Global Prevalence of Macroprolactinemia among Patients with Hyperprolactinemia: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Protocol and Registration
2.2. Data Sources and Search Strategies
2.3. Study Eligibility
2.4. Data Extraction
2.5. Quality Assessment
2.6. Statistical Analyses
3. Results
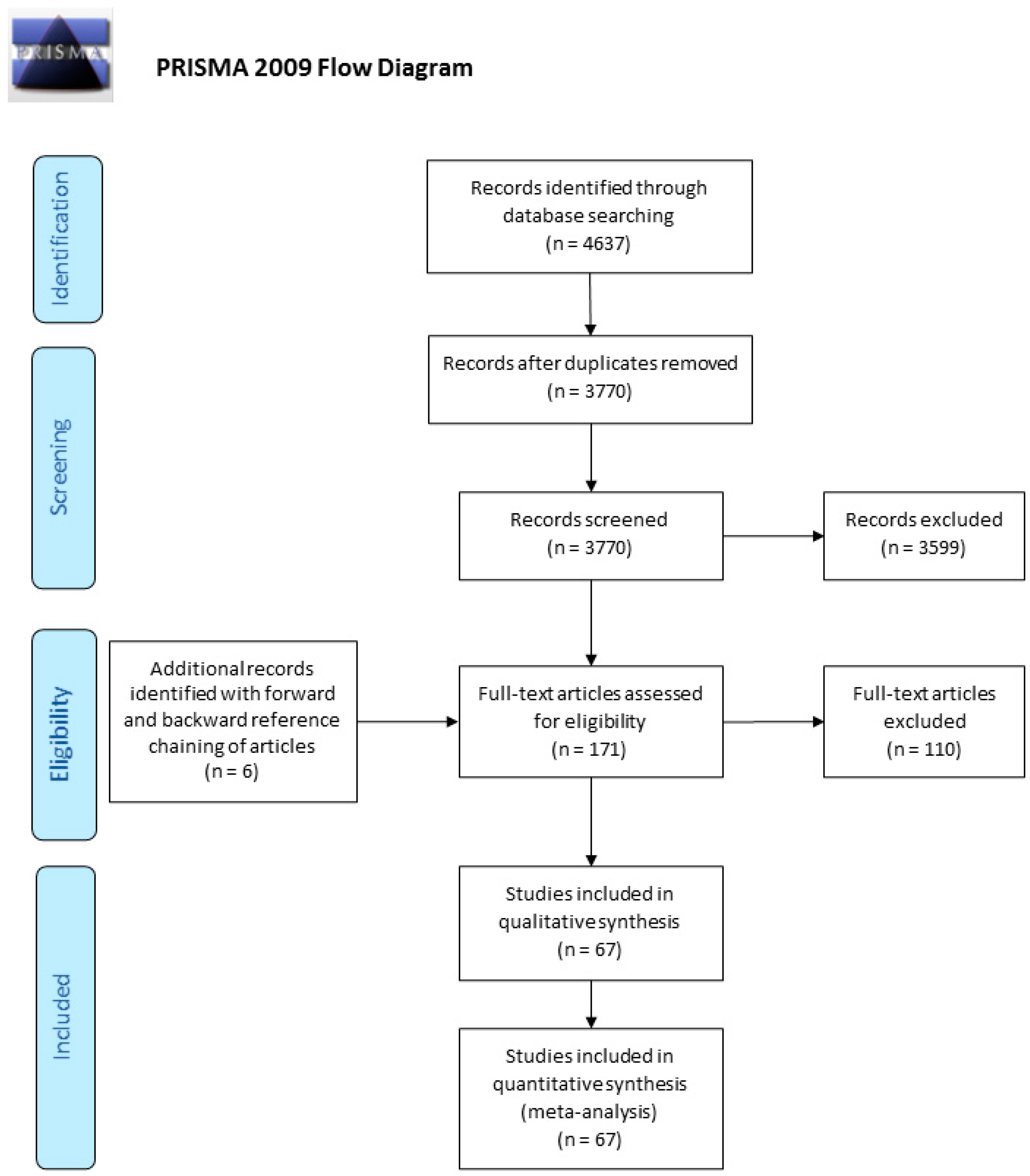
3.1. Study Selection and Characteristics
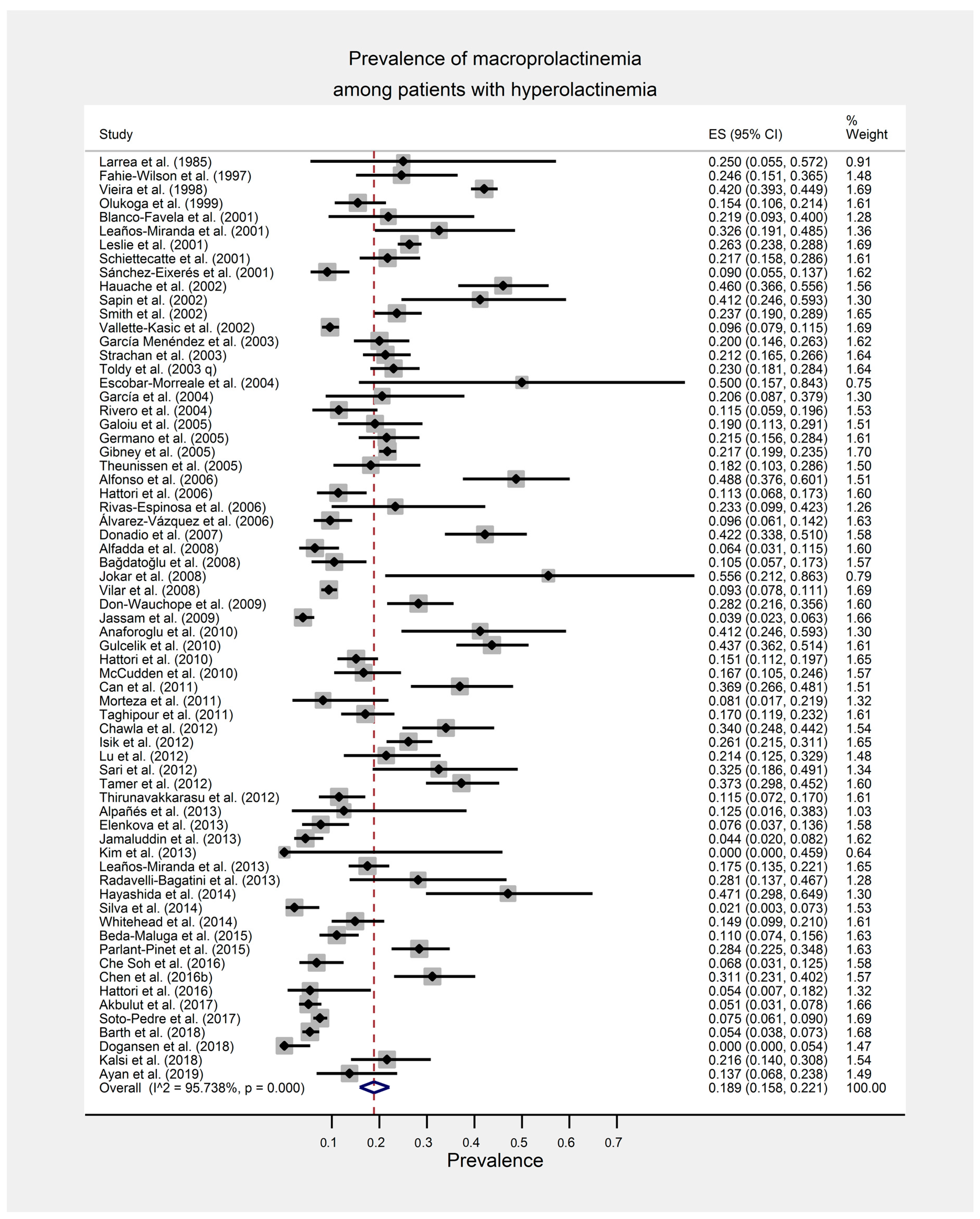
3.2. Prevalence of Macroprolactinemia among Patients with Hyperprolactinemia
3.3. Quality Assessment and Publication Bias
3.4. Subgroup and Meta-Regression Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Delitala, G. Hyperprolactinaemia: Causes, biochemical diagnosis and tests of prolactin secretion. In Clinical Endocrinology; Blackwell Science Ltd.: London, UK, 1998; pp. 138–147. [Google Scholar]
- Melmed, S.; Kleinberg, D. Anterior pituitary. Williams Textb. Endocrinol. 2003, 11, 155–261. [Google Scholar]
- Kasum, M.; Oreskovic, S.; Zec, I.; Jezek, D.; Tomic, V.; Gall, V.; Adzic, G. Macroprolactinemia: New insights in hyperprolactinemia. Biochem. Med. 2012, 22, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Kasum, M.; Orešković, S.; Čehić, E.; Šunj, M.; Lila, A.; Ejubović, E. Laboratory and clinical significance of macroprolactinemia in women with hyperprolactinemia. Taiwan J. Obstet. Gynecol. 2017, 56, 719–724. [Google Scholar] [CrossRef] [PubMed]
- Hattori, N.; Ikekubo, K.; Nakaya, Y.; Kitagawa, K.; Inagaki, C. Immunoglobulin G Subclasses and Prolactin (PRL) Isoforms in Macroprolactinemia Due to Anti-PRL Autoantibodies. J. Clin. Endocrinol. Metab. 2005, 90, 3036–3044. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kavanagh-Wright, L.; Smith, T.P.; Gibney, J.; McKenna, T.J. Characterization of macroprolactin and assessment of markers of autoimmunity in macroprolactinaemic patients. Clin. Endocrinol. 2009, 70, 599–605. [Google Scholar] [CrossRef]
- Vaishya, R.; Gupta, R.; Arora, S. Macroprolactin; A Frequent Cause of Misdiagnosed Hyperprolactinemia in Clinical Practice. J. Reprod. Infertil. 2010, 11, 161–167. [Google Scholar]
- Bonhoff, A.; Vuille, J.-C.; Gomez, F.; Gellersen, B. Identification of macroprolactin in a patient with asymptomatic hyperprolactinemia as a stable PRL-IgG complex. Exp. Clin. Endocrinol. Diabetes 1995, 103, 252–255. [Google Scholar] [CrossRef] [PubMed]
- Hattori, N.; Ishihara, T.; Saiki, Y.; Shimatsu, A. Macroprolactinaemia in patients with hyperprolactinaemia: Composition of macroprolactin and stability during long-term follow-up. Clin. Endocrinol. 2010, 73, 792–797. [Google Scholar] [CrossRef] [PubMed]
- Can, M.; Guven, B.; Atmaca, H.; Acıkgoz, S.; Mungan, G. Clinical characterization of patients with macroprolactinemia and monomeric hyperprolactinemia. Kaohsiung J. Med. Sci. 2011, 27, 173–176. [Google Scholar] [CrossRef]
- McCudden, C.R.; Sharpless, J.L.; Grenache, D.G. Comparison of multiple methods for identification of hyperprolactinemia in the presence of macroprolactin. Clin. Chim. Acta 2010, 411, 155–160. [Google Scholar] [CrossRef]
- Schneider, W.; Marcovitz, S.; Al-Shammari, S.; Yago, S.; Chevalier, S. Reactivity of macroprolactin in common automated immunoassays. Clin. Biochem. 2001, 34, 469–473. [Google Scholar] [CrossRef]
- Cavaco, B.; Prazeres, S.; Santos, M.; Sobrinho, L.; Leite, V. Hyperprolactinemia due to big big prolactin is differently detected by commercially available immunoassays. J. Endocrinol. Investig. 1999, 22, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Fahie-Wilson, M.N. Detection of Macroprolactin Causing Hyperprolactinemia in Commercial Assays for Prolactin. Clin. Chem. 2000, 46, 2022–2023. [Google Scholar] [CrossRef] [PubMed]
- Beltran, L.; Fahie-Wilson, M.N.; McKenna, T.J.; Kavanagh, L.; Smith, T.P. Serum Total Prolactin and Monomeric Prolactin Reference Intervals Determined by Precipitation with Polyethylene Glycol: Evaluation and Validation on Common ImmunoAssay Platforms. Clin. Chem. 2008, 54, 1673–1681. [Google Scholar] [CrossRef]
- McKenna, T.J. Should macroprolactin be measured in all hyperprolactinaemic sera? Clin. Endocrinol. 2009, 71, 466–469. [Google Scholar] [CrossRef]
