A Systematic Review and Meta-Analysis of Hospitalised Current Smokers and COVID-19
Abstract
1. Introduction
2. Methods
2.1. Literature Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Data Extraction
2.4. Statistical Analysis
3. Results
3.1. Literature Retrieval
3.2. China
3.3. USA and Italy
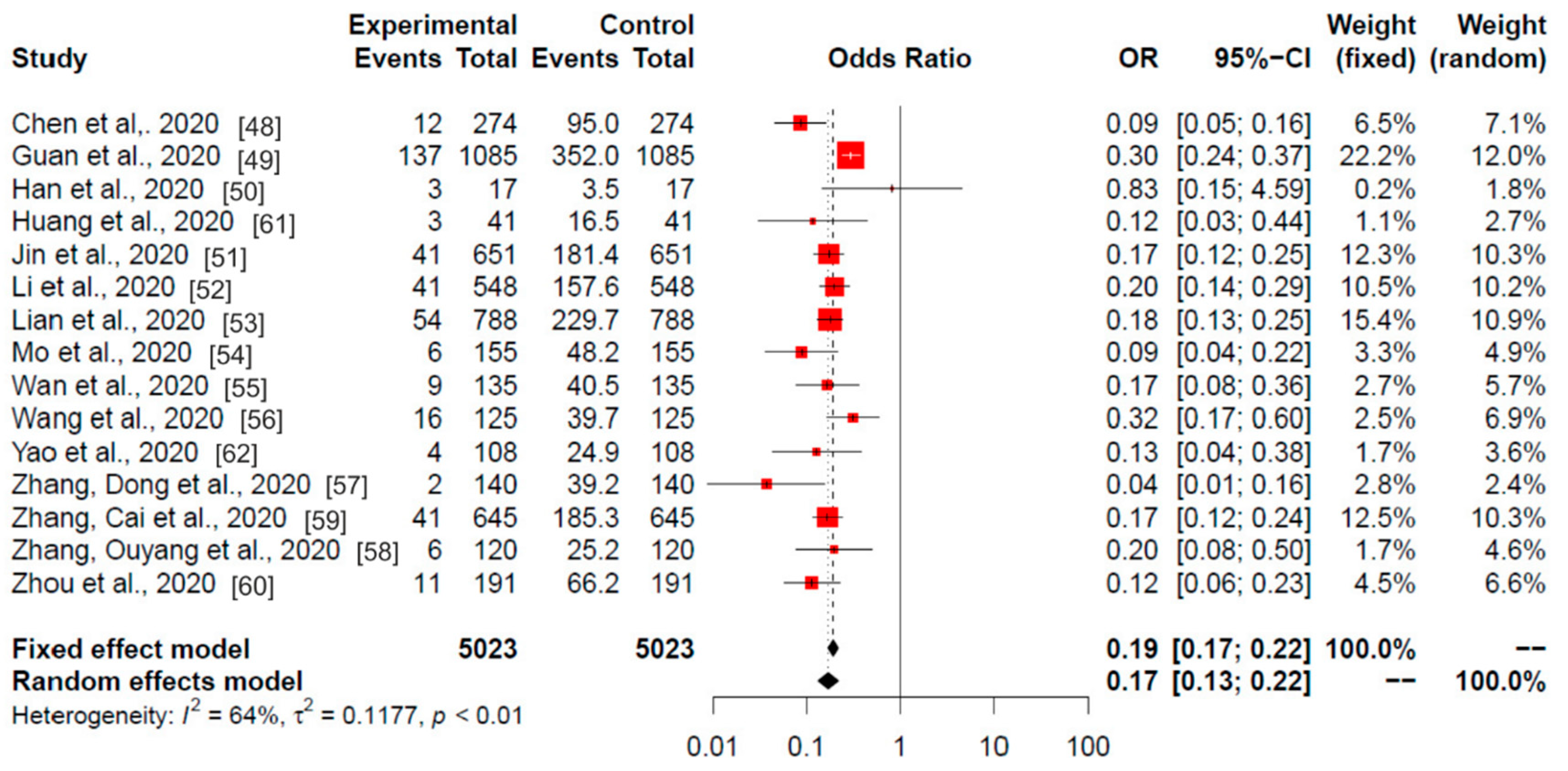
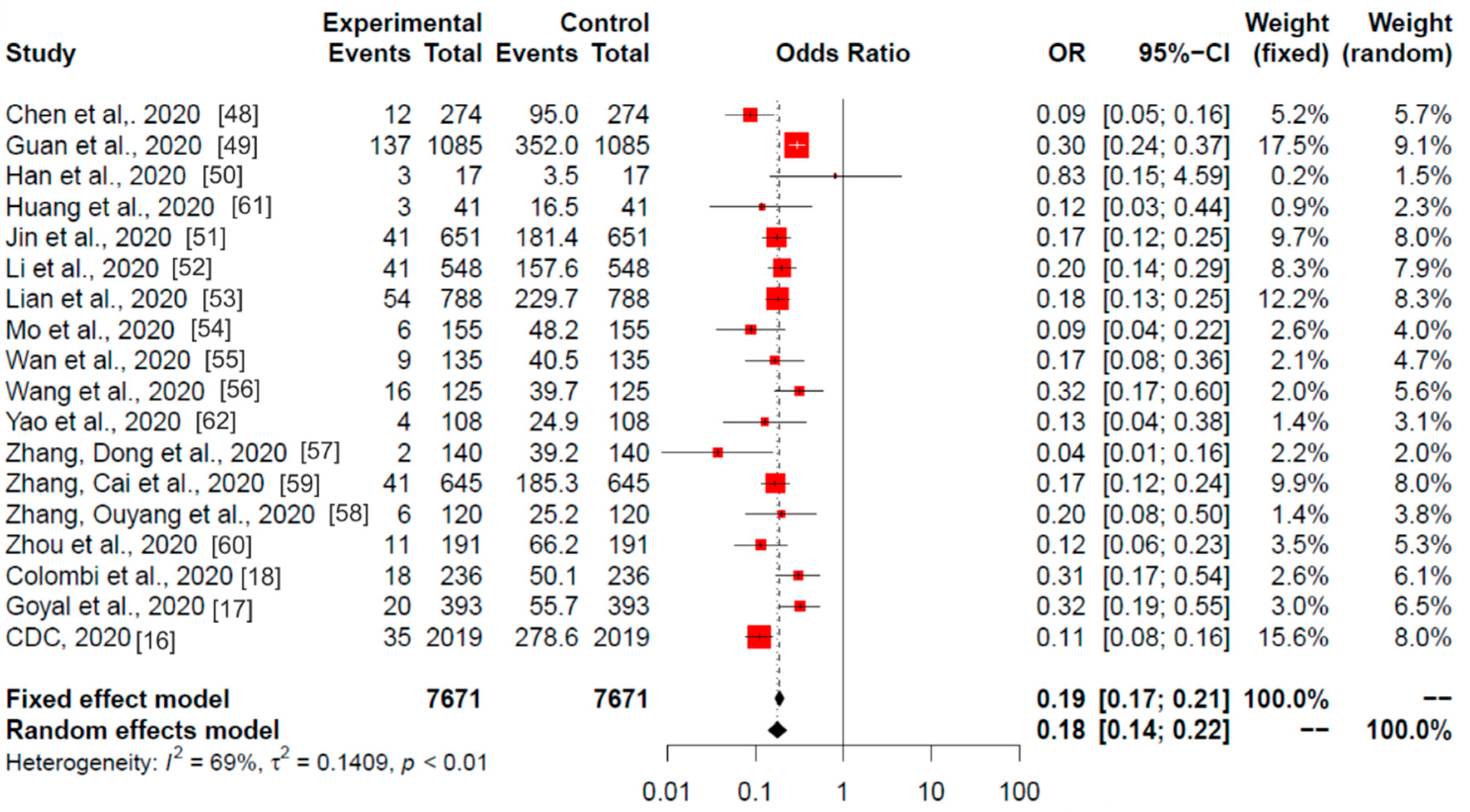
3.4. Global Meta-Analysis
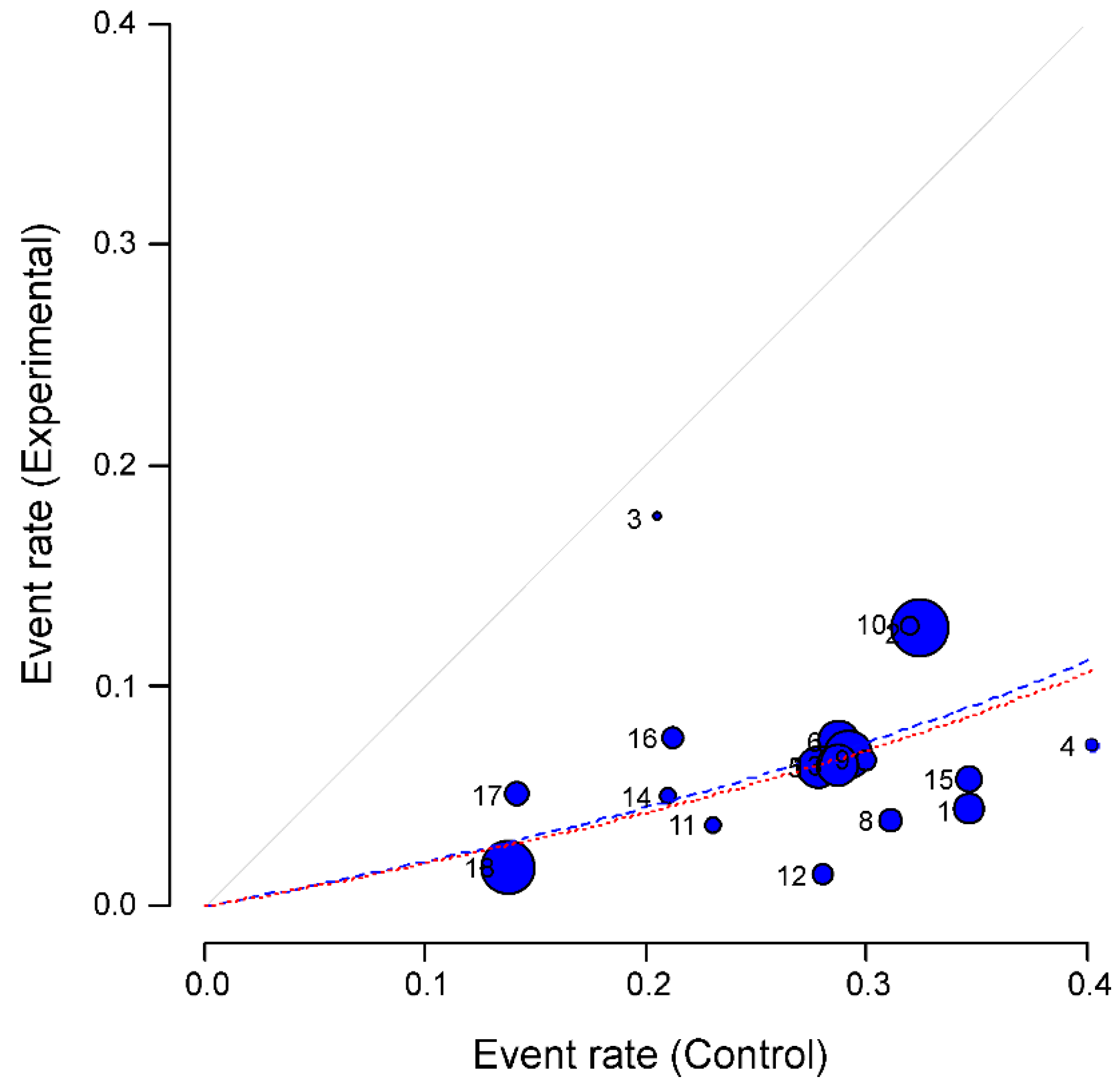
3.5. Heterogeneity
4. Discussion
4.1. Limitations and Biases
4.2. Physiological Substrate for Anti-Inflammatory Pulmonary Effect
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Ksiazek, T.G.; Erdman, D.; Goldsmith, C.S.; Zaki, S.R.; Peret, T.; Emery, S.; Tong, S.X.; Urbani, C.; Comer, J.A.; Lim, W.; et al. A novel coronavirus associated with severe acute respiratory syndrome. N. Engl. J. Med. 2003, 348, 1953–1966. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Li, F.; Shi, Z.-L. Origin and evolution of pathogenic coronaviruses. Nat. Rev. Microbiol. 2019, 17, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, S.F.; Ho, Y.-C. SARS-CoV-2: A storm is raging. J. Clin. Investig. 2020. [Google Scholar] [CrossRef]
