Characteristics of Cancer Patients in the World Trade Center Environmental Health Center
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Enrollment
2.2. Exposure Assessment at WTC EHC
2.3. Clinical and Cancer Characteristics in the WTC EHC
2.4. Analysis
3. Results
3.1. Participants
3.2. Cancer Types
3.3. Cancer Diagnoses and Sex
3.4. Cancer Diagnoses and Age
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Landrigan, P.J.; Lioy, P.J.; Thurston, G.; Berkowitz, G.; Chen, L.C.; Chillrud, S.N.; Gavett, S.H.; Georgopoulos, P.G.; Geyh, A.S.; Levin, S.; et al. Health and environmental consequences of the world trade center disaster. Environ. Health Perspect. 2004, 112, 731–739. [Google Scholar] [CrossRef] [PubMed]
- Leeman, J.E.; McBride, S.M.; Spielsinger, D.; Sherman, E.J.; Wong, R.; Riaz, N.; Lee, N.Y.; Tsai, C.J. Head and neck cancers associated with exposure to the September 11, 2001 World Trade Center terrorist attacks. Int. J. Cancer 2018, 142, 2485–2490. [Google Scholar] [CrossRef] [PubMed]
- Reibman, J.; Levy-Carrick, N.; Miles, T.; Flynn, K.; Hughes, C.; Crane, M.; Lucchini, R.G. Destruction of the World Trade Center Towers. Lessons Learned from an Environmental Health Disaster. Ann. Am. Thorac. Soc. 2016, 13, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Reibman, J.; Liu, M.; Cheng, Q.; Liautaud, S.; Rogers, L.; Lau, S.; Berger, K.I.; Goldring, R.M.; Marmor, M.; Fernandez-Beros, M.E.; et al. Characteristics of a residential and working community with diverse exposure to World Trade Center dust, gas, and fumes. J. Occup. Environ. Med. 2009, 51, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Reibman, J.; Bowers, J.A.; Hwang, S.A.; Hoerning, A.; Gomez, M.I.; Fitzgerald, E.F. Upper respiratory symptoms and other health effects among residents living near the World Trade Center site after September 11, 2001. Am. J. Epidemiol. 2005, 162, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Farfel, M.; DiGrande, L.; Brackbill, R.; Prann, A.; Cone, J.; Friedman, S.; Walker, D.J.; Pezeshki, G.; Thomas, P.; Galea, S.; et al. An overview of 9/11 experiences and respiratory and mental health conditions among World Trade Center Health Registry enrollees. J. Urban Health 2008, 85, 880–909. [Google Scholar] [CrossRef]
- Zeig-Owens, R.; Webber, M.P.; Hall, C.B.; Schwartz, T.; Jaber, N.; Weakley, J.; Rohan, T.E.; Cohen, H.W.; Derman, O.; Aldrich, T.K.; et al. Early assessment of cancer outcomes in New York City firefighters after the 9/11 attacks: An observational cohort study. Lancet 2011, 378, 898–905. [Google Scholar] [CrossRef]
- Brackbill, R.M.; Hadler, J.L.; DiGrande, L.; Ekenga, C.C.; Farfel, M.R.; Friedman, S.; Perlman, S.E.; Stellman, S.D.; Walker, D.J.; Wu, D.; et al. Asthma and posttraumatic stress symptoms 5 to 6 years following exposure to the World Trade Center terrorist attack. JAMA 2009, 302, 502–516. [Google Scholar] [CrossRef]
- Prezant, D.J.; Weiden, M.; Banauch, G.I.; McGuinness, G.; Rom, W.N.; Aldrich, T.K.; Kelly, K.J. Cough and bronchial responsiveness in firefighters at the World Trade Center site. N. Engl. J. Med. 2002, 347, 806–815. [Google Scholar] [CrossRef]
- Aldrich, T.K.; Gustave, J.; Hall, C.B.; Cohen, H.W.; Webber, M.P.; Zeig-Owens, R.; Cosenza, K.; Christodoulou, V.; Glass, L.; Al-Othman, F.; et al. Lung function in rescue workers at the World Trade Center after 7 years. N. Engl. J. Med. 2010, 362, 1263–1272. [Google Scholar] [CrossRef]
- Lioy, P.J.; Weisel, C.P.; Millette, J.R.; Eisenreich, S.; Vallero, D.; Offenberg, J.; Buckley, B.; Turpin, B.; Zhong, M.; Cohen, M.D.; et al. Characterization of the dust/smoke aerosol that settled east of the World Trade Center (WTC) in lower Manhattan after the collapse of the WTC 11 September 2001. Environ. Health Perspect. 2002, 110, 703–714. [Google Scholar] [CrossRef] [PubMed]
- Perlman, S.E.; Friedman, S.; Galea, S.; Nair, H.P.; Eros-Sarnyai, M.; Stellman, S.D.; Hon, J.; Greene, C.M. Short-term and medium-term health effects of 9/11. Lancet 2011, 378, 925–934. [Google Scholar] [CrossRef]
- Pleil, J.D.; Vette, A.F.; Johnson, B.A.; Rappaport, S.M. Air levels of carcinogenic polycyclic aromatic hydrocarbons after the World Trade Center disaster. Proc. Natl. Acad. Sci. USA 2004, 101, 11685–11688. [Google Scholar] [CrossRef] [PubMed]
- Weakley, J.; Webber, M.P.; Gustave, J.; Kelly, K.; Cohen, H.W.; Hall, C.B.; Prezant, D.J. Trends in respiratory diagnoses and symptoms of firefighters exposed to the World Trade Center disaster: 2005-2010. Prev. Med. 2011, 53, 364–369. [Google Scholar] [CrossRef]
- Wisnivesky, J.P.; Teitelbaum, S.L.; Todd, A.C.; Boffetta, P.; Crane, M.; Crowley, L.; de la Hoz, R.E.; Dellenbaugh, C.; Harrison, D.; Herbert, R.; et al. Persistence of multiple illnesses in World Trade Center rescue and recovery workers: A cohort study. Lancet 2011, 378, 888–897. [Google Scholar] [CrossRef]
- Joseph Murphy, M.A. World Trade Center Health Registry Explanation and Calculation of Outcome Rates. Available online: http://www.nyc.gov/html/doh/wtc/downloads/pdf/wtc/wtc-outcome-explanation.pdf (accessed on 15 July 2020).
