Low-Cost Robotic Guide Based on a Motor Imagery Brain–Computer Interface for Arm Assisted Rehabilitation
Abstract
1. Introduction
2. Materials and Methods
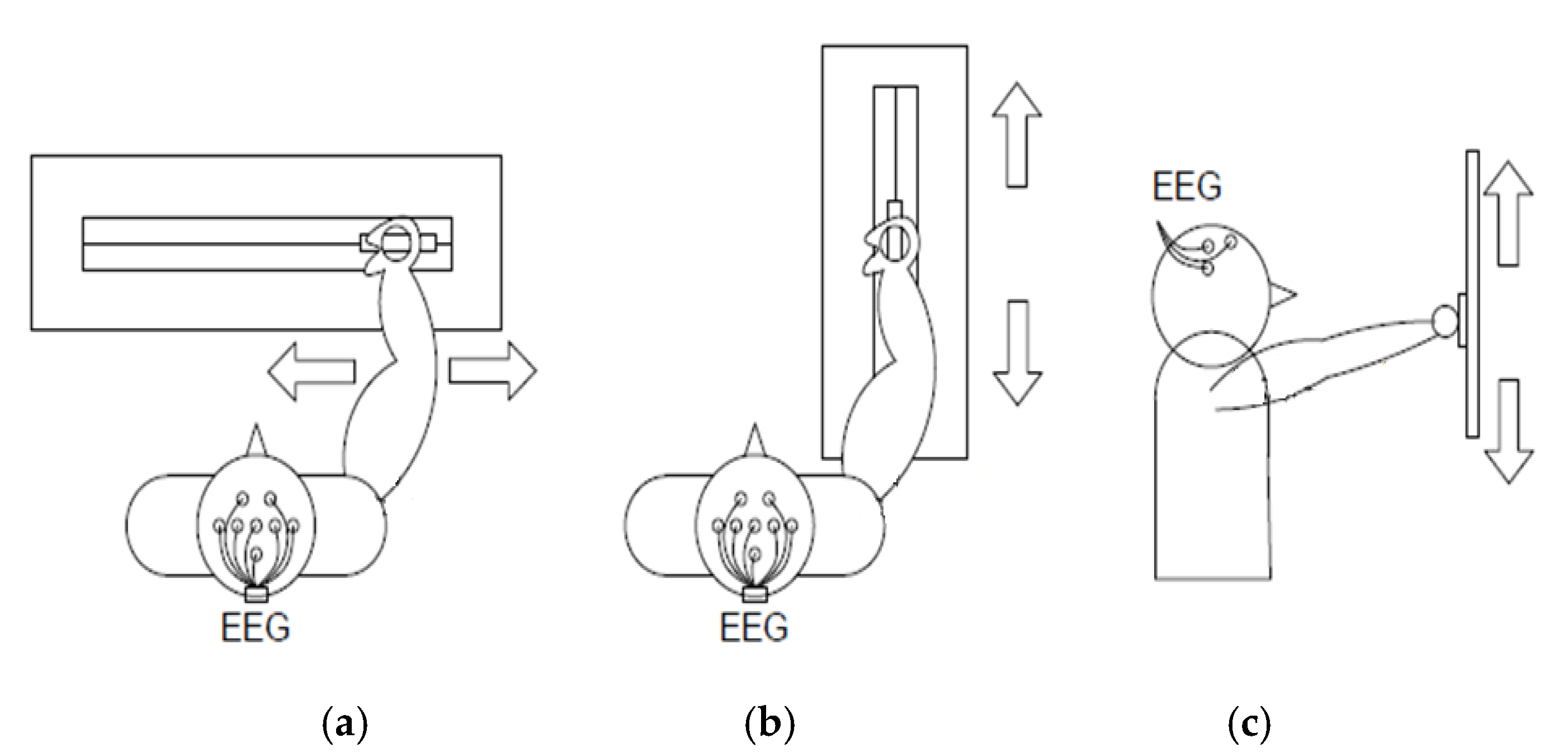
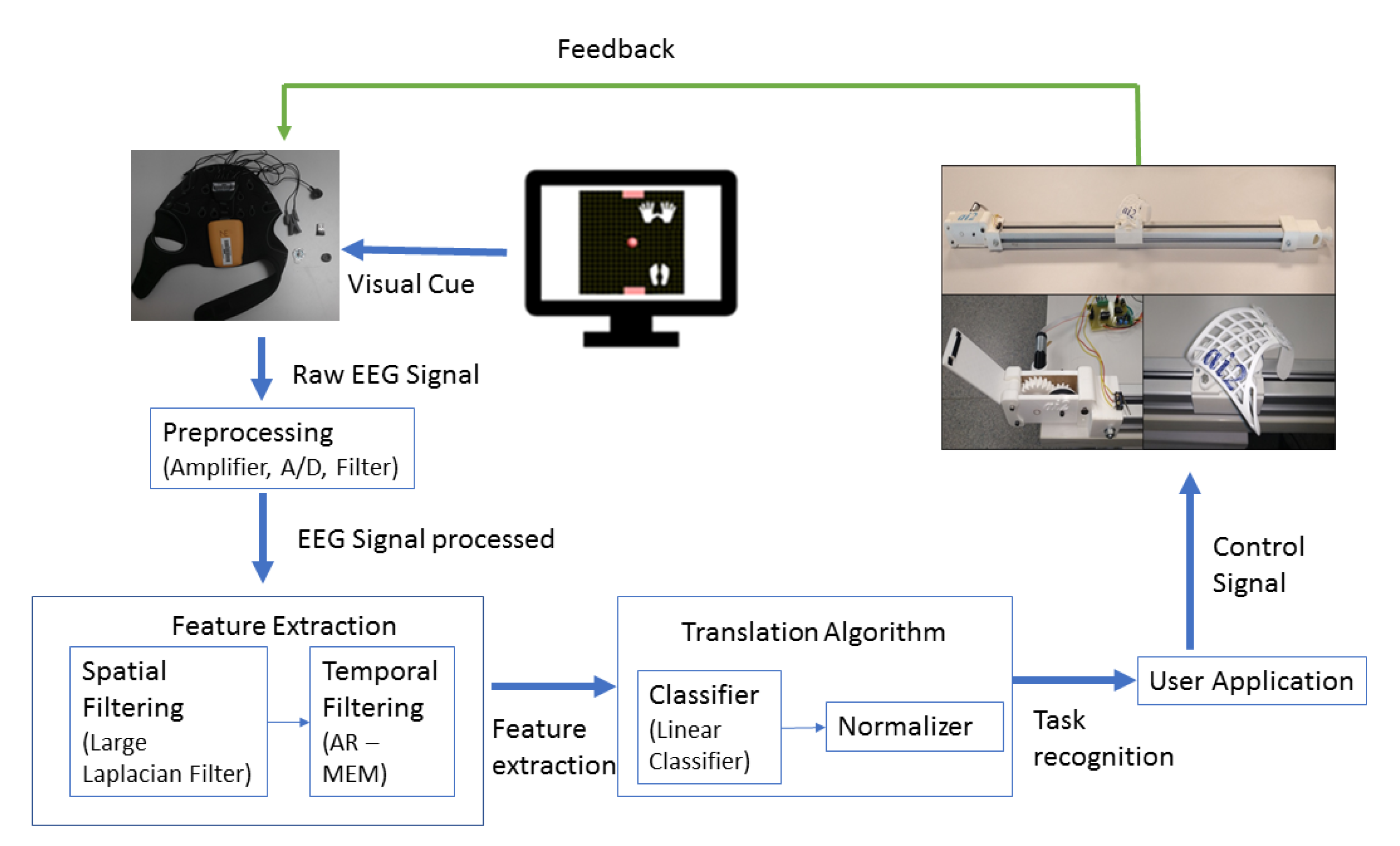
2.1. Rehabilitation Guide
2.2. EEG-Based Control
2.3. Participants
2.4. Experimental Procedure
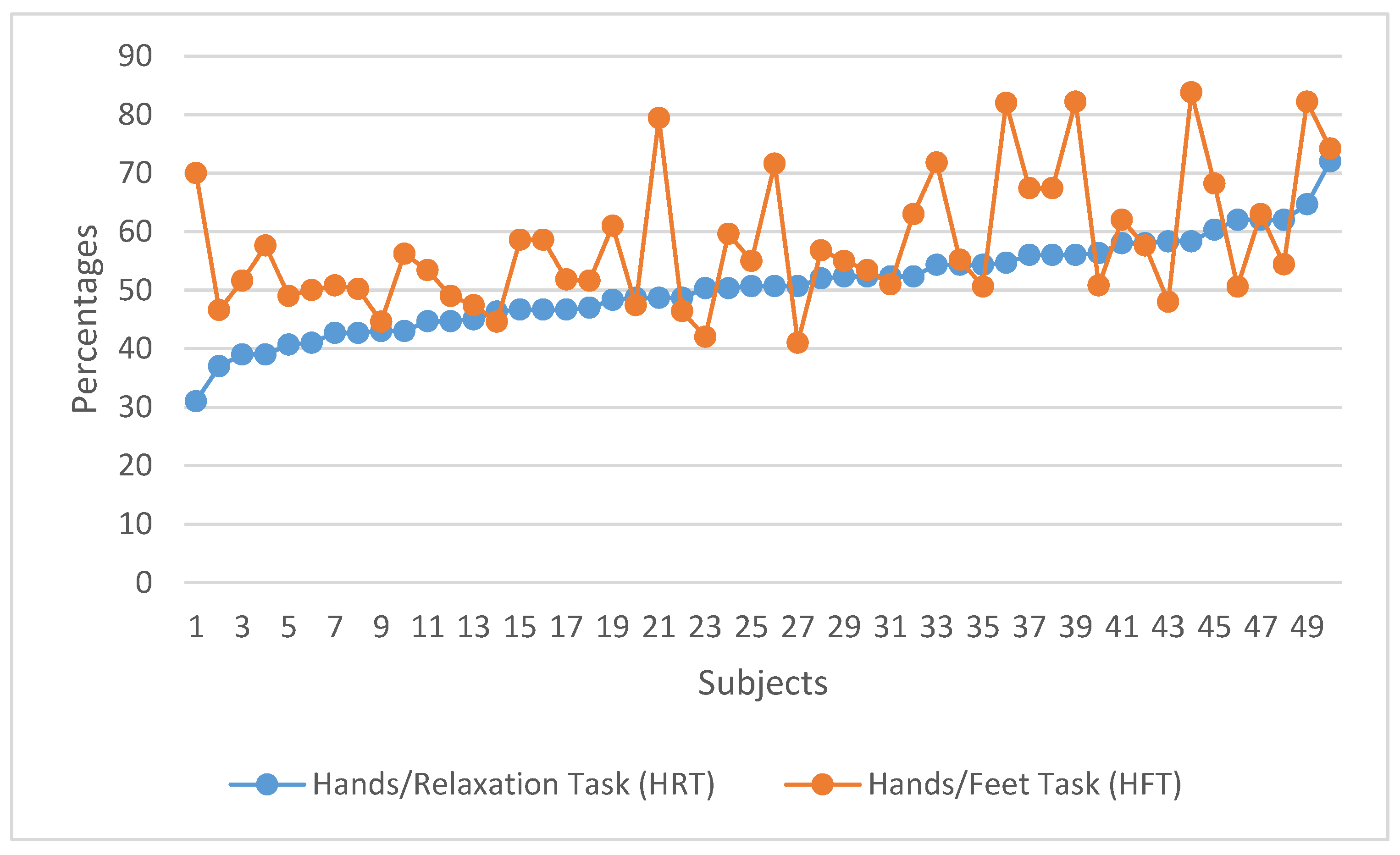
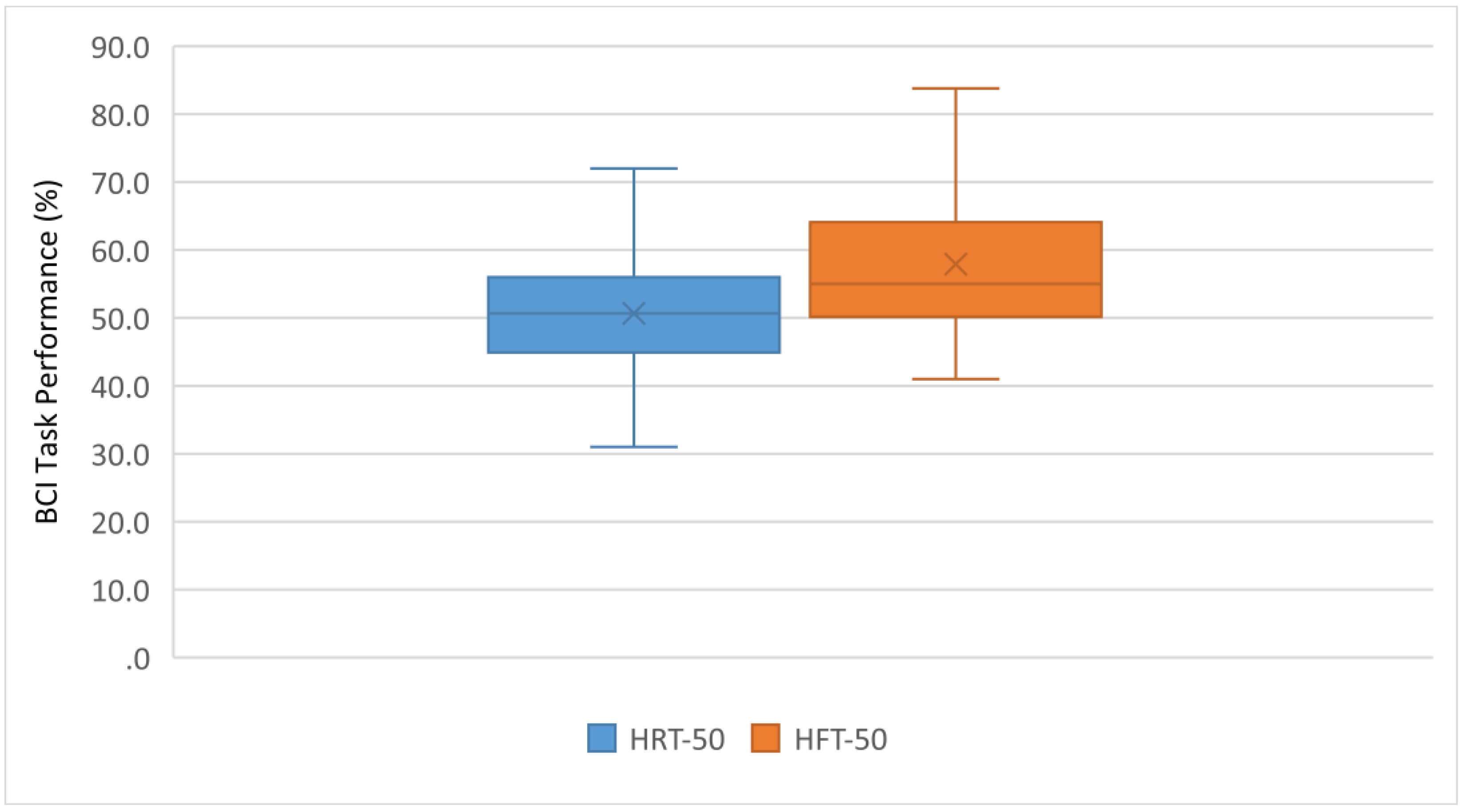
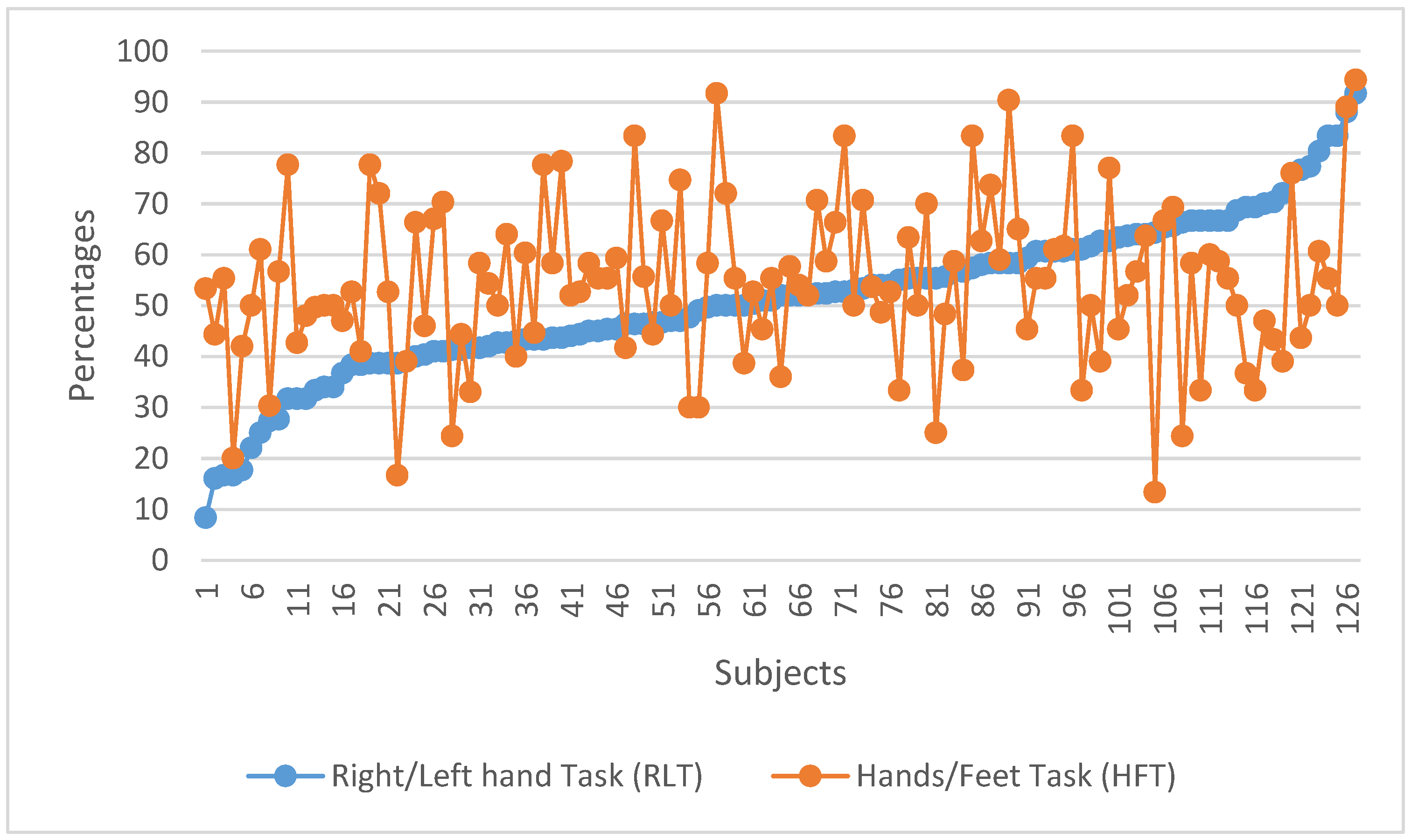
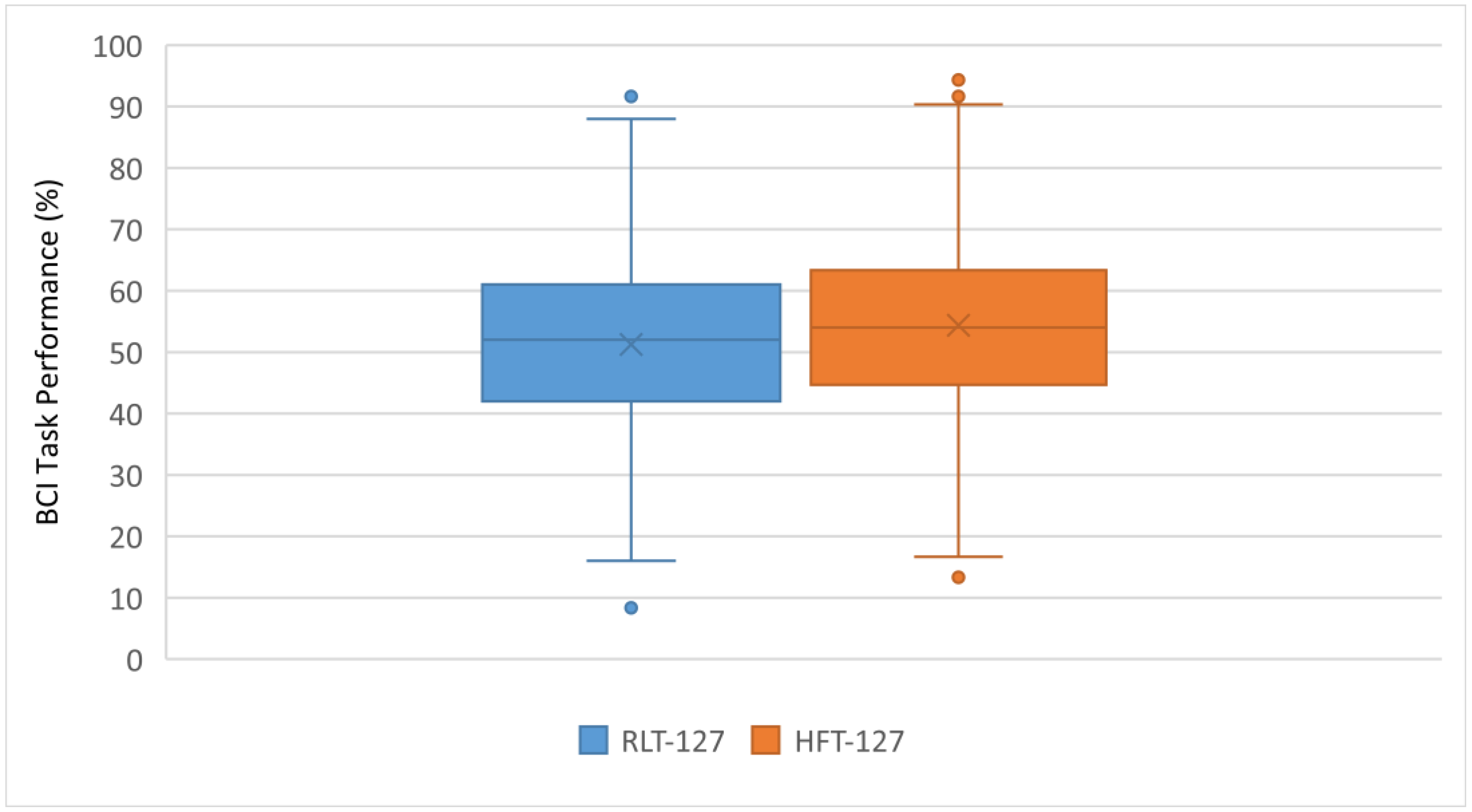
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AMT | Active motor training |
| BCI | Brain–computer interface |
| EEG | Electroencephalography |
| HFT | Hand/feet task |
| HRT | Hand/relax task |
| MI | Motor imagery |
| PWM | Pulse width modulation |
| RLT | Right hand/left hand task |
| SCI | Spinal cord injury |
| SPSS | Statistical Package for the Social Sciences |
| SSVEP | Steady-state visual evoked potentials |
| UDP | User datagram protocol |
References
- Ang, K.K.; Guan, C. Brain-computer interface in stroke rehabilitation. J. Comput. Sci. Eng. 2013, 7, 139–146. [Google Scholar] [CrossRef]
- Langhorne, P.; Bernhardt, J.; Kwakkel, G. Stroke rehabilitation. Lancet 2015, 377, 1693–1702. [Google Scholar] [CrossRef]
- Noser, A.; Ro, T.; Boake, C.; Levin, H.; Aronowski, J.; Schallert, T. Constraint-induced movement therapy. Stroke 2004, 32, 2699–2701. [Google Scholar]
- Pfurtscheller, G.; Neuper, C. Motor imagery activates primary sensorimotor area in humans. Neurosci. Lett. 1997, 239, 65–68. [Google Scholar] [CrossRef]
- McFarland, D.; Miner, L.; Vaughan, T.; Wolpaw, J. Mu and Beta Rhythm Topographies During Motor Imagery and Actual Movements. Brain Topogr. 2000, 12, 177–186. [Google Scholar] [CrossRef]
- Kilteni, K.; Andersson, B.; Houborg, C.; Ehrsson, H. Motor imagery involves predicting the sensory consequences of the imagined movement. Nat. Commun. 2018, 1617, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Sharma, N.; Pomeroy, V.M.; Baron, J.C. Motor imagery a backdoor to the motor system after stroke? Stroke 2006, 37, 1941–1952. [Google Scholar] [CrossRef]
