A Historical Analysis of Randomized Controlled Trials in Rotator Cuff Tears
Abstract
1. Introduction
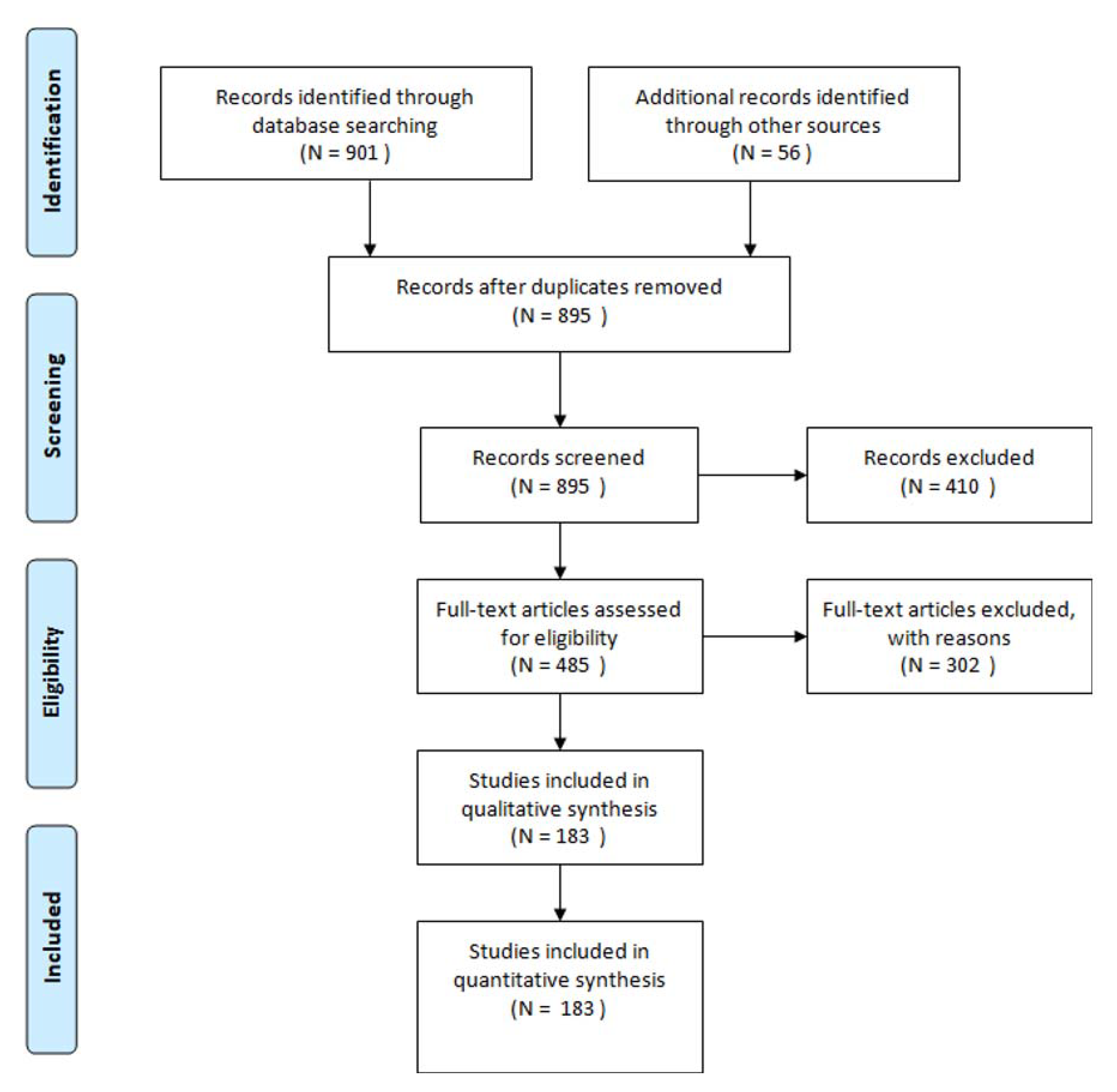
2. Materials and Methods
2.1. Search Strategy and Study Eligibility
2.2. Data Abstraction
2.3. Methodological Quality Assessment
2.4. Assessment of Agreement
2.5. Statistical Analysis
3. Results
3.1. Study Characteristics
3.2. Topic
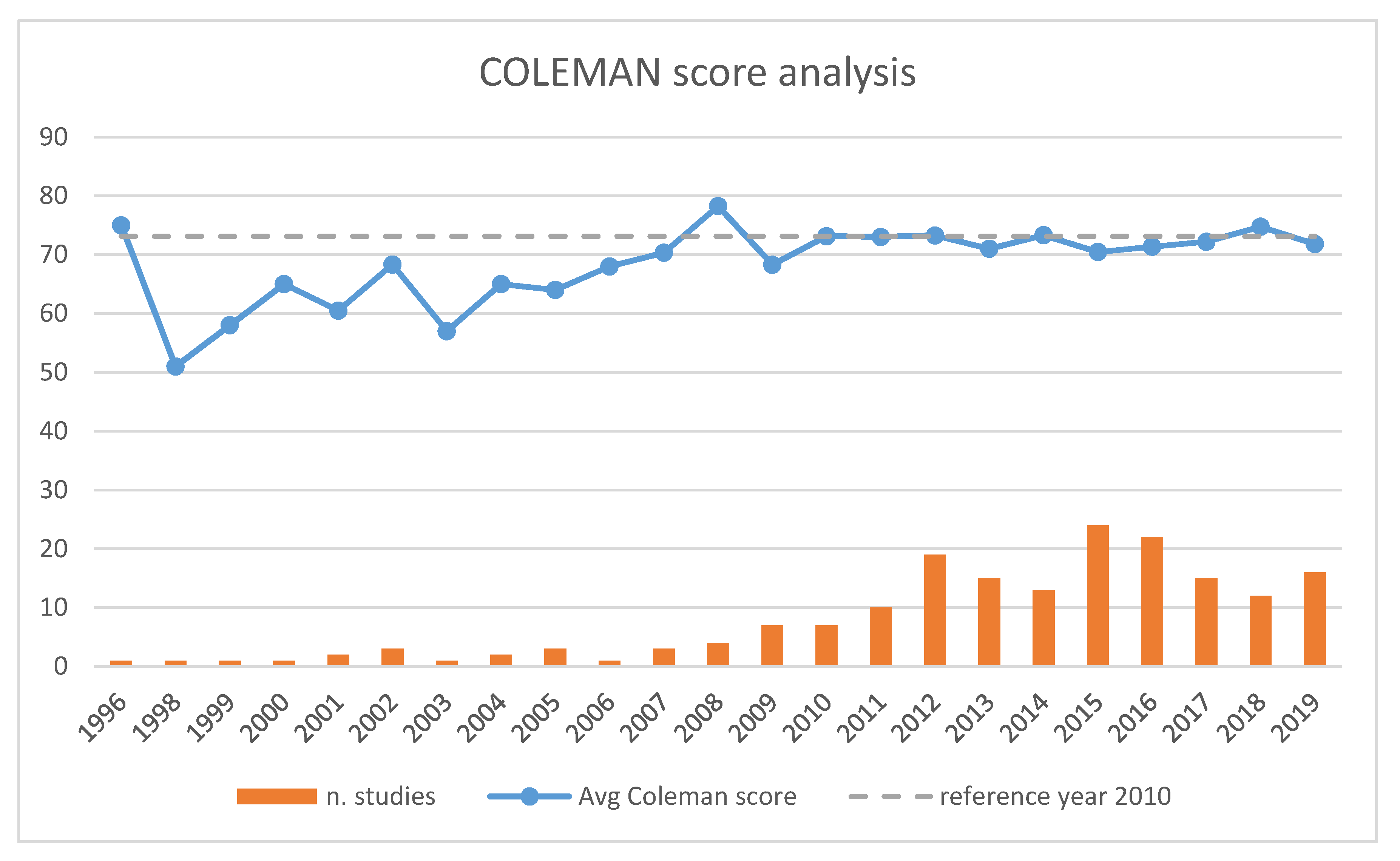
3.3. Modified Coleman Methodology Score (MCMS)
3.4. Trends
3.5. Other Methodological Factors on the Consolidated Standards of Reporting Trials (CONSORT) Checklist
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Abrams, G.D.; Gupta, A.K.; Hussey, K.E.; Tetteh, E.S.; Karas, V.; Bach, B.R.; Cole, B.J.; Romeo, A.A.; Verma, N.N. Arthroscopic Repair of Full-Thickness Rotator Cuff Tears With and Without Acromioplasty: Randomized Prospective Trial With 2-Year Follow-up. Am. J. Sports Med. 2014, 42, 1296–1303. [Google Scholar] [CrossRef]
- Dezaly, C.; Sirveaux, F.; Philippe, R.; Wein-Remy, F.; Sedaghatian, J.; Roche, O.; Molé, D. Arthroscopic treatment of rotator cuff tear in the over-60s: Repair is preferable to isolated acromioplasty-tenotomy in the short term. Orthop. Traumatol. Surg. Res. 2011, 97, S125–S130. [Google Scholar] [CrossRef]
- Jacquot, A.; Dezaly, C.; Goetzmann, T.; Roche, O.; Sirveaux, F.; Molé, D.; French Society for Shoulder, E.b.S. Is rotator cuff repair appropriate in patients older than 60 years of age? prospective, randomised trial in 103 patients with a mean four-year follow-up. Orthop. Traumatol. Surg. Res. 2014, 100, S333–S338. [Google Scholar] [CrossRef]
- MacDonald, P.; McRae, S.; Leiter, J.; Mascarenhas, R.; Lapner, P. Arthroscopic rotator cuff repair with and without acromioplasty in the treatment of full-thickness rotator cuff tears: A multicenter, randomized controlled trial. J. Bone Join. Surg. Am. Vol. 2011, 93, 1953–1960. [Google Scholar] [CrossRef]
- Mohtadi, N.G.; Hollinshead, R.M.; Sasyniuk, T.M.; Fletcher, J.A.; Chan, D.S.; Li, F.X. A randomized clinical trial comparing open to arthroscopic acromioplasty with mini-open rotator cuff repair for full-thickness rotator cuff tears: Disease-specific quality of life outcome at an average 2-year follow-up. Am. J. Sports Med. 2008, 36, 1043–1051. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.J.; Oh, J.H.; Chung, S.W.; Song, M.H. The efficacy of acromioplasty in the arthroscopic repair of small- to medium-sized rotator cuff tears without acromial spur: Prospective comparative study. Arthroscopy 2012, 28, 628–635. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Sun, Z.; Liao, B.; Zhanqiang, S.; Xiao, T.; Zhu, P. Sodium Hyaluronate and Platelet-Rich Plasma for Partial-Thickness Rotator Cuff Tears. Med. Sci. Sports Exerc. 2018. [Google Scholar] [CrossRef] [PubMed]
- Castricini, R.; Longo, U.G.; De Benedetto, M.; Panfoli, N.; Pirani, P.; Zini, R.; Maffulli, N.; Denaro, V. Platelet-rich plasma augmentation for arthroscopic rotator cuff repair: A randomized controlled trial. Am. J. Sports Med. 2011, 39, 258–265. [Google Scholar] [CrossRef]
- Aagaard, K.E.; Lunsjö, K.; Frobell, R. Early repair of trauma-related full-thickness rotator cuff tears does not eliminate the problem of healing failure. Bone Joint J. 2019, 101-B, 603–609. [Google Scholar] [CrossRef]
- Akbaba, Y.A.; Mutlu, E.K.; Altun, S.; Turkmen, E.; Birinci, T.; Celik, D. The effectiveness of trigger point treatment in rotator cuff pathology: A randomized controlled double-blind study. J. Back Musculoskelet Rehabil. 2019, 32, 519–527. [Google Scholar] [CrossRef]
- Alvarez, C.M.; Litchfield, R.; Jackowski, D.; Griffin, S.; Kirkley, A. A prospective, double-blind, randomized clinical trial comparing subacromial injection of betamethasone and xylocaine to xylocaine alone in chronic rotator cuff tendinosis. Am. J. Sports Med. 2005, 33, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Analan, P.D.; Leblebici, B.; Adam, M. Effects of therapeutic ultrasound and exercise on pain, function, and isokinetic shoulder rotator strength of patients with rotator cuff disease. J. Phys. Ther. Sci. 2015, 27, 3113–3117. [Google Scholar] [CrossRef] [PubMed]