- Lawrence, H.; Hunter, A.; Murray, R.; Lim, W.S.; McKeever, T. Cigarette smoking and the occurrence of influenza—Systematic review. J. Infect. 2019, 79, 401–406. [Google Scholar] [CrossRef]
- Alraddadi, B.M.; Watson, J.T.; Almarashi, A.; Abedi, G.R.; Turkistani, A.; Sadran, M.; Housa, A.; Almazroa, M.A.; Alraihan, N.; Banjar, A.; et al. Risk Factors for Primary Middle East Respiratory Syndrome Coronavirus Illness in Humans, Saudi Arabia, 2014. Emerg. Infect. Dis. 2016, 22, 49–55. [Google Scholar] [CrossRef]
- Cai, H. Sex difference and smoking predisposition in patients with COVID-19. Lancet Respir. Med. 2020, 8, E26. [Google Scholar] [CrossRef]
- Lippi, G.; Henry, B.M. Active smoking is not associated with severity of coronavirus disease 2019 (COVID-19). Eur. J. Intern. Med. 2020. [Google Scholar] [CrossRef]
- Kabbani, N.; Olds, J.L. Does COVID19 infect the brain? If so, smokers might be at a higher risk. Mol. Pharmacol. 2020. [Google Scholar] [CrossRef]
- Berlin, I.; Thomas, D.; Le Faou, A.-L.; Cornuz, J. COVID-19 and smoking. Nicotine Tob. Res. 2020. [Google Scholar] [CrossRef]
