Factors Associated with Latent Tuberculosis Infection among the Hospital Employees in a Tertiary Hospital of Northeastern Thailand
Abstract
1. Introduction
2. Methods
2.1. Study Design
2.2. Study Population and Samples
2.2.1. Case Definition
2.2.2. Control Definition
2.3. Study Tool and Data Collection
2.4. Data Analyses
2.5. Operational Definitions
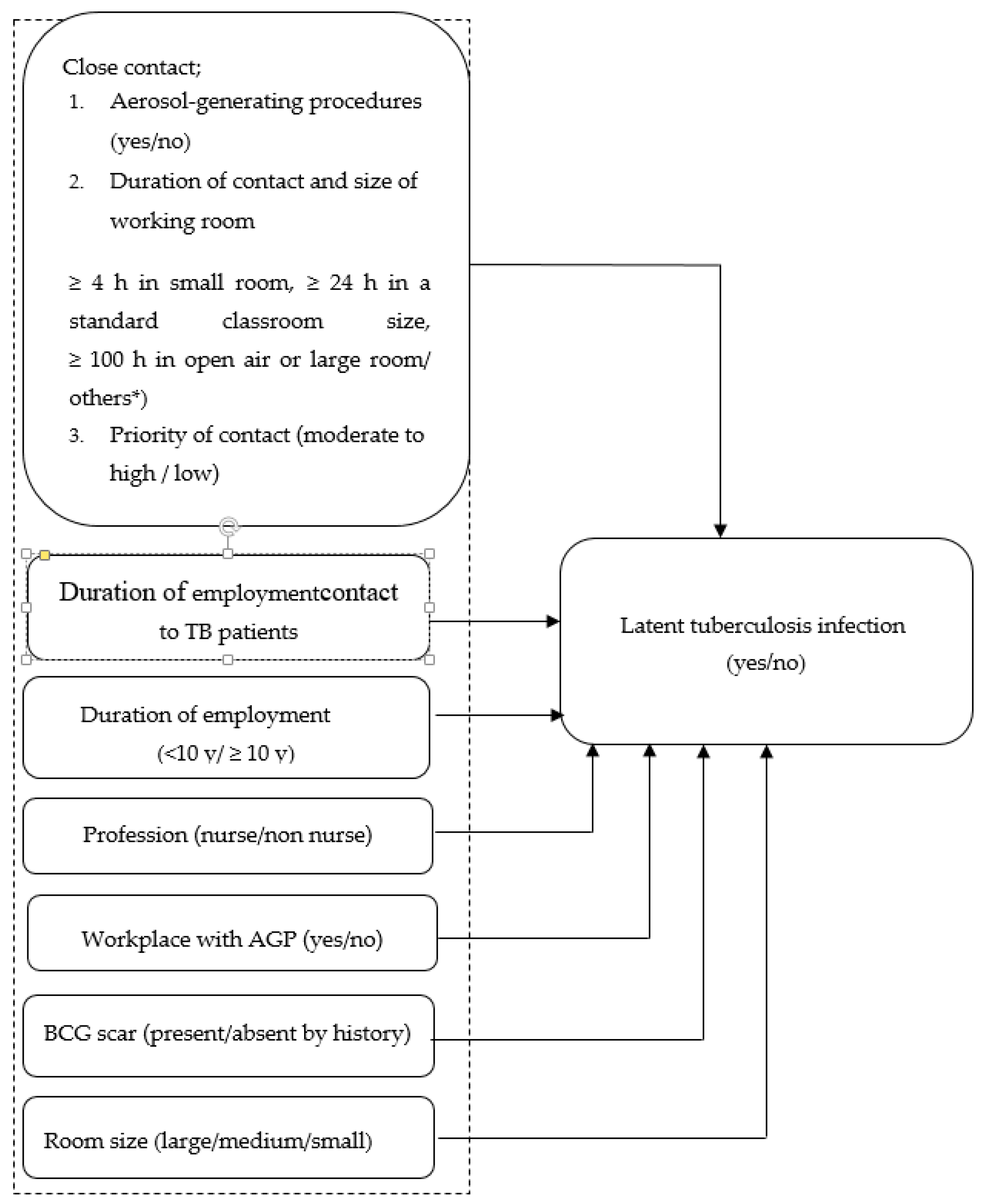
2.5.1. Priority of Contact with TB
2.5.2. Aerosol Generating Procedures (AGPs)
2.6. Ethical Considerations
3. Results
3.1. Demographic Characteristics
3.2. Crude Analysis
3.3. Multivariable Conditional Logistic Regression
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Introduction. In Global Tuberculosis Report 2019; World Health Organization: Geneva, Switzerland, 2019; pp. 7–8. [Google Scholar]
- Agyeman, A.; Ofori-Asenso, R. Tuberculosis—An overview. J. Public Health Emerg. 2017, 1, 1–12. [Google Scholar]
- Rao, M.; Ippolito, G.; Mfinanga, S.; Ntoumi, F.; Yeboah-Manu, D.; Vilaplana, C.; Zumla, A.; Maeurer, M. Latent TB Infection (LTBI)—Mycobacterium tuberculosis pathogenesis and the dynamics of the granuloma battleground. Int. J. Infect. Dis. 2019, 80, S58–S61. [Google Scholar]
- World Health Organization Latent Tuberculosis Infection (LTBI). Available online: https://kku.world/nvb7- (accessed on 25 August 2019).