- Tong, Y.; Pendy, J., Jr.; Li, W.; Du, H.; Zhang, T.; Geng, X.; Ding, Y. Motor Imagery-Based Rehabilitation: Potential Neural Correlates and Clinical Apllication for Functional Recovery of Motor Deficits after Stroke. Aging Dis. 2017, 8, 364–371. [Google Scholar] [CrossRef]
- Nicolas-Alonso, L.F.; Gomez-Gil, J. Brain computer interfaces, a review. Sensors 2012, 12, 1211–1279. [Google Scholar] [CrossRef]
- Clerc, M.; Bougrain, L.; Lotte, F. Brain-Computer Interfaces 1: Foundations and Methods; ISTE-Wiley: London, UK; Hoboken, NJ, USA, 2016. [Google Scholar]
- Clerc, M.; Bougrain, L.; Lotte, F. Brain-Computer Interfaces 2: Technology and Applications; ISTE-Wiley: London, UK; Hoboken, NJ, USA, 2016. [Google Scholar]
- Wolpaw, J.R.; Birbaumer, N.; McFarland, D.J.; Pfurtscheller, G.; Vaughan, T.M. Vaughan, Brain-computer interfaces for communication and control. Clin. Neurophysiol. 2002, 113, 767–791. [Google Scholar] [CrossRef]
- Placidi, G.; Petracca, A.; Spezialetti, M.; Iacoviello, D. A Modular Framework for EEG Web Based Binary Brain Computer Interfaces to Recover Communication Abilities in Impaired People. J. Med. Syst. 2016, 40, 34. [Google Scholar] [CrossRef] [PubMed]
- Hortal, E.; Planelles, D.; Costa, A.; Iáñez, E.; Úbeda, A.; Azorín, J.M.; Fernández, E. SVM-Based Brain-Machine interface for controlling a Robot Arm through four Mental Tasks. Neurocomputing 2015, 151, 116–121. [Google Scholar] [CrossRef]
- Collinger, J.L.; Wodlinger, B.; Downey, J.; Wang, W.; Tyler-Kabara, E.C.; Weber, D.J.; Schwartz, A.B. High-performance neuroprosthetic control by an individual with tetraplegia. Lancet 2013, 381, 557–564. [Google Scholar] [CrossRef]
- Iturrate, I.; Antelis, J.M.; Kubler, A.; Minguez, J.A. A noninvasive brain-actuated wheelchair based on a P300 neurophysiological protocol and automated navigation. IEEE Trans. Robot. 2009, 25, 614–627. [Google Scholar] [CrossRef]
