Contribution of Solid Food to Achieve Individual Nutritional Requirement during a Continuous 438 km Mountain Ultramarathon in Female Athlete
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Population
2.3. Mountain Ultramarathon Course
2.4. Running Pace Data Collection and Standardization
- E: the estimated running time of the segment (min);
- A: the actual running time of the segment (min).
2.5. Glucose Data Collection and Standardization
2.6. Diet Supply Data Collection
2.7. Statistics
3. Results
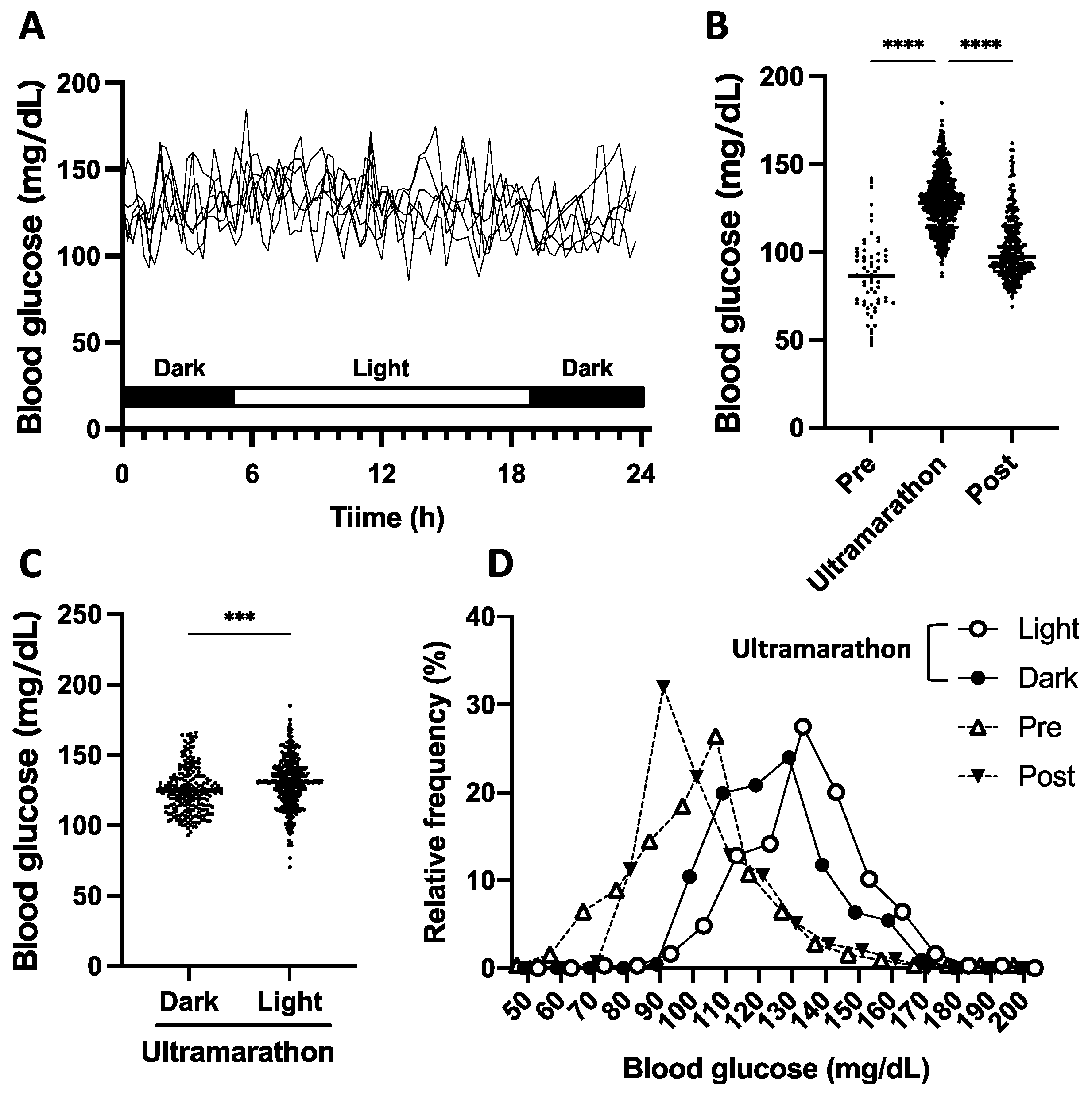
3.1. General Results of Blood Glucose Fluctuation during the Ultramarathon
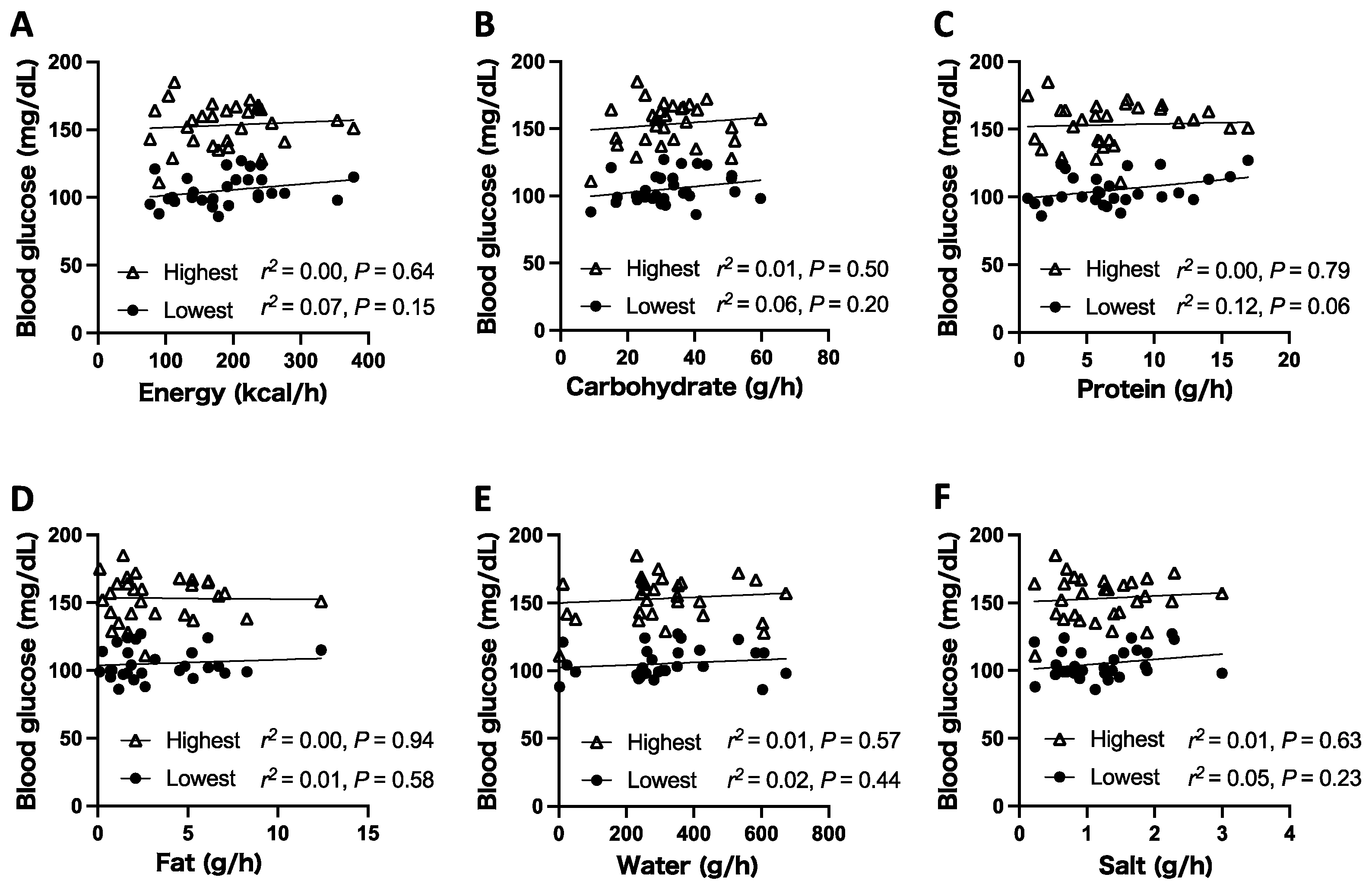
3.2. Relationship between the Amount of Nutrient Intake and Maintenance of Glucose Level during the Ultramarathon
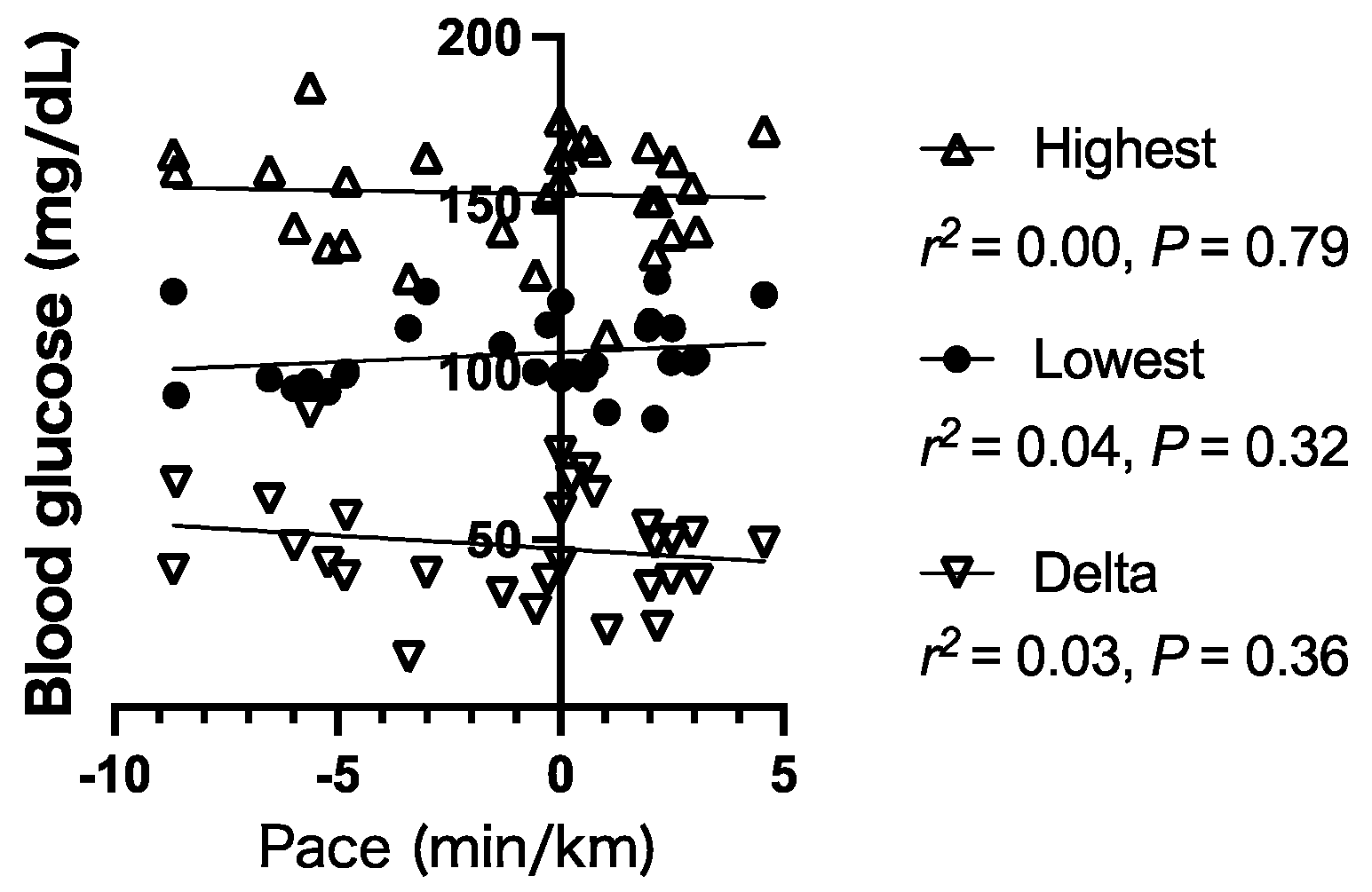
3.3. Relationship between Glucose Level and Running Pace
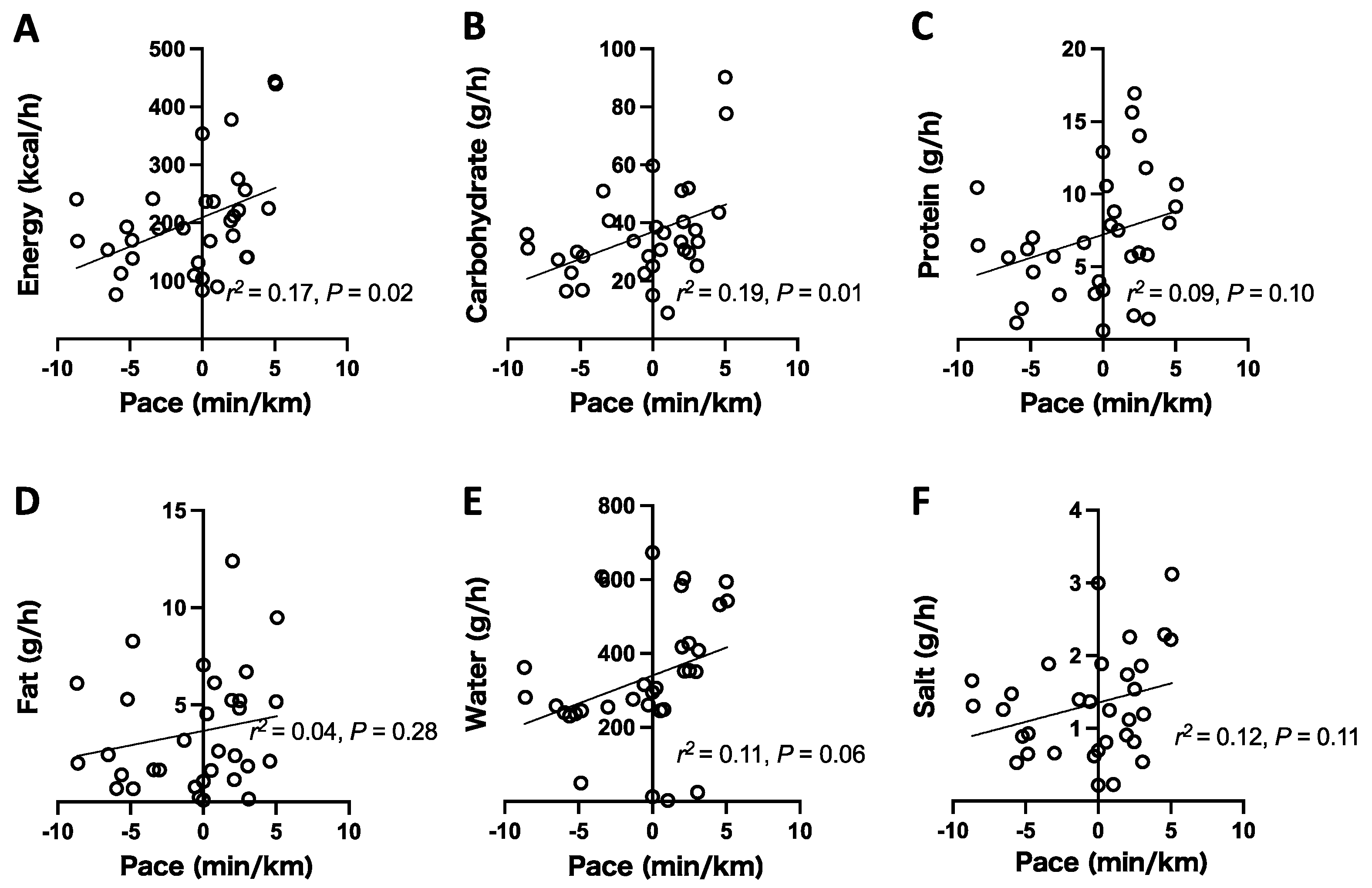
3.4. Relationship between the Amount of Nutrient Intake and Running Pace during the Ultramarathon
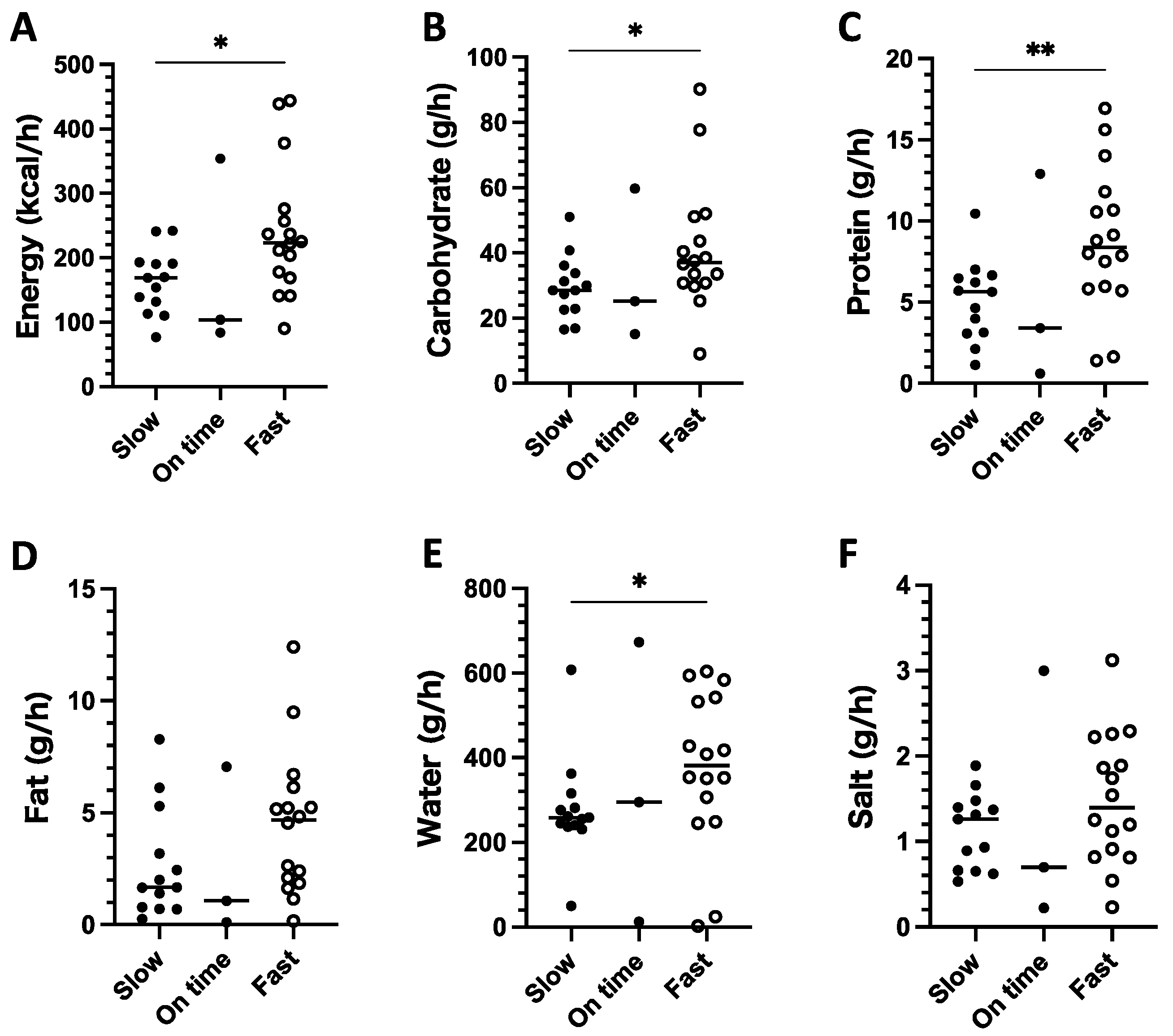
3.5. Comparison of Nutrient Intake between Fast and Slow Running Paces
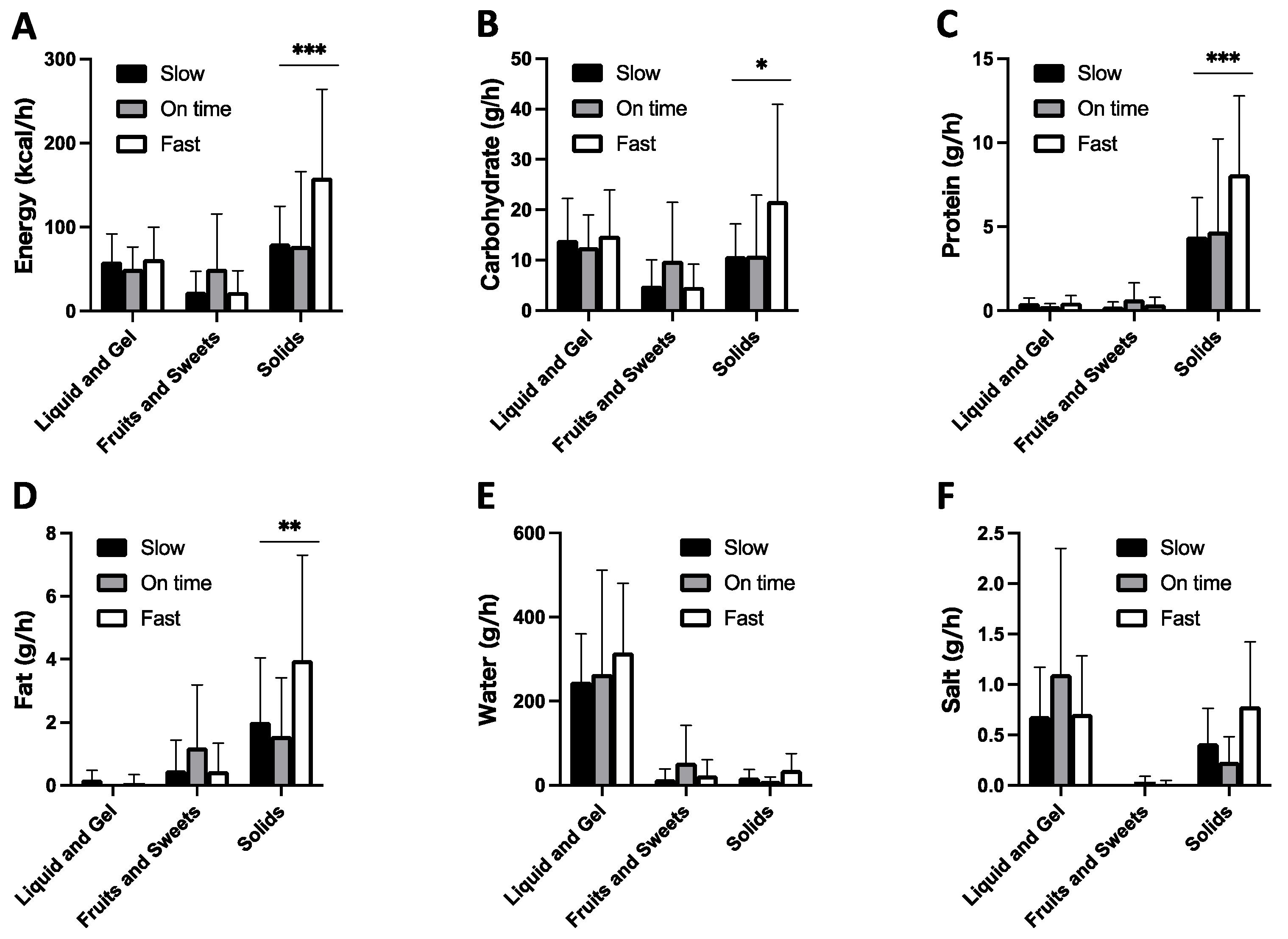
3.6. Comparison of Food Type between Fast and Slow Running Paces
3.7. Change of Serum Parameters Pre- and Post-Ultramarathon
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Stöhr, A.; Nikolaidi, P.T.; Villiger, E.; Sousa, C.V.; Scheer, V.; Hill, L.; Knechtle, B. An analysis of participation and performance of 2067 100-km ultra-marathons worldwide. Int. J. Environ. Res. Public Health 2021, 18, 362. [Google Scholar] [CrossRef]
- Arribalzaga, M.; Ruano, M.; Saiz, S. Review of the Food Guidelines in Continuous Ultramarathon. J. Nutr. Food Sci. 2017, 7. [Google Scholar] [CrossRef]
- Wilson, G.; Drust, B.; Morton, J.P.; Close, G.L. Weight-making Strategies in Professional Jockeys: Implications for Physical and Mental Health and Well-being. Sport. Med. 2014, 44, 785–796. [Google Scholar] [CrossRef]