- Getahun, H.; Matteelli, A.; Chaisson, R.E.; Raviglione, M. Latent Mycobacterium tuberculosis infection. N. Engl. J. Med. 2015, 372, 2127–2135. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization the End TB Strategy. Global Strategy and Targets for Tuberculosis Prevention, Care and Control after 2015; WHO: Geneva, Switzerland, 2014.
- Das, P.; Horton, R. Tuberculosis—Getting to zero. Lancet 2015, 386, 2231–2232. [Google Scholar] [CrossRef] [PubMed]
- Uden, L.; Barber, E.; Ford, N.; Cooke, G.S. Risk of tuberculosis infection and disease for health care workers: An updated meta-analysis. Open Forum Infect. Dis. 2017, 4, ofx137. [Google Scholar] [CrossRef] [PubMed]
- Joshi, R.; Reingold, A.L.; Menzies, D.; Pai, M. Tuberculosis among health-care workers in low- and middle-income countries: A systematic review. PLoS Med. 2006, 3, e494. [Google Scholar] [CrossRef]
- Baussano, I.; Nunn, P.; Williams, B.; Pivetta, E.; Bugiani, M.; Scano, F. Tuberculosis among health care workers. Emerg. Infect. Dis. 2011, 17, 488–494. [Google Scholar]
- Wiwanitkit, V. Prevalence rate of positive tuberculin test among Thai hospital personnel: A summary. J. Hosp. Infect. 2006, 62, 119–120. [Google Scholar] [CrossRef]
- Chaiear, N. Medical surveillance program for health workers. In Occupational Health Service: Important Issues in Occupational Medicine; Division of Occupational Medicine, Department of Community Medicine, Faculty of Medicine, Khon Kaen University: Khon Kaen, Thailand, 2018; pp. 235–249. [Google Scholar]
- Ar-karachaiphong, K.; Chaiear, N.; Reechaipichitkul, W.; Faksri, K.; Lerdruampattana, S. Agreement of tuberculin skin test and quantiFERON®-TB gold-in-tube for screening mycobacterium tuberculosis infection in healthcare workers in a university hospital. J. Med. Assoc. Thai. 2019, 102, S13–S19. [Google Scholar]
- Sawanyawisuth, K.; Chaiear, N.; Sawanyawisuth, K.; Limpawattana, P.; Bourpoern, J.; Reechaipichitkul, W.; Takahashi, K. Can workplaces be predictors for recent onset latent tuberculosis in health care workers? J. Occup. Med. Toxicol. 2009, 4, 20. [Google Scholar] [CrossRef]
- Inchai, J.; Liwsrisakun, C.; Bumroongkit, C.; Euathrongchit, J.; Tajarernmuang, P.; Pothirat, C. Tuberculosis among healthcare workers at Chiang Mai University Hospital, Thailand: Clinical and microbiological characteristics and treatment outcomes. Jpn. J. Infect. Dis. 2018, 71, 214–219. [Google Scholar] [CrossRef] [PubMed]
- Wiwanitkit, V. Prevalence rate of active tuberculosis from chest radiography among Thai hospital personnel: A summary. Am. J. Infect. Control 2005, 5, 313–314. [Google Scholar] [CrossRef] [PubMed]
- Pongwittayapanu, P.; Anothaisintawee, T.; Malathum, K.; Wongrathanandha, C. Incidence of newly diagnosed tuberculosis among healthcare workers in a teaching hospital, Thailand. Ann. Glob. Health 2018, 84, 342–347. [Google Scholar] [CrossRef]
- Nasreen, S.; Shokoohi, M.; Malvankar-Mehta, M.S. Prevalence of latent tuberculosis among health care workers in high burden countries: A systematic review and meta-analysis. PLoS ONE 2016, 11, e0164034. [Google Scholar] [CrossRef] [PubMed]
- Trakultaweesuk, P.; Niyompattama, A.; Boonbamroe, S.; Chaiear, N. Tuberculosis among hospital staffs in a tertiary care hospital, northeastern Thailand. Srinagarind Med. J. 2017, 32, 204–213. [Google Scholar]
