Bivariate Joint Spatial Modeling to Identify Shared Risk Patterns of Hypertension and Diabetes in South Africa: Evidence from WHO SAGE South Africa Wave 2
Abstract
1. Introduction
2. Materials and Methods
Statistical Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lalkhen, H.; Mash, R. Multimorbidity in non-communicable diseases in South African primary healthcare. South Afr. Med. J. 2015, 105, 134. [Google Scholar] [CrossRef] [PubMed]
- Mayosi, B.M.; Flisher, A.J.; Lalloo, U.G.; Sitas, F.; Tollman, S.M.; Bradshaw, D. The burden of non-communicable diseases in South Africa. Lancet 2009, 374, 934–947. [Google Scholar] [CrossRef]
- Alwan, A.; MacLean, D.R.; Riley, L.M.; D’Espaignet, E.T.; Mathers, C.D.; Stevens, G.A.; Bettcher, D. Monitoring and surveillance of chronic non-communicable diseases: Progress and capacity in high-burden countries. Lancet 2010, 376, 1861–1868. [Google Scholar] [CrossRef]
- Gouda, H.N.; Charlson, F.; Sorsdahl, K.; Ahmadzada, S.; Ferrari, A.J.; Erskine, H.; Leung, J.; Santamauro, D.; Lund, C.; Aminde, L.N.; et al. Burden of non-communicable diseases in sub-Saharan Africa, 1990–2017: Results from the Global Burden of Disease Study 2017. Lancet Glob. Health 2019, 7, e1375–e1387. [Google Scholar] [CrossRef]
- Mathers, C.D.; Loncar, D. Projections of Global Mortality and Burden of Disease from 2002 to 2030. PLoS Med. 2006, 3, 442. [Google Scholar] [CrossRef]
- Salomon, J.A.; Murray, C.J.L. The Epidemiologic Transition Revisited: Compositional Models for Causes of Death by Age and Sex. Popul. Dev. Rev. 2002, 28, 205–228. [Google Scholar] [CrossRef]
- Wyk, V.P.V.; Msemburi, W.; Laubscher, R.; Dorrington, R.E.; Groenewald, P.; Glass, T.; Nojilana, B.; Joubert, J.D.; Matzopoulos, R.; Prinsloo, M.; et al. Mortality trends and differentials in South Africa from 1997 to 2012: Second National Burden of Disease Study. Lancet Glob. Health 2016, 4, e642–e653. [Google Scholar] [CrossRef]