- de Bruin, E.C.; Cowell, C.; Warne, P.H.; Jiang, M.; Saunders, R.E.; Melnick, M.A.; Gettinger, S.; Walther, Z.; Wurtz, A.; Heynen, G.J.; et al. Reduced NF1 expression confers resistance to EGFR inhibition in lung cancer. Cancer Discov. 2014, 4, 606–619. [Google Scholar] [CrossRef]
- Nadler, D.L.; Zurbenko, I.G. Developing a Weibull Model Extension to Estimate Cancer Latency. ISRN Epidemiol. 2013, 2013, 750857. [Google Scholar] [CrossRef]
- Yachida, S.; Jones, S.; Bozic, I.; Antal, T.; Leary, R.; Fu, B.; Kamiyama, M.; Hruban, R.H.; Eshleman, J.R.; Nowak, M.A.; et al. Distant metastasis occurs late during the genetic evolution of pancreatic cancer. Nature 2010, 467, 1114–1117. [Google Scholar] [CrossRef]
- CDC. Minimum Latency & Types or Categories of Cancer. Available online: https://www.cdcs.gov/wtc/pdfs/policies/WTCHP-Minimum-Cancer-Latency-PP-01062015-508.pdf (accessed on 20 June 2020).
- Moline, J.M.; Herbert, R.; Crowley, L.; Troy, K.; Hodgman, E.; Shukla, G.; Udasin, I.; Luft, B.; Wallenstein, S.; Landrigan, P.; et al. Multiple myeloma in World Trade Center responders: A case series. J. Occup. Environ. Med. 2009, 51, 896–902. [Google Scholar] [CrossRef]
- Li, J.; Brackbill, R.M.; Liao, T.S.; Qiao, B.; Cone, J.E.; Farfel, M.R.; Hadler, J.L.; Kahn, A.R.; Konty, K.J.; Stayner, L.T.; et al. Ten-year cancer incidence in rescue/recovery workers and civilians exposed to the September 11, 2001 terrorist attacks on the World Trade Center. Am. J. Ind. Med. 2016, 59, 709–721. [Google Scholar] [CrossRef]
- Li, J.; Cone, J.E.; Kahn, A.R.; Brackbill, R.M.; Farfel, M.R.; Greene, C.M.; Hadler, J.L.; Stayner, L.T.; Stellman, S.D. Association between World Trade Center exposure and excess cancer risk. JAMA 2012, 308, 2479–2488. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, M.Z.; Wallenstein, S.R.; Dasaro, C.R.; Lucchini, R.G.; Sacks, H.S.; Teitelbaum, S.L.; Thanik, E.S.; Crane, M.A.; Harrison, D.J.; Luft, B.J.; et al. Cancer in General Responders Participating in World Trade Center Health Programs, 2003-2013. JNCI Cancer Spectr. 2020, 4, pkz090. [Google Scholar] [CrossRef] [PubMed]
- Solan, S.; Wallenstein, S.; Shapiro, M.; Teitelbaum, S.L.; Stevenson, L.; Kochman, A.; Kaplan, J.; Dellenbaugh, C.; Kahn, A.; Biro, F.N.; et al. Cancer incidence in world trade center rescue and recovery workers, 2001–2008. Environ. Health Perspect. 2013, 121, 699–704. [Google Scholar] [CrossRef] [PubMed]
- Tuminello, S.; van Gerwen, M.A.G.; Genden, E.; Crane, M.; Lieberman-Cribbin, W.; Taioli, E. Increased Incidence of Thyroid Cancer among World Trade Center First Responders: A Descriptive Epidemiological Assessment. Int. J. Environ. Res. Public Health 2019, 16, 1258. [Google Scholar] [CrossRef]
- Shao, Y.; Durmus, N.; Zhang, Y.; Pehlivan, S.; Fernandez-Beros, M.E.; Umana, L.; Corona, R.; Abbott, S.; Smyth-Giambanco, S.; Arslan, A.A.; et al. The Development of a WTC Environmental Health Center Pan-cancer Database. 2020; under review. [Google Scholar]
- Niosh, C. World Trade Center Health Program. Available online: https://www.ecfr.gov/cgi-bin/text-idx?SID=5b2666d4940f00b38d815af01a2e7044&mc=true&node=pt42.1.88&rgn=div5 (accessed on 15 September 2020).