- Analay Akbaba, Y.; Kaya Mutlu, E.; Altun, S.; Celik, D. Does the patients' expectations on kinesiotape affect the outcomes of patients with a rotator cuff tear? A randomized controlled clinical trial. Clin. Rehabil. 2018, 32, 1509–1519. [Google Scholar] [CrossRef] [PubMed]
- Arias-Buría, J.L.; Valero-Alcaide, R.; Cleland, J.A.; Salom-Moreno, J.; Ortega-Santiago, R.; Atín-Arratibel, M.A.; Fernández-de-las-Peñas, C. Inclusion of trigger point dry needling in a multimodal physical therapy program for postoperative shoulder pain: A randomized clinical trial. J. Manipulat. Physiol. Ther. 2015, 38, 179–187. [Google Scholar] [CrossRef]
- Arndt, J.; Clavert, P.; Mielcarek, P.; Bouchaib, J.; Meyer, N.; Kempf, J.F.; the French Society for Shoulder & Elbow (SOFEC). Immediate passive motion versus immobilization after endoscopic supraspinatus tendon repair: A prospective randomized study. Orthop. Traumatol. Surg. Res. 2012, 98, S131–S138. [Google Scholar] [CrossRef]
- Avanzi, P.; Giudici, L.D.; Capone, A.; Cardoni, G.; Lunardi, G.; Foti, G.; Zorzi, C. Prospective randomized controlled trial for patch augmentation in rotator cuff repair: 24-month outcomes. J. Shoulder Elb. Surg. 2019, 28, 1918–1927. [Google Scholar] [CrossRef]
- Banerjee, S.S.; Pulido, P.; Adelson, W.S.; Fronek, J.; Hoenecke, H.R. The efficacy of continuous bupivacaine infiltration following arthroscopic rotator cuff repair. Arthroscopy 2008, 24, 397–402. [Google Scholar] [CrossRef]
- Bang, S.R.; Yu, S.K.; Kim, T.H. Can gabapentin help reduce postoperative pain in arthroscopic rotator cuff repair? A prospective, randomized, double-blind study. Arthroscopy 2010, 26, S106–S111. [Google Scholar] [CrossRef]
- Barber, F.A.; Burns, J.P.; Deutsch, A.; Labbé, M.R.; Litchfield, R.B. A prospective, randomized evaluation of acellular human dermal matrix augmentation for arthroscopic rotator cuff repair. Arthroscopy 2012, 28, 8–15. [Google Scholar] [CrossRef]
- Barber, F.A.; Herbert, M.A. The effectiveness of an anesthetic continuous-infusion device on postoperative pain control. Arthroscopy 2002, 18, 76–81. [Google Scholar] [CrossRef]
- Baumgarten, K.M.; Osborn, R.; Schweinle, W.E.; Zens, M.J.; Helsper, E.A. Are Pulley Exercises Initiated 6 Weeks After Rotator Cuff Repair a Safe and Effective Rehabilitative Treatment? A Randomized Controlled Trial. Am. J. Sports Med. 2016, 44, 1844–1851. [Google Scholar] [CrossRef] [PubMed]
- Behr, A.; Freo, U.; Ori, C.; Westermann, B.; Alemanno, F. Buprenorphine added to levobupivacaine enhances postoperative analgesia of middle interscalene brachial plexus block. J. Anesth. 2012, 26, 746–751. [Google Scholar] [CrossRef] [PubMed]
- Belay, E.S.; Wittstein, J.R.; Garrigues, G.E.; Lassiter, T.E.; Scribani, M.; Goldner, R.D.; Bean, C.A. Biceps tenotomy has earlier pain relief compared to biceps tenodesis: A randomized prospective study. Knee Surg. Sports Traumatol. Arth. Off. J. ESSKA 2019, 27, 4032–4037. [Google Scholar] [CrossRef] [PubMed]
- Berth, A.; Neumann, W.; Awiszus, F.; Pap, G. Massive rotator cuff tears: Functional outcome after debridement or arthroscopic partial repair. J. Orthop. Traumatol. 2010, 11, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Bidwai, A.S.; Birch, A.; Temperley, D.; Odak, S.; Walton, M.J.; Haines, J.F.; Trail, I. Medium- to long-term results of a randomized controlled trial to assess the efficacy of arthoscopic-subacromial decompression versus mini-open repair for the treatment of medium-sized rotator cuff tears. Shoulder Elb. 2016, 8, 101–105. [Google Scholar] [CrossRef]
- Bigoni, M.; Gorla, M.; Guerrasio, S.; Brignoli, A.; Cossio, A.; Grillo, P.; Marinoni, E.C. Shoulder evaluation with isokinetic strength testing after arthroscopic rotator cuff repairs. J. Shoulder Elb. Surg. 2009, 18, 178–183. [Google Scholar] [CrossRef]
- Blanchard, T.K.; Bearcroft, P.W.; Constant, C.R.; Griffin, D.R.; Dixon, A.K. Diagnostic and therapeutic impact of MRI and arthrography in the investigation of full-thickness rotator cuff tears. Eur. Radiol. 1999, 9, 638–642. [Google Scholar] [CrossRef]
- Blum, K.; Chen, A.L.; Chen, T.J.; Waite, R.L.; Downs, B.W.; Braverman, E.R.; Kerner, M.M.; Savarimuthu, S.M.; DiNubile, N. Repetitive H-wave device stimulation and program induces significant increases in the range of motion of post operative rotator cuff reconstruction in a double-blinded randomized placebo controlled human study. BMC Musculoskelet Disord. 2009, 10, 132. [Google Scholar] [CrossRef]