- Oni, T.; Youngblood, E.; Boulle, A.; McGrath, N.; Wilkinson, R.J.; Levitt, N.S. Patterns of HIV, TB, and non-communicable disease multi-morbidity in peri-urban South Africa- a cross sectional study. BMC Infect. Dis. 2015, 15, 1–8. [Google Scholar] [CrossRef]
- Bretzel, R.G. Comorbidity of diabetes mellitus and hypertension in the clinical setting: A review of prevalence, pathophysiology, and treatment perspectives. Clin. Ther. 2007, 29, S35–S43. [Google Scholar] [CrossRef]
- Ware, L.J.; Chidumwa, G.; Charlton, K.; Schutte, A.E.; Kowal, P. Predictors of hypertension awareness, treatment and control in South Africa: Results from the WHO-SAGE population survey (Wave 2). J. Hum. Hypertens. 2019, 33, 157–166. [Google Scholar] [CrossRef]
- Dabney, A.R.; Wakefield, J.C. Issues in the mapping of two diseases. Stat. Methods Med Res. 2005, 14, 83–112. [Google Scholar] [CrossRef] [PubMed]
- Knorr-Held, L.; Best, N.G. A shared component model for detecting joint and selective clustering of two diseases. J. R. Stat. Soc. Ser. A 2001, 164, 73–85. [Google Scholar] [CrossRef]
- Langford, I.H.; Leyland, A.H.; Rasbash, J.; Goldstein, H. Multilevel modelling of the geographical distributions of diseases. J. R. Stat. Soc. Ser. C 1999, 48, 253–268. [Google Scholar] [CrossRef] [PubMed]
- Held, L.; Natário, I.; Fenton, S.E.; Rue, H.; Becker, N. Towards joint disease mapping. Stat. Methods Med Res. 2005, 14, 61–82. [Google Scholar] [CrossRef]
- Kazembe, L.N.; Muula, A.S.; Appleton, C.; Kleinschmidt, I. Modelling the effect of malaria endemicity on spatial variations in childhood fever, diarrhoea and pneumonia in Malawi. Int. J. Health Geogr. 2007, 6, 33. [Google Scholar] [CrossRef]
- Manda, S.O.M.; Feltbower, R.G.; Gilthorpe, M.S. Investigating spatio-temporal similarities in the epidemiology of childhood leukaemia and diabetes. Eur. J. Epidemiol. 2009, 24, 743–752. [Google Scholar] [CrossRef]
- Tarekegne, F.E.; Padyab, M.; Schröders, J.; Williams, J.S. Sociodemographic and behavioral characteristics associated with self-reported diagnosed diabetes mellitus in adults aged 50+ years in Ghana and South Africa: Results from the WHO-SAGE wave 1. BMJ Open Diabetes Res. Care 2018, 6, e000449. [Google Scholar] [CrossRef]
