Dentistry during the COVID-19 Epidemic: An Italian Workflow for the Management of Dental Practice
Abstract
1. Introduction
1.1. COVID-19: Updated Report of the Pandemic
1.1.1. Incubation Period and Symptoms
1.1.2. Transmission Routes
1.1.3. Stability of the Virus
1.1.4. COVID-19 Management
1.2. Covid-19 and Dentistry
1.2.1. Operator’s Risk in Dentistry
1.2.2. COVID-19 Prevention in Dentistry
- (A)
- COVID-19 Symptomatic patient;
- (B)
- Asymptomatic positive patient;
- (C)
- Recovered patient that was previously symptomatic;
- (D)
- Recovered patient that was previously asymptomatic;
- (E)
- Negative Patient:
- -
- Very high systemic risk **
- -
- High systemic risk *
- -
- No systemic risk [36]
1.2.3. Experience of a Single Center
- (1)
- Assessment of the patient risk level based on a multiparameter analysis related to dental chief complaint, history of COVID-19 exposure, and systemic conditions.
- (2)
- Promotion of a two-phase dental procedure management: (a) remote contact via telephone and/or web for preliminary risk level evaluation and telediagnosis, and (b) face-to-face treatment.
- (3)
- Use of the updated preventive measures adopted in a COVID Hub Hospital for dental interventions with patient’s centered spaces and times of health care.
2. Materials and Methods
2.1. Concept of Urgent and Postponable Dental Procedures during Epidemic
- (1A)
- Uncontrolled bleeding.
- (1B)
- Diffused soft tissue infection with intra-oral or extra-oral swelling that potentially compromise patient’s airway.
- (2A)
- Severe dental pain from pulpal inflammation.
- (2B)
- Pericoronitis or third-molar pain.
- (2C)
- Surgical post-operative osteitis.
- (2D)
- Abscess, or localized bacterial infection.
- (2E)
- Tooth fracture resulting in pain or causing soft tissue trauma.
- (2F)
- Dental trauma with avulsion/luxation.
- (2G)
- Dental treatment required prior to critical medical procedures.
- (2H)
- Final crown/bridge cementation if the temporary restoration is lost, broken or causing gum irritation.
- (2I)
- Biopsy of abnormal tissue.
- (3A)
- Extensive dental caries or defective restorations causing pain.
- (3B)
- Suture removal.
- (3C)
- Denture adjustment on radiation/oncology patients.
- (3D)
- Denture adjustments or repairs when function impeded.
- (3E)
- Replacing temporary filling on endo access openings in patients experiencing pain.
- (3F)
- Snipping or adjustment of an orthodontic wire or appliances piercing or ulcerating the oral mucosa.
- (4A)
- Initial or periodic oral examinations and recall visits, including routine radiographs.
- (4B)
- Routine dental cleaning and preventive therapies.
- (4C)
- Orthodontic procedures.
- (4D)
- Extraction of asymptomatic teeth.
- (4E)
- Restorative dentistry including of asymptomatic carious lesions’ treatment.
2.2. Operators Management
- (a)
- One administrative staff member;
- (b)
- One nursing staff member outside the operative area;
- (c)
- One nursing staff member inside the operative area;
- (d)
- One clinician.
