Persistent Post-Traumatic Headache and Migraine: Pre-Clinical Comparisons
Abstract
1. Introduction
2. Methods
2.1. Eligibility Criteria
2.2. Source of Information
2.3. Search Strategy
3. Results
3.1. Sensory Differences between Migraine and PPTH
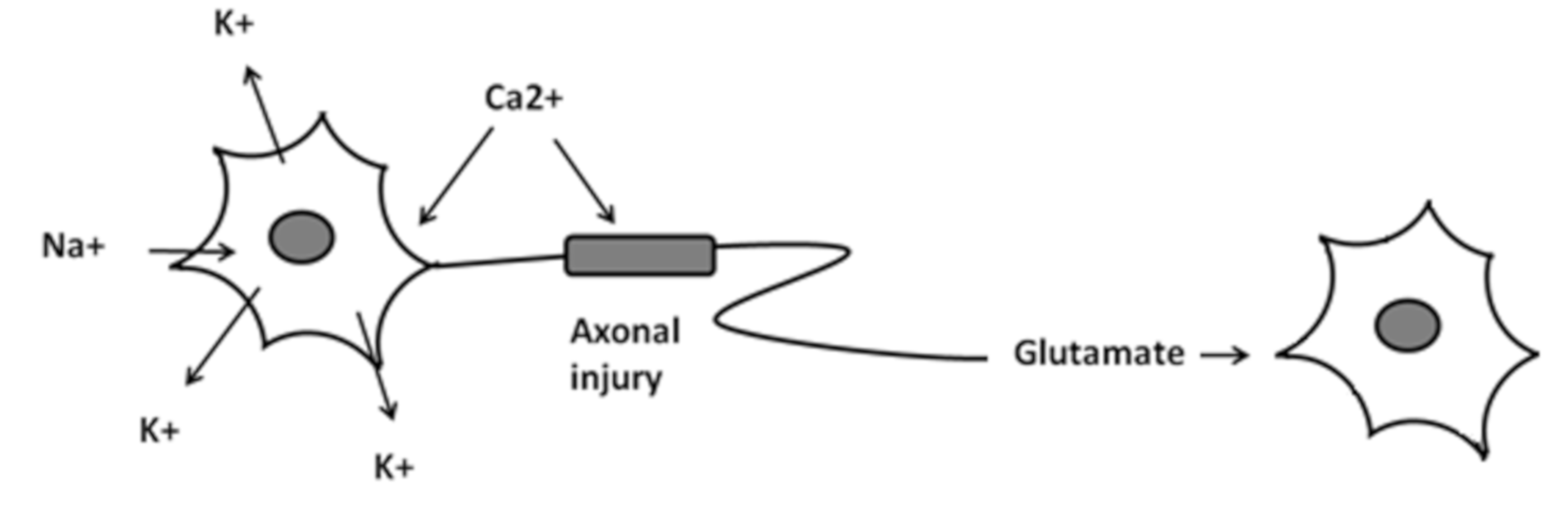
3.2. Biochemical Alteration and Pathophysiology
4. Treatment Approach
4.1. NSAIDs (Non-Steroidal Anti-Inflammatory Drugs) Drugs
4.2. Triptans Drugs
4.3. Tricyclic Antidepressants Drugs
4.4. Antiepileptics Drugs
4.5. Monoclonal Antibodies
5. Discussion
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Headache Classification Subcommittee of the International Headache Society. The International Classification of Headache Disorders: 2nd edition. Cephalalgia 2004, 24, 9–160. [Google Scholar]
- Lucas, S.; Hoffman, J.M.; Bell, K.R.; Walker, W.; Dikmen, S. Characterization of headache after traumatic brain injury. Cephalalgia 2012, 32, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Ashina, H.; Porreca, F.; Anderson, T.; Amin, F.M.; Ashina, M.; Schytz, H.W.; Dodick, D.W. Post-traumatic headache: Epidemiology and pathophysiological insights. Nat. Rev. Neurol. 2019, 15, 607–617. [Google Scholar] [CrossRef] [PubMed]
- Schwedt, T.J.; Chong, C.D.; Peplinski, J.; Ross, K.; Berisha, V. Persistent post-traumatic headache vs. migraine: An MRI study demonstrating differences in brain structure. J. Headache Pain 2017, 18, 8. [Google Scholar] [CrossRef] [PubMed]
- The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia 2013, 33, 629–808. [CrossRef] [PubMed]
- Ruff, R.L.; Blake, K. Pathophysiological links between traumatic brain injury and post-traumatic headaches. F1000 Res. 2016, 5. [Google Scholar] [CrossRef]
- Kamins, J.; Charles, A. Posttraumatic Headache: Basic Mechanisms and Therapeutic Targets. Posttraumatic Headache: Basic Mechanisms and Therapeutic Targets. Headache 2018, 58, 811–826. [Google Scholar] [CrossRef]
- Seifert, T.; Sufrinko, A.; Cowan, R.; Black, W.S.; Watson, D.; Edwards, B.; Livingston, S.; Webster, K.; Akers, D.; Lively, M.; et al. Comprehensive headache experience in collegiate student- athletes: An initial report from the NCAA Headache Task Force. Headache 2017, 57, 877–886. [Google Scholar] [CrossRef]
- Hoffman, J.M.; Lucas, S.; Dikmen, S.; Braden, C.A.; Brown, A.W.; Brunner, R.; Diaz-Arrastia, R.; Walker, W.C.; Watanabe, T.K.; Bell, K.R. Natural history of headache after traumatic brain injury. J. Neurotrauma 2011, 28, 1719–1725. [Google Scholar] [CrossRef]
- Bree, D.; Levy, D. Development of CGRP-dependent pain and headache related behaviours in a rat model of concussion: Implications for mechanisms of post-traumatic headache. Cephalalgia 2018, 38, 246–258. [Google Scholar] [CrossRef]