- Boehm, T.D.; Werner, A.; Radtke, S.; Mueller, T.; Kirschner, S.; Gohlke, F. The effect of suture materials and techniques on the outcome of repair of the rotator cuff: A prospective, randomised study. J. Bone Joint Surg. Br. 2005, 87, 819–823. [Google Scholar] [CrossRef]
- Borgeat, A.; Aguirre, J.; Marquardt, M.; Mrdjen, J.; Blumenthal, S. Continuous interscalene analgesia with ropivacaine 0.2% versus ropivacaine 0.3% after open rotator cuff repair: The effects on postoperative analgesia and motor function. Anesth. Analg. 2010, 111, 1543–1547. [Google Scholar] [CrossRef]
- Burks, R.T.; Crim, J.; Brown, N.; Fink, B.; Greis, P.E. A prospective randomized clinical trial comparing arthroscopic single- and double-row rotator cuff repair: Magnetic resonance imaging and early clinical evaluation. Am. J. Sports Med. 2009, 37, 674–682. [Google Scholar] [CrossRef]
- Cai, Y.Z.; Zhang, C.; Jin, R.L.; Shen, T.; Gu, P.C.; Lin, X.J.; Chen, J.D. Arthroscopic Rotator Cuff Repair With Graft Augmentation of 3-Dimensional Biological Collagen for Moderate to Large Tears: A Randomized Controlled Study. Am. J. Sports Med. 2018, 46, 1424–1431. [Google Scholar] [CrossRef] [PubMed]
- Capito, N.M.; Cook, J.L.; Yahuaca, B.; Capito, M.D.; Sherman, S.L.; Smith, M.J. Safety and efficacy of hyperosmolar irrigation solution in shoulder arthroscopy. J. Shoulder Elb. Surg. 2017, 26, 745–751. [Google Scholar] [CrossRef] [PubMed]
- Carbonel, I.; Martinez, A.A.; Calvo, A.; Ripalda, J.; Herrera, A. Single-row versus double-row arthroscopic repair in the treatment of rotator cuff tears: A prospective randomized clinical study. Int. Orthop. 2012, 36, 1877–1883. [Google Scholar] [CrossRef]
- Carr, A.; Cooper, C.; Campbell, M.K.; Rees, J.; Moser, J.; Beard, D.J.; Fitzpatrick, R.; Gray, A.; Dawson, J.; Murphy, J.; et al. Effectiveness of open and arthroscopic rotator cuff repair (UKUFF): A randomised controlled trial. Bone Joint J. 2017, 99-B, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Castagna, A.; Borroni, M.; Garofalo, R.; Rose, G.D.; Cesari, E.; Padua, R.; Conti, M.; Gumina, S. Deep partial rotator cuff tear: Transtendon repair or tear completion and repair? A randomized clinical trial. Knee Surg. Sports Traumatol. Arth. Off. J. ESSKA 2015, 23, 460–463. [Google Scholar] [CrossRef] [PubMed]
- Chierichini, A.; Frassanito, L.; Vergari, A.; Santoprete, S.; Chiarotti, F.; Saccomanno, M.F.; Milano, G. The effect of norepinephrine versus epinephrine in irrigation fluid on the incidence of hypotensive/bradycardic events during arthroscopic rotator cuff repair with interscalene block in the sitting position. Arthroscopy 2015, 31, 800–806. [Google Scholar] [CrossRef]
- Cho, C.H.; Lee, S.W.; Lee, Y.K.; Shin, H.K.; Hwang, I. Effect of a sleep aid in analgesia after arthroscopic rotator cuff repair. Yonsei Med. J. 2015, 56, 772–777. [Google Scholar] [CrossRef]
- Cho, C.H.; Song, K.S.; Jung, G.H.; Lee, Y.K.; Shin, H.K. Early postoperative outcomes between arthroscopic and mini-open repair for rotator cuff tears. Orthopedics 2012, 35, e1347–e1352. [Google Scholar] [CrossRef]
- Cho, C.H.; Song, K.S.; Min, B.W.; Lee, K.J.; Ha, E.; Lee, Y.C.; Lee, Y.K. Multimodal approach to postoperative pain control in patients undergoing rotator cuff repair. Knee Surg. Sports Traumatol. Arth. Off. J. ESSKA 2011, 19, 1744–1748. [Google Scholar] [CrossRef]
- Cho, N.S.; Ha, J.H.; Rhee, Y.G. Patient-controlled analgesia after arthroscopic rotator cuff repair: Subacromial catheter versus intravenous injection. Am. J. Sports Med. 2007, 35, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Kim, T.; Kwon, Y.S.; Kang, H. Intra-operative effect of interscalene brachial plexus block to arthroscopic rotator cuff repair surgery. Int. Orthop. 2018. [Google Scholar] [CrossRef] [PubMed]
- Chou, W.Y.; Ko, J.Y.; Wang, F.S.; Huang, C.C.; Wong, T.; Wang, C.J.; Chang, H.E. Effect of sodium hyaluronate treatment on rotator cuff lesions without complete tears: A randomized, double-blind, placebo-controlled study. J. Shoulder Elb. Surg. 2010, 19, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Coghlan, J.A.; Forbes, A.; McKenzie, D.; Bell, S.N.; Buchbinder, R. Efficacy of subacromial ropivacaine infusion for rotator cuff surgery. A randomized trial. J. Bone Joint Surg. Am. Vol. 2009, 91, 1558–1567. [Google Scholar] [CrossRef] [PubMed]