- Fu, L.; Wang, B.; Yuan, T.; Chen, X.; Ao, Y.; Fitzpatrick, T.; Li, P.; Zhou, Y.; Lin, Y.; Duan, Q.; et al. Clinical characteristics of coronavirus disease 2019 (COVID-19) in China: A systematic review and meta-analysis. J. Infect. 2020. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Meng, M.; Kumar, R.; Wu, Y.; Huang, J.; Lian, N.; Deng, Y.; Lin, S. The impact of COPD and smoking history on the severity of Covid-19: A systemic review and meta-analysis. J. Med. Virol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Brake, S.J.; Barnsley, K.; Lu, W.; McAlinden, K.D.; Eapen, M.S.; Sohal, S.S. Smoking Upregulates Angiotensin-Converting Enzyme-2 Receptor: A Potential Adhesion Site for Novel Coronavirus SARS-CoV-2 (Covid-19). J. Clin. Med. 2020, 9, 841. [Google Scholar] [CrossRef] [PubMed]
- Vardavas, C.I.; Nikitara, K. COVID-19 and smoking: A systematic review of the evidence. Tob. Induc. Dis. 2020, 18, 20. [Google Scholar] [CrossRef] [PubMed]
- WHO. WHO Report on the Global Tobacco Epidemic. 2019. Available online: http://www.who.int/tobacco/global_report/en/ (accessed on 20 April 2020).
