Who Is Best to Test? A Systematic Review of Chlamydia Infections in Switzerland
Abstract
1. Introduction
2. Materials and Methods
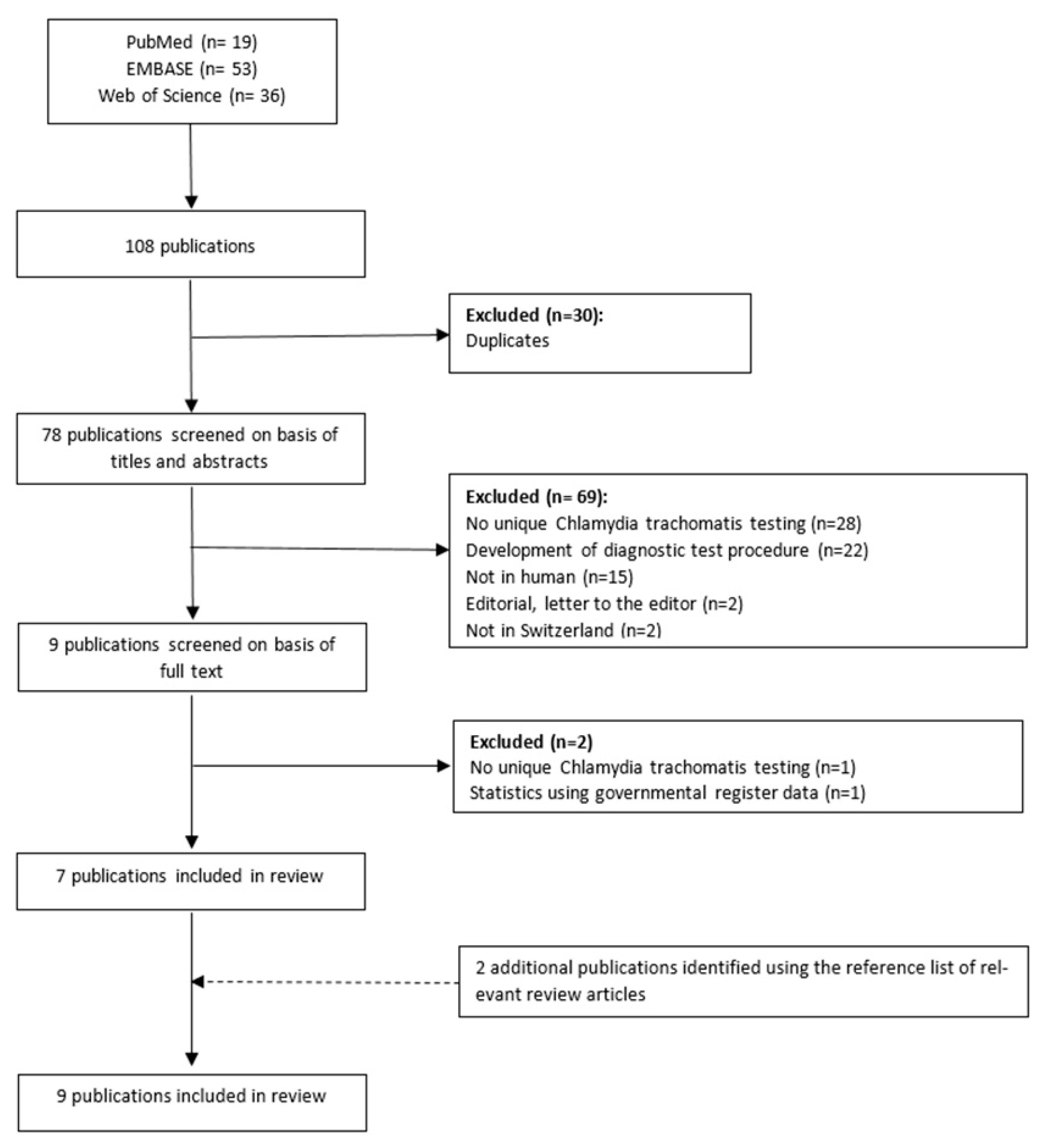
2.1. Search Method
2.2. Study Selection and Data Collection
2.3. Quality Assessment
2.4. Statistical Analysis
3. Results
3.1. Study Population
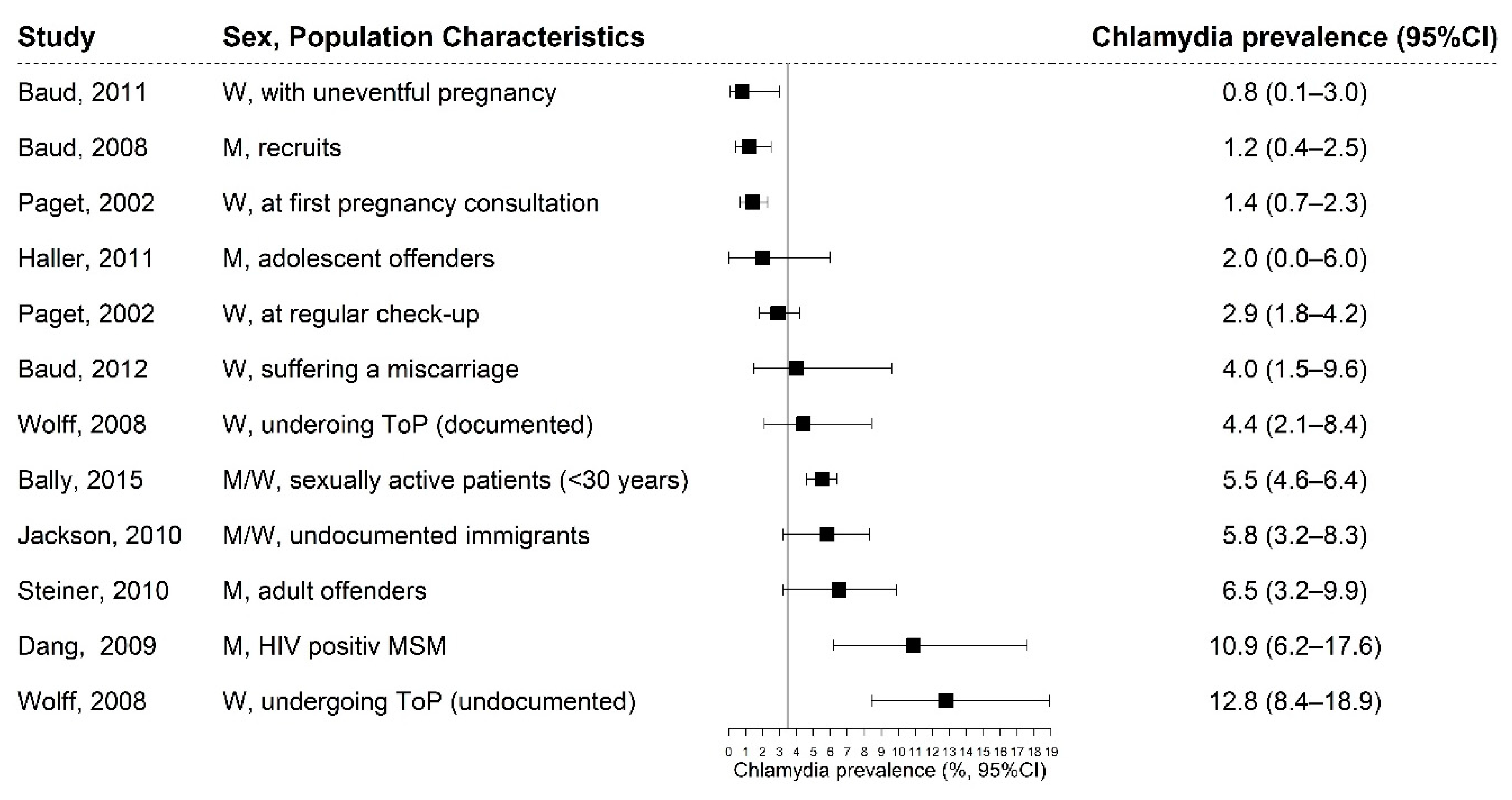
3.2. Chlamydia Prevalence
3.3. Recommendations for Chlamydia Testing
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Database | Search Terms |
|---|---|
| PubMed | chlamydia [MeSH Terms] |
| chlamydia trachomatis [MeSH Terms] | |
| chlamydia infections [MeSH Terms] | |
| 1 OR 2 OR 3 | |
| screening [all Fields] | |
| mass screening [MeSH Terms] | |
| testing [all Fields] | |
| diagnosis [Subheading] | |
| diagnosis [all Fields] | |
| 5 OR 6 OR 7 OR 8 OR 9 | |
| Switzerland [MeSH Terms] | |
| Swiss [all Fields] | |
| 11 OR 12 | |
| 4 AND 10 AND 13 | |
| limit 14 to human | |
| EMBASE | chlamydia [Emtree] |
| chlamydia trachomatis [Emtree] | |
| chlamydia infection * | |
| 1 OR 2 OR 3 | |
| Screening [Emtree] | |
| testing | |
| diagnosis [Emtree] | |
| 5 OR 6 OR 7 | |
| Switzerland [Emtree] | |
| Swiss [Emtree] | |
| 8 OR 9 | |
| 4 and 8 and 11 | |
| limit 12 to human | |
| Web of Science | 1. chlamydia * |
| 2. testing * | |
| 3. screening | |
| 4. 2 OR 3 | |
| 5. Switzerland | |
| 6. Swiss | |
| 7. 5 OR 6 | |
| 8. 1 AND 4 AND 7 | |
| 9. limit 8 to human |
Appendix B
| Criteria | Bally, 2015 | Baud, 2011 | Baud, 2008 | Dang, 2009 | Haller, 2011 | Jackson, 2010 | Paget, 2002 | Steiner, 2010 | Wolff, 2008 |
|---|---|---|---|---|---|---|---|---|---|
| yes | yes | yes | yes | yes | yes | yes | yes | yes |
| yes | yes | yes | yes | yes | yes | yes | yes | yes |
| yes | NR | NR | NR | yes | yes | NR | yes | yes |
| yes | yes | yes | yes | yes | yes | yes | yes | no |
| no | no | no | no | yes | no | no | yes | no |
| NA | NA | NA | NA | NA | NA | NA | NA | NA |
| NA | NA | NA | NA | NA | NA | NA | NA | NA |
| NA | NA | NA | NA | NA | NA | NA | NA | NA |
| yes | NA | yes | yes | no | NR | NR | NR | NA |
| NA | NA | NA | NA | NA | NA | NA | NA | NA |
| yes | yes | yes | yes | yes | yes | yes | yes | yes |
| NA | NA | NA | NA | NA | NA | NA | NA | NA |
| no | NA | NA | NA | NA | NA | NA | NA | NR |
| yes | yes | yes | NA | no | yes | yes | yes | yes |
| Score (yes = 1, no = 0) | 0.78 | 0.71 | 0.75 | 0.71 | 0.75 | 0.75 | 0.71 | 0.88 | 0.62 |
| Quality rating (good, fair poor) | good | fair | good | fair | good | good | fair | good | fair |
| NR = not reported, NA = not applicable | |||||||||
References
- Gauly, J.; Ross, J.; Hall, I.; Soda, I.; Atherton, H. Pharmacy-based sexual health services: A systematic review of experiences and attitudes of pharmacy users and pharmacy staff. Sex. Transm. Infect. 2019, 95, 488–495. [Google Scholar] [CrossRef]
- Gonsalves, L.; Hindin, M.J. Pharmacy provision of sexual and reproductive health commodities to young people: A systematic literature review and synthesis of the evidence. Contraception 2017, 95, 339–363. [Google Scholar] [CrossRef]
- Public Health England. National Chlamydia Screening Programme Standards, 7th ed.; Updated November 2018; PHE: London, UK, 2016.
- Kapadia, M.Z. Chlamydia screening in community pharmacies: A systematic literature review of the characteristics of service users and a meta-analysis of chlamydia prevalence. Sex. Health 2013, 10, 1–8. [Google Scholar] [CrossRef]
- Kettle, H.; Cay, S.; Brown, A.; Glasier, A. Screening for Chlamydia trachomatis infection is indicated for women under 30 using emergency contraception. Contraception 2002, 66, 251–253. [Google Scholar] [CrossRef]
- Gudka, S.; Bourdin, A.; Watkins, K.; Eshghabadi, A.; Everett, A.; Clifford, R. Self-reported risk factors for chlamydia: A survey of pharmacy-based emergency contraception consumers. Int. J. Pharm. Pract. 2014, 22, 13–19. [Google Scholar] [CrossRef]
- Wiesenfeld, H.C. Screening for Chlamydia trachomatis Infections in Women. N. Engl. J. Med. 2017, 376, 765–773. [Google Scholar] [CrossRef]
- Unemo, M.; Bradshaw, C.S.; Hocking, J.S.; de Vries, H.J.C.; Francis, S.C.; Mabey, D.; Marrazzo, J.M.; Sonder, G.J.B.; Schwebke, J.R.; Hoornenborg, E.; et al. Sexually transmitted infections: Challenges ahead. Lancet Infect. Dis. 2017, 17, e235–e279. [Google Scholar] [CrossRef]
- Federal Office of Public Health. HIV, Syphilis, Gonorrhea and Chlamydia in Switzerland in 2018: An epidemiological overview. BAG Bull. 2019, 41, 10–36. [Google Scholar]
- Lanjouw, E.; Ouburg, S.; de Vries, H.J.; Stary, A.; Radcliffe, K.; Unemo, M. 2015 European guideline on the management of Chlamydia trachomatis infections. Int. J. STD AIDS 2016, 27, 333–348. [Google Scholar] [CrossRef] [PubMed]
- Geisler, W.; Lensing, S.; Press, C. Spontaneous resolution of genital Chlamydia trachomatis Infection in women and protection from reinfection. J. Infect. Dis. 2013, 207, 1850–1856. [Google Scholar] [CrossRef] [PubMed]
- Workowski, K.A.; Bolan, G.A. Sexually transmitted diseases treatment guidelines, 2015. MMWR Recomm. Rep. 2015, 64, 1–137. [Google Scholar] [PubMed]
- Notter, J. Sexually transmitted infections with Chlamydia trachomatis. Swiss Med. Forum 2017, 11, 705–711. [Google Scholar] [CrossRef]
- Gudka, S.; Afuwape, F.E.; Wong, B.; Yow, X.L.; Anderson, C.; Clifford, R.M. Chlamydia screening interventions from community pharmacies: A systematic review. Sex. Health 2013, 10, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Arnet, I.; Gudka, S.; Salter, S.; Hersberger, K.E.; Clifford, R. Readiness of pharmacists and consumers for pharmacy-based chlamydia screening in Australia and Switzerland. Sex. Reprod. Healthc. 2018. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. J. Clin. Epidemiol. 2009, 62, e1–e34. [Google Scholar] [CrossRef]
- Steiner, A.S.; Haller, D.; Elger, B.S.; Sebo, P.; Gaspoz, J.M.; Wolff, H. Chlamydia trachomatis infection in a Swiss prison: A cross sectional study. Swiss Med. Wkly. 2010, 140, w13126. [Google Scholar] [CrossRef]
- National Heart Lung and Blood Institute. Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies. Available online: https://www.nhlbi.nih.gov/health-pro/guidelines/in-develop/cardiovascular-risk-reduction/tools/cohort (accessed on 2 August 2020).
- Sommer, A.E.; Golden, B.P.; Peterson, J.; Knoten, C.A.; O′Hara, L.; O′Leary, K.J. Hospitalized Patients′ Knowledge of Care: A Systematic Review. J. Gen. Intern. Med. 2018, 33, 2210–2229. [Google Scholar] [CrossRef]
- RStudio Team. RStudio: Integrated Development for R; RStudio, Inc.: Boston, MA, USA, 2016. [Google Scholar]
- Baud, D.; Goy, G.; Jaton, K.; Osterheld, M.-C.; Blumer, S.; Borel, N.; Vial, Y.; Hohlfeld, P.; Pospischil, A.; Greub, G. Role of Chlamydia trachomatis in Miscarriage. Emerg. Infect. Dis. 2011, 17, 1630–1635. [Google Scholar] [CrossRef]
- Paget, W.J.; Zbinden, R.; Ritzler, E.; Zwahlen, M.; Lengeler, C.; StÜrchler, D.; Matter, H.C. Swiss Sentinel Surveillance Network of Gynecologists. National laboratory reports of Chlamydia trachomatis seriously underestimate the frequency of genital chlamydial infections among women in Switzerland. Sex. Transm. Dis. 2002, 29, 715–720. [Google Scholar] [CrossRef][Green Version]
- Baud, D.; Jaton, K.; Bertelli, C.; Kulling, J.P.; Greub, G. Low prevalence of Chlamydia trachomatis infection in asymptomatic young Swiss men. BMC Infect. Dis. 2008, 8, 45. [Google Scholar] [CrossRef]
- Haller, D.M.; Steiner, A.S.; Sebo, P.; Gaspoz, J.M.; Wolff, H. Chlamydia trachomatis infection in males in a juvenile detention facility in Switzerland. Swiss Med. Wkly. 2011, 141. [Google Scholar] [CrossRef] [PubMed]
- Jackson, Y.; Sebo, P.; Aeby, G.; Bovier, P.; Ninet, B.; Schrenzel, J.; Sudre, P.; Haller, D.; Gaspoz, J.M.; Wolff, H. Prevalence and associated factors for Chlamydia trachomatis infection among undocumented immigrants in a primary care facility in Geneva, Switzerland: A cross-sectional study. J. Immigr. Minor. Health 2010, 12, 909–914. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wolff, H.; Lourenço, A.; Bodenmann, P.; Epiney, M.; Uny, M.; Andreoli, N.; Irion, O.; Gaspoz, J.M.; Dubuisson, J.B. Chlamydia trachomatis prevalence in undocumented migrants undergoing voluntary termination of pregnancy: A prospective cohort study. BMC Public Health 2008, 8. [Google Scholar] [CrossRef]
- Bally, F.; Quach, A.; Greub, G.; Jaton, K.; Petignat, C.; Ambord, C.; Fellay, J.; Masserey, E.; Spencer, B. Opportunistic testing for urogenital infection with Chlamydia trachomatis in south-western Switzerland, 2012: A feasibility study. Eurosurveillance 2015, 20. [Google Scholar] [CrossRef]
- Dang, T.; Jaton-Ogay, K.; Flepp, M.; Kovari, H.; Evison, J.M.; Fehr, J.; Schmid, P.; Boffi El Amari, E.; Cavassini, M.; Odorico, M.; et al. High prevalence of anorectal chlamydial infection in HIV-infected men who have sex with men in Switzerland. Clin. Infect. Dis. 2009, 49, 1532–1535. [Google Scholar] [CrossRef]
- Redmond, S.M.; Alexander-Kisslig, K.; Woodhall, S.C.; van den Broek, I.V.; van Bergen, J.; Ward, H.; Uuskula, A.; Herrmann, B.; Andersen, B.; Gotz, H.M.; et al. Genital chlamydia prevalence in Europe and non-European high income countries: Systematic review and meta-analysis. PLoS ONE 2015, 10, e0115753. [Google Scholar] [CrossRef] [PubMed]
- Schmutz, C.; Burki, D.; Frei, R.; Mausezahl-Feuz, M.; Mausezahl, D. Testing for Chlamydia trachomatis: Time trends in positivity rates in the canton of Basel-Stadt, Switzerland. Epidemiol. Infect. 2013, 141, 1953–1964. [Google Scholar] [CrossRef]
- Haag, M.; Gudka, S.; Hersberger, K.E.; Arnet, I.J.; Care, R.H. Do Swiss community pharmacists address the risk of sexually transmitted infections during a consultation on emergency contraception? A simulated patient study. Eur. J. Contracept. Reprod. Health Care 2019, 24, 407–412. [Google Scholar] [CrossRef]
- Papp, J.R.; Schachter, J.; Gaydos, C.A.; Van Der Pol, B.J.M.R.; Morbidity, R.; Recommendations, M.W.R.; Control, R.C.F.D. Recommendations for the laboratory-based detection of Chlamydia trachomatis and Neisseria gonorrhoeae—2014. MMWR Recomm. Rep. 2014, 63, 1. [Google Scholar]
- Centers for Disease Control and Prevention (CDC). Screening tests to detect Chlamydia trachomatis and Neisseria gonorrhoeae infections—2002. MMWR 2002, 51, 1–39. [Google Scholar]
- Huai, P.; Li, F.; Chu, T.; Liu, D.; Liu, J.; Zhang, F. Prevalence of genital Chlamydia trachomatis infection in the general population: A meta-analysis. BMC Infect. Dis. 2020, 20, 589. [Google Scholar] [CrossRef] [PubMed]
- Hocking, J.S.; Temple-Smith, M.; Guy, R.; Donovan, B.; Braat, S.; Law, M.; Gunn, J.; Regan, D.; Vaisey, A.; Bulfone, L.; et al. Population effectiveness of opportunistic chlamydia testing in primary care in Australia: A cluster-randomised controlled trial. Lancet 2018, 392, 1413–1422. [Google Scholar] [CrossRef]
- Nsuami, M.; Cammarata, C.L.; Brooks, B.N.; Taylor, S.N.; Martin, D.H. Chlamydia and Gonorrhea Co-occurrence in a High School Population. Sex. Transm. Dis. 2004, 31, 424–427. [Google Scholar] [CrossRef] [PubMed]
- Catarino, R.; Cherkaoui, A.; Toutous-Trellu, L.; Yaron, M. Who should be screened for Chlamydia trachomatis infection? Three years’ experience at a University Hospital in Switzerland. J. Infect. Dev. Ctries. 2018, 12, 208–2010. [Google Scholar] [CrossRef]
- Centre for Evidence-Based Medicine (CEBM). Oxford Centre for Evidence-Based Medicin-Levels of Evidence. Available online: https://www.cebm.net/2009/06/oxford-centre-evidence-based-medicine-levels-evidence-march-2009/ (accessed on 23 July 2019).
- Council of Europe. Resolution CM/Res(2020)3 on the Implementation of Pharmaceutical Care for the Benefit of Parients and Health Services. Available online: https://search.coe.int/cm/pages/result_details.aspx?objectid=09000016809cdf26 (accessed on 20 March 2020).
| Author, Year | Location | Population; Venue | Sex; Mean Age in Years (S.D.) | Specimen; Testing Method | Main Findings (Study Design, no. of Participants (% of Females if Applicable), Chlamydia Prevalence (%), Response Rate, Authors’ Conclusions) | Quality Assessment |
|---|---|---|---|---|---|---|
| Bally, 2015 [27] | Canton: Vaud, Valais | Sexually active patients < 30 years; centres of the cantonal public sexual health networks (n = 13) and infection disease outpatient clinics (n = 2) | W/M; 21.4 (N.R.) | First-void urine or self-applied low vagina swab for women according to their preferences; NAAT: PCR analysis with a commercial kit (Roche Diagnostics, Switzerland) or an in house validated test | Cross sectional study with 2455 participants (77% females); overall prevalence: 5.5% (95% CI: 4.6–6.4), W: 5.9% (CI: 4.8–7.0); M: 3.9% (CI: 2.3–5.5); response rate: 85%; opportunistic chlamydia testing is feasible and acceptable. | good |
| Baud, 2011 [21] | Lausanne | Women with an acute miscarriage (S.G.) or an uneventful pregnancy (C.G.); attending the emergency gynaecology ward or labour ward at the university hospital | W; S.G.: 33.3 (6.1) C.G: 31.5 (5.0) | Cervicovaginal swab, placenta, and serum; Placenta and cervicovaginal swab: NAAT: TaqMan real-time PCR analysis. Serum: tested for IgG and IgA using the Ridascreen IgG/IgA kit (R-biopharm, Darmstadt, Germany) | Cohort study with 386 participants; S.G.: 4.0% (CI: 1.5–9.6), C.G.: 0.8% (CI: 0.1–3.0); response rate: N.R.; women suffering a miscarriage should be screened for chlamydia to prevent a recurrence. | fair |
| Baud, 2008 [23] | Lausanne | Swiss recruits; the medical entrance examination at the Army recruitment centre | M; 20.6 (1.4) | First-void urine; NAAT: TaqMan real-time PCR analysis and genotyping of positive samples using ompA sequencing | Cross-sectional study with 517 participants; 1.2% (CI: 0.4–2.5); response rate: N.R.; chlamydia prevalence was extremely low in Swiss male recruits. Further research is required. | fair |
| Dang, 2009 [28] | nationwide | HIV-positive MSM enrolled in the SHCS having had unprotected anal intercourse in the past two years or symptoms of proctitis; outpatient clinics | M; median: 42 (N.R.) | Anal swab taken by study physician; NAAT: TaqMan real-time PCR. Genotyping of positive samples using ompA | Cross-sectional study with 147 participants; 10.9% (95% CI: 6.2–17.6); response rate: N.R.; routine anorectal chlamydia screening is suggested if unprotected intercourse is reported. | fair |
| Haller, 2011 [24] | Geneva | Juvenile offenders (>15 years); a juvenile detention facility | M; 16.2 (0.9) | First-void urine; NAAT: PCR analysis using Abott CT/NG reagent (Abott Molecular Diagnostics, Des Plaintes, IL, USA) | Cross-sectional study with 50 participants, 2% (CI: 0.0–6.0); response rate: 85%; results do not support systematic screening. | fair |
| Jackson, 2010 [25] | Geneva | Patients (18–50 years) with no legal residency permit or health insurance; a community Mobile Care Unit | M/W; 32.4 (8.0) | First-void urine; NAAT: PCR analysis using Abott CT/NG reagents (Abott Molecular Diagnostics, Des Plaintes, IL, USA) | Cross-sectional study with 313 participants (78.4% females); overall prevalence: 5.8% (CI: 3.2–8.3), M: 4% (CI: 1.3–10.6), W: 6.5% (CI: 3.7–11); response rate: 82%; access to testing should be ensured for this vulnerable population at risk. | fair |
| Paget, 2002 [22] | nationwide | Patients < 35 years having a first pregnancy consultation (S.G.) or a routine check-up (C.G.), private gynaecology practices | W; S.G. (median): 29 (N.R.) C.G. (median): 27 (N.R.) | Cervical swab; NAAT: LCR assay analysis (LCX, Abott Laboratories, Chicago, IL, USA) | Cross-sectional study with 1589 participants; S.G.: 1.4% (CI: 0.7–2.3), C.G.: 2.9% (CI: 1.8–4.2); response rate: N.R.; prevalence studies are important to assess chlamydia prevalence in addition to official laboratory reports, which underestimate the frequency of infections. | fair |
| Steiner, 2010 [17] | Geneva | Inmates (18–35 years); at a Swiss remand prison | M; 26.4 (6.4) | First-void urine; NAAT: PCR analysis using Abott CT/NG reagents (Abott Molecular Diagnostics, Des Plaintes, IL, USA) | Cross-sectional study with 214 participants; 6.5% (CI: 2.3–9.9); response rate: 80%; inmates are at particular risk for chlamydia infections and widespread surveillance is recommended. | good |
| Wolff, 2008 [26] | Geneva | Undocumented (S.G.) and documented (C.G.) women were undergoing voluntary ToP, the university hospital | W; S.G.: 28 (5.5) C.G.: 28.2 (7.5) | Cervical swab; NAAT: PCR analysis (test not further specified) | Cohort study with 378 participants; S.G.: 12.8% (CI: 8.4–18.9), C.G.: 4.4% (CI: 2.1–8.4); Response rate: S.G. (69%) and C.G.: (85%); Systematic screening among undocumented women undergoing ToP should be considered and access should be ensured. | fair |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haag, M.; Zemp, E.; Hersberger, K.E.; Arnet, I. Who Is Best to Test? A Systematic Review of Chlamydia Infections in Switzerland. Int. J. Environ. Res. Public Health 2020, 17, 9389. https://doi.org/10.3390/ijerph17249389
Haag M, Zemp E, Hersberger KE, Arnet I. Who Is Best to Test? A Systematic Review of Chlamydia Infections in Switzerland. International Journal of Environmental Research and Public Health. 2020; 17(24):9389. https://doi.org/10.3390/ijerph17249389
Chicago/Turabian StyleHaag, Melanie, Elisabeth Zemp, Kurt E. Hersberger, and Isabelle Arnet. 2020. "Who Is Best to Test? A Systematic Review of Chlamydia Infections in Switzerland" International Journal of Environmental Research and Public Health 17, no. 24: 9389. https://doi.org/10.3390/ijerph17249389
APA StyleHaag, M., Zemp, E., Hersberger, K. E., & Arnet, I. (2020). Who Is Best to Test? A Systematic Review of Chlamydia Infections in Switzerland. International Journal of Environmental Research and Public Health, 17(24), 9389. https://doi.org/10.3390/ijerph17249389







