Pilot Test on Pre-Swim Hygiene as a Factor Limiting Trihalomethane Precursors in Pool Water by Reducing Organic Matter in an Operational Facility
Abstract
1. Introduction
Research Goals
2. Materials and Methods
2.1. Preliminary Research
2.2. Characteristics of Participating Swimming Pools
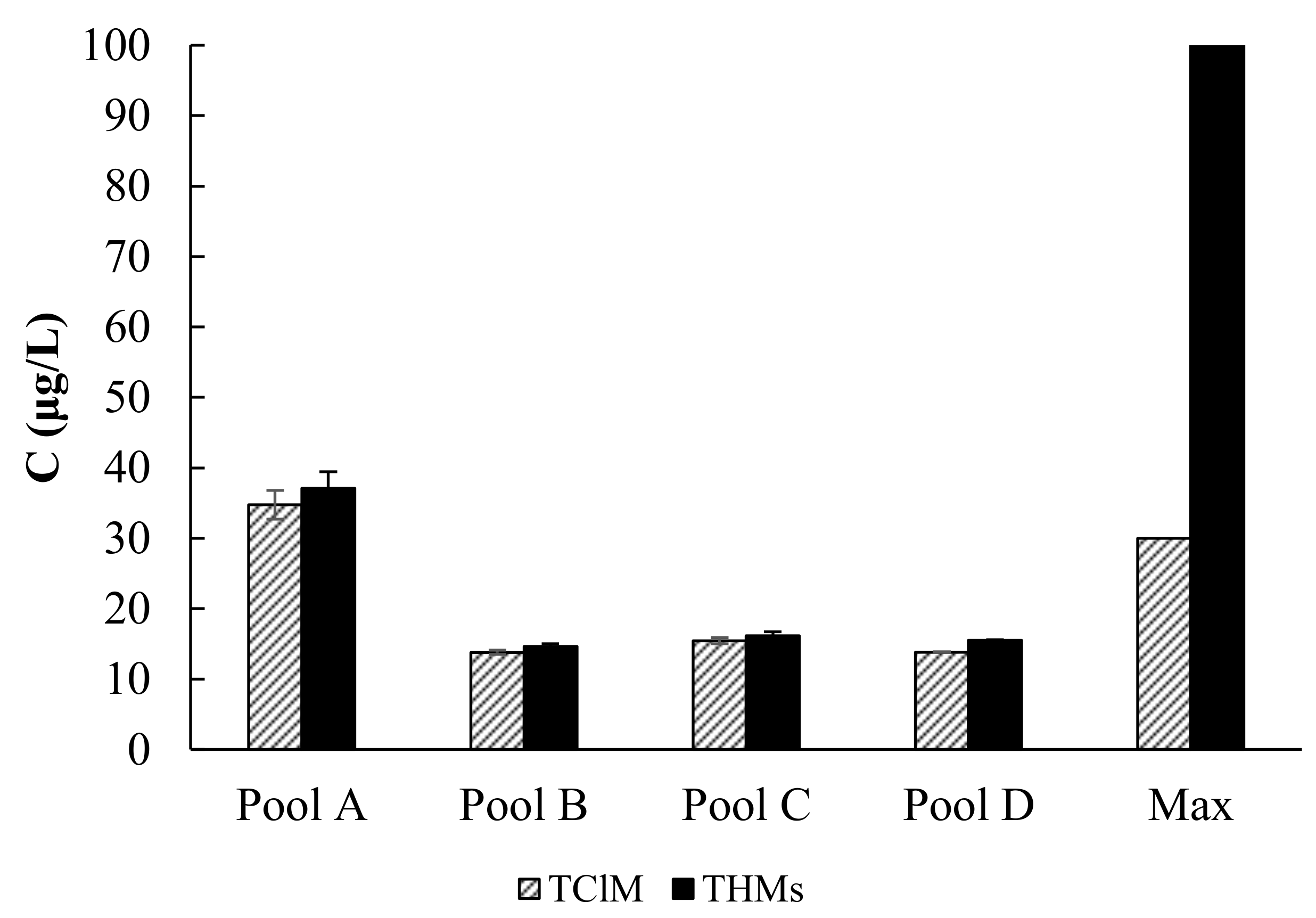
Pool A
Pool B
Pool C
Pool D
Sampling
2.3. Main Experiment
- Stage 1: assessment of DOC and THM (including chloroform) concentrations prior to the introduction of showering control,
- Stage 2: assessment of DOC and THM (including chloroform) concentrations when showering was controlled in the facility,
- Stage 3: assessment of DOC and THM (including chloroform) concentrations after abandoning showering control.
2.4. Method for Determining THMs in Pool Water
- trichloromethane (chloroform): from 0.27 μg/L to 30 μg/L (300 μg/L *),
- bromodichloromethane: from 0.30 μg/L to 30 μg/L (300 μg/L *),
- dibromochloromethane: from 0.23 μg/L to 15 μg/L (150 μg/L *),
- tribromomethane: from 0.31 μg/L to 15 μg/L (150 μg/L *).
