Association between Antidepressant Treatment during Pregnancy and Postpartum Self-Harm Ideation in Women with Psychiatric Disorders: A Cross-Sectional, Multinational Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Postpartum Self-Harm Ideation
2.2. Self-Reported Depression and Anxiety, and Antidepressant Treatment
2.3. Covariates
2.4. Data Analysis
2.5. Sensitivity Analyses
2.6. Ethics Approval and Informed Consent
3. Results
4. Discussion
Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cooper, W.O.; Willy, M.E.; Pont, S.J.; Ray, W.A. Increasing use of antidepressants in pregnancy. Am. J. Obstet. Gynecol. 2007, 196, 544.e1–544.e5. [Google Scholar] [CrossRef]
- Zoega, H.; Kieler, H.; Nørgaard, M.; Furu, K.; Valdimarsdottir, U.A.; Brandt, L.; Haglund, B. Use of SSRI and SNRI Antidepressants during Pregnancy: A Population-Based Study from Denmark, Iceland, Norway and Sweden. PLoS ONE 2015, 10, e0144474. [Google Scholar] [CrossRef]
- Molenaar, N.M.; Bais, B.; Berg, M.P.L.-V.D.; Mulder, C.L.; Howell, E.A.; Fox, N.S.; Rommel, A.-S.; Bergink, V.; Kamperman, A.M. The international prevalence of antidepressant use before, during, and after pregnancy: A systematic review and meta-analysis of timing, type of prescriptions and geographical variability. J. Affect. Disord. 2020, 264, 82–89. [Google Scholar] [CrossRef]
- Freeman, M.P.; Claypoole, L.D.; Burt, V.K.; Sosinsky, A.Z.; Moustafa, D.; Noe, O.B.; Cheng, L.J.; Cohen, L. Course of major depressive disorder after pregnancy and the postpartum period. Depress. Anxiety 2018, 35, 1130–1136. [Google Scholar] [CrossRef] [PubMed]
- Howard, L.M.; Molyneaux, E.; Dennis, C.-L.; Rochat, T.; Stein, A.; Milgrom, J. Non-Psychotic mental disorders in the perinatal period. Lancet 2014, 384, 1775–1788. [Google Scholar] [CrossRef]
- Sharma, V. A closer look at the preventative effect of antidepressants in postpartum depression. Arch. Womens Ment. Heal. 2017, 20, 357–360. [Google Scholar] [CrossRef] [PubMed]
- Molyneaux, E.; A Telesia, L.; Henshaw, C.; Boath, E.; Bradley, E.; Howard, L.M. Antidepressants for preventing postnatal depression. Cochrane Database Syst. Rev. 2018, 4, CD004363. [Google Scholar] [CrossRef] [PubMed]
- Hawton, K.; Zahl, D.; Weatherall, R. Weatherall, Suicide following deliberate self-harm: Long-Term follow-Up of patients who presented to a general hospital. Br. J. Psychiatry 2003, 182, 537–542. [Google Scholar] [CrossRef] [PubMed]
- Zetterqvist, M. The DSM-5 diagnosis of nonsuicidal self-Injury disorder: A review of the empirical literature. Child Adolesc. Psychiatry Ment. Heal. 2015, 9, 31. [Google Scholar] [CrossRef]
- Ayre, K.; Gordon, H.G.; Dutta, R.; Hodsoll, J.; Howard, L.M. The Prevalence and Correlates of Self-Harm in the Perinatal Period: A Systematic Review. J. Clin. Psychiatry 2019, 81, 19r12773. [Google Scholar] [CrossRef]
- Kim, J.J.; La Porte, L.M.; Saleh, M.P.; Allweiss, S.; Adams, M.; Zhou, Y.; Silver, R.K. Suicide risk among perinatal women who report thoughts of self-Harm on depression screens. Obstet. Gynecol. 2015, 125, 885–893. [Google Scholar] [CrossRef] [PubMed]