- Rodriguez, N.N.R.; Di Marco, N.; Langley, S.; DiMarco, N.M. American College of Sports Medicine, American Dietetic Association, and Dietitians of Canada Joint Position Statement: Nutrition and Athletic Performance. Med. Sci. Sports Exerc. 2009, 41, 709–731. [Google Scholar] [CrossRef] [PubMed]
- Tiller, N.B.; Roberts, J.D.; Beasley, L.; Chapman, S.; Pinto, J.M.; Smith, L.; Wiffin, M.; Russell, M.; Sparks, S.A.; Duckworth, L.; et al. International Society of Sports Nutrition Position Stand: Nutritional Considerations for Single-stage Ultra-marathon Training and Racing. J. Int. Soc. Sports Nutr. 2019, 16, 1–23. [Google Scholar] [CrossRef]
- Costa, R.J.S.; Gill, S.K.; Hankey, J.; Wright, A.; Marczak, S. Perturbed Energy Balance and Hydration Status in Ultra-endurance Runners During a 24 h Ultra-marathon. Br. J. Nutr. 2014, 112, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Clark, H.R.; Barker, M.E.; Corfe, B.M. Nutritional Strategies of Mountain Marathon Competitors-An Observational Study. Int. J. Sport Nutr. Exerc. Metab. 2005, 15, 160–172. [Google Scholar] [CrossRef]
- Costa, R.J.S.; Knechtle, B.; Tarnopolsky, M.; Hoffman, M.D. Nutrition for Ultramarathon Running: Trail, Track, and Road. Int. J. Sport Nutr. Exerc. Metab. 2019, 29, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, M.D.; Stuempfle, K.J. Hydration Strategies, Weight Change and Performance in a 161 km Ultramarathon. Res. Sport. Med. 2014, 22, 213–225. [Google Scholar] [CrossRef]
- Zalcman, I.; Guarita, H.V.; Juzwiak, C.R.; Crispim, C.A.; Antunes, H.K.M.; Edwards, B.; Tufik, S.; de Mello, M.T. Nutritional Status of Adventure Racers. Nutrition 2007, 23, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Knechtle, B.; Nikolaidis, P.T. Physiology and Pathophysiology in Ultra-marathon Running. Front. Physiol. 2018, 9, 634. [Google Scholar] [CrossRef] [PubMed]
- Stuempfle, K.J.; Valentino, T.; Hew-Butler, T.; Hecht, F.M.; Hoffman, M.D. Nausea is associated with Endotoxemia during a 161-km Ultramarathon. J. Sports Sci. 2016, 34, 1662–1668. [Google Scholar] [CrossRef]
