Viral Pandemics of the Last Four Decades: Pathophysiology, Health Impacts and Perspectives
Abstract
1. Introduction
2. Gender- and Age-Based Differences in the Susceptibility to Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2) Infection in Comparison with Other Viruses
3. Mechanism of Host Cell Invasion of SARS-CoV-2 in Comparison with Other Viruses
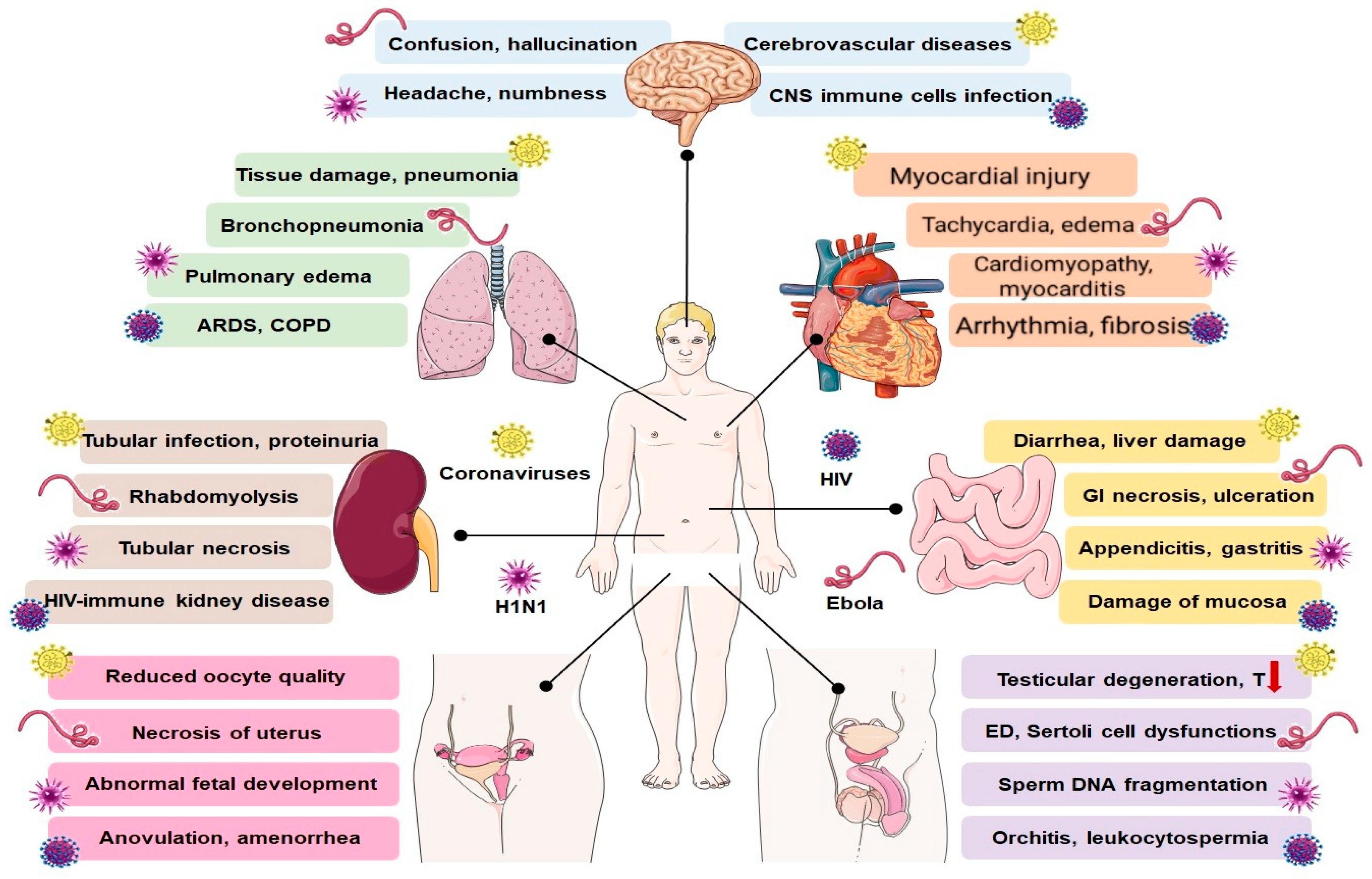
4. Effects of SARS-CoV-2 and Other Viruses on Major Physiological Processes
4.1. Respiratory System
4.2. Cardiovascular System
4.3. Gastrointestinal System
4.4. Nervous System
4.5. Renal System
4.6. Reproductive System
5. Outlooks on Vaccine Development for SARS-CoV-2 in Reference to SARS and Middle East Respiratory Syndrome Coronavirus (MERS-CoV)
6. Lessons Learned from the COVID-19 Pandemic and Other Viral Epidemics
6.1. Prompt Reporting
6.2. International Collaborations
6.3. Strengthening of Healthcare Facilities
6.4. Interventions
7. Perspective
Author Contributions
Funding
Conflicts of Interest
References
- Cascella, M.; Rajnik, M.; Cuomo, A.; Dulebohn, S.C.; Napoli, R.D. Features, evaluation and treatment coronavirus (COVID-19). In Statpearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2020; pp. 1–16. [Google Scholar]
- Bloom, D.E.; Cadarette, D. Infectious disease threats in the twenty-first century: Strengthening the global response. Front. Immunol. 2019, 10, 549. [Google Scholar] [CrossRef] [PubMed]
- Grubaugh, N.D.; Lander, J.T.; Lemey, P.; Pybus, O.G.; Rambaut, A.; Holmes, E.C.; Andersen, K.G. Tracking virus outbreaks in twenty-first century. Nat. Microbiol. 2019, 4, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Zappa, A.; Amendola, A.; Romano, L.; Zanetti, A. Emerging and re-emerging viruses in the era of globalization. Blood Transfus. 2009, 7, 167–171. [Google Scholar] [PubMed]
- Marco, M.D.; Baker, M.L.; Dasjak, P.; Barro, P.D.; Eskew, E.A.; Godde, C.M.; Harwood, T.D.; Herrero, M.; Hoskins, A.J.; Johnson, E.; et al. Sustainable development must account for pandemic risk. Proc. Natl. Acad. Sci. USA 2020, 117, 3888–3892. [Google Scholar] [CrossRef] [PubMed]
- Joint United Nations Programme on HIV/AIDS (UNAIDS). Report on the Global HIV/AIDS Epidemic. Available online: https://data.unaids.org/pub/report/2000/2000_gr_en.pdf (accessed on 29 November 2020).
- Gayle, H.D.; Hill, G.L. Global impacts of human immunodeficiency virus and AIDS. Clin. Microbiol. Rev. 2001, 14, 327–335. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Inadequate Plumbing Systems Likely Contributed to SARS Transmission. 2003. Available online: https://www.who.int/mediacentre/releases/2003/pr70/en/ (accessed on 29 November 2020).
- Abdullah, A.S.M.; Tomlinson, B.; Thomas, G.N.; Cockram, C.S. Impacts of SARS on health care systems and strategies for combating future outbreaks of emerging infectious diseases. In Learning from SARS: Preparing for the Next Disease Outbreak; Knobler, S., Mahmoud, A.D., Lemon, S., Mack, A., Sivitz, L., Oberholtzer, K., Eds.; National Academic Press: Washington, DC, USA, 2004; pp. 83–90. [Google Scholar]
- Cheng, V.C.C.; To, K.K.W.; Tse, H.; Hung, I.F.N.; Yuen, K.-Y. Two tears after pandemic influenza A/2009/H1N1: What have we learned? Clin. Microbiol. Rev. 2012, 25, 223–263. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Middle East Respiratory Syndrome Coronavirus (MERS-CoV)—Update. Disease Outbreak News. 4 October 2013. Available online: https://www.who.int/csr/don/2013_10_04/en/index.html (accessed on 29 November 2020).
- Calnan, M.; Gadsby, E.W.; Konde, M.K.; Diallo, A.; Rossman, J.S. The response to and impact of the ebola epidemic: Towards an agenda for interdisciplinary research. Int. J. Health Policy Manag. 2018, 7, 402–411. [Google Scholar] [CrossRef]
- Guo, Y.-R.; Cao, Q.-D.; Hong, Z.-S.; Tan, Y.-Y.; Chen, S.-D.; Jin, H.-J.; Tan, K.-S.; Wang, D.-Y.; Yan, Y. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak: An update on the status. Mil. Med. Res. 2020, 7, 1–10. [Google Scholar] [CrossRef]
- Li, Q.; Guan, X.; Wu, P.; Wang, X.; Zhou, L.; Tong, Y.; Ren, R.; Leung, K.S.M.; Lau, E.H.Y.; Wong, J.Y.; et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N. Engl. J. Med. 2020, 382, 1199–1207. [Google Scholar] [CrossRef]
- World Health Organization (WHO). WHO Coronavirus Disease (COVID-19) Dashboard. Available online: https://covid19.who.int/ (accessed on 10 December 2020).
- Klein, S.L.; Flanagan, K.L. Sex differences in immune responses. Nat. Rev. Immunol. 2016, 16, 626–638. [Google Scholar] [CrossRef]
- Chen, J.; Subbarao, K. The immunobiology of SARS. Annu. Rev. Immunol. 2007, 25, 443–472. [Google Scholar] [CrossRef] [PubMed]
- Karlberg, J.; Chong, D.S.Y.; Lai, W.Y.Y. Do men have a higher case fatality rate of severe acute respiratory syndrome than women do? Am. J. Epidemiol. 2004, 159, 229–231. [Google Scholar] [CrossRef] [PubMed]
- Alghamdi, I.G.; Hussain, I.I.; Almalki, S.S.; Alghamdi, M.S.; Alghamdi, M.M.; El-Sheemy, M.A. The pattern of Middle East respiratory syndrome coronavirus in Saudi Arabia: A descriptive epidemiological analysis of data from the Saudi Ministry of Health. Int. J. Gen. Med. 2014, 7, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Altamimi, A.; Abu-Saris, R.; El-Metwally, A.; Alaifan, T.; Alamri, A. Demographic variation of MERS-CoV infection among suspected and confirmed cases: An epidemiological analysis of laboratory-based data from Riyadh Regional Laboratory. Biomed. Res. Int. 2020, 2020, 9629747. [Google Scholar] [CrossRef]
- Meleis, A.; Caglia, J.; Langer, A. Women and health: Women’s dual roles as both recipients and providers of healthcare. J. Womens Health 2016, 25, 329–331. [Google Scholar] [CrossRef]
- Wise, J. COVID-19: Low skilled men have highest death rate of working age adults. BMJ 2020, 369, m1906. [Google Scholar] [CrossRef]
- Oksuzyan, A.; Juel, K.; Vaupel, J.W.; Christensen, K. Men: Good health and high mortality. Sex differences in health and aging. Aging Clin. Exp. Res. 2008, 20, 91–102. [Google Scholar] [CrossRef]
- Klein, S.L.; Passaretti, C.; Anker, M.; Olukoya, P.; Pekosz, A. The impact of sex, gender and pregnancy on 2009 H1N1 disease. Biol. Sex Differ. 2010, 1, 5. [Google Scholar] [CrossRef]
- Eshima, N.; Tokumara, O.; Hara, S.; Bacal, K.; Korematsu, S.; Tabata, M.; Karukaya, S.; Yasui, Y.; Okabe, N.; Matsuishi, T. Sex- and age-related differences in morbidity rates of 2009 ppandemic influenza A H1N1 virus of swine origin in Japan. PLoS ONE 2011, 6, e19409. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Sex, Gender and Influenza. Available online: https://apps.who.int/iris/handle/10665/44401 (accessed on 29 November 2020).