2.5. Method of DOC Determination
3. Results
3.1. Preliminary Research Results
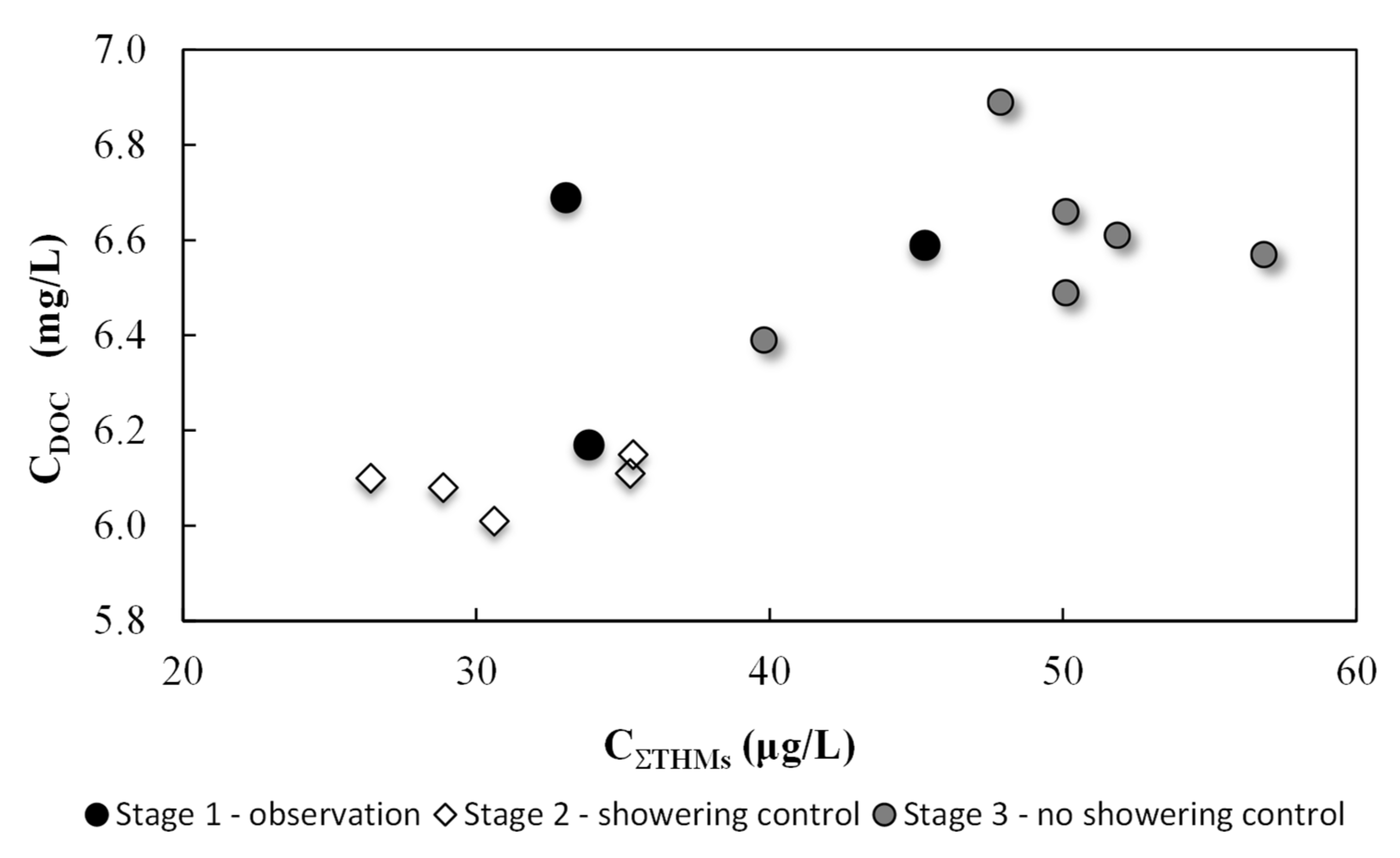
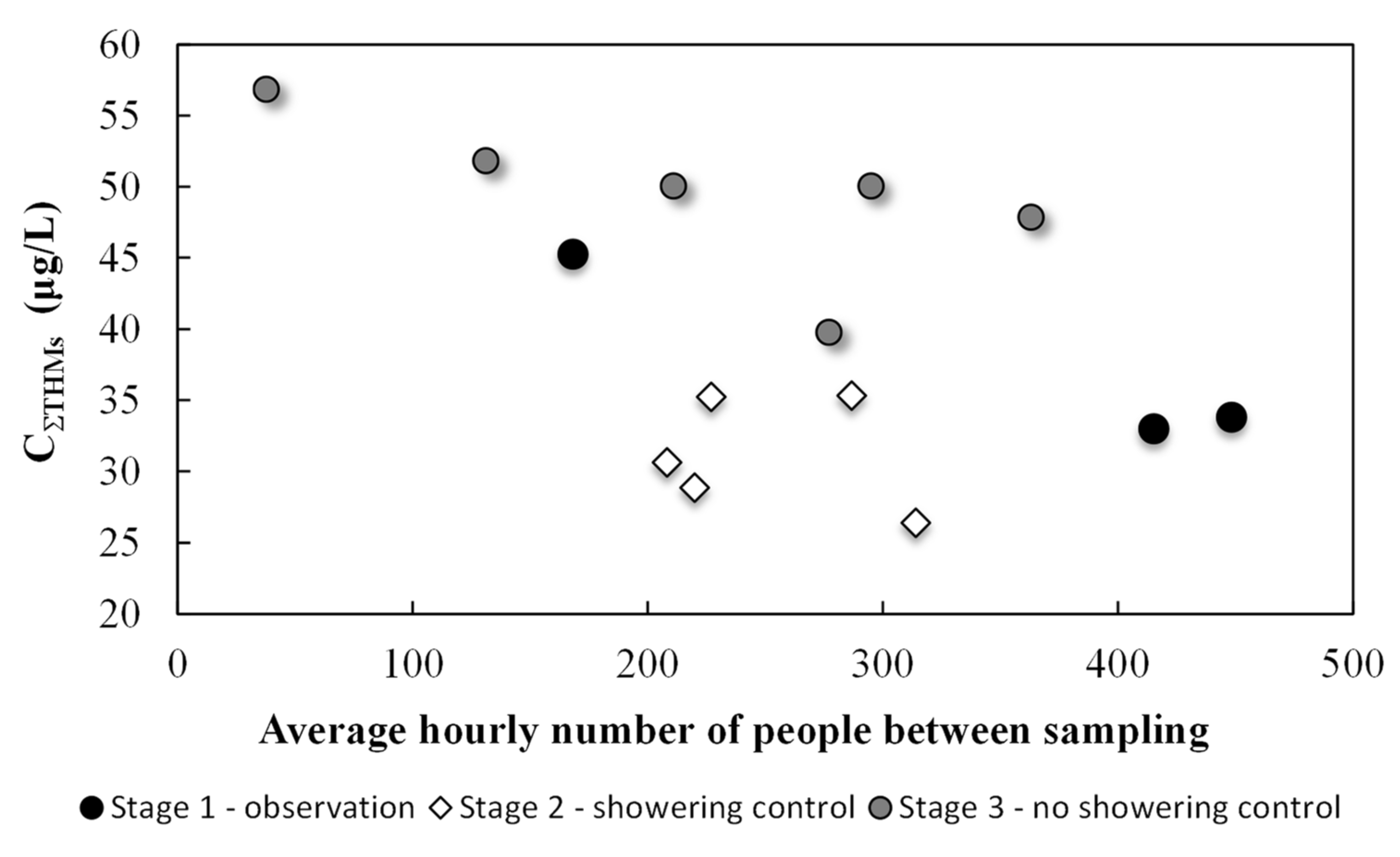
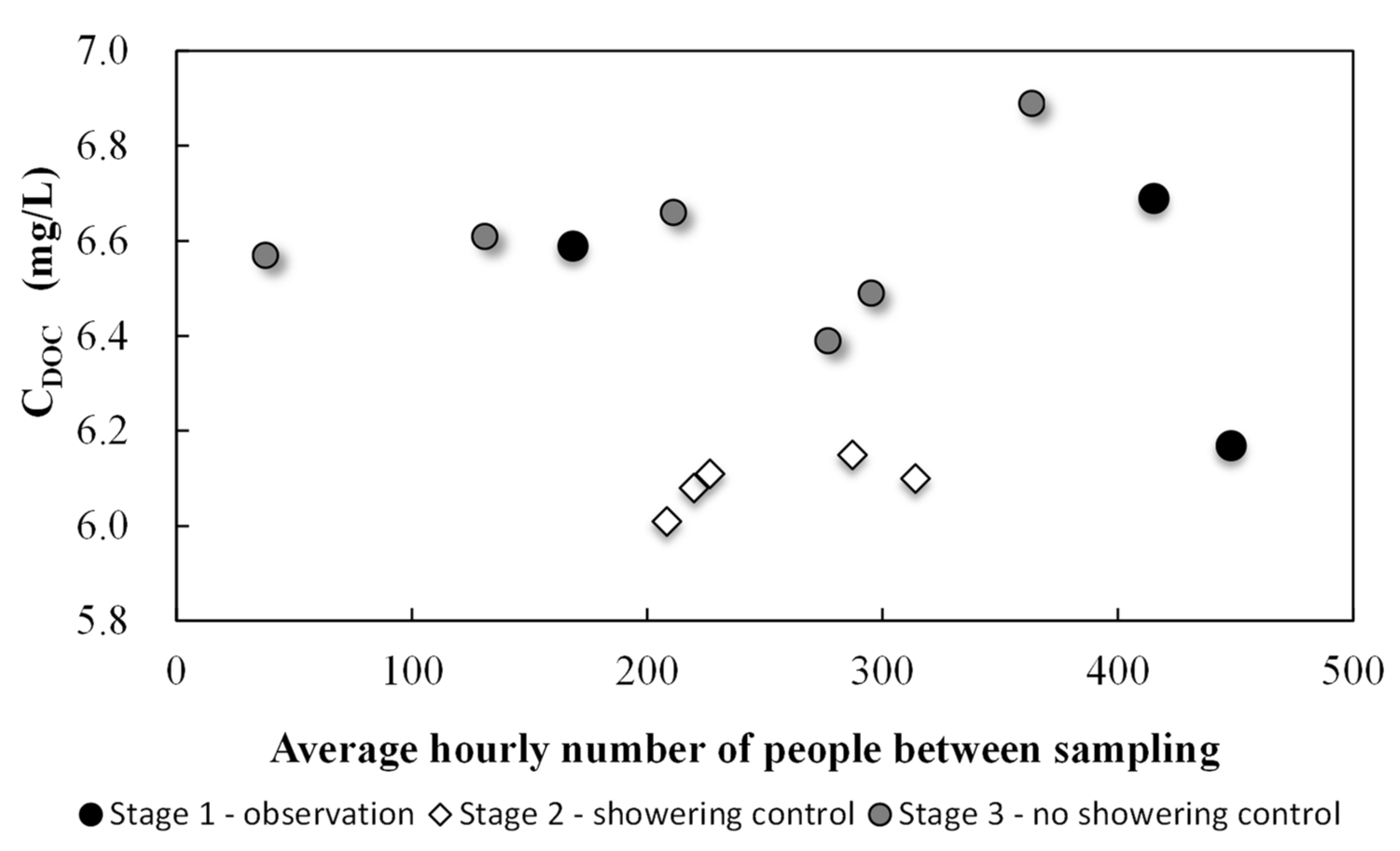
3.2. Main Experiment Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carter, R.A.A.; Joll, C.A. Occurrence and formation of disinfection by-products in the swimming pool environment: A critical review. J. Environ. Sci. 2017, 58, 19–50. [Google Scholar] [CrossRef]
- Panyapinyopol, B.; Marhaba, T.F.; Kanokkantapong, V.; Pavasant, P. Characterization of precursors to trihalomethanes formation in Bangkok source water. J. Hazard Mater. 2005, 120, 229–236. [Google Scholar] [CrossRef]
- Barrett, S.E.; Krasner, S.W.; Amy, G.L. Natural Organic Matter and Disinfection By-Products: Characterization and Control in Drinking Water—An Overview. Am. Chem. Soc. Symp. Ser. 2000, 761, 2–14. [Google Scholar]
- Bessonneau, V.; Derbez, M.; Clement, M.; Thomas, O. Determinants of chlorination by-products in indoor swimming pools. Int. J. Hyg. Environ. Health 2011, 215, 76–85. [Google Scholar] [CrossRef]
- Fantuzzi, G.; Righi, E.; Predieri, G.; Ceppelli, G.; Gobba, C.; Aggazzotti, G. Occupational exposure to trihalomethanes in indoor swimming pools. Sci. Total Environ. 2001, 264, 257–265. [Google Scholar] [CrossRef]
- Li, J.; Blatchley, E.R., III. Volatile Disinfection Byproduct Formation Resulting from Chlorination of Organic−Nitrogen Precursors in Swimming Pools. Environ. Sci. Technol. 2007, 41, 6732–6739. [Google Scholar] [CrossRef] [PubMed]
- Lian, L.E.Y.; Li, J.; Blatchley, E.R., III. Volatile Disinfection Byproducts Resulting from Chlorination of Uric Acid: Implications for Swimming Pools. Environ. Sci. Technol. 2014, 48, 3210–3217. [Google Scholar] [CrossRef]
- Simard, S.; Tardif, R.; Rodriguez, M.J. Variability of chlorination by-product occurrence in water of indoor and outdoor swimming pools. Water Res. 2013, 47, 1763–1772. [Google Scholar] [CrossRef]
