The Relationship between the Migrant Population’s Migration Network and the Risk of COVID-19 Transmission in China—Empirical Analysis and Prediction in Prefecture-Level Cities
Abstract
1. Introduction
2. Methods
2.1. Data
2.1.1. China Migrant Population Dynamic Survey 2017 (CMDS 2017)
2.1.2. COVID-19 Infections Confirmed the Data from Prefecture-Level Cities
2.1.3. Cofactors for Disease Transmission
2.2. Residence–Birthplace Probability Matrix (R-B Matrix)
2.3. Correlation between the Transmission of the Disease and the Birthplace Probability of the Migrant Population by the R-B matrix
2.4. Prediction of Risk Levels in Prefecture-Level Cities and Similar Immigrant Cities and Outbreak Scenarios
3. Results
3.1. Construction of the R-B Matrix
3.2. Bivariate Correlation Test and Regression Results
3.3. Results of Prediction of Risk Levels in Prefecture-Level Cities with Outbreak Scenarios in Similar Immigrant Cities
3.4. Prefecture-Level City Risk Ranking of Disease Transmission
4. Discussion
4.1. COVID-19 Transmission in China is Highly Correlated with Factors that Return Migrants to Their Birthplace
4.2. Different Emergency Management and Control Plans for Different Disease Outbreak Scenarios in Immigrant Cities
4.3. Policy Intervention and Disease Prevention in the Important Migrant Birthplace Regions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Perlman, S. Another decade, another coronavirus. New Engl. J. Med. 2020, 382, 760–762. [Google Scholar] [CrossRef]
- Poletto, C.; Pelat, C.; Levy-Bruhl, D.; Yazdanpanah, Y.; Boelle, P.Y.; Colizza, V. Assessment of the Middle East respiratory syndrome coronavirus (MERS-CoV) epidemic in the Middle East and risk of international spread using a novel maximum likelihood analysis approach. Eurosurveillance 2014, 3. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Yao, L.; Wei, T.; Tian, F.; Jin, D.Y.; Chen, L.; Wang, M. Presumed Asymptomatic Carrier Transmission of COVID-19. J. Am. Med Assoc. 2020, in press. [Google Scholar] [CrossRef] [PubMed]