- Ojanunga, D.N.; Gilbert, C. Women’s access to healthcare in developing countries. Social. Sci. Med. 1992, 35, 613–617. [Google Scholar] [CrossRef]
- Kamate, S.K.; Agarwal, A.; Chaudhary, H.; Singh, K.; Mishra, P.; Asawa, K. Public knowledge, attitude and behavioural changes in an Indian population during the influenza A (H1N1) outbreak. J. Infect. Dev. Ctries. 2010, 4, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Zarychanski, R.; Stuart, T.L.; Kumar, A.; Doucette, S.; Elliott, L.; Kettner, J.; Plummer, F. Correlates of severe disease in patients with 2009 pandemic influenza (H1N1) virus infection. CMAJ 2010, 182, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Quinn, S.C.; Kumar, S.; Freimuth, V.S.; Musa, D.; Casteneda-Angarita, N.; Kidwell, K. Racial disparities in exposure, susceptibility, and access to healthcare in the US H1N1 influenza pandemic. Am. J. Public Health 2011, 101, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Agency for Haelthcare Research and Quality 2007 National Healthcare Quality and Disparities Report. Available online: https://www.arhq.gov/qual/qrdr07.htm (accessed on 1 December 2020).
- Bower, H.; Johnson, S.; Bangura, M.S.; Kamara, A.J.; Kamara, O.; Mansaray, S.H.; Sesay, D.; Turay, C.; Checchi, F.; Glynn, J.R. Exposure-specific and age-specific attack rates for ebola virus disease in ebola-affected households, Sierra Leone. Emerg. Infect. Dis. 2016, 22, 1403–1411. [Google Scholar] [CrossRef]
- Nkangu, M.N.; Olatunde, O.A.; Yaya, S. The perspective of gender on ebola virus using a risk management and population health framework: A scoping review. Infect. Dis. Poverty 2017, 6, 135. [Google Scholar] [CrossRef]
- United Nations (UN). Gender Inequality Index, UN Dev. Programme. Available online: https://hdr.undp.org/en/content/table-4-gender-inequality-index (accessed on 1 December 2020).
- Human Rights Watch (HRW). West Africa: Respect Rights in Ebola Response. Available online: https://www.hrw.org/news/2014/09/15/west-africa-respect-rights-ebola-response (accessed on 1 December 2020).
- World Health Organization (WHO). Addressing Sex and Gender in Epidemic-Prone Infectious Diseases. 2007. Available online: https://www.who.int/csr/resources/publications/SexGenderInfectDis.pdf (accessed on 1 December 2020).
- British Broadcasting Corporation (BBC). “The Number of Deaths Could Have Been Avoided. Better Engagement of Women at the Grassroots Level Who Are Midwives, Who Are Death Attendants, Who Are Traditional Healers, Who Were Not Aware of All the Details. Just Respecting Their Leadership and Engagement Could Have Decreased the Number of People Who Died.” Phumzile Mlambo-Ngcuka, Exec. Dir., U.N. Women, Remarks at the BBC World Debate in Accra, Ghana: Ebola—What Next? 25 March 2015. Available online: http://www.bbc.co.uk/programmes/pozmcz5g (accessed on 1 December 2020).
- Singh, G.K.; Rodriguez-Lainz, A.; Kogan, M.D. Immigrant health inequalities in the United States: Use of eight major national data systems. Sci. World J. 2013, 2013, 512313. [Google Scholar] [CrossRef]
- Azuine, R.E.; Ekejiuba, S.E.; Singh, G.K.; Azuine, M.A. Ebola Virus Disease Epidemic: What Can the World Learn and Not Learn from West Africa? Int. J. MCH AIDS 2015, 3, 1–6. [Google Scholar] [CrossRef]
- Singh, G.K.; Lin, S.C. Marked ethnic, nativity, and socioeconomic disparities in disability and health insurance among US children and adults: The 2008–2010 American community survey. Biomed. Res. Int. 2013, 2013, 627412. [Google Scholar] [CrossRef]
- Girum, T.; Wasie, A.; Lentiro, K.; Muktar, E.; Shumbej, T.; Difer, M.; Shegaze, M.; Worku, A. Gender disparity in epidemiological trend of HIV/AIDS infection and treatment in Ethiopia. Arch. Public Health 2018, 76, 51. [Google Scholar] [CrossRef]
- Magadi, M.A. Understanding the gender disparity in HIV infection across countries in sub-Saharan Africa: Evidence from the demographic and health surveys. Sociol. Health Illn. 2011, 33, 522–539. [Google Scholar] [CrossRef]
- Richardson, E.T.; Collins, S.E.; Kung, T.; Jones, J.H.; Tram, K.H.; Boggiano, V.L.; Bekker, L.-G.; Zolopa, A.R. Gender inequality and HIV transmission: A global analysis. J. Int. AIDS Soc. 2014, 17, 19035. [Google Scholar] [CrossRef] [PubMed]
- WHO (World Health Organization). AIDS Epidemic Update: December 2006. Available online: https://www.who.int/hiv/pub/epidemiology/epiupdate2006/en/ (accessed on 25 September 2020).
- Haley, D.F.; Justman, J.E. The HIV epidemic among women in the United States: A persistent puzzle. J. Women’s Health 2013, 22, 715–717. [Google Scholar] [CrossRef] [PubMed]
- Anugwom, E.; Anugwom, K. Socio-cultural factors in the access of women to HIV/AIDS prevention and treatment services in South-southern Nigeria. Iran. J. Public Health 2016, 45, 754–760. [Google Scholar] [PubMed]
- Heslin, K.C.; Andersen, R.M.; Ettner, S.L.; Cunningham, W.E. Racial and ethnic disparities in access to physicians with HIV-related expertise. J. Gen. Intern. Med. 2005, 20, 283–289. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Diagnoses of HIV Infection in the United States and Dependent Areas. 2018. Available online: www.cdc.gov/hiv/library/reports/hiv-surveillance/vol-31/index.html (accessed on 1 December 2020).
- Liu, Y.; Mao, B.; Liang, S.; Yang, J.-W.; Lu, H.-W.; Chai, Y.-H.; Wang, L.; Zhang, L.; Li, Q.-H.; Zhao, L.; et al. Association between age and clinical characteristics and outcomes of COVID-19. Eur. Respir. J. 2020, 55, 2001112. [Google Scholar] [CrossRef]
- Yang, X.; Yu, Y.; Xu, J.; Shu, H.; Xia, J.; Liu, H.; Wu, Y.; Zhang, L.; Yu, Z.; Fang, M.; et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: A single-centered, retrospective, observational study. Lancet Respir. Med. 2020, 8, 475–481. [Google Scholar] [CrossRef]
- Klein, S.L.; Dhakal, S.; Ursin, R.L.; Deshpande, S.; Sandberg, K.; Mauvais-Jarvis, F. Biological sex impacts COVID-19 outcomes. PLoS Pathog. 2020, 16, e1008570. [Google Scholar] [CrossRef]
- Gebhard, C.; Regitz-Zagrosek, V.; Neuhauser, H.K.; Morgan, R.; Klein, S.L. Impact of sex and gender on COVID-19 outcomes in Europe. Biol. Sex Differ. 2020, 11, 29. [Google Scholar] [CrossRef]
- Chen, T.; Wu, D.; Chen, H.; Yan, W.; Yang, D.; Chen, G.; Ma, K.; Xu, D.; Yu, H.; Wang, H.; et al. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: Retrospective study. BMJ 2020, 368, m1091. [Google Scholar] [CrossRef]
- Dudley, J.P.; Lee, N.T. Disparities in age-specific morbidity and mortality from SARS-CoV-2 in China and the Republic of Korea. Clin. Infect. Dis. 2020, 71, 863–865. [Google Scholar] [CrossRef]
- Borghesi, A.; Zigliani, A.; Masciullo, R.; Golemi, S.; Maculotti, P.; Farina, D.; Maroldi, R. Radiographic severity index in COVID-19 pneumonia: Relationship to age and sex in 783 Italian patients. Radiol. Med. 2020, 125, 461–464. [Google Scholar] [CrossRef] [PubMed]
- Gemmati, D.; Bramanti, B.; Serino, M.L.; Secchiero, P.; Zauli, G.; Tisato, V. COVID-19 and Individual Genetic susceptibility/receptivity: Role of ACE1/ACE2 genes, immunity, inflammation and coagulation. might the double X-chromosome in females be protective against SARS-CoV-2 compared to the single X-chromosome in males? Int. J. Mol. Sci. 2020, 21, 3474. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.-M.; Bai, P.; He, W.; Wu, F.; Liu, X.-F.; Han, D.-M.; Liu, S.; Yang, J.-K. Gender differences in patients with COVID-19: Focus on severity and mortality. Front. Public Health 2020, 8, 152. [Google Scholar] [CrossRef] [PubMed]
- Meng, Y.; Wu, P.; Lu, W.; Liu, K.; Ma, K.; Huang, L.; Cai, J.; Zhang, H.; Qin, Y.; Sun, H.; et al. Sex-specific clinical characteristics and prognosis of coronavirus disease-19 infection in Wuhan, China: A retrospective study of 168 severe patients. PLoS Pathog. 2020, 16, e1008520. [Google Scholar] [CrossRef]
- Lechien, J.R.; Chiesa-Estomba, C.M.; Laethem, Y.V.; Cabaraux, P.; Mat, Q.; Huet, K.; Pizak, J.; Horoi, M.; Hans, S.; Barillari, M.R.; et al. Clinical and epidemiological characteristics of 1420 European patients with mild-to-moderate coronavirus disease 2019. J. Int. Med. 2020, 288, 335–344. [Google Scholar] [CrossRef]
- Cohen, F.S. How viruses invade cells. Biophys. J. 2016, 110, 1028–1032. [Google Scholar] [CrossRef]
- Cohen, F.S.; Melikyan, G.B. The energetic of membrane fusion from binding, through hemifusion, pore formation, and pore enlargement. J. Membr. Biol. 2004, 199, 1–14. [Google Scholar] [CrossRef]
- Rogers, G.N.; Paulson, J.C. Receptor determinants of human and animal influenza virus isolates: Differences in receptor specificity of the H3 hemagglutinin based on species of origin. Virology 1983, 127, 361–373. [Google Scholar] [CrossRef]
- Gamblin, S.J.; Skehel, J.J. Influenza hemagglutinin and neuraminidase membrane glycoproteins. J. Biol. Chem. 2010, 285, 28403–28409. [Google Scholar] [CrossRef]
- Roy, A.M.; Parker, J.S.; Parrish, C.R.; Whittaker, G.R. Early stages of influenza virus entry into Mv-1 lung cells: Involvement of dynamin. Virology 2000, 267, 17–28. [Google Scholar] [CrossRef]
- Rust, M.J.; Lakadamyali, M.; Zhang, F.; Zhuang, X. Assembly of endocytic machinery around individual influenza viruses during viral entry. Nat. Struct. Mol. Biol. 2004, 11, 567–573. [Google Scholar] [CrossRef] [PubMed]
- Martin, K.; Helenius, A. Nuclear transport of influenza virus ribonucleoproteins: The viral matrix protein (M1) promotes export and inhibits import. Cell 1991, 67, 117–130. [Google Scholar] [CrossRef]
- Choi, J.H.; Croyle, M.A. Emerging targets and novel approaches to ebola virus prophylaxis and treatment. BioDrugs 2013, 27, 565–583. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, C.P.; Lasala, F.; Carrillo, J.; Muniz, O.; Corbi, A.L.; Delgado, R. C-type lectins DC-SIGN and L-SIGN mediate cellular entry by ebola virus in cis and in trans. J. Virol. 2002, 76, 6841–6844. [Google Scholar] [CrossRef]
