Effect of Kelulut Honey on the Cellular Dynamics of TGFβ-Induced Epithelial to Mesenchymal Transition in Primary Human Keratinocytes
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Consideration
2.2. Isolation of Primary Human Keratinocytes
2.3. Scratch Wound Healing Assay
2.4. Cellular Dynamic Parameters
2.4.1. Wound Closure Rate Analysis
2.4.2. Migration Speed Analysis
2.4.3. Migration Directionality Analysis
2.5. Cell Circularity
2.6. Immunocytochemistry
2.7. Honey Preparation
2.8. MTT Cell Viability Assay
2.9. Statistical Analysis
3. Results
3.1. TGFβ Induction Enhance In Vitro Keratinocytes Wound Closure
3.2. TGFβ Induction Increased Individual Keratinocytes Migration Velocity
3.3. TGFβ Induction Did Not Affect the Directionality of the Keratinocytes Migration
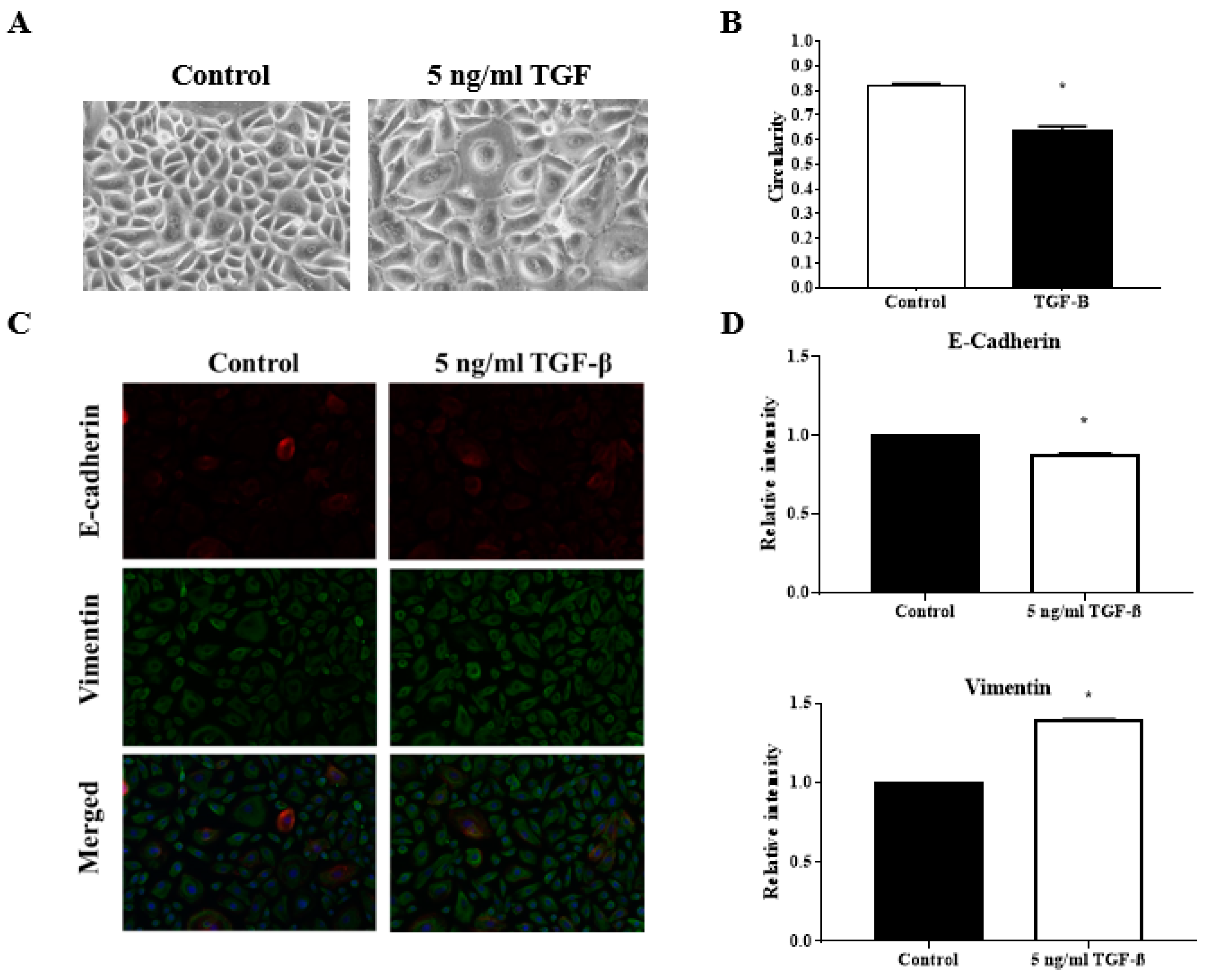
3.4. TGFβ Induction Reduced the Circularity of the Keratinocytes
3.5. TGFβ Induction Exhibit Classical EMT Markers Expression
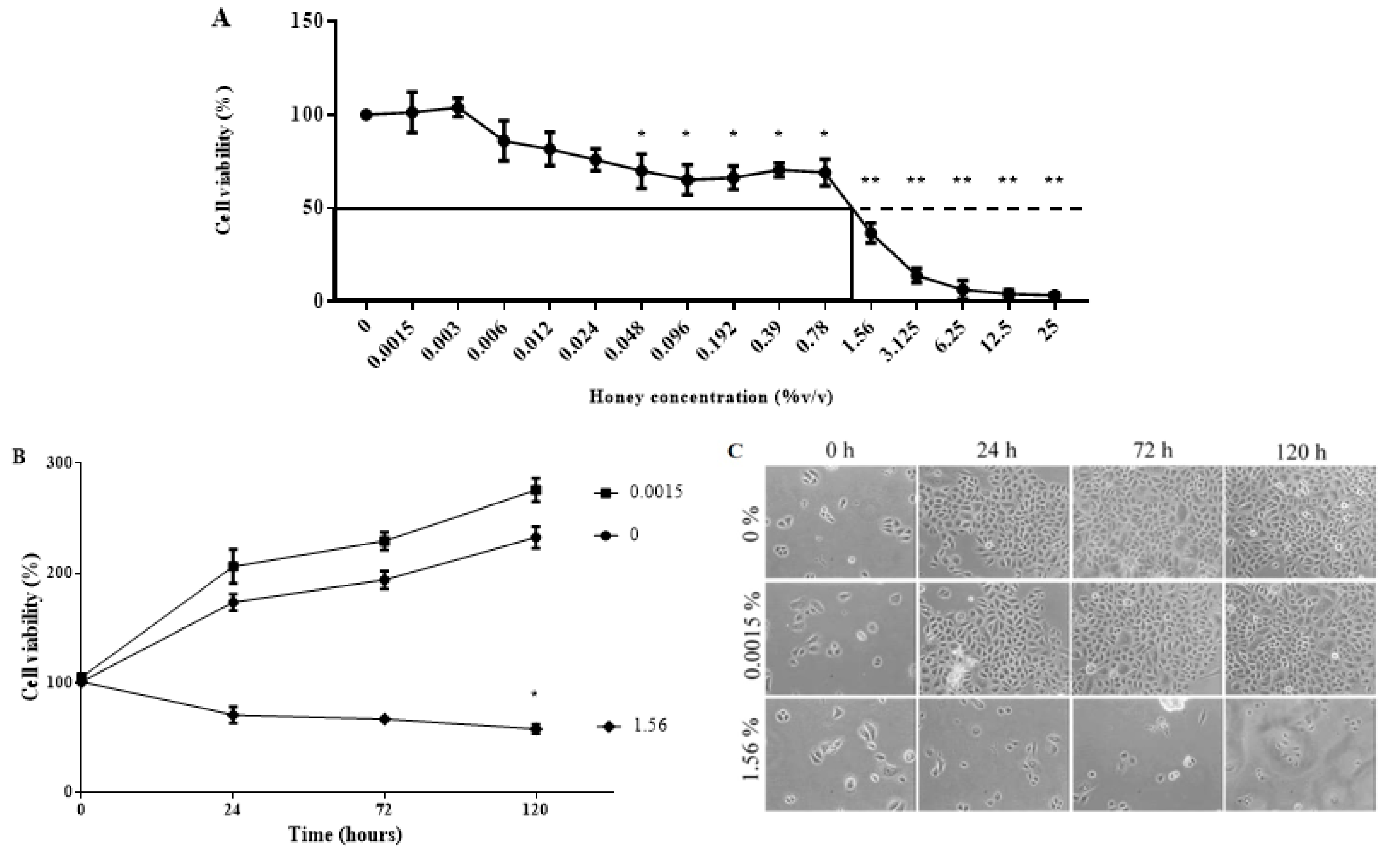
