Non-Malignant Respiratory Illnesses in Association with Occupational Exposure to Asbestos and Other Insulating Materials: Findings from the Alberta Insulator Cohort
Abstract
1. Introduction
2. Methodologies
2.1. Design and Participants
2.2. Questionnaire Assessment
2.3. Clinical Assessments
2.4. Statistical Analyses
2.4.1. Main Analyses
2.4.2. Secondary Analyses
3. Results
3.1. Characteristics of the Study Population
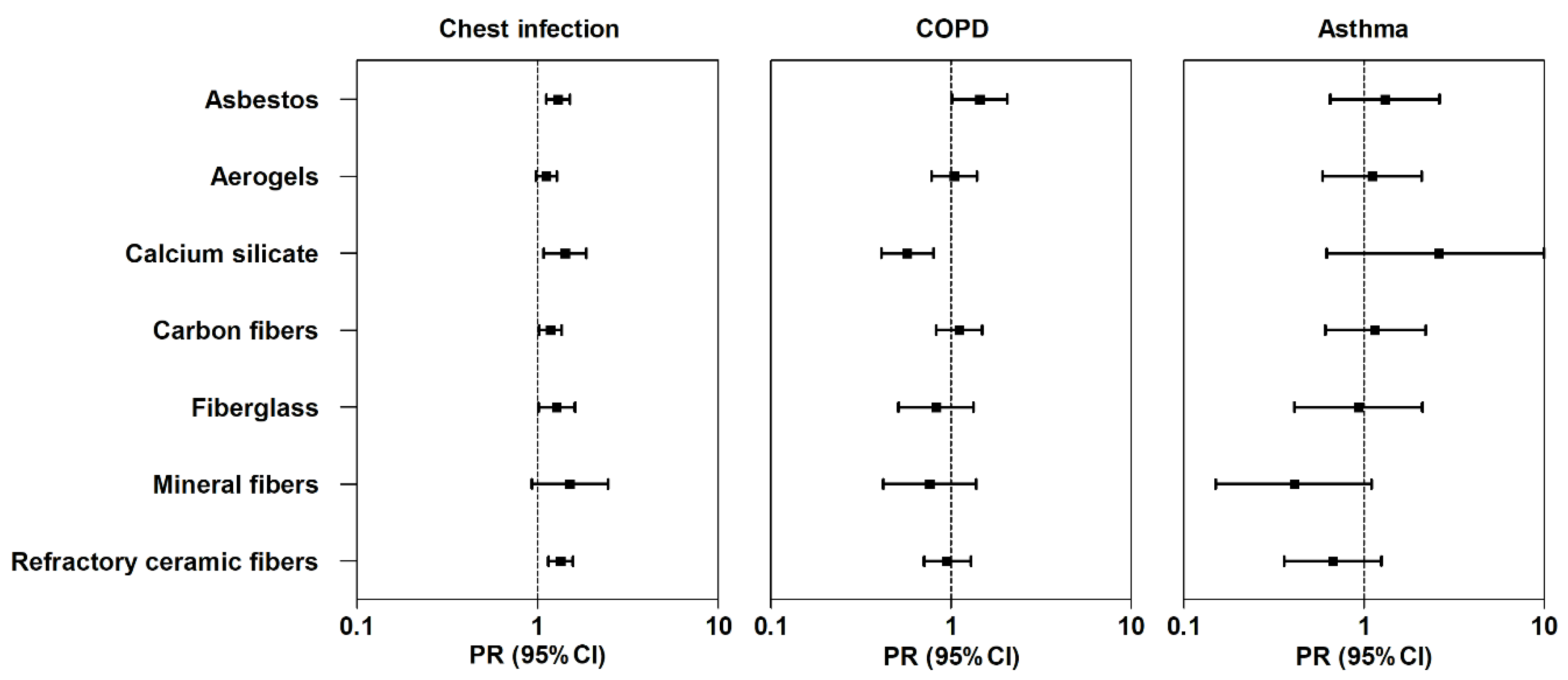
3.2. Association between Exposure and Respiratory Conditions
3.3. Sensitivity Analyses
4. Discussion
4.1. Interpretation of Results in Context of Available Evidence
4.2. Clinical and Public Health Implications
4.3. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data Sharing Policy
References
- Government of Alberta. Occupations in Alberta. Available online: https://alis.alberta.ca/occinfo/occupations-in-alberta/occupation-profiles/insulator/ (accessed on 4 April 2020).