- Preliminary Estimates of the Prevalence of Selected Underlying Health Conditions Among Patients with Coronavirus Disease 2019—United States, February 12-March 28, 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 382–386. [CrossRef]
- Goyal, P.; Choi, J.J.; Pinheiro, L.C.; Schenck, E.J.; Chen, R.; Jabri, A.; Satlin, M.J.; Campion, T.R.J.; Nahid, M.; Ringel, J.B.; et al. Clinical Characteristics of Covid-19 in New York City. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Colombi, D.; Bodini, F.C.; Petrini, M.; Maffi, G.; Morelli, N.; Milanese, G.; Silva, M.; Sverzellati, N.P.; Michieletti, E. Well-aerated Lung on Admitting Chest CT to Predict Adverse Outcome in COVID-19 Pneumonia. Radiology 2020, 201433. [Google Scholar] [CrossRef]
- Guan, W.-J.; Liang, W.-H.; Zhao, Y.; Liang, H.-R.; Chen, Z.-S.; Li, Y.-M.; Liu, X.-Q.; Chen, R.-C.; Tang, C.-L.; Wang, T.; et al. Comorbidity and its impact on 1590 patients with Covid-19 in China: A Nationwide Analysis. Eur. Resp. J. 2020. [Google Scholar] [CrossRef]
- Liu, W.; Tao, Z.-W.; Lei, W.; Ming-Li, Y.; Kui, L.; Ling, Z.; Shuang, W.; Yan, D.; Jing, L.; Liu, H.-G.; et al. Analysis of factors associated with disease outcomes in hospitalized patients with 2019 novel coronavirus disease. Chin. Med. J. (Engl.) 2020. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, M.; Yang, L.; Li, Y.; Wang, L.; Huang, Z.; Wang, L.; Chen, Z.; Zhou, M. Prevalence and patterns of tobacco smoking among Chinese adult men and women: Findings of the 2010 national smoking survey. J. Epidemiol. Community Health 2017, 71, 154–161. [Google Scholar] [CrossRef]
- CDC Current Cigarette Smoking among Adults in the United States. Available online: https://www.cdc.gov/tobacco/data_statistics/fact_sheets/adult_data/cig_smoking/index.htm (accessed on 22 April 2020).
- Salute, M. Della Rapporto 2018 Sulla Prevenzione e Controllo del Tabagismo. Available online: http://www.salute.gov.it/portale/documentazione/p6_2_2_1.jsp?lingua=italiano&id=2851 (accessed on 24 April 2020).
- Giannouchos, T.V.; Sussman, R.A.; Mier, J.M.; Poulas, K.; Farsalinos, K. Characteristics and risk factors for COVID-19 diagnosis and adverse outcomes in Mexico: An analysis of 89,756 laboratory-confirmed COVID-19 cases. Eur. Respir. J. 2020. [Google Scholar] [CrossRef] [PubMed]
- Baradaran, A.; Ebrahimzadeh, M.H.; Baradaran, A.; Kachooei, A.R. Prevalence of Comorbidities in COVID-19 Patients: A Systematic Review and Meta-Analysis. Arch Bone Jt. Surg. 2020, 8, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Emami, A.; Javanmardi, F.; Pirbonyeh, N.; Akbari, A. Prevalence of Underlying Diseases in Hospitalized Patients with COVID-19: A Systematic Review and Meta-Analysis. Arch. Acad. Emerg. Med. 2020, 8, e35. [Google Scholar] [CrossRef]
- Zheng, Z.; Peng, F.; Xu, B.; Zhao, J.; Liu, H.; Peng, J.; Li, Q.; Jiang, C.; Zhou, Y.; Liu, S.; et al. Risk factors of critical & mortal COVID-19 cases: A systematic literature review and meta-analysis. J. Infect. 2020, 81, e16–e25. [Google Scholar] [CrossRef] [PubMed]
- Alqahtani, J.S.; Oyelade, T.; Aldhahir, A.M.; Alghamdi, S.M.; Almehmadi, M.; Alqahtani, A.S.; Quaderi, S.; Mandal, S.; Hurst, J.R. Prevalence, Severity and Mortality associated with COPD and Smoking in patients with COVID-19: A Rapid Systematic Review and Meta-Analysis. PLoS ONE 2020, 15, e0233147. [Google Scholar] [CrossRef] [PubMed]
- Reddy, R.K.; Charles, W.N.; Sklavounos, A.; Dutt, A.; Seed, P.T.; Khajuria, A. The effect of smoking on COVID-19 severity: A systematic review and meta-analysis. J. Med. Virol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Patanavanich, R.; Glantz, S.A. Smoking Is Associated with COVID-19 Progression: A Meta-analysis. Nicotine Tob. Res. 2020, 22, 1653–1656. [Google Scholar] [CrossRef]
- Sanchez-Ramirez, D.C.; Mackey, D. Underlying respiratory diseases, specifically COPD, and smoking are associated with severe COVID-19 outcomes: A systematic review and meta-analysis. Respir. Med. 2020, 171, 106096. [Google Scholar] [CrossRef]
- Jiménez-Ruiz, C.A.; López-Padilla, D.; Alonso-Arroyo, A.; Aleixandre-Benavent, R.; Solano-Reina, S.; de Granda-Orive, J.I. COVID-19 and Smoking: A Systematic Review and Meta-Analysis of the Evidence. Arch. Bronconeumol. 2020. [Google Scholar] [CrossRef]
