Effects of an Indoor Cycling Program on Cardiometabolic Factors in Women with Obesity vs. Normal Body Weight
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Participants
2.3. Anthropometric Measurements and Body Composition Assessments
2.4. Cardiorespiratory Fitness
2.5. Blood Analysis
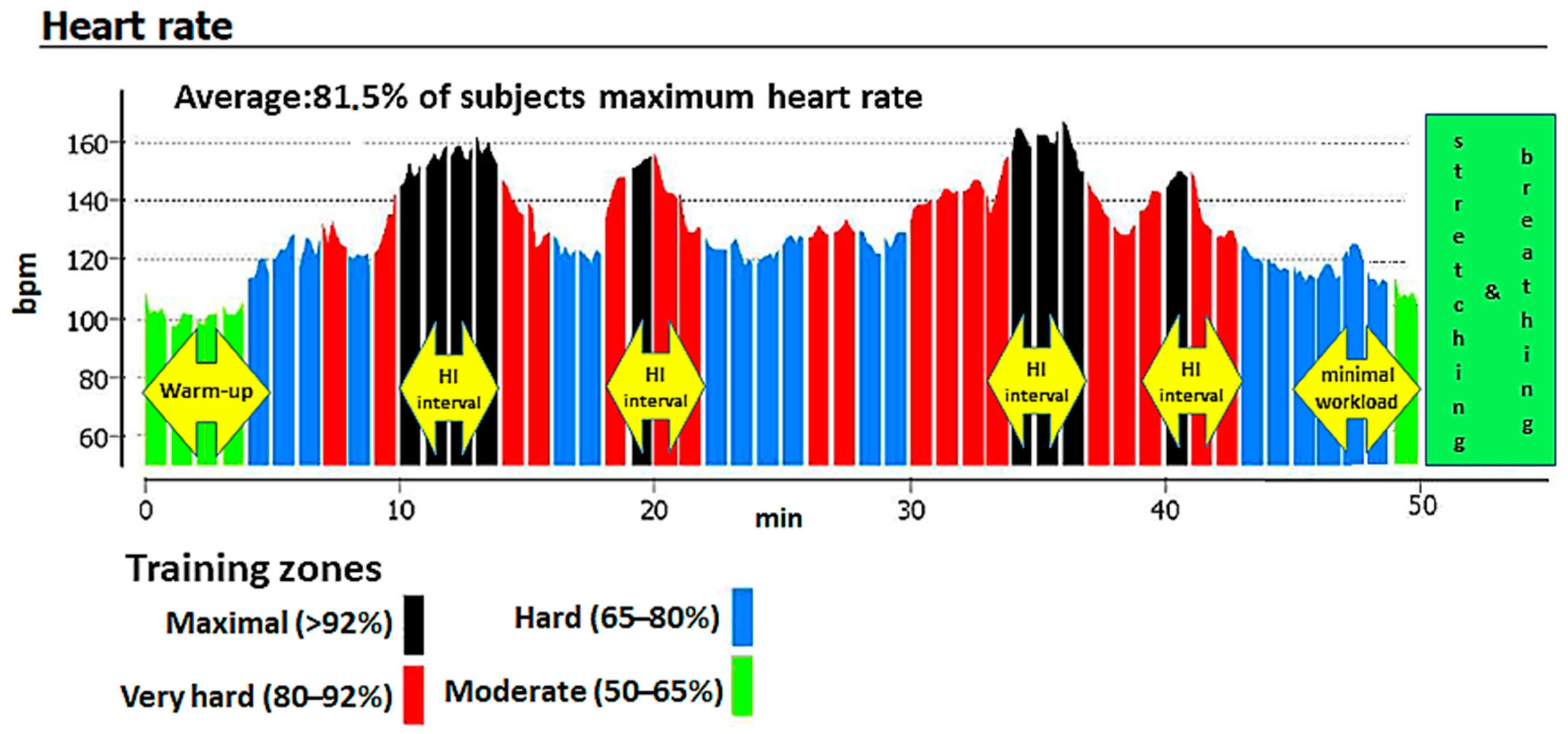
2.6. Exercise Training Protocol
2.7. Statistical Analyses
2.8. Ethical Considerations
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Strasser, B.; Schobersberger, W. Evidence for resistance training as a treatment therapy in obesity. J. Obes. 2011, 2011, 482564. [Google Scholar] [CrossRef]
- Garber, C.E.; Blissmer, B.; Deschenes, M.R.; Franklin, B.A.; Lamonte, M.J.; Lee, I.M.; Nieman, D.C.; Swain, D.P. Quantity and Quality of Exercise for Developing and Maintaining Cardiorespiratory, Musculoskeletal, and Neuromotor Fitness in Apparently Healthy Adults: Guidance for Prescribing Exercise. Med. Sci. Sports Exerc. 2011, 43, 1334–1359. [Google Scholar] [CrossRef]
- Garcia-Hermoso, A.; Cerrillo-Urbina, A.J.; Herrera-Valenzuela, T.; Cristi-Montero, C.; Saavedra, J.M.; Martínez-Vizcaíno, V. Is high-intensity interval training more effective on improving cardiometabolic risk and aerobic capacity than other forms of exercise in overweight and obese youth? A meta-analysis. Obes. Rev. 2016, 17, 531–540. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Fu, J.; Sun, S.; Zhao, G.; Cheng, W.; Dou, C.; Quan, M. Effects of HIIT and MICT on cardiovascular risk factors in adults with overweight and/or obesity: A meta-analysis. PLoS ONE 2019, 14, e0210644. [Google Scholar] [CrossRef] [PubMed]
- Dâmaso, A.R.; Campos, R.M.D.S.; Caranti, D.A.; De Piano, A.; Fisberg, M.; Foschini, D.; Sanches, P.D.L.; Tock, L.; Lederman, H.M.; Tufik, S.; et al. Aerobic plus resistance training was more effective in improving the visceral adiposity, metabolic profile and inflammatory markers than aerobic training in obese adolescents. J. Sports Sci. 2014, 32, 1435–1445. [Google Scholar] [CrossRef] [PubMed]
