Use of Nanoscale Zero-Valent Iron for Remediation of Clayey Soil Contaminated with Hexavalent Chromium: Batch and Column Tests
Abstract
1. Introduction
2. Materials and Methods
2.1. Soil
2.2. Batch Tests
2.3. Column Tests
2.4. Nanoscale Zero-Valent Iron (nZVI)
2.5. Contaminant Analysis
2.6. Statistical Analysis
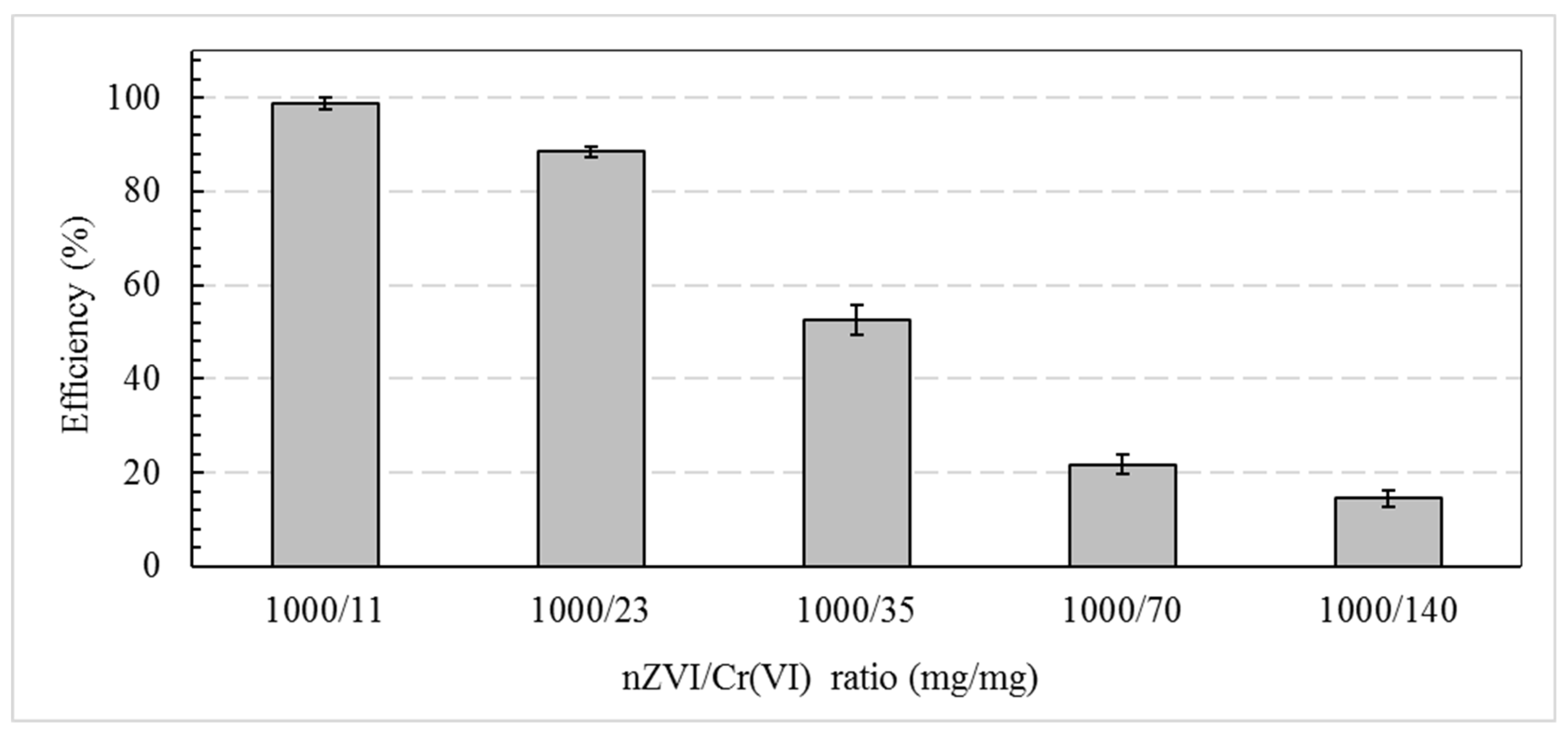
