Myalgia in 30 Patients with Suspected Myopathy
Abstract
1. Introduction
2. Experimental Section
2.1. Patients and Controls
2.2. Questionnaires and HADS
2.3. Pressure Pain Threshold
2.4. Muscle Histopathology and Molecular Genetic Studies
2.5. Statistical Analysis
2.6. Standard Protocol Approvals and Participant Consent
3. Results
3.1. Muscle Diseases
3.2. Localization of Myalgia
3.3. Description of Myalgia
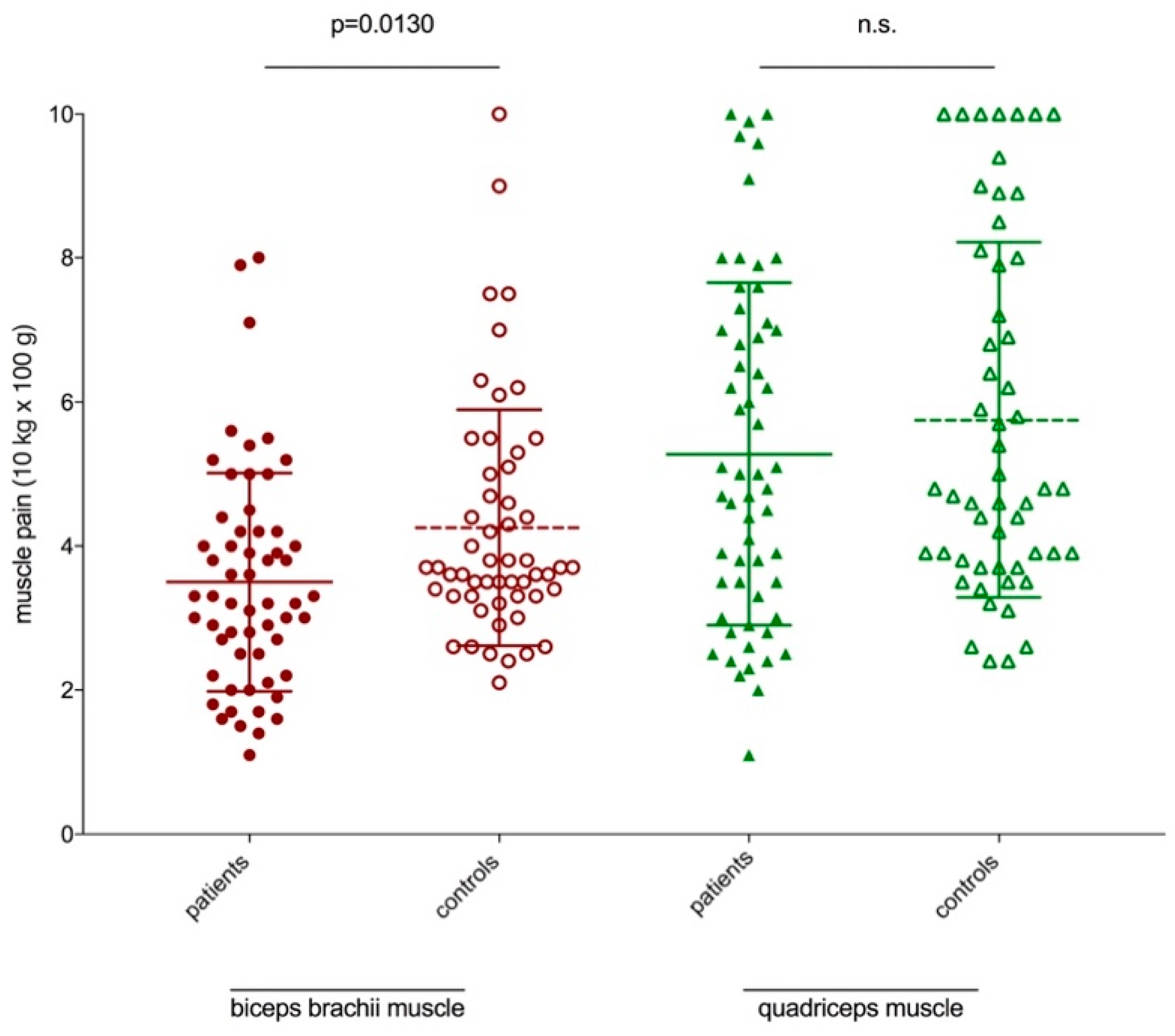
3.4. Pain Scores
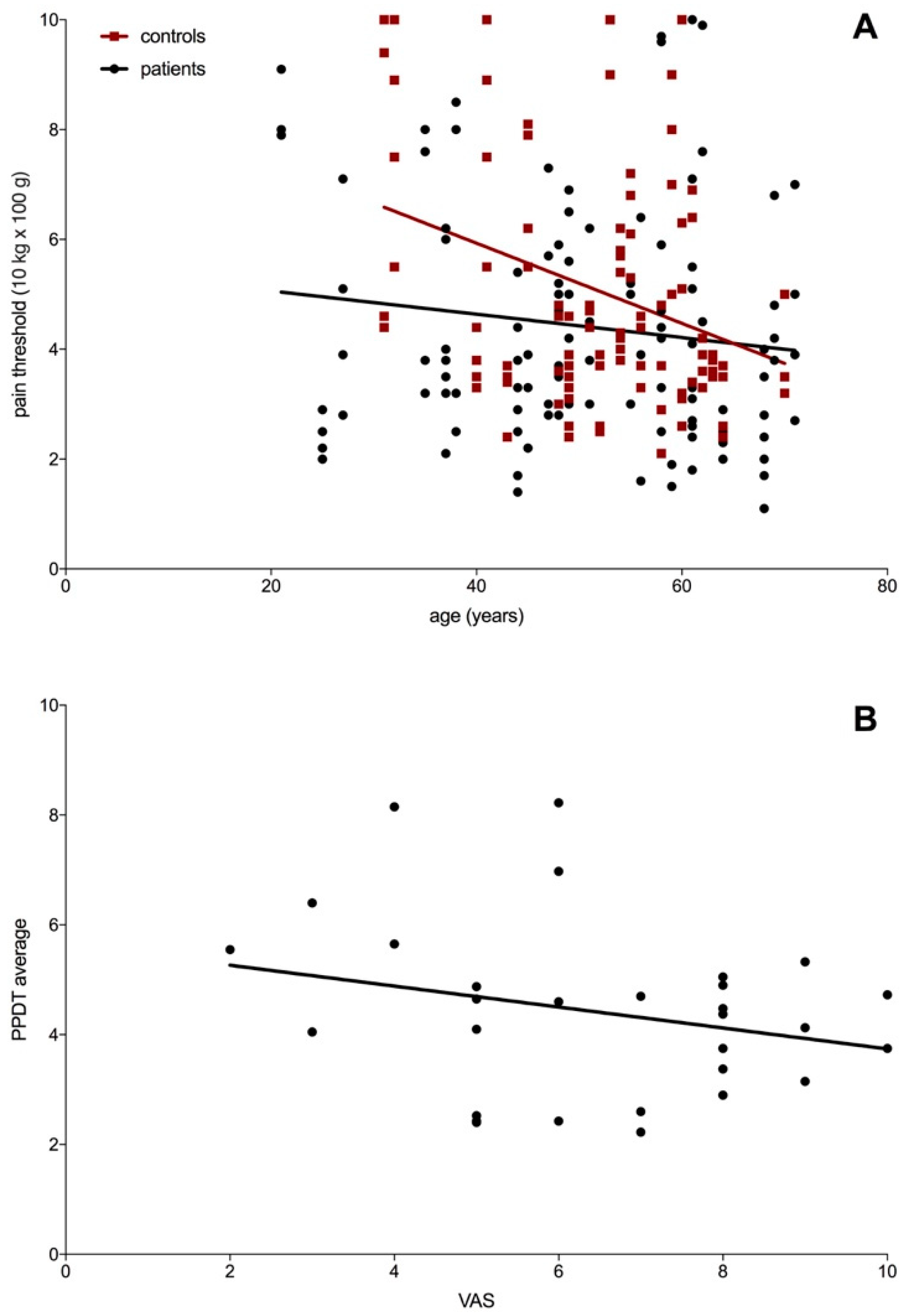
3.5. Pressure Pain Threshold
3.6. HADS
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- de Groot, I.J.; Voet, N.B.; van Middendorp, H.; Knoop, H.J.; Rahbek, J.; van Engelen, B.G. 184th enmc international workshop: Pain and fatigue in neuromuscular disorders: 20–22 may 2011, naarden, the netherlands. Neuromuscul. Disord. 2013, 23, 1028–1032. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.P.; Hoffman, A.J.; Cardenas, D.D. Chronic pain in individuals with spinal cord injury: A survey and longitudinal study. Spinal Cord 2005, 43, 704–712. [Google Scholar] [CrossRef] [PubMed]
- Bassez, G.; Attarian, S.; Laforet, P.; Azulay, J.P.; Rouche, A.; Ferrer, X.; Urtizberea, J.A.; Pellissier, J.F.; Duboc, D.; Fardeau, M.; et al. proximal myotonial myopathy (promm): Clinical and histology study. Rev. Neurol. 2001, 157, 209–218. [Google Scholar] [PubMed]
- van der Sluijs, B.M.; Knoop, H.; Bleijenberg, G.; van Engelen, B.G.; Voermans, N.C. The dutch patients’ perspective on oculopharyngeal muscular dystrophy: A questionnaire study on fatigue, pain and impairments. Neuromuscul. Disord. 2016, 26, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.H.; Huh, K.H.; Kho, H.S. Non-infectious myositis of the lateral pterygoid muscle: A report of four cases. Int. J. Oral Maxillofac. Surg. 2015, 44, 226–228. [Google Scholar] [CrossRef] [PubMed]