- Lambers, S.; Van Laethem, C.; Van Acker, K.; Calders, P. Influence of combined exercise training on indices of obesity, diabetes and cardiovascular risk in type 2 diabetes patients. Clin. Rehabil. 2008, 22, 483–492. [Google Scholar] [CrossRef]
- Sandstad, J.; Stensvold, D.; Hoff, M.; Nes, B.M.; Arbo, I.; Bye, A. The effects of high intensity interval training in women with rheumatic disease: A pilot study. Eur. J. Appl. Physiol. 2015, 115, 2081–2089. [Google Scholar] [CrossRef]
- Vilaça-Alves, J.; Bottaro, M.; Santos, C. Energy expenditure combining strength and aerobic training. J. Hum. Kinet. 2011, 29A, 21–25. [Google Scholar] [CrossRef]
- Chavarrias, M.; Carlos-Vivas, J.; Collado-Mateo, D.; Pérez-Gómez, J. Health benefits of indoor cycling: A systematic review. Medicina 2019, 55, 452. [Google Scholar] [CrossRef]
- Caria, M.A.; Tangianu, F.; Concu, A.; Crisafulli, A.; Mameli, O. Quantification of spinning® bike performance during a standard 50-min class. J. Sports Sci. 2007, 25, 421–429. [Google Scholar] [CrossRef]
- Chukhlantseva, N. Effectiveness of an indoor cycling program in improving the physical condition of young women. Pol. J. Sport Tour. 2019, 26, 14–19. [Google Scholar] [CrossRef]
- Bianco, A.; Bellafiore, M.; Battaglia, G.; Paoli, A.; Caramazza, G.; Farina, F.; Palma, A. The effects of indoor cycling training in sedentary overweight women. J. Sports Med. Phys. Fit. 2010, 50, 159–165. [Google Scholar]
- Faulkner, S.H.; Pugh, J.K.; Hood, T.M.; Menon, K.; King, J.A.; Nimmo, M.A. Group studio cycling; an effective intervention to improve cardio-metabolic health in overweight physically inactive individuals. J. Fit. Res. 2015, 4, 16–25. [Google Scholar]
- Kim, J.; Wang, Z.; Heymsfield, S.B.; Baumgartner, R.N.; Gallagher, D. Total-body skeletal muscle mass: Estimation by a new dual-energy X-ray absorptiometry method. Am. J. Clin. Nutr. 2002, 76, 378–383. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Kahn, H.S. The “lipid accumulation product” performs better than the body mass index for recognizing cardiovascular risk: A population-based comparison. BMC Cardiovasc. Disord. 2005, 5, 26. [Google Scholar] [CrossRef] [PubMed]