- Kowal, P.; Chatterji, S.; Naidoo, N.; Biritwum, R.; Fan, W.; Ridaura, R.L.; Maximova, T.; Arokiasamy, P.; Phaswana-Mafuya, N.; Williams, S.; et al. Data Resource Profile: The World Health Organization Study on global AGEing and adult health (SAGE). Int. J. Epidemiol. 2012, 41, 1639–1649. [Google Scholar] [CrossRef]
- Naude, C.E.; Zani, B.; Ongolo-Zogo, P.; Wiysonge, C.S.; Dudley, L.; Kredo, T.; Garner, P.; Young, T. Research evidence and policy: Qualitative study in selected provinces in South Africa and Cameroon. Implement. Sci. 2015, 10, 126. [Google Scholar] [CrossRef]
- South African National Department of Health. National Health Act 61 of 2003; Government Gazette: Cape Town, South Africa, 2003. [Google Scholar]
- Vyas, S.; Kumaranayake, L. Constructing socio-economic status indices: How to use principal components analysis. Health Policy Plan. 2006, 21, 459–468. [Google Scholar] [CrossRef]
- Filmer, D.; Pritchett, L.H. Estimating Wealth Effects Without Expenditure Data--Or Tears: An Application to Educational Enrollments in States of India. Demography 2001, 38, 115–132. [Google Scholar] [CrossRef] [PubMed]
- Lunn, D.; Spiegelhalter, D.; Thomas, A.; Best, N. The BUGS project: Evolution, critique and future directions. Stat. Med. 2009, 28, 3049–3067. [Google Scholar] [CrossRef] [PubMed]
- Downing, A.; Forman, D.; Gilthorpe, M.S.; Edwards, K.L.; Manda, S.O.M. Joint disease mapping using six cancers in the Yorkshire region of England. Int. J. Health Geogr. 2008, 7, 41. [Google Scholar] [CrossRef] [PubMed]
- Kandala, N.B.; Manda, S.O.M.; Tigbe, W.W.; Mwambi, H.; Stranges, S. Geographic distribution of cardiovascular comorbidities in South Africa: A national cross-sectional analysis. J. Appl. Stat. 2013, 41, 1203–1216. [Google Scholar] [CrossRef]
- Ware, L.J.; Charlton, K.E.; Schutte, A.E.; Cockeran, M.; Naidoo, N.; Kowal, P. Associations between dietary salt, potassium and blood pressure in South African adults: WHO SAGE Wave 2 Salt & Tobacco. Nutr. Metab. Cardiovasc. Dis. 2017, 27, 784–791. [Google Scholar] [CrossRef]
