Sources of and Solutions to Toxic Metal and Metalloid Contamination in Small Rural Drinking Water Systems: A Rapid Review
Abstract
1. Introduction
2. Materials and Methods
- Identify reviews that include relevant information.
- Identify grey literature that includes or synthesizes relevant information.
- Screen identified literature for relevance, emphasizing the evidence most relevant to small rural drinking water systems.
- Synthesize all relevant literature included after steps 1–3.
3. Results
3.1. Search Results
3.2. Sources of Toxic Metals and Metalloids in Drinking Water
3.3. Solutions to Prevent and Correct Toxic Metals and Metalloids in Drinking Water
3.3.1. Prevention: Best Practices for Siting and Planning New Water Sources
3.3.2. Prevention: Preventing and Reducing Catchment Pollution
3.3.3. Prevention: Using Appropriate Parts and Materials
3.3.4. Prevention: Best Practices for Installation, Construction, and Maintenance
3.3.5. Prevention: Conditioning Water
3.3.6. Correction: Source Substitution/Blending
3.3.7. Correction: Treating Water
3.3.8. Correction: Correcting Existing Pollution
3.3.9. Correction: Replacing, Modifying, or Cleaning Parts
4. Discussion
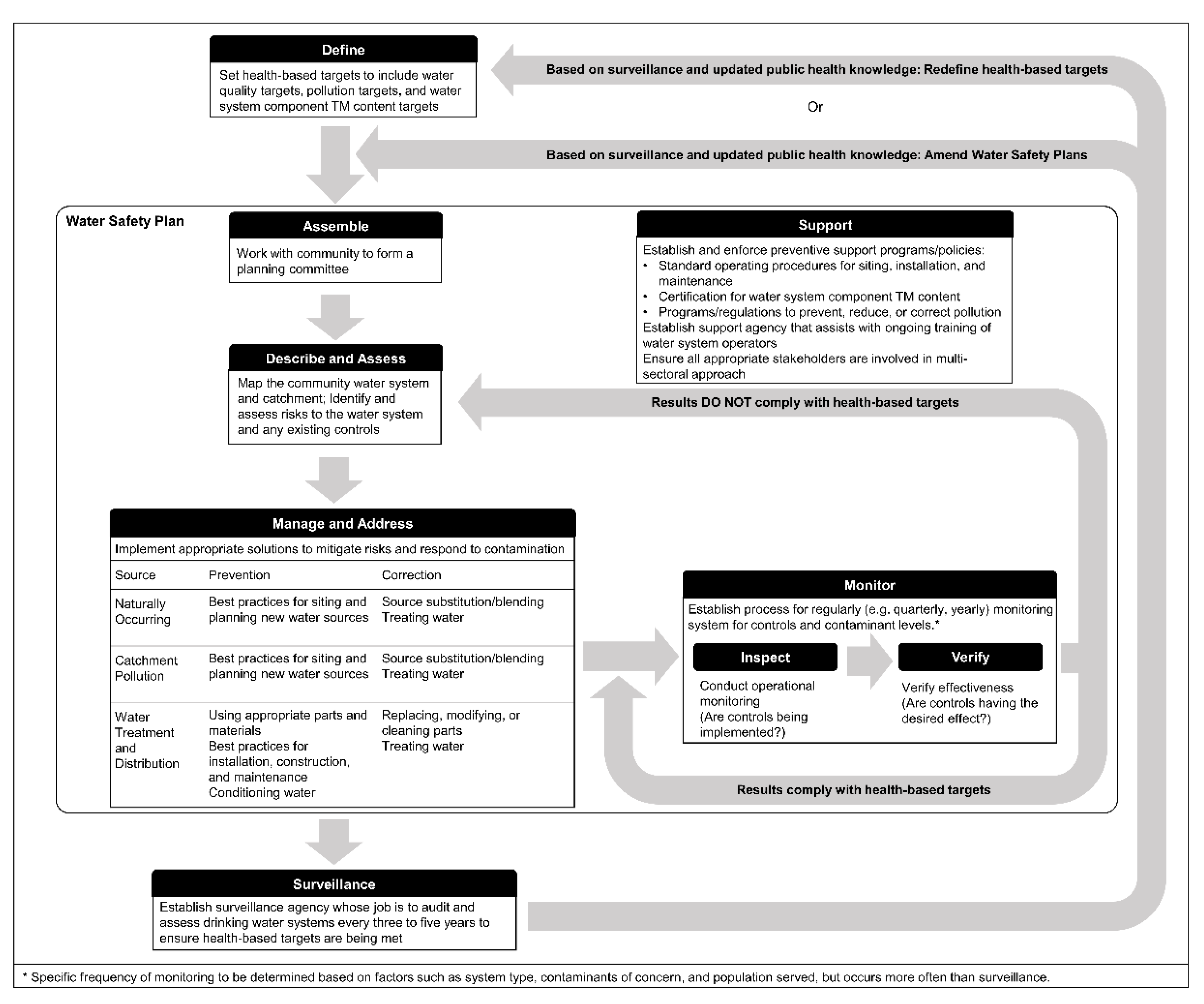
4.1. Toxic Metal and Metalloid Management Framework
4.2. Limitations of the Review
4.3. Next Steps
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lim, S.S.; Vos, T.; Flaxman, A.D.; Danaei, G.; Shibuya, K.; Adair-Rohani, H.; A AlMazroa, M.; Amann, M.; Anderson, H.R.; Andrews, K.G.; et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2224–2260. [Google Scholar] [CrossRef]
- Thompson, T.; Fawell, J.; Kunikane, S.; Jackson, D.; Appleyard, S.; Callan, P.; Bartram, J.; Kingston, P. Chemical Safety of Drinking Water: Assessing Priorities for Risk Management; WHO: Geneva, Switzerland, 2007. [Google Scholar]