- Shen, M.; Peng, Z.; Xiao, Y.; Zhang, L. Modelling the disease trend of the 2019 novel coronavirus outbreak in China. bioRxiv 2020, in press. [Google Scholar]
- Wu, J.T.; Leung, K.; Eung, G.M. Nowcasting and forecasting the potential domestic and international spread of the 2019-nCoV outbreak originating in Wuhan, China: A modelling study. Lancet 2020, in press. [Google Scholar] [CrossRef]
- Keeling, M.J.; Eames, K.T. Networks and epidemic models. J. R. Soc. Interface 2005, 2, 295–307. [Google Scholar] [CrossRef]
- Firestone, S.M.; Christley, R.M.; Ward, M.P.; Dhand, N.K. Adding the spatial dimension to the social network analysis of an epidemic: Investigation of the 2007 outbreak of equine influenza in Australia. Prev. Vet. Med. 2012, 106, 123–135. [Google Scholar] [CrossRef]
- Handcock, M.S.; Hunter, D.R.; Butts, C.T.; Goodreau, S.M.; Morris, M. Statnet: Software tools for the Statistical Modeling of Network Data. SeattleWA. Version 2003, 2. [Google Scholar] [CrossRef]
- Diekmann, O.; De Jong, M.C.M.; Metz, J.A.J. A deterministic epidemic model taking account of repeated contacts between the same individuals. J. Appl. Probab. 1998, 35, 448–462. [Google Scholar] [CrossRef]
- Randolph, S.E.; Rogers, D.J. The arrival, establishment and spread of exotic diseases: Patterns and predictions. Nat. Rev. Microbiol. 2010, 8, 361–371. [Google Scholar] [CrossRef]
- Tatem, A.J.; Hay, S.I.; Rogers, D.J. Global traffic and disease vector dispersal. Proc. Natl. Acad. Sci. USA 2006, 103, 6242–6247. [Google Scholar] [CrossRef] [PubMed]
- Tatem, A.J.; Huang, Z.; Das, A.; Qi, Q.; Roth, J.; Qiu, Y. Air travel and vector-borne disease movement. Parasitology 2012, 139, 1816–1830. [Google Scholar] [CrossRef] [PubMed]