- Chen, C.; Zhu, T.; Wang, Z.; Peng, H.; Kong, W.; Zhou, Y.; Shao, Y.; Zhu, L.; Lu, W. High latent TB infection rate and associated risk factors in the Eastern China of low TB incidence. PLoS ONE 2015, 10, e0141511. [Google Scholar] [CrossRef][Green Version]
- Nonghanphithak, D.; Reechaipichitkul, W.; Chaiyasung, T.; Faksri, K. Risk factors for latent tuberculosis infection among health-care workers in Northeastern Thailand. Southeast Asian J. Trop. Med. Public Health 2016, 47, 1198–1208. [Google Scholar]
- Mathew, A.; David, T.; Thomas, K.; Kuruvilla, P.J.; Balaji, V.; Jesudason, M.V.; Samuel, P. Risk factors for tuberculosis among health care workers in South India: A nested case-control study. J. Clin. Epidemiol. 2013, 66, 67–74. [Google Scholar] [CrossRef]
- Sabri, A.; Quistrebert, J.; Naji Amrani, H.; Abid, A.; Zegmout, A.; Abderrhamani Ghorfi, I.; Souhi, H.; Boucaid, A.; Benali, A.; Abilkassem, R.; et al. Prevalence and risk factors for latent tuberculosis infection among healthcare workers in Morocco. PLoS ONE 2019, 14, e0221081. [Google Scholar] [CrossRef]
- He, G.X.; van denHof, S.; van der Werf, M.J.; Wang, G.J.; Ma, S.W.; Zhao, D.Y.; Hu, Y.L.; Yu, S.C.; Borgdorff, M.W. Infection control and the burden of tuberculosis infection and disease in health care workers in china: A cross-sectional study. BMC Infect. Dis. 2010, 10, 313. [Google Scholar] [CrossRef]
- Reichler, M.R.; Khan, A.; Yuan, Y.; Chen, B.; McAuley, J.; Mangura, B.; Sterling, T.R. Duration of exposure among close contacts of patients with infectious tuberculosis and risk of latent tuberculosis infection. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Lamberti, M.; Muoio, M.; Arnese, A.; Borrelli, S.; Di Lorenzo, T.; Garzillo, E.M.; Signoriello, G.; De Pascalis, S.; Coppola, N.; Nienhaus, A. Prevalence of latent tuberculosis infection in healthcare workers at a hospital in Naples, Italy, a low-incidence country. J. Occup. Med. Toxicol. 2016, 11, 53. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhou, F.; Zhang, L.; Gao, L.; Hao, Y.; Zhao, X.; Liu, J.; Lu, J.; Li, X.; Yang, Y.; Chen, J.; et al. Latent tuberculosis infection and occupational protection among health care workers in two types of public hospitals in China. PLoS ONE 2014, 9, e104673. [Google Scholar] [CrossRef][Green Version]
- Jelip, J.; Mathew, G.G.; Yusin, T.; Dony, J.F.; Singh, N.; Ashaari, M.; Lajanin, N.; Shanmuga Ratnam, C.; Yusof Ibrahim, M.; Gopinath, D. Risk factors of tuberculosis among health care workers in Sabah, Malaysia. Tuberculosis 2004, 84, 19–23. [Google Scholar] [CrossRef]
- Claassens, M.M.; Van Schalkwyk, C.; Du Toit, E.; Roest, E.; Lombard, C.J.; Enarson, D.A.; Beyers, N.; Borgdorff, M.W. Tuberculosis in healthcare workers and infection control measures at primary healthcare facilities in South Africa. PLoS ONE 2013, 8, e76272. [Google Scholar] [CrossRef]
- Muangchana, C.; Thamapornpilas, P.; Karnkawinpong, O. Immunization policy development in Thailand: The role of the Advisory Committee on Immunization Practice. Vaccine 2010, 28 (Suppl. S1), A104–A109. [Google Scholar] [CrossRef][Green Version]
- Division of Vaccine Preventable Diseases, Department of Disease Control. Report of National Immunization Coverage Survey 2018. Available online: https://kku.world/h3gcr (accessed on 25 August 2019).