- Weng, S.C.; Weaver, W.A.; Zare Afifi, M.; Blatchley, T.N.; Cramer, J.S.L.; Chen, J.; Blatchley, E.R., III. Dynamics of gas-phase trichloramine (NCl3) in chlorinated, indoor swimming pool facilities. Indoor Air 2011, 21, 391–399. [Google Scholar] [CrossRef]
- Zwiener, C.; Richardson, S.D.; De Marini, D.M.; Grummr, T.; Glauner, T.; Frimmel, F.H. Drowning in Disinfection Byproducts? Assessing Swimming Pool Water. Environ. Sci. Technol. 2007, 41, 363–372. [Google Scholar] [CrossRef]
- Schmalz, C.; Frimmel, F.H.; Zwiener, C. Trichloramine in swimming pools–Formation and mass transfer. Water Res. 2011, 45, 2681–2690. [Google Scholar] [CrossRef] [PubMed]
- Weng, S.C.; Sun, P.; Ben, W.; Huang, C.H.; Lee, L.T.; Blatchley, E.R., III. The Presence of Pharmaceuticals and Personal Care Products (PPCPs) in Swimming Pools. Environ. Sci. Technol. Lett. 2014, 1, 495–498. [Google Scholar] [CrossRef]
- Caro, J.H.; Gallego, M. Alveolar air and urine analyses as biomarkers of exposure to trihalomethanes in an indoor swimming pool. Environ. Sci. Technol. 2008, 42, 5002–5007. [Google Scholar] [CrossRef]
- Dyck, R.; Sadiq, R.; Rodriguez, M.J.; Simard, S.; Tardif, R. Trihalomethane exposures in indoor swimming pools: A level III fugacity model. Water Res. 2011, 45, 5084–5098. [Google Scholar] [CrossRef] [PubMed]
- Lourencetti, C.; Grimalt, J.O.; Marco, E.; Fernandez, P.; Font-Ribera, L.; Villanueva, C.M.; Kogevinas, M. Trihalomethanes in chlorine and bromine disinfected swimming pools: Air-water distributions and human exposure. Environ. Int. 2012, 45, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Font-Ribera, L.; Kogevinas, M.; Schmaltz, C.; Zwiener, C.; Marco, E.; Grimalt, J.O.; Liu, J.; Zhang, X.; Mitch, W.; Critelli, R.; et al. Environmental and personal determination of the uptake of disinfection by-products during swimming. Environ. Res. 2016, 149, 206–215. [Google Scholar] [CrossRef]
- Daiber, E.J.; DeMarini, D.M.; Ravuri, S.A.; Liberatore, H.K.; Cuthbertson, A.A.; Thompson-Klemish, A.; Byer, J.D.; Schmid, J.E.; Afifi, M.A.; Blatchley, E.R., III; et al. Progressive increase in disinfection byproducts and mutagenicity from source to tap to swimming pool and spa water: Impact of human inputs. Environ. Sci. Technol. 2016, 50, 6652–6662. [Google Scholar] [CrossRef]