- Conti, M.; Garofalo, R.; Castagna, A. Does a brace influence clinical outcomes after arthroscopic rotator cuff repair? Musculoskelet Surg. 2015, 99 (Suppl. 1), S31–S35. [Google Scholar] [CrossRef]
- Coory, J.A.; Parr, A.F.; Wilkinson, M.P.; Gupta, A. Efficacy of suprascapular nerve block compared with subacromial injection: A randomized controlled trial in patients with rotator cuff tears. J. Shoulder Elb. Surg. 2019, 28, 430–436. [Google Scholar] [CrossRef]
- Cuff, D.J.; Pupello, D.R. Prospective randomized study of arthroscopic rotator cuff repair using an early versus delayed postoperative physical therapy protocol. J. Shoulder Elb. Surg. 2012, 21, 1450–1455. [Google Scholar] [CrossRef]
- Culebras, X.; Van Gessel, E.; Hoffmeyer, P.; Gamulin, Z. Clonidine combined with a long acting local anesthetic does not prolong postoperative analgesia after brachial plexus block but does induce hemodynamic changes. Anesth. Analg. 2001, 92, 199–204. [Google Scholar] [CrossRef]
- D'Ambrosi, R.; Palumbo, F.; Paronzini, A.; Ragone, V.; Facchini, R.M. Platelet-rich plasma supplementation in arthroscopic repair of full-thickness rotator cuff tears: A randomized clinical trial. Musculoskelet Surg. 2016, 100, 25–32. [Google Scholar] [CrossRef]
- De Carli, A.; Vadalà, A.; Zanzotto, E.; Zampar, G.; Vetrano, M.; Iorio, R.; Ferretti, A. Reparable rotator cuff tears with concomitant long-head biceps lesions: Tenotomy or tenotomy/tenodesis? Knee Surg. Sports Traumatol. Arth. Off. J. ESSKA 2012, 20, 2553–2558. [Google Scholar] [CrossRef]
- De Roo, P.J.; Muermans, S.; Maroy, M.; Linden, P.; Van den Daelen, L. Passive mobilization after arthroscopic rotator cuff repair is not detrimental in the early postoperative period. Acta Orthop. Belg. 2015, 81, 485–492. [Google Scholar] [PubMed]
- Delaunay, L.; Souron, V.; Lafosse, L.; Marret, E.; Toussaint, B. Analgesia after arthroscopic rotator cuff repair: Subacromial versus interscalene continuous infusion of ropivacaine. Reg. Anesth. Pain Med. 2005, 30, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Desmet, M.; Vanneste, B.; Reynvoet, M.; Van Cauwelaert, J.; Verhelst, L.; Pottel, H.; Missant, C.; Van de Velde, M. A randomised controlled trial of intravenous dexamethasone combined with interscalene brachial plexus blockade for shoulder surgery. Anaesthesia 2015, 70, 1180–1185. [Google Scholar] [CrossRef] [PubMed]
- Desroches, A.; Klouche, S.; Schlur, C.; Bauer, T.; Waitzenegger, T.; Hardy, P. Suprascapular Nerve Block Versus Interscalene Block as Analgesia After Arthroscopic Rotator Cuff Repair: A Randomized Controlled Noninferiority Trial. Arthroscopy 2016, 32, 2203–2209. [Google Scholar] [CrossRef] [PubMed]
- Düzgün, I.; Baltacı, G.; Atay, O.A. Comparison of slow and accelerated rehabilitation protocol after arthroscopic rotator cuff repair: Pain and functional activity. Acta Orthop. Traumatol. Turc. 2011, 45, 23–33. [Google Scholar] [CrossRef]
- Düzgün, İ.; Baltacı, G.; Turgut, E.; Atay, O.A. Effects of slow and accelerated rehabilitation protocols on range of motion after arthroscopic rotator cuff repair. Acta Orthop. Traumatol. Turc. 2014, 48, 642–648. [Google Scholar] [CrossRef]
- Ebert, J.R.; Wang, A.; Smith, A.; Nairn, R.; Breidahl, W.; Zheng, M.H.; Ackland, T. A Midterm Evaluation of Postoperative Platelet-Rich Plasma Injections on Arthroscopic Supraspinatus Repair: A Randomized Controlled Trial. Am. J. Sports Med. 2017, 45, 2965–2974. [Google Scholar] [CrossRef]
- Flurin, P.H.; Hardy, P.; Abadie, P.; Desmoineaux, P.; Essig, J.; Joudet, T.; Sommaire, C.; Thelu, C.E.; French Arthroscopy Society (SFA). Rotator cuff tears after 70 years of age: A prospective, randomized, comparative study between decompression and arthroscopic repair in 154 patients. Orthop. Traumatol. Surg. Res. 2013, 99, S371–S378. [Google Scholar] [CrossRef]
- Flury, M.; Rickenbacher, D.; Schwyzer, H.K.; Jung, C.; Schneider, M.M.; Stahnke, K.; Goldhahn, J.; Audigé, L. Does Pure Platelet-Rich Plasma Affect Postoperative Clinical Outcomes After Arthroscopic Rotator Cuff Repair? A Randomized Controlled Trial. Am. J. Sports Med. 2016, 44, 2136–2146. [Google Scholar] [CrossRef]