- Brown, M.J.; Margolis, S. Lead in drinking water and human blood lead levels in the United States. Morb. Mortal. Wkly. Rep. 2012, 61 (Suppl. S61), 1–9. [Google Scholar]
- World Health Organization. Health Aspects of Plumbing; WHO: Geneva, Switzerland, 2006. [Google Scholar]
- World Health Organization. Developing Drinking-Water Quality Regulations and Standards: General Guidance with a Special Focus on Countries with Limited Resources; WHO: Geneva, Switzerland, 2018. [Google Scholar]
- Khalequzzaman, M.; Faruque, F.; Mitra, A. Assessment of Arsenic Contamination of Groundwater and Health Problems in Bangladesh. Int. J. Environ. Res. Public Health 2005, 2, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Belabed, B.E.; Meddour, A.; Samraoui, B.; Chenchouni, H. Modeling seasonal and spatial contamination of surface waters and upper sediments with trace metal elements across industrialized urban areas of the Seybouse watershed in North Africa. Environ. Monit. Assess. 2017, 189, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Tricco, A.C.; Langlois, E.V.; Straus, S.E. Rapid Reviews to Strengthen Health Policy and Systems: A Practical Guide; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- Bower, M.; Hayes, C. Best Practice Guide on the Management of Metals in Small Water Supplies; IWA: London, UK, 2016. [Google Scholar]
- World Health Organization. Selenium in Drinking-Water: Background Document for Development of WHO Guidelines for Drinking-Water Quality; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Arsenic in Drinking Water. Available online: https://www.lboro.ac.uk/research/wedc/well/water-supply/ws-factsheets/arsenic/ (accessed on 18 June 2019).