- Stutts, W.C. Physical activity determinants in adults. Perceived benefits, barriers, and self efficacy. AAOHN J. 2002, 50, 499–507. [Google Scholar] [CrossRef]
- MacInnis, M.J.; Gibala, M.J. Physiological adaptations to interval training and the role of exercise intensity. J. Physiol. 2017, 595, 2915–2930. [Google Scholar] [CrossRef]
- Beals, J.W.; Sukiennik, R.A.; Nallabelli, J.; Emmons, R.S.; Van Vliet, S.; Young, J.R.; Ulanov, A.V.; Li, Z.; Paluska, S.A.; De Lisio, M.; et al. Anabolic sensitivity of postprandial muscle protein synthesis to the ingestion of a protein-dense food is reduced in overweight and obese young adults. Am. J. Clin. Nutr. 2016, 104, 1014–1022. [Google Scholar] [CrossRef]
- Beals, J.W.; Skinner, S.K.; McKenna, C.F.; Poozhikunnel, E.G.; Farooqi, S.A.; Van Vliet, S.; Martinez, I.G.; Ulanov, A.V.; Li, Z.; Paluska, S.A.; et al. Altered anabolic signalling and reduced stimulation of myofibrillar protein synthesis after feeding and resistance exercise in people with obesity. J. Physiol. 2018, 596, 5119–5133. [Google Scholar] [CrossRef]
- Racil, G.; Ben Ounis, O.; Hammouda, O.; Kallel, A.; Zouhal, H.; Chamari, K.; Amri, M. Effects of high vs. moderate exercise intensity during interval training on lipids and adiponectin levels in obese young females. Eur. J. Appl. Physiol. 2013, 113, 2531–2540. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.W.; Konz, E.C. Obesity and disease management: Effects of weight loss on comorbid conditions. Obes. Res. 2001, 9 (Suppl. 4), 326s–334s. [Google Scholar] [CrossRef]