- Larsen, E.L.; Ashina, H.; Iljazi, A.; Al-Khazali, H.M.; Seem, K.; Ashina, M.; Ashina, S.; Schytz, H.W. Acute and preventive pharmacological treatment of post-traumatic headache: A systematic review. J. Headache Pain 2019, 20, 98. [Google Scholar] [CrossRef] [PubMed]
- Baandrup, L.; Jensen, R. Chronic post-traumatic headache—A clinical analysis in relation to the international headache classification 2nd edition. Cephalalgia 2005, 25, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Kjeldgaard, D.; Forchhammer, H.; Teasdale, T.; Jensen, R.H. Chronic post-traumatic headache aftermild head injury: A descriptive study. Cephalalgia 2014, 34, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Pomes, L.M.; Guglielmetti, M.; Bertamino, E.; Simmaco, M.; Borro, M.; Martelletti, P. Optimising migraine treatment: From drug-drug interactions to personalized medicine. J. Headache Pain 2019, 20, 56. [Google Scholar] [CrossRef] [PubMed]
- Dumkrieger, G.; Chong, C.D.; Ross, K.; Berisha, V.; Schwedt, T.J. Static and dynamic functional connectivity differences between migraine and persistent post-traumatic headache: A resting-state magnetic resonance imaging study. Cephalalgia 2019, 39, 1366–1381. [Google Scholar] [CrossRef] [PubMed]
- Van der Horn, H.J.; Liemburg, E.J.; Scheenen, M.E.; de Koning, M.E.; Marsman, J.B.; Spikman, J.M.; van der Naalt, J. Brain network dysregulation, emotion, and complaints after mild traumatic brain injury. Hum. Brain Mapp. 2016, 37, 1645–1654. [Google Scholar] [CrossRef]
- Levy, D.; Gruener, H.; Riabinin, M.; Feingold, Y.; Schreiber, S.; Pick, C.G.; Defrin, R. Different clinical phenotypes of persistent post-traumatic headache exhibit distinct sensory profiles. Cephalalgia 2019, 1–14. [Google Scholar] [CrossRef]
- Kelman, L. Migraine pain location: A tertiary care study of 1283 migraineurs. Headache 2005, 45, 1038–1047. [Google Scholar] [CrossRef]
- Nordhaug, L.H.; Hagen, K.; Vik, A.; Stovner, L.J.; Follestad, T.; Pedersen, T.; Gravdahl, G.B.; Linde, M. Headache following head injury: A population-based longitudinal cohort study (HUNT). J. Headache Pain 2018, 19, 8. [Google Scholar] [CrossRef]
- Pettus, E.H.; Povlishock, J.T. Characterization of a distinct set of intra-axonal ultrastructural changes associated with traumatically induced alteration in axolemmal permeability. Brain Res. 1996, 722, 1–11. [Google Scholar] [CrossRef]
- Povlishock, J.T. Traumatically induced axonal injury: Pathogenesis and pathobiological implications. Brain Pathol. 1992, 2, 1–12. [Google Scholar] [PubMed]
- Dalkara, T.; Nozari, A.; Moskowitz, M.A. Migraine aura pathophysiology: The role of blood vessels and microembolisation. Lancet Neurol. 2010, 9, 309–317. [Google Scholar] [CrossRef]
- Sanchez-Del-Rio, M.; Reuter, U.; Moskowitz, M.A. New insights into migraine pathophysiology. Curr. Opin. Neurol. 2006, 19, 294–298. [Google Scholar] [CrossRef] [PubMed]
