Impact of Electronic Alternatives to Tobacco Cigarettes on Indoor Air Particular Matter Levels
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Tests
2.2. Statistical Analysis
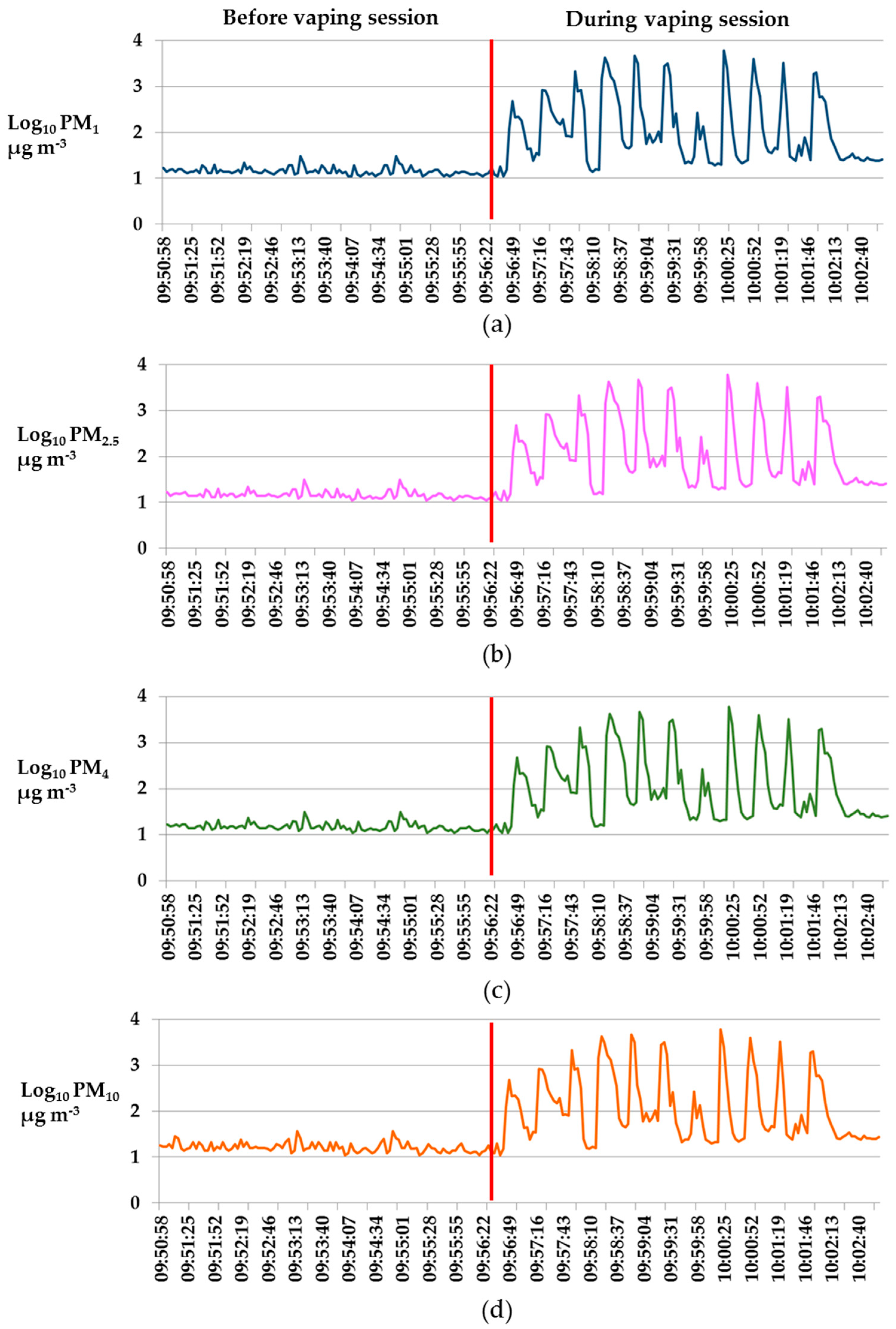
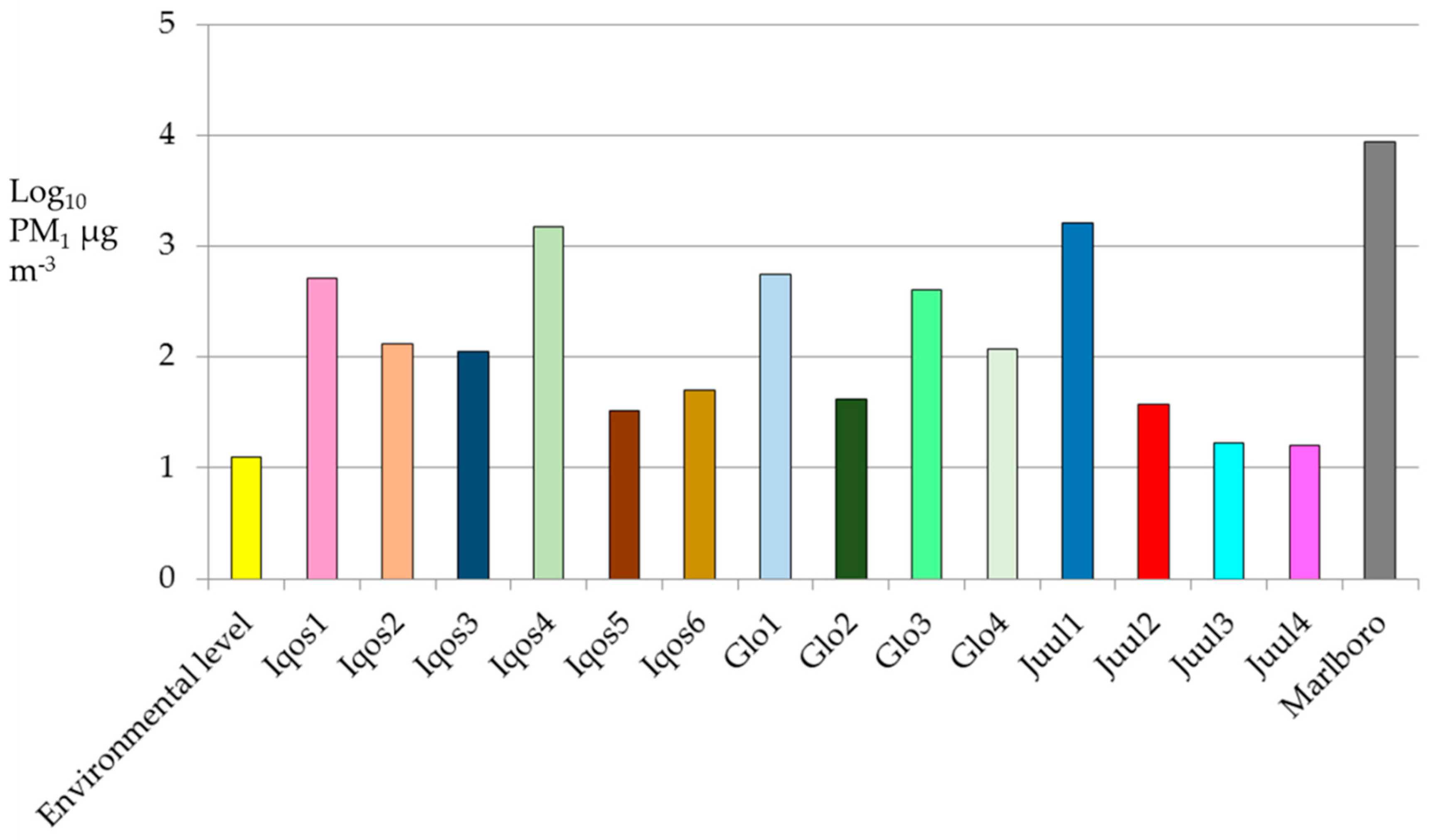
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Protano, C.; Di Milia, L.M.; Orsi, G.B.; Vitali, M. Electronic cigarette: A threat or an opportunity for public health? State of the art and future perspectives. Clin. Ter. 2015, 166, 32–37. [Google Scholar] [CrossRef]
- Protano, C.; Avino, P.; Manigrasso, M.; Vivaldi, V.; Perna, F.; Valeriani, F.; Vitali, M. Environmental Electronic Vape Exposure from Four Different Generations of Electronic Cigarettes: Airborne Particulate Matter Levels. Int. J. Environ. Res. Public Health 2018, 15, 2172. [Google Scholar] [CrossRef] [PubMed]
- Simonavicius, E.; McNeill, A.; Shahab, L.; Brose, L.S. Heat-not-burn tobacco products: A systematic literature review. Tob. Control 2019, 28, 582–594. [Google Scholar] [CrossRef] [PubMed]
- Barrington-Trimis, J.L.; Leventhal, A.M. Adolescents’ Use of “Pod Mod” E-Cigarettes—Urgent Concerns. N. Engl. J. Med. 2018, 379, 1099–1102. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Duncan, D.T.; El Shahawy, O.; Lee, L.; Shearston, J.A.; Tamura, K.; Sherman, S.E.; Weitzman, M. Characteristics of Adults Who Switched From Cigarette Smoking to E-cigarettes. Am. J. Prev. Med. 2017, 53, 652–660. [Google Scholar] [CrossRef]
- Choi, K.; Chen-Sankey, J.C. Will Electronic Nicotine Delivery System (EATC) use reduce smoking disparities? Prevalence of daily EATC use among cigarette smokers. Prev. Med. Rep. 2019, 17, 101020. [Google Scholar] [CrossRef]
- Spears, C.A.; Jones, D.M.; Weaver, S.R.; Huang, J.; Yang, B.; Pechacek, T.F.; Eriksen, M.P. Sociodemographic Correlates of Electronic Nicotine Delivery Systems (EATC) Use in the United States, 2016–2017. Am. J. Public Health 2019, 109, 1224–1232. [Google Scholar] [CrossRef]
- Jenssen, B.P.; Walley, S.C. Section on tobacco control. E-Cigarettes and Similar Devices. Pediatrics 2019, 143, e20183652. [Google Scholar] [CrossRef]