- Alcock, R.; McCubbin, A.; Camões-Costa, V.; Costa, R.J.S. Case Study: Providing Nutritional Support to an Ultraendurance Runner in Preparation for a Self-Sufficient Multistage Ultramarathon: Rationed Versus Full Energy Provisions. Wilderness Environ. Med. 2018, 29, 508–520. [Google Scholar] [CrossRef] [PubMed]
- Kerschan-Schindl, K.; Thalmann, M.M.; Weiss, E.; Tsironi, M.; Föger-Samwald, U.; Meinhart, J.; Skenderi, K.; Pietschmann, P. Changes in Serum Levels of Myokines and Wnt-Antagonists After an Ultramarathon Race. PLoS ONE 2015, 10, e0132478. [Google Scholar] [CrossRef]
- Edwards, K.H.; Elliott, B.T.; Kitic, C.M. Carbohydrate Intake and Ketosis in Self-sufficient Multi-stage Ultramarathon Runners. J. Sports Sci. 2019. [Google Scholar] [CrossRef] [PubMed]
- Seal, A.D.; Anastasiou, C.A.; Skenderi, K.P.; Echegaray, M.; Yiannakouris, N.; Tsekouras, Y.E.; Matalas, A.L.; Yannakoulia, M.; Pechlivani, F.; Kavouras, S.A. Incidence of Hyponatremia During a Continuous 246-km Ultramarathon Running Race. Front. Nutr. 2019, 6, 161. [Google Scholar] [CrossRef]
- David Cotter, J.; Gatterer, H.; Vernillo, G.; Savoldelli, A.; Skafidas, S.; Zignoli, A.; La Torre, A.; Pellegrini, B.; Giardini, G.; Trabucchi, P.; et al. An Extreme Mountain Ultra-Marathon Decreases the Cost of Uphill Walking and Running. Front. Physiol. 2016, 7, 530. [Google Scholar] [CrossRef][Green Version]
- Fallon, K.E.; Sivyer, G.; Sivyer, K.; Dare, A. The Biochemistry of Runners in a 1600 km Ultramarathon. Br. J. Sport. Med. 1999, 33, 264–269. [Google Scholar] [CrossRef]
- Abernethy, P.J.; Eden, B. Changes in Blood Glucose Levels during a 1005-km Running Race: A Case Study. Br. J. Sports Med. 1992, 26, 66–68. [Google Scholar] [CrossRef]
- Dempster, S.; Britton, R.; Murray, A.; Costa, R.J.S. Case Study: Nutrition and Hydration Status during 4,254 km of Running Over 78 Consecutive Days. Int. J. Sport Nutr. Exerc. Metab. 2013, 23, 533–541. [Google Scholar] [CrossRef]