- Karanasos, A.; Aznaouridis, K.; Latsios, G.; Synetos, A.; Plitaria, S.; Tousoulis, D.; Toutouzas, K. Impact of Smoking Status on Disease Severity and Mortality of Hospitalized Patients With COVID-19 Infection: A Systematic Review and Meta-analysis. Nicotine Tob. Res. 2020, 22, 1657–1659. [Google Scholar] [CrossRef]
- Del Sole, F.; Farcomeni, A.; Loffredo, L.; Carnevale, R.; Menichelli, D.; Vicario, T.; Pignatelli, P.; Pastori, D. Features of severe COVID-19: A systematic review and meta-analysis. Eur. J. Clin. Investig. 2020, 50, e13378. [Google Scholar] [CrossRef] [PubMed]
- Gülsen, A.; Yigitbas, B.A.; Uslu, B.; Drömann, D.; Kilinc, O. The Effect of Smoking on COVID-19 Symptom Severity: Systematic Review and Meta-Analysis. Pulm. Med. 2020, 2020, 7590207. [Google Scholar] [CrossRef] [PubMed]
- Farsalinos, K.; Barbouni, A.; Poulas, K.; Polosa, R.; Caponnetto, P.; Niaura, R. Current smoking, former smoking, and adverse outcome among hospitalized COVID-19 patients: A systematic review and meta-analysis. Ther. Adv. Chronic. Dis. 2020, 11, 2040622320935765. [Google Scholar] [CrossRef] [PubMed]
- Farsalinos, K.; Niaura, R.; Le Houezec, J.; Barbouni, A.; Tsatsakis, A.; Kouretas, D.; Vantarakis, A.; Poulas, K. Editorial: Nicotine and SARS-CoV-2: COVID-19 may be a disease of the nicotinic cholinergic system. Toxicol. Rep. 2020. [Google Scholar] [CrossRef] [PubMed]
- Tizabi, Y.; Getachew, B.; Copeland, R.L.; Aschner, M. Nicotine and the nicotinic cholinergic system in COVID-19. FEBS J. 2020, 287, 3656–3663. [Google Scholar] [CrossRef] [PubMed]
- Farsalinos, K.; Barbouni, A.; Niaura, R. Systematic review of the prevalence of current smoking among hospitalized COVID-19 patients in China: Could nicotine be a therapeutic option? Intern. Emerg. Med. 2020, 15, 845–852. [Google Scholar] [CrossRef] [PubMed]
- Rajput, D.V. Systematic review of the prevalence of current smoking among hospitalized COVID-19 patients in China: Could nicotine be a therapeutic option? Comment. Intern. Emerg. Med. 2020. [Google Scholar] [CrossRef]
- Takagi, H. Systematic review of the prevalence of current smoking among hospitalized COVID-19 patients in China: Could nicotine be a therapeutic option? Intern. Emerg. Med. 2020. [Google Scholar] [CrossRef]
- Changeux, J.-P.; Amoura, Z.; Rey, F.A.; Miyara, M. A nicotinic hypothesis for Covid-19 with preventive and therapeutic implications. C. R. Biol. 2020, 343, 33–39. [Google Scholar] [CrossRef]
- Gonzalez-Rubio, J.; Navarro-Lopez, C.; Lopez-Najera, E.; Lopez-Najera, A.; Jimenez-Diaz, L.; Navarro-Lopez, J.D.; Najera, A. Cytokine Release Syndrome (CRS) and Nicotine in COVID-19 Patients: Trying to Calm the Storm. Front. Immunol. 2020, 11. [Google Scholar] [CrossRef]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; PRISMA-P Group. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: Chichester, UK, 2008; ISBN 978-0-470-51845-8. [Google Scholar]
- L’Abbé, K.A.; Detsky, A.S.; O’Rourke, K. Meta-analysis in clinical research. Ann. Intern. Med. 1987, 107, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Wu, D.; Chen, H.; Yan, W.; Yang, D.; Chen, G.; Ma, K.; Xu, D.; Yu, H.; Wang, H.; et al. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: Retrospective study. BMJ Br. Med. J. 2020, 368, m1091. [Google Scholar] [CrossRef]
- Guan, W.-J.; Ni, Z.-Y.; Hu, Y.; Liang, W.-H.; Ou, C.-Q.; He, J.-X.; Liu, L.; Shan, H.; Lei, C.-L.; Hui, D.S.C.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- Han, X.; Cao, Y.; Jiang, N.; Chen, Y.; Alwalid, O.; Zhang, X.; Gu, J.; Dai, M.; Liu, J.; Zhu, W.; et al. Novel Coronavirus Pneumonia (COVID-19) Progression Course in 17 Discharged Patients: Comparison of Clinical and Thin-Section CT Features During Recovery. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Jin, X.; Lian, J.-S.; Hu, J.-H.; Gao, J.; Zheng, L.; Zhang, Y.-M.; Hao, S.-R.; Jia, H.-Y.; Cai, H.; Zhang, X.-L.; et al. Epidemiological, clinical and virological characteristics of 74 cases of coronavirus-infected disease 2019 (COVID-19) with gastrointestinal symptoms. Gut 2020, 69, 1002–1009. [Google Scholar] [CrossRef]