- Buchan, D.; Ollis, S.; Young, J.D.; Thomas, N.E.; Cooper, S.-M.; Tong, T.K.; Nie, J.; Malina, R.M.; Baker, J.S. The effects of time and intensity of exercise on novel and established markers of CVD in adolescent youth. Am. J. Hum. Biol. 2011, 23, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, M.R.; Waclawovsky, G.; Perin, L.; Camboim, I.; Eibel, B.; Lehnen, A.M. Effects of high-intensity interval training on endothelial function, lipid profile, body composition and physical fitness in normal-weight and overweight-obese adolescents: A clinical trial. Physiol. Behav. 2020, 213, 112728. [Google Scholar] [CrossRef] [PubMed]
- Välimäki, I.A.; Vuorimaa, T.; Ahotupa, M.; Vasankari, T.J. Strenuous physical exercise accelerates the lipid peroxide clearing transport by HDL. Eur. J. Appl. Physiol. 2016, 116, 1683–1691. [Google Scholar] [CrossRef] [PubMed]
- Tiainen, S.; Luoto, R.; Ahotupa, M.; Raitanen, J.; Vasankari, T. 6-mo aerobic exercise intervention enhances the lipid peroxide transport function of HDL. Free. Radic. Res. 2016, 50, 1279–1285. [Google Scholar] [CrossRef] [PubMed]
- Leon, A.S.; Sanchez, O.A. Response of blood lipids to exercise training alone or combined with dietary intervention. Med. Sci. Sports Exerc. 2001, 33, S502–S515, discussion S528–S529. [Google Scholar] [CrossRef]
- Nascimento-Ferreira, M.V.; Rendo-Urteaga, T.; Vilanova-Campelo, R.C.; Carvalho, H.B.; Oliveira, G.D.P.; Landim, M.B.P.; Torres-Leal, F.L. The lipid accumulation product is a powerful tool to predict metabolic syndrome in undiagnosed Brazilian adults. Clin. Nutr. 2017, 36, 1693–1700. [Google Scholar] [CrossRef]
- Stein, E.; Silva, I.M.; Dorneles, G.P.; Lira, F.S.; Romão, P.; Peres, A. Reduced fat oxidation during exercise in post-menopausal overweight-obese women with higher lipid accumulation product index. Exp. Clin. Endocrinol. Diabetes 2018, 128, 556–562. [Google Scholar] [CrossRef]
- Owen, M.K.; Noblet, J.N.; Sassoon, D.J.; Conteh, A.M.; Goodwill, A.G.; Tune, J.D. Perivascular adipose tissue and coronary vascular disease. Arter. Thromb. Vasc. Biol. 2014, 34, 1643–1649. [Google Scholar] [CrossRef]
- Bastard, J.-P.; Jardel, C.; Bruckert, E.; Blondy, P.; Capeau, J.; Laville, M.; Vidal, H.; Hainque, B. Elevated levels of interleukin 6 are reduced in serum and subcutaneous adipose tissue of obese women after weight loss. J. Clin. Endocrinol. Metab. 2000, 85, 3338–3342. [Google Scholar] [CrossRef] [PubMed]