- Nanbo, A.; Imai, M.; Watanabe, S.; Noda, T.; Takhashi, K.; Neumann, G.; Halfmann, P.; Kawaoka, Y. Ebolavirus is internalized into host cells via macropinocytosis in a viral glycoprotein-dependent manner. PLoS Pathog. 2010, 23, e1001121. [Google Scholar] [CrossRef]
- Moller-Tank, S.; Maury, W. Phosphatidylserine receptors: Enhancers of enveloped virus entry and infection. Virology 2014, 468, 565–580. [Google Scholar] [CrossRef]
- Lee, J.; Gregory, S.M.; Nelson, E.A.; White, J.M.; Tamm, L.K. The roles of histidines and charged residues as potential triggers of a conformational change in the fusion loop of ebola virus glycoprotein. PLoS ONE 2016, 11, e0152527. [Google Scholar] [CrossRef] [PubMed]
- Furuta, Y.; Eriksson, K.; Svennerholm, B.; Fredman, P.; Horal, P.; Jeansson, S.; Vahlne, A.; Holmgren, J.; Czerkinsky, C. Infection of vaginal and colonic epithelial cells by the human immunodeficiency virus type 1 is neutralized by antibodies raised against conserved epitopes in the envelope glycoprotein gp 120. Proc. Natl. Acad. Sci. USA 1994, 91, 12559–12563. [Google Scholar] [CrossRef] [PubMed]
- Bomsel, M. Transcytosis of infectious human immunodeficiency virus across a tight human epithelial cell line barrier. Nat. Med. 1997, 3, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Yeaman, G.R.; Asin, S.; Weldon, S.; Demian, D.J.; Collins, J.E.; Gonzalez, J.L.; Wira, C.R.; Fanger, M.W.; Howell, A.L. Chemokine receptor expression in the human ectocervix: Implications for infection by the human immunodeficiency virus-type 1. Immunology 2004, 113, 524–533. [Google Scholar] [CrossRef]
- Hladik, F.; McElrath, M.J. Setting the stage- HIV host invasion. Nat. Rev. Immunol. 2008, 8, 447–457. [Google Scholar] [CrossRef]
- Suresh, P.; Wanchu, A. Chemokines and chemokine receptors in HIV infection: Role in pathogenesis and therapeutics. J. Postgrad. Med. 2006, 52, 210–217. [Google Scholar] [PubMed]
- Willey, J.M.; Sherwood, L.M.; Woolverton, C.J. Human diseases caused by viruses and prions. In Prescott’s Microbiology, 10th ed.; Willey, J.M., Sherwood, L.M., Woolverton, C.J., Eds.; Mc Graw Hill: New York, NY, USA, 2017; pp. 827–858. [Google Scholar]
- Cheng, P.K.C.; Wong, D.A.; Tong, L.K.L.; Ip, S.-M.; Lo, A.C.T.; Lau, C.-S.; Yeung, E.Y.H.; Lim, W.W.L. Viral shedding patterns of coronavirus in patients with probable severe acute respiratory syndrome. Lancet 2004, 363, 1699–1700. [Google Scholar] [CrossRef]
- Li, W.; Moore, M.J.; Vasilieva, N.; Sui, J.; Wong, S.K.; Berne, M.A.; Somasundaran, M.; Sullivan, J.L.; Luzuriaga, K.; Greenough, T.C.; et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature 2003, 426, 250–454. [Google Scholar] [CrossRef] [PubMed]
- Kuba, K.; Imai, Y.; Ohto-Nakanishi, T.; Penninger, J.M. Trilogy of ACE2: A peptidase in the rennin-angiotensin system, a SARS receptor, and a partner for the amino acid transporters. Pharmacol. Ther. 2010, 128, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Raj, V.S.; Mou, H.; Smits, S.L.; Dekkers, D.H.W.; Muller, M.A.; Dijkman, R.; Muth, D.; Demmers, J.A.A.; Zaki, A.; Fouchier, R.A.M.; et al. Dipeptidyl peptidase 4 is a functional receptor for the emerging human coronavirus-EMC. Nature 2013, 495, 251–254. [Google Scholar] [CrossRef] [PubMed]
- Bosch, B.J.; van der Zee, R.; de Haan, C.A.M.; Rottier, P.J.M. The coronavirus spike protein is a class I virus fusion protein: Structural and functional characterization of the fusion core complex. J. Virol. 2003, 77, 8801–8811. [Google Scholar] [CrossRef]
- Fehr, A.R.; Perlman, S. Coronaviruses: An overview of their replication and pathogenesis. Coronaviruses 2015, 1282, 1–23. [Google Scholar]
- Rabi, F.A.; Al Zoubi, M.S.; Kasasbeh, G.A.; Salameh, D.M.; Al-Nasser, A.D. SARS-CoV-2 and coronavirus disease 2019: What we know so far. Pathogens 2020, 9, 231. [Google Scholar] [CrossRef]
- Walls, A.C.; Park, Y.-J.; Tortorici, M.A.; Wall, A.; McGuire, A.T.; Veesler, D. Structure, function and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell 2020, 181, 281–292. [Google Scholar] [CrossRef]
- Simmons, G.; Zmora, P.; Gierer, S.; Heurich, A.; Pohlmann, S. Proteolytic activation of the SARS-coronavirus spike protein: Cutting enzymes at the cutting edge of antiviral research. Antiviral. Res. 2013, 100, 605–614. [Google Scholar] [CrossRef] [PubMed]
- Sungnak, W.; Huang, N.; Becavin, C.; Berg, M. HCA Lung Biological Network. SARS-CoV-2 entry genes are most highly expressed in nasal goblet and ciliated cells within human airways. arXiv 2020, arXiv:2003.06122. [Google Scholar]
- Hou, Y.J.; Okuda, K.; Edwards, C.E.; Martinez, D.R.; Asakura, T.; Dinnon, K.H.; Kato, T.; Lee, R.E.; Yount, B.L.; Mascenik, T.M.; et al. SARS-CoV-2 reverse genetics reveals a variable infection gradient in the respiratory tract. Cell 2020, 182, 429–446. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Geng, X.; Tan, Y.; Li, Q.; Xu, C.; Xu, J.; Hao, L.; Zeng, Z.; Luo, X.; Liu, F.; et al. New understanding of the damage of SARS-CoV-2 infection outside the respiratory system. Biomed. Pharmacother. 2020, 127, 110195. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, M.; Saito, J.; Zhao, H.; Sakamoto, A.; Hirota, K.; Ma, D. Inflammation triggered by SARS-CoV-2 and ACE2 augment deives multiple organ failure of severe COVID-19: Molecular mechanisms and implications. Inflammation 2020, 43, 1–22. [Google Scholar]
- Astuti, I. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): An overview of viral structure and host response. Diabetes Metab. Syndr. 2020, 14, 407–412. [Google Scholar] [CrossRef]
- Wang, Q.; Wu, J.; Wang, H.; Gao, Y.; Liu, Q.; Mu, A.; Ji, W.; Yan, L.; Zhu, Y.; Zhu, C.; et al. Structural basis for RNA replication by the SARS-CoV-2 polymerase. Cell 2020, 182, 417–428. [Google Scholar] [CrossRef]
- Renu, K.; Prasanna, P.L.; Gopalakrishnan, A.V. Coronaviruses pathogenesis, comorbidities and multiple-organ damage—A review. Life Sci. 2020, 255, 117839. [Google Scholar] [CrossRef]
- Zaragoulidis, P.; Kouliatsis, G.; Papanas, N.; Spyratos, D.; Constantinidis, T.C.; Kouroumichakis, I.; Steiropoulos, P.; Mabroudi, M.; Matthaios, D.; Kerenidi, T.; et al. Long-term respiratory follow-up of H1N1 infection. Virol. J. 2011, 8, 319. [Google Scholar] [CrossRef]
- Wolf, T.; Kann, G.; Becker, S.; Stephan, C.; Brodt, H.-R.; Leuw, P.D.; Grunewald, T.; Vogl, T.; Kempf, V.A.J.; Keppler, O.T.; et al. Severe ebola virus disease with vascular leakage and multiorgan failure: Treatment of a patient in intensive care. Lancet 2015, 385, 1428–1435. [Google Scholar] [CrossRef]
- Martines, R.B.; Ng, D.L.; Greer, P.W.; Rollin, P.E.; Zaki, S.R. Tissue and cellular tropism, pathology and pathogenesis of ebola and Marburg viruses. J. Pathol. 2015, 235, 153–174. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, M.E.; Kunisaki, K.M.; Morris, A. Pulmonary disease in HIV-infected adults in the era of antiretroviral therapy. AIDS 2018, 32, 277–292. [Google Scholar] [CrossRef]
- Gu, J.; Gong, E.; Zhang, B.; Zheng, J.; Gao, Z.; Zhong, Y.; Zou, W.; Zhan, J.; Wang, S.; Xie, Z.; et al. Multiple organ infection and the pathogenesis of SARS. J. Exp. Med. 2005, 202, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Ramadan, N.; Shaib, H. Middle east respiratory syndrome coronavirus (MERS-CoV): A review. Germs 2019, 9, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, S.K.; Satapathy, A.; Naidu, M.M.; Mukhopadhyay, S.; Sharma, S.; Barton, L.M.; Stroberg, E.; Duval, E.J.; Pradhan, D.; Tzankov, A.; et al. Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) and coronavirus disease 19 (COVID-19)- anatomic pathology perspective on current knowledge. Diagn. Pathol. 2020, 15, 103. [Google Scholar] [CrossRef]
- Ciceri, F.; Beretta, L.; Scandroglio, A.M.; Colombo, S.; Landoni, G.; Ruggeri, A.; Peccatori, J.; D’Angelo, A.; De Cobelli, F.; Rovere-Querini, P.; et al. Microvascular COVID-19 lung vessels obstructive thromboinflammatory syndrome (MicroCLOTS): An atypical acute respiratory distress syndrome working hypothesis. Crit. Care Resusc. 2020, 22, 95–97. [Google Scholar]
- Gupta, M.D.; Girish, M.P.; Yadav, G.; Shankar, A.; Yadav, R. Coronavirus disease 2019 and the cardiovascular system: Impacts and implications. Indian Heart J. 2020, 72, 1–6. [Google Scholar] [CrossRef]
- Richardson, P.; McKenna, W.; Bristow, M.; Maisch, B.; Mautner, B.; O’Connell, J.; Olsen, E.; Thiene, G.; Goodwin, J.; Gyarfas, I.; et al. Reprt of the 1995 World Health Organization/International Society and Federation of Cardiology Task Force on the definition and classification of cardiomyopathies. Circulation 1996, 93, 841. [Google Scholar]
- Onitsuka, H.; Imamura, T.; Miyamoto, N.; Shibata, Y.; Kashiwagi, T.; Ayabe, T.; Kawagoe, J.; Matsuda, J.; Ishikawa, T.; Unoki, T.; et al. Clinical manifestations of influenza a myocarditis during the influenza epidemic of winter 1998–1999. J. Cardiol. 2001, 37, 315–323. [Google Scholar]
- Golabchi, A.; Sarrafzadegan, N. What every cardiologist should know about H1N1? ARYA Atheroscler. 2010, 6, 118–121. [Google Scholar]
- Kortepeter, M.G.; Lawler, J.V.; Honko, A.; Bray, M.; Johnson, J.C.; Purcell, B.K.; Olinger, G.G.; Rivard, R.; Hepburn, M.J.; Hensley, L.E. Real-time monitoring of cardiovascular function in rhesus macaques infected with Zaire ebolavirus. J. Infect. Dis. 2011, 204, S1000–S1010. [Google Scholar] [CrossRef] [PubMed]
- Barnes, R.P.; Lacson, J.C.A.; Bahrami, H. HIV infection and risk of cardiovascular diseases beyond coronary artery disease. Curr. Atheroscler. Rep. 2017, 19, 20. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.-M.; Wong, R.S.-M.; Wu, E.B.; Kong, S.-L.; Wong, J.; Yip, G.W.-K.; Soo, Y.O.Y.; Chiu, M.L.S.; Chan, Y.-S.; Hui, D.; et al. Cardiovascular complications of severe acute respiratory syndrome. Postgrad. Med. J. 2006, 82, 140–144. [Google Scholar] [CrossRef]