- Sesso, H.D.; Cook, N.R.; Buring, J.E.; Manson, J.E.; Gaziano, J.M. Alcohol Consumption and the Risk of Hypertension in Women and Men. Hypertension 2008, 51, 1080–1087. [Google Scholar] [CrossRef]
- Gatimu, S.M.; Milimo, B.; Sebastián, M.S. Prevalence and determinants of diabetes among older adults in Ghana. BMC Public Health 2016, 16, 1–12. [Google Scholar] [CrossRef]
- Ayah, R.; Joshi, M.D.; Wanjiru, R.; Njau, E.K.; Otieno, F.C.; Njeru, E.K.; Mutai, K.K. A population-based survey of prevalence of diabetes and correlates in an urban slum community in Nairobi, Kenya. BMC Public Health 2013, 13, 371. [Google Scholar] [CrossRef]
- Amoah, A.G.; Owusu, S.K.; Adjei, S. Diabetes in Ghana: A community based prevalence study in Greater Accra. Diabetes Res. Clin. Pr. 2002, 56, 197–205. [Google Scholar] [CrossRef]
- Wolf-Maier, K.; Cooper, R.S.; Banegas, J.R.; Giampaoli, S.; Hence, H.W.; Joffres, M.; Kastarinen, M.; Poulter, N.; Primatesta, P.; Rodríguez-Artalejo, F. Hypertension prevalence and blood pressure levels in 6 European countries, Canada, and the United States. J. Am. Med Assoc. 2003, 289, 2363–2369. [Google Scholar] [CrossRef]
- Abaerei, A.A.; Ncayiyana, J.; Levin, J. Health-care utilization and associated factors in Gauteng province, South Africa. Glob. Health Action 2017, 10. [Google Scholar] [CrossRef] [PubMed]
- Otwombe, K.; Dietrich, J.; Laher, F.; Hornschuh, S.; Nkala, B.; Chimoyi, L.; Kaida, A.; Gray, G.E.; Miller, C.L. Health-seeking behaviours by gender among adolescents in Soweto, South Africa. Glob. Health Action 2015, 8, 25670. [Google Scholar] [CrossRef] [PubMed]
- Gerber, L.M.; Stern, P.M. Relationship of body size and body mass to blood pressure: Sex-specific and developmental influences. Hum. Biol. 1999, 71. [Google Scholar]