- Division Tuberculosis, Department of Disease Control. Prevention of tuberculosis transmission. In National Tuberculosis Control Programme Guideline, Thailand, 2018; Division Tuberculosis, Department of Disease Control: Bangkok, Thailand, 2018; pp. 141–154. (In Thai) [Google Scholar]
- Centers for Disease Control and Prevention Transmission and Pathogenesis of Tuberculosis. In Core Curriculum on Tuberculosis: What the Clinician Should Know; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2013; pp. 19–44.
- Centers for Disease Control and Prevention Testing for Tuberculosis Infection and Disease. In Core Curriculum on Tuberculosis: What the Clinician Should Know; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2013; pp. 45–74.
- Diel, R.; Loddenkemper, R.; Nienhaus, A. Predictive value of interferon-γ release assays and tuberculin skin testing for progression from latent TB infection to disease state: A meta-analysis. Chest 2012, 142, 63–75. [Google Scholar] [CrossRef]
- Mazurek, G.H.; Jereb, J.; Vernon, A.; LoBue, P.; Goldberg, S.; Castro, K. Updated guidelines for using interferon gamma release assays to detect mycobacterium tuberculosis infection—United States, 2010. MMWR Recomm. Rep. 2010, 59, 1–25. [Google Scholar]
- Yeon, J.H.; Seong, H.; Hur, H.; Park, Y.; Kim, Y.A.; Park, Y.S.; Han, C.H.; Lee, S.M.; Seo, J.H.; Kang, J.G. Prevalence and risk factors of latent tuberculosis among Korean healthcare workers using whole-blood interferon-γ release assay. Sci. Rep. 2018, 8, 10113. [Google Scholar] [CrossRef]
- Jong Lee, K.; Ae Kang, Y.; Mi Kim, Y.; Cho, S.-N.; Wook Moon, J.; Suk Park, M.; Kyu Kim, S.; Chang, J.; Sam Kim, Y. Screening for latent tuberculosis infection in South Korean healthcare workers using a tuberculin skin test and whole blood interferon-gamma assay. Scand. J. Infect. Dis. 2010, 42, 672–678. [Google Scholar] [CrossRef]
- National Tuberculosis Controllers Association; Centers for Disease Control and Prevention. Guidelines for the investigation of contacts of persons with infectious tuberculosis. Recommendations from the National Tuberculosis Controllers Association and CDC. MMWR Recomm. Rep. 2005, 54, 1–47. [Google Scholar]
- O’Malley, M.; Brown, A.G.; Colmers, J.M. Maryland TB Guidelines for Prevention and Treatment of Tuberculosis; Maryland Department of Health and Mental Hygiene: Baltimore, MD, USA, 2007.
- Panthong, J.; Chaiear, N.; Jongkumchok, W.; Janpho, P. In a hospital setting, is there any benefit in prioritizing risk following exposure to tuberculosis?—A preliminary report. J. Med. Assoc. Thai. 2019, 102 (Suppl. S1), S27–S32. [Google Scholar]
- Schlesselman, J.J.; Stolley, P.D. Case Control Studies: Design, Conduct, Analysis; Oxford University Press: New York, NY, USA, 1982; ISBN 978-0-19-502933-8. Available online: https://kku.world/el05u (accessed on 10 August 2020).