- Bolay, H.; Reuter, U.; Dunn, A.K.; Huang, Z.; Boas, D.A.; Moskowitz, M.A. Intrinsic brain activity triggers trigeminal meningeal afferents in a migraine model. Nat. Med. 2002, 8, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Gursoy-Ozdemir, Y.; Qiu, J.; Matsuoka, N.; Bolay, H.; Bermpohl, D.; Jin, H.; Wang, X.; Rosenberg, G.A.; Lo, E.H.; Moskowitz, M.A. Cortical spreading depression activates and upregulates MMP-9. J. Clin. Investig. 2004, 113, 1447–1455. [Google Scholar] [CrossRef] [PubMed]
- Karatas, H.; Erdener, S.E.; Gursoy-Ozdemir, Y.; Lule, S.; Eren-Koçak, E.; Sen, Z.D.; Dalkara, T. Spreading depression triggers headache by activating neuronal Panx1 channels. Science 2013, 339, 1092–1095. [Google Scholar] [CrossRef]
- Martins-Oliveira, A.; Gonçalves, F.M.; Speciali, J.G.; Fontana, V.; Izidoro-Toledo, T.C.; Belo, V.A.; Dach, F.; Tanus-Santos, J.E. Specific matrix metalloproteinase 9 (MMP-9) haplotype affect the circulating MMP-9 levels in women with migraine. J. Neuroimmunol. 2012, 252, 89–94. [Google Scholar] [CrossRef]
- Chen, G.; Shi, J.; Hu, Z.; Hang, C. Inhibitory effect on cerebral inflammatory response following traumatic brain injury in rats: A potential neuroprotective mechanism of N-acetylcysteine. Mediators Inflamm. 2008, 2008, 716458. [Google Scholar] [CrossRef]
- Eakin, K.; Baratz-Goldstein, R.; Pick, C.G.; Zindel, O.; Balaban, C.D.; Hoffer, M.E.; Lockwood, M.; Miller, J.; Hoffer, B.J. Efficacy of N-acetyl cysteine in traumatic brain injury. PLoS ONE 2014, 9, e90617. [Google Scholar] [CrossRef]
- Hoffer, M.E.; Balaban, C.; Slade, M.D.; Tsao, J.W.; Hoffer, B. Amelioration of acute sequelae of blast induced mild traumatic brain injury by N- acetyl cysteine: A double- blind, placebo controlled study. PLoS ONE 2013, 8, e54163. [Google Scholar] [CrossRef]
- Lucchesi, C.; Baldacci, F.; Cafalli, M.; Chico, L.; Lo Gerfo, A.; Bonuccelli, U.; Siciliano, G.; Gori, S. Evidences of Reduced Antioxidant Activity in Patients with Chronic Migraine and Medication-Overuse Headache. Headache 2015, 55, 984–991. [Google Scholar] [CrossRef]
- Sparaco, M.; Feleppa, M.; Lipton, R.B.; Rapoport, A.M.; Bigal, M.E. Mitochondrial dysfunction and migraine: Evidence and hypotheses. Cephalalgia 2006, 26, 361–372. [Google Scholar] [CrossRef]
- Okada, H.; Araga, S.; Takeshima, T.; Nakashima, K. Plasma lactic acid and pyruvic levels in migraine and tension type headache. Headache 1998, 38, 39–42. [Google Scholar] [CrossRef]
- Razeghi Jahromi, S.; Ghorbani, Z.; Martelletti, P.; Lampl, C.; Togha, M. School of Advanced Studies of the European Headache Federation (EHF-SAS). Association of diet and headache. J. Headache Pain 2019, 20, 106. [Google Scholar] [CrossRef]