3.6. KH Is Non-Toxic at Low Concentrations
3.7. KH Modulates TGFβ Induced Enhancement of In Vitro Keratinocytes Wound Healing
3.8. KH Modulates TGFβ Induced Enhancement of Keratinocytes Migration Velocity
3.9. KH alone Improves the Keratinocytes Migration Directionality
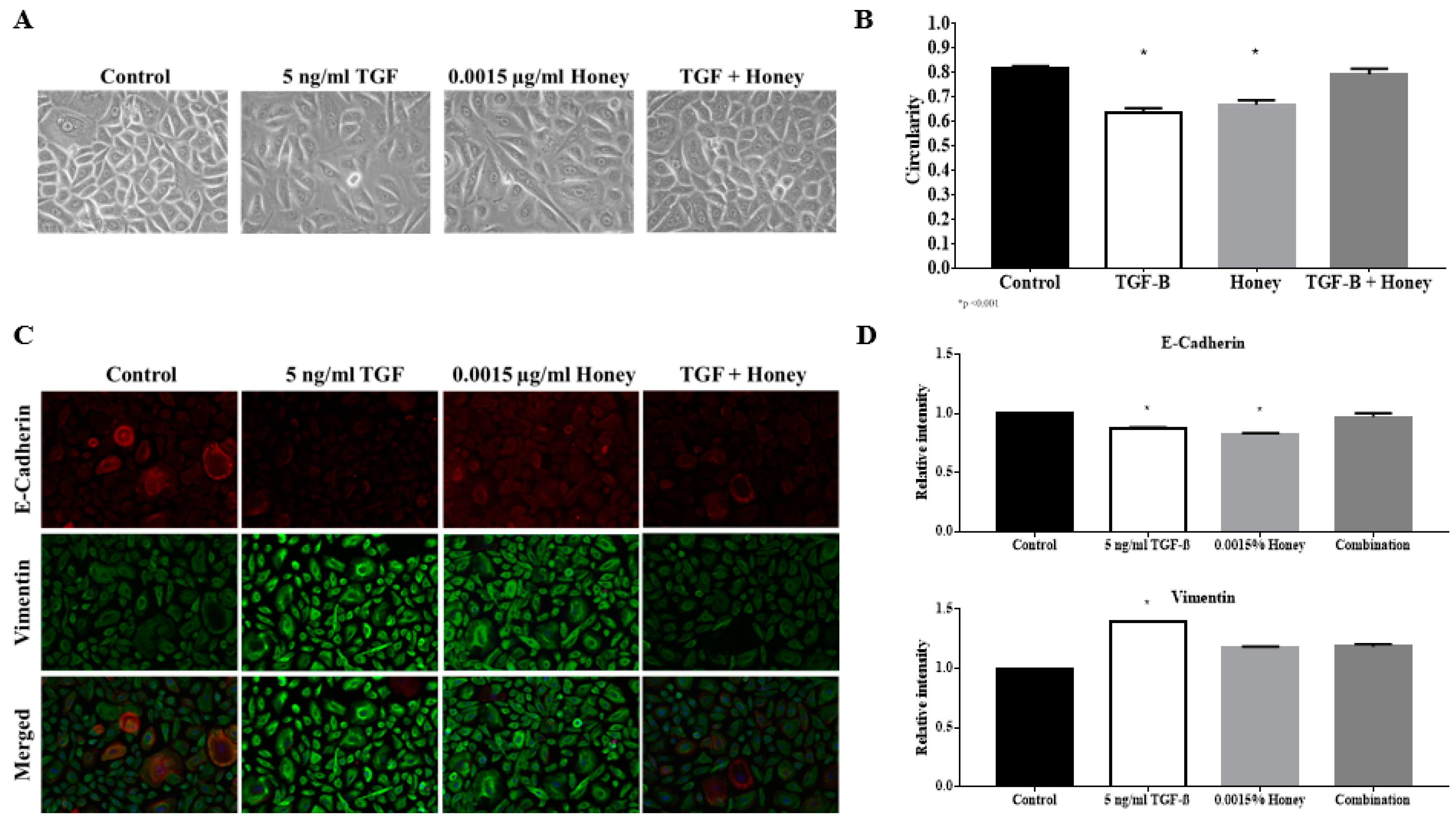
3.10. KH Reverses TGFβ Induced Reduction of the Keratinocytes Circularity
3.11. KH Reverses TGFβ Induced EMT Protein Expression
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ANOVA | One-way analysis of variance |
| bHLH | Basic helix-loop-helix |
| DMSO | Dimethyl sulfoxide |
| EMT | Epithelial to mesenchymal transition |
| IC50 | Half maximal inhibitory concentration |
| KH | Kelulut honey |
| MAPK | Mitogen-activated kinase |
| MTT | 3-(4, 5-dimethylthiazolyl-2)-2, 5-diphenyltetrazolium bromide |
| PI3K | Phosphoinositide 3-kinase |
| RI | Relative intensity |
| TGFβ | Tumor growth factor beta |
| UKMREC | Universiti Kebangsaan Malaysia Research Ethics Committee |
| ZEB | Zinc-finger E-box-binding |