- Knight, M.; Bunch, K.; Tuffnell, D.; Shakespeare, J.; Kotnis, R.; Kenyon, S.; Kurinczuk, J.J.; on behalf of MBRRACE-UK (Eds.) Saving Lives, Improving Mothers’ Care-Lessons Learned to Inform Maternity Care from the UK and Ireland Confidential Enquiries into Maternal Deaths and Morbidity 2015-17; National Perinatal Epidemiology Unit, University of Oxford: Oxford, UK, 2019. [Google Scholar]
- Mangla, K.; Hoffman, M.C.; Trumpff, C.; O’Grady, S.; Monk, C. Maternal self-Harm deaths: An unrecognized and preventable outcome. Am. J. Obstet. Gynecol. 2019, 221, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Van Heeringen, K.; Mann, J.J. The neurobiology of suicide. Lancet. Psychiatry 2014, 1, 63–72. [Google Scholar] [CrossRef]
- Barbui, C.; Esposito, E.; Cipriani, A. Selective serotonin reuptake inhibitors and risk of suicide: A systematic review of observational studies. CMAJ 2009, 180, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Hawton, K.; Witt, K.G.; Salisbury, T.L.T.; Arensman, E.; Gunnell, D.; Hazell, P.; Townsend, E.; Heeringen, K.V. Pharmacological interventions for self-Harm in adults. Cochrane Database Syst. Rev. 2015, 6, CD011777. [Google Scholar] [CrossRef] [PubMed]
- Cipriani, A.; Barbui, C.; Geddes, J.R. Suicide, depression, and antidepressants. BMJ 2005, 330, 373–374. [Google Scholar] [CrossRef]
- Noma, H.; A Furukawa, T.; Maruo, K.; Imai, H.; Shinohara, K.; Tanaka, S.; Ikeda, K.; Yamawaki, S.; Cipriani, A. Exploratory analyses of effect modifiers in the antidepressant treatment of major depression: Individual-participant data meta-Analysis of 2803 participants in seven placebo-Controlled randomized trials. J. Affect. Disord. 2019, 250, 419–424. [Google Scholar] [CrossRef]
- Miller, M.; Pate, V.; Swanson, S.A.; Azrael, D.; White, A.; Stürmer, T. Antidepressant class, age, and the risk of deliberate self-Harm: A propensity score matched cohort study of SSRI and SNRI users in the USA. CNS Drugs 2014, 28, 79–88. [Google Scholar] [CrossRef][Green Version]
- Miller, M.; Swanson, S.A.; Azrael, D.; Pate, M.V.; Stürmer, T. Antidepressant dose, age, and the risk of deliberate self-Harm. JAMA Intern. Med. 2014, 174, 899–909. [Google Scholar] [CrossRef]
- Bayrampour, H.; Kapoor, A.; Bunka, M.; Ryan, D. The Risk of Relapse of Depression During Pregnancy After Discontinuation of Antidepressants: A Systematic Review and Meta-Analysis. J. Clin. Psychiatry 2020, 81, 19r13134. [Google Scholar] [CrossRef]
- Swanson, S.A.; Hernandez-Diaz, S.; Palmsten, K.; Mogun, H.; Olfson, M.; Huybrechts, K.F. Methodological considerations in assessing the effectiveness of antidepressant medication continuation during pregnancy using administrative data. Pharmacoepidemiol. Drug Saf. 2015, 24, 934–942. [Google Scholar] [CrossRef] [PubMed]
- Lindahl, V.; Pearson, J.L.; Colpe, L. Prevalence of suicidality during pregnancy and the postpartum. Arch. Womens Ment. Heal. 2005, 8, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Molenaar, N.M.; Kamperman, A.M.; Boyce, P.; Bergink, V. Guidelines on treatment of perinatal depression with antidepressants: An international review. Aust. N. Z. J. Psychiatry 2018, 52, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Lupattelli, A.; Spigset, O.; Twigg, M.J.; Zagorodnikova, K.; Mardby, A.C.; Moretti, M.E.; Drozd, M.; Panchaud, A.; Hameen-Anttila, K.; Rieutord, A.; et al. Medication use in pregnancy: A cross-Sectional, multinational web-Based study. BMJ Open 2014, 4, e004365. [Google Scholar] [CrossRef]