- Rontoyannis, G.P.; Skoulis, T.; Pavlou, K.N. Energy Balance in Ultramarathon Running. Am. J. Clin. Nutr. 1989, 49, 976–979. [Google Scholar] [CrossRef]
- Costa, R.J.S.; Hoffman, M.D.; Stellingwerff, T. Considerations for Ultra-endurance Activities: Part 1- Nutrition. Res. Sport. Med. 2019. [Google Scholar] [CrossRef] [PubMed]
- Ishihara, K.; Uchiyama, N.; Kizaki, S.; Mori, E.; Nonaka, T.; Oneda, H. Application of Continuous Glucose Monitoring for Assessment of Individual Carbohydrate Requirement During Ultramarathon Race. Nutrients 2020, 12, 1121. [Google Scholar] [CrossRef] [PubMed]
- Nikolaidis, P.T.; Knechtle, B. Ultramarathon with Type 1 Diabetes. Praxis 2018, 107, 777–781. [Google Scholar] [CrossRef][Green Version]
- Sengoku, Y.; Nakamura, K.; Ogata, H.; Nabekura, Y.; Nagasaka, S.; Tokuyama, K. Continuous Glucose Monitoring During a 100-km Race: A Case Study in an Elite Ultramarathon Runner. Int. J. Sports Physiol. Perform. 2015, 10, 124–127. [Google Scholar] [CrossRef]
- Arakawa, K.; Hosono, A.; Shibata, K.; Ghadimi, R.; Fuku, M.; Goto, C.; Imaeda, N.; Tokudome, Y.; Hoshino, H.; Marumoto, M.; et al. Changes in blood biochemical markers before, during, and after a 2-day ultramarathon. Open Access J. Sport Med. 2016, 7, 43–50. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hoffman, M.D.; Ingwerson, J.L.; Rogers, I.R.; Hew-Butler, T.; Stuempfle, K.J. Increasing creatine kinase concentrations at the 161-km western states endurance run. Wilderness Environ. Med. 2012, 23, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Viribay, A.; Arribalzaga, S.; Mielgo-Ayuso, J.; Castañeda-Babarro, A.; Seco-Calvo, J.; Urdampilleta, A. Effects of 120 g/h of Carbohydrates Intake During a Mountain Marathon on Exercise-induced Muscle Damage in Elite Runners. Nutrients 2020, 12, 1367. [Google Scholar] [CrossRef]