- Franceschi, F.; Longo, U.G.; Ruzzini, L.; Rizzello, G.; Maffulli, N.; Denaro, V. No advantages in repairing a type II superior labrum anterior and posterior (SLAP) lesion when associated with rotator cuff repair in patients over age 50: A randomized controlled trial. Am. J. Sports Med. 2008, 36, 247–253. [Google Scholar] [CrossRef]
- Franceschi, F.; Papalia, R.; Del Buono, A.; Vasta, S.; Costa, V.; Maffulli, N.; Denaro, V. Articular-sided rotator cuff tears: Which is the best repair? A three-year prospective randomised controlled trial. Int. Orthop. 2013, 37, 1487–1493. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, F.; Papalia, R.; Franceschetti, E.; Palumbo, A.; Del Buono, A.; Paciotti, M.; Maffulli, N.; Denaro, V. Double-Row Repair Lowers the Retear Risk After Accelerated Rehabilitation. Am. J. Sports Med. 2016, 44, 948–956. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, F.; Ruzzini, L.; Longo, U.G.; Martina, F.M.; Zobel, B.B.; Maffulli, N.; Denaro, V. Equivalent clinical results of arthroscopic single-row and double-row suture anchor repair for rotator cuff tears: A randomized controlled trial. Am. J. Sports Med. 2007, 35, 1254–1260. [Google Scholar] [CrossRef] [PubMed]
- Ganokroj, P.; Matrakool, L.; Limsuwarn, P.; Sissaynarane, T.; Yimvassana, C.; Laoratanavoraphong, S.; Ratanawarinchai, J. A Prospective Randomized Study Comparing the Effectiveness of Midlateral and Posterior Subacromial Steroid Injections. Orthopedics 2019, 42, e44–e50. [Google Scholar] [CrossRef]
- Garofalo, R.; Conti, M.; Notarnicola, A.; Maradei, L.; Giardella, A.; Castagna, A. Effects of one-month continuous passive motion after arthroscopic rotator cuff repair: Results at 1-year follow-up of a prospective randomized study. Musculoskelet Surg. 2010, 94 (Suppl. 1), S79–S83. [Google Scholar] [CrossRef]
- Gartsman, G.M.; Drake, G.; Edwards, T.B.; Elkousy, H.A.; Hammerman, S.M.; O'Connor, D.P.; Press, C.M. Ultrasound evaluation of arthroscopic full-thickness supraspinatus rotator cuff repair: Single-row versus double-row suture bridge (transosseous equivalent) fixation. Results of a prospective, randomized study. J. Shoulder Elb. Surg. 2013, 22, 1480–1487. [Google Scholar] [CrossRef]
- Gartsman, G.M.; O'connor, D.P. Arthroscopic rotator cuff repair with and without arthroscopic subacromial decompression: A prospective, randomized study of one-year outcomes. J. Shoulder Elb. Surg. 2004, 13, 424–426. [Google Scholar] [CrossRef]
- Gervasi, E.; Maman, E.; Dekel, A.; Cautero, E. Fluoroscopy-guided biodegradable spacer implantation using local anesthesia: Safety and efficacy study in patients with massive rotator cuff tears. Musculoskelet Surg. 2016, 100, 19–24. [Google Scholar] [CrossRef]
- Ghandour, T.M.; Ibrahim, A.; Abdelrahman, A.A.; Elgammal, A.; Hammad, M.H. Does The Type of Shoulder Brace Affect Postoperative Pain and Clinical Outcome After Arthroscopic Rotator Cuff Repair? Arthroscopy 2019, 35, 1016–1023. [Google Scholar] [CrossRef]
- Gialanella, B.; Prometti, P. Effects of corticosteroids injection in rotator cuff tears. Pain Med. 2011, 12, 1559–1565. [Google Scholar] [CrossRef]
- Grasso, A.; Milano, G.; Salvatore, M.; Falcone, G.; Deriu, L.; Fabbriciani, C. Single-row versus double-row arthroscopic rotator cuff repair: A prospective randomized clinical study. Arthroscopy 2009, 25, 4–12. [Google Scholar] [CrossRef] [PubMed]
- Greiner, S.; Ide, J.; Van Noort, A.; Mochizuki, Y.; Ochi, H.; Marraffino, S.; Sridharan, S.; Rudicel, S.; Itoi, E. Local rhBMP-12 on an Absorbable Collagen Sponge as an Adjuvant Therapy for Rotator Cuff Repair–A Phase 1, Randomized, Standard of Care Control, Multicenter Study: Safety and Feasibility. Am. J. Sports Med. 2015, 43, 1994–2004. [Google Scholar] [CrossRef] [PubMed]
- Gumina, S.; Campagna, V.; Ferrazza, G.; Giannicola, G.; Fratalocchi, F.; Milani, A.; Postacchini, F. Use of platelet-leukocyte membrane in arthroscopic repair of large rotator cuff tears: A prospective randomized study. J. Bone Joint Surg. Am. Vol. 2012, 94, 1345–1352. [Google Scholar] [CrossRef] [PubMed]
- Gumina, S.; Passaretti, D.; Gurzì, M.D.; Candela, V. Arginine L-alpha-ketoglutarate, methylsulfonylmethane, hydrolyzed type I collagen and bromelain in rotator cuff tear repair: A prospective randomized study. Curr. Med. Res. Opin. 2012, 28, 1767–1774. [Google Scholar] [CrossRef] [PubMed]