- Sobreira, C.; Marques, W., Jr.; Barreira, A.A. Myalgia as the revealing symptom of multicore disease and fibre type disproportion myopathy. J. Neurol. Neurosurg. Psychiatry 2003, 74, 1317–1319. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sharp, L.J.; Haller, R.G. Metabolic and mitochondrial myopathies. Neurol. Clin. 2014, 32, 777–799, ix. [Google Scholar] [CrossRef] [PubMed]
- Melzack, R. The mcgill pain questionnaire: Major properties and scoring methods. Pain 1975, 1, 277–299. [Google Scholar] [CrossRef]
- Wolfe, F.; Smythe, H.A.; Yunus, M.B.; Bennett, R.M.; Bombardier, C.; Goldenberg, D.L.; Tugwell, P.; Campbell, S.M.; Abeles, M.; Clark, P.; et al. The american college of rheumatology 1990 criteria for the classification of fibromyalgia. Report of the multicenter criteria committee. Arthritis Rheum. 1990, 33, 160–172. [Google Scholar] [CrossRef] [PubMed]
- Hawker, G.A.; Mian, S.; Kendzerska, T.; French, M. Measures of adult pain: Visual analog scale for pain (vas pain), numeric rating scale for pain (nrs pain), mcgill pain questionnaire (mpq), short-form mcgill pain questionnaire (sf-mpq), chronic pain grade scale (cpgs), short form-36 bodily pain scale (sf-36 bps), and measure of intermittent and constant osteoarthritis pain (icoap). Arthritis Rheum. 2011, 63 (Suppl. 11), S240–S252. [Google Scholar]
- Zigmond, A.S.; Snaith, R.P. The hospital anxiety and depression scale. Acta Psychiatr. Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, C.; Kalepu, R.; Ponfick, M.; Reichel, H.; Cakir, B.; Zierz, S.; Gdynia, H.J.; Kassubek, J.; Ludolph, A.C.; Rosenbohm, A. Histological characterization and biochemical analysis of paraspinal muscles in neuromuscularly healthy subjects. Muscle Nerve 2015, 52, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Deschauer, M.; Joshi, P.R.; Glaser, D.; Hanisch, F.; Stoltenburg, G.; Zierz, S. Muscular dystrophy due to mutations in anoctamin 5: Clinical and molecular genetic findings. Nervenarzt 2011, 82, 1596–1603. [Google Scholar] [CrossRef] [PubMed]