- World Health Organization. Arsenic in Drinking-Water: Background Document for Development of WHO Guidelines for Drinking-Water Quality; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- World Health Organization. Manganese in Drinking-Water: Background Document for Development of WHO Guidelines for Drinking-Water Quality; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- World Health Organization. Uranium in Drinking-Water: Background Document for Development of WHO Guidelines for Drinking-Water Quality; WHO: Geneva, Switzerland, 2012. [Google Scholar]
- World Health Organization. Protecting Surface Water for Health: Identifying, Assessing and Managing Drinking-Water Quality Risks in Surface-Water Catchments; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- Committee on Environmental Health and Committee on Infectious Diseases. Drinking water from private wells and risks to children. Pediatrics 2009, 123, 1599–1605. [Google Scholar] [CrossRef] [PubMed]
- Stewart, C.; Johnston, D.; Leonard, G.; Horwell, C.; Thordarson, T.; Cronin, S. Contamination of water supplies by volcanic ashfall: A literature review and simple impact modelling. J. Vulcanol. Geotherm. Res. 2006, 158, 296–306. [Google Scholar] [CrossRef]
- Mushak, P. Potential impact of acid precipitation on arsenic and selenium. Environ. Health Perspect. 1985, 63, 105–113. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Davison, A.; Howard, G.; Stevens, M.; Callan, P.; Fewtrell, L.; Deere, D.; Bartram, J. Water Safety Plans: Managing Drinking-Water Quality from Catchment to Consumer; WHO: Geneva, Switzerland, 2005. [Google Scholar]
- World Bank. Pollution Prevention and Abatement Handbook, 1998: Toward Cleaner Production; World Bank: Washington, DC, USA, 1999. [Google Scholar]
- Schmoll, O.; Howard, G.; Chilton, J.; Chorus, I. Protecting Groundwater for Health: Managing the Quality of Drinking-Water Sources; WHO: Geneva, Switzerland, 2006. [Google Scholar]
- Foster, S.; Hirata, R.; Gomes, D.; D’Elia, M.; Paris, M. Groundwater Quality Protection: A Guide for Water Service Companies, Municipal Authorities, and Environment Agencies; World Bank: Washington, DC, USA, 2002. [Google Scholar]
- Schwarzenbach, R.P.; Egli, T.; Hofstetter, T.B.; von Gunten, U.; Wehrli, B. Global water pollution and human health. Annu. Rev. Environ. Resour. 2010, 35, 109–136. [Google Scholar] [CrossRef]
- Triantafyllidou, S.; Edwards, M. Lead (Pb) in tap water and in blood: Implications for lead exposure in the United States. Crit. Rev. Environ. Sci. Technol. 2012, 42, 1297–1352. [Google Scholar] [CrossRef]
- Gonzalez, S.; Lopez-Roldan, R.; Cortina, J.-L. Presence of metals in drinking water distribution networks due to pipe material leaching: A review. Toxicol. Environ. Chem. 2013, 95, 870–889. [Google Scholar] [CrossRef]
- Dietrich, A.M.; Burlingame, G.A. Critical review and rethinking of USEPA secondary standards for maintaining organoleptic quality of drinking water. Environ. Sci. Technol. 2015, 49, 708–720. [Google Scholar] [CrossRef] [PubMed]
- Merkel, T.; Pehkonen, S. General corrosion of copper in domestic drinking water installations: Scientific background and mechanistic understanding. Corros. Eng. Sci. Technol. 2006, 41, 21–37. [Google Scholar] [CrossRef]
- World Health Organization. Lead in Drinking-Water: Background Document for Development of WHO Guidelines for Drinking-Water Quality; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- World Health Organization. Cadmium in Drinking-Water: Background Document for Development of WHO Guidelines for Drinking-Water Quality; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- World Health Organization. Guidelines for Drinking-Water Quality: First Addendum to the Fourth Edition; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- Liu, G.; Zhang, Y.; Knibbe, W.-J.; Feng, C.; Liu, W.; Medema, G.; van der Meer, W. Potential impacts of changing supply-water quality on drinking water distribution: A review. Water Res. 2017, 116, 135–148. [Google Scholar] [CrossRef] [PubMed]