- Suliman, A.M.; Smith, T.P.; Gibney, J.; McKenna, T.J. Frequent Misdiagnosis and Mismanagement of Hyperprolactinemic Patients before the Introduction of Macroprolactin Screening: Application of a New Strict Laboratory Definition of Macroprolactinemia. Clin. Chem. 2003, 49, 1504–1509. [Google Scholar] [CrossRef]
- Gibney, J.; Smith, T.; McKenna, T. The Impact on Clinical Practice of Routine Screening for Macroprolactin. J. Clin. Endocrinol. Metab. 2005, 90, 3927–3932. [Google Scholar] [CrossRef]
- Schiettecatte, J.; De Schepper, J.; Velkeniers, B.; Smitz, J.; Van Steirteghem, A. Rapid Detection of Macroprolactin in tHe Form of Prolactin-Immunoglobulin G Complexes by Immunoprecipitation with Anti-human IgG-Agarose. Clin. Chem. Lab. Med. 2001, 39, 1244–1248. [Google Scholar] [CrossRef]
- Prazeres, S.; Santos, M.A.; Ferreira, H.G.; Sobrinho, L. A practical method for the detection of macroprolactinaemia using ultrafiltration. Clin. Endocrinol. 2003, 58, 686–690. [Google Scholar] [CrossRef]
- Sapin, R.; Kertesz, G. Macroprolactin Detection by Precipitation with Protein A-Sepharose: A Rapid Screening Method Compared with Polyethylene Glycol Precipitation. Clin. Chem. 2003, 49, 502–505. [Google Scholar] [CrossRef] [PubMed]
- Fahie-Wilson, M.; Halsall, D. Polyethylene glycol precipitation: Proceed with care. Ann. Clin. Biochem. 2008, 45, 233–235. [Google Scholar] [CrossRef]
- Hoy, D.; Brooks, P.; Woolf, A.; Blyth, F.; March, L.; Bain, C.; Baker, P.; Smith, E.; Buchbinder, R. Assessing risk of bias in prevalence studies: Modification of an existing tool and evidence of interrater agreement. J. Clin. Epidemiol. 2012, 65, 934–939. [Google Scholar] [CrossRef]
- Larrea, F.; Villanueva, C.; Cravioto, M.C.; Escorza, A.; Del Real, O. Further evidence that big, big prolactin is preferentially secreted in women with hyperprolactinemia and normal ovarian function. Fertil. Steril. 1985, 44, 25–30. [Google Scholar] [CrossRef]
- Fahie-Wilson, M.; Soule, S. Macroprolactinaemia: Contribution to hyperprolactinaemia in a district general hospital and evaluation of a screening test based on precipitation with polyethylene glycol. Ann. Clin. Biochem. 1997, 34, 252–258. [Google Scholar] [CrossRef]
- Vieira, J.G.H.; Tachibana, T.T.; Obara, L.H.; Maciel, R.M. Extensive Experience and Validation of Polyethylene Glycol Precipitation as a Screening Method for Macroprolactinemia. Clin. Chem. 1998, 44, 1758–1759. [Google Scholar] [CrossRef] [PubMed]
- Olukoga, A.; Kane, J. Macroprolactinaemia: Validation and application of the polyethylene glycol precipitation test and clinical characterization of the condition. Clin. Endocrinol. 1999, 51, 119–126. [Google Scholar] [CrossRef]
- Blanco-Favela, F.; Quintal, M.G.; Chavez-Rueda, A.; Leanos-Miranda, A.; Berron-Peres, R.; Baca-Ruiz, V.; Lavalle-Montalvo, C. Anti-prolactin autoantibodies in paediatric systemic lupus erythematosus patients. Lupus 2001, 10, 803–808. [Google Scholar] [CrossRef]
- Leanos-Miranda, A.; Pascoe-Lira, D.; Chávez-Rueda, K.; Blanco-Favela, F. Detection of macroprolactinemia with the polyethylene glycol precipitation test in systemic lupus erythematosus patients with hyperprolactinemia. Lupus 2001, 10, 340–345. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Eixerés, M.R.; Mauri, M.; Alfayate, R.; Graells, M.L.; Miralles, C.; López, A.; Picó, A. Prevalence of macroprolactin detected by Elecsys® 2010. Horm. Res. Paediatr. 2001, 56, 87–92. [Google Scholar] [CrossRef]
- Leslie, H.; Courtney, C.; Bell, P.; Hadden, D.; McCance, D.; Ellis, P.; Sheridan, B.; Atkinson, A. Laboratory and clinical experience in 55 patients with macroprolactinemia identified by a simple polyethylene glycol precipitation method. J. Clin. Endocrinol. Metab. 2001, 86, 2743–2746. [Google Scholar] [CrossRef][Green Version]
- Smith, T.P.; Suliman, A.M.; Fahie-Wilson, M.N.; McKenna, T.J. Gross Variability in the Detection of Prolactin in Sera Containing Big Big Prolactin (Macroprolactin) by Commercial Immunoassays. J. Clin. Endocrinol. Metab. 2002, 87, 5410–5415. [Google Scholar] [CrossRef]
- Hauache, O.M.; Rocha, A.J.; Maia, A.C., Jr.; Maciel, R.M.; Vieira, J.G.H. Screening for macroprolactinaemia and pituitary imaging studies. Clin. Endocrinol. 2002, 57, 327–331. [Google Scholar] [CrossRef]