- Han, S.S.; Lee, Y.H.; Oh, J.H.; Aminzai, S.; Kim, S.H. Randomized, controlled trial of multimodal shoulder injection or intravenous patient-controlled analgesia after arthroscopic rotator cuff repair. Knee Surg. Sports Traumatol. Arth. Off. J. ESSKA 2013, 21, 2877–2883. [Google Scholar] [CrossRef] [PubMed]
- Hartrick, C.T.; Tang, Y.S.; Siwek, D.; Murray, R.; Hunstad, D.; Smith, G. The effect of initial local anesthetic dose with continuous interscalene analgesia on postoperative pain and diaphragmatic function in patients undergoing arthroscopic shoulder surgery: A double-blind, randomized controlled trial. BMC Anesthesiol. 2012, 12, 6. [Google Scholar] [CrossRef] [PubMed]
- Hayes, K.; Ginn, K.A.; Walton, J.R.; Szomor, Z.L.; Murrell, G.A. A randomised clinical trial evaluating the efficacy of physiotherapy after rotator cuff repair. Aust. J. Physiother. 2004, 50, 77–83. [Google Scholar] [CrossRef]
- Hollman, F.; Wolterbeek, N.; Zijl, J.A.C.; van Egeraat, S.P.M.; Wessel, R.N. Abduction Brace Versus Antirotation Sling After Arthroscopic Cuff Repair: The Effects on Pain and Function. Arthroscopy 2017, 33, 1618–1626. [Google Scholar] [CrossRef]
- Holtby, R.; Christakis, M.; Maman, E.; MacDermid, J.C.; Dwyer, T.; Athwal, G.S.; Faber, K.; Theodoropoulos, J.; Woodhouse, L.J.; Razmjou, H. Impact of Platelet-Rich Plasma on Arthroscopic Repair of Small- to Medium-Sized Rotator Cuff Tears: A Randomized Controlled Trial. Orthop. J. Sports Med. 2016, 4, 2325967116665595. [Google Scholar] [CrossRef]
- Hufeland, M.; Wicke, S.; Verde, P.E.; Krauspe, R.; Patzer, T. Biceps tenodesis versus tenotomy in isolated LHB lesions: A prospective randomized clinical trial. Arch. Orthop. Trauma Surg. 2019, 139, 961–970. [Google Scholar] [CrossRef]
- Iannotti, J.P.; Codsi, M.J.; Kwon, Y.W.; Derwin, K.; Ciccone, J.; Brems, J.J. Porcine small intestine submucosa augmentation of surgical repair of chronic two-tendon rotator cuff tears. A randomized, controlled trial. J. Bone Joint Surg. Am. Vol. 2006, 88, 1238–1244. [Google Scholar] [CrossRef]
- Ide, J.; Mochizuki, Y.; van Noort, A.; Ochi, H.; Sridharan, S.; Itoi, E.; Greiner, S. Local rhBMP-12 on an Absorbable Collagen Sponge as an Adjuvant Therapy for Rotator Cuff Repair–A Phase 1, Randomized, Standard of Care Control, Multicenter Study: Part 2-A Pilot Study of Functional Recovery and Structural Outcomes. Orthop. J. Sports Med. 2017, 5, 2325967117726740. [Google Scholar] [CrossRef] [PubMed]
- Ikemoto, R.Y.; Murachovsky, J.; Prata Nascimento, L.G.; Bueno, R.S.; Oliveira Almeida, L.H.; Strose, E.; de Mello, S.C.; Saletti, D. Prospective Randomized Study Comparing Two Anesthetic Methods for Shoulder Surgery. Rev. Bras. Ortop. 2010, 45, 395–399. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ilhanli, I.; Guder, N.; Gul, M. Platelet-Rich Plasma Treatment With Physical Therapy in Chronic Partial Supraspinatus Tears. Iran. Red. Crescent Med. J. 2015, 17, e23732. [Google Scholar] [CrossRef] [PubMed]
- Jenssen, K.K.; Lundgreen, K.; Madsen, J.E.; Kvakestad, R.; Pripp, A.H.; Dimmen, S. No Functional Difference Between Three and Six Weeks of Immobilization After Arthroscopic Rotator Cuff Repair: A Prospective Randomized Controlled Non-Inferiority Trial. Arthroscopy 2018, 34, 2765–2774. [Google Scholar] [CrossRef]
- Jo, C.H.; Shin, J.S.; Huh, J. Multimodal analgesia for arthroscopic rotator cuff repair: A randomized, placebo-controlled, double-blind trial. Eur. J. Orthop. Surg. Traumatol. 2014, 24, 315–322. [Google Scholar] [CrossRef]
- Jo, C.H.; Shin, J.S.; Lee, Y.G.; Shin, W.H.; Kim, H.; Lee, S.Y.; Yoon, K.S.; Shin, S. Platelet-rich plasma for arthroscopic repair of large to massive rotator cuff tears: A randomized, single-blind, parallel-group trial. Am. J. Sports Med. 2013, 41, 2240–2248. [Google Scholar] [CrossRef]
- Jo, C.H.; Shin, J.S.; Shin, W.H.; Lee, S.Y.; Yoon, K.S.; Shin, S. Platelet-rich plasma for arthroscopic repair of medium to large rotator cuff tears: A randomized controlled trial. Am. J. Sports Med. 2015, 43, 2102–2110. [Google Scholar] [CrossRef]
- Keener, J.D.; Galatz, L.M.; Stobbs-Cucchi, G.; Patton, R.; Yamaguchi, K. Rehabilitation following arthroscopic rotator cuff repair: A prospective randomized trial of immobilization compared with early motion. J. Bone Joint Surg. Am. Vol. 2014, 96, 11–19. [Google Scholar] [CrossRef]