- Armand, H.; Stoianov, I.; Graham, N. A holistic assessment of discolouration processes in water distribution networks. Urban Water J. 2017, 14, 263–277. [Google Scholar] [CrossRef]
- McNeill, L.; Edwards, M. Iron pipe corrosion in distribution systems. J. Am. Water Works Assoc. 2001, 93, 88–100. [Google Scholar] [CrossRef]
- Sarin, P.; Snoeyink, V.; Lytle, D.; Kriven, W. Iron corrosion scales: Model for scale growth, iron release, and colored water formation. J. Environ. Eng. 2004, 130, 364–373. [Google Scholar] [CrossRef]
- Farooqi, O.; Loganathan, G.; Edwards, M.; Bosch, D.; Lee, J.; Scardina, P. Copper pinhole failures: Plumbing susceptibility and management. J. Water Resour. Plan. Manag. 2009, 135, 227–236. [Google Scholar] [CrossRef]
- Vargas, I.T.; Fischer, D.A.; Alsina, M.A.; Pavissich, J.P.; Pasten, P.A.; Pizarro, G.E. Copper corrosion and biocorrosion events in premise plumbing. Materials 2017, 10, 1036. [Google Scholar] [CrossRef]
- Furey, S. Handpumps: Where now? A Synthesis of Online Discussions (2012–2014); Skat Foundation, RWSN: St. Gallen, Switzerland, 2014. [Google Scholar]
- World Health Organization. Water Safety in Distribution Systems; WHO: Geneva, Switzerland, 2014. [Google Scholar]
- Danert, K.; Armstrong, T.; Adekile, D.; Duffau, B.; Ouedraogo, I.; Kwei, C. Code of Practice for Cost Effective Boreholes; RWSN: St. Gallen, Switzerland, 2010. [Google Scholar]
- World Health Organization. Water Safety Planning for Small Community Water Supplies: Step-by-Step Risk Management Guidance for Drinking-Water Supplies in Small Communities; WHO: Geneva, Switzerland, 2012. [Google Scholar]
- Batstone, R.; Smith, J.E.J.; Wilson, D. The safe Disposal of Hazardous Wastes: The Special Needs and Problems of Developing Countries; World Health Organization; UN Environment Programme; World Bank: Washington, DC, USA, 1989. [Google Scholar]
- Bernstein, J.D. Alternative Approaches to Pollution Control and Waste Management: Regulatory and Economic Instruments; World Bank: Washington, DC, USA, 1993. [Google Scholar]
- Tuinhof, A.; Foster, S.; van Steenbergen, F.; Talbi, A.; Wishart, M. Appropriate Groundwater Management Policy for Sub-Saharan Africa: In Face of Demographic Pressure and Climatic Variability; Water Papers; World Bank: Washington, DC, USA, 2011. [Google Scholar]
- IAPMO. Uniform Plumbing Code, 28th ed.; IAPMO: Ontario, CA, USA, 2018. [Google Scholar]
- Rigal, S.; Baron, J. State of the art in organic piping systems: Alternative solutionis for avoiding internal corrosion. Water Sci. Technol. Water Supply 2001, 1, 33–38. [Google Scholar] [CrossRef]
- Stern, B.R.; Lagos, G. Are there health risks from the migration of chemical substances from plastic pipes into drinking water? A review. Hum. Ecol. Risk Assess. 2008, 14, 753–779. [Google Scholar] [CrossRef]
- Roy, S.; Coyne, J.M.; Novak, J.A.; Edwards, M.A. Flow-induced failure mechanisms of copper pipe in potable water systems. Corros. Rev. 2018, 36, 449–481. [Google Scholar] [CrossRef]
- Danert, K. What are We Talking About? Experiences and Ideas from RWSN’s Sustainable Groundwater Community; RWSN: St. Gallen, Switzerland, 2013. [Google Scholar]
- Hussain, A.-K.M.; Sanoussi, A.; Hussain, H. Pollution of drinking water transported by corroded metallic pipelines. Water Pollut. X 2010, 135, 61–69. [Google Scholar]
- Maas, R.P.; Patch, S.C.; Morgan, D.M.; Pandolfo, T.J. Reducing lead exposure from drinking water: Recent history and current status. Public Health Rep. 2005, 120, 316–321. [Google Scholar] [CrossRef] [PubMed]