- Sapin, R.; Gasser, F.; Grucker, D. Free prolactin determinations in hyperprolactinemic men with suspicion of macroprolactinemia. Clin. Chim. Acta 2002, 316, 33–41. [Google Scholar] [CrossRef]
- Vallette-Kasic, S.; Morange-Ramos, I.; Selim, A.; Gunz, G.; Morange, S.; Enjalbert, A.; Martin, P.-M.; Jaquet, P.; Brue, T. Macroprolactinemia revisited: A study on 106 patients. J. Clin. Endocrinol. Metab. 2002, 87, 581–588. [Google Scholar] [CrossRef] [PubMed]
- Toldy, E.; Löcsei, Z.; Szabolcs, I.; Góth, M.I.; Kneffel, P.; Szöke, D.; Kovács, G.L. Macroprolactinemia. Endocrine 2003, 22, 267–273. [Google Scholar] [CrossRef]
- Strachan, M.W.; Teoh, W.L.; Don-Wauchope, A.C.; Seth, J.; Stoddart, M.; Beckett, G.J. Clinical and radiological features of patients with macroprolactinaemia. Clin. Endocrinol. 2003, 59, 339–346. [Google Scholar] [CrossRef] [PubMed]
- García, L.M.; Díez, A.H.; de los Ríos Ciriza, C.; Delgado, M.G.; Orejas, A.G.; Fernández, A.E.; González, C.M.; Fernández, M.F. Macroprolactin as etiology of hyperprolactinemia. Method for detection and clinical characterization of the entity in 39 patients. Rev. Clin. Esp. 2003, 203, 459–464. [Google Scholar]
- Garcia, M.; Colombani-Vidal, M.; Zylbersztein, C.; Testi, A.; Marcos, J.; Arturi, A.; Babini, J.; Scaglia, H. Analysis of molecular heterogeneity of prolactin in human systemic lupus erythematosus. Lupus 2004, 13, 575–583. [Google Scholar] [CrossRef]
- Escobar-Morreale, H.F. Macroprolactinemia in women presenting with hyperandrogenic symptoms: Implications for the management of polycystic ovary syndrome. Fertil. Steril. 2004, 82, 1697–1699. [Google Scholar] [CrossRef]
- Rivero, A.; Alonso, E.; Grijalba, A. Decision cut-off of the polyethylene glycol precipitation technique in screening for macroprolactinemia on Immulite 2000. Clin. Chem. Lab. Med. 2004, 42, 566–568. [Google Scholar] [CrossRef]
- Galoiu, S.; Kertesz, G.; Somma, C.; Coculescu, M.; Brue, T. Clinical espression of big-big prolactin and influence of macroprolactinemia upon immunodiagnostic tests. Acta Endocrinol. (1841-0987) 2005, 1, 31–42. [Google Scholar]
- Germano, L.; Mormile, A.; Filtri, L.; Cocciardi, E.; Di Grazia, M.; Marranca, D.; Limone, P.; Migliardi, M. Evaluation of polyethylene glycol precipitation as screening test for macroprolactinemia using Architect immunoanalyser. Immuno-Anal. Biol. Spécialisée 2005, 20, 402–407. [Google Scholar] [CrossRef]
- Theunissen, C.; De Schepper, J.; Schiettecatte, J.; Verdood, P.; Hooghe-Peeters, E.; Velkeniers, B. Macroprolactinemia: Clinical Significance and Characterization of the Condition. Acta Clin. Belg. 2005, 60, 190–197. [Google Scholar] [CrossRef]
- Hattori, N.; Nakayama, Y.; Kitagawa, K.; Ishihara, T.; Saiki, Y.; Inagaki, C. Anti-prolactin (PRL) autoantibody-binding sites (epitopes) on PRL molecule in macroprolactinemia. J. Endocrinol. 2006, 190, 287–293. [Google Scholar] [CrossRef][Green Version]
- Alfonso, A.; Rieniets, K.I.; Vigersky, R.A. Incidence and Clinical Significance of Elevated Macroprolactin Levels in Patients with Hyperprolactinemia. Endocr. Pract. 2006, 12, 275–280. [Google Scholar] [CrossRef]
- Álvarez-Vázquez, P.; Pérez, D.R.; García, E.A.; Fernández, C.P.; Abad, E.H.; Olivié, M.A.A. Significación clínica de la macroprolactina. Endocrinol. Nutr. 2006, 53, 374–378. [Google Scholar] [CrossRef]
- Rivas-Espinosa, J.; Trigos-Landa, Á.; Bocanegra-García, V.; Acosta-González, R.I.; Bocanegra-Alonso, A.; Rivera-Sánchez, G. Estimación de macroprolactina después de precipitación con polietilenglicol en dos inmunoensayos comerciales. Bioquimia 2006, 31, 140–145. [Google Scholar]
- Donadio, F.; Barbieri, A.; Angioni, R.; Mantovani, G.; Beck-Peccoz, P.; Spada, A.; Lania, A.G. Patients with macroprolactinaemia: Clinical and radiological features. Eur. J. Clin. Investig. 2007, 37, 552–557. [Google Scholar] [CrossRef]
- Jokar, M.; Maybodi, N.T.; Amini, A.; Fard, M.H. Prolactin and macroprolactin in patients with systemic lupus erythematosus. Int. J. Rheum. Dis. 2008, 11, 257–262. [Google Scholar] [CrossRef]
- Polat, G.; Dolgun, H.; Karatafi, M.A. The importance of macroprolactinemia in the differential diagnosis of hyperprolactinemic patients. Turk. Neurosurg. 2008, 18, 223–227. [Google Scholar]