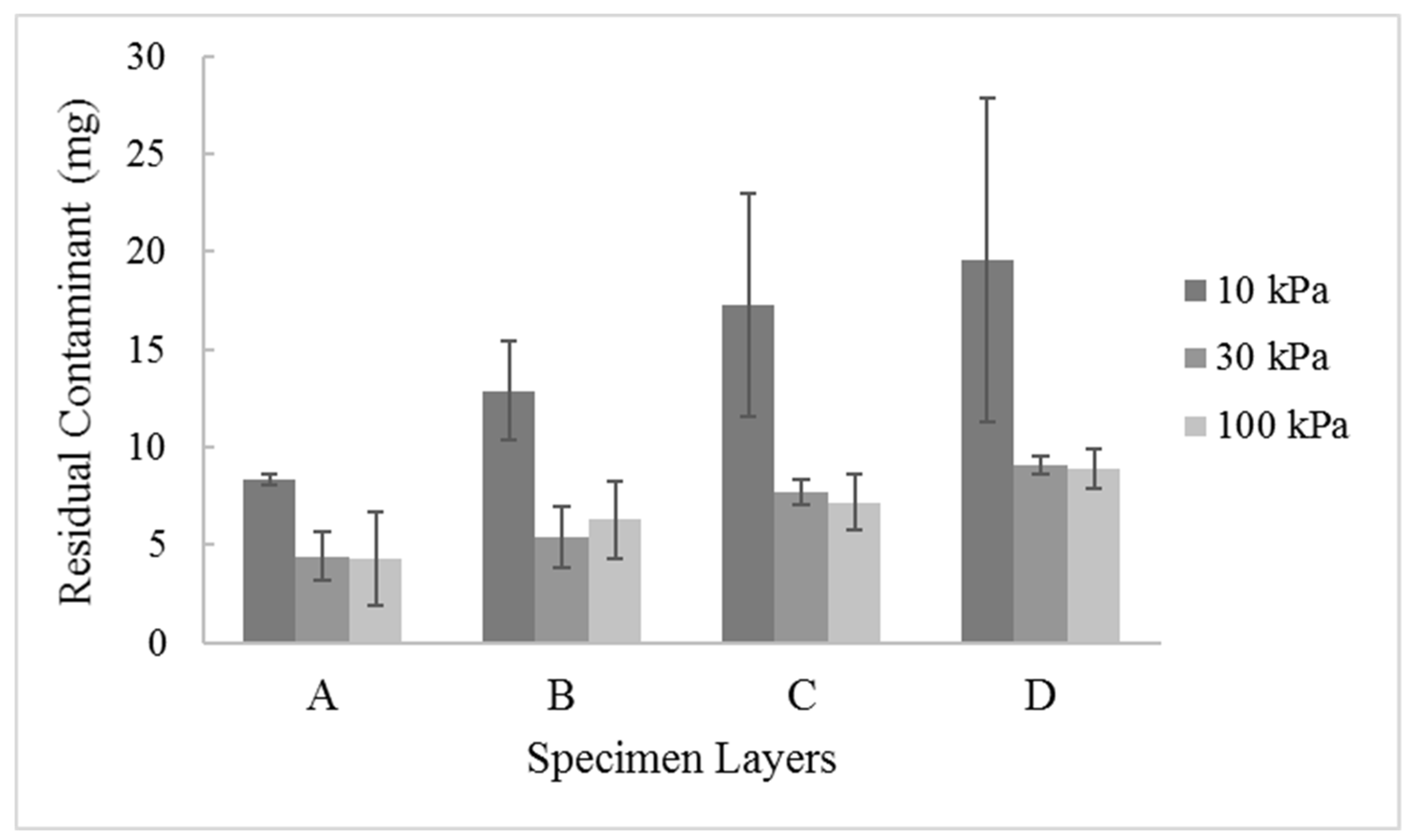
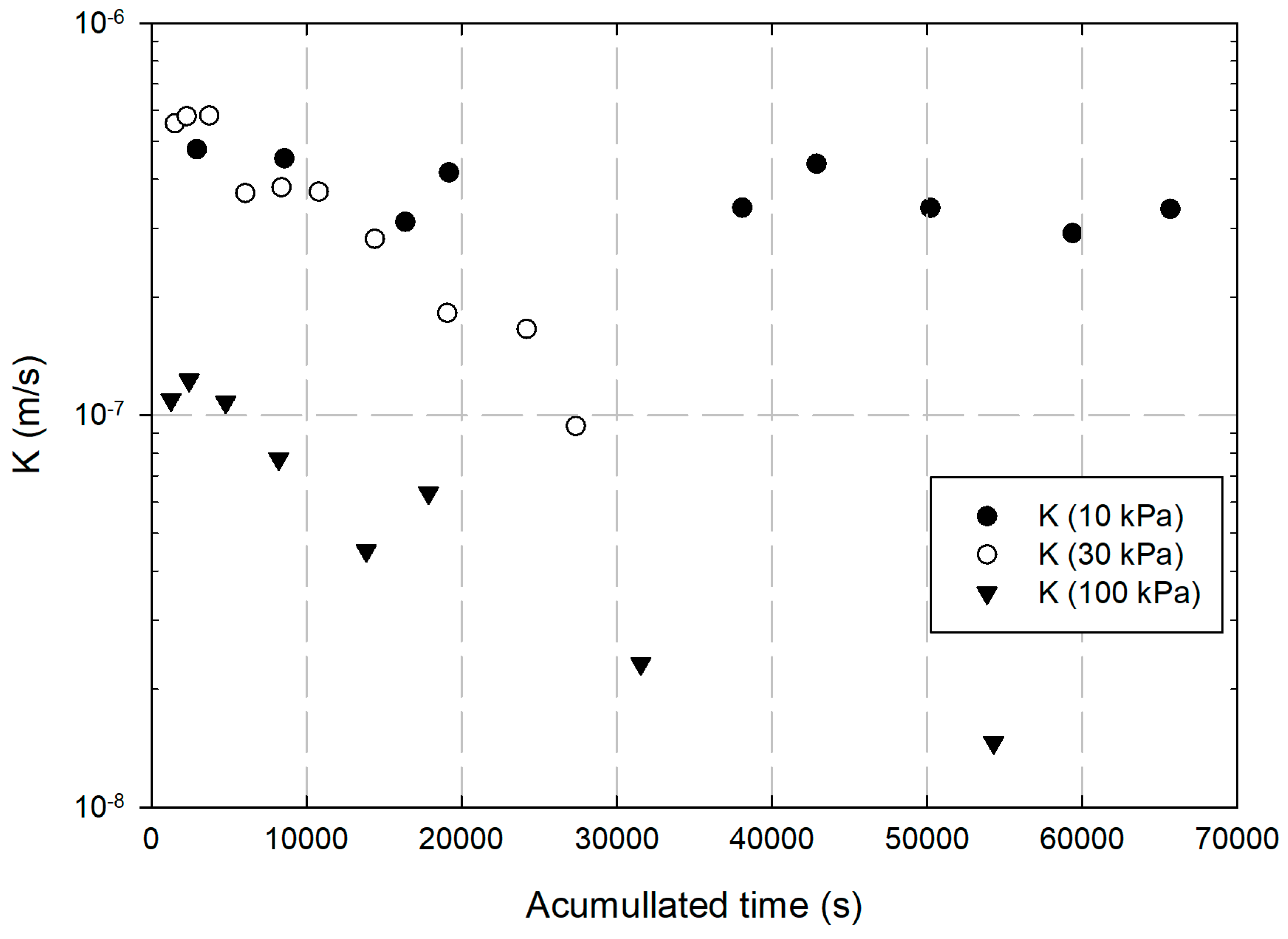
3. Results and Discussions
4. Conclusions
- nZVI is efficient for reducing Cr(VI) in clay soils.