- Edlinger, G.; Holzner, C.; Guger, C.; Groenegress, C.; Slater, M. Brain-computer interfaces for goal orientated control of a virtual smart home environment. In Proceedings of the 4th International IEEE/EMBS Conference on Neural Engineering, Antalya, Turkey, 29 April–2 May 2009. [Google Scholar]
- Mihajlović, V.; Grundlehner, B.; Vullers, R.; Penders, J. Wearable, wireless EEG solutions in daily life applications: What are we missing? IEEE J. Biomed. Health Inf. 2015, 19, 6–21. [Google Scholar] [CrossRef]
- Lightbody, G.; Galway, L.; McCullagh, P. The brain computer interface: Barriers to becoming pervasive. In Pervasive Health; Holzinger, A., Ziefle, M., Röcker, C., Eds.; Springer: London/England, UK, 2014; pp. 101–129. [Google Scholar]
- Tangermann, M.; Lotte, F.; Van Erp, J. Brain-Computer Interfaces: Beyond Medical Applications. IEEE Comput. Soc. 2012, 45, 26–34. [Google Scholar]
- Úbeda, A.; Azorín, J.M.; Chavarriaga, R.; Millán, J.d.R. Classification of upper limb center-out reaching tasks by means of EEG-based continuous decoding techniques. J. Neuroeng. Rehabil. 2017, 14, 9. [Google Scholar] [CrossRef]
- Wolpaw, J.R.; McFarland, D.J. Control of a two-dimensional movement signal by a noinvasive brain-computer interface in humans. Proc. Natl. Acad. Sci. USA 2004, 101, 17849–17854. [Google Scholar] [CrossRef]
- Lotte, F.; Congedo, M.; Lécuyer, A.; Lamarche, F.; Arnaldi, B. A review of classification algorithms for eeg-based brain-computer interfaces. J. Neural Eng. 2007, 4, R1–R14. [Google Scholar] [CrossRef]
- Li, M.; Luo, X.; Yang, J.; Sun, Y. Applying a locally linear embedding algorithm for feature extraction and visualization of MI-EEG. J. Sens. 2016, 2016, 7481946. [Google Scholar] [CrossRef]
- Soekadar, S.R.; Birbaumer, N.; Slutzky, M.W.; Cohen, L.G. Brain-machine interfaces in neurorehabilitation of stroke. Neurobiol. Dis. 2015, 83, 172–179. [Google Scholar] [CrossRef]
- Pfurtscheller, G.; Guger, C.; Müller, G.; Krausz, G.; Neuper, C. Brain oscillations control hand orthosis in a tetraplegic. Neurosci. Lett. 2000, 292, 211–214. [Google Scholar] [CrossRef]