- WTC Health Program. Regulations. Available online: https://www.cdc.gov/wtc/regulations2.html (accessed on 20 June 2020).
- Trasande, L.; Fiorino, E.K.; Attina, T.; Berger, K.; Goldring, R.; Chemtob, C.; Levy-Carrick, N.; Shao, Y.; Liu, M.; Urbina, E.; et al. Associations of World Trade Center exposures with pulmonary and cardiometabolic outcomes among children seeking care for health concerns. Sci. Total Environ. 2013, 444, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)--a metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Harris, P.A.; Taylor, R.; Minor, B.L.; Elliott, V.; Fernandez, M.; O’Neal, L.; McLeod, L.; Delacqua, G.; Delacqua, F.; Kirby, J.; et al. The REDCap consortium: Building an international community of software platform partners. J. Biomed. Inform. 2019, 95, 103208. [Google Scholar] [CrossRef]
- Vogt, A.; Schmid, S.; Heinimann, K.; Frick, H.; Herrmann, C.; Cerny, T.; Omlin, A. Multiple primary tumours: Challenges and approaches, a review. ESMO Open 2017, 2, e000172. [Google Scholar] [CrossRef]
- Daniels, R.D.; Kubale, T.L.; Yiin, J.H.; Dahm, M.M.; Hales, T.R.; Baris, D.; Zahm, S.H.; Beaumont, J.J.; Waters, K.M.; Pinkerton, L.E. Mortality and cancer incidence in a pooled cohort of US firefighters from San Francisco, Chicago and Philadelphia (1950–2009). Occup. Environ. Med. 2014, 71, 388–397. [Google Scholar] [CrossRef]