2.3. Patient Management
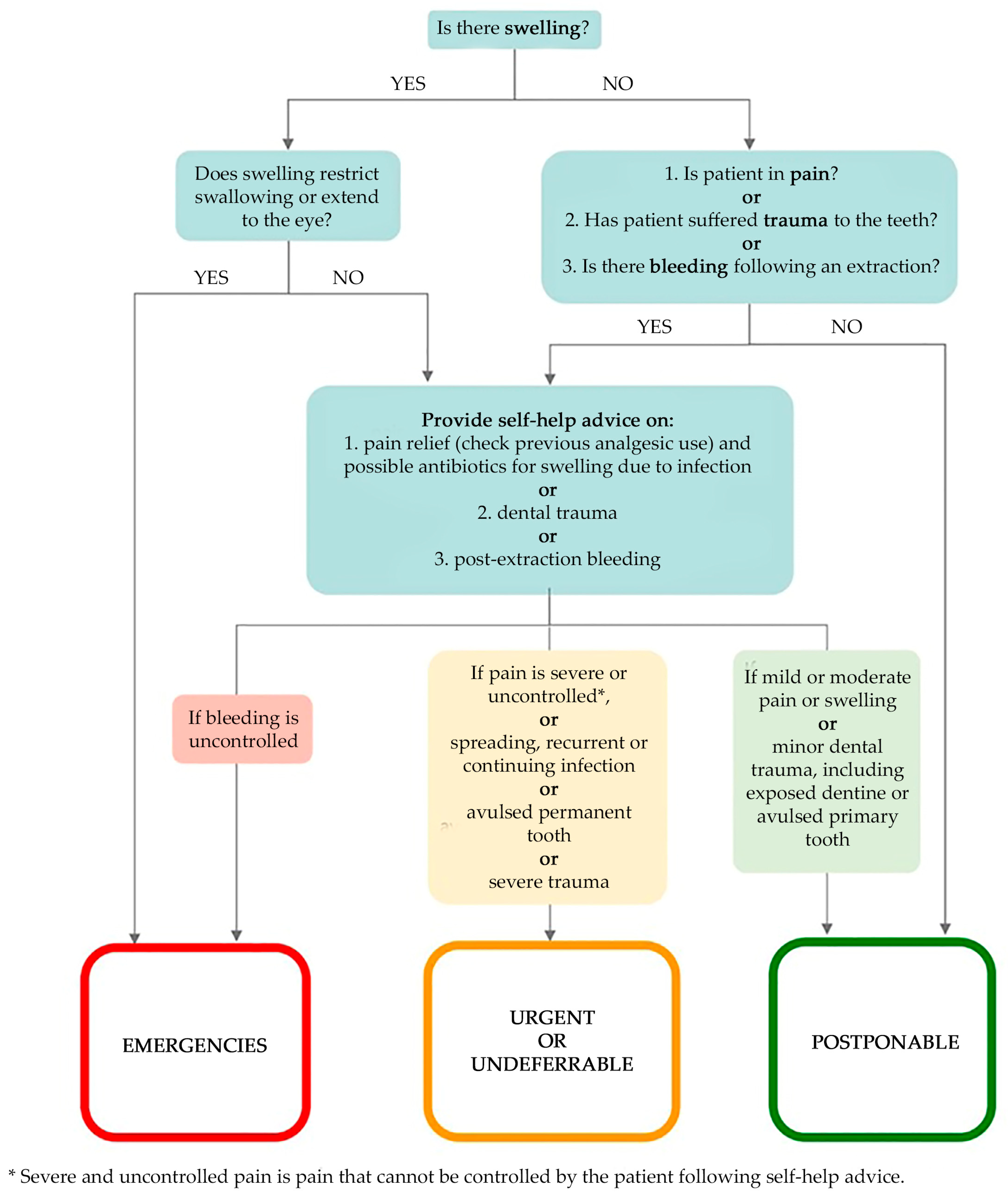
- Chief complaint in order to identify emergencies, urgent, undeferrable and/or postponable dental procedures (Figure 1):
- “Emergencies” (Category 1 ADA): within 1 h, managed through E.R;
- “Urgent” (Category 2 ADA): within 24 h;
- “Undeferrable” (Category 3 ADA): possibly more than 24 h;
- “Postponable” (Category 4 ADA): to be treated remotely.
- COVID-19 personal history obtained through the following questionnaire:
- (a)
- Are you or were you infected with COVID-19?
- (b)
- Have you had a fever, cough, cold, breathing difficulties, muscle pain or headache in the last 28 days?
- (c)
- Have you had contacts with individuals who have had these symptoms in the last 28 days?
- (d)
- Have you been in contact with infected individuals in the last 28 days?
- (e)
- Did you undergo a swab test that returned a positive result for COVID-19?
- (f)
- Have you been in a previously quarantined area?
- (g)
- Have you had contact with individuals coming from quarantined areas?
- Systemic risk category, as mentioned above:
- (A)
- COVID-19 Symptomatic patient;
- (B)
- Asymptomatic positive patient;
- (C)
- Recovered patient that was previously symptomatic;
- (D)
- Recovered patient that was previously asymptomatic;
- (E)
- Negative Patient:
- -
- Very high systemic risk
- -
- High systemic risk
- -
- No systemic risk [36]
2.4. Environments Management
2.5. Instruments Management
2.6. COVID-19 Prevention in Home Environments
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
- Wet and lather both hands and wrists, thoroughly clean all parts, including interdigital and nail spaces for 60 s, rinse thoroughly with running water, dry properly with a disposable towel, and use the disposable towel to close the tap.