- Plotnikoff, R.C.; Eves, N.; Jung, M.; Sigal, R.J.; Padwal, R.S.; Karunamuni, N. Multicomponent, home-based resistance training for obese adults with type 2 diabetes: A randomized controlled trial. Int. J. Obes. 2010, 34, 1733–1741. [Google Scholar] [CrossRef] [PubMed]
- Plaisance, E.P.; Grandjean, P.W. Physical activity and high-sensitivity c-reactive protein. Sports Med. 2006, 36, 443–458. [Google Scholar] [CrossRef] [PubMed]
- Selvin, E.; Paynter, N.P.; Erlinger, T.P. The effect of weight loss on c-reactive protein: A Systematic Review. Arch. Intern. Med. 2007, 167, 31–39. [Google Scholar] [CrossRef]
- Meyers, M.R.; Gokce, N. Endothelial dysfunction in obesity: Etiological role in atherosclerosis. Curr. Opin. Endocrinol. Diabetes Obes. 2007, 14, 365–369. [Google Scholar] [CrossRef]
- Di Francescomarino, S.; Sciartilli, A.; Di Valerio, V.; Di Baldassarre, A.; Gallina, S. The effect of physical exercise on endothelial function. Sports Med. 2009, 39, 797–812. [Google Scholar] [CrossRef]
- Giustarini, D.; Rossi, R.; Milzani, A.; Dalle-Donne, I. Nitrite and nitrate measurement by griess reagent in human plasma: Evaluation of interferences and standardization. Methods Enzymol. 2008, 440, 361–380. [Google Scholar] [CrossRef]
- Tinken, T.M.; Thijssen, D.H.J.; Hopkins, N.; Dawson, E.A.; Cable, N.T.; Green, D.J. Shear stress mediates endothelial adaptations to exercise training in humans. Hypertension 2010, 55, 312–318. [Google Scholar] [CrossRef]
- Ormazabal, V.; Nair, S.; Elfeky, O.; Aguayo, C.; Salomon, C.; Zuñiga, F.A. Association between insulin resistance and the development of cardiovascular disease. Cardiovasc. Diabetol. 2018, 17, 122. [Google Scholar] [CrossRef]
- Bonora, E.; Kiechl, S.; Willeit, J.; Oberhollenzer, F.; Egger, G.; Meigs, J.B.; Bonadonna, R.C.; Muggeo, M. Insulin resistance as estimated by homeostasis model assessment predicts incident symptomatic cardiovascular disease in caucasian subjects from the general population: The bruneck study. Diabetes Care 2007, 30, 318–324. [Google Scholar] [CrossRef]