- Yu, D.; Berlin, J.A.; Penning, T.M.; Field, J. Reactive oxygen species generated by PAH o-quinones cause change-in-function mutations in p53. Chem. Res. Toxicol. 2002, 15, 832–842. [Google Scholar] [CrossRef]
- Kuan, P.F.; Mi, Z.; Georgopoulos, P.; Hashim, D.; Luft, B.J.; Boffetta, P. Enhanced exposure assessment and genome-wide DNA methylation in World Trade Center disaster responders. Eur. J. Cancer Prev. 2019, 28, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Zeig-Owens, R.; Moir, W.; Hall, C.B.; Schwartz, T.; Vossbrinck, M.; Jaber, N.; Webber, M.P.; Kelly, K.J.; Ortiz, V.; et al. Estimation of Future Cancer Burden Among Rescue and Recovery Workers Exposed to the World Trade Center Disaster. JAMA Oncol. 2018, 4, 828–831. [Google Scholar] [CrossRef] [PubMed]
- NIH. Surveillance, Epidemiology, and End Results Program. Available online: https://seer.cancer.gov/index.html (accessed on 20 June 2020).
- ACS. American Cancer Society Cancer Statistics. Available online: https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2020.html (accessed on 15 July 2020).
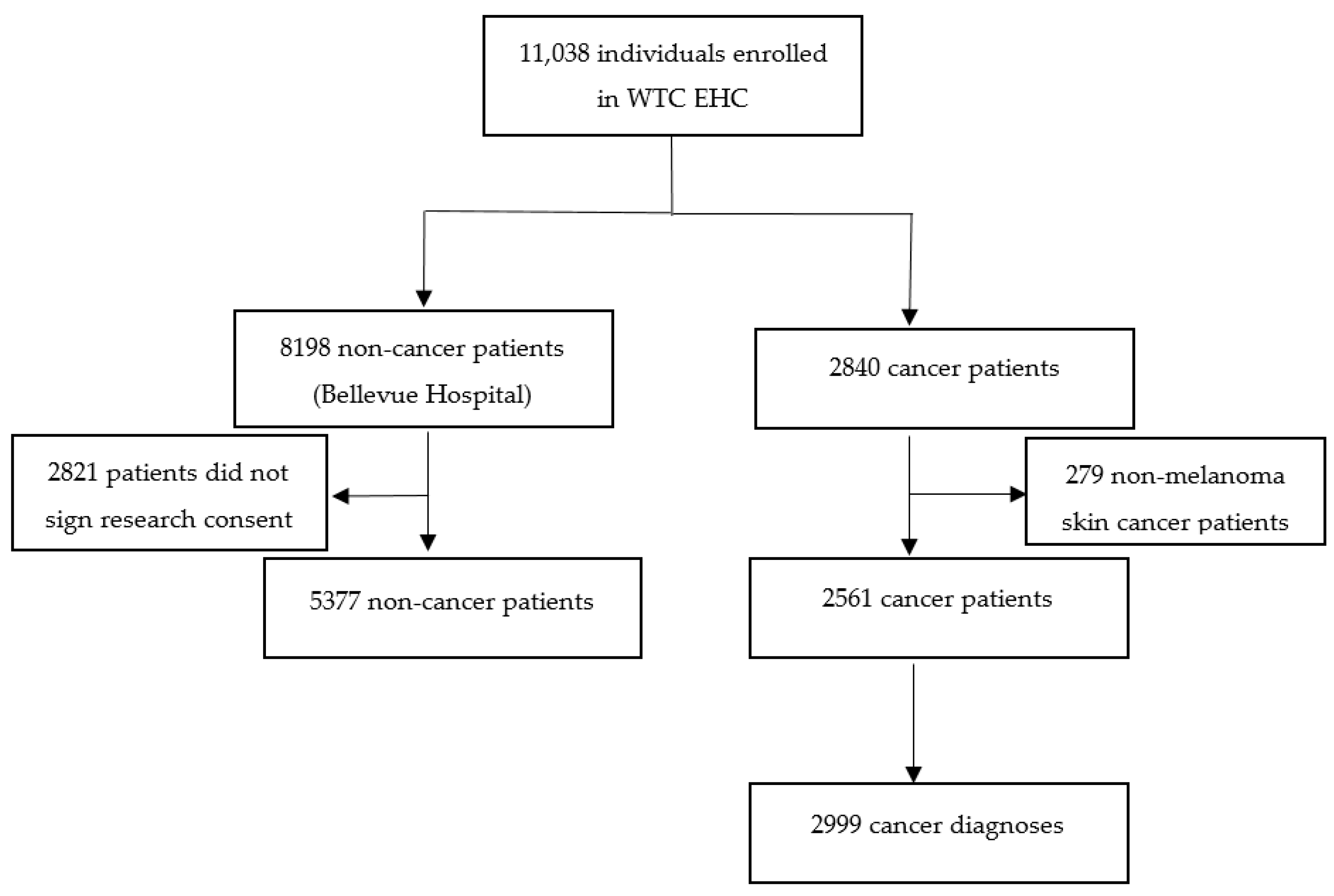
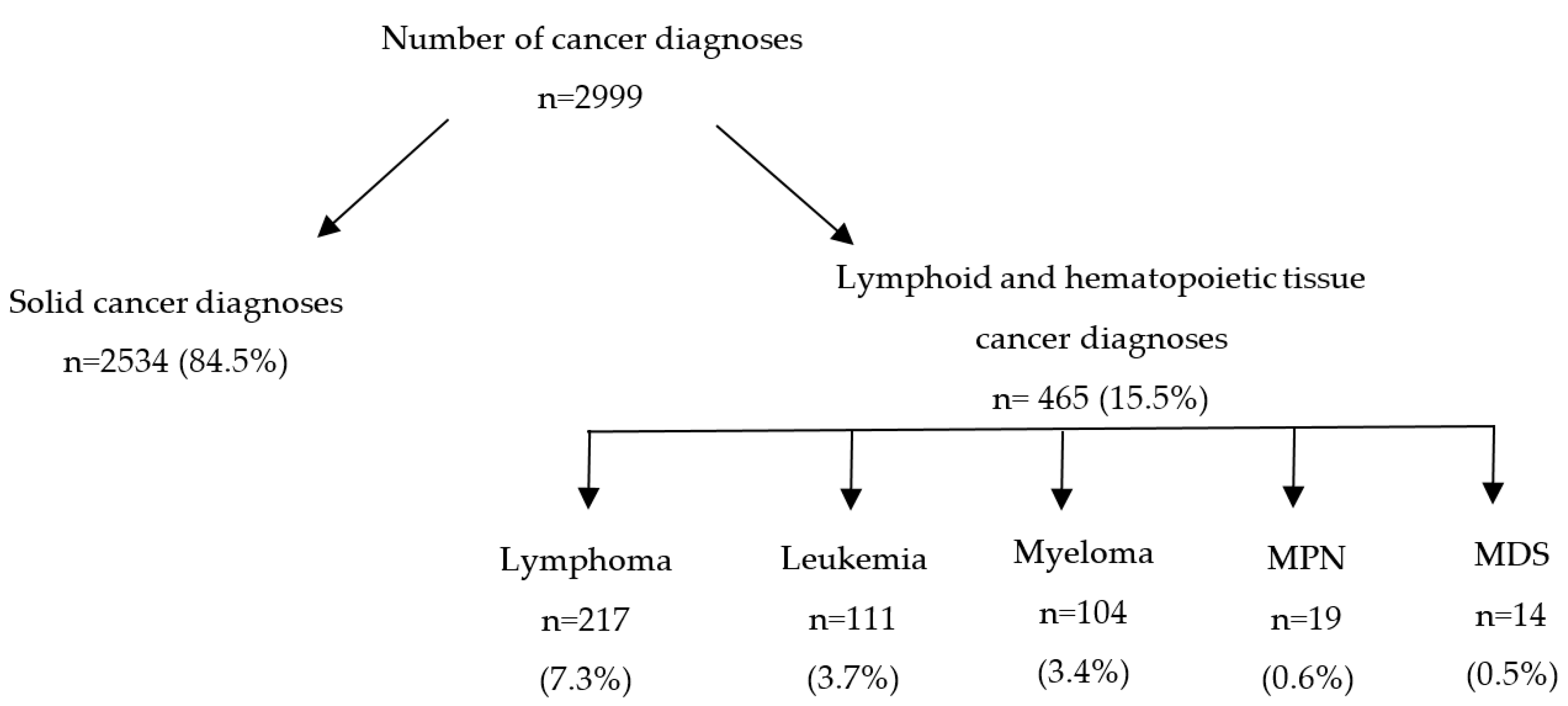
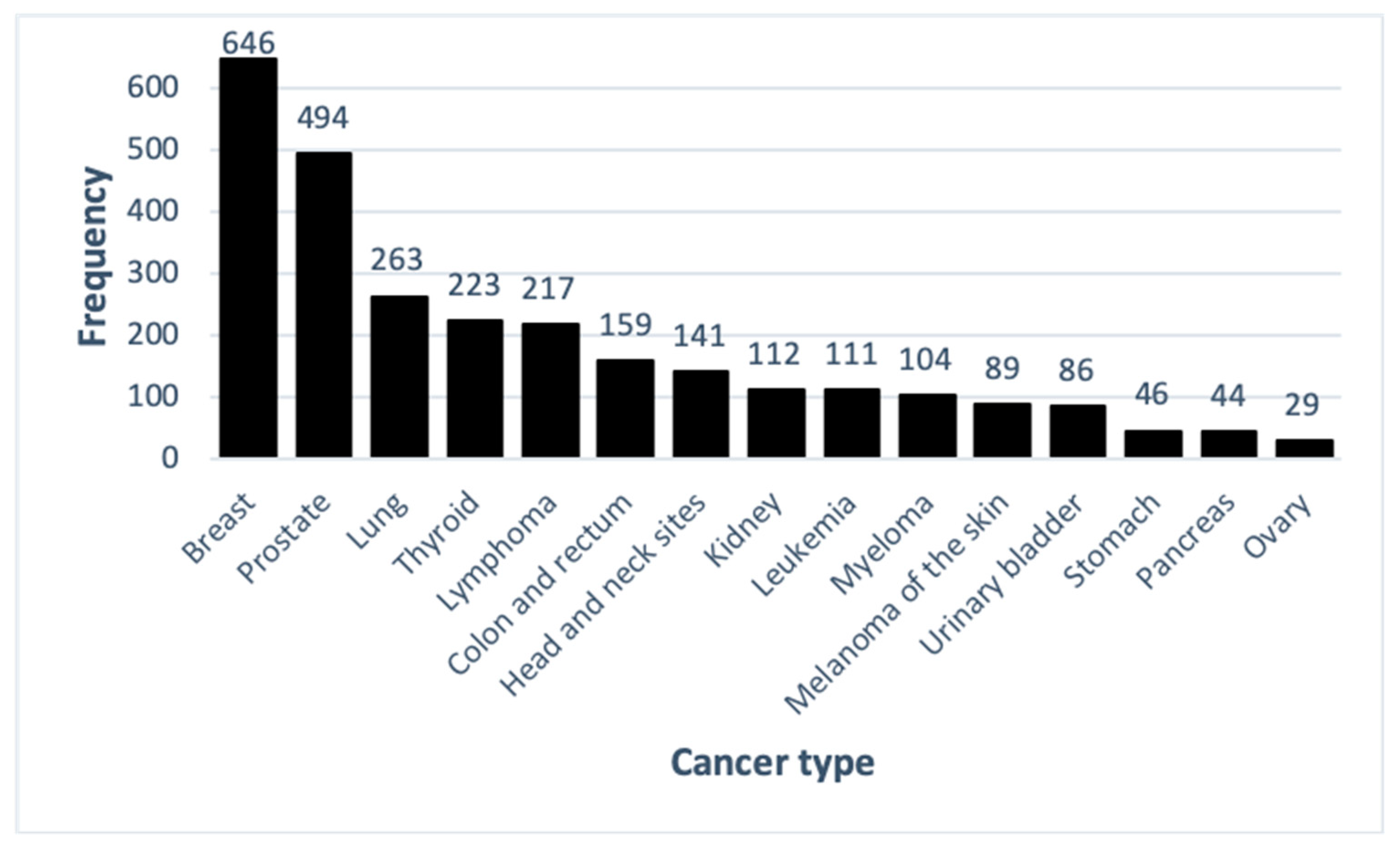
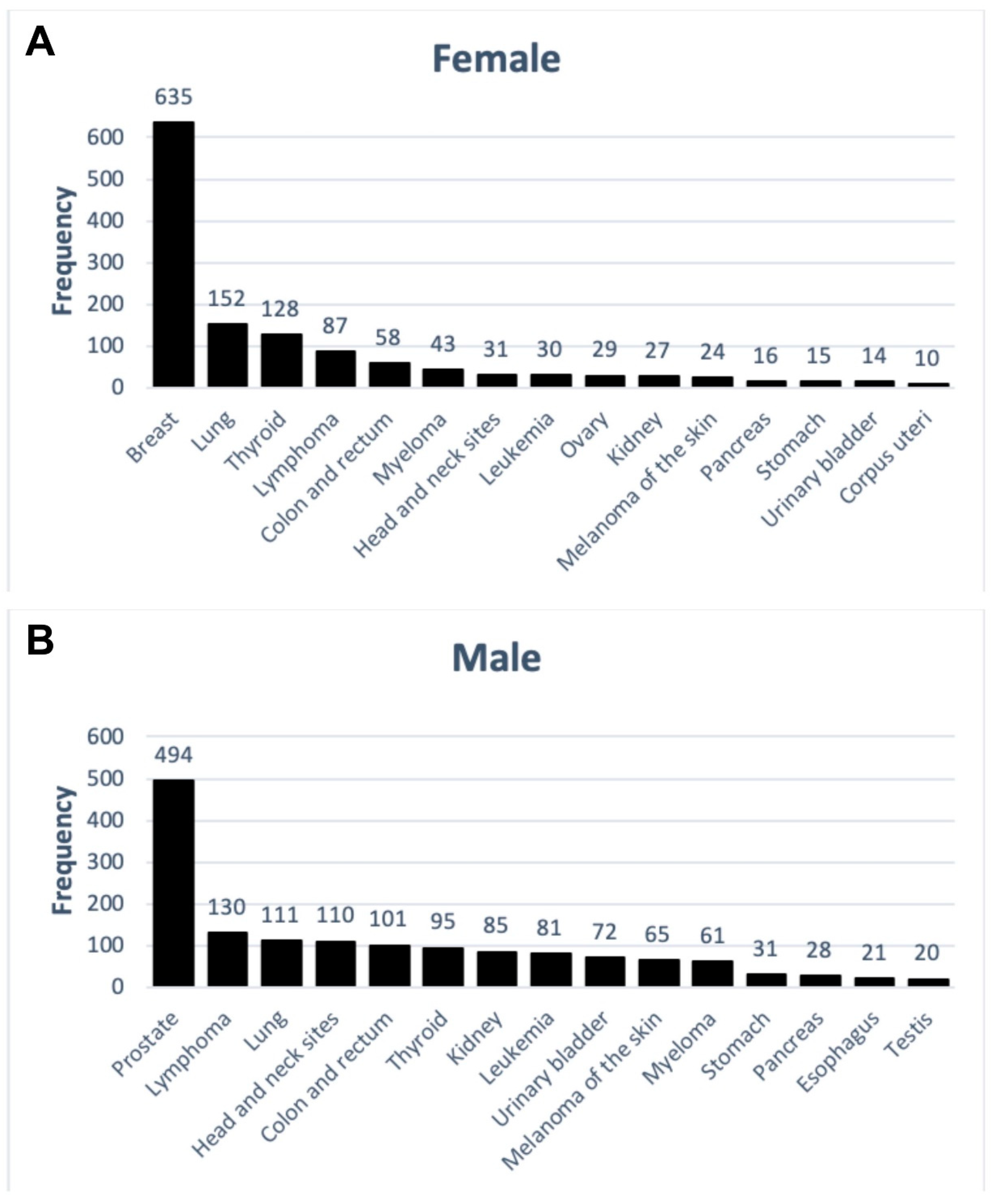
| Non-Cancer | Cancer | ||
|---|---|---|---|
| n | 5377 | 2561 | |
| Sex, n (%) | F | 2678 (49.8) | 1195 (46.7) |
| M | 2699 (50.2) | 1366 (53.3) | |
| Age on 9/11 (years), median [range] | 41.6 [0.0, 82.6] | 46.2 [0.0, 81.9] | |
| Age on 9/11 (years), n (%) | ≤20 | 309 (5.7) | 28 (1.1) |