- Pfadenhauer, L.M.; Burns, J.; Rohwer, A.; Rehfuess, E.A. Effectiveness of interventions to reduce exposure to lead through consumer products and drinking water: A systematic review. Environ. Res. 2016, 147, 525–536. [Google Scholar] [CrossRef]
- Edmunds, W.; Ahmed, K.; Whitehead, P. A review of arsenic and its impacts in groundwater of the Ganges-Brahmaputra-Meghna delta, Bangladesh. Environ. Sci. Process. Impacts 2015, 17, 1032–1046. [Google Scholar] [CrossRef]
- Ambashta, R.D.; Sillanpaa, M. Water purification using magnetic assistance: A review. J. Hazard. Mater. 2010, 180, 38–49. [Google Scholar] [CrossRef]
- Brown, K.W.; Gessesse, B.; Butler, L.J.; MacIntosh, D.L. Potential effectiveness of point-of-use filtration to address risks to drinking water in the United States. Environ. Health Insights 2017, 11, 1–8. [Google Scholar] [CrossRef]
- Chappells, H.; Parker, L.; Fernandez, C.V.; Conrad, C.; Drage, J.; O’Toole, G.; Campbell, N.; Dummer, T.J.B. Arsenic in private drinking water wells: An assessment of jurisdictional regulations and guidelines for risk remediation in North America. J. Water Health 2014, 12, 372–392. [Google Scholar] [CrossRef]
- Hartmann, P. Iron & Manganese Removal: Simple Methods for Drinking Water; Working Papers on Water Supply and Environmental Sanitation; Skat: St. Gallen, Switzerland, 2001. [Google Scholar]
- Mhlanga, S.D.; Nxumalo, E.N. Advances in nanotechnologies for point-of-use and point-of-entry water purification. In Application of Nanotechnology in Water Research; Mishra, A.K., Ed.; Scrivener Publishing LLC: Salem, MA, USA, 2014; pp. 229–267. [Google Scholar]
- Mukherjee, D.C.; Chowdhury, S.B.; Paul, A.; Das, P. A review on arsenic pollution in West Bengal and Bihar: Cause, effects and remedial measures following latest technology. J. Indian Chem. Soc. 2012, 89, 9–18. [Google Scholar]
- Patil, A.; Rao, M. Low cost adsorption, phytoremediation and sludge management technologies of arsenic remediation adoptable in India: A review. Nat. Environ. Pollut. Technol. 2014, 13, 473–481. [Google Scholar]
- Malik, A.H.; Khan, Z.M.; Mahmood, Q.; Nasreen, S.; Bhatti, Z.A. Perspectives of low cost arsenic remediation of drinking water in Pakistan and other countries. J. Hazard. Mater. 2009, 168, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Pandit, A.B.; Kumar, J.K. Clean water for developing countries. Annu. Rev. Chem. Biomol. Eng. 2015, 6, 217–246. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, A.; Paul, B. The global menace of arsenic and its conventional remediation: A critical review. Chemosphere 2016, 158, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Boyd, G.R.; Tarbert, N.K.; Oliphant, R.J.; Kirmeyer, G.J.; Murphy, B.M.; Serpente, R.F. Lead pipe rehabilitation and replacement techniques for drinking water service: Review of available and emerging technologies. Tunn. Undergr. Space Technol. 2000, 15, 13–24. [Google Scholar] [CrossRef]
- Sarver, E.; Zhang, Y.; Edwards, M. Review of brass dezincification corrosion in potable water systems. Corros. Rev. 2010, 28, 155–196. [Google Scholar] [CrossRef]
- Postawa, A. Best Practice Guide on Sampling and Monitoring of Metals in Drinking Water; IWA: London, UK, 2012. [Google Scholar]
- Tang, X.; Po-Yen, W.; Buchter, G. Ion-selective electrodes for detection of lead (II) in drinking water: A mini-review. Environments 2018, 5, 95. [Google Scholar] [CrossRef]

| Keywords | Search Terms |
|---|---|
| Water | Water, (Drink* OR Potable) |
| Source Type | tap, tap stand, piped, standpipe, spigot, hand pump, handpump, borehole, tubewell, tube well, private well |
| Toxic Metals and Metalloids | toxic metal, trace metal, heavy metal, metal, antimony, arsenic, cadmium, chromium, copper, iron, lead, manganese, mercury, nickel, selenium, tin, uranium, zinc |