- Al-Hamdani, M.; Hopkins, D.B.; Park, T. Vaping among youth and young adults: A “red alert” state. J. Public Health Policy 2020, 41, 63–69. [Google Scholar] [CrossRef]
- Vallone, D.M.; Bennett, M.; Xiao, H.; Pitzer, L.; Hair, E.C. Prevalence and correlates of JUUL use among a national sample of youth and young adults. Tob. Control 2019, 28, 603–609. [Google Scholar] [CrossRef]
- Willett, J.G.; Bennett, M.; Hair, E.C.; Xiao, H.; Greenberg, M.S.; Harvey, E.; Cantrell, J.; Vallone, D. Recognition, use and perceptions of JUUL among youth and young adults. Tob. Control 2019, 28, 115–116. [Google Scholar] [CrossRef] [PubMed]
- Miech, R.; Johnston, L.; O’Malley, P.M.; Bachman, J.G.; Patrick, M.E. Adolescent vaping and nicotine use in 2017-2018—U.S. National Estimates. N. Engl. J. Med. 2019, 380, 192–193. [Google Scholar] [CrossRef] [PubMed]
- Cardenas, V.M.; Fischbach, L.A.; Chowdhury, P. The use of electronic nicotine delivery systems during pregnancy and the reproductive outcomes: A systematic review of the literature. Tob. Induc. Dis. 2019, 17, 52. [Google Scholar] [CrossRef] [PubMed]
- Mravec, B.; Tibensky, M.; Horvathova, L.; Babal, P. E-Cigarettes and Cancer Risk. Cancer Prev. Res. (Phila) 2020, 13, 137–144. [Google Scholar] [CrossRef]
- Walley, S.C.; Wilson, K.M.; Winickoff, J.P.; Groner, J. A Public Health Crisis: Electronic Cigarettes, Vape, and JUUL. Pediatrics 2019, 143, e20182741. [Google Scholar] [CrossRef]
- Ratajczak, A.; Jankowski, P.; Strus, P.; Feleszko, W. Heat Not Burn Tobacco Product-A New Global Trend: Impact of Heat-Not-Burn Tobacco Products on Public Health, a Systematic Review. Int. J. Environ. Res. Public Health 2020, 17, 409. [Google Scholar] [CrossRef]
- Jankowski, M.; Brożek, G.M.; Lawson, J.; Skoczyński, S.; Majek, P.; Zejda, J.E. New ideas, old problems? Heated tobacco products—A systematic review. Int. J. Occup. Med. Environ. Health 2019, 32, 595–634. [Google Scholar] [CrossRef]
- Marcham, C.L.; Springston, J.P. Electronic cigarettes in the indoor environment. Rev. Environ. Health 2019, 34, 105–124. [Google Scholar] [CrossRef]
