Distribution Characteristics of Indoor PM2.5 Concentration Based on the Water Type and Humidification Method
Abstract
1. Introduction
2. Experimental Methods and Materials
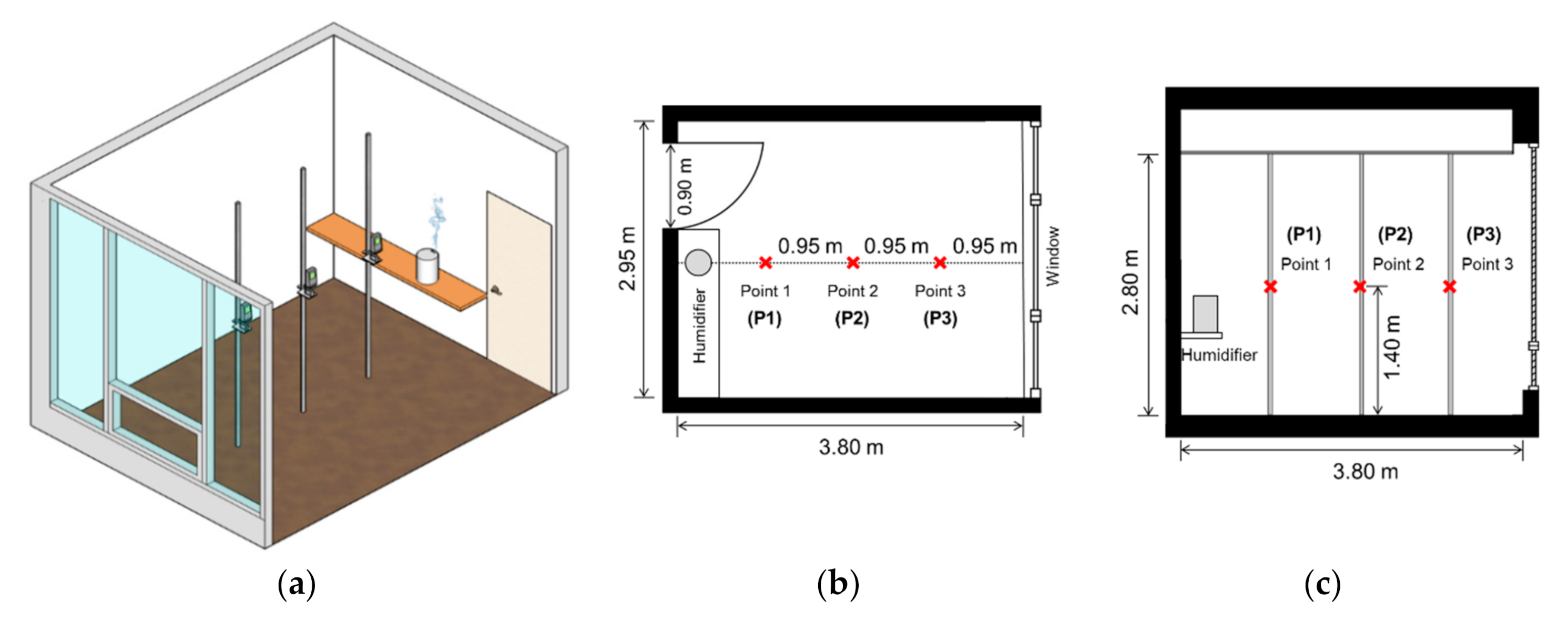
2.1. Experimental Space and Measuring Equipment
2.2. Basic Principle of the Calibration Method
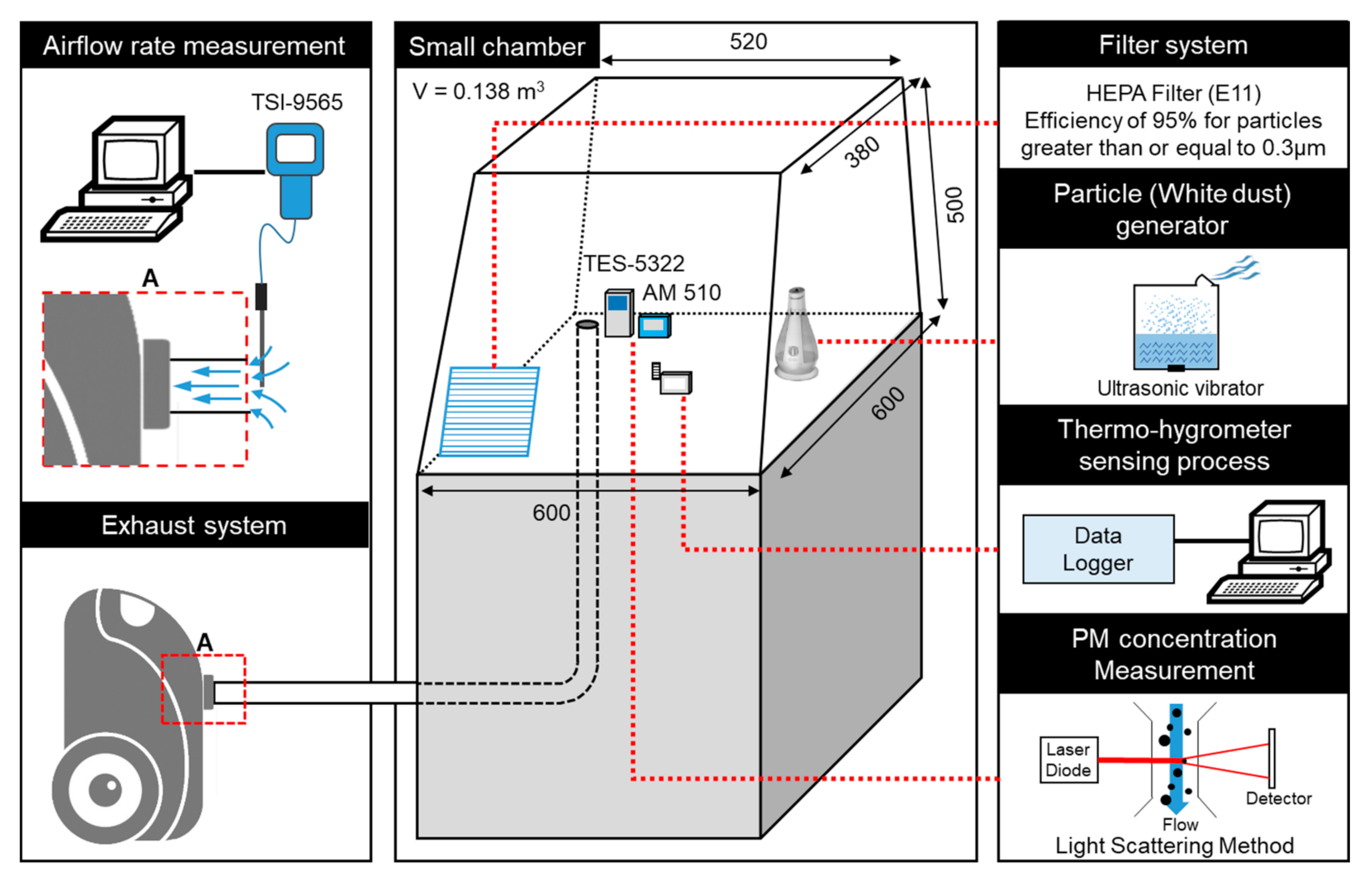
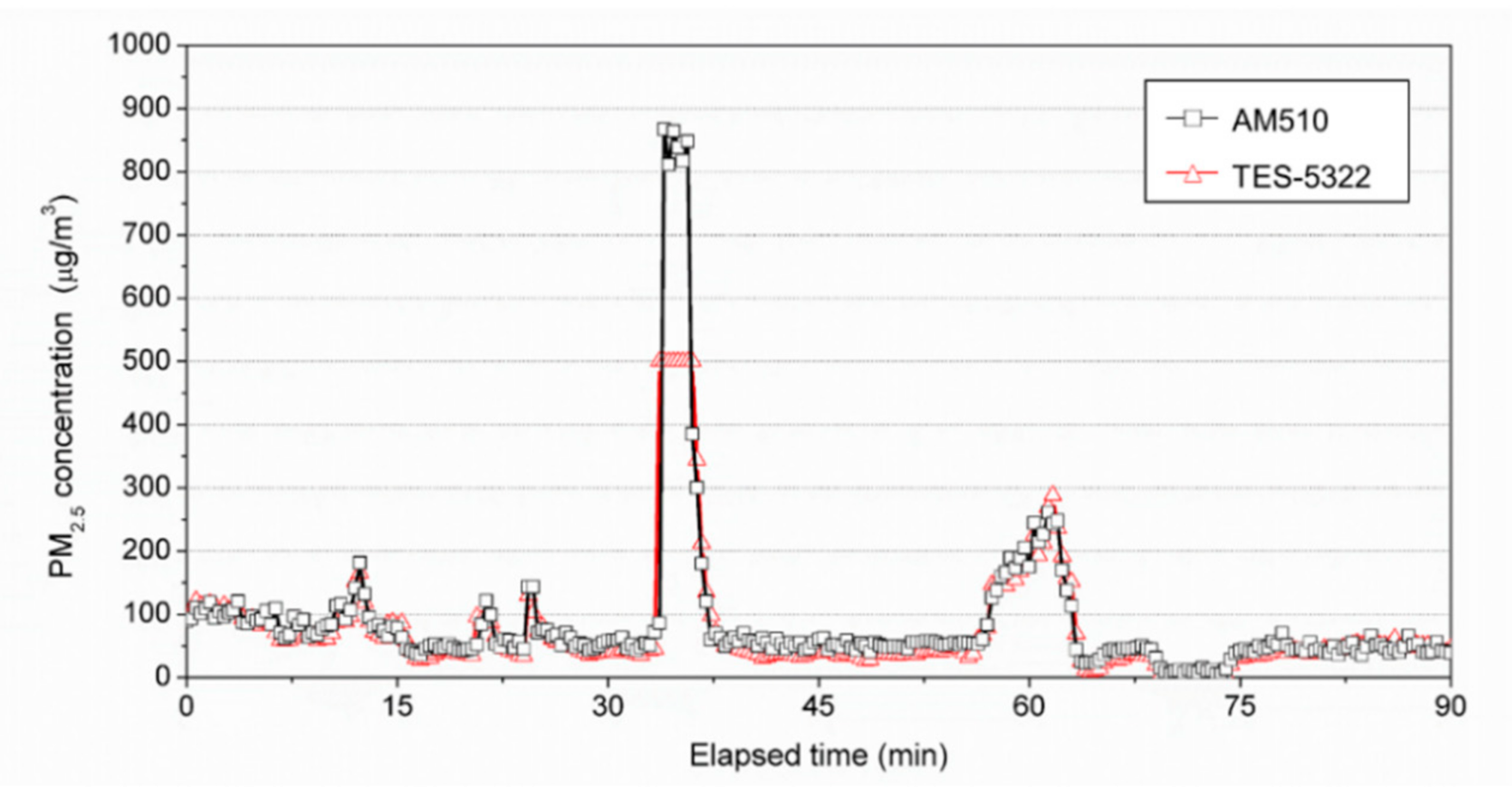
2.3. Calibration Method of the PM Measurements Using a Small Chamber
3. Results and Discussion
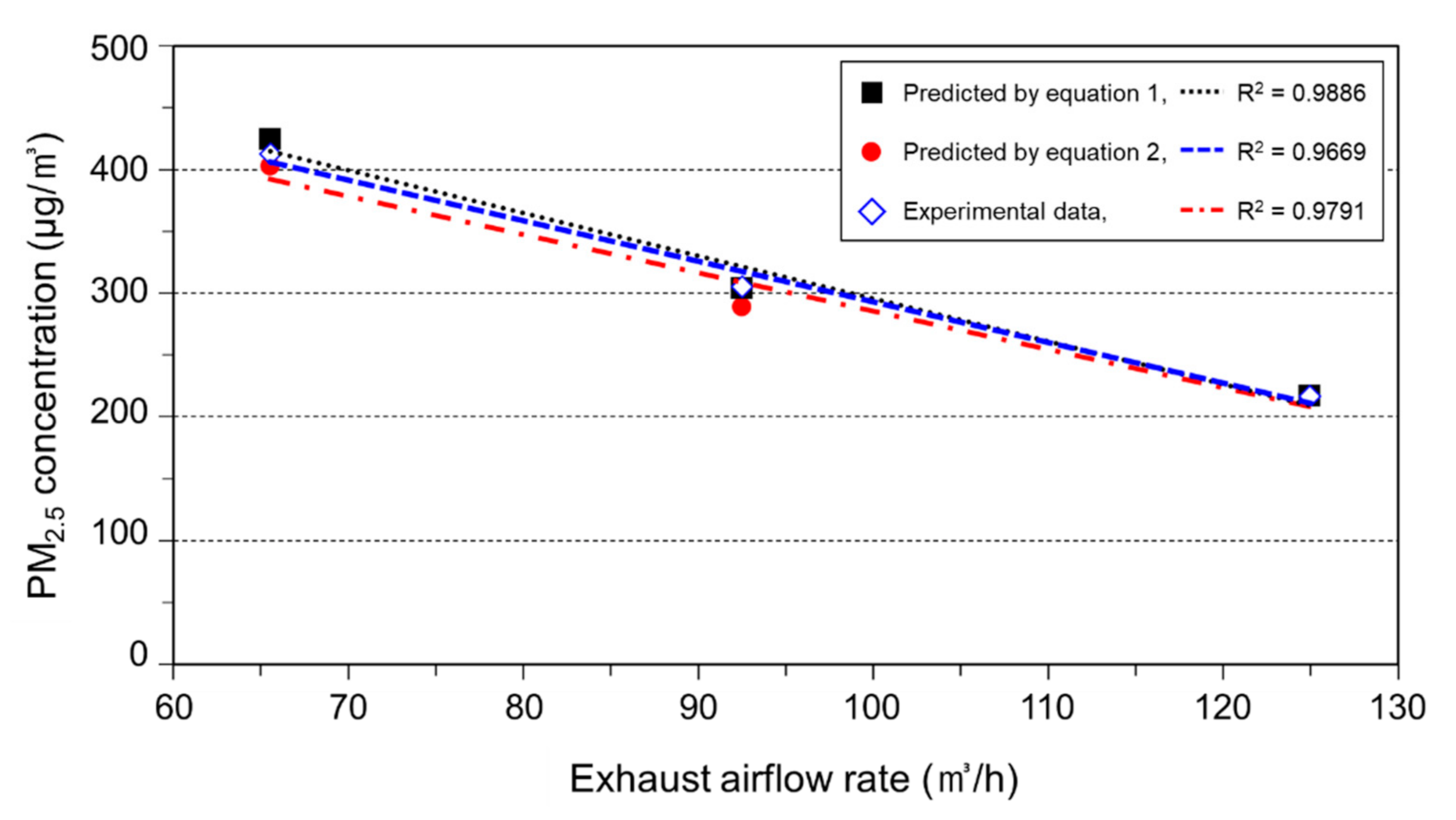
3.1. Calibration Result of the PM Measuring Equipment
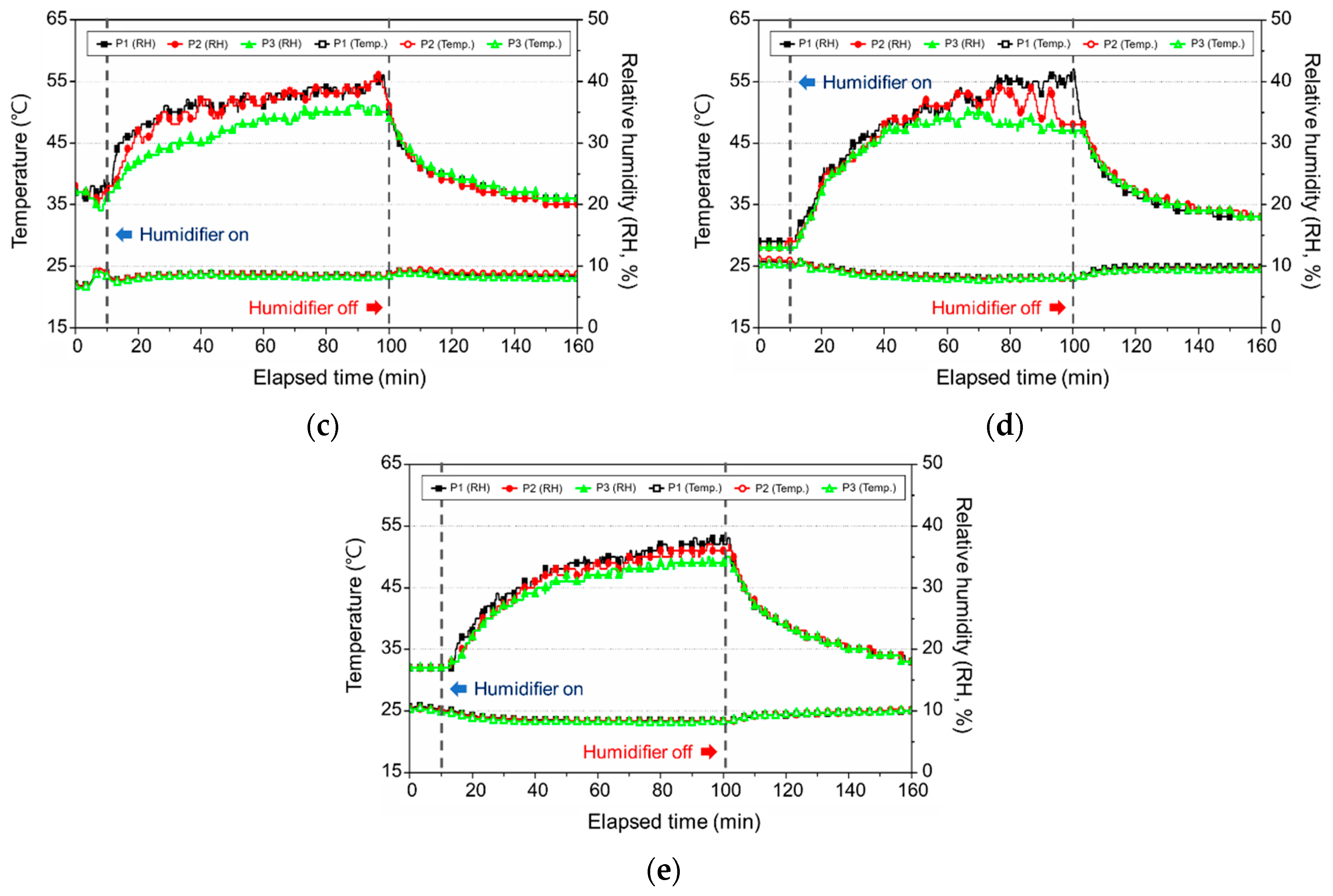
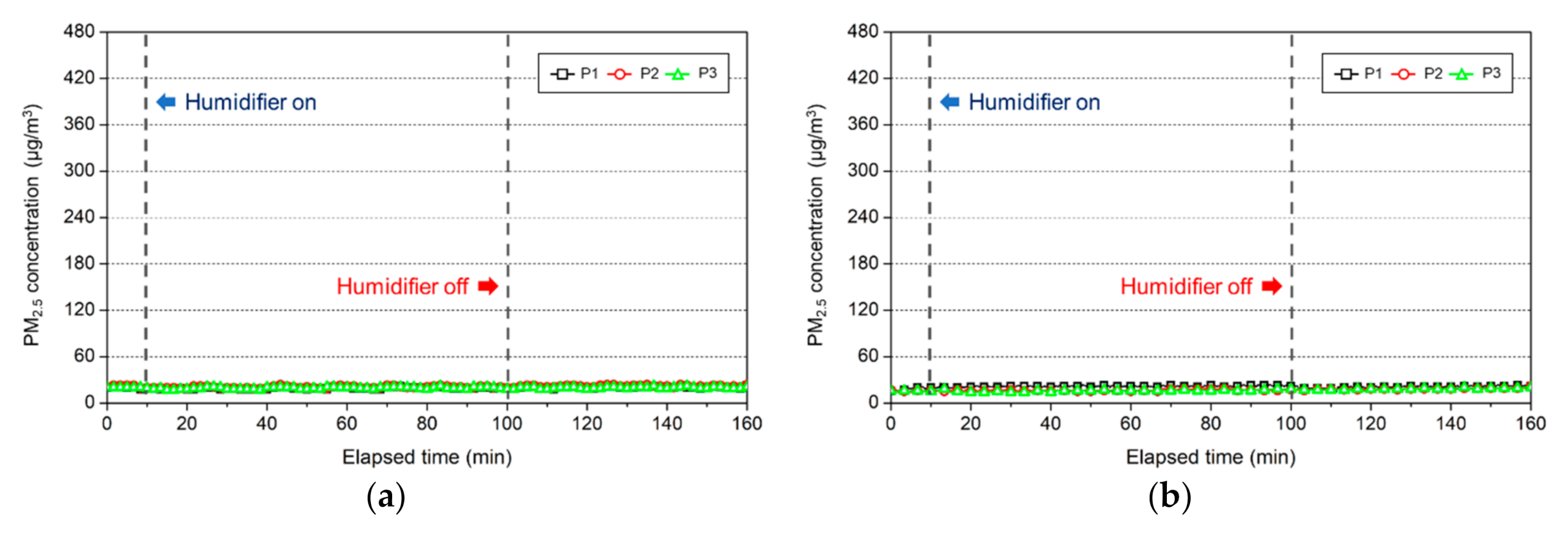
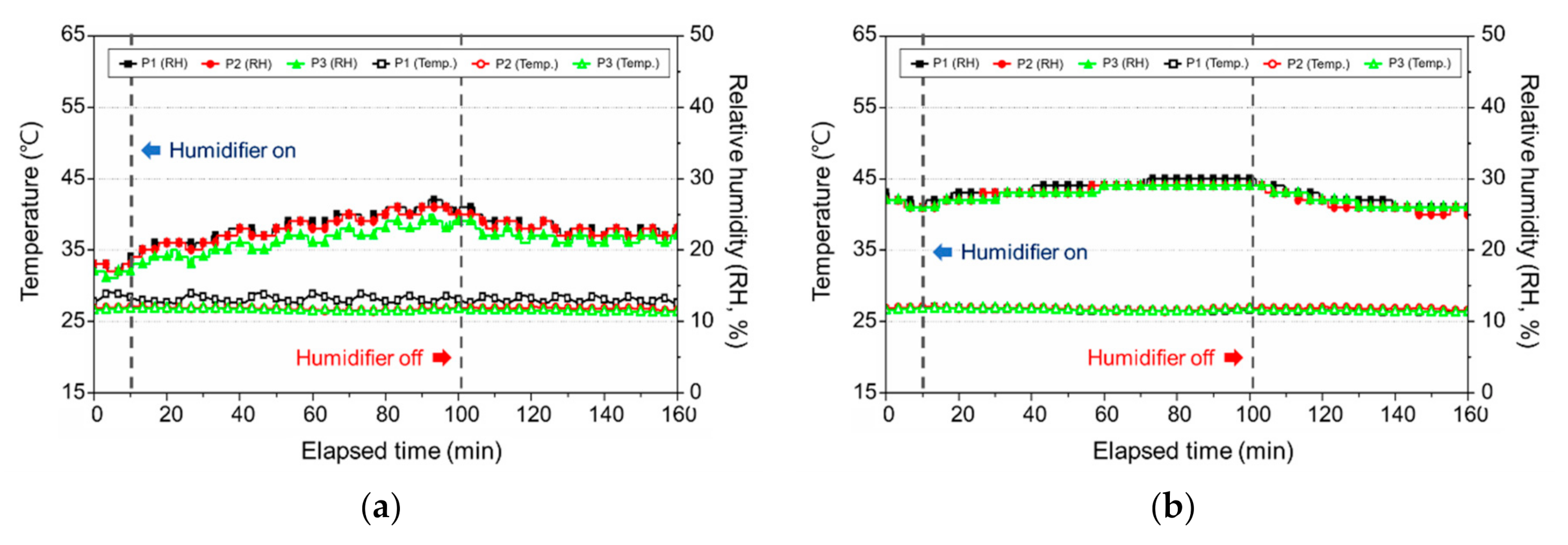