- Urdampilleta, A.; Arribalzaga, S.; Viribay, A.; Castañeda-Babarro, A.; Seco-Calvo, J.; Mielgo-Ayuso, J. Effects of 120 vs. 60 and 90 g/h Carbohydrate Intake During a Trail Marathon on Neuromuscular Function and High Intensity Run Capacity Recovery. Nutrients 2020, 12, 2094. [Google Scholar] [CrossRef]
- Watson, A.M. Sleep and Athletic Performance. Curr. Sports Med. Rep. 2017, 16, 413–418. [Google Scholar] [CrossRef]
- Knutson, K.L.; Spiegel, K.; Penev, P.; Van Cauter, E. The metabolic consequences of sleep deprivation. Sleep Med. Rev. 2007, 11, 163–178. [Google Scholar] [CrossRef]
- Williamson, E. Nutritional Implications for Ultra-endurance Walking and Running Events. Extrem. Physiol. Med. 2016, 5, 1–18. [Google Scholar] [CrossRef]
- Pruitt, K.A.; Hill, J.M. Optimal Pacing and Carbohydrate Intake Strategies for Ultramarathons. Eur. J. Appl. Physiol. 2017, 117, 2527–2545. [Google Scholar] [CrossRef] [PubMed]
- Gawrecki, A.; Zozulinska-Ziolkiewicz, D.; Matejko, B.; Hohendorff, J.; Malecki, M.T.; Klupa, T. Safe Completion of a Trail Running Ultramarathon by Four Men with Type 1 Diabetes. Diabetes Technol. Ther. 2018, 20, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Kordonouri, O.; Vazeou, A.; Scharf, M.; Würsig, M.; Battelino, T. Striving for Control: Lessons Learned from a Successful International Type 1 Diabetes Youth Challenge. Acta Diabetol. 2017, 54, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Belli, T.; de Macedo, D.V.; Scariot, P.P.M.; de Araújo, G.G.; dos Reis, I.G.M.; Lazarim, F.L.; Nunes, L.A.S.; Brenzikofer, R.; Gobatto, C.A. Glycemic Control and Muscle Damage in 3 Athletes with Type 1 Diabetes During a Successful Performance in a Relay Ultramarathon: A Case Report. Wilderness Environ. Med. 2017, 28, 239–245. [Google Scholar] [CrossRef]
- Emig, T.; Peltonen, J. Human Running Performance from Real-world Big Data. Nat. Commun. 2020, 11, 1–9. [Google Scholar] [CrossRef]