- Manigrasso, M.; Vitali, M.; Protano, C.; Avino, P. Temporal evolution of ultrafine particles and of alveolar deposited surface area from main indoor combustion and non-combustion sources in a model room. Sci. Total Environ. 2017, 598, 1015–1026. [Google Scholar] [CrossRef]
- Protano, C.; Manigrasso, M.; Avino, P.; Vitali, M. Second-hand smoke generated by combustion and electronic smoking devices used in real scenarios: Ultrafine particle pollution and age-related dose assessment. Environ. Int. 2017, 107, 190–195. [Google Scholar] [CrossRef]
- Protano, C.; Manigrasso, M.; Avino, P.; Sernia, S.; Vitali, M. Second-hand smoke generated by new electronic device (IQOS and e-cigs) and traditional cigarettes: Submicron particle behaviour in human respiratory system. Ann. Ig. 2016, 28, 109–112. [Google Scholar] [CrossRef] [PubMed]
- Abdo, N.; Khader, Y.S.; Abdelrahman, M.; Graboski-Bauer, A.; Malkawi, M.; Al-Sharif, M.; Elbetieha, A.M. Respiratory health outcomes and air pollution in the Eastern Mediterranean Region: A systematic review. Rev. Environ. Health 2016, 31, 259–280. [Google Scholar] [CrossRef] [PubMed]
- An, Z.; Jin, Y.; Li, J.; Li, W.; Wu, W. Impact of particulate air pollution on cardiovascular health. Curr. Allergy Asthma Rep. 2018, 18, 15. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xiong, L.; Tang, M. Toxicity of inhaled particulate matter on the central nervous system: Neuroinflammation, neuropsychological effects and neurodegenerative disease. J. Appl. Toxicol. 2017, 37, 644–667. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer. Monographs on the Evaluation of Carcinogenic Risks to Humans; WHO Press: Lyon, France, 2015. [Google Scholar]
- Fernández, E.; Ballbè, M.; Sureda, X.; Fu, M.; Saltó, E.; Martínez-Sánchez, J.M. Particulate Matter from Electronic Cigarettes and Conventional Cigarettes: A Systematic Review and Observational Study. Curr. Environ. Health Rep. 2015, 2, 423–429. [Google Scholar] [CrossRef]