- Wood, M.E.; Lapane, K.L.; Van Gelder, M.M.; Rai, D.; Nordeng, H.M. Making fair comparisons in pregnancy medication safety studies: An. overview of advanced methods for confounding control. Pharmacoepidemiol. Drug Saf. 2018, 27, 140–147. [Google Scholar] [CrossRef]
- Khalifeh, H.; Hunt, I.M.; Appleby, L.; Howard, L.M. Suicide in perinatal and non-Perinatal women in contact with psychiatric services: 15 year findings from a UK national inquiry. Lancet Psychiatry 2016, 3, 233–242. [Google Scholar] [CrossRef]
- Cox, J.L.; Holden, J.M.; Sagovsky, R. Detection of Postnatal Depression: Development of the 10-Item Edinburgh Postnatal Depression Scale. Br. J. Psychiatry 1987, 150, 782–786. [Google Scholar] [CrossRef]
- Cox, J.L.; Holden, J. Perinatal Mental Health: A Guide to the Edinburgh Postnatal Depression Scale (EPDS); RCPsych Publications: London, UK, 2003. [Google Scholar]
- Wisner, K.L.; Sit, D.K.Y.; McShea, M.C.; Rizzo, D.M.; Zoretich, R.A.; Hughes, C.L.; Eng, H.F.; Luther, J.F.; Wisniewski, S.R.; Costantino, M.L.; et al. Onset timing, thoughts of self-Harm, and diagnoses in postpartum women with screen-Positive depression findings. JAMA Psychiatry 2013, 70, 490–498. [Google Scholar] [CrossRef]
- Pope, C.J.; Xie, B.; Sharma, V.; Campbell, M.K. A prospective study of thoughts of self-Harm and suicidal ideation during the postpartum period in women with mood disorders. Arch. Womens Ment. Heal. 2013, 16, 483–488. [Google Scholar] [CrossRef]
- Hendrick, V. Psychiatric Disorders in Pregnancy and the Postpartum: Principles and Treatment; Humana Press: Totowa, NJ, USA, 2006. [Google Scholar]
- John, O.P.; Srivastava, S. Handbook of personality: Theory and research. In The Big Five Trait Taxonomy: History, Measurement, and Theoretical Perspectives; Pervin, I.L.A., John, O.P., Eds.; Guilford: New York, NY, USA, 1999; pp. 102–138. [Google Scholar]
- Fanous, A.; Gardner, C.O.; Prescott, C.A.; Cancro, R.; Kendler, K.S. Neuroticism, major depression and gender: A population-based twin study. Psychol. Med. 2002, 32, 719–728. [Google Scholar] [CrossRef]
- Eurostat. Population by Educational Attainment Level, Sex and Age (%-Main Indicators. 2012. Available online: http://ec.europa.eu/eurostat/web/products-datasets/-/edat_lfse_03 (accessed on 14 August 2017).
- Statistics Canada. Births. 2010. Available online: http://www.statcan.gc.ca/daily-quotidien/111220/dq111220g-eng.htm (accessed on 12 November 2012).
- Australian Bureau of Statistics. Births, Australia. 2011. Available online: http://www.abs.gov.au/AUSSTATS/abs@.nsf/mf/3301.0 (accessed on 13 November 2012).
- Hamilton, B.; Martin, J.; Ventura, S. National Vital Statistics Reports. Births: Preliminary Data for 2010; Department of health and human services. Centers for Disease Control and Prevention. National Center for Health Statistics. National Vital Statistics System: Hyattsville, MD, USA, 2011. [Google Scholar]
- Applied Survey Methods-A Statistical Perspective. 2017. Available online: http://applied-survey-methods.com/weight.html (accessed on 14 August 2017).
