Evaluation of Soft Tissue Mobilization in Patients with Temporomandibular Disorder-Myofascial Pain with Referral
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Issues
2.2. Subjects and the Size of the Sample
2.2.1. Inclusion Criteria
- -
- Pain in the craniofacial and/or craniomandibular areas at the level of 8 points or more with respect to VAS (visual analog scale);
- -
- Full natural dentition (class I according to angle’s molar classification and canine relations);
- -
- Lack of orthodontic treatment history or retention status after completion of treatment exceeding 3 years.
2.2.2. Exclusion Criteria
- -
- Traumas or surgical procedures within the craniofacial region;
- -
- Occlusal splint therapy;
- -
- Prosthetic treatment before recruitment to the study;
- -
- Metabolic diseases;
- -
- Cases in which medication or possible health concerns could affect the functioning of the masticatory muscles;
- -
- Previous physiotherapeutic treatment within the craniofacial, craniomandibular and/or craniocervical areas.
2.3. General Description of the Method
- Functional examination of temporomandibular joints and muscles of the stomatognathic system with respect to the diagnostic criteria for temporomandibular disorders (DC/TMD) [14]—axes I and II;
- Electromyography (BioEMG, BioResearch, Inc., Milwaukee, WI, USA);
- Soft tissues mobilization;
- Statistical analysis using the Statistica 13.1 software (TIBCO Software Inc., Statsoft, Cracow, Poland), IBM SPSS Statistics 26.0 (IBM Corporation, Warsaw, Poland) and PQStat Software v. 1.6.8. (PQStat Software, Poznan, Poland)
