits of exercise. Br. J. Pharmacol. 2012, 167, 1–12. [Google Scholar] [CrossRef]
- Chen, Z.; Lan, W.; Yang, G.; Li, Y.; Ji, X.; Chen, L.; Zhou, Y.; Li, S. Exercise Intervention in Treatment of Neuropsychological Diseases: A Review. Front. Psychol. 2020, 11, 569206. [Google Scholar] [CrossRef]
- Marques-Aleixo, I.; Beleza, J.; Sampaio, A.; Stevanović, J.; Coxito, P.; Gonçalves, I.; Ascensão, A.; Magalhães, J. Preventive and Therapeutic Potential of Physical Exercise in Neurodegenerative Diseases. Antioxid. Redox Signal. 2021, 34, 674–693. [Google Scholar] [CrossRef] [PubMed]
- Jia, R.-X.; Liang, J.-H.; Xu, Y.; Wang, Y.-Q. Effects of physical activity and exercise on the cognitive function of patients with Alzheimer disease: A meta-analysis. BMC Geriatr. 2019, 19, 1–14. [Google Scholar] [CrossRef]
- Fu, Q.; Levine, B.D. Exercise in the postural orthostatic tachycardia syndrome. Auton. Neurosci. 2015, 188, 86–89. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.; VanGundy, T.B.; Shibata, S.; Auchus, R.J.; Williams, G.H.; Levine, B.D. Exercise Training Versus Propranolol in the Treatment of the Postural Orthostatic Tachycardia Syndrome. Hypertension 2011, 58, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Eöry, A.; Békési, D.; Rózsa, S. Physical Exercise as a Resilience Factor to Mitigate COVID-Related Allostatic Overload. Psychother. Psychosom. 2021, 90, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Courel-Ibáñez, J.; Martínez-Cava, A.; Morán-Navarro, R.; Escribano-Peñas, P.; Chavarren-Cabrero, J.; González-Badillo, J.J.; Pallarés, J.G. Reproducibility and Repeatability of Five Different Technologies for Bar Velocity Measurement in Resistance Training. Ann. Biomed. Eng. 2019, 47, 1523–1538. [Google Scholar] [CrossRef] [PubMed]
- Pallarés, J.G.; Cava, A.M.; Courel-Ibáñez, J.; González-Badillo, J.J.; Morán-Navarro, R. Full squat produces greater neuromuscular and functional adaptations and lower pain than partial squats after prolonged resistance training. Eur. J. Sport Sci. 2020, 20, 115–124. [Google Scholar] [CrossRef]
- Martínez-Cava, A.; Hernández-Belmonte, A.; Courel-Ibáñez, J.; Conesa-Ros, E.; Morán-Navarro, R.; Pallarés, J.G. Effect of Pause Versus Rebound Techniques on Neuromuscular and Functional Performance After a Prolonged Velocity-Based Training. Int. J. Sports Physiol. Perform. 2021, 1, 1–7. [Google Scholar] [CrossRef]
- Morán-Navarro, R.; Courel-Ibáñez, J.; Martínez-Cava, A.; Conesa-Ros, E.; Sánchez-Pay, A.; Mora-Rodriguez, R.; Pallarés, J.G. Validity of Skin, Oral and Tympanic Temperatures During Exercise in the Heat: Effects of Wind and Sweat. Ann. Biomed. Eng. 2019, 47, 317–331. [Google Scholar] [CrossRef]
- Rodrigo-Carranza, V.; González-Mohíno, F.; Turner, A.P.; Rodriguez-Barbero, S.; González-Ravé, J.M. Using a Portable Near-infrared Spectroscopy Device to Estimate The Second Ventilatory Threshold. Int. J. Sports Med. 2021. [Google Scholar] [CrossRef]
- Molina-Molina, A.; Ruiz-Malagón, E.J.; Carrillo-Pérez, F.; Roche-Seruendo, L.E.; Damas, M.; Banos, O.; García-Pinillos, F. Validation of mDurance, a Wearable Surface Electromyography System for Muscle Activity Assessment. Front. Physiol. 2020, 11, 11. [Google Scholar] [CrossRef]
- Rigamonti, L.; Albrecht, U.-V.; Lutter, C.; Tempel, M.; Wolfarth, B.; Back, D.A. Potentials of Digitalization in Sports Medicine. Curr. Sports Med. Rep. 2020, 19, 157–163. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jimeno-Almazán, A.; Pallarés, J.G.; Buendía-Romero, Á.; Martínez-Cava, A.; Franco-López, F.; Sánchez-Alcaraz Martínez, B.J.; Bernal-Morel, E.; Courel-Ibáñez, J. Post-COVID-19 Syndrome and the Potential Benefits of Exercise. Int. J. Environ. Res. Public Health 2021, 18, 5329. https://doi.org/10.3390/ijerph18105329
Jimeno-Almazán A, Pallarés JG, Buendía-Romero Á, Martínez-Cava A, Franco-López F, Sánchez-Alcaraz Martínez BJ, Bernal-Morel E, Courel-Ibáñez J. Post-COVID-19 Syndrome and the Potential Benefits of Exercise. International Journal of Environmental Research and Public Health. 2021; 18(10):5329. https://doi.org/10.3390/ijerph18105329
Chicago/Turabian StyleJimeno-Almazán, Amaya, Jesús G. Pallarés, Ángel Buendía-Romero, Alejandro Martínez-Cava, Francisco Franco-López, Bernardino J. Sánchez-Alcaraz Martínez, Enrique Bernal-Morel, and Javier Courel-Ibáñez. 2021. "Post-COVID-19 Syndrome and the Potential Benefits of Exercise" International Journal of Environmental Research and Public Health 18, no. 10: 5329. https://doi.org/10.3390/ijerph18105329
APA StyleJimeno-Almazán, A., Pallarés, J. G., Buendía-Romero, Á., Martínez-Cava, A., Franco-López, F., Sánchez-Alcaraz Martínez, B. J., Bernal-Morel, E., & Courel-Ibáñez, J. (2021). Post-COVID-19 Syndrome and the Potential Benefits of Exercise. International Journal of Environmental Research and Public Health, 18(10), 5329. https://doi.org/10.3390/ijerph18105329