| 21–29 | 613 (11.4) | 169 (6.6) | |
| 30–39 | 1454 (27.0) | 510 (19.9) | |
| 40–49 | 1781 (33.1) | 913 (35.7) | |
| 50–59 | 963 (17.9) | 755 (29.5) | |
| 60–69 | 202 (3.8) | 172 (6.7) | |
| 70–79 | 50 (0.9) | 13 (0.5) | |
| ≥80 | 5 (0.1) | 1 (0.0) | |
| Race/Ethnicity, n (%) | Hispanic | 1592 (30.3) | 246 (11.8) |
| NH *-White | 1992 (37.9) | 1099 (52.9) | |
| NH *-Black | 1051 (20.0) | 394 (19.0) | |
| Asian | 610 (11.6) | 334 (16.1) | |
| Native American | 14 (0.3) | 3 (0.1) | |
| BMI **, n (%) | Normal weight (<25) | 1365 (28.7) | 486 (30.3) |
| Overweight (25–30) | 1717 (36.1) | 616 (38.4) | |
| Obese (≥30) | 1671 (35.2) | 504 (31.4) | |
| Income, n (%) | ≤$30,000/year | 2842 (54.8) | 860 (42.4) |
| >$30,000/year | 2340 (45.2) | 1169 (57.6) | |
| Education, n (%) | High school or less | 1801 (33.5) | 578 (27.3) |
| More than high school | 3572 (66.5) | 1541 (72.7) | |
| Exposure information | |||
| Dust cloud exposure, n (%) | No | 2506 (47.0) | 1018 (48.1) |
| Yes | 2824 (53.0) | 1098 (51.9) | |
| Type of community members, n (%) | Local Worker Resident Student | 2766 (51.6) 1329 (24.8) 132 (2.5) | 1352 (64.0) 607 (28.8) 40 (1.9) |
| Clean-up worker | 619 (11.5) | 17 (0.8) | |
| Other | 516 (9.6) | 95 (4.5) | |
| Smoking, n (%) | Never (≤1 p-y ***) | 3661 (69.2) | 1363 (65.0) |
| Former (>1 p-y) | 1076 (20.3) | 621 (29.6) | |
| Current (>1 p-y) | 555 (10.5) | 114 (5.4) | |
| Pack year, n (%) | ≤5 p-y | 4167 (78.7) | 1513 (72.1) |