- Bird, S.R.; Hawley, J.A. Update on the effects of physical activity on insulin sensitivity in humans. BMJ Open Sport Exerc. Med. 2017, 2, e000143. [Google Scholar] [CrossRef] [PubMed]
- Russell, R.D.; Hu, D.; Greenaway, T.; Blackwood, S.J.; Dwyer, R.M.; Sharman, J.E.; Jones, G.; Squibb, K.A.; Brown, A.A.; Otahal, P.; et al. Skeletal muscle microvascular-linked improvements in glycemic control from resistance training in individuals with type 2 diabetes. Diabetes Care 2017, 40, 1256–1263. [Google Scholar] [CrossRef] [PubMed]
- Hamed, N.; Raoof, N. Effect of high intensity interval training on diabetic obese women with polyneuropathy: A randomized controlled clinical trial. Phys. Ther. Rehabil. 2014, 1, 4. [Google Scholar] [CrossRef][Green Version]
- Whyte, L.J.; Gill, J.M.R.; Cathcart, A.J. Effect of 2 weeks of sprint interval training on health-related outcomes in sedentary overweight/obese men. Metabolism 2010, 59, 1421–1428. [Google Scholar] [CrossRef] [PubMed]
- Keshel, T.E. Exercise training and insulin resistance: A current review. J. Obes. Weight. Loss Ther. 2015, 5, S5-003. [Google Scholar] [CrossRef]
| Variables | Normal Weight n = 8 | With Obesity n = 18 | p |
|---|---|---|---|
| Age (years) | 47 ± 5.04 | 51 ± 6.66 | 0.167 |
| Height (m) | 1.65 ± 0.07 | 1.62 ± 0.06 | 0.237 |
| Body mass (kg) | 66.06 ± 5.48 | 95.22 ± 19.6 | 0.000 |
| BMI (kg/m2) | 24.3 ± 1.31 | 36.2 ± 5.64 | 0.000 |
| Before Training | After Training | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Normal Weight Women n = 8 | Women with Obesity n = 18 | NW vs. OW | Normal Weight n = 8 | With Obesity n = 18 | NW vs. OW | ∆NW vs. ∆OW | |||
| Post-Training vs. Baseline | Post-Training vs. Baseline | ||||||||
| p | p | p | p | p | |||||
| Body mass (kg) | 66.06 ± 5.48 | 95.22 ± 19.6 | 0.000 | 64.63 ± 4.35 | 0.093 | 92.98 ± 19.05 | 0.001 | 0.000 | 0.343 |
| BMI (kg/m2) | 24.3 ± 1.31 | 36.2 ± 5.64 | 0.000 | 23.79 ± 1.21 | 0.050 | 35.36 ± 5.52 | 0.001 | 0.000 | 0.275 |
| Waist circumference (cm) | 85.06 ± 5.7 | 111.67 ± 11.5 | 0.000 | 82.06 ± 4.3 | 0.050 | 106.42 ± 12.14 | 0.000 | 0.000 | 0.145 |
| Hip circumference (cm) | 99.13 ± 3.16 | 117.89 ± 12.61 | 0.000 | 97.5 ± 3.3 | 0.039 | 114.31 ±12.67 | 0.000 | 0.000 | 0.043 |
| TBFM (%) | 34.46 ± 4.54 | 47.49 ± 3.53 | 0.000 | 33.44 ± 5 | 0.030 | 45.64 ± 3.51 | 0.000 | 0.000 | 0.927 |
| TBSMM (kg) | 19.89 ± 1.98 | 21.71 ± 2.95 | 0.149 | 20.25 ± 1.93 | 0.462 | 22.27 ± 3.49 | 0.038 | 0.141 | 0.422 |
| VO2 peak [mL/(kg×min)] | 24.73 ± 2.81 | 16.93 ± 2.43 | 0.000 | 29.26 ± 5.22 | 0.002 | 20.59 ± 3.41 | 0.001 | 0.000 | 0.254 |
| VT heart rate (bpm) | 144.63 ± 12.82 | 129.56 ± 16.16 | 0.029 | 146 ± 9.23 | 0.673 | 133.65 ± 14.51 | 0.334 | 0.038 | 0.977 |
| Before Training | After Training | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Normal Weight Women n = 8 | Women with Obesity n = 18 | NW vs. OW | Normal Weight n = 8 | With Obesity n = 18 | NW vs. OW | ∆NW vs. ∆OW | |||
| Post-Training vs. Baseline | Post-Training vs. Baseline | ||||||||
| p | p | p | p | p | |||||
| TC (mmol/L) | 5.84 ± 0.91 | 5.75 ± 1.05 | 0.935 | 5.68 ± 0.5 | 0.401 | 5.36 ± 0.95 | 0.035 | 0.375 | 0.261 |
| HDL-C (mmol/L) | 1.8 ± 0.26 | 1.33 ± 0.39 | 0.005 | 1.82 ± 0.31 | 0.805 | 1.46 ± 0.37 | 0.006 | 0.026 | 0.000 |
| LDL-C (mmol/L) | 3.32 ± 0.61 | 3.45 ± 0.85 | 0.708 | 3.27 ± 0.48 | 0.754 | 3.18 ± 0.90 | 0.139 | 0.780 | 0.397 |
| TG (mmol/L) | 0.77 ± 0.24 | 1.55 ± 0.64 | 0.000 | 0.91 ± 0.25 | 0.097 | 1.6 ± 0.78 | 0.677 | 0.004 | 0.054 |
| OLAb (U/L)* | 10.02 ± 4.93 | 13.95 ± 14.7 | 0.628 | 15.11 ± 13.35 | 0.278 | 9.34 ± 13.5 | 0.008 | 0.066 | 0.031 |
| LAP (cm×mmol/L) | 20.92 ± 7.66 | 84.07 ± 38.9 | 0.000 | 21.73 ± 6.28 | 0.724 | 79.95 ± 54.36 | 0.589 | 0.000 | 0.534 |
| GLUCOSE (mmol/L) | 4.56 ± 0.39 | 4.67 ± 0.61 | 0.656 | 4.67 ± 0.60 | 0.766 | 4.89 ± 0.61 | 0.260 | 0.644 | 0.674 |
| INSULIN (µLU/mL) | 5.75 ± 2.95 | 15.32 ± 7.24 | 0.000 | 8.15 ± 3.91 | 0.120 | 15.57 ± 4.71 | 0.887 | 0.001 | 0.435 |
| HOMA IR | 1.19 ± 0.7 | 3.25 ± 1.73 | 0.000 | 1.67 ± 0.84 | 0.212 | 3.34 ± 0.99 | 0.832 | 0.000 | 0.582 |
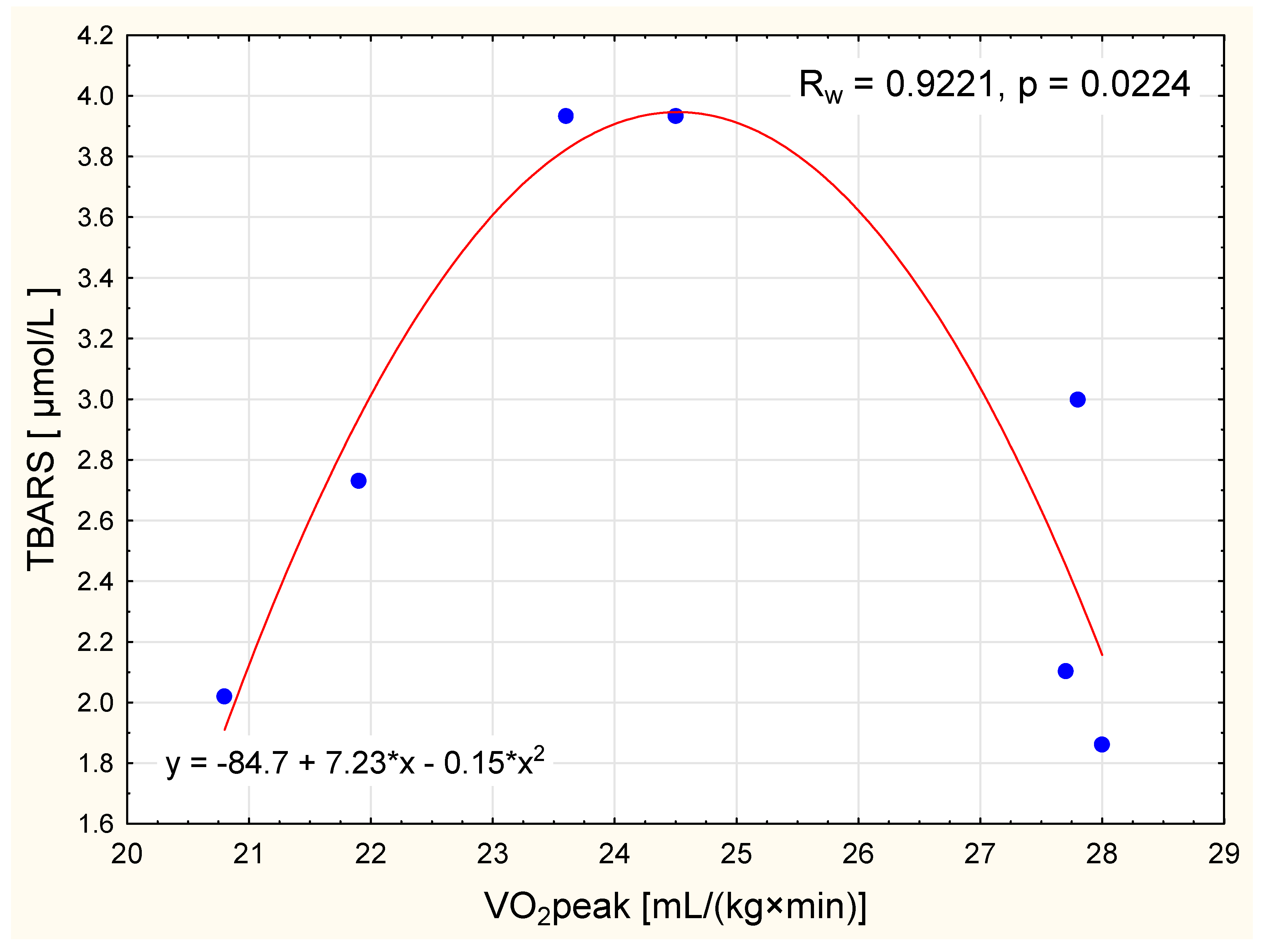
| TBARS (μmol/L) | 2.66 ± 0.89 | 3.05 ± 0.79 | 0.284 | 2.13 ± 0.46 | 0.112 | 2.46 ± 0.67 | 0.016 | 0.226 | 0.884 |
| TAC (mmolCRE·L−1) | 1.15 ± 0.23 | 1.33 ± 0.22 | 0.086 | 1.03 ± 0.23 | 0.208 | 1.35 ± 0.19 | 0.723 | 0.000 | 0.190 |
| eNOS (ng/mL) | 20.98 ± 18.27 | 22.26 ± 23.74 | 0.440 | 24.21 ± 20.32 | 0.123 | 26.13 ± 20.41 | 0.098 | 1.000 | 0.977 |
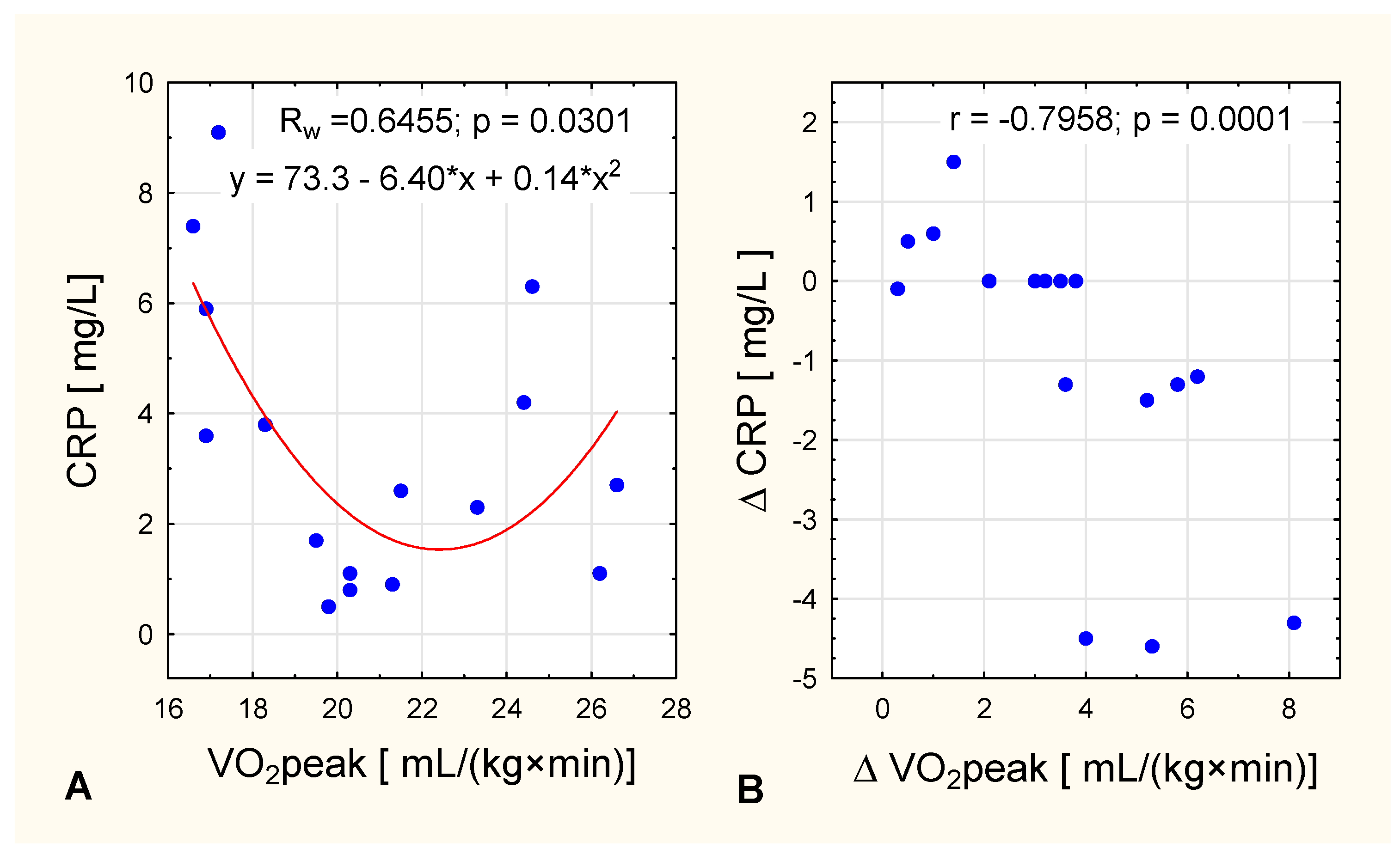
| CRP (mg/L) | 1.2 ± 1.24 | 4.39 ± 2.64 | 0.006 | 1.21 ± 1.19 | 0.463 | 3.37 ± 2.60 | 0.049 | 0.011 | 0.492 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ratajczak, M.; Skrypnik, D.; Krutki, P.; Karolkiewicz, J. Effects of an Indoor Cycling Program on Cardiometabolic Factors in Women with Obesity vs. Normal Body Weight. Int. J. Environ. Res. Public Health 2020, 17, 8718. https://doi.org/10.3390/ijerph17238718
Ratajczak M, Skrypnik D, Krutki P, Karolkiewicz J. Effects of an Indoor Cycling Program on Cardiometabolic Factors in Women with Obesity vs. Normal Body Weight. International Journal of Environmental Research and Public Health. 2020; 17(23):8718. https://doi.org/10.3390/ijerph17238718
Chicago/Turabian StyleRatajczak, Marzena, Damian Skrypnik, Piotr Krutki, and Joanna Karolkiewicz. 2020. "Effects of an Indoor Cycling Program on Cardiometabolic Factors in Women with Obesity vs. Normal Body Weight" International Journal of Environmental Research and Public Health 17, no. 23: 8718. https://doi.org/10.3390/ijerph17238718
APA StyleRatajczak, M., Skrypnik, D., Krutki, P., & Karolkiewicz, J. (2020). Effects of an Indoor Cycling Program on Cardiometabolic Factors in Women with Obesity vs. Normal Body Weight. International Journal of Environmental Research and Public Health, 17(23), 8718. https://doi.org/10.3390/ijerph17238718