2.4. Electromyography (EMG)
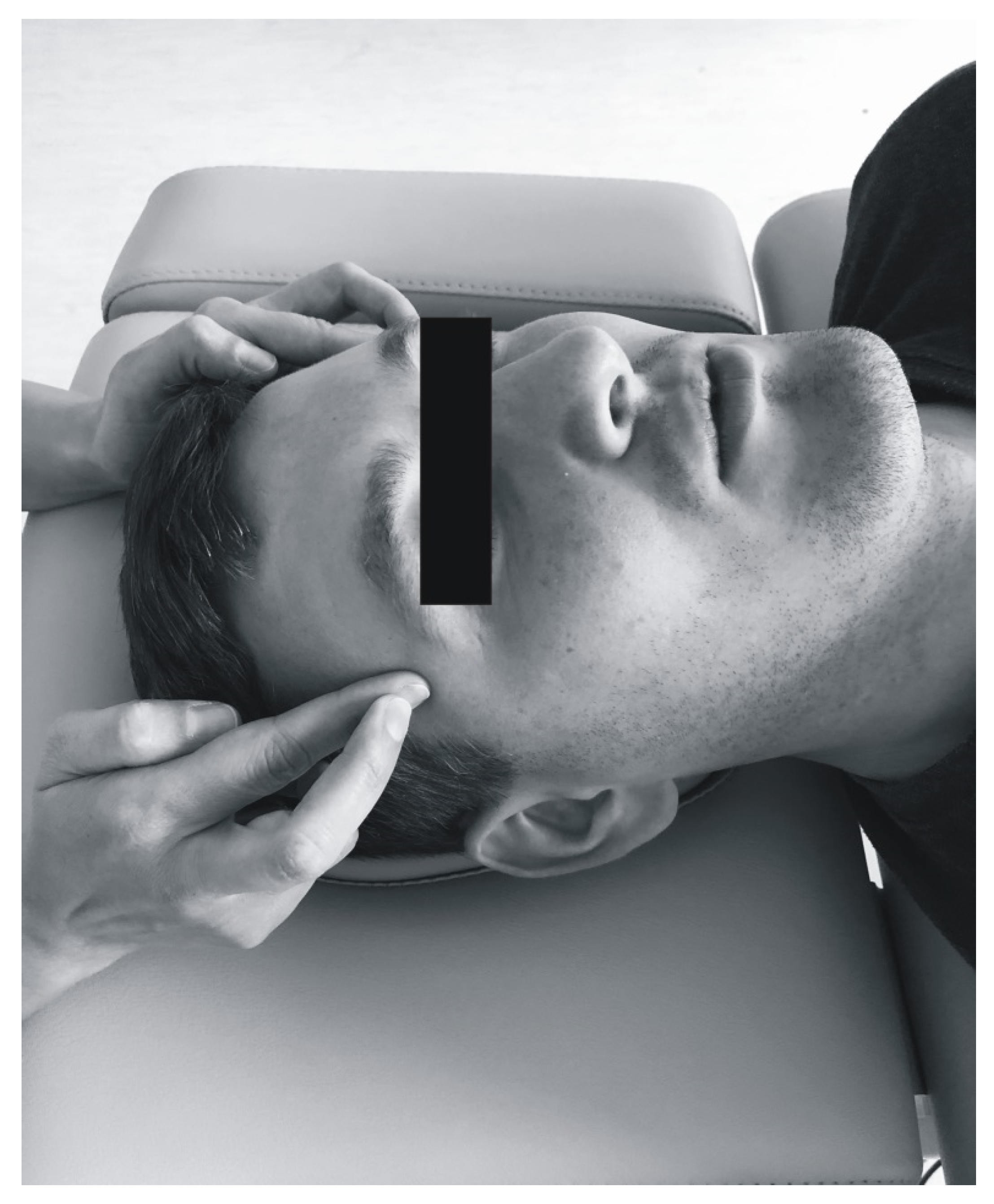
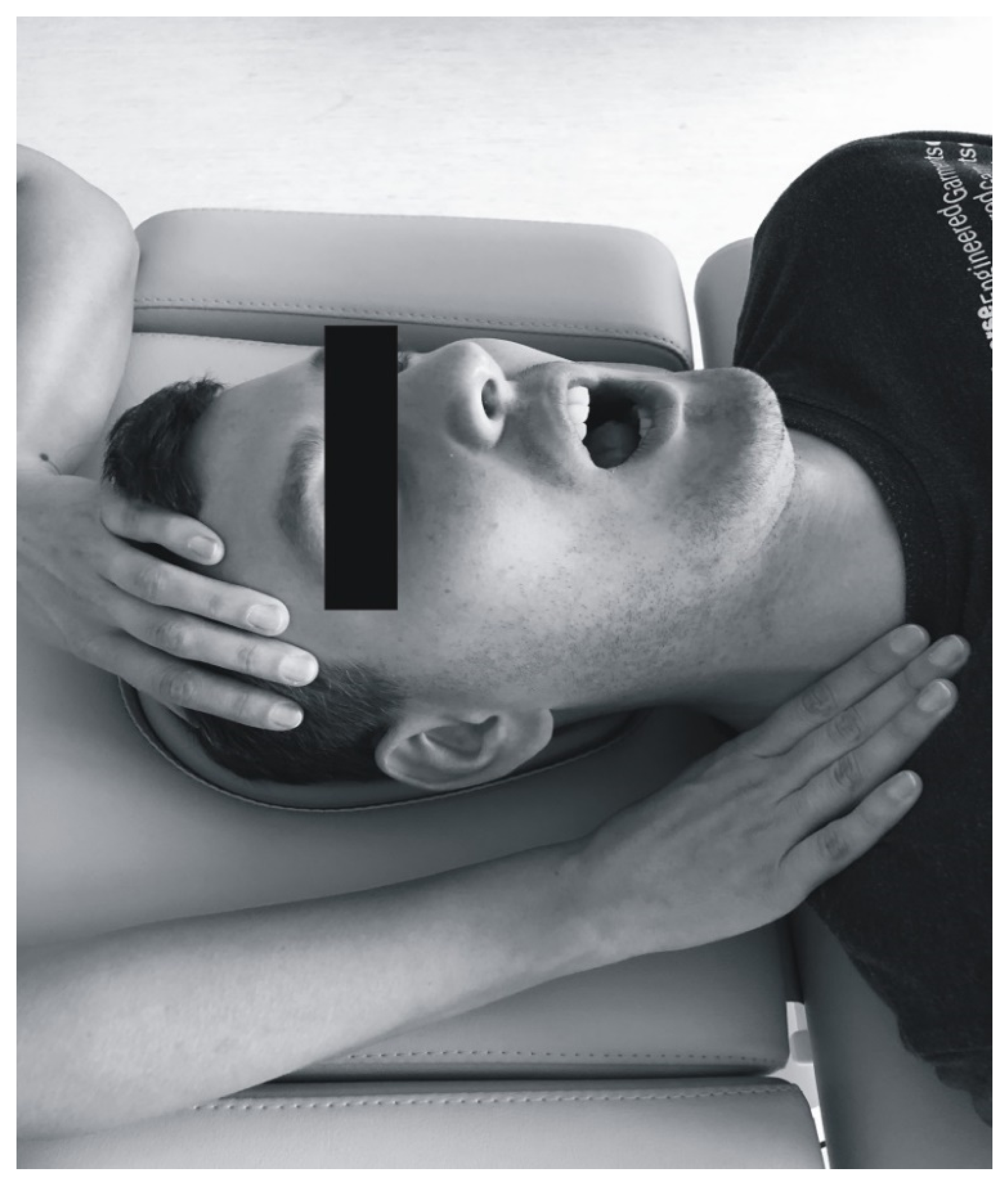
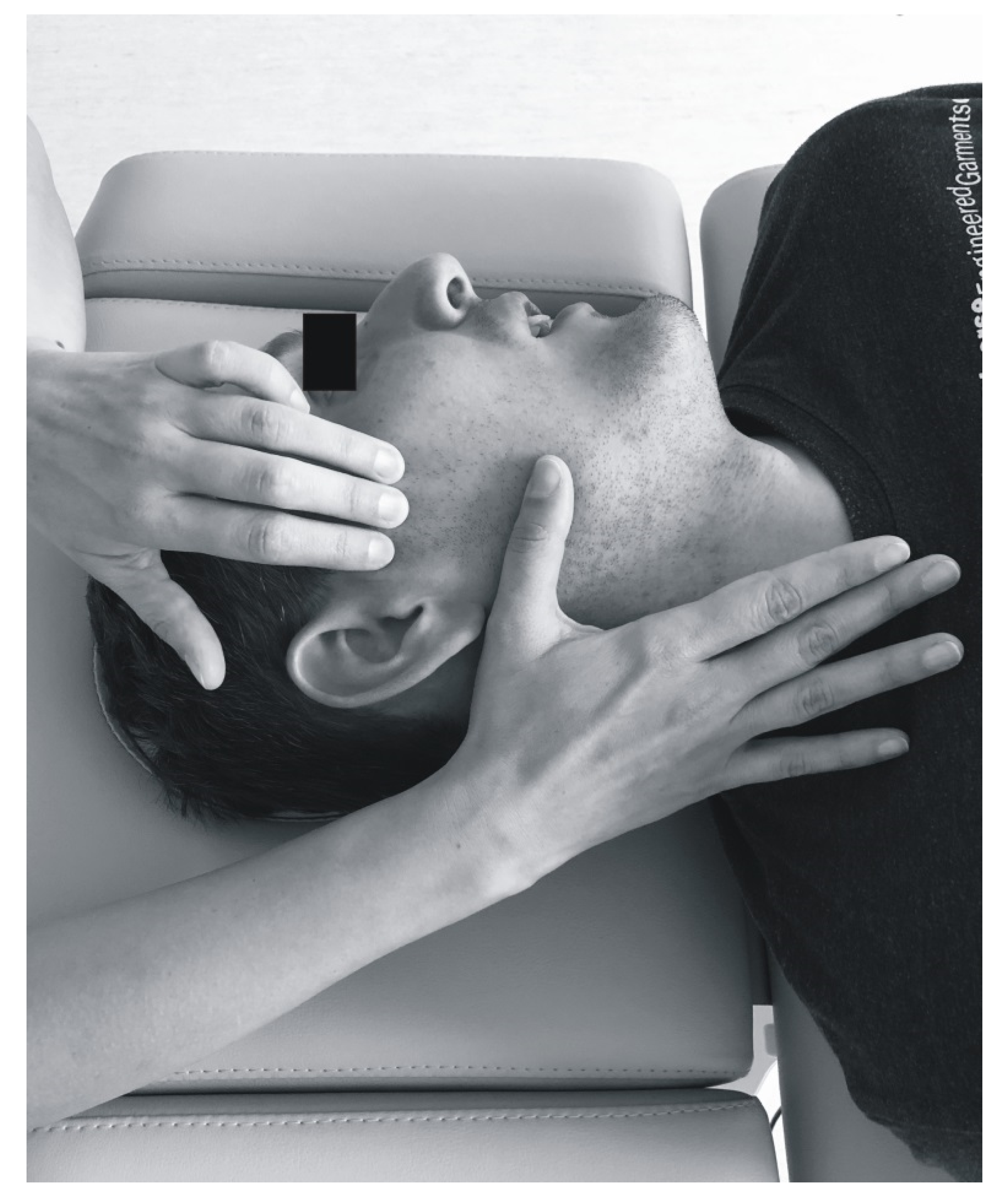
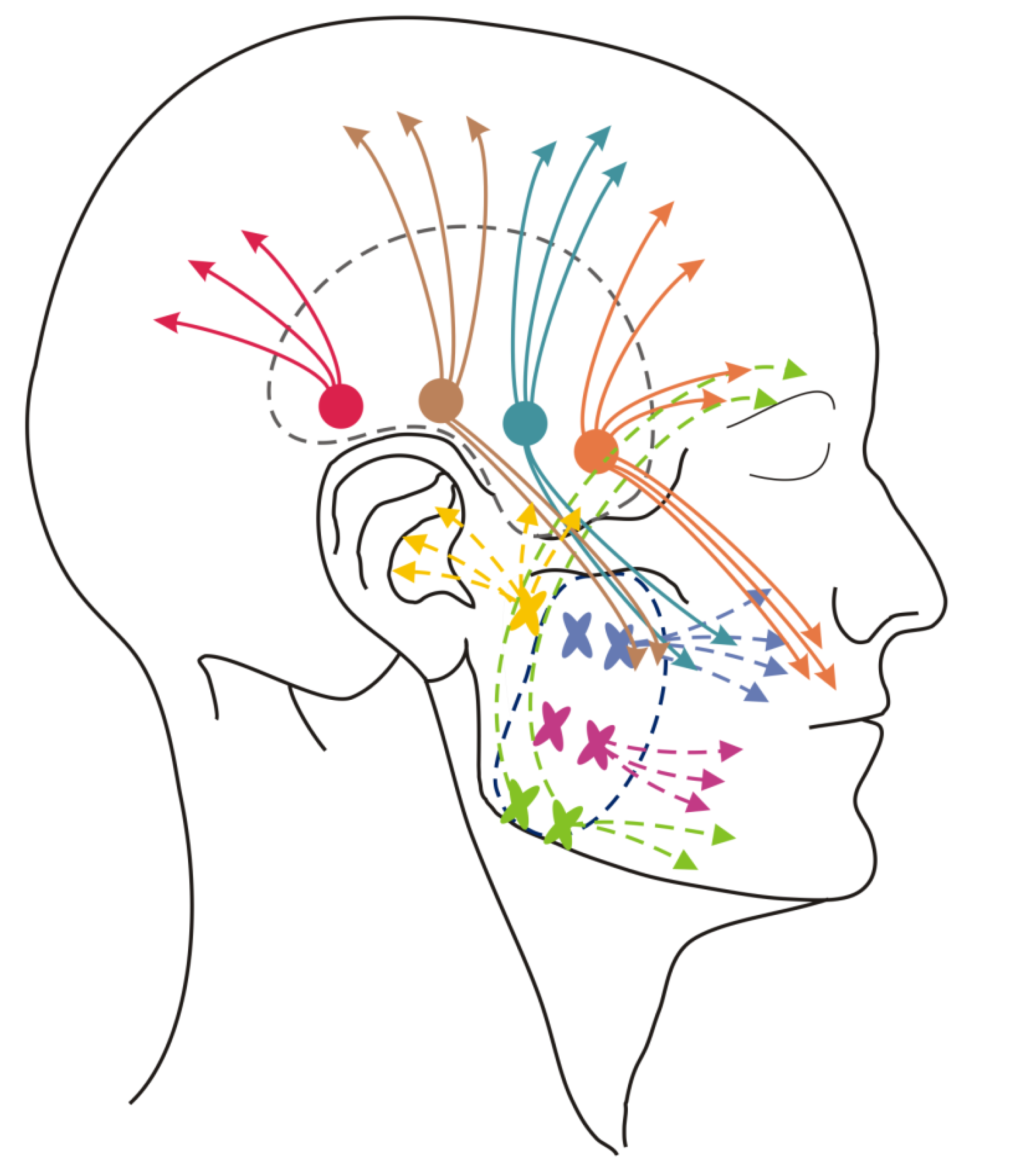
2.5. Soft Tissue Mobilization
2.6. Statistical Analysis
3. Results
4. Discussion
Strengths and Limitations of the Study
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Travell, J.; Rinzler, S.H. The myofascial genesis of pain. Postgrad. Med. 1952, 11, 425–434. [Google Scholar] [CrossRef]
- Khan, M.; Nishi, S.E.; Hassan, S.N.; Islam, M.; Gan, S.H. Trigeminal neuralgia, glossopharyngeal neuralgia, and myofascial pain dysfunction syndrome: An update. Pain Res. Manag. 2017, 2017, 7438326. [Google Scholar] [CrossRef]
- Simons, D.G.; Travell, J.G.; Simons, L.S. Travell & Simons’ Myofascial Pain and Dysfunction: Upper Half of Body; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 1999; Volume 1. [Google Scholar]
- Mense, S.; Gerwin, R.D. Muscle Pain: Diagnosis and Treatment; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2010. [Google Scholar]
- Jafri, M.S. Mechanisms of myofascial pain. Int. Sch. Res. Not. 2014, 2014, 523924. [Google Scholar] [CrossRef] [PubMed]
- Gerber, L.H.; Sikdar, S.; Armstrong, K.; Diao, G.; Heimur, J.; Kopecky, J.; Turo, D.; Otto, P.; Gebreab, T.; Shah, J. A systematic comparison between subjects with no pain and pain associated with active myofascial trigger points. PMR 2013, 5, 931–938. [Google Scholar] [CrossRef] [PubMed]