- Volesky, K.D.; Maki, A.; Scherf, C.; Watson, L.; Van Ryswyk, K.; Fraser, B.; Weichenthal, S.A.; Cassol, E.; Villeneuve, P.J. The influence of three e-cigarette models on indoor fine and ultrafine particulate matter concentrations under real-world conditions. Environ. Pollut. 2018, 243, 882–889. [Google Scholar] [CrossRef]
- Ruprecht, A.A.; De Marco, C.; Saffari, A.; Pozzi, P.; Mazza, R.; Veronese, C.V.; Angellotti, G.; Munarini, E.; Ogliari, A.C.; Westerdahl, D.; et al. Environmental pollution and emission factors of electronic cigarettes, heat-not-burn tobacco products, and conventional cigarettes. Aero Sci. Technol. 2017, 51, 674–684. [Google Scholar] [CrossRef]
- World Health Organization (WHO). WHO Air quality guidelines for Particulate Matter, Ozone, Nitrogen Dioxide, and Sulfur Dioxide; Global Update, 2005; Summary of Risk Assessment; WHO Press: Geneva, Switzerland, 2006. [Google Scholar]
- Manigrasso, M.; Protano, C.; Vitali, M.; Avino, P. Where Do Ultrafine Particles and Nano-Sized Particles Come From? J. Alzheimers Dis. 2019, 68, 1371–1390. [Google Scholar] [CrossRef]
- Manigrasso, M.; Vitali, M.; Protano, C.; Avino, P. Ultrafine particles in domestic environments: Regional doses deposited in the human respiratory system. Environ. Int. 2018, 118, 134–145. [Google Scholar] [CrossRef]
- Manigrasso, M.; Natale, C.; Vitali, M.; Protano, C.; Avino, P. Pedestrians in Traffic Environments: Ultrafine Particle Respiratory Doses. Int. J. Environ. Res. Public Health 2017, 14, 288. [Google Scholar] [CrossRef]
- Peters, A.; Dockery, D.W.; Muller, J.E.; Mittleman, M.A. Increased particulate air pollution and the triggering of myocardial infarction. Circulation 2001, 103, 2810–2815. [Google Scholar] [CrossRef] [PubMed]
- European Environment Agency. Air Quality in Europe—2019 Report No 10/2019. Available online: https://www.eea.europa.eu/publications/air-quality-in-europe-2019 (accessed on 1 March 2020).
- Kubesch, N.; De Nazelle, A.; Guerra, S.; Westerdahl, D.; Martinez, D.; Bouso, L.; Carrasco-Turigas, G.; Hoffmann, B.; Nieuwenhuijsen, M.J. Arterial blood pressure responses to short-term exposure to low and high traffic-related air pollution with and without moderate physical activity. Eur. J. Prev. Cardiol. 2015, 22, 548–557. [Google Scholar] [CrossRef] [PubMed]