| >5 p-y | 1125 (21.3) | 585 (27.9) | |
| Number of Cancers | n | Percent (%) |
|---|---|---|
| 1 | 2226 | 86.90 |
| 2 | 299 | 11.70 |
| 3 | 31 | 1.20 |
| 4 | 4 | 0.20 |
| 5 | 1 | 0.04 |
| Total | 2561 | 100 |
| All (n = 2999) | Male (n = 1623) | Female (n = 1376) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Cancer Type | n (%) | Median Age of Diagnosis (Range) (Year) | Median Latency (Range) (Year) | n (%) | Median Age of Diagnosis (Range) (Year) | Median Latency (Range) (Year) | n (%) | Median Age of Diagnosis (Range) (Year) | Median Latency (Range) (Year) |
| Breast | 646 (22) | 55 (27, 86) | 12.4 (3.3, 17.8) | 11 (1) | 58 (41, 82) | 11.5 (4.7, 16.6) | 635 (46) | 55 (27, 86) | 12.4 (3.3, 17.8) |
| Prostate | 494 (16) | 62 (40, 83) | 12.3 (3.9, 18.2) | 494 (30) | 62 (40, 83) | 12.3 (3.9, 18.2) | |||
| Lung | 263 (9) | 64 (34, 89) | 14 (3.3, 18.1) | 111 (7) | 66 (38, 89) | 14.4 (5.3, 18.1) | 152 (11) | 63 (34, 85) | 13.7 (3.3, 18.1) |
| Thyroid | 223 (7) | 52 (13, 80) | 11.8 (2.7, 17.9) | 95 (6) | 52 (22, 75) | 12.4 (2.7, 17.7) | 128 (9) | 49 (13, 80) | 11.4 (3.0, 17.9) |
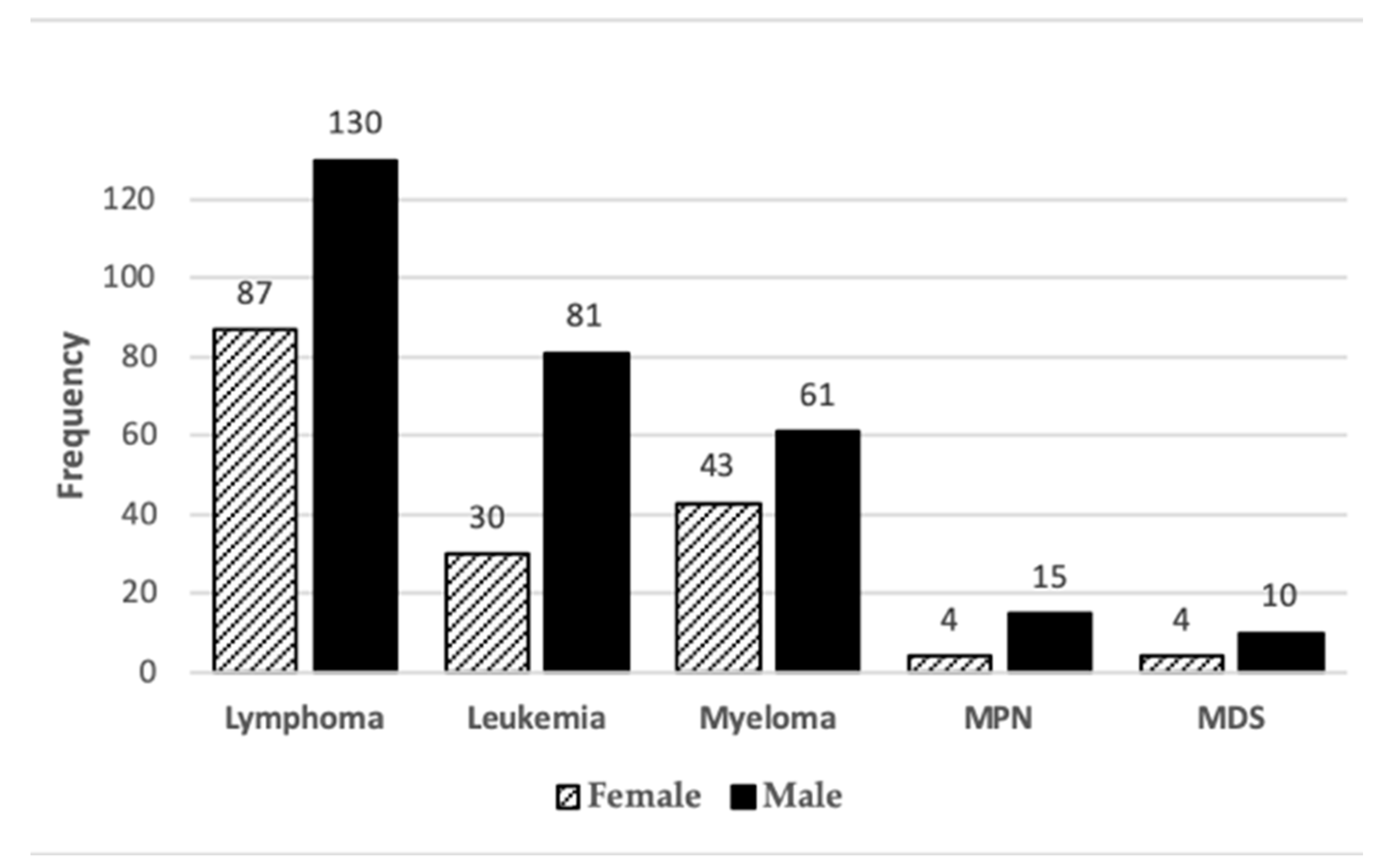
| Lymphoma | 217 (7) | 57 (21, 91) | 12 (0.9, 17.8) | 130 (8) | 56 (21, 82) | 11.8 (1.5, 17.5) | 87 (6) | 59 (22, 91) | 12.7 (0.9, 17.8) |
| Colon and rectum | 159 (5) | 59 (29, 84) | 13.4 (4.1, 18.1) | 101 (6) | 59 (36, 84) | 13.7 (4.1, 18.1) | 58 (4) | 59 (29, 82) | 12.4 (4.9, 17.2) |