References
- World Health Organization Official Website. Rolling Updates on Coronavirus Disease (COVID-19). Updated 18 April 2020. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/events-as-they-happen?fbclid=IwAR2Kk1KFnISKO-TTnWdgEi3rCni0QmJyIo98Pqw-vlPGkq9kf23mR6D4wR4 (accessed on 20 April 2020).
- Wang, C.; Horby, P.W.; Hayden, F.G.; Gao, G.F. A novel coronavirus outbreak of global health concern. Lancet 2020, 395, 470–473. [Google Scholar] [CrossRef]
- Liu, T.; Hu, J.; Kang, M.; Lin, L.; Zhong, H.; Xiao, J.; He, G.; Song, T.; Huang, Q.; Rong, Z.; et al. Transmission Dynamics of 2019 Novel Coronavirus (2019-nCoV); Social Science Research Network: Rochester, NY, USA, 2020. [Google Scholar]
- Wu, F.; Zhao, S.; Yu, B.; Chen, Y.-M.; Wang, W.; Song, Z.-G.; Hu, Y.; Tao, Z.-W.; Tian, J.-H.; Pei, Y.-Y.; et al. A new coronavirus associated with human respiratory disease in China. Nature 2020, 579, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Ministero della Salute: COVID-19 Situazione nel Mondo. Available online: http://www.salute.gov.it/portale/nuovocoronavirus/dettaglioContenutiNuovoCoronavirus.jsp?lingua=italiano&id=5338&area=nuovoCoronavirus&menu=vuoto (accessed on 23 April 2020).
- Ministero della Salute: COVID-19 Situazione in Italia. Available online: http://www.salute.gov.it/portale/nuovocoronavirus/dettaglioContenutiNuovoCoronavirus.jsp?lingua=italiano&id=5351&area=nuovoCoronavirus&menu=vuoto&fbclid=IwAR1yyEwc6dB8lyYoDmp5S0sc61uaParq-P2eVxee3ZBy1lsJLPJKhmzFDEY (accessed on 20 April 2020).
- World Health Organization Official Website. Coronavirus Disease 2019 (COVID-19) Situation Reports. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports/ (accessed on 23 April 2020).
- Covid-19: Bollettino Protezione Civile 23/07/2020, ore 18.00. Available online: http://www.salute.gov.it/portale/news/p3_2_1_1_1.jsp?lingua=italiano&menu=notizie&p=dalministero&id=4583 (accessed on 23 April 2020).
- Li, R.; Pei, S.; Chen, B.; Song, Y.; Zhang, T.; Yang, W.; Shaman, J. Substantial undocumented infection facilitates the rapid dissemination of novel coronavirus (SARS-CoV2). Science 2020, 368, 489–493. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- World Health Organization Official Website. Coronavirus Disease 2019 (COVID-19) Situation Report 7. Available online: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200127-sitrep-7-2019--ncov.pdf (accessed on 20 April 2020).
- China’s National Health Commission News Conference on Coronavirus. Available online: https://www.aljazeera.com/news/2020/01/chinas-national-health-commission-news-conference-coronavirus-200126105935024.html (accessed on 10 April 2020).