- Moitra, S.; Puri, R.; Paul, D.; Huang, Y.C. Global perspectives of emerging occupational and environmental lung diseases. Curr. Opin. Pulm. Med. 2015, 21, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Cullinan, P.; Munoz, X.; Suojalehto, H.; Agius, R.; Jindal, S.; Sigsgaard, T.; Blomberg, A.; Charpin, D.; Annesi-Maesano, I.; Gulati, M.; et al. Occupational lung diseases: From old and novel exposures to effective preventive strategies. Lancet Respir. Med. 2017, 5, 445–455. [Google Scholar] [CrossRef]
- CAREX. Asbestos Canada. Available online: https://www.carexcanada.ca/profile/asbestos/ (accessed on 13 June 2020).
- Ruff, K. How Canada Changed from Exporting Asbestos to Banning Asbestos: The Challenges That Had to Be Overcome. Int. J. Environ. Res. Public Health 2017, 14, 1135. [Google Scholar] [CrossRef]
- Abratt, R.P.; Vorobiof, D.A.; White, N. Asbestos and mesothelioma in South Africa. Lung Cancer 2004, 45, S3–S6. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, C.; Bianchi, T. Malignant mesothelioma: Global incidence and relationship with asbestos. Ind. Health 2007, 45, 379–387. [Google Scholar] [CrossRef]
- Vangelova, K.; Dimitrova, I. Asbestos exposure and mesothelioma incidence and mortality in Bulgaria. Rev. Environ. Health 2016, 31, 203–209. [Google Scholar] [CrossRef]
- Dragani, T.A.; Colombo, F.; Pavlisko, E.N.; Roggli, V.L. Malignant mesothelioma diagnosed at a younger age is associated with heavier asbestos exposure. Carcinogenesis 2018, 39, 1151–1156. [Google Scholar] [CrossRef]
- Ohar, J.; Sterling, D.A.; Bleecker, E.; Donohue, J. Changing patterns in asbestos-induced lung disease. Chest 2004, 125, 744–753. [Google Scholar] [CrossRef]
- Delpierre, S.; Delvolgo-Gori, M.J.; Faucher, M.; Jammes, Y. High prevalence of reversible airway obstruction in asbestos-exposed workers. Arch. Environ. Health 2002, 57, 441–445. [Google Scholar] [CrossRef]
- Gothi, D.; Gahlot, T.; Sah, R.; Saxena, M.; Ojha, U.C.; Verma, A.; Spalgais, S. Asbestos-induced lung disease in small-scale clutch manufacturing workers. Indian J. Occup. Environ. Med. 2016, 20, 95. [Google Scholar] [CrossRef]
- Schwartz, D.A.; Davis, C.S.; Merchant, J.A.; Bunn, W.B.; Galvin, J.R.; Van Fossen, D.S.; Dayton, C.S.; Hunninghake, G.W. Longitudinal changes in lung function among asbestos-exposed workers. Am. J. Respir. Crit. Care Med. 1994, 150, 1243–1249. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.D. Failure to prove asbestos exposure produces obstructive lung disease. Chest 2004, 126, 1000. [Google Scholar] [CrossRef] [PubMed]
- Baur, X.; Manuwald, U.; Wilken, D. [Does long-term asbestos exposure cause an obstructive ventilation pattern?]. Pneumologie 2010, 64, 736–744. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.; Magun, B.E.; Wood, L.J. Lung inflammation caused by inhaled toxicants: A review. Int. J. Chron. Obs. Pulmon Dis. 2016, 11, 1391–1401. [Google Scholar] [CrossRef]
- Thapliyal, P.C.; Singh, K. Aerogels as Promising Thermal Insulating Materials: An Overview. J. Mater. 2014, 2014, 127049. [Google Scholar] [CrossRef]
- Long, L.-Y.; Weng, Y.-X.; Wang, Y.-Z. Cellulose Aerogels: Synthesis, Applications, and Prospects. Polymers 2018, 10, 623. [Google Scholar] [CrossRef]
- NIOSH. Health Hazard Evaluation Report: Evaluation of Aerogel Insulation Particulate at a Union Training Facility. Available online: https://www.cdc.gov/niosh/hhe/reports/pdfs/2014-0026-3230.pdf?id=10.26616/NIOSHHETA201400263230 (accessed on 31 March 2015).