- Jensen, P.A.; Lambert, L.A.; Iademarco, M.F.; Ridzon, R. Guidelines for preventing the transmission of Mycobacterium tuberculosis in health-care settings, 2005. MMWR Recomm. Rep. 2005, 54, 1–141. [Google Scholar]
- Tran, K.; Cimon, K.; Severn, M.; Pessoa-Silva, C.L.; Conly, J. Aerosol generating procedures and risk of transmission of acute respiratory infections to healthcare workers: A systematic review. PLoS ONE 2012, 7, e35797. [Google Scholar] [CrossRef]
- Prado, T.N.D.; Riley, L.W.; Sanchez, M.; Fregona, G.; Nóbrega, R.L.P.; Possuelo, L.G.; Zandonade, E.; Locatelli, R.L.; de Souza, F.M.; Rajan, J.V.; et al. Prevalence and risk factors for latent tuberculosis infection among primary health care workers in Brazil. Cadernos Saúde Pública. 2017, 33, e00154916. [Google Scholar] [CrossRef]
- Sterne, J.A.; Rodrigues, L.C.; Guedes, I.N. Does the efficacy of BCG decline with time since vaccination? Int. J. Tuberc. Lung Dis. 1998, 2, 200–207. [Google Scholar]
- Yothasuphap, A.; Udol, K.; Dumavibhat, N.; Chierakul, N.; Sujirarat, D. Factors associated with pulmonary tuberculosis among health care wokers at Siriraj hospital. In Microbial Diversity: Literacy and Applications, Proceedings of the 11st Ubon Ratchathani University Research, Ubon Ratchathani, Thailand, 3–14 July 2017; Office of the President, Ubon Ratchathani University: Ubon Ratchathani, Thailand, 2017; pp. 1–10. (In Thai) [Google Scholar]
- Catanzaro, A. Nosocomial tuberculosis. Am. Rev. Respir. Dis. 1982, 125, 559–562. [Google Scholar]
| Variables | Case | Control | ||
|---|---|---|---|---|
| n = 85 | % | n = 170 | % | |
| Sex | ||||
| Female | 79 | 92.9 | 158 | 92.9 |
| Male | 6 | 7.1 | 12 | 7.1 |
| Age (yr) | ||||
| <30 | 25 | 29.4 | 50 | 29.4 |
| ≥30 | 60 | 70.6 | 120 | 70.6 |
| Mean (sd) | 37.5 | 37.4 | ||
| (9.6) | (9.6) | |||
| Median | 36 | 36 | ||
| (min:max) | (25:57) | (25:57) | ||
| Characteristic | Case | Control | ||
|---|---|---|---|---|
| n = 85 | % | n = 170 | % | |
| Duration of employment (y) | 38 | 44.7 | 82 | 48.2 |
| <10 | ||||
| ≥10 | 47 | 55.3 | 88 | 51.8 |
| Mean (sd) | 14.0 (10.4) | - | 13.0 (10.6) | - |
| Median(min: max) | 10 (1:38) | - | 10 (1:37) | - |
| Position Nurses | 53 | 62.4 | 97 | 57.1 |
| Assistant nurses | 26 | 30.6 | 52 | 30.6 |
| others | 6 | 7.1 | 21 | 12.4 |
| Workplace | ||||
| Intensive care unit | 27 | 31.8 | 49 | 28.8 |
| Surgery ward | 19 | 22.4 | 18 | 10.6 |
| Medicine ward | 13 | 15.3 | 21 | 12.4 |
| Special ward | 13 | 15.3 | 33 | 19.4 |
| Emergency ward | 5 | 5.9 | 28 | 16.5 |
| other | 8 | 9.4 | 21 | 12.4 |
| Workplace of AGPs | ||||
| No | 16 | 18.8 | 52 | 30.6 |
| Yes | 69 | 81.2 | 118 | 69.4 |
| Duration of contact | ||||
| < 8 h | 19 | 22.4 | 53 | 31.2 |
| ≥ 8 h | 66 | 77.7 | 117 | 68.8 |
| Close contact | ||||
| No | 5 | 5.9 | 19 | 11.2 |
| Yes | 80 | 94.1 | 151 | 88.8 |
| Priority of contact | ||||
| Low | 67 | 78.8 | 122 | 71.8 |
| Moderate to High | 18 | 21.2 | 48 | 28.2 |
| Performed AGPs | ||||
| No | 35 | 41.2 | 101 | 59.4 |
| Yes | 50 | 58.8 | 69 | 40.6 |
| Duration of contact and size of working room | ||||
| others ≥4 h in a small room, or ≥24 h in a standard classroom or ≥100 h in a large room | 29 56 | 34.1 65.9 | 70 100 | 41.2 58.8 |
| Room size | ||||
| Large | 7 | 8.2 | 7 | 4.1 |
| Medium | 20 | 23.5 | 48 | 28.2 |
| Small | 58 | 68.2 | 115 | 67.7 |
| BCG scar | ||||