- Hanisch, F.; Joshi, P.; Zierz, S. Amp deaminase deficiency in skeletal muscle is unlikely to be of clinical relevance. J. Neurol. 2008, 255, 318–322. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, M.; Glaser, D.; Joshi, P.R.; Zierz, S.; Wenninger, S.; Schoser, B.; Deschauer, M. Utility of a next-generation sequencing-based gene panel investigation in german patients with genetically unclassified limb-girdle muscular dystrophy. J. Neurol. 2016, 263, 743–750. [Google Scholar] [CrossRef] [PubMed]
- van Vliet, J.; Tieleman, A.A.; Verrips, A.; Timmerman, H.; van Dongen, R.T.M.; van Engelen, B.G.M.; Wilder-Smith, O.H.G. Qualitative and quantitative aspects of pain in patients with myotonic dystrophy type 2. J. Pain 2018, 19, 920–930. [Google Scholar] [CrossRef] [PubMed]
- Lautenbacher, S.; Peters, J.H.; Heesen, M.; Scheel, J.; Kunz, M. Age changes in pain perception: A systematic-review and meta-analysis of age effects on pain and tolerance thresholds. Neurosci. Biobehav. Rev. 2017, 75, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Petrini, L.; Matthiesen, S.T.; Arendt-Nielsen, L. The effect of age and gender on pressure pain thresholds and suprathreshold stimuli. Perception 2015, 44, 587–596. [Google Scholar] [CrossRef] [PubMed]
- Stuginski-Barbosa, J.; Santos Silva, R.; Ortigosa Cunha, C.; Rigoldi Bonjardim, L.; de Castro Ferreira Conti, A.C.; Rodrigues Conti, P.C. Pressure pain threshold and pain perception in temporomandibular disorder patients: Is there any correlation? Rev. Dor 2015, 16, 22–26. [Google Scholar] [CrossRef]