- Maxim, L.D.; Utell, M.J. Review of refractory ceramic fiber (RCF) toxicity, epidemiology and occupational exposure. Inhal. Toxicol. 2018, 30, 49–71. [Google Scholar] [CrossRef]
- Kilburn, K.H.; Powers, D.; Warshaw, R.H. Pulmonary effects of exposure to fine fibreglass: Irregular opacities and small airways obstruction. Br. J. Ind. Med. 1992, 49, 714–720. [Google Scholar] [CrossRef][Green Version]
- Dumortier, P.; Gocmen, A.; Laurent, K.; Manco, A.; De Vuyst, P. The role of environmental and occupational exposures in Turkish immigrants with fibre-related disease. Eur. Respir. J. 2001, 17, 922–927. [Google Scholar] [CrossRef]
- Fireman, E. Man-made mineral fibers and interstitial lung diseases. Curr. Opin. Pulm. Med. 2014, 20, 194–198. [Google Scholar] [CrossRef]
- National Health and Nutrition Examination Survey. NHANES Questionnaires, Datasets, and Related Documentation. Available online: https://wwwn.cdc.gov/nchs/nhanes/Default.aspx (accessed on 4 April 2020).
- Miller, M.R.; Hankinson, J.; Brusasco, V.; Burgos, F.; Casaburi, R.; Coates, A.; Crapo, R.; Enright, P.; van der Grinten, C.P.; Gustafsson, P.; et al. Standardisation of spirometry. Eur. Respir. J. 2005, 26, 319–338. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.C.; Bourbeau, J.; Hernandez, P.; Chapman, K.; Cowie, R.; FitzGerald, M.J.; Aaron, S.; Marciniuk, D.D.; Maltais, F.; O’Donnell, D.E.; et al. Canadian Prediction Equations of Spirometric Lung Function for Caucasian Adults 20 to 90 Years of Age: Results from the Canadian Obstructive Lung Disease (COLD) Study and the Lung Health Canadian Environment (LHCE) Study. Can. Respir. J. 2011, 18, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Vestbo, J.; Hurd, S.S.; Agusti, A.G.; Jones, P.W.; Vogelmeier, C.; Anzueto, A.; Barnes, P.J.; Fabbri, L.M.; Martinez, F.J.; Nishimura, M.; et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am. J. Respir. Crit. Care Med. 2013, 187, 347–365. [Google Scholar] [CrossRef] [PubMed]
- Zou, G. A modified poisson regression approach to prospective studies with binary data. Am. J. Epidemiol. 2004, 159, 702–706. [Google Scholar] [CrossRef] [PubMed]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gotzsche, P.C.; Vandenbroucke, J.P.; Initiative, S. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. Lancet 2007, 370, 1453–1457. [Google Scholar] [CrossRef]
- Vehmas, T.; Pallasaho, P.; Oksa, P. Lung and pleural fibrosis in asbestos-exposed workers: A risk factor for pneumonia mortality. Epidemiol. Infect. 2012, 140, 1987–1992. [Google Scholar] [CrossRef]
- Toren, K.; Qvarfordt, I.; Bergdahl, I.A.; Jarvholm, B. Increased mortality from infectious pneumonia after occupational exposure to inorganic dust, metal fumes and chemicals. Thorax 2011, 66, 992–996. [Google Scholar] [CrossRef]
- Kilburn, K.H.; Warshaw, R.H. Abnormal lung function associated with asbestos disease of the pleura, the lung, and both: A comparative analysis. Thorax 1991, 46, 33–38. [Google Scholar] [CrossRef][Green Version]