- Li, X.; Xu, S.; Yu, M.; Wang, K.; Tao, Y.; Zhou, Y.; Shi, J.; Zhou, M.; Wu, B.; Yang, Z.; et al. Risk factors for severity and mortality in adult COVID-19 inpatients in Wuhan. J. Allergy Clin. Immunol. 2020. [Google Scholar] [CrossRef]
- Lian, J.; Jin, X.; Hao, S.; Cai, H.; Zhang, S.; Zheng, L.; Jia, H.; Hu, J.; Gao, J.; Zhang, Y.; et al. Analysis of Epidemiological and Clinical features in older patients with Corona Virus Disease 2019 (COVID-19) out of Wuhan. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Mo, P.; Xing, Y.; Xiao, Y.; Deng, L.; Zhao, Q.; Wang, H.; Xiong, Y.; Cheng, Z.; Gao, S.; Liang, K.; et al. Clinical characteristics of refractory COVID-19 pneumonia in Wuhan, China. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Wan, S.; Xiang, Y.; Fang, W.; Zheng, Y.; Li, B.; Hu, Y.; Lang, C.; Huang, D.; Sun, Q.; Xiong, Y.; et al. Clinical features and treatment of COVID-19 patients in northeast Chongqing. J. Med. Virol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Pan, M.; Zhang, X.; Fan, X.; Han, M.; Zhao, F.; Miao, M.; Xu, J.; Guan, M.; Deng, X.; et al. Epidemiological and clinical features of 125 Hospitalized Patients with COVID-19 in Fuyang, Anhui, China. Int. J. Infect. Dis. 2020. [CrossRef] [PubMed]
- Zhang, J.-J.; Dong, X.; Cao, Y.-Y.; Yuan, Y.-D.; Yang, Y.-B.; Yan, Y.-Q.; Akdis, C.A.; Gao, Y.-D. Clinical characteristics of 140 patients infected with SARS-CoV-2 in Wuhan, China. Allergy 2020. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Ouyang, H.; Fu, L.; Wang, S.; Han, J.; Huang, K.; Jia, M.; Song, Q.; Fu, Z. CT features of SARS-CoV-2 pneumonia according to clinical presentation: A retrospective analysis of 120 consecutive patients from Wuhan city. Eur. Radiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Cai, H.; Hu, J.; Lian, J.; Gu, J.; Zhang, S.; Ye, C.; Lu, Y.; Jin, C.; Yu, G.; et al. Epidemiological, clinical characteristics of cases of SARS-CoV-2 infection with abnormal imaging findings. Int. J. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Yao, Q.; Wang, P.; Wang, X.; Qie, G.; Meng, M.; Tong, X.; Bai, X.; Ding, M.; Liu, W.; Liu, K.; et al. Retrospective study of risk factors for severe SARS-Cov-2 infections in hospitalized adult patients. Pol. Arch. Intern. Med. 2020. [Google Scholar] [CrossRef]
- Liu, Q.H.; Wang, W.; Tang, M.; Zhang, H.F. Impacts of complex behavioral responses on asymmetric interacting spreading dynamics in multiplex networks. Sci. Rep. 2016, 6, 25617. [Google Scholar] [CrossRef]
- Zhan, X.-X.; Liu, C.; Zhou, G.; Zhang, Z.-K.; Sun, G.-Q.; Zhu, J.J.H.; Jin, Z. Coupling dynamics of epidemic spreading and information diffusion on complex networks. Appl. Math. Comput. 2018, 332, 437–448. [Google Scholar] [CrossRef]
- Zhang, Z.-K.; Liu, C.; Zhan, X.-X.; Lu, X.; Zhang, C.-X.; Zhang, Y.-C. Dynamics of information diffusion and its applications on complex networks. Phys. Rep. 2016, 651, 1–34. [Google Scholar] [CrossRef]
- Jordan, R.E.; Adab, P.; Cheng, K.K. Covid-19: Risk factors for severe disease and death. BMJ-Br. Med J. 2020, 368, m1198. [Google Scholar] [CrossRef] [PubMed]
- Kukull, W.A. The association between smoking and Alzheimer’s disease: Effects of study design and bias. Biol. Psychiatry 2001, 49, 194–199. [Google Scholar] [CrossRef]
- Carroll, H.A.; Toumpakari, Z.; Johnson, L.; Betts, J.A. The perceived feasibility of methods to reduce publication bias. PLoS ONE 2017, 12, e0186472. [Google Scholar] [CrossRef] [PubMed]
- Ruan, Q.; Yang, K.; Wang, W.; Jiang, L.; Song, J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.W.; Gardner, R.; Porter, D.L.; Louis, C.U.; Ahmed, N.; Jensen, M.; Grupp, S.A.; Mackall, C.L. Current concepts in the diagnosis and management of cytokine release syndrome. Blood 2014, 124, 188–195. [Google Scholar] [CrossRef]
- Zhang, C.; Wu, Z.; Li, J.-W.; Zhao, H.; Wang, G.-Q. The cytokine release syndrome (CRS) of severe COVID-19 and Interleukin-6 receptor (IL-6R) antagonist Tocilizumab may be the key to reduce the mortality. Int. J. Antimicrob. Agents 2020, 55, 105954. [Google Scholar] [CrossRef]
- Zhang, W.; Zhao, Y.; Zhang, F.; Wang, Q.; Li, T.; Liu, Z.; Wang, J.; Qin, Y.; Zhang, X.; Yan, X.; et al. The use of anti-inflammatory drugs in the treatment of people with severe coronavirus disease 2019 (COVID-19): The experience of clinical immunologists from China. Clin. Immunol. 2020, 214, 108393. [Google Scholar] [CrossRef]