- Vilar, L.; Freitas, M.; Naves, L.; Casulari, L.; Azevedo, M.; Montenegro, R.; Barros, A.; Faria, M.; Nascimento, G.; Lima, J.; et al. Diagnosis and management of hyperprolactinemia: Results of a Brazilian multicenter study with 1234 patients. J. Endocrinol. Investig. 2008, 31, 436–444. [Google Scholar] [CrossRef]
- Alfadda, A. Macroprolactin as a Cause of Hyperprolactinemia: Clinical and Radiological Features. Turk. J. Endocrinol. Metab. 2008, 12, 46–49. [Google Scholar]
- Jassam, N.; Paterson, A.; Lippiatt, C.; Barth, J. Macroprolactin on the Advia Centaur: Experience with 409 patients over a three-year period. Ann. Clin. Biochem. 2009, 46, 501–504. [Google Scholar] [CrossRef]
- Don-Wauchope, A.C.; Hoffmann, M.; Le Riche, M.; Ascott-Evans, B.H. Review of the prevalence of macroprolactinaemia in a South African hospital. Clin. Chem. Lab. Med. 2009, 47, 882–884. [Google Scholar] [CrossRef]
- Anaforoglu, I.; Ertorer, M.E.; Kozanoglu, I.; Unal, B.; Haydardedeoglu, F.E.; Bakiner, O.; Bozkirli, E.; Tutuncu, N.B.; Demirag, N.G. Macroprolactinemia, like hyperprolactinemia, may promote platelet activation. Endocrine 2010, 37, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Gulcelik, N.E.; Usman, A. Macroprolactinaemia in diabetic patients. Neuro Endocrinol. Lett. 2010, 31, 270–274. [Google Scholar]
- Taghipour, H.; Esmaili, H.A. Detection of macroprolactinemia in hyperprolactinemic patients by PEG precipitation using Elecsys 2010 immunoanalyser. Pak. J. Med. Sci. 2011, 27, 430–433. [Google Scholar]
- Morteza, T.; Samira, Z. Macroprolactinemia in Patients Presenting with Stalk Compressing Masses. Neurosurg. Q. 2011, 21, 42–43. [Google Scholar] [CrossRef]
- Thirunavakkarasu, K.; Dutta, P.; Sridhar, S.; Dhaliwal, L.; Prashad, G.R.V.; Gainder, S.; Sachdeva, N.; Bhansali, A. Macroprolactinemia in hyperprolactinemic infertile women. Endocrine 2013, 44, 750–755. [Google Scholar] [CrossRef]
- Sari, F.; Sari, R.; Ozdem, S.; Sarikaya, M.; Cetinkaya, R. Serum prolactin and macroprolactin levels in diabetic nephropathy. Clin. Nephrol. 2012, 78, 33–39. [Google Scholar] [CrossRef]
- Isik, S.; Berker, D.; Tutuncu, Y.A.; Ozuguz, U.; Gokay, F.; Erden, G.; Ozcan, H.N.; Kucukler, F.K.; Aydin, Y.; Guler, S. Clinical and radiological findings in macroprolactinemia. Endocrine 2012, 41, 327–333. [Google Scholar] [CrossRef]
- Tamer, G.; Telci, A.; Mert, M.; Uzum, A.K.; Aral, F.; Tanakol, R.; Yarman, S.; Boztepe, H.; Colak, N.; Alagöl, F. Prevalence of pituitary adenomas in macroprolactinemic patients may be higher than it is presumed. Endocrine 2012, 41, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Chawla, R.; Antonios, T.; Berhanu, E.; Ayana, G. Detection of Macroprolactinemia and Molecular Characterization of Prolactin Isoforms in Blood Samples of Hyperprolactinemic Women. J. Med. Biochem. 2012, 31, 19–26. [Google Scholar] [CrossRef]
- Lu, C.-C.; Hsieh, C.-J. The importance of measuring macroprolactin in the differential diagnosis of hyperprolactinemic patients. Kaohsiung J. Med. Sci. 2012, 28, 94–99. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Park, Y.-M. Serum Prolactin and Macroprolactin Levels among Outpatients with Major Depressive Disorder Following the Administration of Selective Serotonin-Reuptake Inhibitors: A Cross-Sectional Pilot Study. PLoS ONE 2013, 8, e82749. [Google Scholar] [CrossRef]
- Leaños-Miranda, A.; Ramírez-Valenzuela, K.L.; Campos-Galicia, I.; Chang-Verdugo, R.; Chinolla-Arellano, L.Z. Frequency of Macroprolactinemia in Hyperprolactinemic Women Presenting with Menstrual Irregularities, Galactorrhea, and/or Infertility: Etiology and Clinical Manifestations. Int. J. Endocrinol. 2013, 2013, 1–7. [Google Scholar] [CrossRef]
- Alpañés, M.; Sanchón, R.; Martínez-García, M.Á.; Martínez-Bermejo, E.; Escobar-Morreale, H.F. Prevalence of hyperprolactinaemia in female premenopausal blood donors. Clin. Endocrinol. 2013, 79, 545–549. [Google Scholar] [CrossRef]
- Radavelli-Bagatini, S.; Lhullier, F.; Mallmann, E.S.; Spritzer, P.M. Macroprolactinemia in women with hyperprolactinemia: A 10-year follow-up. Neuro Endocrinol. Lett. 2013, 34, 207–211. [Google Scholar]