- Al Hayek, A.A.; Robert, A.A.; Al Dawish, M.A. Evaluation of FreeStyle Libre Flash Glucose Monitoring System on Glycemic Control, Health-Related Quality of Life, and Fear of Hypoglycemia in Patients with Type 1 Diabetes. Clin. Med. Insights Endocrinol. Diabetes 2017, 10. [Google Scholar] [CrossRef]
- Fokkert, M.J.; Van Dijk, P.R.; Edens, M.A.; Abbes, S.; De Jong, D.; Slingerland, R.J.; Bilo, H.J.G. Performance of the Freestyle Libre Flash Glucose Monitoring System in Patients with Type 1 and 2 Diabetes Mellitus. BMJ Open Diabetes Res. Care 2017, 5. [Google Scholar] [CrossRef]
- Fokkert, M.J.; Damman, A.; Van Dijk, P.R.; Edens, M.A.; Abbes, S.; Braakman, J.; Slingerland, R.J.; Dikkeschei, L.D.; Dille, J.; Bilo, H.J.G. Use of FreeStyle Libre Flash Monitor Register in the Netherlands (FLARE-NL1): Patient Experiences, Satisfaction, and Cost Analysis. Int. J. Endocrinol. 2019, 2019. [Google Scholar] [CrossRef]
- Aberer, F.; Hajnsek, M.; Rumpler, M.; Zenz, S.; Baumann, P.M.; Elsayed, H.; Puffing, A.; Treiber, G.; Pieber, T.R.; Sourij, H.; et al. Evaluation of subcutaneous glucose monitoring systems under routine environmental conditions in patients with type 1 diabetes. Diabetes. Obes. Metab. 2017, 19, 1051–1055. [Google Scholar] [CrossRef] [PubMed]
- Moser, O.; Mader, J.K.; Tschakert, G.; Mueller, A.; Groeschl, W.; Pieber, T.R.; Koehler, G.; Messerschmidt, J.; Hofmann, P. Accuracy of continuous glucose monitoring (CGM) during continuous and high-intensity interval exercise in patients with type 1 diabetes mellitus. Nutrients 2016, 8, 489. [Google Scholar] [CrossRef]
- Pfeiffer, B.; Stellingwerff, T.; Zaltas, E.; Jeukendrup, A.E.; Pfeeffer, B.; Stellingwerff, T.; Zaltas, E.; Jeukendrup, A.E.; Pfeiffer, A.B.; Stellingwertf, T.; et al. Oxidation of Solid Versus Liquid CHO Sources During Exercise. Med. Sci. Sports Exerc. 2010, 42, 2030–2037. [Google Scholar] [CrossRef] [PubMed]
- Office for Resources, P.D.S.T. P.B. Standard Tables of Food Composition in Japan-2015-(Seventh Revised Version); Ministry of Education, Culture, Sports, Science and Technology: Tokyo, Japan, 2015.