- Duncan, M.S.; Freiberg, M.S.; Greevy, R.A., Jr.; Kundu, S.; Vasan, R.S.; Tindle, H.A. Association of smoking cessation with subsequent risk of cardiovascular disease. JAMA 2019, 322, 642–650. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Ruiz, C.A.; Andreas, S.; Lewis, K.; Tonnesen, P.; van Schayck, C.P.; Hajek, P.; Tonstad, S.; Dautzenberg, B.; Fletcher, M.; Masefield, S.; et al. Statement on smoking cessation in COPD and other pulmonary diseases and in smokers with comorbidities who find it difficult to quit. Eur. Respir. J. 2015, 46, 61–79. [Google Scholar] [CrossRef]
- Saito, E.; Inoue, M.; Tsugane, S.; Ito, H.; Matsuo, K.; Wakai, K.; Wada, K.; Nagata, C.; Tamakoshi, A.; Sugawara, Y.; et al. Smoking cessation and subsequent risk of cancer: A pooled analysis of eight population-based cohort studies in Japan. Cancer Epidemiol. 2017, 51, 98–108. [Google Scholar] [CrossRef]
- Mastrangeli, S.; Carnevale, R.; Cavarretta, E.; Sciarretta, S.; Peruzzi, M.; Marullo, A.G.M.; De Falco, E.; Chimenti, I.; Valenti, V.; Bullen, C.; et al. Predictors of oxidative stress and vascular function in an experimental study of tobacco versus electronic cigarettes: A post hoc analysis of the SUR-VAPES 1 Study. Tob. Induc. Dis. 2018, 16, 18. [Google Scholar] [CrossRef]
- Biondi-Zoccai, G.; Sciarretta, S.; Bullen, C.; Nocella, C.; Violi, F.; Loffredo, L.; Pignatelli, P.; Perri, L.; Peruzzi, M.; Marullo, A.G.M.; et al. Acute Effects of Heat-Not-Burn, Electronic Vaping, and Traditional Tobacco Combustion Cigarettes: The Sapienza University of Rome-Vascular Assessment of Proatherosclerotic Effects of Smoking (SUR—VAPES) 2 Randomized Trial. J. Am. Heart Assoc. 2019, 8, e010455. [Google Scholar] [CrossRef]
- Frati, G.; Carnevale, R.; Nocella, C.; Peruzzi, M.; Marullo, A.G.M.; De Falco, E.; Chimenti, I.; Cammisotto, V.; Valenti, V.; Cavarretta, E.; et al. Profiling the acute effects of modified risk products: Evidence from the SUR-VAPES (Sapienza University of Rome-Vascular Assessment of Proatherosclerotic Effects of Smoking) Cluster Study. Curr. Atheroscler. Rep. 2020, 22, 8. [Google Scholar] [CrossRef]