- Jmaiff, B.L.K.; Wang, W.; Vemula, S.; Jaeger, B.T.; Li, X.F. Sweetened swimming pools and hot tubes. Environ. Sci. Technol. Lett. 2017, 4, 149–153. [Google Scholar] [CrossRef]
- Kim, H.; Shim, J.; Lee, S. Formation of disinfection by-products in chlorinated swimming pool water. Chemosphere 2002, 46, 123–130. [Google Scholar] [CrossRef]
- Weaver, W.A.; Li, J.; Wen, Y.; Johnston, J.; Blatchley, M.R.; Blatchley, E.R., III. Volatile disinfection by-product analysis from chlorinated indoor swimming pools. Water Res. 2009, 43, 3308–3318. [Google Scholar] [CrossRef]
- Weng, S.C.; Blatchley, E.R., III. Disinfection by-product dynamics in a chlorinated, indoor swimming pool under conditions of heavy use: National swimming competition. Water Res. 2011, 45, 5241–5248. [Google Scholar] [CrossRef]
- Afifi, M.Z.; Blatchley, E.R., III. Seasonal dynamics of water and air chemistry in an indoor chlorinated swimming pool. Water Res. 2015, 68, 771–783. [Google Scholar] [CrossRef] [PubMed]
- Erdinger, L.; Kuhn, K.P.; Kirsch, F.; Feldhues, R.; Frobel, T.; Nohynek, B.; Gabrio, T. Pathways of trihalomethane uptake in swimming pools. Int. J. Hyg. Environ. Health 2004, 207, 571–575. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Ha, K.T.; Zoh, K.D. Characteristic of trihalomethane (THM) production and associated health risk assessment in swimming pool waters treated with different disinfection methods. Sci. Total Environ. 2009, 407, 1990–1997. [Google Scholar] [CrossRef] [PubMed]
- Goma, A.; de Lluis, R.; Roca-Ferrer, J.; Lafuente, J.; Picado, C. Respiratory, ocular and skin health in recreational and competitive swimmers: Beneficial effect of a new method to reduce chlorine oxidant derivatives. Environ. Res. 2017, 152, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Tardif, R.; Rodriguez, M.; Catto, C.; Charest-Tardif, G.; Simard, S. Concentrations of disinfection by-products in swimming pool following modifications of the water treatment process: An exploratory study. J. Environ. Sci. 2017, 58, 163–172. [Google Scholar] [CrossRef]