- Dugoff, E.H.; Schuler, M.; Stuart, E.A. Generalizing observational study results: Applying propensity score methods to complex surveys. Heal. Serv. Res. 2014, 49, 284–303. [Google Scholar] [CrossRef] [PubMed]
- Austin, P.C.; Stuart, E.A. Moving towards best practice when using inverse probability of treatment weighting (IPTW) using the propensity score to estimate causal treatment effects in observational studies. Stat. Med. 2015, 34, 3661–3679. [Google Scholar] [CrossRef] [PubMed]
- Zou, G.Y. A modified poisson regression approach to prospective studies with binary data. Am. J. Epidemiol. 2004, 159, 702–706. [Google Scholar] [CrossRef] [PubMed]
- Rubin, D.B. Multiple Imputation for Nonresponse in Surveys; Wiley Series in Probability and Mathematical Statistics; Applied Probability and Statistics; Wiley: New York, NY, USA, 1987. [Google Scholar]
- Lupattelli, A.; Twigg, M.J.; Zagorodnikova, K.; Moretti, M.; Drozd, M.; Panchaud, A.; Rieutord, A.; Juraski, R.G.; Odalovic, M.; Kennedy, D.; et al. Self-Reported perinatal depressive symptoms and postnatal symptom severity after treatment with antidepressants in pregnancy: A cross-Sectional study across 12 European countries using the Edinburgh Postnatal Depression Scale. Clin. Epidemiol. 2018, 10, 655–669. [Google Scholar] [CrossRef] [PubMed]
- Friedman, R.A.; Leon, A.C. Expanding the black box-depression, antidepressants, and the risk of suicide. N. Engl. J. Med. 2007, 356, 2343–2346. [Google Scholar] [CrossRef] [PubMed]
- Leon, A.C. The revised warning for antidepressants and suicidality: Unveiling the black box of statistical analyses. Am. J. Psychiatry 2007, 164, 786–789. [Google Scholar] [CrossRef] [PubMed]
- Fournier, J.C.; DeRubeis, R.J.; Hollon, S.D.; Dimidjian, S.; Amsterdam, J.D.; Shelton, R.C.; Fawcett, J. Antidepressant drug effects and depression severity: A patient-level meta-Analysis. JAMA 2010, 303, 47–53. [Google Scholar] [CrossRef]
- Mulder, R.; Joyce, P.R.; Frampton, C.M.A.; Luty, S.E. Antidepressant treatment is associated with a reduction in suicidal ideation and suicide attempts. Acta Psychiatr. Scand. 2008, 118, 116–122. [Google Scholar] [CrossRef]
- Cipriani, A.; A Furukawa, T.; Salanti, G.; Chaimani, A.; Atkinson, L.Z.; Ogawa, Y.; Leucht, S.; Ruhe, H.G.; Turner, E.H.; Higgins, J.P.T.; et al. Comparative efficacy and acceptability of 21 antidepressant drugs for the acute treatment of adults with major depressive disorder: A systematic review and network meta-Analysis. Lancet 2018, 391, 1357–1366. [Google Scholar] [CrossRef]
- Petersen, I.; Gilbert, R.E.; Evans, S.J.W.; Man, S.-L.; Nazareth, I. Pregnancy as a major determinant for discontinuation of antidepressants: An analysis of data from The Health Improvement Network. J. Clin. Psychiatry 2011, 72, 979–985. [Google Scholar] [CrossRef]
- Valuck, R.J.; Orton, H.D.; Libby, A.M. Antidepressant discontinuation and risk of suicide attempt: A retrospective, nested case-Control study. J. Clin. Psychiatry 2009, 70, 1069–1077. [Google Scholar] [CrossRef] [PubMed]
- Biffi, A.; Rea, F.; Locatelli, A.; Cetin, I.; Filippelli, A.; Corrao, G. Misleading meta-analyses of observational studies may generate unjustified alarms: The case of medications for nausea and vomiting in pregnancy. Pharmacol. Res. 2020, 105229. [Google Scholar] [CrossRef] [PubMed]
- Rothman, K.J.; Greenland, S.; Lash, T.L. Modern Epidemiology; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2008. [Google Scholar]
- Ekman, A.; Dickman, P.W.; Klint, Å.; Weiderpass, E.; Litton, J.-E. Feasibility of Using Web-Based Questionnaires in Large Population-based Epidemiological Studies. Eur. J. Epidemiol. 2006, 21, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Van Gelder, M.M.H.J.; Bretveld, R.W.; Roeleveld, N. Web-Based Questionnaires: The Future in Epidemiology? Am. J. Epidemiol. 2010, 172, 1292–1298. [Google Scholar] [CrossRef]
- Seybert, H. Internet Use in Households and by Individuals in 2011. Eurostat Statistics in Focus. 2011. Available online: http://ec.europa.eu/eurostat/documents/3433488/5579964/KS-SF-11-066-EN.PDF/090e071f-c3a9-45d8-aa90-9b142251fd3a?version=1.0 (accessed on 13 November 2015).