- He, X.; Lau, E.H.Y.; Wu, P.; Deng, X.; Wang, J.; Hao, X.; Lau, Y.C.; Wong, J.Y.; Guan, Y.; Tan, X.; et al. Temporal dynamics in viral shedding and transmissibility of COVID-19. Nat. Med. 2020, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Wölfel, R.; Corman, V.M.; Guggemos, W.; Seilmaier, M.; Zange, S.; Müller, M.A.; Niemeyer, D.; Jones, T.C.; Vollmar, P.; Rothe, C.; et al. Virological assessment of hospitalized patients with COVID-2019. Nature 2020, 1–5. [Google Scholar] [CrossRef]
- Backer, J.A.; Klinkenberg, D.; Wallinga, J. Incubation period of 2019 novel coronavirus (2019-nCoV) infections among travellers from Wuhan, China, 20–28 January 2020. Eurosurveillance 2020, 25, 2000062. [Google Scholar] [CrossRef]
- Guan, W.; Ni, Z.; Hu, Y.; Liang, W.; Ou, C.; He, J.; Liu, L.; Shan, H.; Lei, C.; Hui, D.S.C.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Wang, D.; Hu, B.; Hu, C.; Zhu, F.; Liu, X.; Zhang, J.; Wang, B.; Xiang, H.; Cheng, Z.; Xiong, Y.; et al. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus–Infected Pneumonia in Wuhan, China. JAMA 2020, 323, 1061–1069. [Google Scholar] [CrossRef]
- Lu, C.-W.; Liu, X.-F.; Jia, Z.-F. 2019-nCoV transmission through the ocular surface must not be ignored. Lancet 2020, 395, e39. [Google Scholar] [CrossRef]
- Belser, J.A.; Rota, P.A.; Tumpey, T.M. Ocular tropism of respiratory viruses. Microbiol. Mol. Biol. Rev. 2013, 77, 144–156. [Google Scholar] [CrossRef] [PubMed]
- To, K.K.-W.; Tsang, O.T.-Y.; Yip, C.C.-Y.; Chan, K.-H.; Wu, T.-C.; Chan, J.M.-C.; Leung, W.-S.; Chik, T.S.-H.; Choi, C.Y.-C.; Kandamby, D.H.; et al. Consistent Detection of 2019 Novel Coronavirus in Saliva. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Cheng, V.C.C.; Wong, S.-C.; Chen, J.H.K.; Yip, C.C.Y.; Chuang, V.W.M.; Tsang, O.T.Y.; Sridhar, S.; Chan, J.F.W.; Ho, P.-L.; Yuen, K.-Y. Escalating infection control response to the rapidly evolving epidemiology of the coronavirus disease 2019 (COVID-19) due to SARS-CoV-2 in Hong Kong. Infect. Control Hosp. Epidemiol. 2020, 41, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Ong, S.W.X.; Tan, Y.K.; Chia, P.Y.; Lee, T.H.; Ng, O.T.; Wong, M.S.Y.; Marimuthu, K. Air, Surface Environmental, and Personal Protective Equipment Contamination by Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) From a Symptomatic Patient. JAMA 2020. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, K.L.; Murti, M.; Finkelstein, M.; Leis, J.; Fitzgerald-Husek, A.; Bourns, L.; Meghani, H.; Saunders, A.; Allen, V.; Yaffe, B. Lack of COVID-19 Transmission on an International Flight. CMAJ 2020, 192, E410. [Google Scholar] [CrossRef]
- Xu, J.; Li, Y.; Gan, F.; Du, Y.; Yao, Y. Salivary Glands: Potential Reservoirs for COVID-19 Asymptomatic Infection. J. Dent. Res. 2020, 22034520918518. [Google Scholar] [CrossRef]
- Azzi, L.; Carcano, G.; Gianfagna, F.; Grossi, P.; Gasperina, D.D.; Genoni, A.; Fasano, M.; Sessa, F.; Tettamanti, L.; Carinci, F.; et al. Saliva Is A Reliable Tool To Detect SARS-CoV-2. J. Infect. 2020. [Google Scholar] [CrossRef]
- van Doremalen, N.; Bushmaker, T.; Morris, D.H.; Holbrook, M.G.; Gamble, A.; Williamson, B.N.; Tamin, A.; Harcourt, J.L.; Thornburg, N.J.; Gerber, S.I.; et al. Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1. N. Engl. J. Med. 2020, 382, 1564–1567. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control. Disinfection of Environments in Healthcare and Nonhealthcare Settings Potentially Contaminated with SARS-CoV-2; ECDC: Stockholm, Sweden, 2020. [Google Scholar]
- Bennardo, F.; Buffone, C.; Giudice, A. New therapeutic opportunities for COVID-19 patients with Tocilizumab: Possible correlation of interleukin-6 receptor inhibitors with osteonecrosis of the jaws. Oral Oncol. 2020, 104659. [Google Scholar] [CrossRef]
- Occupational Safety and Health Administration Official Website. Worker Exposure Risk to COVID-19. Available online: https://www.osha.gov/Publications/OSHA3993.pdf (accessed on 20 April 2020).
- Gamio, L. The Workers Who Face the Greatest Coronavirus Risk. Available online: https://www.nytimes.com/interactive/2020/03/15/business/economy/coronavirus-worker-risk.html?action=click&module=Top+Stories&pgtype=Homepage (accessed on 20 April 2020).