- Khashan, M.; Dolkart, O.; Amar, E.; Chechik, O.; Sharfman, Z.; Mozes, G.; Maman, E.; Weinbroum, A. Effect of preemptive intra-articular morphine and ketamine on pain after arthroscopic rotator cuff repair: A prospective, double-blind, randomized controlled study. Arch. Orthop. Trauma Surg. 2016, 136, 233–239. [Google Scholar] [CrossRef]
- Kim, J.; Chung, J.; Ok, H. Asymptomatic acromioclavicular joint arthritis in arthroscopic rotator cuff tendon repair: A prospective randomized comparison study. Arch. Orthop. Trauma Surg. 2011, 131, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Ha, D.H.; Kim, S.M.; Kim, K.W.; Han, S.Y.; Kim, Y.S. Does arthroscopic preemptive extensive rotator interval release reduce postoperative stiffness after arthroscopic rotator cuff repair?: A prospective randomized clinical trial. J. Shoulder Elb. Surg. 2019, 28, 1639–1646. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Koh, H.J.; Kim, D.K.; Lee, H.J.; Kwon, K.H.; Lee, K.Y.; Kim, Y.S. Interscalene brachial plexus bolus block versus patient-controlled interscalene indwelling catheter analgesia for the first 48 hours after arthroscopic rotator cuff repair. J. Shoulder Elb. Surg. 2018, 27, 1243–1250. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Lee, J.S.; Park, C.W. Extracorporeal shock wave therapy is not useful after arthroscopic rotator cuff repair. Knee Surg. Sports Traumatol. Arth. Off. J. ESSKA 2012, 20, 2567–2572. [Google Scholar] [CrossRef]
- Kim, J.Y.; Song, K.S.; Kim, W.J.; Park, Y.H.; Kang, H.; Woo, Y.C.; Shin, H.Y. Analgesic efficacy of two interscalene blocks and one cervical epidural block in arthroscopic rotator cuff repair. Knee Surg. Sports Traumatol. Arth. Off. J. ESSKA 2016, 24, 931–939. [Google Scholar] [CrossRef]
- Kim, J.Y.; Song, S.H.; Cho, J.H.; Cho, H.R. Comparison of clinical efficacy among remifentanil, nicardipine, and remifentanil plus nicardipine continuous infusion for hypotensive anesthesia during arthroscopic shoulder surgery. J. Orthop. Surg. 2017, 25, 2309499017716251. [Google Scholar] [CrossRef]
- Kim, Y.S.; Chung, S.W.; Kim, J.Y.; Ok, J.H.; Park, I.; Oh, J.H. Is early passive motion exercise necessary after arthroscopic rotator cuff repair? Am. J. Sports Med. 2012, 40, 815–821. [Google Scholar] [CrossRef]
- Kim, Y.S.; Lee, H.J.; Jin, H.K.; Kim, S.E.; Lee, J.W. Conventional En Masse Repair Versus Separate Double-Layer Double-Row Repair for the Treatment of Delaminated Rotator Cuff Tears. Am. J. Sports Med. 2016, 44, 1146–1152. [Google Scholar] [CrossRef]
- Kim, Y.S.; Lee, H.J.; Kim, J.H.; Noh, D.Y. When Should We Repair Partial-Thickness Rotator Cuff Tears? Outcome Comparison Between Immediate Surgical Repair Versus Delayed Repair After 6-Month Period of Nonsurgical Treatment. Am. J. Sports Med. 2018, 46, 1091–1096. [Google Scholar] [CrossRef]
- Klintberg, I.H.; Gunnarsson, A.C.; Svantesson, U.; Styf, J.; Karlsson, J. Early loading in physiotherapy treatment after full-thickness rotator cuff repair: A prospective randomized pilot-study with a two-year follow-up. Clin. Rehabil. 2009, 23, 622–638. [Google Scholar] [CrossRef]
- Ko, S.H.; Cho, S.D.; Lee, C.C.; Choi, J.K.; Kim, H.W.; Park, S.J.; Bae, M.H.; Cha, J.R. Comparison of Arthroscopically Guided Suprascapular Nerve Block and Blinded Axillary Nerve Block vs. Blinded Suprascapular Nerve Block in Arthroscopic Rotator Cuff Repair: A Randomized Controlled Trial. Clin. Orthop. Surg. 2017, 9, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Ko, S.H.; Lee, C.C.; Friedman, D.; Park, K.B.; Warner, J.J. Arthroscopic single-row supraspinatus tendon repair with a modified mattress locking stitch: A prospective, randomized controlled comparison with a simple stitch. Arthroscopy 2008, 24, 1005–1012. [Google Scholar] [CrossRef] [PubMed]
- Koh, K.H.; Kang, K.C.; Lim, T.K.; Shon, M.S.; Yoo, J.C. Prospective randomized clinical trial of single- versus double-row suture anchor repair in 2- to 4-cm rotator cuff tears: Clinical and magnetic resonance imaging results. Arthroscopy 2011, 27, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Koh, K.H.; Lim, T.K.; Shon, M.S.; Park, Y.E.; Lee, S.W.; Yoo, J.C. Effect of immobilization without passive exercise after rotator cuff repair: Randomized clinical trial comparing four and eight weeks of immobilization. J. Bone Joint Surg. Am. Vol. 2014, 96, e44. [Google Scholar] [CrossRef]