- Injection of nZVI under high pressures could lead to Cr(VI) leaching, and its mobility in soil is low due to its reaction with the contaminant.
- Clogging of soil voids with nZVI could occur, resulting in reduced soil permeability.
- Low contaminant reduction is due to nZVI’s poor stability and mobility and its tendency to aggregate further reduce reduction reactivity.
- To remedy metal polluted soils, further studies are needed to improve mobility and reactivity of nZVI, especially when field application is desired.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hu, N.; Li, Z.; Huang, P.; Tao, C. Distribution and mobility of metals in agricultural soils near a copper smelter in South China. Environ. Geochem. Health 2006, 28, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Thomé, A.; Reddy, K.R.; Reginatto, C.; Cecchin, I. Review of nanotechnology for soil and groundwater remediation: Brazilian perspectives. Water Air Soil Pollut. 2015, 226, 2–20. [Google Scholar] [CrossRef]
- Jiang, D.; Guangming, Z.; Danlian, H.; Ming, C.; Chen, Z.; Chao, H.; Jia, W. Remediation of contaminated soils by enhanced nanoscale zero valent iron. Environ. Res. 2018, 163, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Wei, L.; Weiping, S.; Mingxin, G. Remediation techniques for heavy metal-contaminated soils: Principles and applicability. Sci. Total Environ. 2018, 633, 206–219. [Google Scholar] [CrossRef] [PubMed]
- Cotton, F.A.; Wilkinson, G. Advanced Inorganic Chemistry, 6th ed.; John Wiley & Sons: New York, NY, USA, 1999; p. 1376. [Google Scholar]
- Padmavathy, K.S.; Madhub, G.; Haseena, P.V. Study on effects of pH, adsorbent dosage, time, initial concentration and adsorption isotherm study for the removal of hexavalent chromium (Cr (VI)) from wastewater by magnetite nanoparticles. Proc. Technol. 2016, 24, 585–594. [Google Scholar] [CrossRef]
- Megharaj, M.; Avudainayagam, S.; Naidu, R. Toxicity of hexavalent chromium and its reduction by bacteria isolated from soil contaminated with tannery waste. Curr. Microbiol. 2003, 47, 51–54. [Google Scholar] [CrossRef]
- Di Palma, L.; Gueye, M.T.; Petrucci, E. Hexavalent chromium reduction in contaminated soil: A comparison between ferrous sulphate and nanoscale zero-valent iron. J. Hazard. Mater. 2015, 281, 70–76. [Google Scholar] [CrossRef]
- Grace Pavithra, K.; Jaikumar, V.; Senthil, K.P.; Panneer, S.S. A Review on Cleaner Strategies for Chromium Industrial Wastewater: Present Research and Future Perspective. J. Clean. Prod. 2019, 228, 580–593. [Google Scholar] [CrossRef]
- USEPA (U.S. Environmental Protection Agency). Nanotechnology: Applications for Environmental Remediation. 2019. Available online: http://www.cluin.org/techfocus/default.focus/sec/Nanotechnology:_Applications_for_Environmental_Remediation/cat/Application/#3 (accessed on 6 August 2019).
- Nanorem. Nanotechnological Remediation Processes from Lab Scale to End User Applications for the Restoration of a Clean Environment. 2018. Available online: http://www.nanorem.eu/index.aspx (accessed on 20 June 2018).