References
- Hahn, J.M.; McFarland, K.L.; Combs, K.A.; Supp, D.M. Partial epithelial-mesenchymal transition in keloid scars: Regulation of keloid keratinocytes gene expression by transforming growth factor-β1. Burn. Trauma 2016, 4, 30. [Google Scholar] [CrossRef] [PubMed]
- Moore, A.L.; Marshall, C.D.; Nauta, A.; Lorenz, H.P.; Longaker, M.T. Scarless wound healing: From experimental target to clinical reality. In Principles of Regenerative Medicine, 3rd ed.; Atala, A., Lanza, R., Mikos, A.G., Nerem, R., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 65–92. [Google Scholar]
- Hahn, J.M.; Glaser, K.; McFarland, K.L.; Aronow, B.J.; Boyce, S.T.; Supp, D.M. Keloid-derived keratinocytes exhibit an abnormal gene expression profile consistent with a distinct causal role in keloid pathology. Wound Repair Regen. 2013, 21, 530–544. [Google Scholar] [CrossRef] [PubMed]
- Nordin, A.; Chowdhury, S.R.; Saim, A.; Hj Idrus, R. Epithelial to mesenchymal transition and reepithelialisation in wound healing: A review of comparison. Sains Malays. 2018, 47, 2463–2471. [Google Scholar] [CrossRef]
- Kalluri, R.; Weinberg, R. The basics of epithelial-mesenchymal transition. J. Clin. Invest. 2009, 119, 1420–1428. [Google Scholar] [CrossRef] [PubMed]
- Devaraj, V.; Bose, B. Morphological state transition dynamics in EGF-induced epithelial to mesenchymal transition. J. Clin. Med. 2019, 8, 911. [Google Scholar] [CrossRef]
- Kimmel, J.C.; Chang, A.Y.; Brack, A.S.; Marshall, W.F. Inferring cell state by quantitative motility analysis reveals a dynamic state system and broken detailed balance. PLoS Comput. Biol. 2018, 14, e1005927. [Google Scholar] [CrossRef]
- Rueden, C.T.; Eliceiri, K.W. The ImageJ ecosystem: An open and extensible platform for biomedical image analysis. In Microscopy Histopathology and Analytics, Proceedings of the Biophotonics Congress: Biomedical Optics Congress, Hollywood, FL, USA, 3–6 April 2018; Optical Society of America: Washington, DC, USA, MTh2A-3.