- Assiri, A.; Al-Tawfiq, J.A.; Al-Rabeeah, A.A.; Al-Rabiah, F.A.; Al-Hajjar, S.; Al-Barrak, A.; Flemban, H.; Al-Nassir, W.N.; Balkhy, H.H.; Al-Hakeem, R.F.; et al. Epidemiological, demographic, and clinical characteristics of 47 cases of Middle East respiratory syndrome coronavirus disease from Saudi Arabia: A descriptive study. Lancet Infect. Dis. 2013, 13, 752–761. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Landi, K.K.; Coleman, A.T. Sudden death in toddlers caused by influenza B infection: A report of two cases and a review of the literature. J. Forensic Sci. 2008, 53, 213–215. [Google Scholar] [CrossRef]
- Adalja, A.A.; Henderson, D.A. Original antigenic sin and pandemic (H1N1) 2009. Emerg. Infect. Dis. 2010, 16, 1028–1029. [Google Scholar] [CrossRef]
- Reisler, R.B.; Zeng, X.; Schellhase, C.W.; Bearss, J.J.; Warren, T.K.; Trefry, J.C.; Christopher, G.W.; Kortepeter, M.G.; Bavari, S.; Cardile, A.P. Ebola virus causes intestinal tract architecrural disruption and bacterial invasion in non-human primates. Viruses 2018, 10, 513. [Google Scholar] [CrossRef]
- Sestak, K. Chronic diarrhea and AIDS: Insights into studies with non-human primates. Curr. HIV Res. 2005, 3, 199–205. [Google Scholar] [CrossRef]
- Zhang, J.-Z. Severe acute respiratory syndrome and its lesions in digestive system. World J. Gastroenterol. 2003, 9, 1135–1138. [Google Scholar] [CrossRef]
- Chan, J.F.W.; Lau, S.K.P.; To, K.K.W.; Cheng, V.C.C.; Woo, P.C.Y.; Yuen, K.-Y. Middle East respiratory syndrome coronavirus: Another zoonotic betacoronavirus causing SARS-like disease. Clin. Microbiol. Rev. 2015, 28, 465–522. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Li, C.; Zhao, G.; Chu, H.; Wang, D.; Yan, H.H.-N.; Poon, V.K.-M.; Wen, L.; Wong, B.H.-Y.; Zhao, X.; et al. Human intestinal tract serves as an alternative infection route for Middle East respiratory syndrome coronavirus. Sci. Adv. 2017, 15, eaao4966. [Google Scholar] [CrossRef]
- Hajifathalian, K.; Mahadev, S.; Schwartz, R.E.; Shah, S.; Sampath, K.; Schnoll-Sussman, F.; Brown, R.S., Jr.; Carr-Locke, D.; Cohen, D.E.; Sharaiha, R.Z. SARS-CoV-2-infection (coronavirus disease 2019) for the gastrointestinal consultant. World J. Gastroenterol. 2020, 26, 1546–1553. [Google Scholar] [CrossRef] [PubMed]
- Sulkava, R.; Rissanen, A.; Pyhala, R. Post-influenzal encephalitis during the influenza A outbreak in 1979/1980. J. Neurol. Neurosurg. Psychiatry 1981, 44, 161–163. [Google Scholar] [CrossRef]
- Asadi-Pooya, A.A.; Yaghoubi, E.; Niksereshi, A.; Moghadami, M.; Honarvar, B. The neurological manifestations of H1N1 influenza infection; diagnostic challenges and recommendations. Iran J. Med. Sci. 2011, 36, 36–39. [Google Scholar]
- West, T.E.; Arnim, A.V.S.A.-V. Clinical presentation and management of severe ebola virus disease. Ann. Am. Thorac. Soc. 2014, 11, 1341–1350. [Google Scholar] [CrossRef] [PubMed]
- Billioux, B.J.; Smith, B.; Nath, A. Neurological complications of ebola virus infection. Neurotherapeutics 2016, 13, 461–470. [Google Scholar] [CrossRef]
- Ellis, R.J.; Calero, P.; Stockin, M.D. HIV infection and the central nervous system: A primer. Neuropsychol. Rev. 2009, 19, 144–151. [Google Scholar] [CrossRef]
- Lau, K.-K.; Yu, W.-C.; Chu, C.-M.; Lau, S.-T.; Sheng, B.; Yuen, K.-Y. Possible central nervous system infection by SARS coronavirus. Emerg. Infect. Dis. 2004, 10, 342–344. [Google Scholar] [CrossRef]
- Xu, J.; Zhong, S.; Liu, J.; Li, L.; Li, Y.; Wu, X.; Li, Z.; Deng, P.; Zhang, J.; Zhong, N.; et al. Detection of severe acute respiratory syndrome coronavirus in the brain: Potential role of the chemokine mig in pathogenesis. Clin. Infect. Dis. 2005, 41, 1089–1096. [Google Scholar] [CrossRef]
- Kim, J.E.; Heo, J.H.; Kim, H.O.; Song, S.H.; Park, S.S.; Park, T.H.; Ahn, J.Y.; Kim, M.K.; Choi, J.P. Neurological complications during treatment of Middle East respiratory syndrome. J. Clin. Neurol. 2017, 13, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Moriguchi, T.; Harii, N.; Goto, J.; Harada, D.; Sugawara, H.; Takamino, J.; Ueno, M.; Sakata, H.; Kondo, K.; Myose, N.; et al. A first case of meningitis/ encephalitis associated with SARS-coronavirus-2. Int. J. Infect. Dis. 2020, 94, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Mao, L.; Jin, H.; Wang, M.; HU, Y.; Chen, S.; He, Q.; Chang, J.; Hong, C.; Zhou, Y.; Wang, D.; et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020, 77, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T. Renal complications of seasonal and pandemic influenza A virus infections. Eur. J. Pediatr. 2013, 172, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Prasad, N.; Novak, J.E.; Patel, M.R. Kidney diseases associated with parvovirus B19, hanta, ebola, and dengue virus infection: A brief review. Adv. Chronic Kidney Dis. 2019, 26, 207–219. [Google Scholar] [CrossRef] [PubMed]
- Wyatt, C.M. Kidney diseases and HIV infection. Top Antivir. Med. 2017, 25, 13–16. [Google Scholar]
- Huang, J.-W.; Chen, K.-Y.; Tsai, H.-B.; Wu, V.-C.; Yang, Y.-F.; Wu, M.-S.; Chu, T.-S.; Wu, K.-D. Acute renal failure in patients with severe acute respiratory syndrome. J. Formos. Med. Assoc. 2005, 104, 891–896. [Google Scholar]
- Memish, Z.A.; Zumla, A.I.; Al-Hakeem, R.F.; Al-Rabeeah, A.A.; Stephens, G.M. Family cluster of Middle East respiratory syndrome coronavirus infections. N. Engl. J. Med. 2013, 368, 2487–2494. [Google Scholar] [CrossRef]
- Su, H.; Yang, M.; Wan, C.; Yi, L.-X.; Tang, F.; Zhu, H.-Y.; Yi, F.; Yang, H.-C.; Fogo, A.B.; Nie, X.; et al. Renal histopathological analysis of 26 postmortem findings of patients with COVID-19 in China. Kidney Int. 2020, 98, 219–227. [Google Scholar] [CrossRef]
- Martinez-Rojas, M.A.; Vega-Vega, O.; Bobadilla, N.A. Is the kidney a target of SARS-CoV-2? Am. J. physiol. Renal. Physiol. 2020, 318, F1454–F1462. [Google Scholar] [CrossRef]
- Evenson, D.P.; Jost, L.K.; Corzett, M.; Balhorn, R. Characteristics of human sperm chromatin structure following an episode of influenza and high fever: A case study. J. Androl. 2000, 21, 739–746. [Google Scholar] [PubMed]
- Sergerie, M.; Mieusset, R.; Croute, F.; Daudin, M.; Bujan, L. High risk of temporary alteration of semen parameters after recent acute febrile illness. Fertil. Steril. 2007, 88, e1–e7. [Google Scholar] [CrossRef] [PubMed]
- Shinde, V.; Bridges, C.B.; Uyeki, T.M.; Shu, B.; Balish, A.; Xu, X.; Lindstrom, S.; Gubareva, L.V.; Deyde, V.; Garten, R.J.; et al. Triple-reassortant swine influenza A (H1) in humans in the United States, 2005–2009. N. Engl. J. Med. 2009, 360, 2616–2625. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, S.A.; Jamieson, D.J.; Macfarlane, K.; Cragan, J.D.; Williams, J.; Henderson, Z. Pandemic influenza and pregnant women: Summary of a meeting of experts. Am. J. Public Health 2009, 99, S248–S254. [Google Scholar] [CrossRef] [PubMed]
- Perry, D.L.; Huzella, L.M.; Bernbaum, J.G.; Holbrook, M.R.; Jahrling, P.B.; Hagen, K.R.; Schnell, M.J.; Johnson, R.F. Ebola virus localization in the macaque reproductive tract during acute ebola virus disease. Am. J. Pathol. 2018, 188, 550–558. [Google Scholar] [CrossRef]
- Connolly, B.M.; Steele, K.E.; Davis, K.J.; Geisbert, T.W.; Kell, W.M.; Jaax, N.K.; Jahrling, P.B. Pathogenesis of experimental ebola virus infection in guinea pig. J. Infect. Dis. 1999, 179, S203–S217. [Google Scholar] [CrossRef]
- Muehlenbachs, A.; Vasquez, O.D.L.R.; Bausch, D.G.; Schafer, I.J.; Paddock, C.D.; Nyakio, J.P.; Lame, P.; Bergeron, E.; McCollum, A.M.; Goldsmith, C.S.; et al. Ebola virus disease in pregnancy: Clinical, histopathologic, and immunohistochemical findings. J. Infect. Dis. 2017, 215, 64–69. [Google Scholar] [CrossRef]
- Sissoko, D.; Keita, M.; Diallo, B.; Aliabadi, N.; Fitter, D.L.; Dahl, B.A.; Bore, J.A.; Koundouno, F.R.; Singethan, K.; Meisel, S.; et al. Ebola virus persistence in breast milk after no reported illness: A likely source of virus transmission from mother to child. Clin. Infect. Dis. 2017, 64, 513–516. [Google Scholar] [CrossRef]
- Leruez-Ville, M.; Almeida, M.D.; Tachet, A.; Dulioust, E.; Guibert, J.; Mandelbrot, L.; Salmon, D.; Jouannet, P.; Rouzioux, C. Asssited reproduction in HIV-1 serodifferent couples: The need for viral validation of processed semen. AIDS 2002, 16, 2267–2273. [Google Scholar] [CrossRef]
- Tindall, B.; Forde, S.; Goldstein, D.; Ross, M.W.; Cooper, D.A. Sexual dysfunction in advanced HIV disease. AIDS Care 1994, 6, 105–107. [Google Scholar] [CrossRef]
- Cejtin, H.E.; Kalinowski, A.; Bacchetti, P.; Taylor, R.N.; Watts, D.H.; Kim, S.; Massad, L.S.; Preston-Martin, S.; Anastos, K.; Moxley, M.; et al. Effects of human immunodeficiency virus on protracted amenorrhea and ovarian dysfunction. Obstet. Gynecol. 2006, 108, 1423–1431. [Google Scholar] [CrossRef] [PubMed]
- Paxton, L.A.; Kiwanuka, N.; Nalugoda, F.; Gray, R.; Wawer, M.J. Community based study of treatment seeking among subjects with symptoms of sexually transmitted disease in rural Uganda. BMJ 1998, 317, 1630–1631. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gray, R.H.; Wawer, M.J.; Serwadda, D.; Sewankambo, N.; Li, C.; Wabwire-Mangen, F.; Paxton, L.; Kiwanuka, N.; Kigozi, G.; Konde-Lule, J.; et al. Population-based study of fertility in women with HIV-1 infection in Uganda. Lancet 1998, 351, 98–103. [Google Scholar] [CrossRef]
- Payne, D.C.; Iblam, I.; Alqasrawi, S.; Nsour, M.A.; Rha, B.; Tohme, R.A.; Abedi, G.R.; Farag, N.H.; Haddadin, A.; Sanhouri, T.A.; et al. Stillbirth during infection with Middle East respiratory syndrome coronavirus. J. Infect. Dis. 2014, 209, 1870–1872. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Middle East Respiratory Syndrome Coronavirus (MERS-CoV)—Update. Disease Outbreak News. 2 December 2013. Available online: https://www.who.int/csr/don/2013_12_02/en/ (accessed on 21 August 2020).
- Xu, J.; Qi, L.; Chi, X.; Yang, J.; Wei, X.; Gong, E.; Peh, S.; Gu, J. Orchitis: A complication of severe acute respiratory syndrome (SARS). Biol. Reprod. 2006, 74, 410–416. [Google Scholar] [CrossRef]
- Paoli, D.; Pallotti, F.; Colangelo, S.; Basilico, F.; Mazzuti, L.; Turriziani, O.; Antonelli, G.; Lenzi, A.; Lombardo, F. Study of SARS-CoV-2 in semen and urine samples of a volunteer with positive naso-pharyngeal swab. J. Endocrinol. Invest. 2020, 43, 1819–1822. [Google Scholar] [CrossRef]
- Holtmann, N.; Edimiris, P.; Andree, M.; Doehmen, C.; Baston-Buest, D.; Adams, O.; Kruessel, J.-S.; Bielfeld, A.P. Assessment of SARS-CoV-2 in human semen- a cohort study. Fertil. Steril. 2020, 114, 233–238. [Google Scholar] [CrossRef]
- Li, D.; Jin, M.; Bao, P.; Zhao, W.; Zhang, S. Clinical characteristics and results of semen tests among men with coronavirus disease 2019. JAMA Netw. Open 2020, 3, e208292. [Google Scholar] [CrossRef]
- Ma, L.; Xie, W.; Li, D.; Shi, L.; Mao, Y.; Xiong, Y.; Zhang, Y.; Zhang, M. Effect of SARS-CoV-2 infection upon male gonadal function: A single center-based study. MedRxiv 2020. [Google Scholar] [CrossRef]
- Schroeder, M.; Tuku, B.; Jarczak, D.; Nierhaus, A.; Bai, T.; Jacobsen, H.; Zickler, M.; Mueller, Z.; Bertram-Stanelle, S.; Meinhardt, A.; et al. The majority of male patients with COVID-19 present low testosterone levels on admission to intensive care in Hamburg, Germany: A retrospective cohort study. medRxiv 2020. [Google Scholar] [CrossRef]
- Dutta, S.; Sengupta, P. SARS-CoV-2 and male infertility: Possible multifaceted pathology. Reprod. Sci. 2020, 7, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Blendon, R.J.; Benson, J.M.; DesRoches, C.M.; Raleigh, E.; Taylor-Clark, K. The public’s response to severe acute respiratory syndrome in Toronto and the United States. Clin. Infect. Dis. 2004, 38, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Wheatland, R. Molecular mimicry of ACTH in SARS–implications for corticosteroid treatment and prophylaxis. Med. Hypotheses 2004, 63, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Dutta, S.; Sengupta, P. SARS-CoV-2 infection, oxidative stress and male reproductive hormones: Can testicular-adrenal crosstalks be ruled out? J. Basic Clin. Physiol. Pharmacol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Bouayed, J.; Rammal, H.; Soulimani, R. Oxidative stress and anxiety. Oxid. Med. Cell Longev. 2009, 2, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, P.; Dutta, S. Does SARS-CoV-2 infection cause sperm DNA fragmentation? Possible link with oxidative stress. Eur. J. Contracept. Reprod. Health Care 2020, 10, 1787376. [Google Scholar] [CrossRef] [PubMed]
- Nillni, Y.I.; Wesselink, A.K.; Hatch, E.E.; Mikkelsen, E.M.; Gradus, J.L.; Rothman, K.J.; Wise, L.A. Mental health, psychotropic medication use, and menstrual cycle characteristics. Clin. Epidemiol. 2018, 10, 1073–1082. [Google Scholar] [CrossRef]
- Segars, J.; Katler, Q.; McQueen, D.B.; Kotlyar, A.; Glenn, T.; Knight, Z.; Feinberg, E.C.; Taylor, H.S.; Toner, J.P.; Kawwass, J.F. Prior and novel coronaviruses, coronavirus disease 2019 (COVID-19), and human reproduction: What is known? Fertil. Steril. 2020, 113, 1140–1149. [Google Scholar] [CrossRef]
- Li, R.; Yin, T.; Fang, F.; Li, Q.; Chen, J.; Wang, Y.; Hao, Y.; Wu, G.; Duan, P.; Wang, Y.; et al. Potential risk of SARS-CoV-2 infection on reproductive health. Reprod. Biomed. Online 2020, 41, 89–95. [Google Scholar] [CrossRef]
- de Souza Silva, G.A.; da Silva, S.P.; da Costa, M.A.S.; da Silva, A.R.; de Vasconcelos Alves, R.R.; Tenorio, F.D.C.A.M.; da Silva Melo, A.R.; de Freitas, C.; de Melo, C.M.L. SARS-CoV, MERS-CoV and SARS-CoV-2 infections in pregnancy and fetal development. J. Gynecol. Obstet. Hum. Reprod. 2020, 49, 101846. [Google Scholar] [CrossRef]
- Berkowitz, K.; LaSala, A. Risk factors associated with the increasing prevalence of pneumonia during pregnancy. Am. J. Obstet. Gynecol. 1990, 163, 981–985. [Google Scholar] [CrossRef]
- Chen, H.; Guo, J.; Wang, C.; Luo, F.; Yu, X.; Zhang, W.; Li, J.; Zhao, D.; Xu, D.; Gong, Q.; et al. Clinical characteristics and intrauterine vertical transmission potential of COVID-19 infection in nine pregnant women: A retrospective review of medical records. Lancet 2020, 395, 809–815. [Google Scholar] [CrossRef]
- Zhu, H.; Wang, L.; Fang, C.; Peng, S.; Zhang, L.; Chang, G.; Xia, S.; Zhou, W. Clinical analysis of 10 neonates born to mother with 2019-nCoV pneumonia. Transl. Pediatr. 2020, 9, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Evans-Hoeker, E.A.; Eisenberg, E.; Diamond, M.P.; Legro, R.S.; Alvero, R.; Coutifaris, C.; Casson, P.R.; Christman, G.M.; Hansen, K.R.; Zhang, H.; et al. Major depression, antidepressant use and male and female infertility. Fertil. Steril. 2018, 109, 879–887. [Google Scholar] [CrossRef]
- Nargund, V.H. Effects of psychological stress on male fertility. Nat. Rev. Urol. 2015, 12, 373–382. [Google Scholar] [CrossRef]
- Greene, W.C. A history of AIDS: Looking back to see ahead. Eur. J. Immunol. 2007, 37, S94–S102. [Google Scholar] [CrossRef]
- Wang, L.; Wang, Y.; Ye, D.; Liu, Q. Review of the 2019 novel coronavirus (SARS-CoV-2) based on current evidence. Int. J. Antimicrob. 2020, 55, 105948. [Google Scholar] [CrossRef]
- Central Intelligence Agency (CIA). The World Factbook. Available online: https://www.cia.gov (accessed on 21 August 2020).
- Sharp, P.M.; Hahn, B.H. The evolution of HIV-1 and the origin of AIDS. Philoc. Trans. R. Soc. Lond. B. Bio. Sci. 2010, 365, 2487–2494. [Google Scholar] [CrossRef]
- Tortorici, M.A.; Walls, A.C.; Lang, Y.; Wang, C.; Li, Z.; Koerhuis, D.; Boons, G.-J.; Bosch, B.-J.; Rey, F.A.; de Groot, R.J.; et al. Structural basis for human coronavirus attachment to sialic acid receptors. Nat. Struct. Mol. Biol. 2019, 26, 481–489. [Google Scholar] [CrossRef]
- Matrosovich, M.; Herrler, G.; Klenk, H.D. Sialic acid receptors of viruses. Top Curr. Chem. 2015, 367, 1–28. [Google Scholar]
- Wilen, C.B.; Tilton, J.C.; Doms, R.W. HIV: Cell binding and entry. Cold Spring Harb. Perspect. Med. 2012, 2, a006866. [Google Scholar] [CrossRef]
- Chen, J. Pathogenicity and transmissibility of 2019-nCoV-A quick overview and comparison with other emerging viruses. Microbes Infect. 2020, 22, 69–71. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, J.; Li, H.; Cao, B. A novel coronavirus (COVID-19) outbreak: A call for action. Chest 2020, 157, e99–e101. [Google Scholar] [CrossRef] [PubMed]
- The US Centres for Disease Control and Prevention (CDC). Available online: https://www.cdc.gov/H1N1flu/hosp_deaths_ahdra.html (accessed on 21 August 2020).
- World Health Organization (WHO). Ebola Virus Disease. Available online: https://www.who.int/health-topics/ebola/#tab=tab_1 (accessed on 21 August 2020).
- Ghate, M.V.; Tripathy, S.P.; Kumar, B.K.; Godbole, S.V.; Chittake, A.; Nyayanirgune, P.; Gangakhedkar, R.R.; Divekar, A.D.; Thakar, M.R.; Risbud, A.R.; et al. Rate of hospitalization and inpatient care costs for HIV-1-infected patients in Pune, India. Natl. Med. J. India 2006, 19, 10–14. [Google Scholar] [PubMed]
- Brooks, J.T.; Robbins, K.E.; Youngpairoj, A.S.; Rotblatt, H.; Kerndt, P.R.; Taylor, M.M.; Daar, E.S.; Kalish, M.L. Molecular analysis of HIV strains from a cluster of worker infections in the adult film industry, Los Angeles 2004. AIDS 2006, 20, 923–928. [Google Scholar] [CrossRef] [PubMed]
- Nsubuga, R.N.; White, R.G.; Mayanja, B.N.; Shafer, L.A. Estimation of the HIV basic reproduction number in rural south west Uganda: 1991–2008. PLoS ONE 2014, 9, e83778. [Google Scholar] [CrossRef]
- Bassetti, M.; Vena, A.; Giacobbe, D.R. The novel Chinese coronavirus (2019-nCoV) infections: Challenges for fighting the storm. Eur. J. Clin. Invest. 2020, 50, e13209. [Google Scholar] [CrossRef]
- Vanhems, P.; Hirschel, B.; Phillips, A.N.; Cooper, D.A.; Vizzard, J.; Brassard, J.; Perrin, L. Incubation time of acute human immunodeficiency virus (HIV) infection and duration of acute HIV infection are independent prognostic factors of progression to AIDS. J. Infect. Dis. 2000, 182, 334–337. [Google Scholar] [CrossRef]
- Lai, C.C.; Shih, T.P.; Ko, W.C.; Tang, H.J.; Hsueh, P.R. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): The epidemic and the challenges. Int. J. Antimicrob. Agents. 2020, 55, 105924. [Google Scholar] [CrossRef]
- Bleibtreu, A.; Bertine, M.; Bertin, C.; Houhou-Fidouh, N.; Visseaux, B. Focus on Middle East respiratory syndrome coronavirus (MERS-CoV). Med. Mal. Infect. 2020, 50, 243–251. [Google Scholar] [CrossRef]
- Wilson, N.L.; Azuero, A.; Vance, D.E.; Richman, J.S.; Moneyham, L.D.; Raper, J.L.; Heath, S.L.; Kempf, M.-C. Identifying symptom patterns in people living with HIV disease. J. Assoc. Nurses AIDS Care 2016, 27, 121–132. [Google Scholar] [CrossRef] [PubMed]
- Radiopaedia. H1N1 Influenza. Available online: https://radiopaedia.org/articles/h1n1-influenza (accessed on 21 August 2020).
- Allen, C.M.; Al-Jahdali, H.H.; Irion, K.L.; Ghanem, S.A.; Gouda, A.; Khan, A.N. Imaging lung manifestation of HIV/AIDS. Ann. Thorac. Med. 2010, 5, 201–216. [Google Scholar] [PubMed]
- Banadyga, L.; Siragam, V.; Zhu, W.; He, S.; Cheng, K.; Qiu, X. The cytokine response profile of ebola virus disease in a large cohort of rhesus macaques treated with monoclonal antibodies. Open Forum Infect. Dis. 2019, 6, ofz046. [Google Scholar] [CrossRef] [PubMed]
- Gao, R.; Bhatnagar, J.; Blau, D.M.; Greer, P.; Rollin, D.C.; Denison, A.M.; Deleon-Carnes, M.; Shieh, W.-J.; Sambhara, S.; Tumpey, T.M.; et al. Cytokine and chemokine profiles in lung tissues from fatal cases of 2009 pandemic influenza A (H1N1): Role of the host immune response in pathogenesis. Am. J. Pathol. 2013, 183, 1258–1268. [Google Scholar] [CrossRef]
- Kedzierska, K.; Crowe, S.M. Cytokine and HIV-1: Interaction and clinical implications. Antivir. Chem. Chemother. 2001, 12, 133–150. [Google Scholar] [CrossRef]
- Stockman, L.J.; Bellamy, R.; Garner, P. SARS: Systematic review of treatment effects. PLoS Med. 2006, 3, e343. [Google Scholar] [CrossRef]
- The US Centres for Disease Control and Prevention (CDC). Ebola (Ebola Virus Disease). Available online: https://www.cdc.gov/vhf/ebola/index.html (accessed on 21 August 2020).
- Kemnic, T.R.; Gulick, P.G. HIV antiretroviral therapy. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Padron-Regalado, E. Vaccines for SARS-CoV-2: Lessons from other coronavirus strains. Infect. Dis. Ther. 2020, 9, 255–274. [Google Scholar] [CrossRef]
- Orellana, C. Phase I SARS vaccine trial in China. Lancet Infect. Dis. 2004, 4, 388. [Google Scholar] [CrossRef]
- Tang, L.; Zhu, Q.; Qin, E.; Yu, M.; Ding, Z.; Shi, H.; Cheng, X.; Wang, C.; Chang, G.; Zhu, Q.; et al. Inactivated SARS-CoV vaccine prepared from whole virus induces a high level of neutralizing antibodies in BALB/c mice. DNA Cell Biol. 2004, 23, 391–394. [Google Scholar] [CrossRef]
- Lin, J.-T.; Zhang, J.-S.; Su, N.; Xu, J.-G.; Wang, N.; Chen, J.-T.; Chen, X.; Liu, Y.-X.; Gao, H.; Jia, Y.-P.; et al. Safety and immunogenicity from a phase I trial of inactivated severe acute respiratory syndrome coronavirus vaccine. Antivir. Ther. 2007, 12, 1107–1113. [Google Scholar]
- Bolles, M.; Deming, D.; Long, K.; Agnihothram, S.; Whitmore, A.; Ferris, M.; Funkhouser, W.; Gralinski, L.; Totura, A.; Heise, M.; et al. A double-inacivated severe acute respiratory syndrome coronavirus vaccine provides incomplete protection in mice and induces increased eosinophilic proinflammatory pulmonary response upon challenge. J. Virol. 2011, 85, 12201–12215. [Google Scholar] [CrossRef] [PubMed]
- Modjarrad, K.; Roberts, C.C.; Mills, K.T.; Castellano, A.R.; Paolini, K.; Muthumani, K.; Reuschel, E.L.; Robb, M.L.; Racine, T.; Oh, M.-D.; et al. Safety and immunogenicity of an anti-Middle East respiratory syndrome coronavirus DNA vaccine: A phase I, open-label, single-arm, dose-escalation trial. Lancet Infect. Dis. 2019, 19, 1013–1022. [Google Scholar] [CrossRef]
- Sutton, T.C.; Subbarao, K. Development of animal models against emerging coronaviruses: From SARS to MERS coronavirus. Virology 2015, 479, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Netland, J.; DeDiego, M.L.; Zhao, J.; Fett, C.; Alvarez, E.; Nieto-Torres, J.L.; Enjuanes, L.; Perlman, S. Immunization with an attenuated severe acute respiratory syndrome coronavirus deleted in E protein protects against lethal respiratory disease. Virology 2010, 399, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Tang, F.; Quan, Y.; Xin, Z.-T.; Wrammert, J.; Ma, M.-J.; Lv, H.; Wang, T.-B.; Yang, H.; Richardus, J.H.; Liu, W.; et al. Lack of peripheral memory B cell responses in recovered patients with severe acute respiratory syndrome: A six-year follow-up study. J. Immunol. 2011, 186, 7264–7268. [Google Scholar] [CrossRef]
- Payne, D.C.; Iblam, I.; Rha, B.; Alqasrawi, S.; Haddadin, A.; Nsour, M.A.; Alsanouri, T.; Ali, S.S.; Harcourt, J.; Miao, C.; et al. Persistence of antibodies against Middle East respiratory syndrome coronavirus. Emerg. Infect. Dis. 2016, 22, 1824–1826. [Google Scholar] [CrossRef]
- Zhao, J.; Alshukari, A.N.; Baharoon, S.A.; Ahmed, W.A.; Bokhari, A.A.; Nehdi, A.M.; Layqah, L.A.; Alghamdi, M.G.; Gethamy, M.M.A.; Dada, A.M.; et al. Recovery from the Middle East respiratory syndrome is associated with antibody and T-cell response. Sci. Immunol. 2017, 2, eaan5393. [Google Scholar] [CrossRef]
- World Health Organization (WHO). DRAFT Landscape of COVID-19 Candidate Vaccines—3 September 2020. Available online: https://www.who.int/docs/default-source/coronaviruse/novel-coronavirus-landscape-covid-19-(4).pdf?sfvrsn=bc448477_1&download=true (accessed on 6 September 2020).
- Pfizer. Pfizer and BioNTech Announce Vaccine Candidate Against COVID-19 Achieved in First Interim Analysis from Phase 3 Study. Available online: https://www.pfizer.com/news/press-release/press-release-detail/pfizer-and-biontech-announce-vaccine-candidate-against (accessed on 9 December 2020).
- Moderna. Moderna’s Fully Phase 3 COVE Study of mRNA-1273. Available online: https://www.modernatx.com/cove-study (accessed on 9 December 2020).
- National Geographic. Dozens of COVID-19 Vaccines Are in Development. Here Are the Ones to Follow—3 September 2020. Available online: https://www.nationalgeographic.com/science/health-and-human-body/human-diseases/coronavirus-vaccine-tracker-how-they-work-latest-developments-cvd/ (accessed on 6 September 2020).
- British Braodcasting Corporation (BBC). Covid-19 Vaccine: First Person Receives Pfizer Jab in UK. Available online: https://www.bbc.com/news/uk-55227325 (accessed on 10 December 2020).
- Novavax. Novavax Announces COVID-19 Vaccine Clinical Development Progress. Available online: https://ir.novavax.com/news-releases/news-release-details/novavax-announces-covid-19-vaccine-clinical-development-progress (accessed on 9 December 2020).
- National Institute of Health (NIH). A Phase III Clinical Trial to Determine the Safety and Efficacy of ZF 2001 for Prevention of COVID-19. Available online: https://clinicaltrials.gov/ct2/show/results/NCT04646590?view=results (accessed on 9 December 2020).
- YICAI. China’s Zhifei Finishes Covid-19 Vaccine’s Phase II Clinical Trial in Three Months in Race to the Market. Available online: https://www.yicaiglobal.com/news/china-zhifei-finishes-covid-19-vaccine-phase-ii-clinical-trial-in-three-months-in-race-to-the-market (accessed on 9 December 2020).
- National Affairs Professionals Society (RAPS). COVID-19 Vaccine Tracker. Available online: https://www.raps.org/news-and-articles/news-articles/2020/3/covid-19-vaccine-tracker (accessed on 8 November 2020).
- Mahase, E. COVID-19: Moderna vaccine is nearly 95% effective, trial involving high risk and elderly people shows. BMJ 2020, 371, m4471. [Google Scholar] [CrossRef]
- Mahase, E. COVID-19: Oxford vaccine is up to 90% effective, interim analysis indicates. BMJ 2020, 371, m4564. [Google Scholar] [CrossRef]
- Neucrad Health India (NHI). Covaxin and ZyCoV-D: Recent Update of Covid-19 Vaccine Candidates in India. Available online: https://neucradhealth.in/language/en/covaxin-and-zycov-d-recent-update-of-covid-19-vaccine-candidates-in-india/ (accessed on 8 November 2020).
- Mercado, N.B.; Zahn, R.; Wegmann, F.; Loos, C.; Chandrashekar, A.; Yu, J.; Liu, J.; Peter, L.; McMahan, K.; Tostanoski, L.H.; et al. Single-shot Ad26 vaccine protects against SARS-CoV-2 in rhesus macaques. Nature 2020, 586, 583–588. [Google Scholar] [CrossRef]
- Heymann, D.L.; Rodier, G. SARS: Lessons from a new disease. In Learning From SARS: Preparing for the Next Disease Outbreak; Knobler, S., Mahmoud, A., Lemon, S., Mack, A., Sivitz, L., Oberholtzer, K., Eds.; The National Academic Press: Washington, DC, USA, 2004; pp. 234–246. [Google Scholar]
- Global Health Security Agenda (GHSA). The 6th GHSA Ministerial Meeting. Available online: https://www.ghsagenda.org (accessed on 7 December 2020).
- Filho, W.L.; Brandli, L.L.; Salvia, A.L.; Rayman-Bacchus, L.; Platje, J. COVID-19 and the UN Sustainable Development Goals: Threat to solidarity or an opportunity. Sustainability 2020, 12, 5343. [Google Scholar] [CrossRef]
- The World Bank. Reversing Setbacks to Poverty Reduction Requires Nations to Work Together for a Resilient Recovery. Available online: https://www.worldbank.org/en/news/immersive-story/2020/11/09/reversing-setbacks-to-poverty-reduction-requires-nations-to-work-together-for-a-resilient-recovery (accessed on 2 December 2020).
- The World Bank. The Impact of COVID-19 on Global Poverty under Worsening Growth and Inequality. Available online: https://blogs.worldbank.org/opendata/impact-covid-19-global-poverty-under-worsening-growth-and-inequality (accessed on 2 December 2020).
- Khanna, R.C.; Cicinelli, M.V.; Gilbert, S.S.; Honavar, S.G.; Murthy, G.S. COVID-19 pandemic: Lessons learned and future directions. Indian J. Opthalmol. 2020, 68, 703–710. [Google Scholar] [CrossRef] [PubMed]
- WHO (World Health Organization). WHO Statement on the Tenth Meeting of the IHR Emergency Committee Regarding MERS. 3 September 2016. Available online: https://www.who.int/mediacentre/news/statements/2015/ihr-emergency-committee-mers/en/ (accessed on 21 August 2020).
- Saeed, A.A.B.; Abedi, G.R.; Alzahrani, A.G.; Salameh, I.; Abdirizak, F.; Alhakeem, R.; Algarmi, H.; Nil, O.A.E.; Mohammed, M.; Assiri, A.M.; et al. Surveillance and testing for Middle East respiratory syndrome coronavirus, Saudi Arabia, April 2015–February 2016. Emerg. Infect. Dis. 2017, 23, 682–685. [Google Scholar] [CrossRef]
- World Health Organization (WHO). WHO ad hoc Scientific Teleconference on the Current Influenza A (H1N1) Situation. Available online: https://www.who.int/csr/resources/publications/swineflu/tc_report_2009_04_29/en/ (accessed on 21 August 2020).
- Chong, K.C.; Fong, H.F.; Zee, C.Y. Estimating the incidence reporting rates of new influenza pandemics at an early stage using travel data from the source country. Epidemiol. Infect. 2014, 142, 955–963. [Google Scholar] [CrossRef] [PubMed]
- Woolhouse, M.E.J.; Rambaut, A.; Kellam, P. Lessons from ebola: Improving infectious disease surveillance to inform outbreak management. Sci. Transl. Med. 2015, 7, 307rv5. [Google Scholar] [CrossRef] [PubMed]
- Clerc, O.; Greub, G. Routine use of point-of-care tests: Usefulness and application in clinical microbiology. Clin. Microbiol. Infect. 2010, 16, 1054–1061. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, P.; Gardner, L.I.; Buchacz, K.; Garland, P.M.; Mugavero, M.J.; Bosshart, J.T.; Shouse, R.L.; Bertolli, J. Shifting the paradigm: Using HIV surveillance data as a foundation for improving HIV care and preventing HIV infection. Milbank Q. 2013, 91, 558–603. [Google Scholar] [CrossRef]
- Kramer, J.B.; Brown, D.E.; Kopar, P.K. Ethics in the time of coronavirus: Recommendations in the COVID-19 pandemic. J. Am. Coll. Surg. 2020, 230, 1114–1118. [Google Scholar] [CrossRef]
- Kim, J.Y.; Choe, P.G.; Oh, Y.; Kim, J.; Park, S.J. The first case of 2019 novel coronavirus pneumonia imported into Korea from Wuhan, China: Implication for infection prevention and control measures. J. Korean Med. Sci. 2020, 35, e61. [Google Scholar] [CrossRef]
- Patel, A.; Jernigan, D.B. 2019-nCOV CDC response team initial public health response and interim clinical guidance for the 2019 novel coronavirus outbreak—United States, 31 December 2019—4 February 2020. MMWR Morb. Mortal Wkly. Rep. 2020, 69, 140–146. [Google Scholar] [CrossRef]
- World Economic Forum. Coronavirus—This Is How the World Is Responding. Available online: https://www.weforum.org/agenda/2020/03/coronavirus-this-is-how-the-world-is-responding/ (accessed on 21 August 2020).
- World Health Organization (WHO). Novel Coronavirus (2019-nCOV) Situation Report 1. Available online: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200121-sitrep-1-2019-ncov.pdf?sfvrsn=20a99c10_4 (accessed on 21 August 2020).
- Zyoud, S.H. Global research trends of Middle East respiratory syndrome coronavirus: A bibliometric analysis. BMC Infect. Dis. 2016, 16, 255. [Google Scholar] [CrossRef] [PubMed]
- Xue, L.; Zeng, G. Golbal strategies and response measures to the influenza A (H1N1) pandemic. In A Comprehensive Evaluation on Emergency Response in China. Research Series on the Chinese Dream and China’s Development Path; Xue, L., Zeng, G., Eds.; Springer: Singapore, 2019; pp. 15–44. [Google Scholar]
- People.cn. Developed Countries Rush to Buy Influenza (H1N1) Vaccines, Leaving Poor Countries Worried about Epidemic Control. Available online: http://medicine.people.com.cn/GB/9732375.html (accessed on 3 October 2020).
- Keusch, G.; McAdam, K.; Cuff, P.A.; Mancher, M.; Busta, E.R. Strengthening capacity for response and research. In Integrating Clinical Research into Epidemic Response: The Ebola Experience; Keusch, G., McAdam, K., Cuff, P.A., Mancher, M., Busta, E.R., Eds.; The National Academic Press: Washington, DC, USA, 2017; pp. 155–198. [Google Scholar]
- Balachova, T.N.; Batluk, J.V.; Bryant, K.J.; Shaboltas, A.V. International collaboration in HIV prevention research: Evidence from a research seminar in Russia. AIDS Res. Hum. Retrovir. 2015, 31, 163–172. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, K.K.; Solomon, P.; Ibanez-Carrasco, F.; Chegwidden, W.; McDonnell, E.; Brown, D.; Harding, R.; Bergin, C.; Worthington, C.; Tattle, S.; et al. Evolution of an international research collaborative in HIV and rehabilitation: Community engaged process, lessons learned, and recommendations. Prog. Community Health Partnersh. 2018, 12, 395–408. [Google Scholar]
- Frazer, I.H. Collaboration in the war against viruses: A multidisciplinary international effort. Innovation 2020, 1, 100011. [Google Scholar] [CrossRef]
- Moradian, N.; Ochs, H.D.; Sedikies, C.; Hamblin, M.R.; Camargo, C.A., Jr.; Martinez, J.A.; Biamonte, J.D.; Abdollahi, M.; Torres, P.J.; Nieto, J.J.; et al. The urgent need for integrated science to fight COVID-19 pandemic and beyond. J. Transl. Med. 2020, 18, 205. [Google Scholar] [CrossRef] [PubMed]
- World Economic Forum. COVID-19: Collaboration Is the Engine of Global Science—Especially for Developing Countries. Available online: https://www.weforum.org/agenda/2020/05/global-science-collaboration-open-source-covid-19/ (accessed on 21 August 2020).
- Soheilypour, M.; Mofrad, M.R.K. Agent-based modeling in molecular systems biology. BioEssays 2018, 40, e1800020. [Google Scholar] [CrossRef]
- Ming, R.-X.; Liu, J.; Cheung, W.K.W.; Wan, X. Stochastic modeling of infectious diseases for heterogeneous populations. Infect. Dis. Poverty 2016, 5, 107. [Google Scholar] [CrossRef]
- Adamu, H.; Muhammad, M.M.; Jingi, A.; Usman, M. Mathematical modeling using improved SIR model with more realistic assumptions. Int. J. Eng. Appl. Sci. 2019, 6, 64–69. [Google Scholar]
- Verbeek, J.H.; Ijaz, S.; Mischke, C.; Routsalainen, J.H.; Makela, E.; Neuvonen, K.; Edmond, M.B.; Sauni, R.; Balci, F.S.K.; Mihalache, R.C. Personal protective equipment for preventing highly infectious disease due to exposure to contaminated body fluids in healthcare staff. Cochrane Database Syst. Rev. 2016, 4, CD011621. [Google Scholar] [CrossRef]
- Rubinson, L.; Mutter, R.; Viboud, C.; Hupert, N.; Uyeki, T.; Creanga, A.; Finelli, L.; Iwashyna, T.J.; Carr, B.; Merchant, R.; et al. Impact of the fall 2009 influenza A(H1N1)pdm09 pandemic on US hospitals. Med. Care. 2013, 51, 259–265. [Google Scholar] [CrossRef]
- Dentico, N. Ebola and the global governance of health. Recenti. Prog. Med. 2014, 105, 405–406. [Google Scholar] [PubMed]
- GSDRC. Impact and Implications of the Ebola Crisis. Available online: https://www.gsdrc.org/docs/open/HDQ1177.pdf (accessed on 26 September 2020).
- Studies IfS. Ebola Threatens Social and Political Stability in Affected Countries. Available online: https://www.issafrica.org/iss-today/ebola-threatens-social-and-political-stability-in-affected-countries (accessed on 26 September 2020).
- Regmi, K.; Gilbert, R.; Thunhurst, C. How can health systems be strengthened to control and prevent an ebola outbreak? A narrative review. Infect. Ecol. Epidemiol. 2015, 5, 28877. [Google Scholar] [CrossRef] [PubMed]
- Koto, M.V.; Maharaj, P. Difficulties facing healthcare workers in the era of AIDS treatment in Lesotho. SAHARA J. 2016, 13, 53–59. [Google Scholar] [CrossRef]
- Kretchy, I.A.; Asiedu-Danso, M.; Kretchy, J. Medication management and adherence during the COVID-19 pandemic: Perspectives and experiences from low- and middle-income countries. Res. Soc. Adm. Pharm. 2020, 17, 2023–2026. [Google Scholar] [CrossRef] [PubMed]
- Gallup. COVID-19 Quickly Becomes Most Important US Problem. Available online: https://news.gallup.com/poll/309038/covid-quickly-becomes-important-problem.aspx (accessed on 21 August 2020).
- China Global Television Network (CGTN). COVID-19 UK Roundup: Latest Developments and Challenges. Available online: https://news-cgtn-com.cdn.ampproject.org/v/s/news.cgtn.com/news/2020-05-04/COVID-19-UK-Roundup-Latest-developments-and-challenges-QdglVgctEs/share_amp.html?amp_js_v=a6&_gsa=1&usqp=mq331AQHKAFQArABIA%3D%3D#aoh=16081908079024&referrer=https%3A%2F%2Fwww.google.com&_tf=From%20%251%24s&share=https%3A%2F%2Fnews.cgtn.com%2Fnews%2F2020-05-04%2FCOVID-19-UK-Roundup-Latest-developments-and-challenges-QdglVgctEs%2Findex.html (accessed on 21 August 2020).
- Financial Magazine. 6 Key COVID-19 Challenges Facing UK Businesses Today. Available online: https://www.fm-magazine.com/news/2020/may/key-coronavirus-challenges-for-uk-businesses-cima-andrew-harding.html (accessed on 21 August 2020).
- Garg, S.; Basu, S.; Rustagi, R.; Borle, A. Primary health care facility preparedness for outpatient service provision during the COVID-19 pandemic in India: Cross-sectional study. JMIR Public Health Surveill. 2020, 6, e19927. [Google Scholar] [CrossRef] [PubMed]
- Halder, N.; Kelso, J.K.; Milne, G.J. Analysis of the effectiveness of interventions used during the 2009 A/H1N1 influenza pandemic. BMC Public Health 2010, 10, 168. [Google Scholar] [CrossRef]
- Kirsch, T.D.; Moseson, H.; Massaquoi, M.; Nyenswah, T.; Goodermote, R.; Rodriguez-Barrquer, I.; Lessler, J.; Cumings, D.A.T.; Peters, D.H. Impact of interventions and the incidence of ebola virus disease in Liberia-implications for future epidemics. Health Policy Plan 2017, 32, 205–214. [Google Scholar] [CrossRef]
- Brown, J.L.; Sales, J.M.; DiClemente, R.J. Combination HIV prevention interventions: The potential of integrated behavioral and biomedical approaches. Curr. HIV/AIDS Rep. 2014, 11, 363–375. [Google Scholar] [CrossRef]
- Maier, B.F.; Brockmann, D. Effective containment explains subexponential growth in recent confirmed COVID-19 cases in China. Science 2020, 368, 742–746. [Google Scholar] [CrossRef]
- Dong, L.; Hu, S.; Gao, J. Discovering drugs to treat coronavirus disease 2019 (COVID-19). Drug Discov. Ther. 2020, 14, 58–60. [Google Scholar] [CrossRef]
- Frediansyah, A.; Nainu, F.; Dhama, K.; Mudatsir, M.; Harapan, H. Remdesivir and its antiviral activity against COVID-19: A systematic review. Clin. Epidemiol. Glob. Health 2020, in press. [Google Scholar] [CrossRef] [PubMed]
- Beigel, J.H.; Tomashek, K.M.; Dodd, L.E.; Mehta, A.K.; Zingman, B.S.; Kalil, A.C.; Hohmann, E.; Chu, H.Y.; Luetkemeyer, A.; Kline, S.; et al. Remdesivir for the treatment of Covid-19- preliminary report. N. Engl. J. Med. 2020, 383, 994. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, D.; Du, G.; Du, R.; Zhao, J.; Jin, Y.; Fu, S.; Gao, L.; Cheng, Z.; Lu, Q.; et al. Remdesivir in adults with severe COVID-19: A randomized, double-bling, placebo-controlled, multicentre trial. Lancet 2020, 395, 1569–1578. [Google Scholar] [CrossRef]
- National Institute of Health (NIH). NIH Clinical Trial Shows Remdesivir Accelerates Recovery from Advanced COVID-19. Available online: https://www.nih.gov/news-events/news-releases/nih-clinical-trial-shows-remdesivir-accelerates-recovery-advanced-covid-19 (accessed on 27 September 2020).
- World Health Organization (WHO). Coronavirus Disease (COVID-19): Dexamethasone. Available online: https://www.who.int/news-room/q-a-detail/coronavirus-disease-covid-19-dexamethasone (accessed on 27 October 2020).
- Rohrich, R.J.; Hamilton, K.L.; Avashia, Y.; Savetsky, I. The COVID-19 pandemic: Changing lives and lessons learned. Plast. Reconstr. Surg. Glob. Open 2020, 8, e2854. [Google Scholar] [CrossRef]
- Bong, C.-L.; Brasher, C.; Chikumba, E.; McDougall, R.; Mellin-Olsen, J.; Enright, A. The COVID-19 pandemic: Effects on low- and middle-income countries. Anesth. Anal. 2020, 131, 86–92. [Google Scholar] [CrossRef]
- Li, H.; Liu, S.-M.; Yu, X.-H.; Tang, S.-L.; Tang, C.-K. Coronavirus disease 2019 (COVID-19): Current status and future perspectives. Int. J. Antimicrob. Agents 2020, 55, 105951. [Google Scholar] [CrossRef]
- Wang, F.; Kream, R.M.; Stefano, G.B. An evidence based perspective on mRNA- SARS-CoV-2 vaccine development. Med. Sci. Monit. 2020, 26, e924700. [Google Scholar]
- Gennaro, F.D.; Pizzol, D.; Marotta, C.; Antunes, M.; Racalbuto, V.; Veronese, N.; Smith, L. Coronavirus disease (COVID-19) current status and future perspectives: A narrative review. Int. J. Environ. Res. Public Health 2020, 17, 2690. [Google Scholar] [CrossRef]
| SARS-CoV-2 | SARS-CoV | MERS-CoV | Ebola | H1N1 | HIV | References | |
|---|---|---|---|---|---|---|---|
| Outbreak year and location of first reported cases | 2019 (Wuhan, China) | 2003 (Southern China) | 2012 (Saudi Arabia) | 1976 onwards (Central Africa) | 2009 (North America) | 1981 onwards (West Central Africa) | [172] |
| Outbreak countries | More than 215 countries, including the USA, India, Brazil, China, Japan, Korea, Italy, etc. | 29 countries, including China, Vietnam, Singapore and Canada | More than 27 countries, mainly in Saudi Arabia, South Korea, Jordan and Qatar | Africa, the Americas, South East Asia, Europe, Eastern Mediterranean, Western Pacific | Africa, Europe, the Americas, South-East Asia | More than 130 countries, including the USA, China, India, etc. | [173,174] |
| Natural reservoir | Not identified | Bat | Bat | Fruit bats, porcupines and non-human primates | Human, avian, swine | Chimpanzee | [173,175] |
| Receptor | ACE2, TMPRSS2, sialic acid | ACE2, CD206, sialic acid | DPP4 (CD26), sialic acid | TIM1 (NPC1) | sialic acid | CD4 | [79,80,81,176,177,178] |
| Case Fatality Rate | Not identified, at least 2–3% | 10% | 34.4–37% | 50–63% | 0.02–0.4% | 80–90% | [179,180] |
| Hospitalization rate | ~19% | Most cases | Most cases | 25–90% | 16.19–58.76% | >34.2% | [181,182,183] |
| Community attack rate | 30–40% | 10–60% | 4–13% | 5–30% | 10–20% | 23% | [184] |
| Basic reproductive number (R0) | 1.4–6.4 | 2–5 | <1 | 1.9 | 1.3 | 2–5 | [179,185] |
| Median incubation time | 5.2 days | 5 days | 5 days | 2-21 days | 1–7 days | 5–70 | [186,187] |
| Clinical symptoms | Fever (98%), cough (77%), dyspnea (63.5%), myalgia (11.5%), malaise (35%) and so on | Fever (>99%), cough (62%–100%), chills or rigor (15%–73%), diarrhea 20%, dyspnea (40%) | Fever (77%), cough (90%), dyspnea (68%), sputum production (40%), odynophagia (39%), digestive system/signs (20%), hemoptysis (4.3%), myalgia (43%) and headache (20%) | Fever, fatigue, muscle, pain, headache, sore throat, vomiting, diarrhea, rash, kidney and liver impairments and, in some cases, internal and external bleeding (e.g., oozing from the gums, blood in the stools). Laboratory findings include low WBCs and platelet counts and elevated liver enzymes. | Fever, chills, cough, sore throat, runny or stuffy nose, red eyes, body aches, headache, fatigue, diarrhea, nausea and vomiting | Muscle aches (85%), fatigue (84%), bloating (82%), fever (79%), headache (73%), memory loss (73%), cough (74%), poor appetite (74%), diarrhea (71%) and nausea (72%) | [50,182,188,189,190] |
| Radiology | Critically ill with bilateral multiple lobular and subsegmental areas of consolidation; mild ill with bilateral ground-glass opacity and subsegment alareas of consolidation, almost 100% of patients with abnormal CT | Unilateral/bilateral ground-glass opacities or focal unilateral/bilateral consolidation. The rate of abnormal chest radiography or CT was >94% | Unilateral/bilateral patchy densities or infiltrates, bilateral hilar infiltration, segmented/lobar opacities, ground-glass opacities and possible small pleural effusions. The rate of abnormal chest radiography or CT was between 90% and100% | Aerosolized virus would be unlikely to produce discrete, radiographically visible, pulmonary lesions. | Initial chest radiographs show central or peripheral pulmonary GGO and consolidations with patchy or nodular appearance; multizonal and bilateral peripheral opacities are associated with adverse prognosis. | Bronchiectasis, with ill-defined centrilobular micronodularity and branching structures to mucous impaction in the bronchioles, along with cavitation. | [186,191,192] |
| Cytokines | Increased levels of IL-1β, IL1RA, IL-7, IL-8, IL-9, IL-10, basic FGF, GCSF, GMCSF, IFN-Ɣ, IP10, MCP1, MIP1α, MIP1β, PDGF, TNF-α and VEGF; Critically ill patients have high levels of GCSF, TNF-α and Th2 cytokines (e.g., IL-4 and IL 10) | Increased levels of IL-1β, IL-6, IL-12, IFN-Ɣ, IP10 and MCP-1 | Increased concentrations of proinflammatory cytokines (IFN-Ɣ, TNF-α, IL-15 and IL-17) | TNF-α, IFN-γ, IL-1RA, IL-6, IL-15, MIG, MIF, MIP-1α, MIP-1β, MCP-1, IP-10, ITAC, eotaxin, IL-2, IL-1β, IL-8, HGF, VEGF, GM-CSF and G-CSF | IL1RA, IL-6, TNF-α, IL-8, MCP-1, MIP1β and interferon-inducing protein-10 | Increased levels of TNF-α, TNF-β, IFN-Ɣ, IL-1, IL-2, IL-6, IL-7, IL-10, IL-13, IL-15 and IL-16 | [148,186,193,194,195] |
| Treatment | Corticosteroids, remdesivir, combination of lopinavir and ritonavir, type I interferon and so on | Lopinavir and ritonavir, corticosteroids, IFN-Ɣ, IVIG | IFN-Ɣ, lopinavir and ritonavir, mycophenolic acid | During the 2018 eastern Democratic Republic of the Congo outbreak, two out of four investigational treatments initially available to treat patients with confirmed Ebola, are still in use. These two are REGN-EB3 and mAb114. In addition, treatments include fluid intake and intravenous electrolytes, oxygen therapy and using medication to manage blood pressure, vomiting, diarrhea, fever and pain. | Oseltamivir (Tamiflu),peramivir (Rapivab) andzanamivir (Relenza) appeartowork best, although some types of swine flu do not respond to oseltamivir. | Anti-retroviral therapy, which includes medications such as abacavir, efavirenz, enfuvirtide, atazanavir, maraviroc, dolutegavir, ibalizumab, cobicistat, etc. | [180,196,197,198] |
| Name of Vaccine | Developer Country | Institute and Company | Mode of Action | Results to Date | References |
|---|---|---|---|---|---|
| Sputnik V | Russia | The Gamaleya National Center for Epidemiology and Microbiology | A viral vector vaccine that uses a weakened version of the common cold-causing adenovirus to introduce the SARS-CoV-2 spike protein to the body. | Researchers claim that the vaccine can induce strong antibody and cellular immune responses. However, published data on the clinical trials are not available yet. | [210,213,214,215,216,217,218,219,220,221,222] |
| mRNA-1273 | USA | Moderna Therapeutics | An mRNA-based vaccine that mimics the coronavirus, thus training the immune system to recognize its presence. | Phase III clinical trials. Trials involving high risk and elderly showed that it is nearly 95% effective. | |
| BNT162b2 | USA | Pfizer and BioNTech (Germany) | A nucleoside-modified mRNA that encodes an optimized SARS-CoV-2 full-length spike protein antigen. It contains a piece of the spike protein that elicits an antibody response. | Patients demonstrated a favorable overall tolerability during phase I/II trials and induction of a favorable viral-specific CD4+ and CD8+ T-cell response. Received temporary authorization from the UK Medicines and Healthcare products Regulatory Agency on 2 December, 2020. On 8 December 2020 at 6.31 am local time in London, UK, 334 days after the first reported Covid-19 death in China, Margaret Keenan, 90, became the first person in the world to receive a clinically approved vaccine. | |
| AZD1222 | UK | The University of Oxford; AstraZeneca; IQVIA; Serum Institute of India | A non-replicating viral vector with the viral spike protein, which induces an immune response. | Currently undergoing phase III clinical trials. Phase III interim results, based on 131 cases, as declared via press release (23 November 2020) suggest that it can be up to 90% effective when a half dose is given, followed by a full dose one month later. | |
| Covaxin | India | Bharat Biotech; National Institute of Virology | An inactivated vaccine to trigger specific T-lymphocytes and neutralizing antibodies by the host’s immune system. | Currently undergoing phase III clinical trials. The participants of the clinical trials are reportedly healthy, adeno adverse impacts of the vaccine have been found to date. | |
| CoronaVac | China | Sinovac and Butanan (Brazil) | An inactivated vaccine that initiates an immune response without producing the disease. | Currently undergoing phase III clinical trials. Subjects in the phase II human trial produced antibodies with no severe adverse reactions. | |
| No name announced | China | Sinopharm and Wuhan Institute of Biological Products | An inactivated vaccine that is renderednon-infectious but retains enough surface proteins to set off an immune response. | Undergoing phase III clinical trials. Earlier trial phases have shown that the vaccine can trigger an antibody response with no serious adverse effects. | |
| JNJ-78436735 | USA | Johnson and Johnson | Non-replicating viral vector. Optimal Ad26 vector-based vaccine for SARS-CoV-2 | Currently undergoing phase III clinical trials. Initial data demonstrated that a single shot of the vaccine provided protection against SARS-CoV-2 in non-human primates. | |
| Ad5-nCoV | China | Cansino Biologics | A viral vector vaccine made using a weakened version of the adenovirus (with faulty replication mechanism) as a vehicle for introducing the SARS-CoV-2 spike protein to the body. | Currently undergoing phase III clinical trials. Phase II trials showed that the vaccine produces significant immune responses in the majority of recipients after a single immunization. | |
| NVX-CoV2373 | USA | Novavax | It is a protein subunit vaccine made with full-length recombinant SARS-CoV-2 glycoprotein nanoparticles, adjuvated with Matrix M, which enhances immune response and stimulates high levels of neutralizing antibodies by increasing the rate of antigen-presentation in the local lymph nodes. | Currently under phase III clinical trials, this vaccine candidates demonstrated efficient binding with receptors targeted by the virus, which is a critical aspect of effective vaccine action. | |
| ZF2001 | China | Anhui Zhifei Longcom Biopharmaceutical and Institute of Microbiology, Chinese Academy of Sciences | It is an adjuvated recombinant protein subunit vaccine expressed in CHO cells. It probably elicits protective action against the virus by increasing the level of neutralizing antibody and IgG antibody. | Currently under phase III clinical trials, the vaccine candidate showed promising results during the earlier phases by generating immune response. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roychoudhury, S.; Das, A.; Sengupta, P.; Dutta, S.; Roychoudhury, S.; Choudhury, A.P.; Ahmed, A.B.F.; Bhattacharjee, S.; Slama, P. Viral Pandemics of the Last Four Decades: Pathophysiology, Health Impacts and Perspectives. Int. J. Environ. Res. Public Health 2020, 17, 9411. https://doi.org/10.3390/ijerph17249411
Roychoudhury S, Das A, Sengupta P, Dutta S, Roychoudhury S, Choudhury AP, Ahmed ABF, Bhattacharjee S, Slama P. Viral Pandemics of the Last Four Decades: Pathophysiology, Health Impacts and Perspectives. International Journal of Environmental Research and Public Health. 2020; 17(24):9411. https://doi.org/10.3390/ijerph17249411
Chicago/Turabian StyleRoychoudhury, Shubhadeep, Anandan Das, Pallav Sengupta, Sulagna Dutta, Shatabhisha Roychoudhury, Arun Paul Choudhury, A. B. Fuzayel Ahmed, Saumendra Bhattacharjee, and Petr Slama. 2020. "Viral Pandemics of the Last Four Decades: Pathophysiology, Health Impacts and Perspectives" International Journal of Environmental Research and Public Health 17, no. 24: 9411. https://doi.org/10.3390/ijerph17249411
APA StyleRoychoudhury, S., Das, A., Sengupta, P., Dutta, S., Roychoudhury, S., Choudhury, A. P., Ahmed, A. B. F., Bhattacharjee, S., & Slama, P. (2020). Viral Pandemics of the Last Four Decades: Pathophysiology, Health Impacts and Perspectives. International Journal of Environmental Research and Public Health, 17(24), 9411. https://doi.org/10.3390/ijerph17249411