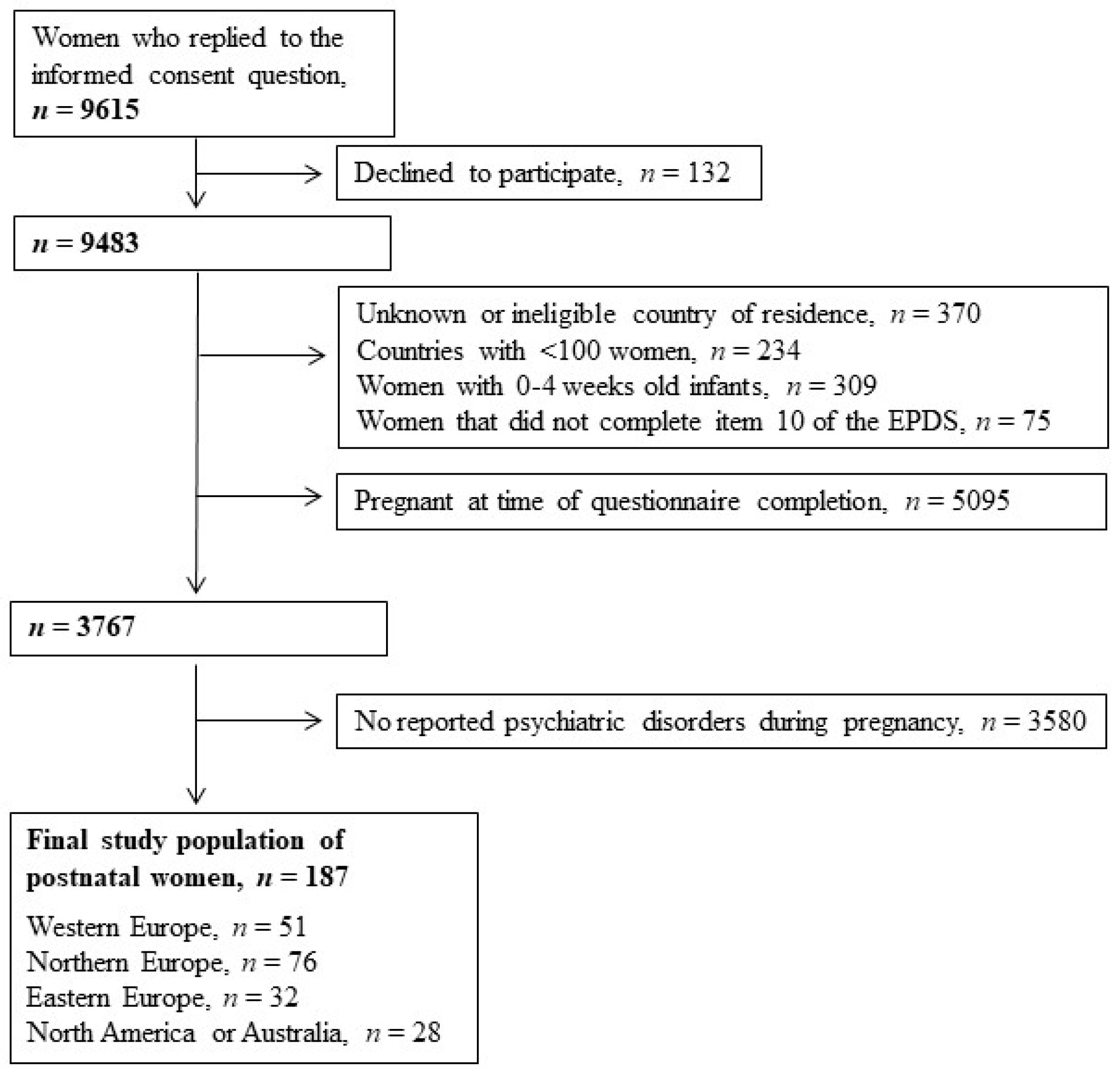
| Sociodemographic, Life-Style and Health Characteristics | Study Population | Antidepressant Treatment during Pregnancy | |
|---|---|---|---|
| Total, n = 187 | No, n = 88 | Yes, n = 99 | |
| n (%) | n (%) | n (%) | |
| Pregnancy characteristics | |||
| Weeks since childbirth | |||
| 5–16 | 42 (22.5) | 19 (21.6) | 23 (23.2) |
| 17–28 | 38 (20.3) | 17 (19.3) | 21 (21.2) |
| 29+ | 107 (57.2) | 52 (59.1) | 55 (55.6) |
| Nulliparous (yes) | 83 (44.4) | 40 (45.5) | 43 (43.4) |
| No perinatal use of folate | 11 (5.9) | 4 (4.6) | 7 (7.1) |
| Unplanned pregnancy (yes) | 33 (17.7) | 15 (17.1) | 18 (18.2) |
| Sociodemographic and life-style characteristics | |||
| Region of residency 2 | |||
| Western Europe | 51 (27.3) | 21 (23.9) | 30 (30.3) |
| Northern Europe | 76 (40.6) | 27 (30.7) | 49 (49.5) |
| Eastern Europe | 32 (17.1) | 28 (31.8) | 4 (4.0) |
| North America | 20 (10.7) | 10 (11.4) | 10 (10.1) |
| Australia | 8 (4.3) | 2 (2.3) | 6 (6.1) |
| Maternal age (years) | |||
| 17–24 | 38 (20.3) | 28 (31.8) | 10 (10.1) |
| 25–30 | 66 (35.3) | 30 (34.1) | 36 (36.4) |
| >30 | 83 (44.4) | 30 (34.1) | 53 (53.5) |
| Married or cohabitating (yes) | 163 (87.2) | 74 (84.1) | 89 (89.9) |
| Working status at conception | |||
| Employed | 99 (52.9) | 43 (48.9) | 56 (56.6) |
| Student | 30 (16.0) | 18 (20.5) | 12 (12.1) |
| Homemaker | 30 (16.0) | 15 (17.1) | 15 (15.2) |
| Job seeker/other | 28 (15.0) | 12 (13.6) | 16 (16.2) |
| Educational Attainment | |||
| Less than high school | 26 (13.9) | 15 (17.1) | 11 (11.1) |
| High school | 74 (39.6) | 37 (42.1) | 37 (37.4) |