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet 2020, 395, 1033–1034. [Google Scholar] [CrossRef]
- Borovikova, L.V.; Ivanova, S.; Zhang, M.; Yang, H.; Botchkina, G.I.; Watkins, L.R.; Wang, H.; Abumrad, N.; Eaton, J.W.; Tracey, K.J. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature 2000, 405, 458–462. [Google Scholar] [CrossRef]
- Mabley, J.; Gordon, S.; Pacher, P. Nicotine exerts an anti-inflammatory effect in a murine model of acute lung injury. Inflammation 2011, 34, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Gomes, J.P.; Watad, A.; Shoenfeld, Y. Nicotine and autoimmunity: The lotus’ flower in tobacco. Pharmacol. Res. 2018, 128, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Bernardo, D.; Vallejo-Díez, S.; Mann, E.R.; Al-Hassi, H.O.; Martínez-Abad, B.; Montalvillo, E.; Tee, C.T.; Murugananthan, A.U.; Núñez, H.; Peake, S.T.C.; et al. IL-6 promotes immune responses in human ulcerative colitis and induces a skin-homing phenotype in the dendritic cells and Tcells they stimulate. Eur. J. Immunol. 2012, 42, 1337–1353. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Lin, S.; Zhang, J.; Otmishi, P.; Guardiola, J.J. Airway nociceptors activated by pro-inflammatory cytokines. Respir. Physiol. Neurobiol. 2007, 156, 116–119. [Google Scholar] [CrossRef]
- Wine, J.J. Parasympathetic control of airway submucosal glands: Central reflexes and the airway intrinsic nervous system. Auton. Neurosci. 2007, 133, 35–54. [Google Scholar] [CrossRef]
- Yamada, M.; Ichinose, M. The cholinergic anti-inflammatory pathway: An innovative treatment strategy for respiratory diseases and their comorbidities. Curr. Opin. Pharmacol. 2018, 40, 18–25. [Google Scholar] [CrossRef]
- Hayashi, S.; Hamada, T.; Kadowaki, M. Mo1960 Nicotine Suppresses Colonic Tumorigenesis Associated With Chronic Colitis in Mice Through the Inhibition of IL-6 and TNF-α Expression. Gastroenterology 2013, 144, S-705. [Google Scholar] [CrossRef]
- Sandborn, W.J.; Tremaine, W.J.; Offord, K.P.; Lawson, G.M.; Petersen, B.T.; Batts, K.P.; Croghan, I.T.; Dale, L.C.; Schroeder, D.R.; Hurt, R.D. Transdermal nicotine for mildly to moderately active ulcerative colitis—A randomized, double-blind, placebo-controlled trial. Ann. Intern. Med. 1997, 126, 364–371. [Google Scholar] [CrossRef]
- Pullan, R.D.; Rhodes, J.; Ganesh, S.; Mani, V.; Morris, J.S.; Williams, G.T.; Newcombe, R.G.; Russell, M.A.; Feyerabend, C.; Thomas, G.A. Transdermal nicotine for active ulcerative colitis. N. Engl. J. Med. 1994, 330, 811–815. [Google Scholar] [CrossRef]
- Oakes, J.M.; Fuchs, R.M.; Gardner, J.D.; Lazartigues, E.; Yue, X. Nicotine and the renin-angiotensin system. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2018, 315, R895–R906. [Google Scholar] [CrossRef]
- Zhang, H.; Penninger, J.M.; Li, Y.; Zhong, N.; Slutsky, A.S. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: Molecular mechanisms and potential therapeutic target. Intensive Care Med. 2020, 46, 586–590. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.C.; Sausville, E.L.; Girish, V.; Yuan, M.L.; Vasudevan, A.; John, K.M.; Sheltzer, J.M. Cigarette Smoke Exposure and Inflammatory Signaling Increase the Expression of the SARS-CoV-2 Receptor ACE2 in the Respiratory Tract. Dev. Cell 2020, 53, 514–529. [Google Scholar] [CrossRef] [PubMed]
- Alifano, M.; Alifano, P.; Forgez, P.; Iannelli, A. Renin-angiotensin system at the heart of COVID-19 pandemic. Biochimie 2020. [Google Scholar] [CrossRef] [PubMed]
- Farsalinos, K.; Angelopoulou, A.; Alexandris, N.; Poulas, K. COVID-19 and the nicotinic cholinergic system. Eur. Respir. J. 2020, 56. [Google Scholar] [CrossRef]
- Richardson, P.; Griffin, I.; Tucker, C.; Smith, D.; Oechsle, O.; Phelan, A.; Stebbing, J. Baricitinib as potential treatment for 2019-nCoV acute respiratory disease. Lancet 2020, 395, e30–e31. [Google Scholar] [CrossRef]
- Pu, S.-Y.; Xiao, F.; Schor, S.; Bekerman, E.; Zanini, F.; Barouch-Bentov, R.; Nagamine, C.M.; Einav, S. Feasibility and biological rationale of repurposing sunitinib and erlotinib for dengue treatment. Antiviral Res. 2018, 155, 67–75. [Google Scholar] [CrossRef]