- Srikiatkhachorn, A.; le Grand, S.M.; Supornsilpchai, W.; Storer, R.J. Pathophysiology of medication over use headache—An update. Headache 2014, 54, 204–210. [Google Scholar] [CrossRef]
- Ashina, H.; Schytz, H.W.; Ashina, M. CGRP in human models of migraine. In Handbook of Experimental Pharmacology; Springer: Cham, Switzerland, 2018. [Google Scholar]
- Daiutolo, B.V.; Tyburski, A.; Clark, S.W.; Elliott, M.B. Trigeminal pain molecules, allodynia, and photosensitivity are pharmacologically and genetically modulated in a model of traumatic brain injury. J. Neurotrauma 2016, 33, 748–760. [Google Scholar] [CrossRef]
- Tyburski, A.L.; Cheng, L.; Assari, S.; Darvish, K.; Elliott, M.B. Frequent mild head injury promotes trigeminal sensitivity concomitant with microglial proliferation, astrocytosis, and increased neuropeptide levels in the trigeminal pain system. J. Headache Pain 2017, 18, 16. [Google Scholar] [CrossRef]
- Eftekhari, S.; Salvatore, C.A.; Calamari, A.; Kane, S.A.; Tajti, J.; Edvinsson, L. Differential distribution of calcitonin gene-related peptide and its receptor components in the human trigeminal ganglion. Neuroscience 2010, 169, 683–696. [Google Scholar] [CrossRef]
- Eftekhari, S.; Gaspar, R.C.; Roberts, R.; Chen, T.-B.; Zeng, Z.; Villarreal, S.; Edvinsson, L.; Salvatore, C.A. Localization of CGRP receptor components and receptor binding sites in rhesus monkey brainstem: A detailed study using in situ hybridization, immunofluorescence, and autoradiography. J. Comp. Neurol. 2016, 524, 90–118. [Google Scholar] [CrossRef]
- Manning, B.M.; Gruba, S.M.; Meyer, A.F.; Haynes, C.L. Neuropeptide-induced mast cell degranulation and characterization of signaling modulation in Response to IgE conditioning. ACS Chem. Biol. 2016, 11, 3077–3083. [Google Scholar] [CrossRef]
- Mayer, C.L.; Huber, B.R.; Peskind, E. Traumatic brain injury, neuroinflammation, and post-traumatic headaches. Headache 2013, 53, 1523–1530. [Google Scholar] [CrossRef]
- Hansen, J.M.; Hauge, A.W.; Olesen, J.; Ashina, M. Calcitonin generelated peptide triggers migraine-like attacks in patients with migraine with aura. Cephalalgia 2010, 30, 1179–1186. [Google Scholar] [CrossRef]
- Navratilova, E.; Rau, J.; Oyarzo, J.; Tien, J.; Mackenzie, K.; Stratton, J.; Remeniuk, B.; Schwedt, T.; Anderson, T.; Dodick, D.; et al. CGRP-dependent and independent mechanisms of acute and persistent post-traumatic headache following mild traumatic brain injury in mice. Cephalalgia 2019, 39, 1762–1775. [Google Scholar] [CrossRef]
- Wang, X.; Fang, Y.; Liang, J.; Yin, Z.; Miao, J.; Luo, N. Selective inhibition of 5-HT7 receptor reduces CGRP release in an experimental model for migraine. Headache 2010, 50, 579–587. [Google Scholar] [CrossRef]