3.2. Change in the Indoor Environment by Using Ultrasonic Humidifiers
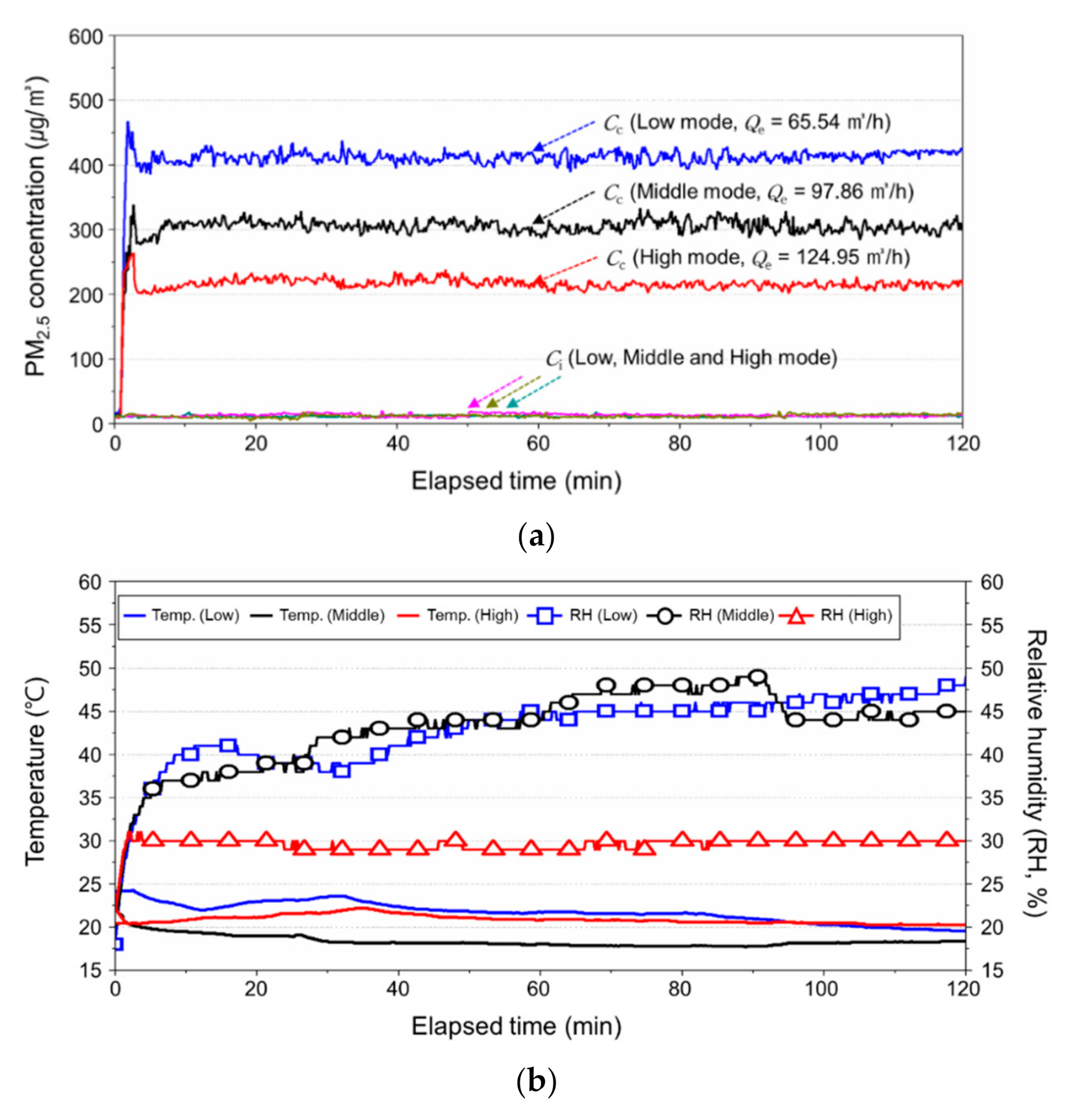
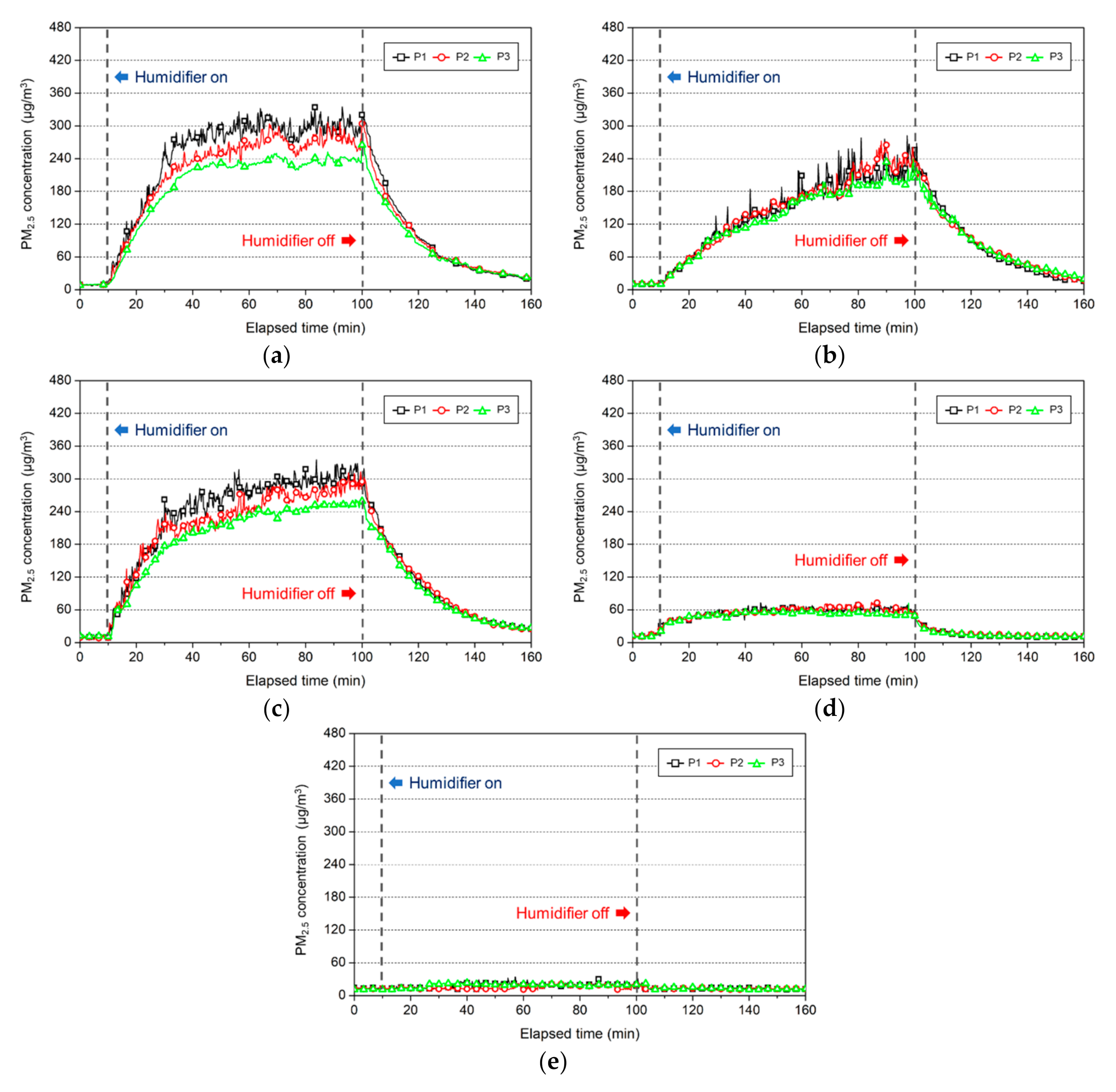
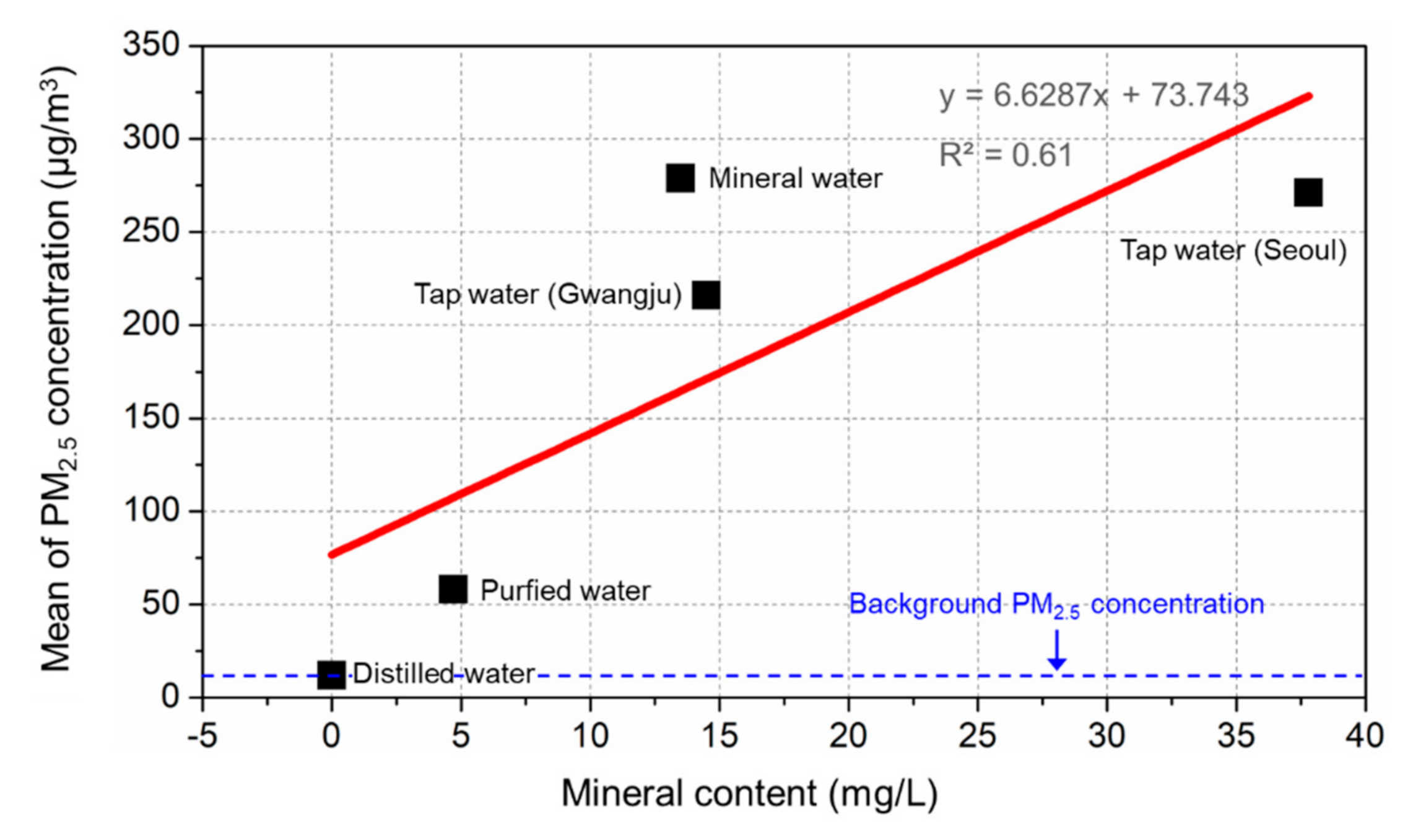
3.2.1. Change in the Indoor PM2.5 Concentration
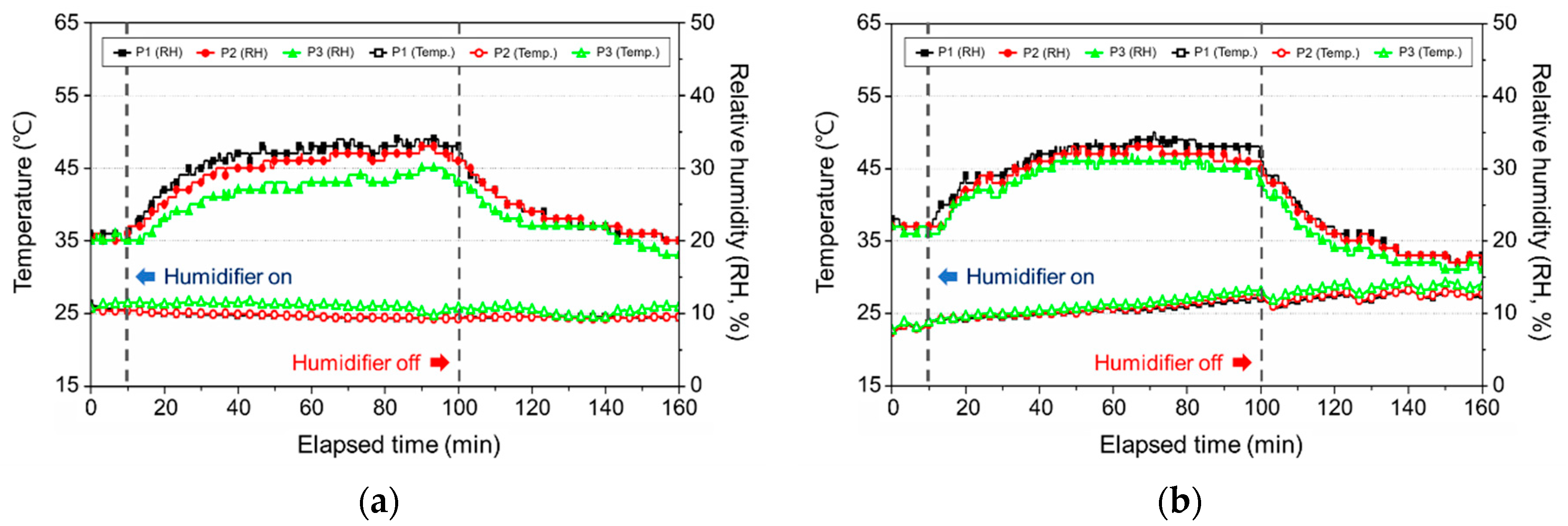
3.2.2. Change in the Temperature and Relative Humidity
3.3. Change in the Indoor Environment by Using a Natural Evaporative Humidifier
3.3.1. Change in the Indoor PM2.5 Concentration
3.3.2. Change in the Temperature and Relative Humidity
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Moreno-Rangel, A.; Sharpe, T.; McGill, G.; Musau, F. Indoor air quality in Passivhaus dwellings: A literature review. Int. J. Environ. Res. Public Health 2020, 17, 4749. [Google Scholar] [CrossRef] [PubMed]