- Kao, H.F.; Chang-Chien, P.W.; Chang, W.T.; Yeh, T.M.; Wang, J.Y. Propolis inhibits TGFβ1-induced epithelial-mesenchymal transition in human alveolar epithelial cells via PPARγ activation. Int. Immunopharmacol. 2013, 15, 565–574. [Google Scholar] [CrossRef]
- Guo, X.; Hutcheon, A.E.K.; Tran, J.A.; Zieske, J.D. TGFβ-target genes are differentially regulated in corneal epithelial cells and fibroblasts. New Front. Ophthalmol. 2017, 3. [Google Scholar] [CrossRef]
- O’Kane, D.; Jackson, M.V.; Kissenpfennig, A.; Spence, S.; Damkat-Thomas, L.; Tolland, J.P.; Smyth, A.E.; Denton, C.P.; Stuart Elborn, J.; McAuley, D.F.; et al. SMAD inhibition attenuates epithelial to mesenchymal transition by primary keratinocytes in vitro. Exp. Dermatol. 2014, 23, 497–503. [Google Scholar] [CrossRef]
- Kuropatnicki, A.K.; Kłósek, M.; Kucharzewski, M. Honey as medicine: Historical perspectives. J. Apic. Res. 2018, 57, 113–118. [Google Scholar] [CrossRef]
- Saikaly, S.K.; Khachemoune, A. Honey and wound healing: An update. Am. J. Clin. Derm. 2017, 18, 237–251. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, A.; Bag, S.; Mandal, M.; Krishna Karri, S.P.; Barui, A.; Rajput, M.; Banerjee, P.; Sheet, D.; Chatterjee, J. Modulating prime molecular expressions and in vitro wound healing rate in keratinocytes (HaCaT) population under characteristic honey dilutions. J. Ethnopharmacol. 2015, 166, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Barui, A.; Khare, R.; Dhara, S.; Banerjee, P.; Chatterjee, J. Ex vivo bio-compatibility of honey-alginate fibrous matrix for HaCaT and 3T3 with prime molecular expressions. J. Mater. Sci. Mater. Med. 2014, 25, 2659–2667. [Google Scholar] [CrossRef] [PubMed]
- Raynaud, A.; Ghezali, L.; Gloaguen, V.; Liagre, B.; Quero, F.; Petit, J.M. Honey-induced macrophage stimulation: AP-1 and NF-κB activation and cytokine production are unrelated to LPS content of honey. Int. Immunopharmacol. 2013, 17, 874–879. [Google Scholar] [CrossRef]
- Erejuwa, O.O.; Sulaiman, S.A.; Ab Wahab, M.S. Effects of honey and its mechanisms of action on the development and progression of cancer. Molecules 2014, 19, 2497–2522. [Google Scholar] [CrossRef]
- Du Toit, D.F.; Page, B.J. An in vitro evaluation of the cell toxicity of honey and silver dressings. J. Wound Care 2009, 18, 383–389. [Google Scholar] [CrossRef]
- Martinotti, S.; Calabrese, G.; Ranzato, E. Honeydew honey: Biological effects on skin cells. Mol. Cell Biochem. 2017, 435, 185–192. [Google Scholar] [CrossRef]
- Ranzato, E.; Martinotti, S.; Burlando, B. Epithelial mesenchymal transition traits in honey-driven keratinocytes wound healing: Comparison among different honeys. Wound Repair Regen 2012, 20, 778–785. [Google Scholar] [CrossRef]
- Nordin, A.; Omar, N.; Sainik, N.Q.A.V.; Chowdhury, S.R.; Omar, E.; Bin Saim, A.; Bt Hj Idrus, R. Low dose stingless bee honey increases viability of human dermal fibroblasts that could potentially promote wound healing. Wound Med. 2018, 23, 22–27. [Google Scholar] [CrossRef]
- Manira, M.; Khairul Anuar, K.; Seet, W.T.; Ahmad Irfan, A.W.; Ng, M.H.; Chua, K.H.; Mohd Heikal, M.Y.; Aminuddin, B.S.; Ruszymah, B.H.I. Comparison of the effects between animal-derived trypsin and recombinant trypsin on human skin cells proliferation, gene and protein expression. Cell Tissue Bank 2014, 15, 41–49. [Google Scholar] [CrossRef]
- Bobadilla, A.V.P.; Arévalo, J.; Sarró, E.; Byrne, H.; Maini, P.K.; Carraro, T.; Balocco, S.; Meseguer, A.; Alarcón, T. Local migration quantification method for scratch assays. arXiv 2018, arXiv:1806.09219. [Google Scholar]
- Norazzila, O.; Yogeswaran, L.; Ruszymah, B.H.I. Comparison of cytotoxicity measurements of Centella asiatica (L.) on human Wharton’s jelly-derived mesenchymal stem cells in vitro via MTT and PrestoBlue assay. Regen. Res. 2017, 5, 10–18. [Google Scholar]