- De Felice, M.; Ossipov, M.H.; Wang, R.; Lai, J.; Chichorro, J.; Meng, I.; Dodick, D.W.; Vanderah, T.W.; Dussor, G.; Porreca, F. Triptaninduced latent sensitization: A possible basis for medication overuse headache. Ann. Neurol. 2010, 67, 325–337. [Google Scholar]
- Defrin, R.; Gruener, H.; Schreiber, S.; Pick, C.G. Quantitative somatosensory testing of subjects with chronic post- traumatic headache: Implications on its mechanisms. Eur. J. Pain 2010, 14, 924–931. [Google Scholar] [CrossRef]
- Burstein, R.; Yarnitsky, D.; Goor-Aryeh, I.; Ransil, B.J.; Bajwa, Z.H. An association between migraine and cutaneous allodynia. Ann. Neurol. 2000, 47, 614–624. [Google Scholar] [CrossRef]
- Mustafa, G.; Hou, J.; Tsuda, S.; Nelson, R.; Sinharoy, A.; Wilkie, Z.; Pandey, R.; Caudle, R.M.; Neubert, J.K.; Thompson, F.J.; et al. Trigeminal neuroplasticity underlies allodynia in a preclinical model of mild closed head traumatic brain injury (cTBI). Neuropharmacology 2016, 107, 27–39. [Google Scholar] [CrossRef]
- Walker, W.C.; Franke, L.M.; McDonald, S.D.; Sima, A.P.; Keyser-Marcus, L. Prevalence of mental health conditions after military blast exposure, their co-occurrence, and their relation to mild traumatic brain injury. Brain Inj. 2015, 29, 1581–1588. [Google Scholar] [CrossRef]
- Chrisman, S.P.; Richardson, L.P. Prevalence of diagnosed depression in adolescents with history of concussion. J. Adolesc. Health 2014, 54, 582–586. [Google Scholar] [CrossRef]
- Jaramillo, C.A.; Eapen, B.C.; McGeary, C.A.; McGeary, D.D.; Robinson, J.; Amuan, M.; Pugh, M.J. A cohort study examining headaches among veterans of Iraq and Afghanistan wars: Associations with traumatic brain injury, PTSD, and depression. Headache 2016, 56, 528–539. [Google Scholar] [CrossRef]
- Capi, M.; Gentile, G.; Lionetto, L.; Salerno, G.; Cipolla, F.; Curto, M.; Borro, M.; Martelletti, P. Pharmacogenetic considerations for migraine therapies. Expert Opin. Drug Metab. Toxicol. 2018, 14, 1161–1167. [Google Scholar] [CrossRef]
- Vandenbussche, N.; Laterza, D.; Lisicki, M.; Lloyd, J.; Lupi, C.; Tischler, H.; Toom, K.; Vandervorst, F.; Quintana, S.; Paemeleire, K.; et al. Medication-overuse headache: A widely recognized entity amidst ongoing debate. J. Headache Pain 2018, 19, 50. [Google Scholar] [CrossRef]
- Benemei, S.; Cortese, F.; Labastida-Ramírez, A.; Marchese, F.; Pellesi, L.; Romoli, M.; Vollesen, A.L.; Lampl, C.; Ashina, M. School of Advanced Studies of the European Headache Federation (EHF-SAS). Triptans and CGRP blockade—Impact on the cranial vasculature. J. Headache Pain 2017, 18, 103. [Google Scholar] [CrossRef]
- Erickson, J.C. Treatment outcomes of chronic post-traumatic headaches after mild head trauma in US soldiers: An observational study. Headache 2011, 51, 932–944. [Google Scholar] [CrossRef]
- Lords, Q.; Greene, J.P. Traumatic migraine versus concussion: A case report. Sports Health 2014, 6, 406–409. [Google Scholar] [CrossRef]