- Cao, T.; Shen, J.; Wang, Q.; Li, H.; Xu, C.; Dong, H. Characteristics of VOCs released from plywood in airtight environments. Forests 2019, 10, 709. [Google Scholar] [CrossRef]
- Tran, V.V.; Park, D.; Lee, Y.-C. Indoor air pollution, related human diseases, and recent trends in the control and improvement of indoor air quality. Int. J. Environ. Res. Public Health 2020, 17, 2927. [Google Scholar] [CrossRef] [PubMed]
- Diapouli, E.; Chaloulakou, A.; Mihalopoulos, N.; Spyrellis, N. Indoor and outdoor PM mass and number concentrations at schools in the Athens area. Environ. Monit. Assess. 2008, 136, 13–20. [Google Scholar] [CrossRef]
- Choi, D.S.; Youn, J.-S.; Lee, I.H.; Park, Y.-K.; Choi, B.J.; Jeon, K.-J. Analysis of national PM2.5 (FPM and CPM) emissions by past, current, and future energy mix scenarios in the Republic of Korea. Sustainability 2019, 11, 4289. [Google Scholar] [CrossRef]
- Lu, C.M.; Dat, N.D.; Lien, C.K.; Chi, K.H.; Chang, M.B. Characteristics of fine particulate matter and Polycyclic Aromatic Hydrocarbons emitted from coal combustion processes. Energy Fuels 2019, 33, 10247–10254. [Google Scholar] [CrossRef]
- Yang, H.-H.; Arafath, S.M.; Lee, K.-T.; Hsieh, Y.-S.; Han, Y.-T. Chemical characteristics of filterable and condensable PM2.5 emissions from industrial boilers with five different fuels. Fuel 2018, 232, 415–422. [Google Scholar] [CrossRef]