- Suk, J.E.; Van Cangh, T.; Beaute, J.; Bartels, C.; Tsolova, S.; Pharris, A.; Semenza, J.C. The interconnected and cross-border nature of risks posed by infectious diseases. Glob. Health Action 2014, 7, 25287. [Google Scholar] [CrossRef] [PubMed]
- Wan, X.; Liu, J.; Cheung, W.K.; Tong, T. Inferring epidemic network topology from surveillance data. PLoS ONE 2014, 9, e100661. [Google Scholar] [CrossRef][Green Version]
- Dangerfield, C.E.; Ross, J.V.; Keeling, M.J. Integrating stochasticity and network structure into an epidemic model. J. R. Soc. Interface 2009, 6, 761–774. [Google Scholar] [CrossRef]
- Eames, K.T.; Read, J.M.; Edmunds, W.J. Epidemic prediction and control in weighted networks. Epidemics 2009, 1, 70–76. [Google Scholar] [CrossRef]
- Wilder-Smith, A.; Leong, W.Y. Importation of yellow fever into China: Assessing travel patterns. J. Travel Med. 2017, 24, tax008. [Google Scholar] [CrossRef]
- Wang, L.; Li, X.; Zhang, Y.Q.; Zhang, Y.; Zhang, K. Evolution of scaling emergence in large-scale spatial epidemic spreading. PLoS ONE 2011, 6, e21197. [Google Scholar] [CrossRef]
- Fadel, F.; Khalil El Karoui, M.D.; Knebelmann, B. Spread of a novel influenza A (H1N1) virus via global airline transportation. Nat. Rev. Immunol. 2008, 8, 153–160. [Google Scholar]
- Lopez, L.F.; Amaku, M.; Coutinho, F.A.B.; Quam, M.; Burattini, M.N.; Struchiner, C.J.; Massad, E. Modeling importations and exportations of infectious diseases via travelers. Bull. Math. Biol. 2016, 78, 185–209. [Google Scholar] [CrossRef]
- World Pop. Preliminary Risk Analysis of 2019 Novel Coronavirus Spread within and Beyond China. Available online: https://www.worldpop.org/events/china (accessed on 15 February 2020).
- Huang, H.; Hailin, Y.; Xin, D.; Peng, Z.; Yong, L.; Zhang, L.; Lei, Z. Influencing Mechanisms of Urban Heat Island on Respiratory Diseases. Iran. J. Public Health 2019, 48, 1636. [Google Scholar] [PubMed]