- Vázquez Delgado, E.; Cascos-Romero, J.; Gay Escoda, C. Myofascial pain syndrome associated with trigger points: A literature review.(I): Epidemiology, clinical treatment and etiopathogeny. Med. Oral Patol. Oral Cir. Bucal 2009, 14, 494–498. [Google Scholar] [CrossRef] [PubMed]
- Svensson, P.; Graven-Nielsen, T. Craniofacial muscle pain: Review of mechanisms and clinical manifestations. J. Orofac. Pain 2001, 15, 117–145. [Google Scholar] [PubMed]
- Mortazavi, H.; Javadzadeh, A.; Delavarian, Z.; Zare Mahmoodabadi, R. Myofascial pain dysfunction syndrome (MPDS). Iran. J. Otorhinolaryngol. 2010, 22, 131–136. [Google Scholar]
- Gerwin, R.D. Classification, epidemiology, and natural history of myofascial pain syndrome. Curr. Pain Headache Rep. 2001, 5, 412–420. [Google Scholar] [CrossRef]
- Osiewicz, M.; Manfredini, D.; Biesiada, G.; Czepiel, J.; Garlicki, A.; Aarab, G.; Pytko-Polończyk, J.; Lobbezoo, F. Prevalence of function-dependent temporomandibular joint and masticatory muscle pain, and predictors of temporomandibular disorders among patients with Lyme disease. J. Clin. Med. 2019, 8, 929. [Google Scholar] [CrossRef] [PubMed]
- Okeson, J.P. Management of Temporomandibular Disorders and Occlusion-E-Book; Elsevier Health Sciences: Beijing, China, 2014. [Google Scholar]
- Fiorillo, L.; Musumeci, G. TMJ Dysfunction and Systemic Correlation. J. Funct. Morphol. Kinesiol. 2020, 5, 20. [Google Scholar] [CrossRef]
- Ohrbach, R.; Gonzalez, Y.; List, T.; Michelotti, A.; Schiffman, E. Diagnostic Criteria for Temporomandibular Disorders (DC/TMD) Clinical Examination Protocol. 2013. Available online: www.rdc-tmdinternational.org (accessed on 2 June 2013).