- Yan, W.; Lien, H.-L.; Koel, B.E.; Zhang, W.-X. Iron nanoparticles for environmental clean-up: Recent developments and future outlook. Environ. Sci. Process. Impacts 2013, 15, 63–77. [Google Scholar] [CrossRef]
- Choppala, G.; Bolan, N.; Park, J.H. Chromium contamination and its risk management in complex environmental settings. Adv. Agron. 2013, 120, 129–172. [Google Scholar] [CrossRef]
- Gueye, M.T.; Elisabetta, P.; Luca, D.P. Chemical Reduction of Hexavalent Chromium (VI) in Soil Slurry by Nano Zero Valent Iron. Chem. Eng. Trans. 2015, 43, 655–660. [Google Scholar] [CrossRef]
- Su, H.; Zhanqiang, F.; Pokeung, E.T.; Liuchun, Z.; Wen, C.; Jianzhang, F.; Dongye, Z. Remediation of hexavalent chromium contaminated soil by biochar supported zero-valent iron nanoparticles. J. Hazard. Mater. 2016, 318, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Gil-Díaz, M.; González, A.; Alonso, J.; Lobo, M.C. Evaluation of the stability of a nanoremediation strategy using barley plants. J. Environ. Manag. 2016, 165, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Wei, W.; Feipeng, L.; Wei-Xian, Z. Heavy metal removal using nanoscale zero-valent iron (nZVI): Theory and application. J. Hazard. Mater. 2017, 322, 163–171. [Google Scholar] [CrossRef]
- Hou, S.; Wu, B.; Peng, D.; Wang, Z.; Wang, Y.; Xu, H. Remediation performance and mechanism of hexavalent chromium in alkaline soil using multi-layer loaded nano-zero-valent iron. Environ. Pollut. 2019, 252, 553–561. [Google Scholar] [CrossRef]
- Vilardi, G.; Di Palma, L.; Verdone, N. A physical-based interpretation of mechanism and kinetics of Cr(VI) reduction in aqueous solution by zero-valent iron nanoparticles. Chemosphere 2019, 220, 590–599. [Google Scholar] [CrossRef] [PubMed]
- Karn, B.; Kuiken, T.; Otto, M. Nanotechnology and in situ remediation: A review of the benefits and potential risks. Environ. Health Perspect. 2009, 117, 1813–1831. [Google Scholar] [CrossRef]
- Reddy, K.; Darnault, C.; Darko-Kagya, K. Transport of Lactate-Modified Nanoscale Iron Particles in Porous Media. J. Geotech. Geoenviron. Eng. 2014, 140. [Google Scholar] [CrossRef]
- Crane, R.A.; Scott, T.B. Nanoscale zero-valent iron: Future prospects for an emerging water treatment technology. J. Hazard. Mater. 2012, 211–212, 112–125. [Google Scholar] [CrossRef]
- Fu, F.; Dionysiou, D.D.; Liu, H. The use of zero-valent iron for groundwater remediation and wastewater treatment: A review. J. Hazard. Mater. 2014, 267, 194–205. [Google Scholar] [CrossRef]
- Wang, M.; Gao, B.; Tang, D. Review of key factors controlling engineered nanoparticle transport in porous media. J. Hazard. Mater. 2016, 318, 233–246. [Google Scholar] [CrossRef]
- Reddy, K.R.; Darko-Kagya, K.; Cameselle, C. Electrokinetic-enhanced transport of lactate-modified nanoscale iron particles for degradation of dinitrotoluene in clayey soils. Sep. Purif. Technol. 2011, 79, 230–237. [Google Scholar] [CrossRef]
- Saleh, N.; Sirk, K.; Liu, Y.; Phenrat, T.; Dufour, B.; Matyjaszewski, K.; Tilton, R.D.; Lowry, G.V. Surface Modifications Enhance Nanoiron Transport and NAPL Targeting in Saturated Porous Media. Environ. Eng. Sci. 2007, 24, 45–57. [Google Scholar] [CrossRef]
- Tosco, T.; Papini, M.P.; Viggi, C.C.; Sethi, R. Nanoscale zerovalent iron particles for groundwater remediation: A review. J. Clean. Prod. 2014, 77, 10–21. [Google Scholar] [CrossRef]