- Müller-Putz, G.R.; Scherer, R.; Pfurtscheller, G.; Rupp, R. EEG-based neuroprosthesis control: A step towards clinical practice. Neurosci. Lett. 2005, 382, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Horki, P.; Solis-Escalante, T.; Neuper, C.; Müller-Putz, G. Combined motor imagery and SSVEP based BCI control of a 2 DoF artificial upper limb. Med. Biol. Eng. Comput. 2011, 49, 567–577. [Google Scholar] [CrossRef]
- Lo, H.; Xie, S. Exoskeleton robots for upper limb rehabilitation: State of the art and future prospects. Med. Eng. Phys. 2012, 34, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Sheng, B.; Zhang, Y.; Meng, W.; Xie, C.D.S. Bilateral robots for upper limb stroke rehabilitation: State of the art and future prospects. Med. Eng. Phys. 2012, 38, 587–606. [Google Scholar] [CrossRef]
- Tang, Z.; Sun, S.; Zhang, S.; Chen, Y.; Li, C.; Chen, S. A Brain-Machine Interface Based on ERD/ERS for an Upper-Limb Exoskeleton Control. Sensors 2016, 16, 2050. [Google Scholar] [CrossRef]
- Ang, K.K.; Guan, C.; Chua, K.; Ang, B.T.; Kuah, C.; Wang, C.; Phua, K.S.; Chin, Z.Y.; Zhang, H. A clinical study of motor imagery-based brain-computer interface for upper limb robotic rehabilitation. In Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Minneapolis, MN, USA, 3–6 September 2009. [Google Scholar]
- Blankertz, B.; Sannelli, C.; Halder, S.; Hammer, E.M.; Kübler, A.; Müller, K.R.; Curio, G.; Dickhaus, T. Neurophysiological predictor of SMR-based BCI performance. Neuroimage 2010, 51, 1303–1309. [Google Scholar] [CrossRef]
- Allison, B.Z.; Neuper, C. Could Anyone Use a BCI. In Brain-Computer Interfaces; Applying our minds to Human Computer Interaction; Springer: London, UK, 2010; pp. 33–54. [Google Scholar]
- Jeunet, C.; N’Kaoua, B.; Subramanian, S.; Hachet, M.; Lotte, F. Predicting Mental Imagery-Based BCI Perfomance from Personality, Cognitive Profile and Neurophysiological Patterns. PLoS ONE 2015, 10, e0143962. [Google Scholar] [CrossRef]