- Werfalli, M.; Kassanjee, R.; Kalula, S.; Kowal, P.; Phaswana-Mafuya, N.; Levitt, N.S. Diabetes in South African older adults: Prevalence and impact on quality of life and functional disability—As assessed using SAGE Wave 1 data. Glob. Health Action 2018, 11, 1449924. [Google Scholar] [CrossRef]
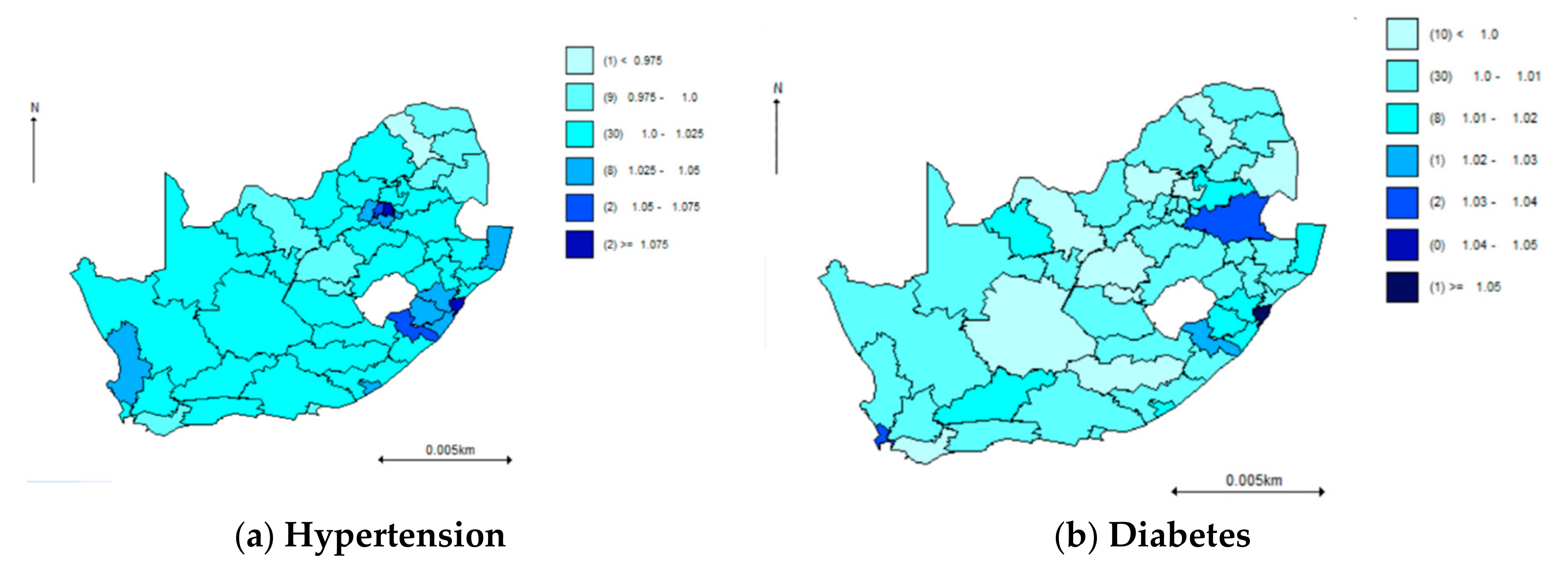
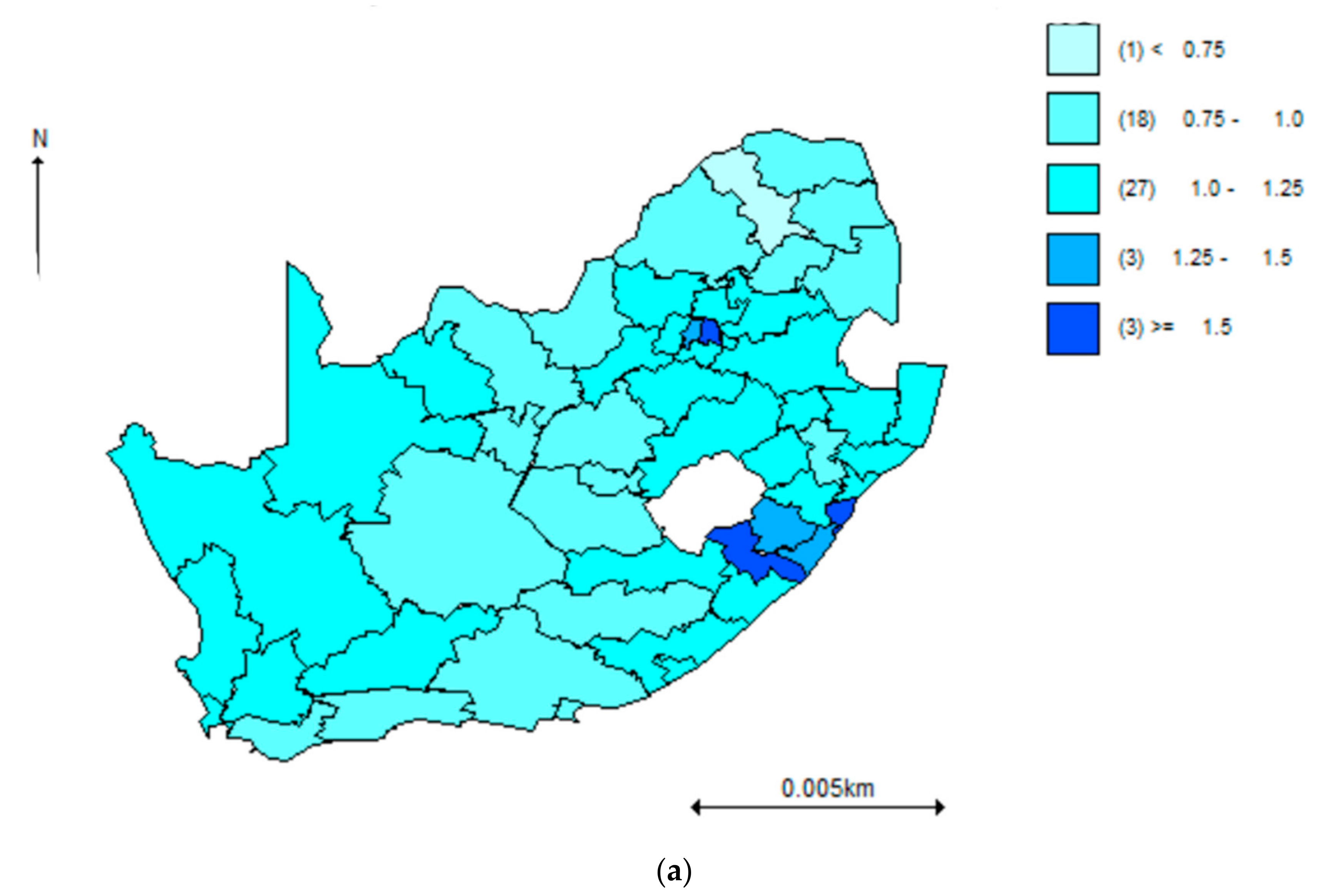
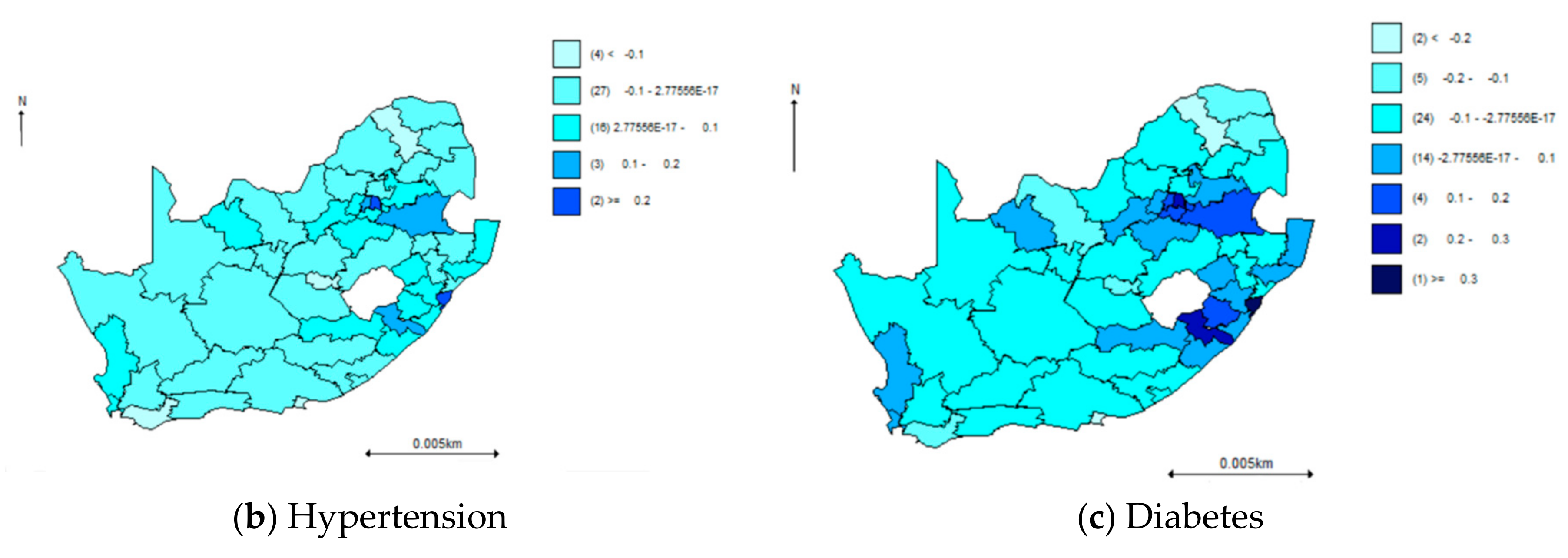
| PROVINCE | Total | Hypertension | Diabetes | Comorbidity |
|---|---|---|---|---|
| N | N (%) | N (%) | N (%) | |
| Eastern Cape | 522 | 82 (15.7) | 31 (5.9) | 18 (3.5) |
| Free State | 216 | 51 (23.6) | 22 (10.2) | 13 (6.0) |
| Gauteng | 528 | 117 (22.2) | 80 (15.2) | 55 (10.4) |
| Kwa-Zulu Natal | 450 | 127 (28.2) | 86 (19.1) | 65 (14.4) |
| Mpumalanga | 142 | 48 (33.8) | 14 (9.9) | 13 (9.2) |
| North West | 318 | 68 (21.4) | 26 (8.2) | 14 (4.4) |
| Northern Cape | 93 | 19 (20.4) | 10 (10.8) | 7 (7.5) |
| Northern Province | 175 | 30 (17.1) | 8 (4.6) | 6 (3.4) |
| Western Cape | 317 | 99 (31.2) | 61 (19.3) | 49 (15.5) |
| Total | No Co-Morbidity | Co-Morbidity | p-Value | |
|---|---|---|---|---|
| (N = 2761) | (N = 2521) | (N = 240) | ||
| Demographic characteristics | ||||
| Age (years) | <0.001 | |||
| Median (Q1, Q3) | 56.0 (40.0, 66.0) | 55.0 (39.0, 65.0) | 64.0 (56.0, 71.0) | |
| Sex | <0.001 | |||
| Male | 915 (33.1) | 866 (34.4) | 49 (20.4) | |
| Female | 1846 (66.9) | 1655 (65.6) | 191 (79.6) | |
| Currently working | <0.001 | |||
| Yes | 532 (34.5) | 509 (36.4) | 23 (16.4) | |
| No | 1008 (65.5) | 891 (63.6) | 117 (83.6) | |
| Residence | 0.611 | |||
| Urban | 1881 (68.1) | 1721 (68.3) | 160 (66.7) | |
| Rural | 880 (31.9) | 800 (31.7) | 80 (33.3) | |
| Socioeconomic characteristics | ||||
| Years of schooling | <0.001 | |||
| Median (Q1, Q3) | 10.0 (7.0, 12.0) | 10.0 (7.0, 12.0) | 8.0 (6.0, 10.0) | |
| Completed high school? | 0.003 | |||
| Yes | 652 (29.5) | 615 (30.3) | 37 (19.9) | |
| No | 1561 (70.5) | 1412 (69.7) | 149 (80.1) | |
| Household wealth tertile | 0.008 | |||
| 1 [lowest] | 751 (33.4) | 665 (32.6) | 86 (41.3) | |
| 2 | 749 (33.3) | 678 (33.2) | 71 (34.1) | |