- Jamaluddin, F.A.; Sthaneshwar, P.; Hussein, Z.; Othman, N.; Peng, C.S. Importance of screening for macroprolactin in all hyperprolactinaemic sera. Malays. J. Pathol. 2013, 35, 59. [Google Scholar]
- Elenkova, A.; Genov, N.; Abadzhieva, Z.; Kirilov, G.; Vasilev, V.; Kalinov, K.; Zacharieva, S. Macroprolactinemia in Patients with Prolactinomas: Prevalence and Clinical Significance. Exp. Clin. Endocrinol. Diabetes 2013, 121, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, S.; Cornes, M.; Ford, C.; Gama, R. Reference ranges for serum total and monomeric prolactin for the current generation Abbott Architect assay. Ann. Clin. Biochem. 2015, 52, 61–66. [Google Scholar] [CrossRef]
- Hayashida, S.A.; Marcondes, J.A.; Soares, J.M., Jr.; Rocha, M.P.; Barcellos, C.R.; Kobayashi, N.K.; Baracat, E.C.; Maciel, G.A. Evaluation of macroprolactinemia in 259 women under investigation for polycystic ovary syndrome. Clin. Endocrinol. 2014, 80, 616–618. [Google Scholar] [CrossRef]
- Silva, A.M.; Da Costa, P.M.; Pacheco, A.; Oliveira, J.C.; Freitas, C. Assessment of macroprolactinemia by polyethylene glycol precipitation method. Rev. Port. Endocrinol. Diabetes Metab. 2014, 9, 25–28. [Google Scholar] [CrossRef]
- Beda-Maluga, K.; Pisarek, H.; Romanowska, I.; Komorowski, J.; Świętosławski, J.; Winczyk, K. Ultrafiltration—An alternative method to polyethylene glycol precipitation for macroprolactin detection. Arch. Med. Sci. 2015, 11, 1001. [Google Scholar]
- Parlant-Pinet, L.; Harthé, C.; Roucher, F.; Morel, Y.; Borson-Chazot, F.; Raverot, G.; Raverot, V. Macroprolactinaemia: A biological diagnostic strategy from the study of 222 patients. Eur. J. Endocrinol. 2015, 172, 687–695. [Google Scholar] [CrossRef]
- Soh, N.A.C.; Omar, J.; Mohamed, W.M.W.; Abdullah, M.R.; Yaacob, N.M. Low prevalence of macroprolactinaemia among patients with hyperprolactinaemia screened using polyethylene glycol 8000. J. Taibah Univ. Med. Sci. 2016, 11, 464–468. [Google Scholar] [CrossRef][Green Version]
- Chen, Y.; Song, G.; Wang, Z. A new criteria for screening macroprolactinemia using polyethylene glycol treatment combined with different assays for prolactin. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 1788–1794. [Google Scholar]
- Hattori, N.; Aisaka, K.; Shimatsu, A. A possible cause of the variable detectability of macroprolactin by different immunoassay systems. Clin. Chem. Lab. Med. 2016, 54, 603–608. [Google Scholar] [CrossRef]
- Akbulut, E.D.; Ercan, M.; Erdoğan, S.; Topçuoğlu, C.; Yılmaz, F.M.; Turhan, T. Assessment of macroprolactinemia rate in a training and research hospital from Turkey. Turk. J. Biochem. 2017, 42, 87–91. [Google Scholar] [CrossRef]
- Soto-Pedre, E.; Newey, P.J.; Bevan, J.S.; Greig, N.; Leese, G.P. The epidemiology of hyperprolactinaemia over 20 years in the Tayside region of Scotland: The Prolactin Epidemiology, Audit and Research Study (PROLEARS). Clin. Endocrinol. 2017, 86, 60–67. [Google Scholar] [CrossRef]
- Dogansen, S.C.; Yalin, G.Y.; Yarman, S. Assessment of macroprolactinemia inpatients with prolactinoma. Turk. J. Biochem. 2017, 43, 71–75. [Google Scholar] [CrossRef]
- Kalsi, A.K.; Halder, A.; Jain, M.; Chaturvedi, P.; Sharma, J. Prevalence and reproductive manifestations of macroprolactinemia. Endocrine 2019, 63, 332–340. [Google Scholar] [CrossRef] [PubMed]
- Barth, J.H.; Lippiatt, C.M.; Gibbons, S.G.; Desborough, R.A. Observational studies on macroprolactin in a routine clinical laboratory. Clin. Chem. Lab. Med. 2018, 56, 1259–1262. [Google Scholar] [CrossRef] [PubMed]
- Ayan, N.N.; Temeloglu, E.K. An approach to screening for macroprolactinemia in all hyperprolactinemic sera. Int. J. Med. Biochem. 2019, 2, 19–23. [Google Scholar] [CrossRef]
- Hattori, N.; Ishihara, T.; Saiki, Y. Macroprolactinaemia: Prevalence and aetiologies in a large group of hospital workers. Clin. Endocrinol. 2009, 71, 702–708. [Google Scholar] [CrossRef] [PubMed]
- Shimatsu, A.; Hattori, N. Macroprolactinemia: Diagnostic, Clinical, and Pathogenic Significance. Clin. Dev. Immunol. 2012, 2012, 1–7. [Google Scholar] [CrossRef]
- Muhtaroglu, S.; Keti, D.B.; Hacıoglu, A. Macroprolactin: An overlooked reason of hyperprolactinemia. J. Lab. Med. 2019, 43, 163–168. [Google Scholar] [CrossRef]