- Wilken, D.; Garrido, M.; Manuwald, U.; Baur, X. Lung function in asbestos-exposed workers, a systematic review and meta-analysis. J. Occup. Med. Toxicol. 2011, 6, 21. [Google Scholar] [CrossRef]
- LeVan, T.D.; Koh, W.P.; Lee, H.P.; Koh, D.; Yu, M.C.; London, S.J. Vapor, dust, and smoke exposure in relation to adult-onset asthma and chronic respiratory symptoms: The Singapore Chinese Health Study. Am. J. Epidemiol. 2006, 163, 1118–1128. [Google Scholar] [CrossRef]
- Follmann, H.D.M.; Oliveira, O.N.; Lazarin-Bidoia, D.; Nakamura, C.V.; Huang, X.; Asefa, T.; Silva, R. Multifunctional hybrid aerogels: Hyperbranched polymer-trapped mesoporous silica nanoparticles for sustained and prolonged drug release. Nanoscale 2018, 10, 1704–1715. [Google Scholar] [CrossRef] [PubMed]
- Mohammadian, M.; Jafarzadeh Kashi, T.S.; Erfan, M.; Soorbaghi, F.P. Synthesis and characterization of silica aerogel as a promising drug carrier system. J. Drug Deliv. Sci. Technol. 2018, 44, 205–212. [Google Scholar] [CrossRef]
- Dong, S.; Zhang, Y.-N.; Wan, J.; Cui, R.; Yu, X.; Zhao, G.; Lin, K. A novel multifunctional carbon aerogel-coated platform for osteosarcoma therapy and enhanced bone regeneration. J. Mater. Chem. B 2020, 8, 368–379. [Google Scholar] [CrossRef] [PubMed]
- Maxim, L.D.; Niebo, R.; Utell, M.J.; McConnell, E.E.; LaRosa, S.; Segrave, A.M. Wollastonite toxicity: An update. Inhal. Toxicol. 2014, 26, 95–112. [Google Scholar] [CrossRef]
- Tátrai, E.; Kováčiková, Z.; Brózik, M.; Six, É. Pulmonary toxicity of wollastonite in vivo and in vitro. J. Appl. Toxicol. 2004, 24, 147–154. [Google Scholar] [CrossRef]
- Hanke, W.; Sepulveda, M.J.; Watson, A.; Jankovic, J. Respiratory morbidity in wollastonite workers. Occup. Environ. Med. 1984, 41, 474–479. [Google Scholar] [CrossRef]
- Walling, B.E.; Lau, G.W. Perturbation of pulmonary immune functions by carbon nanotubes and susceptibility to microbial infection. J. Microbiol. 2014, 52, 227–234. [Google Scholar] [CrossRef]
- Kobayashi, N.; Izumi, H.; Morimoto, Y. Review of toxicity studies of carbon nanotubes. J. Occup. Health 2017, 59, 394–407. [Google Scholar] [CrossRef]
- Shvedova, A.A.; Fabisiak, J.P.; Kisin, E.R.; Murray, A.R.; Roberts, J.R.; Tyurina, Y.Y.; Antonini, J.M.; Feng, W.H.; Kommineni, C.; Reynolds, J.; et al. Sequential Exposure to Carbon Nanotubes and Bacteria Enhances Pulmonary Inflammation and Infectivity. Am. J. Respir. Cell Mol. Biol. 2008, 38, 579–590. [Google Scholar] [CrossRef]
- Beyeler, S.; Steiner, S.; Wotzkow, C.; Tschanz, S.A.; Adhanom Sengal, A.; Wick, P.; Haenni, B.; Alves, M.P.; von Garnier, C.; Blank, F. Multi-walled carbon nanotubes activate and shift polarization of pulmonary macrophages and dendritic cells in an in vivo model of chronic obstructive lung disease. Nanotoxicology 2020, 14, 77–96. [Google Scholar] [CrossRef]