| 3 [highest] | 748 (33.3) | 697 (34.2) | 51 (24.5) | |
| Support from government? | <0.001 | |||
| Yes | 936 (34.7) | 827 (33.5) | 109 (47.6) | |
| No | 1764 (65.3) | 1644 (66.5) | 120 (52.4) | |
| Anthropometric characteristics | ||||
| Waist-to-height ratio | <0.001 | |||
| Median (Q1, Q3) | 0.6 (0.5, 0.7) | 0.6 (0.5, 0.7) | 0.6 (0.6, 0.7) | |
| Body mass index (BMI) category, kg/m2 | 0.012 | |||
| Underweight (<18.5) | 60 (3.2) | 57 (3.3) | 3 (1.9) | |
| Normal weight (18.5–24.9) | 494 (26.1) | 467 (27.0) | 27 (16.7) | |
| Overweight (25–29.9) | 538 (28.5) | 490 (28.3) | 48 (29.6) | |
| Obese (≥30) | 799 (42.3) | 715 (41.4) | 84 (51.9) | |
| Normotensive (<120/80 mmHg) | 0.065 | |||
| Yes | 1342 (48.6) | 1239 (49.1) | 103 (42.9) | |
| No | 1419 (51.4) | 1282 (50.9) | 137 (57.1) | |
| Pre-hypertensive (120/80-139/89mmHg) | 0.466 | |||
| Yes | 1385 (50.2) | 1270 (50.4) | 115 (47.9) | |
| No | 1376 (49.8) | 1251 (49.6) | 125 (52.1) | |
| Hypertensive | <0.001 | |||
| Yes | 1168 (42.3) | 1041 (41.3) | 127 (52.9) | |
| No | 1593 (57.7) | 1480 (58.7) | 113 (47.1) | |
| Behavioral characteristics | ||||
| Add salt at table | <0.001 | |||
| Yes | 1910 (69.4) | 1770 (70.4) | 140 (58.8) | |
| No | 842 (30.6) | 744 (29.6) | 98 (41.2) | |
| Self-reported vigorous intensity physical activity | 0.468 | |||
| Yes | 366 (13.3) | 338 (13.5) | 28 (11.8) | |
| No | 2376 (86.7) | 2167 (86.5) | 209 (88.2) | |
| Ever used alcohol? | 0.221 | |||
| Yes | 523 (19.0) | 485 (19.3) | 38 (16.0) | |
| No | 2227 (81.0) | 2028 (80.7) | 199 (84.0) | |
| Ever used tobacco? | 0.781 | |||
| Yes | 482 (17.5) | 442 (17.6) | 40 (16.9) | |
| No | 2267 (82.5) | 2070 (82.4) | 197 (83.1) |
| VARIABLE | Description | HYPERTENSION | DIABETES |
|---|---|---|---|
| aOR (Credible Interval) | aOR (Credible Interval) | ||
| Demographic characteristics | |||
| Age (years) | 1.06 (1.05; 1.08) | 1.05 (1.03; 1.07) | |
| Sex | |||
| Male | Reference | Reference | |
| Female | 2.45 (1.62; 3.71) | 2.36 (1.41; 4.01) | |
| Socio-economic status characteristics | |||