- Sanches, M.L.; Juliano, Y.; Novo, N.F.; Guimaraes, A.S.; Rodrigues Conti, P.C.; Alonso, L.G. Correlation between pressure pain threshold and pain intensity in patients with temporomandibular disorders who are compliant or non-compliant with conservative treatment. Oral Surg Oral Med. Oral Pathol. Oral Radiol. 2015, 120, 459–468. [Google Scholar] [CrossRef] [PubMed]
| Patient-ID | Age at Study Inclusion | Gender (m/f) | HADS-A/D | VAS (x/10) | EMG | CK (μkat/L) | Biopsy | Diagnosis | |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 61 | f | 4 | 8 | 8 | myopathic | 8.42 ↑ | myopathic | muscle dystrophy (LGMD 1B)—heterozygote mutation in the LMNA-gene (c. 1930C<T) |
| 2 | 25 | f | 5 | 1 | 5 | myopathic | 42.45 ↑ | myopathic | anoctaminopathy–homozygote mutation in the ANO5-Gen (c.191dupA) |
| 3 | 59 | m | 3 | 1 | 6 | normal | <3.20 | myopathic | myoadenylate deaminase deficiency |
| 4 | 71 | f | 2 | 4 | 5 | normal | <2.85 | myopathic | mitochondrial myopathy |
| 5 | 35 | f | 7 | 6 | 4 | myopathic | 6.81 ↑ | myopathic and neurogenic | myopathy |
| 6 | 58 | f | 8 | 9 | 6 | normal | <2.85 | myopathic | myopathy |
| 7 | 64 | m | 3 | 1 | 6 | normal | <3.20 | myopathic | myopathy |
| 8 | 68 | f | 4 | 4 | 5 | normal | <2.85 | myopathic | myopathy |
| 9 | 45 | f | 12 | 8 | 8 | normal | <2.85 | myopathic | myopathy |
| 10 | 37 | f | 4 | 15 | 9 | normal | <2.85 | myopathic | myopathy |
| 11 | 68 | f | 3 | 3 | 7 | normal | <2.85 | n. p. | PMR |
| 12 | 61 | m | 9 | 10 | 5 | normal | <3.20 | n. p. | PMR |
| 13 | 51 | f | 15 | 17 | 8 | myopathic | <2.85 | myopathic/inflammatoric | polymyositis |
| 14 | 62 | f | 3 | 9 | 3 | myopathic/myotonic | 5.2 ↑ | myopathic/inflammatoric | polymyositis |
| 15 | 61 | m | 1 | 1 | 4 | normal | <3.20 | normal | MPFS |
| 16 | 44 | f | 3 | 0 | 7 | normal | <2.85 | normal | MPFS |
| 17 | 49 | f | 7 | 4 | 5 | normal | <2.85 | normal | MPFS |