- Kuć, J.; Szarejko, K.D.; Sierpinska, T. Evaluation of Orofacial and General Pain Location in Patients with Temporomandibular Joint Disorder-Myofascial Pain With Referral. Front. Neurol. 2019, 10, 546. [Google Scholar] [CrossRef] [PubMed]
- Khaled, Y.; Brennan, M.; Napeñas, J.; Quach, J. Using physical therapy to treat temporomandibular disorders. A cohort study. J. Dent. Sci. Oral Maxillofac. Res. 2018, 1, 31–35. [Google Scholar]
- Falsiroli Maistrello, L.; Geri, T.; Gianola, S.; Zaninetti, M.; Testa, M. Effectiveness of trigger point manual treatment on the frequency, intensity, and duration of attacks in primary headaches: A systematic review and meta-analysis of randomized controlled trials. Front. Neurol. 2018, 9, 254. [Google Scholar] [CrossRef] [PubMed]
- Ginszt, M.; Zieliński, G.; Berger, M.; Szkutnik, J.; Bakalczuk, M.; Majcher, P. Acute Effect of the Compression Technique on the Electromyographic Activity of the Masticatory Muscles and Mouth Opening in Subjects with Active Myofascial Trigger Points. Appl. Sci. 2020, 10, 7750. [Google Scholar] [CrossRef]
- Shaffer, S.M.; Brismée, J.-M.; Sizer, P.S.; Courtney, C.A. Temporomandibular disorders. Part 2: Conservative management. J. Man. Manip. Ther. 2014, 22, 13–23. [Google Scholar] [CrossRef]
- Bialosky, J.E.; Bishop, M.D.; Price, D.D.; Robinson, M.E.; George, S.Z. The mechanisms of manual therapy in the treatment of musculoskeletal pain: A comprehensive model. Man. Ther. 2009, 14, 531–538. [Google Scholar] [CrossRef]
- Bishop, M.D.; Torres-Cueco, R.; Gay, C.W.; Lluch-Girbés, E.; Beneciuk, J.M.; Bialosky, J.E. What effect can manual therapy have on a patient’s pain experience? Pain Manag. 2015, 5, 455–464. [Google Scholar] [CrossRef]
- Vigotsky, A.D.; Bruhns, R.P. The role of descending modulation in manual therapy and its analgesic implications: A narrative review. Pain Res. Treat. 2015, 2015, 92805. [Google Scholar] [CrossRef]
- Gil-Martínez, A.; Paris-Alemany, A.; López-de-Uralde-Villanueva, I.; La Touche, R. Management of pain in patients with temporomandibular disorder (TMD): Challenges and solutions. J. Pain Res. 2018, 11, 571. [Google Scholar] [CrossRef]
- Weerapong, P.; Hume, P.A.; Kolt, G.S. The mechanisms of massage and effects on performance, muscle recovery and injury prevention. Sports Med. 2005, 35, 235–256. [Google Scholar] [CrossRef]
- Carlson, C.R.; Okeson, J.P.; Falace, D.A.; Nitz, A.J.; Anderson, D. Stretch-based relaxation and the reduction of EMG activity among masticatory muscle pain patients. J. Craniomandib. Disord. 1991, 5, 205–212. [Google Scholar]
- Biasotto-Gonzalez, D.A.; Bérzin, F. Electromyographic study of patients with masticatory muscles disorders, physiotherapeutic treatment (massage). Braz. J. Oral Sci. 2004, 3, 516–521. [Google Scholar]
- Capellini, V.K.; De Souza, G.S.; De Faria, C.R.S. Massage therapy in the management of myogenic TMD: A pilot study. J. Appl. Oral Sci. 2006, 14, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Manzotti, A.; Viganoni, C.; Lauritano, D.; Bernasconi, S.; Paparo, A.; Risso, R.; Nanussi, A. Evaluation of the Stomatognathic System before and after Osteopathic Manipulative Treatment in 120 Healthy People by Using Surface Electromyography. Int. J. Environ. Res. Public Health 2020, 17, 3250. [Google Scholar] [CrossRef] [PubMed]
- Lerman, M.D. The muscle engram: The reflex that limits conventional occlusal treatment. CRANIO® 2011, 29, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Alajbeg, I.Z.; Valentic-Peruzovic, M.; Alajbeg, I.; Cifrek, M. The influence of age and dental status on elevator and depressor muscle activity. J. Oral Rehabil. 2006, 33, 94–101. [Google Scholar] [CrossRef]
- Gerdi Kittel Ries, L.; Correa Alves, M.; Berzin, F. Asymmetric activation of temporalis, masseter, and sternocleidomastoid muscles in temporomandibular disorder patients. CRANIO® 2008, 26, 59–64. [Google Scholar] [CrossRef]
- Tartaglia, G.M.; da Silva, M.A.M.R.; Bottini, S.; Sforza, C.; Ferrario, V.F. Masticatory muscle activity during maximum voluntary clench in different research diagnostic criteria for temporomandibular disorders (RDC/TMD) groups. Man. Ther. 2008, 13, 434–440. [Google Scholar] [CrossRef]
- Ballenberger, N.; von Piekartz, H.; Danzeisen, M.; Hall, T. Patterns of cervical and masticatory impairment in subgroups of people with temporomandibular disorders—An explorative approach based on factor analysis. CRANIO® 2018, 36, 74–84. [Google Scholar] [CrossRef]