- Zhang, W.-X. Nanoscale iron particles for environmental remediation: An overview. J. Nanopart. Res. 2003, 5, 323–332. [Google Scholar] [CrossRef]
- Schrick, B.; Blough, J.L.; Jones, A.D.; Mallouk, T.E. Hydro dechlorination of trichloroethylene to hydrocarbons using bimetallic nickel–iron nanoparticles. Chem. Mater. 2002, 14, 5140–5147. [Google Scholar] [CrossRef]
- Petosa, R.; Jaisi, D.P.; Quevedo, I.R.; Elimelech, M.; Tufenkji, N. Aggregation and deposition of engineered nanomaterials in aquatic environments: Role of physicochemical interactions. Environ. Sci. Technol. 2010, 44, 6532–6549. [Google Scholar] [CrossRef]
- Yang, F.; Zhang, S.; Sun, Y.; Cheng, K.; Li, J.; Tsang, D.C.W. Fabrication and characterization of hydrophilic corn stalk biochar-supported nanoscale zerovalent iron composites for efficient metal removal. Bioresour. Technol. 2018, 265, 490–497. [Google Scholar] [CrossRef]
- Vilardi, G.; Rodriguez-Rodriguez, J.; Miguel, O.-P.J.; Di Palma, L.; Verdone, N. Fixed-bed reactor scale-up and modelling for Cr(VI) removal using nano iron-based coated biomass as packing material. Chem. Eng. J. 2019, 361, 990–998. [Google Scholar] [CrossRef]
- He, X.; Pei, Z.; Xinhong, Q. Remediation of hexavalent chromium in contaminated soil by Fe(II)-Al layered double hydroxide. Chemosphere 2018, 210, 1157–1166. [Google Scholar] [CrossRef]
- Pei, G.; Yuen, Z.; Junguo, W.; Yanxi, P.; Hua, L. Vinegar residue supported nanoscale zero-valent iron: Remediation of hexavalent chromium in soil. Environ. Pollut. 2020, 256. [Google Scholar] [CrossRef] [PubMed]
- Streck, E.V.; Kampf, N.; Dalmolin, R.S.D.; Klamt, E.; Nascimento, P.C.; Schneider, P.; Giasson, E.; Pinto, L.F.S. Rio Grande do Sul Soils, 2nd ed.; Emater/Rs-Ascar: Porto Alegre, Ufrgs, Brazil, 2008. (In Portuguese) [Google Scholar]
- CONAMA—National Environment Council No 420. In It Provides for Soil Quality Criteria and Guiding Values for the Presence of Chemicals and Establishes Guidelines for the Environmental Management of Areas Contaminated by These Substances as a Result of Anthropic Activities; National Environment Council: Brasília, Brazil, 2009; pp. 81–84.
- ASTM. Standard Test Method for Leaching Solid Material in a Column Apparatus; ASTM D4874; ASTM International: West Conshohocken, PA, USA, 2014; Available online: http://compass.astm.org/ (accessed on 15 November 2018).
- Reginatto, C.; Cecchin, I.; Thomé, A.; Keineck, K.S.; Reddy, K.R. Influence of nanoscale zero-valent iron on hydraulic conductivity of a residual clayey soil and modeling of the filtration parameter. Environ. Sci. Pollut. Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- NANOIRON. Nanoiron, Future Technology. 2019. Available online: http://www.nanoiron.cz (accessed on 25 November 2019).
- USEPA (U.S. Environmental Protection Agency). Method 3060a: Alkaline Digestion for Hexavalent Chromium. 1996. Available online: https://www.epa.gov/sites/production/files/2015-12/documents/3060a.pdf (accessed on 16 December 2018).
- USEPA (U.S. Environmental Protection Agency). Method 7196A: Chromium, Hexavalent (Colorimetric). 1992. Available online: https://www.epa.gov/sites/production/files/2015-12/documents/7196a.pdf (accessed on 5 December 2018).