- Friedrich, E.V.; Neuper, C.; Scherer, R. Whatever works: A systematic user-centered training protocol to optimize brain-computer interfacing individually. PLoS ONE 2013, 8, e76214. [Google Scholar] [CrossRef]
- Pfurtscheller, G.; Neuper, C. Motor Imagery and Direct Brain-Computer Communication. Proc. IEEE 2001, 89, 1123–1134. [Google Scholar] [CrossRef]
- Vuckovic, A.; Osuagwu, B.A. Using a motor imagery questionnaire to estimate the perfomance of a Brain-Computer Interface based on object oriented motor imagery. Clin. Neurophysiol. 2013, 124, 1586–1595. [Google Scholar] [CrossRef]
- Chaudhary, U.; Birbaumer, N.; Ramos-Murguialday, A. Brain computer interfaces for communication and rehabilitation. Nat. Rev. Neurol. 2016, 12, 513–525. [Google Scholar] [CrossRef]
- Jeunet, C.; N’Kaoua, B.; Lotte, F. Advances in User-Training for Mental-Imagery Based BCI Control: Psychological and Cognitive Factors and their Neural Correlates. Progress Brain Res. 2016, 228, 3–35. [Google Scholar]
- Schalk, G.; Brunner, P.; Gerhardt, L.A.; Bischof, H.; Wolpaw, J.R. Brain-computer interfaces (BCIs): detection instead of classification. J. Neurosci. Methods 2008, 167, 51–62. [Google Scholar] [CrossRef]
- Cester, I.; Dunne, S.; Riera, A.; Ruffini, G. ENOBIO: Wearable, Wireless, 4-channel electrophysiology recording system optimized for dry electrodes. In Proceedings of the International Workshop on Wearable Micro and Nanosystems for Personalised Health, Valencia, España, 21–23 May 2008. [Google Scholar]
- Schalk, G.; Mellinger, J. A Practical Guide to Brain-Computer Interfacing with Bci2000; Springer: London, UK, 2010. [Google Scholar]
- Schalk, G.; McFarland, D.J.; Hinterberger, T.; Birbaumer, N.; Wolpaw, J.R. BCI2000: A general-purpose brain-computer interface (BCI) system. IEEE Trans. Biomed. Eng. 2004, 51, 1034–1043. [Google Scholar] [CrossRef]
- Wolpaw, J.R.; McFarland, D.J.; Neat, G.W.; Forneris, C.A. An EEG-based brain-computer interface for cursor control. Electroencephalogr. Clin. Neurophysiol. 1991, 78, 252–259. [Google Scholar] [CrossRef]