- Ratajczak, K.; Piotrowska, A. Disinfection By-Products in Swimming pool water and Possibilities of Limiting Their Impact on Health of Swimmers. Geomat. Environ. Eng. 2019, 13, 71–92. [Google Scholar] [CrossRef]
- Krasner, S.W.; Weinberg, H.S.; Richardson, S.D.; Pastor, S.J.; Chinn, R.; Sclimenti, M.J.; Onstad, G.D.; Thruston, A.D. Occurrence of a new generation of disinfection byproducts. Environ. Sci. Technol. 2006, 40, 7175–7185. [Google Scholar] [CrossRef]
- Rook, J.J. Formation of haloforms during chlorination of natural waters. Water Treat. Exam. 1974, 23, 234–243. [Google Scholar]
- Kanan, A.; Karanfil, T. Formation of disinfection by-products in indoor swimming pool water: The contribution from filling water natural organic matter and swimmer body fluids. Water Res. 2011, 45, 926–932. [Google Scholar] [CrossRef]
- Carraro, S.; Pasquale, M.F.; Da Fre, M.; Rusconi, F.; Bonetto, G.; Zanconato, S.; Baraldi, E. Swimming pool attendance end exhaled nitric oxide in children. J. Allergy Clin. Immunol. 2006, 118, 950–960. [Google Scholar] [CrossRef] [PubMed]
- Fantuzzi, G.; Righi, E.; Predieri, G.; Giacobazzi, P.; Petra, B.; Aggazzotti, G. Airborne trichloramine (NCl3) levels and self-reported health symptoms in indoor swimming pool workers: Dose-response relationships. J. Expo. Sci. Environ. Epidemiol. 2013, 23, 88–93. [Google Scholar] [CrossRef]
- Florentin, A.; Hautemaniere, A.; Hartemann, P. Health effects of disinfection by-products in chlorinated swimming pools. Int. J. Hyg. Environ. Health 2011, 214, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Fornander, L.; Ghafouri, B.; Lindahl, M.; Graff, P. Airway irritation among indoor swimming pool personnel: Trichloramine exposure, exhaled NO and protein profiling of nasal lavage fluids. Int. Arch. Occup. Environ. Health 2013, 86, 571–580. [Google Scholar] [CrossRef]