| More than high school | 87 (46.5) | 36 (40.9) | 51 (51.5) |
| Immigrant status (yes) 3 | 7 (3.7) | 3 (3.4) | 4 (4.0) |
| Smoking during pregnancy (yes) | 30 (16.0) | 16 (18.2) | 14 (14.1) |
| Alcohol use during pregnancy (yes) 4 | 34 (18.2) | 19 (21.6) | 15 (15.2) |
| Health-related factors | |||
| Healthcare contact due to infertility (yes) | 34 (18.2) | 19 (21.6) | 15 (15.2) |
| Neurotic trait (range 8–40), mean (SD) | 28.8 (5.5) | 29.3 (5.4) | 28.4 (5.6) |
| Nausea in pregnancy (yes) | 147 (78.6) | 67 (76.1) | 80 (80.8) |
| Sleeping problems in pregnancy (yes) | 115 (61.5) | 52 (59.1) | 63 (63.6) |
| Non-psychiatric chronic conditions (yes) | 101 (54.0) | 57 (64.8) | 44 (44.4) |
| Co-medication use in pregnancy (yes) | |||
| Antipsychotics | 16 (8.6) | 6 (6.8) | 10 (10.1) |
| Benzodiazepines and z-hypnotics | 27 (14.4) | 9 (10.2) | 18 (18.2) |
| Number of co-morbid psychiatric disorders | |||
| 1 | 103 (55.1) | 50 (56.8) | 53 (53.5) |
| 2 | 80 (42.8) | 37 (42.1) | 43 (43.4) |
| 3 | 4 (2.1) | 1 (1.1) | 3 (3.0) |
| EPDS score (items 1–9; range 0–27), mean (SD) | 11.6 (5.9) | 12.7 (5.8) | 10.6 (5.0) |
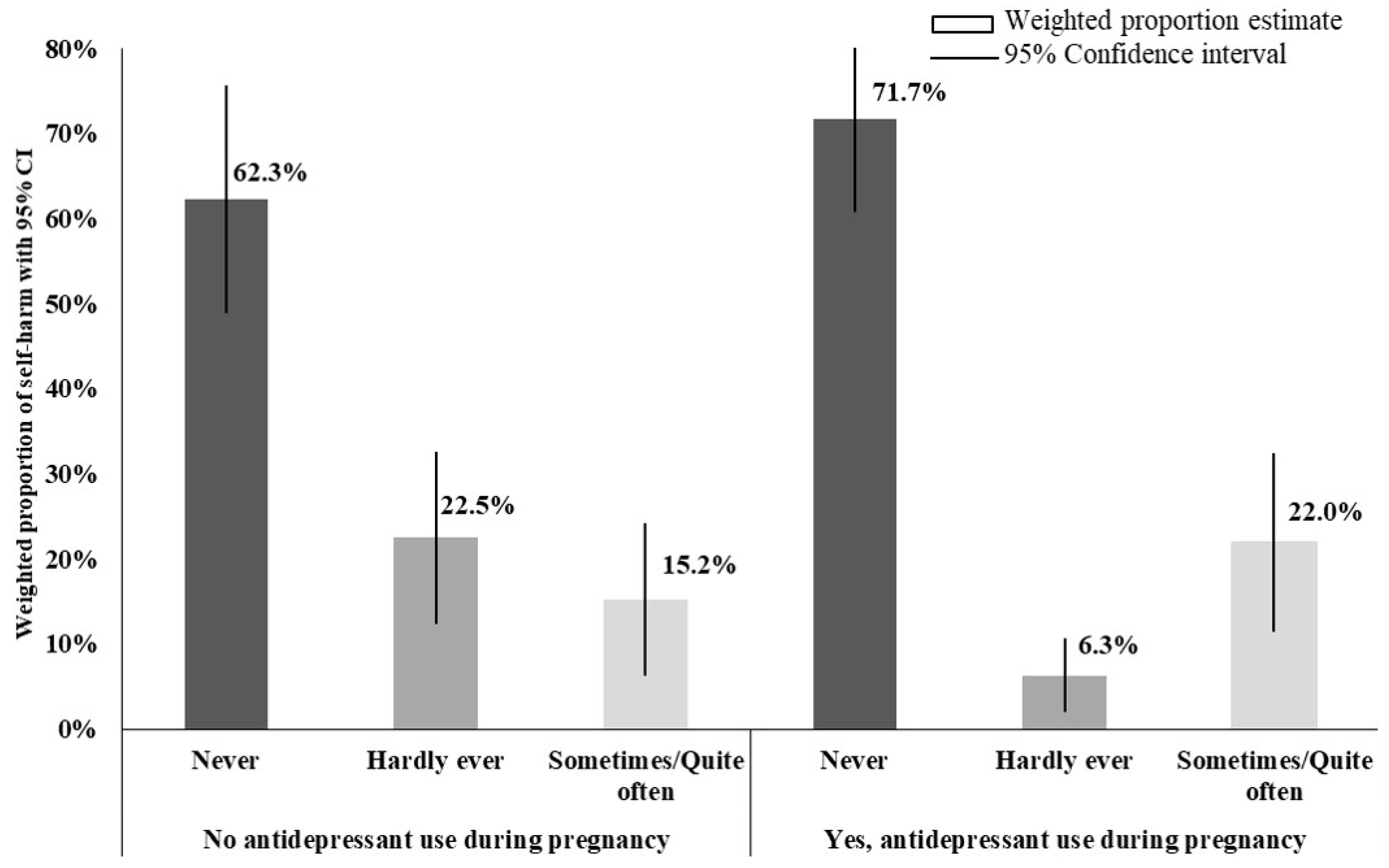
| Frequency of Postnatal Self-Harm | ||||
|---|---|---|---|---|
| Sometimes/ Quite Often | Hardly Ever | Never | ||
| n | % (95% CI) | % (95% CI) | % (95% CI) | |
| Non-medicated in pregnancy | 88 | 15.2 (6.3–24.2) | 22.5 (12.4–32.6) | 62.3 (48.9–75.6) |
| Any antidepressant in 1 trimester | 22 | 36.3 (8.6–64.0) | - | 63.7 (36.0–91.4) |
| Any antidepressant in 2 trimesters | 11 | 10.0 (0.08–28.8) | 18.1 (1.0–41.3) | 71.9 (44.3–99.4) |