| EATC Types | Stick/Pod | Code |
|---|---|---|
| IQOS® | Heets Amber label | Iqos1 |
| Heets Blue label | Iqos2 | |
| Heets Bronze label | Iqos3 | |
| Heets Sienna label | Iqos4 | |
| Heets Turquoise label | Iqos5 | |
| Heets Yellow label | Iqos6 | |
| GLO® | Neo Aegean stick | Glo1 |
| Neo Beryl stick | Glo2 | |
| Neo Ultramarine stick | Glo3 | |
| Neo Yellow stick | Glo4 | |
| JUUL® | Golden Tobacco | Juul1 |
| Mango | Juul2 | |
| Mint | Juul3 | |
| Royal Creme | Juul4 | |
| Marlboro® gold | – | Marlboro |
| Experiment Code | Before Experiment | During Experiment (10–12 Puff for about 5–6 min) | p-Value (Calculated Probability) | ||
|---|---|---|---|---|---|
| Arithmetic Mean [Standard Deviation] | Median [Interquartile Range] | Arithmetic Mean [Standard Deviation] | Median [Interquartile Range] | ||
| Iqos1 | 14.2 [3.7] | 13.0 [3.0] | 517.7 [1048.9] | 70.0 [383.0] | <0.001 |
| Iqos2 | 14.6 [6.5] | 13.0 [2.0] | 132.0 [619.3] | 21.0 [12.0] | <0.001 |
| Iqos3 | 9.2 [1.5] | 9.0 [2.0] | 112.3 [469.4] | 11.0 [5.0] | <0.001 |
| Iqos4 | 9.1 [5.8] | 7.0 [1.0] | 1511.3 [2707.2] | 337.5 [1568.0] | <0.001 |
| Iqos5 | 23.7 [4.6] | 22.0 [6.0] | 32.4 [8.4] | 32.0 [16.0] | <0.001 |
| Iqos6 | 10.7 [2.1] | 10.0 [2.0] | 49.8 [128.8] | 21.0 [19.0] | <0.001 |
| Glo1 | 8.8 [2.4] | 8.0 [2.0] | 552.6 [1477.5] | 66.0 [205.5] | <0.001 |
| Glo2 | 8.7 [0.9] | 8.0 [1.0] | 42.0 [115.1] | 13.0 [9.0] | <0.001 |
| Glo3 | 26.9 [1.7] | 27.0 [2.0] | 406.5 [1335.6] | 31.0 [101.0] | <0.001 |
| Glo4 | 12.1 [1.9] | 12.0 [2.0] | 117.7 [336.8] | 23.0 [38.0] | <0.001 |
| Juul1 | 8.3 [2.3] | 7.5 [1.0] | 1637.9 [6387.6] | 110.0 [289.0] | <0.001 |
| Juul2 | 10.9 [1.5] | 10.0 [1.0] | 37.7 [208.3] | 11.0 [3.0] | <0.001 |
| Juul3 | 13.8 [1.9] | 13.0 [4.0] | 16.7 [5.4] | 15.0 [4.0] | <0.001 |
| Juul4 | 13.3 [1.5] | 13.0 [2.0] | 16.0 [5.0] | 15.0 [4.0] | <0.001 |
| Marlboro | 3.9 [1.2] | 4.0 [1.0] | 8638.5 [1,2215.9] | 3430.0 [8620.0] | <0.001 |