| Head and neck sites | 141 (5) | 58 (26, 87) | 12.9 (3.6, 18.1) | 110 (7) | 58 (37, 87) | 11.9 (3.6, 18.1) | 31 (2) | 57 (26, 73) | 13.2 (7.7, 17.1) |
| Kidney | 112 (4) | 58 (7, 80) | 12.8 (4.2, 17.7) | 85 (5) | 58 (7, 80) | 12.7 (4.2, 17.7) | 27 (2) | 58 (32, 73) | 13.2 (6.4, 17.6) |
| Leukemia | 111 (4) | 59 (16, 81) | 12.4 (0.7, 16.8) | 81 (5) | 59 (16, 81) | 12.2 (0.7, 16.7) | 30 (2) | 58 (34, 72) | 12.6 (2.1, 16.8) |
| Myeloma | 104 (3) | 60 (28, 82) | 13.9 (3.9, 17.8) | 61(4) | 59 (28, 79) | 13.2 (5.2, 17.8) | 43 (3) | 60 (40, 82) | 14.3 (3.9, 17.7) |
| Melanoma of the skin | 89 (3) | 59 (34, 85) | 12.7 (3.2, 17.6) | 65 (4) | 60 (37, 85) | 12.8 (3.2, 17.6) | 24 (2) | 55 (34, 74) | 9.7 (4.2, 16.9) |
| Urinary bladder | 86 (3) | 65 (34, 95) | 13.2 (4.2, 17.4) | 72 (4) | 65 (34, 95) | 13.3 (4.2, 17.4) | 14 (1) | 64 (35, 75) | 12.2 (6.6, 16.8) |
| Gonad (Testis/Ovary) | 49 (2) | 52 (28, 82) | 12.2 (4.3, 17.6) | 20 (1) | 41 (28, 70) | 8.8 (4.3, 16.9) | 29 (2) | 57 (28, 82) | 13.1 (8.2, 17.6) |
| Stomach | 46 (2) | 59 (39, 75) | 13.8 (4.4, 17.8) | 31 (2) | 57 (39, 72) | 13.9 (4.4, 17.8) | 15 (1) | 60 (49, 75) | 12.4 (4.4, 17.0) |
| Pancreas | 44 (1) | 64 (46, 83) | 14.8 (5.7, 17.7) | 28 (2) | 64 (49, 83) | 14.9 (6.9, 17.7) | 16 (1) | 60 (46, 79) | 14.2 (5.7, 17.5) |
| Mesothelioma | 3 (0) | 57 (38, 70) | 15.4 (15.0, 16.7) | 2 (0) | 54 (38, 70) | 16 (15.4, 16.7) | 1 (0) | 57 (57, 57) | 15 (15.0, 15.0) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Durmus, N.; Shao, Y.; Arslan, A.A.; Zhang, Y.; Pehlivan, S.; Fernandez-Beros, M.