| Household wealth tertile | |||
| 1 [lowest] | Reference | Reference | |
| 2 | 0.90 (0.60; 1.34) | 0.59 (0.36; 0.97) | |
| 3 [highest] | 0.83 (0.51; 1.35) | 0.53 (0.28; 0.98) | |
| Support from government? | |||
| No | Reference | ||
| Yes | 3.77 (0; 1.6 × 108) | ||
| Years of schooling | 0.90 (0.85; 0.95) | 0.93 (0.87; 0.99) | |
| Anthropological characteristics | |||
| BMI | 1.03 (1.01; 1.05) | ||
| Behavioral characteristics | |||
| Add salt at table? | No | ||
| Yes | 1.48 (0; 2.9 × 108) | ||
| Ever used tobacco? | No | Reference | |
| Yes | 1.74 (1.04; 2.88) | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chidumwa, G.; Maposa, I.; Kowal, P.; Micklesfield, L.K.; Ware, L.J. Bivariate Joint Spatial Modeling to Identify Shared Risk Patterns of Hypertension and Diabetes in South Africa: Evidence from WHO SAGE South Africa Wave 2. Int. J. Environ. Res. Public Health 2021, 18, 359. https://doi.org/10.3390/ijerph18010359
Chidumwa G, Maposa I, Kowal P, Micklesfield LK, Ware LJ. Bivariate Joint Spatial Modeling to Identify Shared Risk Patterns of Hypertension and Diabetes in South Africa: Evidence from WHO SAGE South Africa Wave 2. International Journal of Environmental Research and Public Health. 2021; 18(1):359. https://doi.org/10.3390/ijerph18010359
Chicago/Turabian StyleChidumwa, Glory, Innocent Maposa, Paul Kowal, Lisa K. Micklesfield, and Lisa J. Ware. 2021. "Bivariate Joint Spatial Modeling to Identify Shared Risk Patterns of Hypertension and Diabetes in South Africa: Evidence from WHO SAGE South Africa Wave 2" International Journal of Environmental Research and Public Health 18, no. 1: 359. https://doi.org/10.3390/ijerph18010359
APA StyleChidumwa, G., Maposa, I., Kowal, P., Micklesfield, L. K., & Ware, L. J. (2021). Bivariate Joint Spatial Modeling to Identify Shared Risk Patterns of Hypertension and Diabetes in South Africa: Evidence from WHO SAGE South Africa Wave 2. International Journal of Environmental Research and Public Health, 18(1), 359. https://doi.org/10.3390/ijerph18010359