- Razali, R.A.; Nik Ahmad Eid, N.A.H.; Jayaraman, T.; Amir Hassan, M.A.; Azlan, N.Q.; Ismail, N.F.; Sainik, N.Q.A.V.; Yazid, M.D.; Lokanathan, Y.; Saim ABin Hj Idrus, R.B. The potential of Olea europaea extracts to prevent TGFβ1-induced epithelial to mesenchymal transition in human nasal respiratory epithelial cells. BMC Complement. Altern. Med. 2018, 18, 197. [Google Scholar] [CrossRef] [PubMed]
- Pastar, I.; Stojadinovic, O.; Krzyzanowska, A.; Barrientos, S.; Stuelten, C.; Zimmerman, K.; Blumenberg, M.; Brem, H.; Tomic-Canic, M. Attenuation of the transforming growth factor β-signaling pathway in chronic venous ulcers. Mol. Med. 2010, 16, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Medici, D.; Hay, E.D.; Olsen, B.R. Snail and Slug promote epithelial-mesenchymal transition through β-catenin–T-cell factor-4-dependent expression of transforming growth factor-β3. Mol. Biol. Cell. 2008, 19, 4875–4887. [Google Scholar] [CrossRef]
- Lamouille, S.; Xu, J.; Derynck, R. Molecular mechanisms of epithelial–mesenchymal transition. Nat. Rev. Mol. Cell Biol. 2014, 15, 178. [Google Scholar] [CrossRef]
- Ko, Y.G.; Ho, C.C. Gradient-free directional cell migration in continuous microchannels. Soft Mater 2013, 9, 2467–2474. [Google Scholar] [CrossRef]
- Haensel, D.; Sun, P.; MacLean, A.L.; Ma, X.; Zhou, Y.; Stemmler, M.P.; Brabletz, S.; Berx, G.; Plikus, M.V.; Nie, Q.; et al. An Ovol2-Zeb1 transcriptional circuit regulates epithelial directional migration and proliferation. EMBO Rep. 2019, 20, e46273. [Google Scholar] [CrossRef]
- Hou, L.; Liu, L.L.; Qu, Z.H.; Wang, X.; Sun, X.X.; Wu, P.; Shi, Y.P. In vitro cytotoxicity evaluation of highly absorbent foam dressings based on silver zirconium phosphate via IC 50 value. J. Biomater. Nanobiotechnol. 2016, 7, 37–44. [Google Scholar]
- Yusof, A.M.; Abd Ghafar, N.; Kamarudin, T.A.; Hui, C.K.; Yusof, Y.A. Gelam honey potentiates ex vivo corneal keratocytes proliferation with desirable phenotype expression. BMC Complement. Altern. Med. 2016, 16, 1–9. [Google Scholar] [CrossRef]
- Nordin, A.; Sainik, N.Q.A.V.; Chowdhury, S.R.; Saim, A.B.; Idrus, R.B.H. Physicochemical properties of stingless bee honey from around the globe: A comprehensive review. J. Food Compos. Anal. 2018, 73, 91–102. [Google Scholar] [CrossRef]
- Tchounwou, C.K.; Yedjou, C.G.; Farah, I.; Tchounwou, P.B. D-glucose-induced cytotoxic, genotoxic, and apoptotic effects on human breast adenocarcinoma (MCF-7) cells. J. Cancer Sci. 2014, 6, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Xiong, H.R. Culture Conditions and Types of Growth Media for Mammalian Cells. In Biomedical Tissue Culture; Ceccherini-Nelli, L., Matteoli, B., Eds.; IntechOpen: Rijeka, Croatia, 2012; pp. 3–18. [Google Scholar]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nordin, A.; Chowdhury, S.R.; Saim, A.B.; Bt Hj Idrus, R. Effect of Kelulut Honey on the Cellular Dynamics of TGFβ-Induced Epithelial to Mesenchymal Transition in Primary Human Keratinocytes. Int. J. Environ. Res. Public Health 2020, 17, 3229. https://doi.org/10.3390/ijerph17093229
Nordin A, Chowdhury SR, Saim AB, Bt Hj Idrus R. Effect of Kelulut Honey on the Cellular Dynamics of TGFβ-Induced Epithelial to Mesenchymal Transition in Primary Human Keratinocytes. International Journal of Environmental Research and Public Health. 2020; 17(9):3229. https://doi.org/10.3390/ijerph17093229
Chicago/Turabian StyleNordin, Abid, Shiplu Roy Chowdhury, Aminuddin Bin Saim, and Ruszymah Bt Hj Idrus. 2020. "Effect of Kelulut Honey on the Cellular Dynamics of TGFβ-Induced Epithelial to Mesenchymal Transition in Primary Human Keratinocytes" International Journal of Environmental Research and Public Health 17, no. 9: 3229. https://doi.org/10.3390/ijerph17093229
APA StyleNordin, A., Chowdhury, S. R., Saim, A. B., & Bt Hj Idrus, R. (2020). Effect of Kelulut Honey on the Cellular Dynamics of TGFβ-Induced Epithelial to Mesenchymal Transition in Primary Human Keratinocytes. International Journal of Environmental Research and Public Health, 17(9), 3229. https://doi.org/10.3390/ijerph17093229