- De Vuyst, P.; Dumortier, P.; Swaen, G.M.; Pairon, J.C.; Brochard, P. Respiratory health effects of man-made vitreous (mineral) fibres. Eur. Respir. J. 1995, 8, 2149–2173. [Google Scholar] [CrossRef]
- Nayebzadeh, A.; Dufresne, A.; Case, B.; Vali, H.; Williams-Jones, A.E.; Martin, R.; Normand, C.; Clark, J. Lung mineral fibers of former miners and millers from Thetford-Mines and asbestos regions: A comparative study of fiber concentration and dimension. Arch. Environ. Health 2001, 56, 65–76. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, C.; Buck, B.; Miller, A.; Lockey, J.; Weis, C.; Weissman, D.; Jonesi, A.; Ryan, P. Exposure to naturally occurring mineral fibers due to off-road vehicle use: A review. Int. J. Hyg. Environ. Health 2017, 220, 1230–1241. [Google Scholar] [CrossRef] [PubMed]
- Naccache, J.M.; Monnet, I.; Nunes, H.; Billon-Galland, M.A.; Pairon, J.C.; Guillon, F.; Valeyre, D. Anthracofibrosis attributed to mixed mineral dust exposure: Report of three cases. Thorax 2008, 63, 655–657. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, K.; Brown, R.C.; Brown, G.M. New perspectives on basic mechanisms in lung disease. 5. Respirable industrial fibres: Mechanisms of pathogenicity. Thorax 1993, 48, 390–395. [Google Scholar] [CrossRef]
- Lockey, J.E.; Levin, L.S.; Lemasters, G.K.; McKay, R.T.; Rice, C.H.; Hansen, K.R.; Papes, D.M.; Simpson, S.; Medvedovic, M. Longitudinal estimates of pulmonary function in refractory ceramic fiber manufacturing workers. Am. J. Respir. Crit. Care Med. 1998, 157, 1226–1233. [Google Scholar] [CrossRef]
- Cowie, H.A.; Wild, P.; Beck, J.; Auburtin, G.; Piekarski, C.; Massin, N.; Cherrie, J.W.; Hurley, J.F.; Miller, B.G.; Groat, S.; et al. An epidemiological study of the respiratory health of workers in the European refractory ceramic fibre industry. Occup. Environ. Med. 2001, 58, 800–810. [Google Scholar] [CrossRef]
- Trethowan, W.N.; Burge, P.S.; Rossiter, C.E.; Harrington, J.M.; Calvert, I.A. Study of the respiratory health of employees in seven European plants that manufacture ceramic fibres. Occup. Environ. Med. 1995, 52, 97–104. [Google Scholar] [CrossRef]
- Lemasters, G.K.; Lockey, J.E.; Levin, L.S.; McKay, R.T.; Rice, C.H.; Horvath, E.P.; Papes, D.M.; Lu, J.W.; Feldman, D.J. An industry-wide pulmonary study of men and women manufacturing refractory ceramic fibers. Am. J. Epidemiol. 1998, 148, 910–919. [Google Scholar] [CrossRef]
- Pahwa, P.; Karunanayake, C.P.; Rennie, D.C.; Lawson, J.A.; Ramsden, V.R.; McMullin, K.; Gardipy, P.J.; MacDonald, J.; Abonyi, S.; Episkenew, J.-A.; et al. Prevalence and associated risk factors of chronic bronchitis in First Nations people. BMC Pulm. Med. 2017, 17. [Google Scholar] [CrossRef]
- Statistics Canada. Table 13-10-0096-19 Chronic Obstructive Pulmonary Disease (COPD), 35 Years and Over. Available online: https://www150.statcan.gc.ca/t1/tbl1/en/tv.action?pid=1310009619 (accessed on 3 September 2020).