| Present | 40 | 47.1 | 119 | 70.0 |
| Absent | 45 | 52.9 | 51 | 30.0 |
| Characteristic | Cases (n = 85) % | Controls (n = 170) % | Crude OR | 95% CI | p-Value |
|---|---|---|---|---|---|
| Duration | 0.383 | ||||
| of employment (y) | |||||
| <10 | 44.7 | 48.1 | 1 | - | |
| ≥10 | 55.3 | 51.8 | 1.47 | 0.61–3.52 | |
| Workplace of AGPs | 0.041 | ||||
| No | 18.8 | 30.6 | 1 | - | |
| Yes | 81.2 | 69.4 | 1.90 | 1.01–3.58 | |
| Duration of contact (h) | 0.148 | ||||
| <8 | 22.4 | 31.2 | 1 | - | |
| ≥8 | 77.6 | 68.8 | 1.53 | 0.85–2.79 | |
| Close contact | - | - | - | - | 0.118 |
| No | 5.9 | 11.2 | 1 | - | |
| Yes | 94.1 | 88.8 | 2.36 | 0.75–7.44 | |
| Priority of contact | 0.443 | ||||
| Low | 78.8 | 71.8 | 1 | - | |
| Moderate to High | 21.2 | 28.2 | 0.67 | 0.36–1.27 | |
| Performed AGPs | 0.007 | ||||
| No | 41.2 | 31.2 | 1 | - | - |
| Yes | 58.8 | 40.6 | 2.04 | 1.20–3.48 | - |
| Duration of contact and size of working room | 0.239 | ||||
| Others | 34.1 | 41.2 | 1 | - | |
| ≥4 h in a small room, or ≥24 h in a standard classroom or ≥100 h in open air or a large room | 65.9 | 58.8 | 1.38 | 0.81–2.35 | |
| Room size | - | - | - | 0.346 | |
| Large | 8.2 | 4.1 | 1 | - | |
| Medium | 23.5 | 28.24 | 0.42 | 0.13–1.36 | - |
| Small | 68.3 | 67.65 | 0.50 | 0.16–1.55 | - |
| BCG scar | - | - | - | - | < 0.001 |
| Present | 47.1 | 70.0 | 1 | - | - |
| Absent | 52.9 | 30.0 | 2.59 | 1.50–4.47 | - |
| Characteristic | Cases (n = 85) | Controls (n = 170) | Crude OR | Adjusted OR | 95% CI | p-Value |
|---|---|---|---|---|---|---|
| n, % | n, % | |||||
| Close contact | 0.123 | |||||
| No | 5, 5.9 | 19, 11.2 | 1 | 1 | - | |
| Yes | 80, 94.1 | 151, 88.8 | 2.36 | 2.44 | 0.74–8.09 | |
| Duration of contact (h) | 0.166 | |||||
| <8 | 19, 22.4 | 53, 31.2 | 1 | 1 | - | |
| ≥8 | 66, 77.6 | 117, 68.8 | 1.53 | 1.55 | 0.82–2.95 | |
| Performed AGPs | 0.035 | |||||
| No | 35,41.2 | 101, 59.4 | 1 | 1 | - | |
| Yes | 50, 58.8 | 69, 40.6 | 2.04 | 1.82 | 1.04–3.20 | |
| BCG scar | <0.001 | |||||
| Present | 40, 47.1 | 119, 70.0 | 1 | 1 | - | |
| Absent | 45, 52.9 | 51, 30.0 | 2.59 | 2.49 | 1.42–4.38 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chanpho, P.; Chaiear, N.; Kamsa-ard, S. Factors Associated with Latent Tuberculosis Infection among the Hospital Employees in a Tertiary Hospital of Northeastern Thailand. Int. J. Environ. Res. Public Health 2020, 17, 6876. https://doi.org/10.3390/ijerph17186876
Chanpho P, Chaiear N, Kamsa-ard S. Factors Associated with Latent Tuberculosis Infection among the Hospital Employees in a Tertiary Hospital of Northeastern Thailand. International Journal of Environmental Research and Public Health. 2020; 17(18):6876. https://doi.org/10.3390/ijerph17186876
Chicago/Turabian StyleChanpho, Patimaporn, Naesinee Chaiear, and Supot Kamsa-ard. 2020. "Factors Associated with Latent Tuberculosis Infection among the Hospital Employees in a Tertiary Hospital of Northeastern Thailand" International Journal of Environmental Research and Public Health 17, no. 18: 6876. https://doi.org/10.3390/ijerph17186876
APA StyleChanpho, P., Chaiear, N., & Kamsa-ard, S. (2020). Factors Associated with Latent Tuberculosis Infection among the Hospital Employees in a Tertiary Hospital of Northeastern Thailand. International Journal of Environmental Research and Public Health, 17(18), 6876. https://doi.org/10.3390/ijerph17186876