| Iqos2 | Iqos3 | Iqos4 | Iqos5 | Iqos6 | Glo1 | Glo2 | Glo3 | Glo4 | Juul1 | Juul2 | Juul3 | Juul4 | Marlboro Gold | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Iqos1 | * | * | * | * | * | * | * | * | * | * | * | * | * | * |
| Iqos2 | * | * | * | * | * | * | * | * | * | * | * | * | * | |
| Iqos3 | * | * | * | * | ns | * | * | * | ns | ns | ns | * | ||
| Iqos4 | * | * | * | * | * | * | * | * | * | * | * | |||
| Iqos5 | * | * | * | * | * | * | * | * | * | * | ||||
| Iqos6 | * | * | * | * | * | * | * | * | * | |||||
| Glo1 | * | * | * | * | * | * | * | * | ||||||
| Glo2 | * | * | * | ns | ns | ns | * | |||||||
| Glo3 | * | * | * | * | * | * | ||||||||
| Glo4 | * | * | * | * | * | |||||||||
| Juul1 | * | * | * | * | ||||||||||
| Juul2 | * | * | * | * | ||||||||||
| Juul3 | * | * | * | |||||||||||
| Juul4 | * | * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Protano, C.; Manigrasso, M.; Cammalleri, V.; Biondi Zoccai, G.; Frati, G.; Avino, P.; Vitali, M. Impact of Electronic Alternatives to Tobacco Cigarettes on Indoor Air Particular Matter Levels. Int. J. Environ. Res. Public Health 2020, 17, 2947. https://doi.org/10.3390/ijerph17082947
Protano C, Manigrasso M, Cammalleri V, Biondi Zoccai G, Frati G, Avino P, Vitali M. Impact of Electronic Alternatives to Tobacco Cigarettes on Indoor Air Particular Matter Levels. International Journal of Environmental Research and Public Health. 2020; 17(8):2947. https://doi.org/10.3390/ijerph17082947
Chicago/Turabian StyleProtano, Carmela, Maurizio Manigrasso, Vittoria Cammalleri, Giuseppe Biondi Zoccai, Giacomo Frati, Pasquale Avino, and Matteo Vitali. 2020. "Impact of Electronic Alternatives to Tobacco Cigarettes on Indoor Air Particular Matter Levels" International Journal of Environmental Research and Public Health 17, no. 8: 2947. https://doi.org/10.3390/ijerph17082947
APA StyleProtano, C., Manigrasso, M., Cammalleri, V., Biondi Zoccai, G., Frati, G., Avino, P., & Vitali, M. (2020). Impact of Electronic Alternatives to Tobacco Cigarettes on Indoor Air Particular Matter Levels. International Journal of Environmental Research and Public Health, 17(8), 2947. https://doi.org/10.3390/ijerph17082947