- Von Piekartz, H.; Lüdtke, K. Effect of treatment of temporomandibular disorders (TMD) in patients with cervicogenic headache: A single-blind, randomized controlled study. CRANIO® 2011, 29, 43–56. [Google Scholar] [CrossRef]
- Cuccia, A.; Caradonna, C.; Annunziata, V.; Caradonna, D. Osteopathic manual therapy versus conventional conservative therapy in the treatment of temporomandibular disorders: A randomized controlled trial. J. Bodyw. Mov. Ther. 2010, 14, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Ismail, F.; Demling, A.; Hessling, K.; Fink, M.; Stiesch-Scholz, M. Short-term efficacy of physical therapy compared to splint therapy in treatment of arthrogenous TMD. J. Oral Rehabil. 2007, 34, 807–813. [Google Scholar] [CrossRef] [PubMed]
- Espí-López, G.V.; Arnal-Gómez, A.; Cuerda del Pino, A.; Benavent-Corai, J.; Serra-Añó, P.; Inglés, M. Effect of manual therapy and splint therapy in people with temporomandibular disorders: A preliminary study. J. Clin. Med. 2020, 9, 2411. [Google Scholar] [CrossRef]
- Calixtre, L.; Moreira, R.; Franchini, G.; Alburquerque-Sendín, F.; Oliveira, A. Manual therapy for the management of pain and limited range of motion in subjects with signs and symptoms of temporomandibular disorder: A systematic review of randomised controlled trials. J. Oral Rehabil. 2015, 42, 847–861. [Google Scholar] [CrossRef] [PubMed]
- Nitecka-Buchta, A.; Nowak-Wachol, A.; Wachol, K.; Walczyńska-Dragon, K.; Olczyk, P.; Batoryna, O.; Kempa, W.; Baron, S. Myorelaxant Effect of Transdermal Cannabidiol Application in Patients with TMD: A Randomized, Double-Blind Trial. J. Clin. Med. 2019, 8, 1886. [Google Scholar] [CrossRef] [PubMed]
- Ashok, A.; Suganya, M.; Arun, B. Comparison of Myofascial Release, Muscle Energy Technique and Cervical Manual Therapy in Postural Neck Pain. Asian J. Orthop. Res. 2019, 2, 1–6. [Google Scholar]
- Fonder, A. The dental distress syndrome quantified. Basal Facts 1987, 9, 141–167. [Google Scholar]
- Adrian, E.D.; Umrath, K. The impulse discharge from the Pacinian corpuscle. J. Physiol. 1929, 68, 139. [Google Scholar] [CrossRef]
- Downey, P.A.; Barbano, T.; Kapur-Wadhwa, R.; Sciote, J.J.; Siegel, M.I.; Mooney, M.P. Craniosacral therapy: The effects of cranial manipulation on intracranial pressure and cranial bone movement. J. Orthop. Sports Phys. Ther. 2006, 36, 845–853. [Google Scholar] [CrossRef]
- Kandasamy, S.; Greene, C.S. The evolution of temporomandibular disorders: A shift from experience to evidence. J. Oral Pathol. Med. 2020, 49, 461–469. [Google Scholar] [CrossRef]


| Variables | Before 1st Treatment | After 1st Treatment | Before 2nd Treatment | After 2nd Treatment | Before 3rd Treatment | After 3rd Treatment | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | |
| MT-R | 108.0 | 48.7 | 99.0 | 82.3 | 27.4 | 77.0 | 84.0 | 28.9 | 81.5 | 79.1 | 33.2 | 70.5 | 86.1 | 32.5 | 83.0 | 77.8 | 27.2 | 76.0 |
| MT-L | 93.1 | 30.3 | 94.0 | 80.5 | 25.4 | 76.0 | 86.2 | 34.0 | 78.5 | 79.2 | 31.9 | 70.5 | 86.0 | 33.8 | 81.5 | 77.8 | 25.1 | 76.5 |
| MM-R | 182.7 | 74.3 | 179.5 | 128.9 | 51.7 | 124.0 | 133.0 | 48.1 | 121.0 | 111.2 | 41.8 | 105.0 | 130.0 | 49.9 | 121.0 | 115.6 | 36.7 | 116.5 |
| MM-L | 168.7 | 75.7 | 177.5 | 129.9 | 54.6 | 125.5 | 134.3 | 52.5 | 125.0 | 115.5 | 45.8 | 111.5 | 129.7 | 52.1 | 126.0 | 119.6 | 42.4 | 117.0 |
| SCM-R | 11.8 | 7.4 | 10.0 | 9.9 | 6.7 | 7.0 | 10.1 | 6.6 | 9.0 | 10.1 | 6.8 | 8.0 | 11.9 | 11.1 | 8.0 | 9.6 | 6.7 | 8.0 |
| SCM-L | 14.3 | 9.8 | 11.0 | 10.7 | 6.6 | 9.0 | 10.5 | 5.8 | 9.0 | 10.8 | 9.7 | 8.0 | 10.6 | 6.9 | 9.0 | 10.1 | 7.3 | 8.0 |
| MD-R | 20.3 | 8.0 | 19.0 | 16.7 | 7.2 | 15.0 | 18.0 | 9.0 | 16.5 | 15.7 | 7.2 | 14.5 | 17.9 | 7.5 | 17.0 | 15.8 | 5.3 | 15.5 |
| MD-L | 19.5 | 7.8 | 18.0 | 15.0 | 7.0 | 13.5 | 15.3 | 6.9 | 15.0 | 13.5 | 5.8 | 12.0 | 16.1 | 7.1 | 14.0 | 13.1 | 3.9 | 14.0 |
| Post Hoc Tests/Pairwise Comparisons with the Bonferroni Correction | ||||||||||||||||||
| Friedman Test | Kendall’s Coefficient of Concordance | Before 1st and after 1st Treatment | Before 1st and after 2nd Treatment | Before 1st and after 3rd Treatment | Before 2nd and after 2nd Treatment | Before 3rd and after 3rd Treatment | ||||||||||||
| Friedman Statistic (Fr) | df | p-Value | W | p-Value | p-Value | p-Value | p-Value | p-Value | ||||||||||
| MT-R | 25.69493 | 5 | 0.00010 * | 0.10278 | 0.014 ** | 0.001 ** | 0.000 ** | 1.000 | 0.978 | |||||||||