| N = 990 | |
|---|---|
| Demographics | |
| Sex (male), n (%) | 875 (88) |
| Age (years), mean (SD) | 45 (14) |
| 1st quartile (n = 239) | 28 (4) |
| 2nd quartile (n = 237) | 39 (4) |
| 3rd quartile (n = 256) | 51 (3) |
| 4th quartile (n = 258) | 62 (6) |
| BMI (kg/m2), mean (SD) | 29.3 (5.5) |
| Ethnicity, n (%) | |
| Caucasian | 792 (80) |
| African/Afro-American | 63 (6) |
| Asian/Middle-Eastern | 38 (4) |
| Hispanic | 65 (7) |
| Aboriginal | 32 (3) |
| Education, n (%) | |
| Up to high school | 163 (16) |
| Trade school | 490 (50) |
| College or beyond | 337 (34) |
| Marital status, n (%) | |
| Married/common law-partner | 592 (60) |
| Smoking history | |
| Never smokers, n (%) | 332 (34) |
| Overall pack-years, median (IQR) | 4.7 (0, 18) |
| Job history (years), median (IQR) | 13 (4, 29) |
| Passively smoke exposure at childhood, n (%) | 661 (67) |
| Parental lung disease, n (%) | 250 (25) |
| Family history of cancer, n (%) | 285 (29) |
| Clinical features | |
| Allergies, n (%) | |
| Any allergies | 416 (42) |
| Hay fever † | 111 (11) |
| Eczema † | 46 (5) |
| Hives † | 36 (4) |
| Other allergies † | 340 (34) |
| FEV1 (% predicted), mean (SD) | 94.0 (15.8) |
| FVC (% predicted), mean (SD) | 100.3 (15.1) |
| FEV1/FVC, mean (SD) | 76.7 (8.5) |
| Exposure details | |
| Ever exposed to asbestos, n (%) | 547 (55) |
| Ever exposed to aerogel, n (%) | 392 (40) |
| Ever exposed to calcium silicate, n (%) | 877 (89) |
| Ever exposed to carbon fibers, n (%) | 597 (60) |
| Ever exposed to fiberglass, n (%) | 849 (86) |
| Ever exposed to mineral fibers, n (%) | 950 (96) |
| Ever exposed to refractory ceramic fibers, n (%) | 632 (64) |
| Use of PPE for | |
| Asbestos, n (%) | 276 (28) |
| Aerogels, n (%) | 303 (31) |
| Calcium silicate, n (%) | NA |
| Carbon fibers, n (%) | NA |
| Fiberglass, n (%) | 105 (11) |
| Mineral fibers, n (%) | 148 (15) |
| Refractory ceramic fibers, n (%) | NA |
| Respiratory conditions | |
| Having a chest infection within past 3 years, n (%) | 457 (46) |
| Frequency of chest infection within past 3 years, median (IQR) ‡ | 2 (1, 3) |
| COPD, n (%) | 156 (16) |
| Asthma, n (%) | 40 (4) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moitra, S.; Farshchi Tabrizi, A.; Idrissi Machichi, K.; Kamravaei, S.; Miandashti, N.; Henderson, L.; Mukherjee, M.; Khadour, F.; Naseem, M.T.; Lacy, P.; et al. Non-Malignant Respiratory Illnesses in Association with Occupational Exposure to Asbestos and Other Insulating Materials: Findings from the Alberta Insulator Cohort. Int. J. Environ. Res. Public Health 2020, 17, 7085. https://doi.org/10.3390/ijerph17197085
Moitra S, Farshchi Tabrizi A, Idrissi Machichi K, Kamravaei S, Miandashti N, Henderson L, Mukherjee M, Khadour F, Naseem MT, Lacy P, et al. Non-Malignant Respiratory Illnesses in Association with Occupational Exposure to Asbestos and Other Insulating Materials: Findings from the Alberta Insulator Cohort. International Journal of Environmental Research and Public Health. 2020; 17(19):7085. https://doi.org/10.3390/ijerph17197085
Chicago/Turabian StyleMoitra, Subhabrata, Ali Farshchi Tabrizi, Kawtar Idrissi Machichi, Samineh Kamravaei, Noushin Miandashti, Linda Henderson, Manali Mukherjee, Fadi Khadour, Muhammad T. Naseem, Paige Lacy, and et al. 2020. "Non-Malignant Respiratory Illnesses in Association with Occupational Exposure to Asbestos and Other Insulating Materials: Findings from the Alberta Insulator Cohort" International Journal of Environmental Research and Public Health 17, no. 19: 7085. https://doi.org/10.3390/ijerph17197085
APA StyleMoitra, S., Farshchi Tabrizi, A., Idrissi Machichi, K., Kamravaei, S., Miandashti, N., Henderson, L., Mukherjee, M., Khadour, F., Naseem, M. T., Lacy, P., & Melenka, L. (2020). Non-Malignant Respiratory Illnesses in Association with Occupational Exposure to Asbestos and Other Insulating Materials: Findings from the Alberta Insulator Cohort. International Journal of Environmental Research and Public Health, 17(19), 7085. https://doi.org/10.3390/ijerph17197085