- Martins, N.R.; da Graça, G.C. Impact of outdoor PM2.5 on natural ventilation usability in California’s nondomestic buildings. Appl. Energy 2017, 189, 711–724. [Google Scholar] [CrossRef]
- Urso, P.; Cattaneo, A.; Garramone, G.; Peruzzo, C.; Cavallo, D.M.; Carrer, P. Identification of particulate matter determinants in residential homes. Build. Environ. 2015, 86, 61–69. [Google Scholar] [CrossRef]
- Clements, N.; Keady, P.; Emerson, J.B.; Fierer, N.; Miller, S.L. Seasonal variability of airborne particulate matter and bacterial concentrations in Colorado homes. Atmosphere 2018, 9, 133. [Google Scholar] [CrossRef]
- Stabile, L.; De Luca, G.; Pacitto, A.; Morawska, L.; Avino, P.; Buonanno, G. Ultrafine particle emission from floor cleaning products. Indoor Air 2020. [Google Scholar] [CrossRef] [PubMed]
- Daftary, A.S.; Deterding, R.R. Inhalational lung injury associated with humidifier “white dust”. Pediatrics 2011, 127, e509–e512. [Google Scholar] [CrossRef] [PubMed]
- Yao, W.; Dal Porto, R.; Gallagher, D.L.; Dietrich, A.M. Human exposure to particles at the air-water interface: Influence of water quality on indoor air quality from use of ultrasonic humidifiers. Environ. Int. 2020, 143, 105902. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Park, S.; Jo, H.; Song, S.; Ham, S.; Yoon, C. Behavioral characteristics of polyhexamethyleneguanidine (PHMG) particles in aqueous solution and air when sprayed into an ultrasonic humidifier. Environ. Res. 2020, 182, 109078. [Google Scholar] [CrossRef]
- Sain, A.; Zook, J.; Davy, B.; Marr, L.; Dietrich, A. Size and mineral composition of airborne particles generated by an ultrasonic humidifier. Indoor Air 2018, 28, 80–88. [Google Scholar] [CrossRef]
- Cheong, H.-K.; Ha, M.; Lee, J.-H. Unrecognized bomb hidden in the babies’ room: Fatal pulmonary damage related with use of biocide in humidifiers. Environ. Health Toxicol. 2012, 27, e2012001. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency. Indoor Air Facts No. 8: Use and Care of Home Humidifiers; EPA: Washington, DC, USA, 1991.