- Sabino-Silva, R.; Jardim, A.C.G.; Siqueira, W.L. Coronavirus COVID-19 impacts to dentistry and potential salivary diagnosis. Clin. Oral Investig. 2020, 24, 1619–1621. [Google Scholar] [CrossRef] [PubMed]
- Spagnuolo, G.; De Vito, D.; Rengo, S.; Tatullo, M. COVID-19 Outbreak: An Overview on Dentistry. Int. J. Environ. Res. Public Health 2020, 17, 2094. [Google Scholar] [CrossRef] [PubMed]
- Ye, G.; Pan, Z.; Pan, Y.; Deng, Q.; Chen, L.; Li, J.; Li, Y.; Wang, X. Clinical characteristics of severe acute respiratory syndrome coronavirus 2 reactivation. J. Infect. 2020, 80, e14–e17. [Google Scholar] [CrossRef] [PubMed]
- Klompas, M. Coronavirus Disease 2019 (COVID-19): Protecting Hospitals From the Invisible. Ann. Intern. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- National Health Service Official Website. COVID-19 Guidance and Standard Operating Procedure. Urgent Dental Care Systems in the Context of Coronavirus. Version 1. 15 April 2020. Available online: https://www.england.nhs.uk/coronavirus/wp-content/uploads/sites/52/2020/04/C0282-covid-19-urgent-dental-care-sop.pdf (accessed on 20 April 2020).
- American Dental Association Official Website. What Constitutes a Dental Emergency? Available online: https://success.ada.org/~/media/CPS/Files/Open%20Files/ADA_COVID19_Dental_Emergency_DDS.pdf (accessed on 20 April 2020).
- American Dental Association. ADA Interim Guidance for Management of Emergency and Urgent Dental Care. Updated 4/1/2020. Available online: https://www.ada.org/~/media/CPS/Files/COVID/ADA_Int_Guidance_Mgmt_Emerg-Urg_Dental_COVID19?utm_source=adaorg&utm_medium=VanityURL&utm_content=interimguidance-flowcharts&utm_campaign=covid-19 (accessed on 20 April 2020).
- SIdP Società Italiana di Parodontologia e Implantologia. COVID-19—Norme per L’attività Odontoiatrica. Available online: https://www.sidp.it/media/taxtbu3.pdf?fbclid=IwAR3woxuY0a5aQ7EAuVGq0dyWfWElCiMdNCkEmCULCnLEA8-F4e41QvpNxZw (accessed on 20 April 2020).
- 3M Report. Comparison of FFP2, KN95, and N95 and Other Filtering Facepiece Respirator Classes. Technical Bulletin, January, 2020, Revision 2. Available online: https://multimedia.3m.com/mws/media/1791500O/comparison-ffp2-kn95-n95-filtering-facepiece-respirator-classes-tb.pdf (accessed on 20 April 2020).
- Fiche Pratique de Sécurité ED 105. Appareils de Protection Respiratoire et Métiers de la Santé. Inrs.fr. INRS. Available online: https://www.esst-inrs.fr/3rb/ressources/ed105.pdf (accessed on 20 April 2020).
- Izzetti, R.; Nisi, M.; Gabriele, M.; Graziani, F. COVID-19 Transmission in Dental Practice: Brief Review of Preventive Measures in Italy. J. Dent. Res. 2020. [Google Scholar] [CrossRef]
- Scottish Dental Clinical Effectiveness Programme Official Website. Drugs for the Management of Dental Problems during COVID-19 Pandemic. Updated 8 April 2020. Available online: http://www.sdcep.org.uk/wp-content/uploads/2020/04/SDCEP-MADP-COVID-19-drug-supplement-080420.pdf (accessed on 20 April 2020).