- Zhang, C.; Wang, M. Origin time and epidemic dynamics of the 2019 novel coronavirus. bioRxiv 2020, in press. [Google Scholar]
- Read, J.M.; Bridgen, J.R.; Cummings, D.A.; Ho, A.; Jewell, C.P. Novel coronavirus 2019-nCoV: Early estimation of epidemiological parameters and epidemic predictions. medRxiv 2020, in press. [Google Scholar]
- Li, Q.; Guan, X.; Wu, P.; Wang, X.; Zhou, L.; Tong, Y.; Xing, X. Early transmission dynamics in Wuhan, China, of novel coronavirus–infected pneumonia. New Engl. J. Med. 2020, in press. [Google Scholar] [CrossRef] [PubMed]
- Getz, W.M.; Pickering, J. Epidemic models: Thresholds and population regulation. Am. Nat. 1983, 121, 892–898. [Google Scholar] [CrossRef]
- Mangen, M.J.; Nielen, M.; Burrell, A.M. Simulated effect of pig-population density on epidemic size and choice of control strategy for classical swine fever epidemics in The Netherlands. Prev. Vet. Med. 2002, 56, 141–163. [Google Scholar] [CrossRef]
- Balcan, D.; Gonçalves, B.; Hu, H.; Ramasco, J.J.; Colizza, V.; Vespignani, A. Modeling the spatial spread of infectious diseases: The GLobal Epidemic and Mobility computational model. J. Comput. Sci. 2010, 1, 132–145. [Google Scholar] [CrossRef]
- Bereitschaft, B.; Debbage, K. Urban form, air pollution, and CO2 emissions in large US metropolitan areas. Prof. Geogr. 2013, 65, 612–635. [Google Scholar] [CrossRef]
- Fan, C.; Tian, L.; Zhou, L.; Hou, D.; Song, Y.; Qiao, X.; Li, J. Examining the impacts of urban form on air pollutant emissions: Evidence from China. J. Environ. Manag. 2018, 212, 405–414. [Google Scholar] [CrossRef]
- Zhou, L.; Tian, L.; Gao, Y.; Ling, Y.; Fan, C.; Hou, D.; Zhou, W. How did industrial land supply respond to transitions in state strategy? An analysis of prefecture-level cities in China from 2007 to 2016. Land Use Policy 2019, 87, 104009. [Google Scholar] [CrossRef]
- Riley, S.; Ferguson, N.M. Smallpox transmission and control: Spatial dynamics in Great Britain. Proc. Natl. Acad. Sci. USA 2006, 103, 12637–12642. [Google Scholar] [CrossRef] [PubMed]