- Highsmith, V.R.; Hardy, R.J.; Costa, D.L.; Germani, M.S. Physical and chemical characterization of indoor aerosols resulting from the use of tap water in portable home humidifiers. Environ. Sci. Technol. 1992, 26, 673–680. [Google Scholar] [CrossRef]
- Umezawa, M.; Sekita, K.; Suzuki, K.-I.; Kubo-Irie, M.; Niki, R.; Ihara, T.; Sugamata, M.; Takeda, K. Effect of aerosol particles generated by ultrasonic humidifiers on the lung in mouse. Part. Fibre Toxicol. 2013, 10, 64. [Google Scholar] [CrossRef]
- Nogueira, R.; Melo, N.; Bastos, H.N.E.; Martins, N.; Delgado, L.; Morais, A.; Mota, P. Hypersensitivity pneumonitis: Antigen diversity and disease implications. Pulmonology 2019, 25, 97–108. [Google Scholar] [CrossRef]
- Morawska, L.; Afshari, A.; Bae, G.; Buonanno, G.; Chao, C.; Hänninen, O.; Hofmann, W.; Isaxon, C.; Jayaratne, E.; Pasanen, P. Indoor aerosols: From personal exposure to risk assessment. Indoor Air 2013, 23, 462–487. [Google Scholar] [CrossRef]
- Highsmith, V.R.; Rodes, C.E.; Hardy, R.J. Indoor particle concentrations associated with use of tap water in portable humidifiers. Environ. Sci. Technol. 1988, 22, 1109–1112. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-T.; Liang, C.-C.; Chien, T.-Y.; Wu, F.-J.; Fan, K.-C.; Wan, G.-H. Effect of ventilation rate on air cleanliness and energy consumption in operation rooms at rest. Environ. Monit. Assess. 2018, 190, 178. [Google Scholar] [CrossRef] [PubMed]
- Sousan, S.; Koehler, K.; Thomas, G.; Park, J.H.; Hillman, M.; Halterman, A.; Peters, T.M. Inter-comparison of low-cost sensors for measuring the mass concentration of occupational aerosols. Aerosol Sci. Technol. 2016, 50, 462–473. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Park, S.; Seo, J. Evaluation on air purifier’s performance in reducing the concentration of fine particulate matter for occupants according to its operation methods. Int. J. Environ. Res. Public Health 2020, 17, 5561. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zhao, B. Reducing human exposure to PM2.5 generated while cooking typical Chinese cuisine. Build. Environ. 2020, 168, 106522. [Google Scholar] [CrossRef]
- Huang, Y.; Bao, M.; Xiao, J.; Qiu, Z.; Wu, K. Effects of PM2.5 on cardio-pulmonary function injury in open manganese mine workers. Int. J. Environ. Res. Public Health 2020, 16, 2017. [Google Scholar] [CrossRef] [PubMed]
- Kallawicha, K.; Chao, H.J.; Kotchasatan, N. Bioaerosol levels and the indoor air quality of laboratories in Bangkok metropolis. Aerobiologia 2019, 35, 1–14. [Google Scholar] [CrossRef]
- Hyland, A.; Travers, M.J.; Dresler, C.; Higbee, C.; Cummings, K.M. A 32-country comparison of tobacco smoke derived particle levels in indoor public places. Tob. Control. 2008, 17, 159–165. [Google Scholar] [CrossRef]
- Soule, E.K.; Maloney, S.F.; Spindle, T.R.; Rudy, A.K.; Hiler, M.M.; Cobb, C.O. Electronic cigarette use and indoor air quality in a natural setting. Tob. Control. 2017, 26, 109–112. [Google Scholar] [CrossRef]
- Agency, U.S.E.P. ENERGY STAR Market & Industry Scoping Report-Residential Humidifiers. 2012. Available online: https://www.energystar.gov/sites/default/files/asset/document/ENERGY_STAR_Scoping_Report_Residential_Humidifiers.pdf (accessed on 19 November 2020).


| Parameter | AM510 | TES-5322 | |
|---|---|---|---|
| Measurement | 90° light scattering | Light scattering | |
| Sensor type | 670 nm laser diode | - | |
| Particle size range (μm) | 0.1 to 10 | 0.1 to 2.5 | |
| Concentration range (μg/m3) | 0.1 to 20,000 | 0.1 to 500 | |
| Accuracy | ≤50 μg/m3 | - | ±5 μg/m3 |
| >50 μg/m3 | - | 10% | |
| Operational condition | Temperature (°C) | 0 to 50 | 0 to 60 |
| RH (%) | 0 to 95 | 0 to 95 | |
| Capacity | Approximately N = 31,000 | MicroSD card 4 GB | |
| Price (USD, $) | Approximately 3800 | Approximately 400 | |
| Parameter | Value | |
|---|---|---|
| Smartmi Air Humidifier | Smartmi Air Humidifier 2 | |
| Type | Ultrasonic | Natural evaporative |
| Capacity (L/h) | 0.355 | 0.24 |
| Tank capacity (L) | 3.5 | 4 |
| Noise level (dB) | Approximately 40.4 | Approximately 34.3 |
| Energy use (W) | 38 | 8 |
| Wireless technology | 802.11 b/g/n, 2.4 GHz | |
| Water Type | Calcium (Ca) | Sodium (Na) | Potassium (K) | Magnesium (Mg) | Lead (Pb) |
|---|---|---|---|---|---|
| Tap water (Seoul) | 20.1 | 2.8 | 3.8 | 11.1 | None |
| Tap water (Gwangju) | 7.3 | 1.6 | 1.9 | 3.7 | None |
| Mineral water | 3.2 | 2.5 | 2.8 | 5.0 | None |
| Purified water | 1.8 | 0.5 | 0.4 | 2.0 | None |
| Distilled water | None | None | None | None | None |
| Component | Model | Parameter | Value |
|---|---|---|---|
| Airflow Measurement | TSI 9565 | Range (m/s) | 0 to 50 |
| Accuracy (m/s) | ±0.015 | ||
| PM measurement | AM510/TES-5322 | Table 1 | |
| Temperature and RH measurement | TR-72WF | Range (°C and %) | 0 to 55 and 10 to 95 |
| Accuracy (°C and %) | ±0.5 and ±5 | ||
| Resolution (°C and %) | 0.1 and 1 | ||
| Exhaust system | WD 5 P | Energy use (W) | 1100 |
| Container capacity (L) | 25 | ||
| Dimensions (mm) | 418 × 382 × 652 | ||
| Filter | GFAH-1005 | Efficiency (%) | 98 (0.5 to 1.0 μm) |
| Size (mm) | 175 × 170 | ||
| Particle generator | DP-2030UH | Humidification type | Ultrasonic |
| Capacity (L/h) | 0.18 | ||
| Tank size (L) | 1 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, S.; Seo, J.; Lee, S. Distribution Characteristics of Indoor PM2.5 Concentration Based on the Water Type and Humidification Method. Int. J. Environ. Res. Public Health 2020, 17, 8638. https://doi.org/10.3390/ijerph17228638
Park S, Seo J, Lee S. Distribution Characteristics of Indoor PM2.5 Concentration Based on the Water Type and Humidification Method. International Journal of Environmental Research and Public Health. 2020; 17(22):8638. https://doi.org/10.3390/ijerph17228638
Chicago/Turabian StylePark, Seonghyun, Janghoo Seo, and Sunwoo Lee. 2020. "Distribution Characteristics of Indoor PM2.5 Concentration Based on the Water Type and Humidification Method" International Journal of Environmental Research and Public Health 17, no. 22: 8638. https://doi.org/10.3390/ijerph17228638
APA StylePark, S., Seo, J., & Lee, S. (2020). Distribution Characteristics of Indoor PM2.5 Concentration Based on the Water Type and Humidification Method. International Journal of Environmental Research and Public Health, 17(22), 8638. https://doi.org/10.3390/ijerph17228638