| MT-L | 10.49828 | 5 | 0.06229 | 0.04199 | 0.634 | 0.045 ** | 0.154 | 1.000 | 1.000 | |||||||||
| MM-R | 75.59161 | 5 | 0.00000 * | 0.30237 | 0.000 ** | 0.000 ** | 0.000 ** | 0.089 | 0.029 * | |||||||||
| MM-L | 36.90299 | 5 | 0.00000 * | 0.14761 | 0.001 ** | 0.000 ** | 0.000 ** | 0.426 | 1.000 | |||||||||
| SCM-R | 18.37309 | 5 | 0.00251 * | 0.07349 | 0.143 | 0.054 ** | 0.001 ** | 1.000 | 0.594 | |||||||||
| SCM-L | 23.06490 | 5 | 0.00033 * | 0.09226 | 0.242 | 0.002 ** | 0.001 ** | 1.000 | 1.000 | |||||||||
| MD-R | 22.35103 | 5 | 0.00045 * | 0.08940 | 0.104 | 0.006 ** | 0.000 ** | 0.766 | 0.323 | |||||||||
| MD-L | 45.25414 | 5 | 0.00000 * | 0.18102 | 0.004 ** | 0.000 ** | 0.000 ** | 0.167 | 0.132 | |||||||||
| Variables | Before 1st Treatment | After 1st Treatment | Before 2nd Treatment | After 2nd Treatment | Before 3rd Treatment | After 3rd Treatment | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | |
| MT-R | 106.2 | 41.5 | 100.0 | 84.2 | 26.5 | 79.0 | 86.7 | 24.4 | 84.0 | 81.8 | 34.5 | 71.0 | 87.0 | 30.8 | 84.0 | 79.4 | 24.4 | 78.0 |
| MT-L | 96.8 | 28.8 | 95.0 | 81.4 | 23.6 | 80.0 | 84.2 | 29.1 | 77.0 | 82.3 | 32.3 | 74.0 | 83.7 | 26.5 | 82.0 | 79.9 | 25.8 | 77.0 |
| MM-R | 189.4 | 79.5 | 188.0 | 126.1 | 46.3 | 122.0 | 132.1 | 42.5 | 121.0 | 114.7 | 44.2 | 105.0 | 134.0 | 46.0 | 127.0 | 120.0 | 32.9 | 120.0 |
| MM-L | 168.2 | 72.0 | 178.0 | 126.8 | 46.5 | 123.0 | 133.1 | 44.5 | 123.0 | 117.9 | 46.8 | 112.0 | 133.5 | 43.7 | 131.0 | 122.2 | 35.1 | 122.0 |
| SCM-R | 11.2 | 6.7 | 10.0 | 8.7 | 6.3 | 7.0 | 9.7 | 6.8 | 8.0 | 9.5 | 6.0 | 8.0 | 10.3 | 9.9 | 8.0 | 8.8 | 5.9 | 7.0 |
| SCM-L | 13.3 | 8.5 | 10.0 | 8.9 | 3.9 | 8.0 | 9.4 | 4.3 | 9.0 | 10.7 | 10.6 | 8.0 | 9.1 | 5.7 | 8.0 | 8.3 | 4.6 | 7.0 |
| MD-R | 21.1 | 8.3 | 20.0 | 16.7 | 7.5 | 15.0 | 18.7 | 8.8 | 17.0 | 16.1 | 6.2 | 15.0 | 18.3 | 7.4 | 16.0 | 15.8 | 5.2 | 15.0 |
| MD-L | 19.6 | 7.8 | 19.0 | 14.7 | 5.9 | 14.0 | 16.0 | 6.9 | 15.0 | 14.6 | 5.8 | 14.0 | 16.8 | 7.7 | 14.0 | 13.0 | 3.9 | 14.0 |
| Post Hoc Tests/Pairwise Comparisons with the Bonferroni Correction | ||||||||||||||||||
| Friedman Test | Kendall’s Coefficient of Concordance | Before 1st and after 1st Treatment | Before 1st and after 2nd Treatment | Before 1st and after 3rd Treatment | Before 2nd and after 2nd Treatment | Before 3rd and after 3rd Treatment | ||||||||||||
| Friedman Statistic (Fr) | df | p-Value | W | p-Value | p-Value | p-Value | p-Value | p-Value | ||||||||||
| MT-R | 17.87051 | 5 | 0.00311 * | 0.09660 | 0.085 | 0.021 ** | 0.002 ** | 1.000 | 1.000 | |||||||||
| MT-L | 9.608830 | 5 | 0.08711 | 0.05194 | 0.379 | 0.178 | 0.273 | 1.000 | 1.000 | |||||||||
| MM-R | 54.16602 | 5 | 0.00000 * | 0.29279 | 0.000 ** | 0.000 ** | 0.000 ** | 0.561 | 0.043 ** | |||||||||
| MM-L | 27.07137 | 5 | 0.00006 * | 0.14633 | 0.003 ** | 0.000 ** | 0.004 ** | 0.702 | 1.000 | |||||||||
| SCM-R | 19.42691 | 5 | 0.00160 * | 0.10501 | 0.021 ** | 0.136 | 0.001 ** | 1.000 | 1.000 | |||||||||
| SCM-L | 21.46962 | 5 | 0.00066 * | 0.11605 | 0.178 | 0.023 ** | 0.000 ** | 1.000 | 1.000 | |||||||||
| MD-R | 17.80926 | 5 | 0.00320 * | 0.09627 | 0.297 | 0.163 | 0.000 ** | 1.000 | 0.000 ** | |||||||||
| MD-L | 32.17826 | 5 | 0.00001 * | 0.17394 | 0.006 ** | 0.000 ** | 0.000 ** | 0.935 | 0.000 ** | |||||||||
| Variables | Before 1st Treatment | After 1st Treatment | Before 2nd Treatment | After 2nd Treatment | Before 3rd Treatment | After 3rd Treatment | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | |
| MT-R | 113.2 | 67.0 | 85.0 | 76.8 | 30.2 | 66.0 | 76.3 | 39.2 | 76.0 | 71.3 | 28.8 | 70.0 | 83.5 | 38.3 | 76.0 | 73.5 | 34.8 | 61.0 |
| MT-L | 82.6 | 33.4 | 74.0 | 78.1 | 30.9 | 76.0 | 91.8 | 46.1 | 81.0 | 70.3 | 30.0 | 65.0 | 92.5 | 49.9 | 81.0 | 72.1 | 23.0 | 74.0 |
| MM-R | 163.9 | 55.3 | 172.0 | 136.9 | 66.3 | 126.0 | 135.8 | 63.4 | 132.0 | 101.4 | 33.3 | 105.0 | 118.5 | 60.1 | 115.0 | 103.2 | 44.7 | 106.0 |
| MM-L | 169.9 | 88.4 | 177.0 | 139.0 | 74.8 | 137.0 | 137.7 | 72.7 | 127.0 | 108.9 | 44.0 | 111.0 | 118.9 | 71.9 | 112.0 | 112.1 | 59.6 | 106.0 |
| SCM-R | 13.3 | 9.3 | 9.0 | 13.2 | 6.9 | 15.0 | 11.2 | 6.4 | 11.0 | 11.7 | 8.7 | 10.0 | 16.6 | 13.5 | 11.0 | 11.9 | 8.4 | 9.0 |
| SCM-L | 17.1 | 12.7 | 12.0 | 16.0 | 9.7 | 13.0 | 13.5 | 8.3 | 15.0 | 11.1 | 6.9 | 10.0 | 14.8 | 8.4 | 11.0 | 15.3 | 10.8 | 13.0 |
| MD-R | 18.2 | 7.2 | 16.0 | 16.8 | 6.7 | 14.0 | 15.9 | 9.7 | 13.0 | 14.5 | 9.9 | 12.0 | 16.9 | 7.9 | 17.0 | 16.0 | 5.8 | 16.0 |