- Jacobs, J.H.; Spaan, S.; van Rooy, G.B.G.J.; Meliefste, C.; Zaat, V.A.C.; Rooyackers, J.M.; Heederik, D. Exposure to trichloramine and respiratory symptoms in indoor swimming pool workers. Eur. Respir. J. 2007, 29, 690–698. [Google Scholar] [CrossRef] [PubMed]
- Abbasnia, A.; Ghoochani, M.; Yousefi, N.; Nazmara, S.; Radfard, M.; Soleimani, H.; Yousefi, M.; Barmar, S.; Alimohammadi, M. Prediction of human exposure and health risk assessment to trihalomethanes in indoor swimming pools and risk reduction strategy. Hum. Ecol. Risk Assess. Int. J. 2018. [Google Scholar] [CrossRef]
- Hamidin, N.; Yu, Q.J.; Connell, D.W. Human health risk assessment of chlorinated disinfection by-products in drinking water using a probabilistic approach. Water Res. 2008, 42, 3263–3274. [Google Scholar] [CrossRef]
- Lourencetti, C.; Ballester, C.; Fernandez, P.; Marco, E.; Prado, C.; Periago, J.F.; Grimalt, J.O. New method for determination of trihalomethanes in exhaled breath: Applications to swimming pool and bath environments. Anal. Chim. Acta 2010, 662, 23–30. [Google Scholar] [CrossRef]
- Villanueva, C.M.; Cantor, K.P.; Grimalt, J.O.; Malats, N.; Silverman, D.; Tardon, A.; Garcia-Closas, R.; Serra, C.; Carrato, A.; Castano-Vinyals, G.; et al. Bladder cancer and exposure to water disinfection by-products through ingestion, bathing, showering and swimming in pool. Am. J. Epidemiol. 2007, 165, 148–156. [Google Scholar] [CrossRef]
- Aggazzotti, G.; Righi, E.; Fantuzzi, G.; Biasotti, B.; Ravera, G.; Kanitz, S.; Barbone, F.; Sansebastiano, G.; Battaglia, M.A.; Leoni, V. Chlorination by-products (CBPs) in drinking water and adverse pregnancy outcomes in Italy. J. Water Health 2004, 2, 233–247. [Google Scholar] [CrossRef]
- Nieuwenhuijsen, M.J.; Northstone, K.; Golding, J. Swimming and birth weight. Epidemiology. 2002, 13, 725–728. [Google Scholar] [CrossRef] [PubMed]
- Rozporządzenie Ministra Zdrowia. Z dnia 9 Listopada 2015 r. w Sprawie Wymagań, Jakim Powinna Odpowiadać Woda na Pływalniach. Dz. U. 2015, poz. 2016; Rozporządzenie Ministra Zdrowia: Warsaw, Poland, 2016.