- McFarland, D.J.; Neat, G.W.; Read, R.F.; Wolpaw, J.R. An EEG-based method for graded cursor control. Psychobiology 1993, 21, 77–81. [Google Scholar]
- Fabiani, G.E.; McFarland, D.J.; Wolpaw, J.R.; Pfurtscheller, G. Conversion of EEG Activity into Cursor Movement by a Brain-Computer Interface (BCI). IEEE Trans. Neural Syst. Rehabil. Eng. 2004, 12, 331–338. [Google Scholar] [CrossRef]
- McFarland, D.J.; McCane, L.M.; David, S.V.; Wolpaw, J.R. Spatial filter selection for EEG-based communication. Electroencephalogr. Clin. Neurophysiol. 1997, 103, 386–394. [Google Scholar] [CrossRef]
- Zhang, Y.; Ji, X.; Zhang, Y. Classification of EEG Signals Based on AR Model and Approximate Entropy. In Proceedings of the International Joint Conference on Neural Networks, Killarney, Ireland, 12–17 July 2015. [Google Scholar]
- Wilson, J.; Williams, J. Massively parallel signal processing using the graphics processing unit for real-time brain-computer interface feature extraction. Front. Neuroeng. 2009, 2, 11. [Google Scholar] [CrossRef] [PubMed]
- Bell, I.; Falcon, J.; Limroth, J.; Robinson, K. Integration of hardware into the Labview environment for rapid prototyping and the development of control design applications. In Proceedings of the Mini Symposia UKACC Control, Bath, England, 8 September 2004. [Google Scholar]
- Candela, G.; Quiles, E.; Chio, N.; Suay, F. Attentional Variables and BCI Performance: Comparing Two Strategies. In Psychology Applications & Developments IV; Science Press: Lisboa, Portugal, 2018; pp. 130–139. [Google Scholar]
- Wilson, J.A.; Schalk, G.; Walton, L.M.; Williams, J.C. Using an EEG-Based Brain-Computer Interface for Virtual Cursor Movement with BCI2000. J. Vis. Exp. JoVE 2009, 29, 1319. [Google Scholar] [CrossRef] [PubMed]
- Clerc, M.; Bougrain, L.; Lotte, F. Physiological Markers for Controlling Active and Reactive BCIs. In Brain-Computer Interfaces 1: Foundations and Methods; ISTE-Wiley: London, UK; Hoboken, NJ, USA, 2016. [Google Scholar]