- Stebbing, J.; Phelan, A.; Griffin, I.; Tucker, C.; Oechsle, O.; Smith, D.; Richardson, P. COVID-19: Combining antiviral and anti-inflammatory treatments. Lancet Infect. Dis. 2020, 20, 400–402. [Google Scholar] [CrossRef]
- Gonzalez-Rubio, J.; Navarro-Lopez, C.; Lopez-Najera, E.; Lopez-Najera, A.; Jiménez-Díaz, L.; Navarro-López, J.D.; Nájera, A. What Is Happening with Smokers and COVID-19? A Systematic Review and a Meta-Analysis; 2020; in press. [Google Scholar] [CrossRef]
| Study | # | n (Male/Female) | Current Smokers | 95%CI (Wilson) | Expected Current Smokers (Male/Female) | Sig. |
|---|---|---|---|---|---|---|
| Chen et al., 2020 [48] | 1 | 274 (171, 103) | 12 (4.4%) | (2.6–7.4) | 95.0 (92.3, 2.7) | p < 0.0001 |
| Guan et al., 2020 [49] | 2 | 1085 (631, 454) | 137 (12.6%) | (10.8–14.7) | 352.5 (340.7, 11.8) | p < 0.0001 |
| Han et al. 2020 [50] | 3 | 17 (6, 11) | 3 (17.6%) | (8.5–38.7) | 3.5 (3.2, 0.3) | p = 0.9999 |
| Huang et al., 2020 [61] | 4 | 41 (30, 11) | 3 (7.3%) | (3.6–18.3) | 16.5 (16.2, 0.3) | p = 0.0006 |
| Jin et al., 2020 [51] | 5 | 651 (320, 331) | 41 (6.3%) | (4.7–8.4) | 181.4 (172.8, 8.6) | p < 0.0001 |
| Li et al., 2020 [52] | 6 | 548 (279, 269) | 41 (7.5%) | (5.6–10.0) | 157.7 (150.7, 7.0) | p < 0.0001 |
| Lian et al., 2020 [53] | 7 | 788 (407, 381) | 54 (6.9%) | (5.3–8.8) | 229.7 (219.8, 9.9) | p < 0.0001 |
| Mo et al., 2020 [54] | 8 | 155 (86, 69) | 6 (3.9%) | (2.0-8.0) | 48.2 (46.4, 1.8) | p < 0.0001 |
| Wan et al., 2020 [55] | 9 | 135 (72, 63) | 9 (6.7%) | (3.8–12.0) | 40.5 (38.9, 1.6) | p < 0.0001 |
| Wang et al. 2020 [56] | 10 | 125 (71, 54) | 16 (12.8%) | (8.2–19.6) | 39.7 (38.3, 1.4) | p = 0.0003 |
| Yao et al., 2020 [62] | 11 | 108 (43, 65) | 4 (3.8%) | (1.8–8.7) | 24.9 (23.2, 1.7) | p < 0.0001 |
| Zhang, Dong et al., 2020 [57] | 12 | 140 (69, 71) | 2 (1.4%) | (0.8–4.6) | 39.1 (37.3, 1.9) | p < 0.0001 |
| Zhang, Cai et al., 2020 [59] | 13 | 645 (328, 317) | 41 (6.4%) | (4.7–8.5) | 185.4 (177.2, 8.2) | p < 0.0001 |
| Zhang, Ouyang et al., 2020 [58] | 14 | 120 (43, 77) | 6 (5.0%) | (2.6–10.2) | 25.2 (23.2, 2.0) | p = 0.0004 |
| Zhou et al., 2020 [60] | 15 | 191 (119, 72) | 11 (5.8%) | (3.4–9.9) | 66.2 (64.3, 1.9) | p < 0.0001 |
| Combined | - | 5023 (2675, 2348) | 386 (7.7%) | (7.0–8.5) | 1505.6 (1444.5, 61.0) | p < 0.0001 |
| Study | # | n (Male/Female) | Current Smokers | 95%CI (Wilson) | Expected Current Smokers (Male/Female) | Sig. |
|---|---|---|---|---|---|---|
| CDC, 2020 [16] | 16 | 2019 1 | 35 (1.7%) | (1.3–2.4) | 278.6 1 | p < 0.0001 |
| Goyal et al., 2020 [17] | 17 | 393 (238, 155) | 20 (5.1%) | (3.4–7.7) | 55.7 (37.1, 18.6) | p < 0.0001 |
| USA, combined | - | 2412 | 55 (2.3%) | (1.8, 3.0) | 334.3 | p < 0.0001 |
| Colombi et al., 2020 [18] | 18 | 236 (177, 59) | 18 (7.6%) | (5.0–11.6) | 50.1 (41.2, 8.9) | p < 0.0001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Rubio, J.; Navarro-López, C.; López-Nájera, E.; López-Nájera, A.; Jiménez-Díaz, L.; Navarro-López, J.D.; Nájera, A. A Systematic Review and Meta-Analysis of Hospitalised Current Smokers and COVID-19. Int. J. Environ. Res. Public Health 2020, 17, 7394. https://doi.org/10.3390/ijerph17207394
González-Rubio J, Navarro-López C, López-Nájera E, López-Nájera A, Jiménez-Díaz L, Navarro-López JD, Nájera A. A Systematic Review and Meta-Analysis of Hospitalised Current Smokers and COVID-19. International Journal of Environmental Research and Public Health. 2020; 17(20):7394. https://doi.org/10.3390/ijerph17207394
Chicago/Turabian StyleGonzález-Rubio, Jesus, Carmen Navarro-López, Elena López-Nájera, Ana López-Nájera, Lydia Jiménez-Díaz, Juan D. Navarro-López, and Alberto Nájera. 2020. "A Systematic Review and Meta-Analysis of Hospitalised Current Smokers and COVID-19" International Journal of Environmental Research and Public Health 17, no. 20: 7394. https://doi.org/10.3390/ijerph17207394
APA StyleGonzález-Rubio, J., Navarro-López, C., López-Nájera, E., López-Nájera, A., Jiménez-Díaz, L., Navarro-López, J. D., & Nájera, A. (2020). A Systematic Review and Meta-Analysis of Hospitalised Current Smokers and COVID-19. International Journal of Environmental Research and Public Health, 17(20), 7394. https://doi.org/10.3390/ijerph17207394