- Yang, L.; Chen, X.; She, Q.; Cao, G.; Liu, Y.; Chang, V.; Tang, C.Y. Regulation, formation, exposure, and treatment of disinfection by-products (DBPs) in swimming pool waters: A critical review. Environ. Int. 2018, 121, 1039–1057. [Google Scholar] [CrossRef] [PubMed]
- Afifi, M.Z.; Blatchley, E.R., III. Effects of UV-based treatment on volatile disinfection byproducts in a chlorinated, indoor swimming pool. Water Res. 2016, 105, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Wyczarska-Kokot, J.; Lempart, A.; Marciniak, M. Research and evaluation of water quality in outdoor swimming pools. E3s Web Conf. 2019, 100, 89. [Google Scholar] [CrossRef]
- Tardif, R.; Catto, C.; Haddad, S.; Simard, S.; Rodriguez, M. Assessment of air and water contamination by disinfection by-products at 41 indoor swimming pools. Environ. Res. 2016, 148, 411–420. [Google Scholar] [CrossRef] [PubMed]
- Kaczmarek, W.; Borys, S.; Panasiuk, J.; Majsterek, M.; Pobudkowska, A. Studies on the work characteristics of amperometric free chlorine probes. Mechatron. Syst. Mater. 2018, 2029, 20026. [Google Scholar] [CrossRef]
- Kaczmarek, W.; Borys, S. Projekt naczynia analitycznego do odwzorowania w małej skali obiegu wody basenowej z wyłączeniem układu filtracji i dezynfekcji. Przegląd Mech. 2018, 9, 33–35. [Google Scholar] [CrossRef]
- Cardador, M.J.; Gallego, M. Haloacetic acids in swimming pools: Swimmer and worker exposure. Environ. Sci. Technol. 2011, 45, 5783–5790. [Google Scholar] [CrossRef]
- Pasquarella, C.; Veronesi, L.; Napoli, C.; Castaldi, S.; Pasquarella, M.L.; Saccani, E.; Colucci, M.E.; Auxilia, F.; Galle, F.; Di Onofrio, V.; et al. Swimming pools and health-related behaviours: Results of an Italian multicentre study on showering habits among pool users. Public Health 2013, 127, 614–619. [Google Scholar] [CrossRef]
- Galle, F.; Dallolio, L.; Marotta, M.; Raggi, A.; Di Onofrio, V.; Liguori, G.; Toni, F.; Leoni, E. Health-related behaviors in swimming pool users: Influence of knowledge of regulations and awareness of health risks. Int. J. Environ. Res. Public Health 2016, 13, 513. [Google Scholar] [CrossRef]
- Pesonen, H.L.; Josko, E.; Hamalainen, S. Improving eco-efficiency of a swimming hall through customer involvement. J. Clean. Prod. 2013, 39, 294–302. [Google Scholar] [CrossRef]
- Marco, E.; Lourencetti, C.; Grimalt, J.O.; Gari, M.; Fernandez, P.; Font-Ribera, L.; Villanueva, C.M.; Kogevinas, M. Influence of physical activity in the intake of trihalomethanes in indoor swimming pools. Environ. Res. 2015, 140, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Bai, E.Y.; Lian, H.; Li, L.; Blatchley, E.R., III. Effect of chloride on the formation of volatile disinfection byproducts in chlorinated swimming pools. Water Res. 2016, 105, 413–420. [Google Scholar]
- Parinet, J.; Tabaries, S.; Coulomb, B.; Vassalo, L.; Boudenne, J.L. Exposure levels to brominated compounds in seawater swimming pools treated with chlorine. Water Res. 2012, 46, 828–836. [Google Scholar] [CrossRef] [PubMed]
| Country | Maximum Concentration | Free Chlorine | Combined Chlorine | pH | |
|---|---|---|---|---|---|
| (μg/L) | (mg/L) | (mg/L) | (−) | ||
| Germany | 20 | chloroform | 0.3–0.6 | 0.2 | 6.5–7.6 |
| Switzerland | 30 | THMs | <3.0 | 0.2 | 7.2–7.8 |
| Belgium | 100 | chloroform | 0.3–1.4 | 0.6 | 6.9–7.7 |
| United Kingdom | 100 | THMs | 1.0–2.0 | 0.2 | − |
| Poland | 100 | THMs | 0.3–0.6 | 0.3 | 6.5–7.6 |
| 30 | chloroform | ||||
| United States | 80 (drinking water) | THMs | 1–5 (2–4) | 7.4–7.6 | |
| Experiment Stage | Date of Water Sample Collection | At the Sampling Point | Air Temperature | Average Hourly Number of People between Sampling | |||