- Alonso-Valerdi, L.; Salido-Ruiz, R.; Ramirez-Mendoza, R. Motor imagery based brain-computer interfaces: An emerging technology to rehabilitate motor deficits. Neuropsychologia 2015, 79, 354–363. [Google Scholar] [CrossRef] [PubMed]
- Monge-Pereira, E.; Ibañez-Pereda, J.; Alguacil-Diego, I.; Serrano, J.; Spottorno-Rubio, M.; Molina-Rueda, F. Use of Electroencephalography Brain-Computer Interface Systems as a Rehabilitative Approach for Upper Limb Function After a Stroke: A System Review. PmR 2017, 9, 918–932. [Google Scholar] [CrossRef]
- Cengiz, B.; Vuralli, D.; Zinnuroğlu, M.; Bayer, G.; Golmohammadzadeh, H.; Günendi, Z.; Turgut, A.; İrfanoğlu, B.; Arikan, K. Analysis of mirror neuron system activation during action observation alone and action observation with motor imagery tasks. Exp. Brain Res. 2018, 236, 497–503. [Google Scholar] [CrossRef]

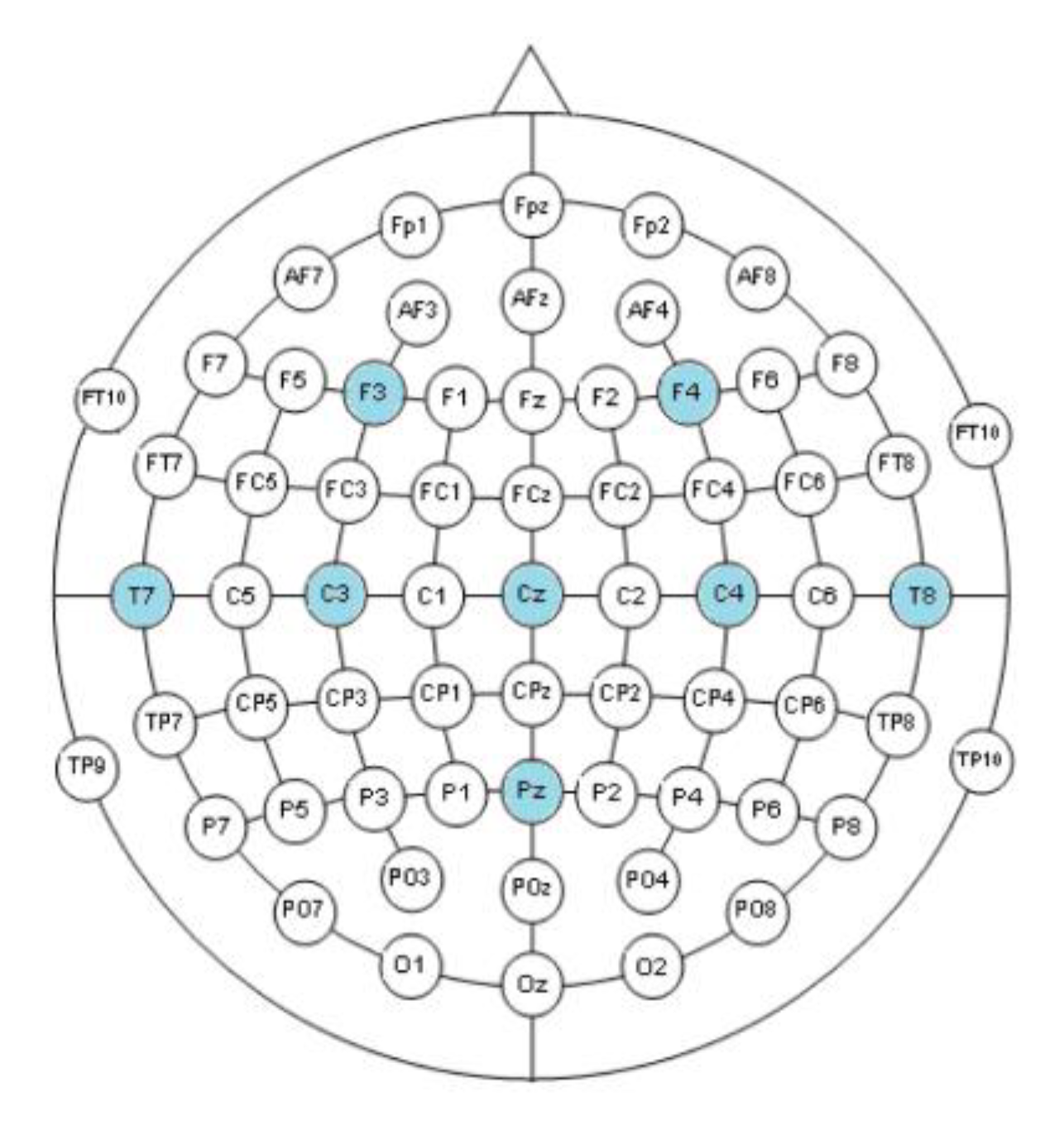

| Ck | ||||||||
|---|---|---|---|---|---|---|---|---|
| Cout | F3 | F4 | T7 | C3 | CZ | C4 | T8 | PZ |
| C3 | −0.25 | 0 | −0.25 | 1 | −0.25 | 0 | 0 | −0.25 |
| CZ | −0.2 | −0.2 | 0 | −0.2 | 1 | −0.2 | 0 | −0.2 |
| C4 | 0 | −0.25 | 0 | 0 | −0.25 | 1 | −0.25 | −0.25 |
| Step | Time (Minutes) | Activities |
|---|---|---|
| Preparation and information | 15 |
|
| Relaxation | 5 |
|
| MI-BCI tasks | 30 |
|
| Opinion questionnaire | 5 |
|
| Experimental end | 5 |
| Experiment | Paradigm | Task | Visual Cue/Description |
|---|---|---|---|
1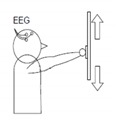 | Hand movement versus relax (HRT) | “↑” imagine opening and closing hand “↓” no movement and relax |  |
1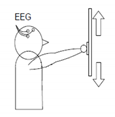 | Hand versus feet movement (HFT) | “↑” imagine opening and closing hand “↓” imagine both feet flexion |  |
2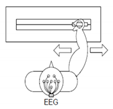 | Right hand versus left hand movement (RLT) | “←” imagine opening and closing left hand “→” imagine opening and closing right hand |  |
2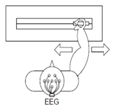 | Hand versus feet movement (HFT) | “←” imagine both feet flexion “→” imagine opening and closing hand |  |