- Vassilatou, E.; Schinochoritis, P.; Marioli, S.; Tzavara, I. Macroprolactinemia in a young man and review of the literature. Hormones 2003, 2, 130–134. [Google Scholar] [CrossRef]
- Sadideen, H.; Swaminathan, R. Macroprolactin: What is it and what is its importance? Int. J. Clin. Pract. 2006, 60, 457–461. [Google Scholar] [CrossRef]
- Veljkovic, K.; Servedio, D.; Don-Wauchope, A.C. Reporting of post-polyethylene glycol prolactin: Precipitation by polyethylene glycol 6000 or polyethylene glycol 8000 will change reference intervals for monomeric prolactin. Ann. Clin. Biochem. 2012, 49, 402–404. [Google Scholar] [CrossRef]

| No | Author | Year | Country | Design | Age group | Sex | Specific Condition of hPRL | Method of PRL Detection | Method of Macroprolactin Detection | Cut off Recovery (%) | n hPRL | n mPRL |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Larrea et al. [24] | 1985 | Mexico | cross-sectional | Adult | Female | No | RIA | GFC | - | 12 | 3 |
| 2 | Fahie-Wilson et al. [25] | 1997 | UK | cross-sectional | Adult | Both | No | FIA, EIA, CLIA | PEG 6000, GFC | - | 69 | 17 |
| 3 | Vieira et al. [26] | 1998 | Brazil | cross-sectional | Unspecified | Both | No | FIA | PEG 6000, GFC | 30 | 1220 | 513 |
| 4 | Olukoga et al. [27] | 1999 | UK | cross-sectional | Adult | Both | No | FIA | PEG 6000 | 40 | 188 | 29 |
| 5 | Blanco-Favela et al. [28] | 2001 | Mexico | cross-sectional | Teenage | Both | SLE patients | IRMA | PEG, Protein G Sepharose | - | 32 | 7 |
| 6 | Leaños-Miranda et al. [29] | 2001 | Mexico | cross-sectional | Unspecified | Both | SLE patients | IRMA | PEG 6000, GFC | - | 43 | 14 |
| 7 | Sánchez-Eixerés et al. [30] | 2001 | Spain | cross-sectional | Adult | Both | No | ECLIA | PEG 6000, GFC | 40 | 211 | 19 |
| 8 | Schiettecatte et al. [19] | 2001 | Belgium | cross-sectional | Unspecified | Both | No | ECLIA | PEG 6000, GFC | 50 | 175 | 38 |
| 9 | Leslie et al. [31] | 2001 | UK | cross-sectional | Adult | Female | No | FIA | PEG 6000 | 40 | 1225 | 322 |
| 10 | Smith et al. [32] | 2002 | UK | cross-sectional | Adult | Both | No | EIA, CLIA, ECLIA, IFMA | PEG, GFC | - | 300 | 71 |
| 11 | Hauache et al. [33] | 2002 | Brazil | cross-sectional | Adult | Both | No | FIA | PEG 6000, GFC | 30 | 113 | 52 |
| 12 | Sapin et al. [34] | 2002 | France | cross-sectional | All age | Male | No | CLIA, ECLIA | PEG 6000 | 40 | 34 | 14 |
| 13 | Vallette-Kasic et al. [35] | 2002 | France | cross-sectional | All age | Both | No | CLIA | GFC | - | 1106 | 106 |
| 14 | Toldy et al. [36] | 2003 | Hungary | cross-sectional | All age | Both | No | ECLIA | PEG 6000 | 40 | 270 | 62 |
| 15 | Strachan et al. [37] | 2003 | UK | cross-sectional | Adult | Both | No | CLIA | PEG 6000 | 50 | 273 | 58 |
| 16 | García Menéndez et al. [38] | 2003 | Spain | cross-sectional | Adult | Both | No | ECLIA | PEG 6000 | 50 | 195 | 39 |
| 17 | García et al. [39] | 2004 | Argentina | cross-sectional | Adult | Both | SLE patients | IRMA | PEG 6000, GFC | - | 34 | 7 |
| 18 | Escobar-Morreale et al. [40] | 2004 | USA | cross-sectional | Adult | Female | Hyperandrogenic | CLIA | PEG 6000 | 40 | 8 | 4 |
| 19 | Rivero et al. [41] | 2004 | Spain | cross-sectional | Adult | Both | No | CLIA | PEG 6000, GFC | 54 | 96 | 11 |
| 20 | Galoiu et al. [42] | 2005 | Romania | cross-sectional | Adult | Both | No | IRMA, ECLIA | GFC, protein A precipitation | - | 84 | 16 |
| 21 | Germano et al. [43] | 2005 | Italy | cross-sectional | Adult | Both | No | CLIA | PEG 6000 | 40 | 172 | 37 |
| 22 | Gibney et al. [18] | 2005 | Ireland | cross-sectional | Adult | Both | No | FIA | PEG 8000 | - | 2089 | 453 |
| 23 | Theunissen et al. [44] | 2005 | Belgium | cross-sectional | Adult | Both | No | EIA, RIA, ECLIA | PEG 6000 | 40 | 77 | 14 |
| 24 | Hattori et al. [45] | 2006 | Japan | cross-sectional | Teenage and adult | Both | No | ELISA | PEG 6000 | 40 | 159 | 18 |
| 25 | Alfonso et al. [46] | 2006 | USA | cross-sectional | Adult | Both | No | ECLIA | PEG | 50 | 82 | 40 |
| 26 | Álvarez-Vázquez et al. [47] | 2006 | Spain | cross-sectional | Teenage and adult | Both | No | CLIA | PEG 6000 | 75 | 228 | 22 |
| 27 | Rivas-Espinosa et al. [48] | 2006 | Mexico | others | Adult | Both | No | EIA | PEG 6000 | 50 | 30 | 7 |