- Couch, J.R. Amitriptyline Versus Placebo Study Group. Amitriptyline in the prophylactic treatment of migraine and chronic daily headache. Headache 2011, 51, 33–35. [Google Scholar] [CrossRef]
- Hurwitz, M.; Lucas, S.; Bell, K.R.; Temkin, N.; Dikmen, S.; Hoffman, J. Use of Amitriptyline in the Treatment of Headache After Traumatic Brain Injury: Lessons Learned from a Clinical Trial. Headache 2020, 60, 713–723. [Google Scholar] [CrossRef]
- Steiner, T.J.; Jensen, R.; Katsarava, Z.; Linde, M.; MacGregor, E.A.; Osipova, V.; Paemeleire, K.; Olesen, J.; Peters, M.; Martelletti, P. Aids to management of headache disorders in primary care (2nd edition): On behalf of the European Headache Federation and Lifting the Burden: The Global Campaign against Headache. J. Headache Pain 2019, 20, 57. [Google Scholar] [CrossRef]
- Sacco, S.; Bendtsen, L.; Ashina, M.; Reuter, U.; Terwindt, G.; Mitsikostas, D.D.; Martelletti, P. European headache federation guideline on the use of monoclonal antibodies acting on the calcitonin gene related peptide or its receptor for migraine prevention. J. Headache Pain 2019, 20, 6. [Google Scholar] [CrossRef]
- Efficacy and Tolerability of Erenumab in the Prophylactic Treatment of Persistent Post-Traumatic Headache, NCT03974360. Available online: https://clinicaltrials.gov/ct2/show/NCT03974360?term=CGRP&cond=Post-Traumatic+Headache&draw=2&rank=3 (accessed on 25 March 2020).
- Charles, J.A. Treatment of posttraumatic headache migraine phenotype with erenumab—An observational study. J. Concussion 2019, 3. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Capi, M.; Pomes, L.M.; Andolina, G.; Curto, M.; Martelletti, P.; Lionetto, L. Persistent Post-Traumatic Headache and Migraine: Pre-Clinical Comparisons. Int. J. Environ. Res. Public Health 2020, 17, 2585. https://doi.org/10.3390/ijerph17072585
Capi M, Pomes LM, Andolina G, Curto M, Martelletti P, Lionetto L. Persistent Post-Traumatic Headache and Migraine: Pre-Clinical Comparisons. International Journal of Environmental Research and Public Health. 2020; 17(7):2585. https://doi.org/10.3390/ijerph17072585
Chicago/Turabian StyleCapi, Matilde, Leda Marina Pomes, Giulia Andolina, Martina Curto, Paolo Martelletti, and Luana Lionetto. 2020. "Persistent Post-Traumatic Headache and Migraine: Pre-Clinical Comparisons" International Journal of Environmental Research and Public Health 17, no. 7: 2585. https://doi.org/10.3390/ijerph17072585
APA StyleCapi, M., Pomes, L. M., Andolina, G., Curto, M., Martelletti, P., & Lionetto, L. (2020). Persistent Post-Traumatic Headache and Migraine: Pre-Clinical Comparisons. International Journal of Environmental Research and Public Health, 17(7), 2585. https://doi.org/10.3390/ijerph17072585