| MD-L | 19.2 | 8.3 | 16.0 | 15.9 | 9.8 | 13.0 | 13.1 | 6.6 | 13.0 | 10.5 | 5.0 | 9.0 | 14.3 | 4.9 | 14.0 | 13.4 | 4.0 | 14.0 |
| Post Hoc Tests/Pairwise Comparisons with theBonferroni Correction | ||||||||||||||||||
| Friedman Test | Kendall’s Coefficient of Concordance | Before 1st and after 1st Treatment | Before 1st and after 2nd Treatment | Before 1st and after 3rd Treatment | Before 2nd and after 2nd Treatment | Before 3rd and after 3rd soft Treatment | ||||||||||||
| Friedman Statistic (Fr) | df | p-Value | W | p-Value | p-Value | p-Value | p-Value | p-Value | ||||||||||
| MT-R | 8.606195 | 5 | 0.12584 | 0.13240 | 0.999 | 0.096 | 0.363 | 1.000 | 1.000 | |||||||||
| MT-L | 7.989011 | 5 | 0.15685 | 0.12291 | 1.000 | 1.000 | 1.000 | 0.416 | 1.000 | |||||||||
| MM-R | 24.49115 | 5 | 0.00017 * | 0.37679 | 0.888 | 0.001 ** | 0.001 ** | 0.888 | 1.000 | |||||||||
| MM-L | 13.35541 | 5 | 0.02027 * | 0.20547 | 1.000 | 0.017 ** | 0.082 | 1.000 | 1.000 | |||||||||
| SCM-R | 6.531323 | 5 | 0.25790 | 0.10048 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |||||||||
| SCM-L | 6.008969 | 5 | 0.30535 | 0.09245 | 1.000 | 0.363 | 0.614 | 1.000 | 1.000 | |||||||||
| MD-R | 7.296380 | 5 | 0.19952 | 0.11225 | 1.000 | 0.153 | 1.000 | 1.000 | 1.000 | |||||||||
| MD-L | 20.87302 | 5 | 0.00086 * | 0.32112 | 1.000 | 0.000 ** | 0.363 | 0.999 | 1.000 | |||||||||
| Variables | Before 1st Treatment | After 1st Treatment | Before 2nd Treatment | After 2nd Treatment | Before 3rd Treatment | After 3rd Treatment | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | |
| MT ↔ | 81.9 | 12.0 | 83.0 | 83.5 | 12.7 | 87.0 | 83.1 | 11.5 | 85.5 | 85.5 | 9.7 | 84.0 | 82.2 | 11.5 | 84.5 | 86.0 | 11.2 | 89.5 |
| MM ↔ | 80.1 | 14.4 | 83.0 | 81.3 | 12.5 | 85.0 | 80.5 | 13.0 | 80.5 | 81.3 | 12.1 | 83.5 | 83.4 | 13.5 | 87.0 | 81.1 | 14.2 | 83.5 |
| SCM ↔ | 72.0 | 17.1 | 73.0 | 72.8 | 21.0 | 78.5 | 75.4 | 16.3 | 74.5 | 75.9 | 15.9 | 79.0 | 72.1 | 20.2 | 76.0 | 81.7 | 15.2 | 83.5 |
| MD ↔ | 82.4 | 16.3 | 86.5 | 80.4 | 15.7 | 82.5 | 79.1 | 15.5 | 81.0 | 83.3 | 12.0 | 83.5 | 82.3 | 13.8 | 86.0 | 83.5 | 11.0 | 84.0 |
| MT-R ↕ MM-R | 60.8 | 17.6 | 59.5 | 59.3 | 17.1 | 58.0 | 59.3 | 17.6 | 62.5 | 61.4 | 19.7 | 61.0 | 59.5 | 17.1 | 58.5 | 63.9 | 19.8 | 68.0 |
| MT-L ↕ MM-L | 58.6 | 18.4 | 59.0 | 61.3 | 19.0 | 63.0 | 58.2 | 16.8 | 59.5 | 63.7 | 19.7 | 62.5 | 61.9 | 19.2 | 62.0 | 62.7 | 20.7 | 59.5 |
| Post Hoc Tests/Pairwise Comparisons with the Bonferroni Correction | ||||||||||||||||||
| Friedman Test | Kendall’s Coefficient of Concordance | Before 1st and after 1st Treatment | Before 1st and after 2nd Treatment | Before 1st and after 3rd Treatment | Before 2nd and after 2nd Treatment | Before 3rd and after 3rd Treatment | ||||||||||||
| Friedman Statistic (Fr) | df | p-Value | W | p-Value | p-Value | p-Value | p-Value | p-Value | ||||||||||
| MT ↔ | 9.770749 | 5 | 0.08200 | 0.03908 | 1.000 | 1.000 | 0.456 | 1.000 | 0.122 | |||||||||
| MM ↔ | 6.036088 | 5 | 0.30273 | 0.02414 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |||||||||
| SCM ↔ | 15.84824 | 5 | 0.00729 * | 0.06339 | 1.000 | 1.000 | 0.004 ** | 1.000 | 0.113 | |||||||||
| MD ↔ | 2.116051 | 5 | 0.83286 | 0.00846 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |||||||||
| MT-R ↕ MM-R | 4.774697 | 5 | 0.44399 | 0.01910 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |||||||||
| MT-L ↕ MM-L | 6.312283 | 5 | 0.27701 | 0.02525 | 1.000 | 1.000 | 1.000 | 0.594 | 1.000 | |||||||||
| Variables | Before 1st Treatment | After 1st Treatment | Before 2nd Treatment | After 2nd Treatment | Before 3rd Treatment | After 3rd Treatment | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | |
| MT ↔ | 81.9 | 12.5 | 86.0 | 83.7 | 13.4 | 87.0 | 83.4 | 12.0 | 86.0 | 84.2 | 10.0 | 83.0 | 84.9 | 11.2 | 88.0 | 86.3 | 11.1 | 90.0 |
| MM ↔ | 83.0 | 11.8 | 85.0 | 81.1 | 12.6 | 85.0 | 82.7 | 12.8 | 83.0 | 82.3 | 10.1 | 84.0 | 86.3 | 11.5 | 89.0 | 83.1 | 12.8 | 85.0 |
| SCM ↔ | 72.2 | 18.2 | 75.0 | 74.9 | 22.0 | 81.0 | 75.1 | 16.6 | 74.0 | 75.1 | 15.1 | 78.0 | 75.4 | 19.7 | 81.0 | 81.6 | 16.8 | 84.0 |
| MD ↔ | 83.0 | 16.2 | 87.0 | 79.2 | 17.5 | 82.0 | 80.2 | 15.5 | 81.0 | 83.8 | 12.5 | 86.0 | 84.8 | 10.8 | 88.0 | 84.2 | 10.3 | 84.0 |
| MT-R ↕ MM-R | 62.4 | 17.1 | 62.0 | 61.0 | 17.1 | 59.0 | 60.2 | 17.0 | 63.0 | 63.7 | 20.1 | 64.0 | 60.0 | 17.0 | 59.0 | 65.3 | 19.3 | 70.0 |