| Any antidepressant in 3 trimesters | 66 | 18.0 (7.5–28.5) | 7.1 (1.5–12.7) | 74.9 (63.4–86.4) |
| SSRI antidepressant, ever in pregnancy | 85 | 11.3 (3.6–18.9) | 6.5 (1.8–11.3) | 82.2 (73.5–91.0) |
| SNRI antidepressant, ever in pregnancy | 14 | 70.1 (44.9–95.4) | 5.1 (1.0–15.4) | 24.7 (1.7–47.8) |
| n | n (%) with SHI | Crude RR (95% CI) | Survey Weighted, 2 RR (95% CI) | Fully Weighted, 3 RR (95% CI) | |
|---|---|---|---|---|---|
| Frequent SHI vs never/hardly ever SHI | |||||
| Non-medicated in pregnancy | 88 | 14 (15.9) | Reference | Reference | Reference |
| Antidepressants, ever in pregnancy | 99 | 18 (18.2) | 1.14 (0.60–2.17) | 1.44 (0.68–3.08) | 1.90 (0.79–4.56) |
| Antidepressants for 3 trimesters | 66 | 11 (16.7) | 1.05 (0.50–2.18) | 1.18 (0.52–2.71) | 0.95 (0.38–2.39) |
| Antidepressants for 1 trimester | 22 | 6 (27.3) | 1.71 (0.73–4.0) | 2.38 (0.90–6.29) | 1.85 (0.60–6.04) |
| Frequent SHI vs never SHI4 | |||||
| Non-medicated in pregnancy | 65 | 14 (21.5) | Reference | Reference | Reference |
| Antidepressants, ever in pregnancy | 90 | 18 (20.0) | 0.93 (0.50–1.74) | 1.19 (0.55–2.57) | 1.66 (0.68–4.02) |
| Antidepressants for 3 trimesters | 59 | 11 (18.6) | 0.87 (0.42–1.78) | 0.99 (0.43–2.29) | 0.85 (0.34–2.17) |
| Antidepressants for 1 trimester | 22 | 6 (27.3) | 1.27 (0.55–2.94) | 1.85 (0.69–4.93) | 1.49 (0.45–4.94) |
| Sporadic SHI vs never” SHI5 | |||||
| Non-medicated in pregnancy | 74 | 23 (31.1) | Reference | Reference | Reference |
| Antidepressants, ever in pregnancy | 81 | 9 (11.1) | 0.36 (0.18–0.73) * | 0.31 (0.13–0.70) * | 0.43 (0.17–1.09) § |