-E.; Umana, L.; Corona, R.; Smyth-Giambanco, S.; Abbott, S.A.; et al. Characteristics of Cancer Patients in the World Trade Center Environmental Health Center. Int. J. Environ. Res. Public Health 2020, 17, 7190. https://doi.org/10.3390/ijerph17197190
Durmus N, Shao Y, Arslan AA, Zhang Y, Pehlivan S, Fernandez-Beros M-E, Umana L, Corona R, Smyth-Giambanco S, Abbott SA, et al. Characteristics of Cancer Patients in the World Trade Center Environmental Health Center. International Journal of Environmental Research and Public Health. 2020; 17(19):7190. https://doi.org/10.3390/ijerph17197190
Chicago/Turabian StyleDurmus, Nedim, Yongzhao Shao, Alan A. Arslan, Yian Zhang, Sultan Pehlivan, Maria-Elena Fernandez-Beros, Lisette Umana, Rachel Corona, Sheila Smyth-Giambanco, Sharon A. Abbott, and et al. 2020. "Characteristics of Cancer Patients in the World Trade Center Environmental Health Center" International Journal of Environmental Research and Public Health 17, no. 19: 7190. https://doi.org/10.3390/ijerph17197190
APA StyleDurmus, N., Shao, Y., Arslan, A. A., Zhang, Y., Pehlivan, S., Fernandez-Beros, M.-E., Umana, L., Corona, R., Smyth-Giambanco, S., Abbott, S. A., & Reibman, J. (2020). Characteristics of Cancer Patients in the World Trade Center Environmental Health Center. International Journal of Environmental Research and Public Health, 17(19), 7190. https://doi.org/10.3390/ijerph17197190