| 18 | 69 | f | 8 | 4 | 8 | normal | <2.85 | n. p. | MPFS |
| 19 | 27 | f | 7 | 5 | 10 | normal | <2.85 | normal | MPFS |
| 20 | 21 | m | 7 | 5 | 6 | normal | <3.20 | normal | MPFS |
| 21 | 49 | f | 4 | 3 | 9 | normal | 7.82 ↑ | normal | MPFS |
| 22 | 47 | m | 5 | 5 | 7 | normal | <3.20 | n. p. | MPFS |
| 23 | 38 | f | 1 | 0 | 2 | normal | 4.19 ↑ | normal | MPFS |
| 24 | 48 | f | 1 | 1 | 8 | normal | <2.85 | n. p. | MPFS |
| 25 | 56 | f | 2 | 3 | 8 | normal | <2.85 | normal | myalgia |
| 26 | 44 | f | 17 | 20 | 10 | normal | <2.85 | normal | myalgia |
| 27 | 48 | f | 9 | 5 | 9 | normal | <2.85 | normal | myalgia |
| 28 | 37 | f | 6 | 5 | 8 | normal | <2.85 | n. p. | myalgia |
| 29 | 58 | f | 9 | 8 | 5 | normal | <2.85 | normal | myalgia |
| 30 | 55 | f | 8 | 5 | 3 | normal | <2.85 | n. p. | myalgia |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lehmann Urban, D.; Lehmann, E.; Motlagh Scholle, L.; Kraya, T. Myalgia in 30 Patients with Suspected Myopathy. Int. J. Environ. Res. Public Health 2020, 17, 2502. https://doi.org/10.3390/ijerph17072502
Lehmann Urban D, Lehmann E, Motlagh Scholle L, Kraya T. Myalgia in 30 Patients with Suspected Myopathy. International Journal of Environmental Research and Public Health. 2020; 17(7):2502. https://doi.org/10.3390/ijerph17072502
Chicago/Turabian StyleLehmann Urban, Diana, Elizabeth Lehmann, Leila Motlagh Scholle, and Torsten Kraya. 2020. "Myalgia in 30 Patients with Suspected Myopathy" International Journal of Environmental Research and Public Health 17, no. 7: 2502. https://doi.org/10.3390/ijerph17072502
APA StyleLehmann Urban, D., Lehmann, E., Motlagh Scholle, L., & Kraya, T. (2020). Myalgia in 30 Patients with Suspected Myopathy. International Journal of Environmental Research and Public Health, 17(7), 2502. https://doi.org/10.3390/ijerph17072502