| Antidepressants for 3 trimesters | 55 | 7 (12.7) | 0.41 (0.19–0.89) † | 0.33 (0.13–0.81) ¶ | 0.57 (0.18–1.80) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vallee, J.; Wong, Y.; Mannino, E.; Nordeng, H.; Lupattelli, A. Association between Antidepressant Treatment during Pregnancy and Postpartum Self-Harm Ideation in Women with Psychiatric Disorders: A Cross-Sectional, Multinational Study. Int. J. Environ. Res. Public Health 2021, 18, 46. https://doi.org/10.3390/ijerph18010046
Vallee J, Wong Y, Mannino E, Nordeng H, Lupattelli A. Association between Antidepressant Treatment during Pregnancy and Postpartum Self-Harm Ideation in Women with Psychiatric Disorders: A Cross-Sectional, Multinational Study. International Journal of Environmental Research and Public Health. 2021; 18(1):46. https://doi.org/10.3390/ijerph18010046
Chicago/Turabian StyleVallee, Jennifer, Yih Wong, Eline Mannino, Hedvig Nordeng, and Angela Lupattelli. 2021. "Association between Antidepressant Treatment during Pregnancy and Postpartum Self-Harm Ideation in Women with Psychiatric Disorders: A Cross-Sectional, Multinational Study" International Journal of Environmental Research and Public Health 18, no. 1: 46. https://doi.org/10.3390/ijerph18010046
APA StyleVallee, J., Wong, Y., Mannino, E., Nordeng, H., & Lupattelli, A. (2021). Association between Antidepressant Treatment during Pregnancy and Postpartum Self-Harm Ideation in Women with Psychiatric Disorders: A Cross-Sectional, Multinational Study. International Journal of Environmental Research and Public Health, 18(1), 46. https://doi.org/10.3390/ijerph18010046