- Koh, W.U.; Kim, H.J.; Park, H.S.; Choi, W.J.; Yang, H.S.; Ro, Y.J. A randomised controlled trial comparing continuous supraclavicular and interscalene brachial plexus blockade for open rotator cuff surgery. Anaesthesia 2016, 71, 692–699. [Google Scholar] [CrossRef]
- Kraeutler, M.J.; Reynolds, K.A.; Long, C.; McCarty, E.C. Compressive cryotherapy versus ice-a prospective, randomized study on postoperative pain in patients undergoing arthroscopic rotator cuff repair or subacromial decompression. J. Shoulder Elb. Surg. 2015, 24, 854–859. [Google Scholar] [CrossRef] [PubMed]
- Kukkonen, J.; Joukainen, A.; Lehtinen, J.; Mattila, K.T.; Tuominen, E.K.; Kauko, T.; Aärimaa, V. Treatment of non-traumatic rotator cuff tears: A randomised controlled trial with one-year clinical results. Bone Joint J. 2014, 96-B, 75–81. [Google Scholar] [CrossRef]
- Lam, P.H.; Hansen, K.; Keighley, G.; Hackett, L.; Murrell, G.A. A Randomized, Double-Blinded, Placebo-Controlled Clinical Trial Evaluating the Effectiveness of Daily Vibration After Arthroscopic Rotator Cuff Repair. Am. J. Sports Med. 2015, 43, 2774–2782. [Google Scholar] [CrossRef]
- Lamas, J.R.; García-Fernández, C.; Tornero-Esteban, P.; Lópiz, Y.; Rodriguez-Rodriguez, L.; Ortega, L.; Fernández-Gutiérrez, B.; Marco, F. Adverse effects of xenogenic scaffolding in the context of a randomized double-blind placebo-controlled study for repairing full-thickness rotator cuff tears. Trials 2019, 20, 387. [Google Scholar] [CrossRef]
- Lambers Heerspink, F.O.; van Raay, J.J.; Koorevaar, R.C.; van Eerden, P.J.; Westerbeek, R.E.; van’t Riet, E.; van den Akker-Scheek, I.; Diercks, R.L. Comparing surgical repair with conservative treatment for degenerative rotator cuff tears: A randomized controlled trial. J. Shoulder Elb. Surg. 2015, 24, 1274–1281. [Google Scholar] [CrossRef]
- Lapner, P.L.; Sabri, E.; Rakhra, K.; McRae, S.; Leiter, J.; Bell, K.; Macdonald, P. A multicenter randomized controlled trial comparing single-row with double-row fixation in arthroscopic rotator cuff repair. J. Bone Joint Surg. Am. Vol. 2012, 94, 1249–1257. [Google Scholar] [CrossRef]
- Lastayo, P.C.; Wright, T.; Jaffe, R.; Hartzel, J. Continuous passive motion after repair of the rotator cuff. A prospective outcome study. J. Bone Joint Surg. Am. Vol. 1998, 80, 1002–1011. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.G.; Cho, N.S.; Rhee, Y.G. Effect of two rehabilitation protocols on range of motion and healing rates after arthroscopic rotator cuff repair: Aggressive versus limited early passive exercises. Arthroscopy 2012, 28, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Jeong, J.Y.; Kim, C.K.; Kim, Y.S. Surgical treatment of lesions of the long head of the biceps brachii tendon with rotator cuff tear: A prospective randomized clinical trial comparing the clinical results of tenotomy and tenodesis. J. Shoulder Elb. Surg. 2016, 25, 1107–1114. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Kim, Y.S.; Park, I.; Ha, D.H.; Lee, J.H. Administration of analgesics after rotator cuff repair: A prospective clinical trial comparing glenohumeral, subacromial, and a combination of glenohumeral and subacromial injections. J. Shoulder Elb. Surg. 2015, 24, 663–668. [Google Scholar] [CrossRef]
- Lee, J.J.; Hwang, J.T.; Kim, D.Y.; Lee, S.S.; Hwang, S.M.; Lee, N.R.; Kwak, B.C. Effects of arthroscopy-guided suprascapular nerve block combined with ultrasound-guided interscalene brachial plexus block for arthroscopic rotator cuff repair: A randomized controlled trial. Knee Surg. Sports Traumatol. Arth. Off. J. ESSKA 2017, 25, 2121–2128. [Google Scholar] [CrossRef]
- Lee, J.J.; Kim, D.Y.; Hwang, J.T.; Lee, S.S.; Hwang, S.M.; Kim, G.H.; Jo, Y.G. Effect of ultrasonographically guided axillary nerve block combined with suprascapular nerve block in arthroscopic rotator cuff repair: A randomized controlled trial. Arthroscopy 2014, 30, 906–914. [Google Scholar] [CrossRef]
- Lee, J.J.; Yoo, Y.S.; Hwang, J.T.; Kim, D.Y.; Jeon, S.J.; Hwang, S.M.; Jang, J.S. Efficacy of direct arthroscopy-guided suprascapular nerve block after arthroscopic rotator cuff repair: A prospective randomized study. