The Toxicity Exerted by the Antibiotic Sulfadiazine on the Growth of Soil Bacterial Communities May Increase over Time
Abstract
1. Introduction
2. Material and Methods
2.1. Experimental Design
2.2. Materials
2.3. Characterization of Soils
2.4. Estimation of Bacterial Community Growth
2.5. Data Analysis and Statistics
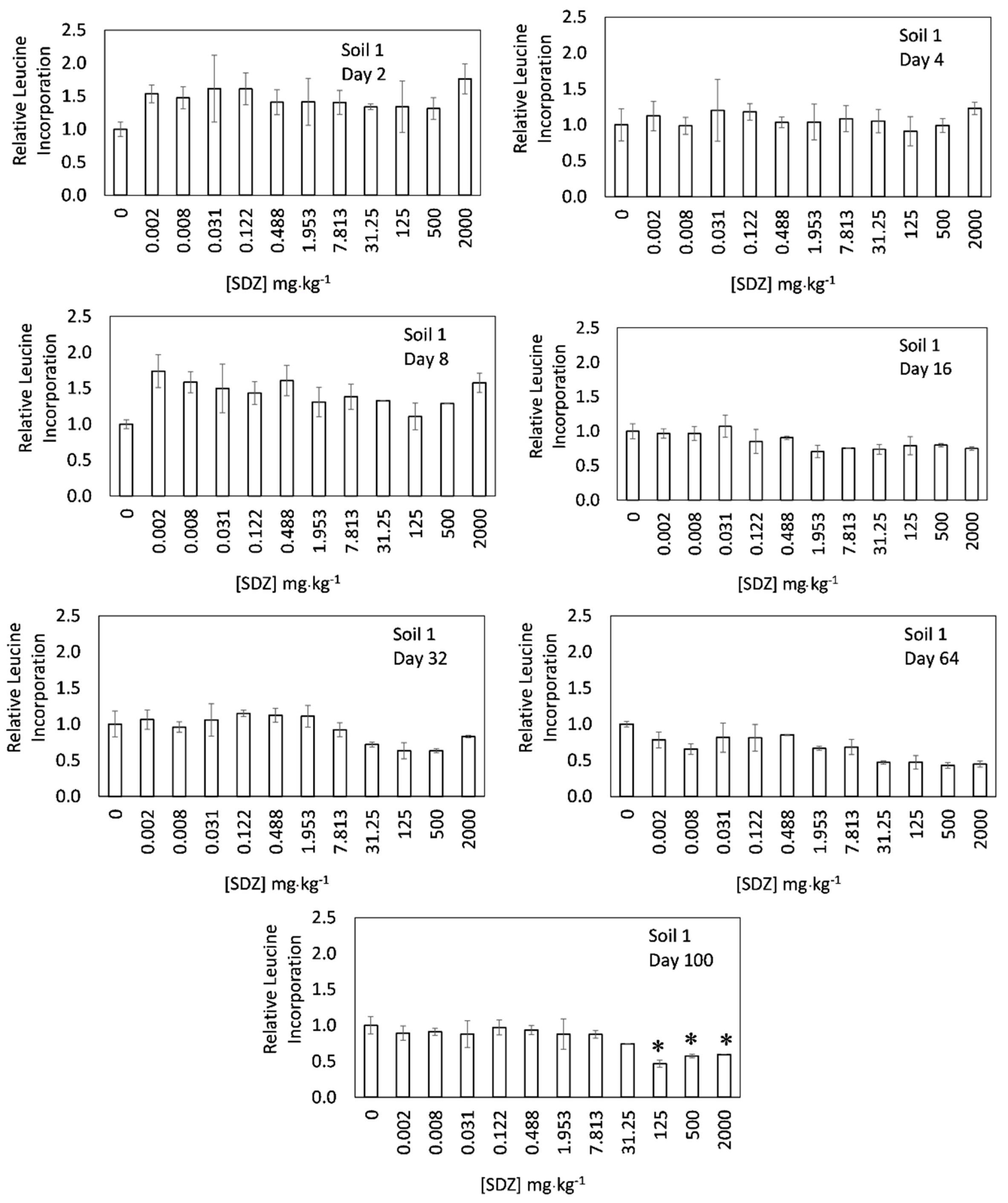
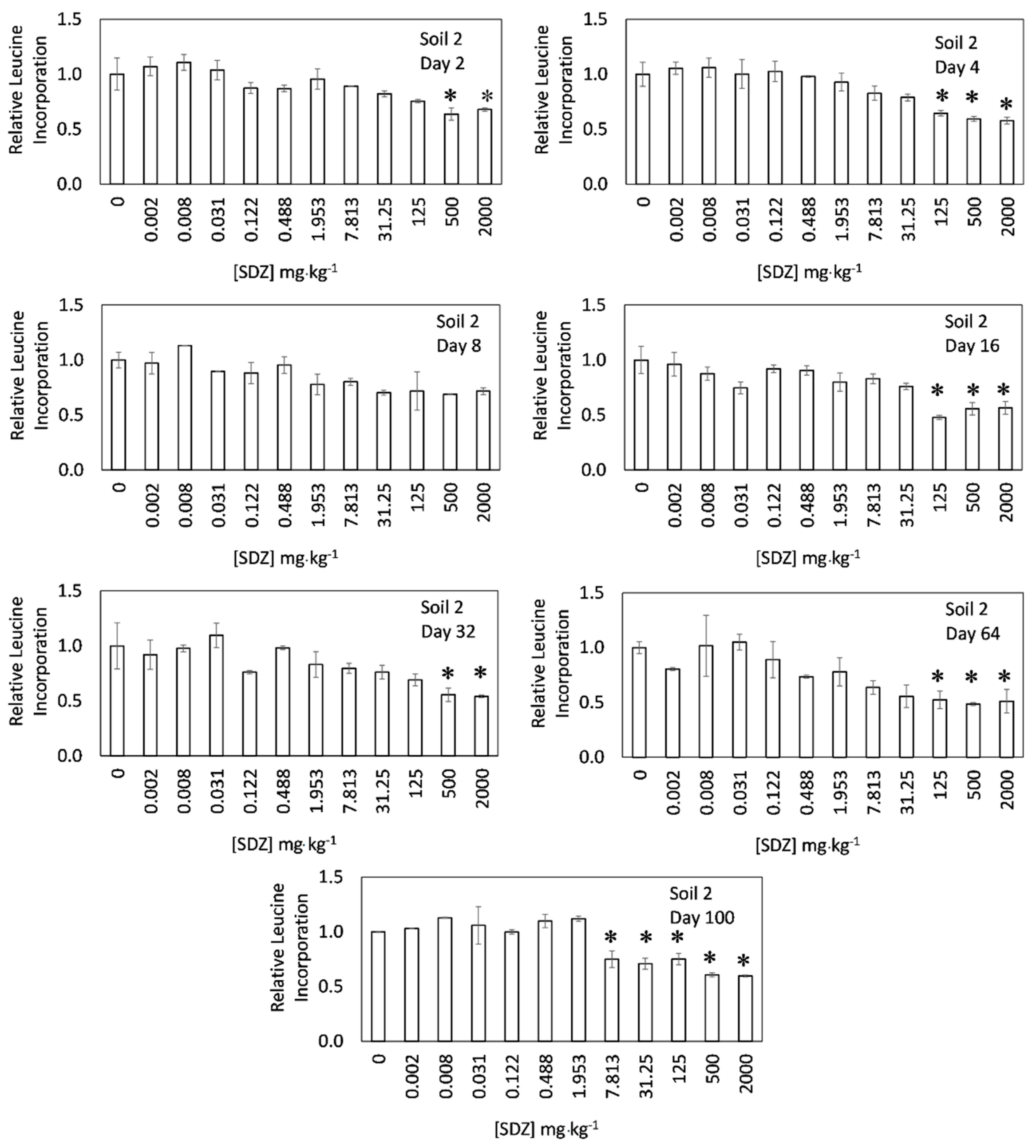
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- European Commission (EC); Directorate-General (DG) XXIV; Consumer Policy and Consumer Health Protection. Opinion of the Scientific Steering Committee on Antimicrobial Resistance; Report of the European Commission, DG XXIV, Consumer Policy and Consumer Health Protection; European Commission: Brussels, Belgium, 1999. [Google Scholar]
- Sarmah, A.K.; Meyer, M.T.; Boxall, A.B. A global perspective on the use, sales, exposure pathways, occurrence, fate and effects of veterinary antibiotics (VAs) in the environment. Chemosphere 2006, 65, 725–759. [Google Scholar] [CrossRef] [PubMed]
- Hamscher, G.; Sczesny, S.; Höper, H.; Nau, H. Determination of persistent tetracycline residues in soil fertilized with liquid manure by high-performance liquid chromatography with electrospray ionization tandem mass spectrometry. Anal. Chem. 2002, 74, 1509–1518. [Google Scholar] [CrossRef] [PubMed]
- Ungemach, F.R. Figures on quantities of antibacterials used for different purposes in the EU countries and interpretation. Acta Vet. Scand. Suppl. 2000, 93, 89–97. [Google Scholar] [PubMed]
- Halling-Sørensen, B.N.N.S.; Nielsen, S.N.; Lanzky, P.F.; Ingerslev, F.; Lützhøft, H.H.; Jørgensen, S.E. Occurrence, fate and effects of pharmaceutical substances in the environment—A review. Chemosphere 1998, 36, 357–393. [Google Scholar] [CrossRef]
- Thiele-Bruhn, S. Pharmaceutical antibiotic compounds in soils—A review. J. Plant Nutr. Soil Sci. 2003, 166, 145–167. [Google Scholar] [CrossRef]
- Ter Laak, T.L.; Gebbink, W.A.; Tolls, J. The effect of pH and ionic strength on the sorption of sulfachloropyridazine, tylosin, and oxytetracycline to soil. Environ. Toxicol. Chem. 2006, 25, 904–911. [Google Scholar] [CrossRef]
- De la Torre, A.; Iglesias, I.; Carballo, M.; Ramírez, P.; Muñoz, M.J. An approach for mapping the vulnerability of European Union soils to antibiotic contamination. Sci. Total Environ. 2012, 414, 672–679. [Google Scholar] [CrossRef]
- Burkhardt, M.; Stamm, C.; Waul, C.; Singer, H.; Müller, S. Surface runoff and transport of sulfonamide antibiotics and tracers on manured grassland. J. Environ. Qual. 2005, 34, 1363–1371. [Google Scholar] [CrossRef]
- Brown, G.M. The biosynthesis of folic acid. 2: Inhibition by sulfonamides. J. Biol. Chem. 1962, 237, 536–540. [Google Scholar]
- Merck. The Merck Index, 13rd ed.; O’Neil, M.J., Smith, A., Heckelmann, P.E., Eds.; Merck: Readington Township, NJ, USA, 2001. [Google Scholar]
- Petrie, W.A. Antimicrobial Agents Sulfonamides, Trimethoprim Sulfamethoxazole, Quinolones, and Agents for Urinary Tract Infections. In Goodman and Gilman’s: The Pharmacological Basis of Therapeutics, 10th ed.; Hardman, J.G., Limbird, L.E., Gilman, A.G., Eds.; McGraw-Hill: New York, NY, USA, 2001; pp. 1171–1188. [Google Scholar]
- Hammesfahr, U.; Bierl, R.; Thiele-Bruhn, S. Combined effects of the antibiotic sulfadiazine and liquid manure on the soil microbial-community structure and functions. J. Plant Nutr. Soil Sci. 2011, 174, 614–623. [Google Scholar] [CrossRef]
- Xu, Y.; Yu, W.; Ma, Q.; Wang, J.; Zhou, H.; Jiang, C. The combined effect of sulfadiazine and copper on soil microbial activity and community structure. Ecotoxicol. Environ. Saf. 2016, 134, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Brandt, K.K.; Sjøholm, O.R.; Krogh, K.A.; Halling-Sørensen, B.; Nybroe, O. Increased pollution-induced bacterial community tolerance to sulfadiazine in soil hotspots amended with artificial root exudates. Environ. Sci. Technol. 2009, 43, 2963–2968. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Mao, D.Q.; Rysz, M.; Zhou, D.X.; Zhang, H.J.; Xu, L.; Alvarez, P.J.J. Trends in antibiotic resistance genes occurrence in the Haihe River, China. Environ. Sci. Technol. 2010, 44, 7220–7225. [Google Scholar] [CrossRef] [PubMed]
- Qiao, M.; Ying, G.-G.; Singer, A.C.; Zhu, Y.-G. Review of antibiotic resistance in China and its environment. Environ. Int. 2018, 110, 160–172. [Google Scholar] [CrossRef] [PubMed]
- Lo, C.-C. Effect of pesticides on soil microbial community. J. Environ. Sci. Health Part B Pestic. Contam. Agric. Wastes 2010, 45, 348–359. [Google Scholar] [CrossRef]
- Zielezny, Y.; Groeneweg, J.; Vereecken, H.; Tappe, W. Impact of sulfadiazine and chlorotetracycline on soil bacterial community structure and respiratory activity. Soil Biol. Biochem. 2006, 38, 2372–2380. [Google Scholar] [CrossRef]
- Hammesfahr, U.; Heuer, H.; Manzke, B.; Smalla, K.; Thiele-Bruhn, S. Impact of the antibiotic sulfadiazine and pig manure on the microbial community structure in agricultural soils. Soil Biol. Biochem. 2008, 40, 1583–1591. [Google Scholar] [CrossRef]
- Cycon, M.; Mrozik, A.; Piotrowska-Seget, Z. Antibiotics in the soil environment—degradation and their impact on microbial activity and diversity. Front. Microbiol. 2019, 10, 338. [Google Scholar] [CrossRef]
- Rousk, J.; Demoling, L.A.; Bååth, E. Contrasting short-term antibiotic effects on respiration and bacterial growth compromises the validity of the selective respiratory inhibition technique to distinguish fungi and bacteria. Microb. Ecol. 2009, 58, 75–85. [Google Scholar] [CrossRef]
- Fernández-Calviño, D.; Rousk, J.; Bååth, E.; Bollmann, U.E.; Bester, K.; Brandt, K.K. Ecotoxicological assessment of propiconazole using soil bacterial and fungal growth assays. Appl. Soil Ecol. 2017, 115, 27–30. [Google Scholar] [CrossRef]
- Fernández-Calviño, D.; Rousk, J.; Brookes, P.C.; Bååth, E. Bacterial pH-optima for growth track soil pH, but are higher than expected at low pH. Soil Biol. Biochem. 2011, 43, 1569–1575. [Google Scholar] [CrossRef]
- Fernández-Calviño, D.; Bååth, E. Interaction between pH and Cu toxicity on fungal and bacterial performance in soil. Soil Biol. Biochem. 2016, 96, 20–29. [Google Scholar] [CrossRef]
- Odukkathil, G.; Vasudevan, N. Toxicity and bioremediation of pesticides in agricultural soil. Rev. Environ. Sci. Bio/Technol. 2013, 12, 421–444. [Google Scholar] [CrossRef]
- Demoling, L.A.; Bååth, E.; Greve, G.; Wouterse, M.; Schmitt, H. Effects of sulfamethoxazole on soil microbial communities after adding substrate. Soil Biol. Biochem. 2009, 41, 840–848. [Google Scholar] [CrossRef]
- Santás-Miguel, V.; Arias-Estévez, M.; Díaz-Raviña, M.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A.; Fernández-Calviño, D. Interactions between soil properties and tetracycline toxicity affecting to bacterial community growth in agricultural soil. Appl. Soil Ecol. 2020, 147, 103437. [Google Scholar] [CrossRef]
- Santás-Miguel, V.; Arias-Estévez, M.; Díaz-Raviña, M.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A.; Fernández-Calviño, D. Effect of oxytetracycline and chlortetracycline on bacterial community growth in agricultural soils. Agronomy 2020, 10, 1011. [Google Scholar] [CrossRef]
- Marsh, J.A.P.; Davies, H.A. Effects of dichlorprop and mecoprop on respiration and transformation of nitrogen in two soils. Bull. Environ. Contam. Toxicol. 1981, 26, 108–115. [Google Scholar] [CrossRef]
- Fernández-Calviño, D.; Rousk, J.; Bååth, E.; Bollmann, U.E.; Bester, K.; Brandt, K.K. Short-term toxicity assessment of a triazine herbicide (terbutryn) underestimates the sensitivity of soil microorganisms. Soil Biol. Biochem. 2020, in press. [Google Scholar]
- Meisner, A.; Bååth, E.; Rousk, J. Microbial growth responses upon rewetting soil dried for four days or one year. Soil Biol. Biochem. 2013, 66, 188–192. [Google Scholar] [CrossRef]
- Rousk, J.; Demoling, L.A.; Bahr, A.; Bååth, E. Examining the fungal and bacterial niche overlap using selective inhibitors in soil. FEMS Microbiol. Ecol. 2008, 63, 350–358. [Google Scholar] [CrossRef]
- Conde-Cid, M.; Álvarez-Esmorís, C.; Paradelo, R.; Nóvoa-Muñoz, J.C.; Arias-Estevez, M.; Álvarez-Rodriguez, E.; Fernández-Sanjurjo, M.J.; Núñez-Delgado, A. Occurrence of tetracyclines and sulfonamides in manures. agricultural soils and crops from different areas in Galicia (NW Spain). J. Clean. Prod. 2018, 197, 491–500. [Google Scholar] [CrossRef]
- Guitián, F.; Carballas, T. Técnicas de Análisis de Suelos; Pico Sacro: Santiago de Compostela, Spain, 1976; ISBN 8485170091, 9788485170098. [Google Scholar]
- Sumner, M.E.; Miller, W.P. Cation exchange capacity and exchange coefficients. In Methods of Soil Analysis—Part 3: Chemical Methods; Soil Science Society of America: Madison, WI, USA, 1996; pp. 1201–1229. [Google Scholar]
- Bertsch, P.M.; Bloom, P.R. Aluminum. In Methods of Soil Analysis—Part 3: Chemical Methods; Soil Science Society of America: Madison, WI, USA, 1996; pp. 517–550. [Google Scholar]
- Olsen, S.R.; Sommers, L.E. Phosphorus. In Methods of Soil Analysis—Part 2: Chemical and Microbiological Properties; Page, A.L., Miller, L.H., Keeney, D.R., Eds.; Soil Science Society of America: Madison, WI, USA, 1996. [Google Scholar]
- Kirchman, D.; K’nees, E.; Hodson, R. Leucine incorporation and its potential as a measure of protein-synthesis by bacteria in natural aquatic systems. Appl. Environ. Microbiol. 1985, 49, 599–607. [Google Scholar] [CrossRef] [PubMed]
- Bååth, E.; Pettersson, M.; Söderberg, K.H. Adaptation of a rapid and economical microcentrifugation method to measure thymidine and leucine incorporation by soil bacteria. Soil Biol. Biochem. 2001, 33, 1571–1574. [Google Scholar] [CrossRef]
- Demoling, L.A.; Bååth, E. No long-term persistence of bacterial pollution-induced community tolerance in tylosin-polluted soil. Environ. Sci. Technol. 2008, 42, 6917–6921. [Google Scholar] [CrossRef] [PubMed]
- Pikkemaat, M.G.; Yassin, H.; van der Fels, K.H.J.; Berendsen, B.J.A. Antibiotic Residues and Resistance in the Environment; RIKILT Report 2016.009; RIKILT Wageningen UR: Wageningen, The Netherlands, 2016. [Google Scholar]
- Ingerslev, F.; Halling-Sørensen, B. Biodegradability properties of sulfonamides in activated sludge. Environ. Toxicol. Chem. 2000, 19, 2467–2473. [Google Scholar] [CrossRef]
- Wang, S.; Wang, H. Adsorption behavior of antibiotic in soil environment: A critical review. Front. Environ. Sci. Eng. 2015, 9, 565–574. [Google Scholar] [CrossRef]
- Chen, M.; Yi, Q.; Hong, J.; Zhang, L.; Lina, K.; Yuana, D. Simultaneous determination of 32 antibiotics and 12 pesticides in sediment using ultrasonic-assisted extraction and high performance liquid chromatography-tandem mass spectrometry. Anal. Methods 2015, 7, 1896. [Google Scholar] [CrossRef]
- Bonfleur, E.J.; Tornisielo, V.L.; Regitano, J.B.; Lavorenti, A. The Effects of Glyphosate and Atrazine Mixture on Soil Microbial Population and Subsequent Impacts on Their Fate in a Tropical Soil. Water Air Soil Pollut. 2015, 226, 21. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhu, L.; Wang, J.; Xie, H.; Wang, J.; Wang, F.; Sun, F. Effects of fomesafen on soil enzyme activity, microbial population, and bacterial community composition. Environ. Monit. Assess. 2014, 186, 2801–2812. [Google Scholar] [CrossRef]
- Kah, M.; Beulke, S.; Brown, C.C. Factors influencing degradation of pesticides in soil. J. Agric. Food Chem. 2007, 55, 4487–4492. [Google Scholar] [CrossRef]
- Hatzinger, P.B.; Alexander, M. Effect of ageing of chemicals in soil on their biodegradability and extractability. Environ. Sci. Technol. 1995, 29, 537–545. [Google Scholar] [CrossRef] [PubMed]
- Blanck, H. A critical review of procedures and approaches used for assessing pollution-induced community tolerance (PICT) in biotic communities. Hum. Ecol. Risk Assess. 2002, 8, 1003–1034. [Google Scholar] [CrossRef]
- Clemens, G.P.; Sisler, H.D. Formation of a fungitoxic derivative from benlate. Phytopathology 1969, 59, 705–706. [Google Scholar]
- Pfeifer, T.; Tuerk, J.; Fuchs, R. Structural Characterization of Sulfadiazine Metabolites Using H/D Exchange Combined with Various MS/MS Experiments. J. Am. Soc. Mass Spectrom. 2005, 16, 1687–1694. [Google Scholar] [CrossRef] [PubMed]
- Schimel, J.; Balser, T.C.; Wallenstein, M. Microbial stress-response physiology and its implications for ecosystem functions. Ecology 2007, 88, 1386–1394. [Google Scholar] [CrossRef]
- OECD. Soil Microorganisms: Nitrogen 194 Transformation Test. In OECD Guideline for the Testing of Chemicals; OECD: Paris, France, 2000. [Google Scholar]
- OECD. Soil Microorganisms: Carbon Transformation Test. In OECD Guideline for the Testing of Chemicals; OECD: Paris, France, 2000. [Google Scholar]
- EFSA Panel on Plant Protection Products and their Residues (PPR); Ockleford, C.; Adriaanse, P.; Berny, P.; Brock, T.; Duquesne, S.; Grilli, S.; Hernandez-Jerez, A.F.; Bennekou, S.H.; Klein, M.; et al. Scientific opinion addressing the state of the science on risk assessment of plant protection products for in-soil organisms. EFSA J. 2017, 15, 4690. [Google Scholar]
- Thiour-Mauprivez, C.; Martin-Laurent, F.; Calvayrac, C.; Barthelmebs, L. Effects of herbicide on non-target microorganisms: Towards a new class of biomarkers? Sci. Total Environ. 2019, 684, 314–325. [Google Scholar] [CrossRef]
| Parameter | Soil | ||
|---|---|---|---|
| 1 | 2 | ||
| Sand (%) | 70 ± 2 | 65 ± 2 | |
| Silt (%) | 12 ± 1 | 13 ± 1 | |
| Clay (%) | 18 ± 1 | 21 ± 2 | |
| Texture | Sandy loam | Sandy clay loam | |
| pHw | 4.8 ± 0.1 | 4.6 ± 0.1 | |
| C (%) | 1.1 ± 0.1 | 3.1 ± 0.2 | |
| N (%) | 0.09 ± 0.01 | 0.25 ± 0.04 | |
| Cae (cmolc kg−1) | 1.53 ± 0.06 | 1.51 ± 0.21 | |
| Mge (cmolc kg−1) | 0.41 ± 0.03 | 0.52 ± 0.02 | |
| Nae (cmolc kg−1) | 0.25 ± 0.03 | 0.21 ± 0.01 | |
| Ke (cmolc kg−1) | 1.27 ± 0.14 | 0.93 ± 0.09 | |
| Ale (cmolc kg−1) | 0.61 ± 0.05 | 2.16 ± 0.07 | |
| eCEC (cmolc kg−1) | 4.08 ± 0.31 | 5.33 ± 0.40 | |
| Pavailable (mg·kg−1) | 225 ± 34 | 169 ± 9 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santás-Miguel, V.; Rodríguez-González, L.; Núñez-Delgado, A.; Díaz-Raviña, M.; Arias-Estévez, M.; Fernández-Calviño, D. The Toxicity Exerted by the Antibiotic Sulfadiazine on the Growth of Soil Bacterial Communities May Increase over Time. Int. J. Environ. Res. Public Health 2020, 17, 8773. https://doi.org/10.3390/ijerph17238773
Santás-Miguel V, Rodríguez-González L, Núñez-Delgado A, Díaz-Raviña M, Arias-Estévez M, Fernández-Calviño D. The Toxicity Exerted by the Antibiotic Sulfadiazine on the Growth of Soil Bacterial Communities May Increase over Time. International Journal of Environmental Research and Public Health. 2020; 17(23):8773. https://doi.org/10.3390/ijerph17238773
Chicago/Turabian StyleSantás-Miguel, Vanesa, Laura Rodríguez-González, Avelino Núñez-Delgado, Montserrat Díaz-Raviña, Manuel Arias-Estévez, and David Fernández-Calviño. 2020. "The Toxicity Exerted by the Antibiotic Sulfadiazine on the Growth of Soil Bacterial Communities May Increase over Time" International Journal of Environmental Research and Public Health 17, no. 23: 8773. https://doi.org/10.3390/ijerph17238773
APA StyleSantás-Miguel, V., Rodríguez-González, L., Núñez-Delgado, A., Díaz-Raviña, M., Arias-Estévez, M., & Fernández-Calviño, D. (2020). The Toxicity Exerted by the Antibiotic Sulfadiazine on the Growth of Soil Bacterial Communities May Increase over Time. International Journal of Environmental Research and Public Health, 17(23), 8773. https://doi.org/10.3390/ijerph17238773