- Kariwa, H.; Fujii, N.; Takashima, I. Inactivation of SARS coronavirus by means of povidone-iodine, physical conditions and chemical reagents. Dermatology (Basel) 2006, 212 (Suppl. 1), 119–123. [Google Scholar] [CrossRef]
- Kirk-Bayley, J.; Combes, J.; Sunkaraneni, S.; Challacombe, S. The Use of Povidone Iodine Nasal Spray and Mouthwash During the Current COVID-19 Pandemic May Reduce Cross Infection and Protect Healthcare Workers; Social Science Research Network: Rochester, NY, USA, 2020. [Google Scholar]
- Meng, L.; Hua, F.; Bian, Z. Coronavirus Disease 2019 (COVID-19): Emerging and Future Challenges for Dental and Oral Medicine. J. Dent. Res. 2020, 99, 481–487. [Google Scholar] [CrossRef]
- Peng, X.; Xu, X.; Li, Y.; Cheng, L.; Zhou, X.; Ren, B. Transmission routes of 2019-nCoV and controls in dental practice. Int. J. Oral Sci. 2020, 12, 1–6. [Google Scholar] [CrossRef]
- Alharbi, A.; Alharbi, S.; Alqaidi, S. Guidelines for dental care provision during the COVID-19 pandemic. Saudi Dent. J. 2020. [Google Scholar] [CrossRef]
- World Health Organization Official Website. Coronavirus Disease (COVID-19) Advice for the Public. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/advice-for-public (accessed on 20 April 2020).
- Ministero della Salute. Previeni le Infezioni con il Corretto Lavaggio Delle Mani. Available online: http://www.salute.gov.it/imgs/C_17_opuscoliPoster_340_allegato.pdf (accessed on 20 April 2020).

| DPI | Outside the Operative Area | Non-aerosol Generating Procedures | Aerosol Generating Procedures |
|---|---|---|---|
| Surgical mask | √ | ||
| FFP2/FFP3 mask | √ | √ | |
| Face shield | √ | ||
| Protective glasses | √ | √ | |
| Gloves | √ | √ | √ |
| Cap | √ | √ | √ |
| Protective waterproof clothing | √ | ||
| Shoe cover | √ | √ |
| Types of Masks | Specifics |
|---|---|
| FFP1 | Aerosol filtration percentage: 80% minimum Internal leak rate: Maximum 22% |
| FFP2 (equivalent to N95) | Aerosol filtration percentage: Not less than 94% Internal leak rate: Maximum 8% |
| FFP3 | Aerosol filtration percentage: Not less than 99% for EN 149-FFP3. And 99.95% for EN 143-P3 Internal leak rate: Maximum 2% |
| Disinfectant | Required Exposure Time |
|---|---|
| 70% ethyl alcohol | 5 min |
| Potassium peroxymonosulphate solution (1/100 dilution) | 5 min |
| 2.5% sodium hypochlorite | 5 min |
| 55% hydroalcoholic solution with quaternary ammonium propionate | 5 min |
| Preventive Measures/Tools | Non Aerosol-Generating Procedures | Aerosol-Generating Procedures |
|---|---|---|
| 1. Sterility rules for any autoclavable instrument | YES | YES |
| 2. Protect dental unit, lamp, handles with disposable films that will be properly disposed at the end of the session | YES | YES |
| 3. Protect handpieces, hoses of handpieces, hoses of suction system, handles, trays and shelves with disposable films | NO | YES |
| 4. Necessary tools only on the shelves | YES | YES |
| 5. Rubber dam | NO | YES |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peditto, M.; Scapellato, S.; Marcianò, A.; Costa, P.; Oteri, G. Dentistry during the COVID-19 Epidemic: An Italian Workflow for the Management of Dental Practice. Int. J. Environ. Res. Public Health 2020, 17, 3325. https://doi.org/10.3390/ijerph17093325
Peditto M, Scapellato S, Marcianò A, Costa P, Oteri G. Dentistry during the COVID-19 Epidemic: An Italian Workflow for the Management of Dental Practice. International Journal of Environmental Research and Public Health. 2020; 17(9):3325. https://doi.org/10.3390/ijerph17093325
Chicago/Turabian StylePeditto, Matteo, Simone Scapellato, Antonia Marcianò, Paola Costa, and Giacomo Oteri. 2020. "Dentistry during the COVID-19 Epidemic: An Italian Workflow for the Management of Dental Practice" International Journal of Environmental Research and Public Health 17, no. 9: 3325. https://doi.org/10.3390/ijerph17093325
APA StylePeditto, M., Scapellato, S., Marcianò, A., Costa, P., & Oteri, G. (2020). Dentistry during the COVID-19 Epidemic: An Italian Workflow for the Management of Dental Practice. International Journal of Environmental Research and Public Health, 17(9), 3325. https://doi.org/10.3390/ijerph17093325