| Reviews | review, systematic review, literature review, meta-analysis, meta analysis, guide |
| Inclusion Criteria | Discusses or reports on metals or metalloids of interest: antimony, arsenic, cadmium, chromium, copper, iron, lead, manganese, mercury, nickel, selenium, tin, uranium, zinc Discusses source of TM contamination and/or solutions for prevention/correction of contamination Relevant to a drinking water source (or a source likely to be used for drinking such as groundwater or fresh surface water documented as being a source of drinking water for human settlements) Relevant or applicable to small rural drinking water systems Subject to peer review, external review (articles); based on expert consensus/review (book chapters, reports, etc.) Written in English |
| Exclusion Criteria | Duplicate Case study (descriptive study unable to be generalized because of small scale or unique sociopolitical/geographical context) Focuses exclusively on risk assessment and/or health outcomes rather than sources of and solutions to contamination Is a commentary, editorial, opinion piece, speech, news article, or minutes from a workshop or meeting Has been superseded by a later publication or edition Has been redacted to the point of being unusable Unavailable in English |
| Metal/Metalloid | Potential Sources of Contamination | |||
|---|---|---|---|---|
| Naturally Occurring | Catchment Pollution | Treatment and Distribution | ||
| Industrial | Human Settlements | |||
| Antimony | X | X | X | |
| Arsenic | X | X | ||
| Cadmium | X | X | X | |
| Chromium | X | X | ||
| Copper | X | X | X | |
| Iron | X | X | X | |
| Lead | X | X | X | |
| Manganese | X | X | ||
| Mercury | X | |||
| Nickel | X | X | X | X |
| Selenium | X | X | ||
| Tin | X | X | ||
| Uranium | X | X | ||
| Zinc | X | X | X | |
| Source | Prevention | Correction |
|---|---|---|
| Naturally occurring | Best practices for siting and planning new water sources | Source substitution/blending Treating water |
| Catchment pollution | Best practices for siting and planning new water sources Preventing and reducing catchment pollution | Correcting existing pollution Source substitution/blending Treating water |
| Water treatment and distribution | Using appropriate parts and materials Best practices for installation, construction, and maintenance Conditioning water | Replacing, modifying, or cleaning parts Treating water |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tracy, J.W.; Guo, A.; Liang, K.; Bartram, J.; Fisher, M. Sources of and Solutions to Toxic Metal and Metalloid Contamination in Small Rural Drinking Water Systems: A Rapid Review. Int. J. Environ. Res. Public Health 2020, 17, 7076. https://doi.org/10.3390/ijerph17197076
Tracy JW, Guo A, Liang K, Bartram J, Fisher M. Sources of and Solutions to Toxic Metal and Metalloid Contamination in Small Rural Drinking Water Systems: A Rapid Review. International Journal of Environmental Research and Public Health. 2020; 17(19):7076. https://doi.org/10.3390/ijerph17197076
Chicago/Turabian StyleTracy, J. Wren, Amy Guo, Kaida Liang, Jamie Bartram, and Michael Fisher. 2020. "Sources of and Solutions to Toxic Metal and Metalloid Contamination in Small Rural Drinking Water Systems: A Rapid Review" International Journal of Environmental Research and Public Health 17, no. 19: 7076. https://doi.org/10.3390/ijerph17197076
APA StyleTracy, J. W., Guo, A., Liang, K., Bartram, J., & Fisher, M. (2020). Sources of and Solutions to Toxic Metal and Metalloid Contamination in Small Rural Drinking Water Systems: A Rapid Review. International Journal of Environmental Research and Public Health, 17(19), 7076. https://doi.org/10.3390/ijerph17197076