| 28 | Donadio et al. [49] | 2007 | Italy | retrospective cohort | Adult | Both | No | FIA | PEG 6000 | 40 | 135 | 57 |
| 29 | Jokar et al. [50] | 2008 | Iran | cross-sectional | Teenage and adult | Both | SLE patients | RIA | PEG | 40 | 9 | 5 |
| 30 | Baǧdatoǧlu et al. [51] | 2008 | Turkey | cross-sectional | All age | Both | No | ECLIA | PEG 6000 | 40 | 124 | 13 |
| 31 | Vilar et al. [52] | 2008 | Brazil | cross-sectional | Adult | Both | No | CLIA, IRMA | PEG | 30 | 1234 | 115 |
| 32 | Alfadda et al. [53] | 2008 | Saudi Arabia | retrospective cohort | All age | Both | No | ECLIA | PEG 6000 | 40 | 156 | 10 |
| 33 | Jassam et al. [54] | 2009 | UK | cross-sectional | Adult | Both | No | CLIA | PEG 6000, GFC | 40 | 409 | 16 |
| 34 | Don-Wauchope et al. [55] | 2009 | South Africa | cross-sectional | All age | Both | No | CLIA | PEG 6000 | 60 | 170 | 48 |
| 35 | Hattori et al. [9] | 2010 | Japan | cross-sectional | Adult | Both | No | EIA | PEG 6000 | 40 | 292 | 44 |
| 36 | Anaforoglu et al. [56] | 2010 | Turkey | case-control | Adult | Female | No | CLIA | PEG 8000 | 40 | 34 | 14 |
| 37 | McCudden et al. [11] | 2010 | USA | cross-sectional | Adult | Female | No | CLIA | PEG 6000 | 40 | 120 | 20 |
| 38 | Gulcelik et al. [57] | 2010 | Turkey | cross-sectional | Adult | Both | No | CLIA | PEG | 40 | 174 | 76 |
| 39 | Taghipour et al. [58] | 2011 | Iran | cross-sectional | Adult | Both | No | ECLIA | PEG 6000 | 40 | 188 | 32 |
| 40 | Can et al. [10] | 2011 | Turkey | cross-sectional | Adult | Female | No | CLIA | PEG 6000 | 40 | 84 | 31 |
| 41 | Morteza et al. [59] | 2011 | Iran | longitudinal | Adult | Both | hPRL due to hypothalamus or stalk compression | IRMA | PEG | 40 | 37 | 3 |
| 42 | Thirunavakkarasu et al. [60] | 2012 | India | cross-sectional | Adult | Female | Infertility | ECLIA | PEG | 40 | 183 | 21 |
| 43 | Sari et al. [61] | 2012 | Turkey | cross-sectional | Adult | Both | Type 2 diabetes | ECLIA | PEG 8000 | 40 | 40 | 13 |
| 44 | Isik et al. [62] | 2012 | Turkey | cross-sectional | Adult | Both | No | CLIA | PEG 6000 | 40 | 337 | 88 |
| 45 | Tamer et al. [63] | 2012 | Turkey | cross-sectional | Adult | Female | No | ECLIA | PEG 6000 | 40 | 161 | 60 |
| 46 | Chawla et al. [64] | 2012 | Ethiopia | cross-sectional | Adult | Female | No | ECLIA | PEG, GFC | 40 | 100 | 34 |
| 47 | Lu et al. [65] | 2012 | Taiwan | cross-sectional | Adult | Both | No | IRMA | PEG 6000 | 40 | 70 | 15 |
| 48 | Kim et al. [66] | 2013 | Korea | cross-sectional | Adult | Both | Major depression on SSRI | CLIA | PEG 8000 | 52.8 | 6 | 0 |
| 49 | Leaños-Miranda et al. [67] | 2013 | Mexico | cross-sectional | Adult | Female | Gynecological disorder | EIA | PEG 6000, GFC | - | 326 | 57 |
| 50 | Alpañés et al. [68] | 2013 | Spain | cross-sectional | Adult | Female | No | CLIA | PEG 6000 | 40 | 16 | 2 |
| 51 | Radavelli-Bagatini et al. [69] | 2013 | Brazil | longitudinal | Adult | Female | No | IRMA | PEG 6000 | 40 | 32 | 9 |
| 52 | Jamaluddin et al. [70] | 2013 | Malaysia | cross-sectional | Adult | Both | No | CLIA | PEG 6000, GFC | 40 | 204 | 9 |
| 53 | Elenkova et al. [71] | 2013 | Bulgaria | case-control | Adult | Both | Prolactinoma | RIA | PEG 8000 | 40 | 131 | 10 |
| 54 | Whitehead et al. [72] | 2014 | Britain | cross-sectional | Unspecified | Both | No | CLIA | PEG 6000 | - | 175 | 26 |
| 55 | Hayashida et al. [73] | 2014 | Brazil | cross-sectional | Adult | Female | PCOS | FIA | PEG 6000 | 30 | 34 | 16 |
| 56 | Silva et al. [74] | 2014 | Portugal | cross-sectional | Unspecified | Both | No | ECLIA | PEG 6000 | 40 | 96 | 2 |
| 57 | Beda-Maluga et al. [75] | 2015 | Poland | cross-sectional | Adult | Both | No | CLIA | PEG, Ultrafiltration, GFC | 40 | 245 | 27 |
| 58 | Parlant-Pinet et al. [76] | 2015 | France | cross-sectional | Adult | Both | No | RIA, ECLIA | PEG 6000, GFC | 30 | 222 | 63 |
| 59 | Che Soh et al. [77] | 2016 | Malaysia | cross-sectional | Adult | Both | No | ECLIA | PEG 8000 | 40 | 133 | 9 |
| 60 | Chen et al. [78] | 2016 | China | cross-sectional | All age | Both | No | CLIA, ECLIA | PEG 6000, GFC | 60 | 122 | 38 |
| 61 | Hattori et al. [79] | 2016 | Japan | cross-sectional | Adult | Female | No | EIA, CLIA | PEG 6000, GFC | 40 | 37 | 2 |