| MT-L ↕ MM-L | 59.8 | 18.5 | 59.0 | 63.2 | 19.0 | 65.0 | 60.3 | 16.7 | 62.0 | 65.9 | 16.8 | 65.0 | 60.5 | 17.8 | 57.0 | 64.2 | 20.3 | 60.0 |
| Post Hoc Tests/Pairwise comparisons with the Bonferroni correction | ||||||||||||||||||
| Friedman Test | Kendall’s Coefficient of Concordance | Before 1st and after 1st Treatment | Before 1st and after 2nd Treatment | Before 1st and after 3rd Treatment | Before 2nd and after 2nd Treatment | Before 3rd and after 3rd Treatment | ||||||||||||
| Friedman Statistic (Fr) | df | p-Value | W | p-Value | p-Value | p-Value | p-Value | p-Value | ||||||||||
| MT ↔ | 5.164835 | 5 | 0.39610 | 0.02792 | 1.000 | 1.000 | 0.811 | 1.000 | 1.000 | |||||||||
| MM ↔ | 7.729068 | 5 | 0.17182 | 0.04178 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |||||||||
| SCM ↔ | 11.70313 | 5 | 0.03909 * | 0.06326 | 1.000 | 1.000 | 0.028 ** | 1.000 | 1.000 | |||||||||
| MD ↔ | 1.043784 | 5 | 0.95896 | 0.00564 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |||||||||
| MT-R ↕ MM-R | 5.261514 | 5 | 0.38481 | 0.02844 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |||||||||
| MT-L ↕ MM-L | 4.823668 | 5 | 0.43778 | 0.02607 | 1.000 | 0.811 | 1.000 | 1.000 | 1.000 | |||||||||
| Variables | Before 1st Treatment | After 1st Treatment | Before 2nd Treatment | After 2nd Treatment | Before 3rd Treatment | After 3rd Treatment | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | Mean | ±SD | Me | |
| MT ↔ | 82.0 | 10.9 | 82.0 | 83.1 | 10.9 | 82.0 | 82.1 | 10.5 | 82.0 | 89.2 | 8.0 | 93.0 | 74.6 | 8.8 | 78.0 | 84.9 | 11.8 | 87.0 |
| MM ↔ | 71.9 | 18.1 | 76.0 | 81.9 | 12.6 | 84.0 | 74.2 | 11.8 | 72.0 | 78.5 | 16.7 | 78.0 | 75.2 | 15.6 | 75.0 | 75.5 | 16.9 | 81.0 |
| SCM ↔ | 71.3 | 14.4 | 70.0 | 66.9 | 17.1 | 65.0 | 76.2 | 16.0 | 75.0 | 78.0 | 18.6 | 80.0 | 62.9 | 19.3 | 70.0 | 82.0 | 9.9 | 80.0 |
| MD ↔ | 80.6 | 17.3 | 83.0 | 83.7 | 8.3 | 83.0 | 75.9 | 15.7 | 78.0 | 82.0 | 10.6 | 79.0 | 75.2 | 18.9 | 81.0 | 81.8 | 13.1 | 84.0 |
| MT-R ↕ MM-R | 56.4 | 19.1 | 53.0 | 54.5 | 16.7 | 53.0 | 56.9 | 19.6 | 62.0 | 54.7 | 17.5 | 58.0 | 58.2 | 18.0 | 58.0 | 59.9 | 21.4 | 58.0 |
| MT-L ↕ MM-L | 55.2 | 18.3 | 59.0 | 56.1 | 18.7 | 53.0 | 52.4 | 16.4 | 55.0 | 57.2 | 25.8 | 45.0 | 65.9 | 23.0 | 67.0 | 58.6 | 22.2 | 59.0 |
| Post Hoc Tests/Pairwise Comparisons with theBonferroni Correction | ||||||||||||||||||
| FRIEDMAN TEST | Kendall’s Coefficient of Concordance | Before 1st and after 1st Treatment | Before 1st and after 2nd Treatment | Before 1st and after 3rd Treatment | Before 2nd and after 2nd Treatment | Before 3rd and after 3rd Treatment | ||||||||||||
| Friedman Statistic (Fr) | df | p-Value | W | p-Value | p-Value | p-Value | p-Value | p-Value | ||||||||||
| MT ↔ | 16.33630 | 5 | 0.00595 * | 0.25133 | 1.000 | 1.000 | 1.000 | 1.000 | 0.021 ** | |||||||||
| MM ↔ | 6.792035 | 5 | 0.23658 | 0.10449 | 0.316 | 1.000 | 1.000 | 1.000 | 1.000 | |||||||||
| SCM ↔ | 13.10155 | 5 | 0.02245 * | 0.20156 | 1.000 | 1.000 | 0.888 | 1.000 | 0.132 | |||||||||
| MD ↔ | 6.898455 | 5 | 0.22831 | 0.10613 | 1.000 | 1.000 | 1.000 | 1.000 | 0.614 | |||||||||
| MT-R ↕ MM-R | 2.033333 | 5 | 0.84451 | 0.03128 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |||||||||
| MT-L ↕ MM-L | 10.07778 | 5 | 0.07306 | 0.15504 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuć, J.; Szarejko, K.D.; Gołębiewska, M. Evaluation of Soft Tissue Mobilization in Patients with Temporomandibular Disorder-Myofascial Pain with Referral. Int. J. Environ. Res. Public Health 2020, 17, 9576. https://doi.org/10.3390/ijerph17249576
Kuć J, Szarejko KD, Gołębiewska M. Evaluation of Soft Tissue Mobilization in Patients with Temporomandibular Disorder-Myofascial Pain with Referral. International Journal of Environmental Research and Public Health. 2020; 17(24):9576. https://doi.org/10.3390/ijerph17249576
Chicago/Turabian StyleKuć, Joanna, Krzysztof Dariusz Szarejko, and Maria Gołębiewska. 2020. "Evaluation of Soft Tissue Mobilization in Patients with Temporomandibular Disorder-Myofascial Pain with Referral" International Journal of Environmental Research and Public Health 17, no. 24: 9576. https://doi.org/10.3390/ijerph17249576
APA StyleKuć, J., Szarejko, K. D., & Gołębiewska, M. (2020). Evaluation of Soft Tissue Mobilization in Patients with Temporomandibular Disorder-Myofascial Pain with Referral. International Journal of Environmental Research and Public Health, 17(24), 9576. https://doi.org/10.3390/ijerph17249576