- StatSoft Version 5.5 [Computer software]; StatSoft, Tulsa: Tulsa, OK, USA, 1999.
- Lyu, H.; Zhao, H.; Tang, J.; Gong, Y.; Huang, Y.; Wu, Q.; Gao, B. Immobilization of hexavalent chromium in contaminated soils using biochar supported nanoscale iron sulfide composite. Chemosphere 2018, 194, 360–369. [Google Scholar] [CrossRef]
- Wang, S.; Zhao, M.; Zhou, M.; Li, Y.C.; Wang, J.; Gao, B.; Sato, S.; Feng, K.; Yin, W.; Igalavithana, A.D.; et al. Biochar-supported nZVI (nZVI/BC) for contaminant removal from soil and water: A critical review. J. Hazard. Mater. 2019, 373, 820–834. [Google Scholar] [CrossRef]
- Ambika, S.; Nambi, I.M. Optimized synthesis of methanol-assisted nZVI for assessing reactivity by systematic chemical speciation approach at neutral and alkaline conditions. J. Water Proc. Eng. 2016, 13, 107–116. [Google Scholar] [CrossRef]
- Zhu, F.; Li, L.; Ren, W.; Deng, X.; Liu, T. Effect of pH, temperature, humic acid and coexisting anions on reduction of Cr(VI) in the soil leachate by nZVI/Ni bimetal material. Environ. Pollut. 2017, 227, 444–450. [Google Scholar] [CrossRef]
| Parameter | Value |
|---|---|
| Clay (%) | 68 |
| Silt (%) | 5 |
| Sand (%) | 27 |
| Natural moisture content (%) | 34 |
| Unit weight (kN/m3) | 16.03 |
| Void ratio | 1.24 |
| Degree of saturation (%) | 73.5 |
| Porosity (%) | 54 |
| pH | 5.4 |
| Organic matter (%) | 0.5 |
| Cation Exchange Capacity (CEC) (cmolc/dm3) | 8.6 |
| Hydraulic conductivity (m/s) | 1.39 × 10−5 |
| nZVI Injection Pressure (kPa) | Cr(VI) Reduction Efficiency (%) | |
|---|---|---|
| Average | Standard Deviation | |
| 10 | 23.6 | 1.5 |
| 30 | 48.7 | 2.0 |
| 100 | 47.2 | 5.2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reginatto, C.; Cecchin, I.; Heineck, K.S.; Thomé, A.; Reddy, K.R. Use of Nanoscale Zero-Valent Iron for Remediation of Clayey Soil Contaminated with Hexavalent Chromium: Batch and Column Tests. Int. J. Environ. Res. Public Health 2020, 17, 1001. https://doi.org/10.3390/ijerph17031001
Reginatto C, Cecchin I, Heineck KS, Thomé A, Reddy KR. Use of Nanoscale Zero-Valent Iron for Remediation of Clayey Soil Contaminated with Hexavalent Chromium: Batch and Column Tests. International Journal of Environmental Research and Public Health. 2020; 17(3):1001. https://doi.org/10.3390/ijerph17031001
Chicago/Turabian StyleReginatto, Cleomar, Iziquiel Cecchin, Karla Salvagni Heineck, Antonio Thomé, and Krishna R. Reddy. 2020. "Use of Nanoscale Zero-Valent Iron for Remediation of Clayey Soil Contaminated with Hexavalent Chromium: Batch and Column Tests" International Journal of Environmental Research and Public Health 17, no. 3: 1001. https://doi.org/10.3390/ijerph17031001
APA StyleReginatto, C., Cecchin, I., Heineck, K. S., Thomé, A., & Reddy, K. R. (2020). Use of Nanoscale Zero-Valent Iron for Remediation of Clayey Soil Contaminated with Hexavalent Chromium: Batch and Column Tests. International Journal of Environmental Research and Public Health, 17(3), 1001. https://doi.org/10.3390/ijerph17031001