| 62 | Akbulut et al. [80] | 2017 | Turkey | cross-sectional | Unspecified | Both | No | CLIA, ECLIA | PEG 6000 | 40 | 376 | 19 |
| 63 | Soto-Pedre et al. [81] | 2017 | UK | longitudinal | Unspecified | Both | No | CLIA, ECLIA | unknown | - | 1301 | 97 |
| 64 | Dogansen et al. [82] | 2018 | Turkey | cross-sectional | Adult | Both | Prolactinomas | ECLIA | PEG 6000 | 40 | 66 | 0 |
| 65 | Kalsi et al. [83] | 2018 | India | cross-sectional | Adult | Both | No | CLIA | PEG 6000 | 25 | 102 | 22 |
| 66 | Barth et al. [84] | 2018 | UK | cross-sectional | Unspecified | Both | No | CLIA | PEG 6000 | 60 | 672 | 36 |
| 67 | Ayan et al. [85] | 2019 | Turkey | cross-sectional | Adult | Both | No | ECLIA | PEG 6000 | 40 | 73 | 10 |
| Study Characteristic | Number of Studies | Random Effect Pooled Prevalence | 95% CI of Pooled Prevalence | Within Group Heterogeneity | Between Group Heterogeneity | |||
|---|---|---|---|---|---|---|---|---|
| I2 (%) | χ2 (df) | p-Value | χ2 (df) | p-Value | ||||
| Region | ||||||||
| European Region | 37 | 17.5 | 14.0, 21.2 | 95.7 | 840.70 (36) | <0.001 | 7.32 (3) | 0.062 |
| Region of the Americas | 14 | 29.1 | 18.5, 41.0 | 97.1 | 455.07 (13) | <0.001 | ||
| Western Pacific Region | 7 | 12.6 | 6.7, 19.9 | 89.3 | 55.94 (6) | <0.001 | ||
| South-East Asian Region | 3 | 12.7 | 4.7, 23.1 | - | - | - | ||
| African Region | 2 | 30.3 | 25.0, 36.0 | - | - | - | ||
| Eastern Mediterranean Region | 4 | 13.9 | 4.8, 26.3 | 83.8 | 18.53 (3) | <0.001 | ||
| Sex | ||||||||
| Both (male and female) | 52 | 17.1 | 13.8, 20.6 | 96.2 | 1359.49 (51) | <0.001 | 6.56 (1) | 0.010 |
| Female only | 14 | 25.4 | 19.6, 31.6 | 84.9 | 86.49 (13) | <0.001 | ||
| Male only | 1 | 41.2 | 24.6, 59.3 | - | - | - | ||
| Age group | ||||||||
| Adults only | 48 | 19.8 | 16.6, 23.2 | 93.3 | 697.08 (47) | <0.001 | 0.23 (1) | 0.630 |
| Teenagers and adults | 10 | 18.0 | 11.9, 25.0 | 92.2 | 114.91 (9) | <0.001 | ||
| Teenagers only | 1 | 21.9 | 9.3, 40.0 | - | - | - | ||
| Year period | ||||||||
| Before 2000 | 4 | 26.5 | 11.2, 45.2 | 95.4 | 64.56 (3) | <0.001 | 2.64 (2) | 0.267 |
| Between 2000 and 2009 | 30 | 20.4 | 16.5, 24.5 | 94.6 | 536.29 (29) | <0.001 | ||
| Between 2010 and 2019 | 33 | 16.4 | 12.4, 20.9 | 94.3 | 557.34 (32) | <0.001 | ||
| PEG type | ||||||||
| PEG 6000 | 47 | 18.8 | 15.0, 23.0 | 95.6 | 1053.67 (46) | <0.001 | 0.06 (1) | 0.801 |
| PEG 8000 | 6 | 16.7 | 7.8, 27.7 | 90.6 | 53.43 (6) | <0.001 | ||
| Study Characteristic | Number of Studies | Regression Coefficient (β) | Standard Error of β | 95% CI of β | t | p-Value |
|---|---|---|---|---|---|---|
| Sample size | 67 | −0.00002 | 0.00003 | −0.00008, 0.00004 | −0.59 | 0.557 |
| Year of the study | 67 | −0.007 | 0.003 | −0.012, −0.002 | −2.66 | 0.010 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Che Soh, N.A.A.; Yaacob, N.M.; Omar, J.; Mohammed Jelani, A.; Shafii, N.; Tuan Ismail, T.S.; Wan Azman, W.N.; Ghazali, A.K. Global Prevalence of Macroprolactinemia among Patients with Hyperprolactinemia: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2020, 17, 8199. https://doi.org/10.3390/ijerph17218199
Che Soh NAA, Yaacob NM, Omar J, Mohammed Jelani A, Shafii N, Tuan Ismail TS, Wan Azman WN, Ghazali AK. Global Prevalence of Macroprolactinemia among Patients with Hyperprolactinemia: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health. 2020; 17(21):8199. https://doi.org/10.3390/ijerph17218199
Chicago/Turabian StyleChe Soh, Noor Azlin Azraini, Najib Majdi Yaacob, Julia Omar, Aniza Mohammed Jelani, Noorazliyana Shafii, Tuan Salwani Tuan Ismail, Wan Norlina Wan Azman, and Anis Kausar Ghazali. 2020. "Global Prevalence of Macroprolactinemia among Patients with Hyperprolactinemia: A Systematic Review and Meta-Analysis" International Journal of Environmental Research and Public Health 17, no. 21: 8199. https://doi.org/10.3390/ijerph17218199
APA StyleChe Soh, N. A. A., Yaacob, N. M., Omar, J., Mohammed Jelani, A., Shafii, N., Tuan Ismail, T. S., Wan Azman, W. N., & Ghazali, A. K. (2020). Global Prevalence of Macroprolactinemia among Patients with Hyperprolactinemia: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health, 17(21), 8199. https://doi.org/10.3390/ijerph17218199