| Questions | Answers | Percentage |
|---|---|---|
| Q1: Dexterity | Right | 92.6 |
| Left | 7.4 | |
| Q2: Do you play any musical instrument? | Yes | 17.1 |
| No | 82.9 | |
| Q3: Do you consider yourself a bilingual person? | Yes | 70.9 |
| No | 29.1 | |
| Q4: Did you sleep well last night? | Yes | 64.2 |
| No | 35.8 |
| Discomfort | None | Little | Moderate | A Lot | Too Much |
|---|---|---|---|---|---|
| Participants (%) | 88 (49.7%) | 79 (44.6%) | 10 (5.7%) | 0 (0%) | 0 (0%) |
| Tolerance Time | <1 h | 1–2 h | 2–4 h | Almost All Day | All Day |
|---|---|---|---|---|---|
| Participants (%) | 30 (17.0%) | 79 (44.6%) | 29 (16.4%) | 29 (16.4%) | 10 (5.6%) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quiles, E.; Suay, F.; Candela, G.; Chio, N.; Jiménez, M.; Álvarez-Kurogi, L. Low-Cost Robotic Guide Based on a Motor Imagery Brain–Computer Interface for Arm Assisted Rehabilitation. Int. J. Environ. Res. Public Health 2020, 17, 699. https://doi.org/10.3390/ijerph17030699
Quiles E, Suay F, Candela G, Chio N, Jiménez M, Álvarez-Kurogi L. Low-Cost Robotic Guide Based on a Motor Imagery Brain–Computer Interface for Arm Assisted Rehabilitation. International Journal of Environmental Research and Public Health. 2020; 17(3):699. https://doi.org/10.3390/ijerph17030699
Chicago/Turabian StyleQuiles, Eduardo, Ferran Suay, Gemma Candela, Nayibe Chio, Manuel Jiménez, and Leandro Álvarez-Kurogi. 2020. "Low-Cost Robotic Guide Based on a Motor Imagery Brain–Computer Interface for Arm Assisted Rehabilitation" International Journal of Environmental Research and Public Health 17, no. 3: 699. https://doi.org/10.3390/ijerph17030699
APA StyleQuiles, E., Suay, F., Candela, G., Chio, N., Jiménez, M., & Álvarez-Kurogi, L. (2020). Low-Cost Robotic Guide Based on a Motor Imagery Brain–Computer Interface for Arm Assisted Rehabilitation. International Journal of Environmental Research and Public Health, 17(3), 699. https://doi.org/10.3390/ijerph17030699