- Arnaoutis, G.; Leveritt, M.; Wardenaar, F.C.; Hoogervorst, D.; Versteegen, J.J.; Van Der Burg, N.; Lambrechtse, K.J.; Bongers, C.C.W.G. Real-Time Observations of Food and Fluid Timing During a 120 km Ultramarathon. Front. Nutr. 2018, 5, 32. [Google Scholar] [CrossRef]
- Wardenaar, F.C.; Dijkhuizen, R.; Ceelen, I.J.M.; Jonk, E.; de Vries, J.H.M.; Witkamp, R.F.; Mensink, M. Nutrient Intake by Ultramarathon Runners: Can They Meet Recommendations? Int. J. Sport Nutr. Exerc. Metab. 2015, 25, 375–386. [Google Scholar] [CrossRef] [PubMed]
- Stuempfle, K.J.; Hoffman, M.D.; Weschler, L.B.; Rogers, I.R.; Hew-Butler, T. Race Diet of Finishers and Non-finishers in a 100 Mile (161 km) Mountain Footrace. J. Am. Coll. Nutr. 2011, 30, 529–535. [Google Scholar] [CrossRef]
- Martinez, S.; Aguilo, A.; Rodas, L.; Lozano, L.; Moreno, C.; Tauler, P. Energy, Macronutrient and Water Intake During a Mountain Ultramarathon Event: The Influence of Distance. J. Sports Sci. 2018, 36, 333–339. [Google Scholar] [CrossRef]
- Hill, R.J.; Davies, P.S. Energy Expenditure during 2 wk of an Ultra-endurance Run around Australia. Med. Sci. Sports Exerc. 2001, 33, 148–151. [Google Scholar] [CrossRef]
- Dumke, C.L.; Shooter, L.; Lind, R.H.; Nieman, D.C. Indirect Calorimetry during Ultradistance Running: A Case Report. J. Sports Sci. Med. 2006, 5, 692–698. [Google Scholar]
- Webster, C.C.; Noakes, T.D.; Chacko, S.K.; Swart, J.; Kohn, T.A.; Smith, J.A.H. Gluconeogenesis During Endurance Exercise in Cyclists Habituated to a Long-term Low Carbohydrate High-fat Diet. Authors. J. Physiol. C 2016, 594, 4389–4405. [Google Scholar] [CrossRef]
- Emhoff, C.A.W.; Messonnier, L.A.; Horning, M.A.; Fattor, J.A.; Carlson, T.J.; Brooks, G.A. Gluconeogenesis and Hepatic Glycogenolysis During Exercise at the Lactate Threshold. J. Appl. Physiol. 2013, 114, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Robergs, R.A.; Mcminn, S.B.; Mermier, C.; Hi, G.L.; Ruby, B.; Quinn, C. Blood Glucose and Glucoregulatory Hormone Responses to Solid and Liquid Carbohydrate Ingestion during Exercise. Int. J. Oí Sport Nutr. 1998, 8, 70–83. [Google Scholar] [CrossRef]
- Peracchi, M.; Santangelo, A.; Conte, D.; Fraquelli, M.; Tagliabue, R.; Gebbia, C.; Porrini, M. The Physical State of a Meal Affects Hormone Release and Postprandial Thermogenesis. Br. J. Nutr. 2000, 83, 623–628. [Google Scholar] [CrossRef][Green Version]
- Mattin, L.R.; Yau, A.M.W.; McIver, V.; James, L.J.; Evans, G.H. The Effect of Exercise Intensity on Gastric Emptying Rate, Appetite and Gut Derived Hormone Responses After Consuming a Standardised Semi-Solid Meal in Healthy Males. Nutrients 2018, 10, 787. [Google Scholar] [CrossRef] [PubMed]
- Ishihara, K.; Taniguchi, H.; Akiyama, N.; Asami, Y. Easy to Swallow Rice Cake as a Carbohydrate Source During Endurance Exercise Suppressed Feelings of Thirst and Hunger Without Changing Exercise Performance. Nutr. Sci. Vitaminol. 2020, 66, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Mears, S.A.; Boxer, B.; Sheldon, D.; Wardley, H.; Tarnowski, C.A.; James, L.J.; Hulston, C.J. Sports Drink Intake Pattern Affects Exogenous Carbohydrate Oxidation During Running. Med. Sci. Sports Exerc. 2020, 52, 1976–1982. [Google Scholar] [CrossRef]
- Geer, E.B.; Shen, W. Gender Differences in Insulin Resistance, Body Composition, and Energy Balance. Gend. Med. 2009, 6, 60–75. [Google Scholar] [CrossRef]
- Wismann, J.; Willoughby, D. Gender Differences in Carbohydrate Metabolism and Carbohydrate Loading. J. Int. Soc. Sports Nutr. 2006, 3, 28–32. [Google Scholar] [CrossRef]
- Soeters, M.R.; Sauerwein, H.P.; Groener, J.E.; Aerts, J.M.; Ackermans, M.T.; Glatz, J.F.C.; Fliers, E.; Serlie, M.J. Gender-related Differences in the Metabolic Response to Fasting. J. Clin. Endocrinol. Metab. 2007, 92, 3646–3652. [Google Scholar] [CrossRef]
- Keramida, G.; Peters, A.M. Fasting Hepatic Glucose Uptake is Higher in Men than Women. Physiol. Rep. 2017, 5. [Google Scholar] [CrossRef]
- Scheen, A.J.; Byrne, M.M.; Plat, L.; Leproult, R.; Van Cauter, E. Relationships between sleep quality and glucose regulation in normal humans. Am. J. Physiol. Endocrinol. Metab. 1996, 271. [Google Scholar] [CrossRef]
- Shapiro, E.T.; Polonsky, K.S.; Copinschi, G.; Bosson, D.; Tillil, H.; Blackman, J.; Lewis, G.; Van Cauter, E. Nocturnal elevation of glucose levels during fasting in noninsulin-dependent diabetes. J. Clin. Endocrinol. Metab. 1991, 72, 444–454. [Google Scholar] [CrossRef]
- Cauter, E.; Van Désir, D.; Decoster, C.; Féry, F.; Balasse, E.O. Nocturnal Decrease in Glucose Tolerance During Constant Glucose Infusion. J. Clin. Endocrinol. Metab. 1989, 69, 604–611. [Google Scholar] [CrossRef]
- Spiegel, K.; Leproult, R.; Van Cauter, E. Impact of sleep debt on metabolic and endocrine function. Lancet 1999, 354, 1435–1439. [Google Scholar] [CrossRef]
- Van Helder, T.; Radoki, M.W. Sleep Deprivation and the Effect on Exercise Performance. Sports Med. 1989, 7, 235–247. [Google Scholar] [CrossRef]
- Martin, B.J. Effect of sleep deprivation on tolerance of prolonged exercise. Eur. J. Appl. Physiol. Occup. Physiol. 1981, 47, 345–354. [Google Scholar] [CrossRef]
- Symons, J.D.; Vanhelder, T.; Myles, W.S. Physical performance and physiological responses following 60 hours of sleep deprivation. Med. Sci. Sports Exerc. 1988, 20, 374–380. [Google Scholar] [CrossRef]
- Janosky, J.E. Use of the single subject design for practice based primary care research. Postgrad. Med. J. 2005, 81, 549–551. [Google Scholar] [CrossRef]
- Bobrovitz, C.D.; Ottenbacher, K.J. Comparison of visual inspection and statistical analysis of single- subject data in rehabilitation research. Am. J. Phys. Med. Rehabil. 1998, 77, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Zhan, S.; Ottenbacher, K.J. Single subject research designs for disability research. Disabil. Rehabil. 2001, 23, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H. N of 1 Randomized Trials for Investigating New Drugs. In Drug Epidemiology and Post-Marketing Surveillance; Springer: Boston, MA, USA, 1992; pp. 125–134. [Google Scholar] [CrossRef]
- McReynolds, L.V.; Thompson, C.K. Flexibility of single-subject experimental designs. Part I: Review of the basics of single-subject designs. J. Speech Hear. Disord. 1986, 51, 194–203. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, T.F. An Examination and Evaluation of Single Subject Designs Used in Behavior Analysis Research in School Settings. Educ. Res. Q. 1983, 7, 35–42. [Google Scholar]
- Brown, J.S.; Connolly, D.A. Selected Human Physiological Responses during Extreme Heat: The Badwater Ultramarathon. J. Strength Cond. Res. 2015, 29, 1729–1736. [Google Scholar] [CrossRef][Green Version]
- Paton, C.D.; Hopkins, W.G. Tests of Cycling Performance. Sport. Med. 2001, 31, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Moser, O.; Eckstein, M.L.; Mueller, A.; Birnbaumer, P.; Aberer, F.; Koehler, G.; Sourij, C.; Kojzar, H.; Holler, P.; Simi, H.; et al. Impact of physical exercise on sensor performance of the FreeStyle Libre intermittently viewed continuous glucose monitoring system in people with Type 1 diabetes: A randomized crossover trial. Diabet. Med. 2019, 36, 606–611. [Google Scholar] [CrossRef] [PubMed]
| Energy, kcal | Protein, g | Fat, g | Carbohydrate, g | Water, g | Salt, g | |
|---|---|---|---|---|---|---|
| Liquids and gels | 7999 | 56 | 17 | 1911 | 35,018 | 84 |
| Sports drink | 1467 | 5 | 0 | 377 | 6617 | 13 |
| Cola | 87 | 0 | 0 | 21 | 103 | 0 |
| Gel | 1255 | 9 | 0 | 306 | 208 | 44 |
| Milk product | 3324 | 32 | 6 | 772 | 11,889 | 8 |
| Tea | 1120 | 0 | 0 | 284 | 14,216 | 14 |
| Soup | 331 | 8 | 11 | 48 | 683 | 5 |
| Other liquids | 415 | 2 | 0 | 103 | 1302 | 0 |
| Fruits and sweets | 3075 | 37 | 59 | 657 | 2329 | 1 |
| Fruit | 1122 | 14 | 3 | 291 | 1601 | 0 |
| Sweet | 1953 | 23 | 56 | 366 | 728 | 1 |
| Solids | 15,516 | 815 | 409 | 2047 | 3579 | 77 |
| Bar | 3120 | 260 | 91 | 312 | 78 | 8 |
| Noodle | 1678 | 53 | 29 | 287 | 1069 | 17 |
| Rice product | 5334 | 125 | 91 | 965 | 1050 | 32 |
| Wheat product | 2164 | 74 | 88 | 250 | 463 | 7 |
| Powder | 651 | 119 | 7 | 22 | 12 | 2 |
| Other solids | 2569 | 184 | 103 | 211 | 907 | 11 |
| Total | 26,590 | 908 | 485 | 4615 | 40,926 | 162 |
| Average per hour | 171 | 5.9 | 3.1 | 29.7 | 263 | 1.1 |
| Normal Range | Ultramarathon | |||
|---|---|---|---|---|
| Mean (95% CI) | Pre | Post | ||
| Total protein | g/dL | 7.4 ± 0.5 (6.5–8.3) | 6.9 | 5.8 L |
| Triglyceride | mg/dL | 89.5 ± 30.4 (30.0–149.0) | 140 | 33 |
| LDL-cholesterol | mg/dL | 104.5 ± 17.6 (70.0–139.0) | 65 L | 42 L |
| AST | U/L | 23.0 ± 7.7 (8.0–38.0) | 31 | 87 H |
| ALT | U/L | 23.5 ± 9.9 (4.0–43.0) | 35 | 84 H |
| ALP | U/L | 232.0 ± 62.2 (110.0–354.0) | 258 | 70 L |
| LDH | U/L | 183.0 ± 31.6 (121.0–245.0) | 184 | 428 H |
| CK | U/L | 117.0 ± 40.3 (38.0–196.0) | 102 | 1312 H |
| Na | mEq/L | 142.5 ± 3.8 (135.0–150.0) | 137 | 142 |
| Cl | mEq/L | 104.0 ± 3.1 (98.0–110.0) | 101 | 106 |
| K | mEq/L | 4.4 ± 0.5 (3.5–5.3) | 4.7 | 3.9 |
| Ca | mg/dL | 9.3 ± 0.5 (8.4–10.2) | 9 | 8.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ishihara, K.; Inamura, N.; Tani, A.; Shima, D.; Kuramochi, A.; Nonaka, T.; Oneda, H.; Nakamura, Y. Contribution of Solid Food to Achieve Individual Nutritional Requirement during a Continuous 438 km Mountain Ultramarathon in Female Athlete. Int. J. Environ. Res. Public Health 2021, 18, 5153. https://doi.org/10.3390/ijerph18105153
Ishihara K, Inamura N, Tani A, Shima D, Kuramochi A, Nonaka T, Oneda H, Nakamura Y. Contribution of Solid Food to Achieve Individual Nutritional Requirement during a Continuous 438 km Mountain Ultramarathon in Female Athlete. International Journal of Environmental Research and Public Health. 2021; 18(10):5153. https://doi.org/10.3390/ijerph18105153
Chicago/Turabian StyleIshihara, Kengo, Naho Inamura, Asuka Tani, Daisuke Shima, Ai Kuramochi, Tsutomu Nonaka, Hiroshi Oneda, and Yasuyuki Nakamura. 2021. "Contribution of Solid Food to Achieve Individual Nutritional Requirement during a Continuous 438 km Mountain Ultramarathon in Female Athlete" International Journal of Environmental Research and Public Health 18, no. 10: 5153. https://doi.org/10.3390/ijerph18105153
APA StyleIshihara, K., Inamura, N., Tani, A., Shima, D., Kuramochi, A., Nonaka, T., Oneda, H., & Nakamura, Y. (2021). Contribution of Solid Food to Achieve Individual Nutritional Requirement during a Continuous 438 km Mountain Ultramarathon in Female Athlete. International Journal of Environmental Research and Public Health, 18(10), 5153. https://doi.org/10.3390/ijerph18105153