|---|---|---|---|---|---|---|---|
| Water Temperature | Free Chlorine ** | Redox | pH | ||||
| (°C) | (ppm) | (mV) | (-) | (°C) | |||
| Stage 1 observation | Mar 18 8:15 a.m. | 27.7 | 0.40 | 682 | 7.10 | 25.0 | 168 |
| Mar 19 3:00 p.m. | 27.7 | 0.25 | 645 | 7.09 | 25.5 | 448 | |
| Mar 21 5:30 p.m. | 27.8 | 0.44 | 678 | 7.10 | 25.0 | 415 | |
| Stage 2 showering control | Mar 25 3:00 p.m.* | 27.5 | 0.29 | 699 | 7.10 | 25.1 | 221 |
| Mar 26 3:00 p.m.* | 27.7 | 0.33 | 677 | 7.13 | 24.9 | 315 | |
| Mar 27 2:50 p.m.* | 27.8 | 0.30 | 730 | 7.12 | 25.4 | 227 | |
| Mar 28 2:30 p.m.* | 27.8 | 0.30 | 709 | 7.12 | 25.1 | 287 | |
| Mar 29 10:00 a.m.* | 27.7 | 0.38 | 714 | 7.07 | 24.9 | 208 | |
| Stage 3 no showering control | Mar 30 4:00 p.m. | 28.2 | 0.39 | 692 | 7.10 | 25.6 | 211 |
| Mar 31 4:00 p.m. | 28.0 | 0.40 | 674 | 7.07 | 25.1 | 38 | |
| Apr 01 3:00 p.m. | 27.7 | 0.50 | 686 | 7.02 | 26.1 | 363 | |
| Apr 02 1:30 p.m. | 28.0 | 0.45 | 690 | 7.10 | 26.1 | 277 | |
| Apr 03 3:00 p.m. | 27.8 | 0.33 | 640 | 7.11 | 26.1 | 296 | |
| Apr 04 11:00 a.m. | 28.2 | 0.40 | 650 | 7.10 | 26.5 | 132 | |
| Average | 27.8 ± 0.2 | 0.37 ± 0.07 | 683.3 ± 26 | 7.1 ± 0.03 | 25.5 ± 0.53 | 350 people/day | |
| Experiment Stages | Date of Water Sample Collection | DOC | TClM | BrDClM | DBrClM | TBrM | ∑THM |
|---|---|---|---|---|---|---|---|
| (mg/L) | (μg/L) | (μg/L) | (μg/L) | (μg/L) | (μg/L) | ||
| Stage 1 observation | Mar 18 8:15 a.m. | 6.59 | 42.5 | 2.79 | <0.31 | <0.30 | 45.3 |
| Mar 19 3:00 p.m. | 6.17 | 32.3 | 1.53 | <0.31 | <0.30 | 33.8 | |
| Mar 21 5:30 p.m. | 6.69 | 31.2 | 1.83 | <0.31 | <0.30 | 33.0 | |
| Stage 2 showering control | Mar 25 3:00 p.m.* | 6.10 | 26.6 | 2.27 | <0.31 | <0.30 | 28.9 |
| Mar 26 3:00 p.m.* | 6.10 | 24.9 | 1.50 | <0.31 | <0.30 | 26.4 | |
| Mar 27 2:50 p.m.* | 6.11 | 33.2 | 2.04 | <0.31 | <0.30 | 35.2 | |
| Mar 28 2:30 p.m.* | 6.15 | 33.3 | 2.04 | <0.31 | <0.30 | 35.3 | |
| Mar 29 10:00 a.m.* | 6.01 | 28.8 | 1.83 | <0.31 | <0.30 | 30.6 | |
| Stage 3 no showering control | Mar 30 4:00 p.m. | 6.66 | 47.4 | 2.67 | <0.31 | <0.30 | 50.1 |
| Mar 31 4:00 p.m. | 6.57 | 53.9 | 2.95 | <0.31 | <0.30 | 56.9 | |
| Apr 01 3:00 p.m. | 6.89 | 45.1 | 2.78 | <0.31 | <0.30 | 47.9 | |
| Apr 02 1:30 p.m. | 6.39 | 37.2 | 2.59 | <0.31 | <0.30 | 39.8 | |
| Apr 03 3:00 p.m. | 6.49 | 46.5 | 3.17 | 0.39 | <0.30 | 49.7 | |
| Apr 04 11:00 a.m. | 6.61 | 48.2 | 3.23 | 0.4 | <0.30 | 50.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ratajczak, K.; Pobudkowska, A. Pilot Test on Pre-Swim Hygiene as a Factor Limiting Trihalomethane Precursors in Pool Water by Reducing Organic Matter in an Operational Facility. Int. J. Environ. Res. Public Health 2020, 17, 7547. https://doi.org/10.3390/ijerph17207547
Ratajczak K, Pobudkowska A. Pilot Test on Pre-Swim Hygiene as a Factor Limiting Trihalomethane Precursors in Pool Water by Reducing Organic Matter in an Operational Facility. International Journal of Environmental Research and Public Health. 2020; 17(20):7547. https://doi.org/10.3390/ijerph17207547
Chicago/Turabian StyleRatajczak, Katarzyna, and Aneta Pobudkowska. 2020. "Pilot Test on Pre-Swim Hygiene as a Factor Limiting Trihalomethane Precursors in Pool Water by Reducing Organic Matter in an Operational Facility" International Journal of Environmental Research and Public Health 17, no. 20: 7547. https://doi.org/10.3390/ijerph17207547
APA StyleRatajczak, K., & Pobudkowska, A. (2020). Pilot Test on Pre-Swim Hygiene as a Factor Limiting Trihalomethane Precursors in Pool Water by Reducing Organic Matter in an Operational Facility. International Journal of Environmental Research and Public Health, 17(20), 7547. https://doi.org/10.3390/ijerph17207547