- Riley, S.; Eames, K.; Isham, V.; Mollison, D.; Trapman, P. Five challenges for spatial epidemic models. Epidemics 2015, 10, 68–71. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Miao, S.; Sarkar, C.; Fan, L.; Li, Z. Do neighborhood ties matter for residents’ mental health in affordable housing: Evidence from Guangzhou, China. Cities 2020, 100, 102666. [Google Scholar] [CrossRef]
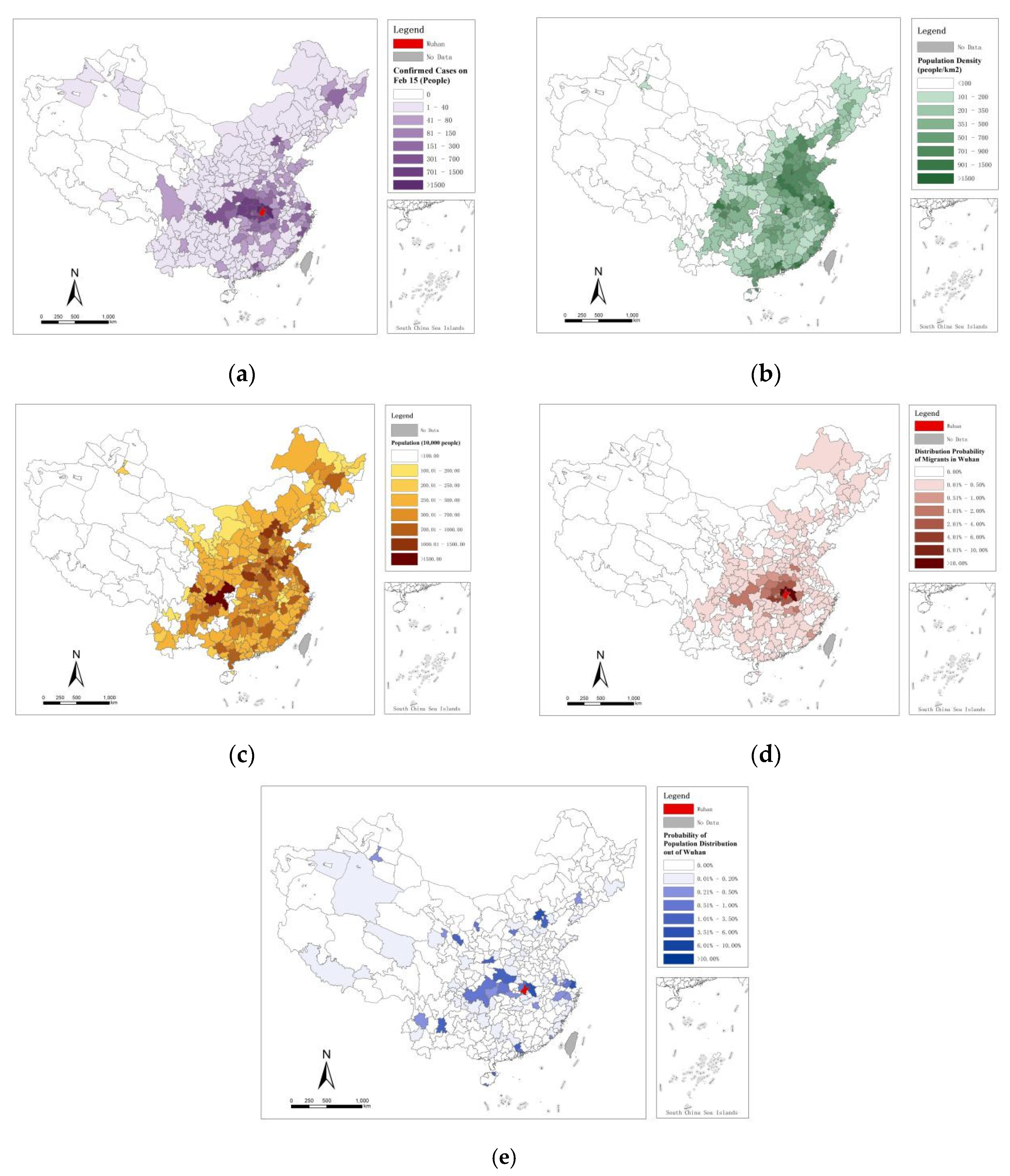
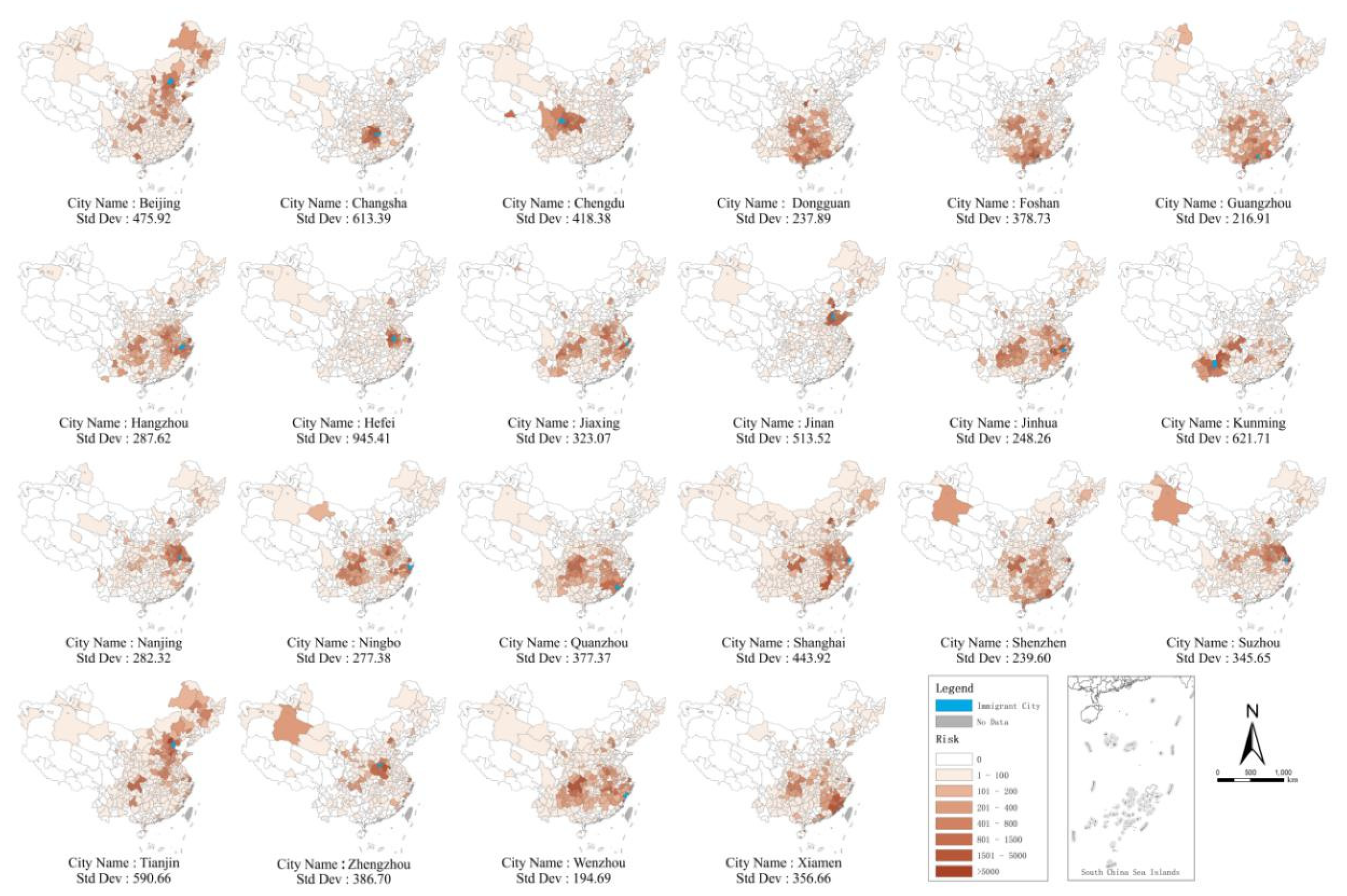
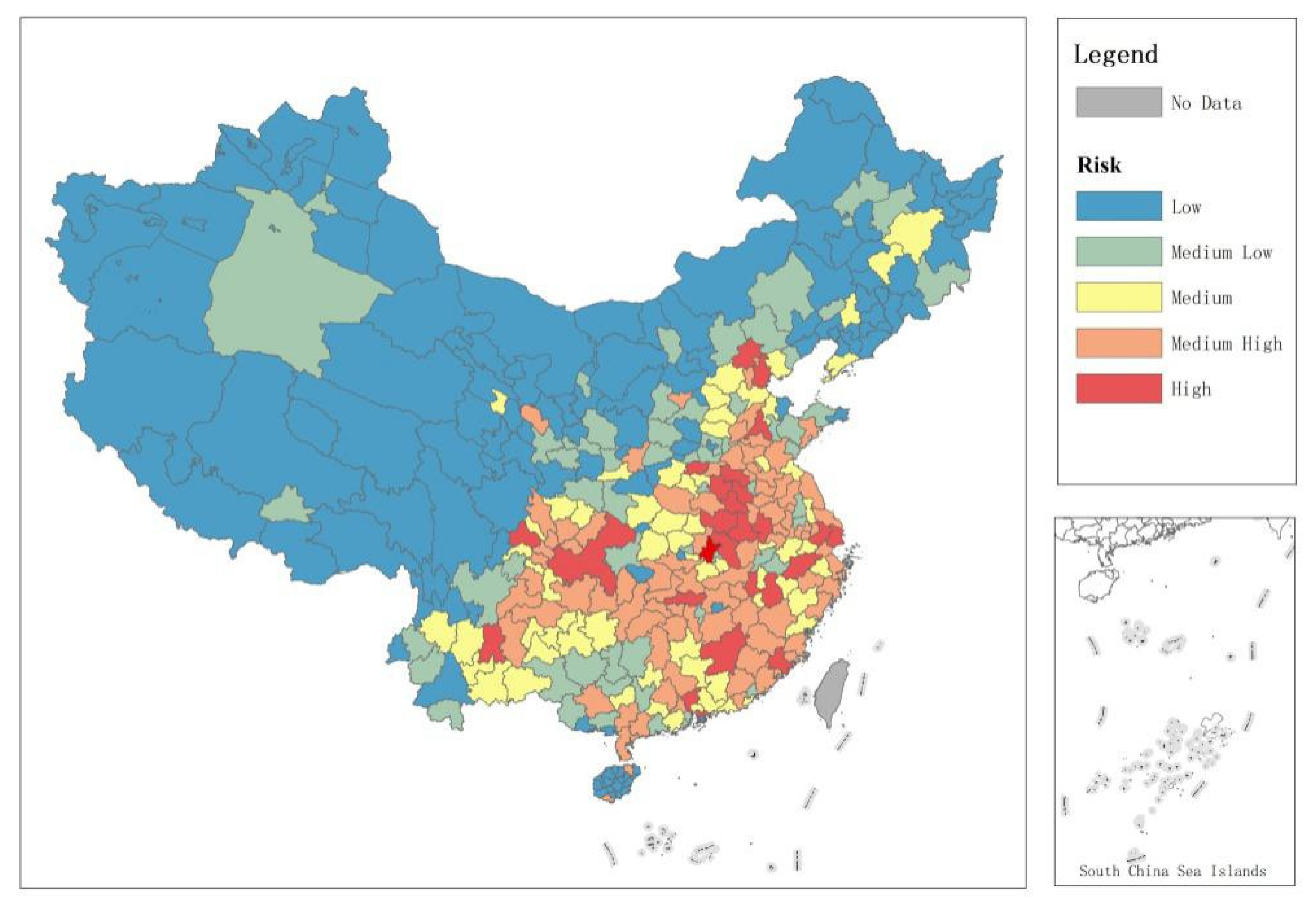
| t1 | t2 | t3 | t4 | t5 | t6 | t7 | t8 | t9 | t10 | t11 | t12 | t13 | t14 | t15 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0.157 ** | 0.146 ** | 0.142 * | 0.136 * | 0.129 * | 0.128 * | 0.128 * | 0.127 * | 0.126 * | 0.125 * | 0.125 * | 0.120 * | 0.117 * | 0.117 * | 0.117 * | |
| 0.232 ** | 0.220 ** | 0.212 ** | 0.200 ** | 0.187 ** | 0.182 ** | 0.177 ** | 0.175 ** | 0.173 ** | 0.172 ** | 0.170 ** | 0.162 ** | 0.158 ** | 0.155 ** | 0.154 ** | |
| 0.836 ** | 0.849 ** | 0.857 ** | 0.883 ** | 0.907 ** | 0.915 ** | 0.917 ** | 0.915 ** | 0.915 ** | 0.915 ** | 0.916 ** | 0.913 ** | 0.915 ** | 0.916 ** | 0.918 ** | |
| 0.543 ** | 0.534 ** | 0.514 ** | 0.500 ** | 0.477 ** | 0.466 ** | 0.463 ** | 0.461 ** | 0.458 ** | 0.456 ** | 0.453 ** | 0.447 ** | 0.445 ** | 0.439 ** | 0.436 ** | |
| t1 | t2 | t3 | t4 | t5 | t6 | t7 | t8 | t9 | t10 | t11 | t12 | t13 | t14 | t15 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Beta | Beta | Beta | Beta | Beta | Beta | Beta | Beta | Beta | Beta | Beta | Beta | Beta | Beta | Beta | |
| 0.003 | −0.003 | 0.003 | 0.008 | 0.017 | 0.024 | 0.029 | 0.030 | 0.031 | 0.031 | 0.034 | 0.034 | 0.034 | 0.038 | 0.040 | |
| 0.119 ** | 0.108 ** | 0.102 * | 0.082 * | 0.063 | 0.056 | 0.049 | 0.048 | 0.047 | 0.046 | 0.044 | 0.036 | 0.032 | 0.029 | 0.027 | |
| 0.715 *** | 0.734 *** | 0.751 *** | 0.786 *** | 0.823 *** | 0.834 *** | 0.837 *** | 0.836 *** | 0.836 *** | 0.837 *** | 0.838 *** | 0.839 *** | 0.842 *** | 0.846 *** | 0.849 *** | |
| 0.303 *** | 0.295 *** | 0.270 *** | 0.250 *** | 0.221 *** | 0.206 *** | 0.204 *** | 0.202 *** | 0.200 *** | 0.198 *** | 0.195 *** | 0.191 *** | 0.190 *** | 0.183 *** | 0.179 *** | |
| R2 | 0.826 | 0.835 | 0.836 | 0.865 | 0.881 | 0.887 | 0.887 | 0.883 | 0.882 | 0.880 | 0.880 | 0.883 | 0.885 | 0.885 | 0.887 |
| N | 329 | ||||||||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fan, C.; Cai, T.; Gai, Z.; Wu, Y. The Relationship between the Migrant Population’s Migration Network and the Risk of COVID-19 Transmission in China—Empirical Analysis and Prediction in Prefecture-Level Cities. Int. J. Environ. Res. Public Health 2020, 17, 2630. https://doi.org/10.3390/ijerph17082630
Fan C, Cai T, Gai Z, Wu Y. The Relationship between the Migrant Population’s Migration Network and the Risk of COVID-19 Transmission in China—Empirical Analysis and Prediction in Prefecture-Level Cities. International Journal of Environmental Research and Public Health. 2020; 17(8):2630. https://doi.org/10.3390/ijerph17082630
Chicago/Turabian StyleFan, Chenjing, Tianmin Cai, Zhenyu Gai, and Yuerong Wu. 2020. "The Relationship between the Migrant Population’s Migration Network and the Risk of COVID-19 Transmission in China—Empirical Analysis and Prediction in Prefecture-Level Cities" International Journal of Environmental Research and Public Health 17, no. 8: 2630. https://doi.org/10.3390/ijerph17082630
APA StyleFan, C., Cai, T., Gai, Z., & Wu, Y. (2020). The Relationship between the Migrant Population’s Migration Network and the Risk of COVID-19 Transmission in China—Empirical Analysis and Prediction in Prefecture-Level Cities. International Journal of Environmental Research and Public Health, 17(8), 2630. https://doi.org/10.3390/ijerph17082630




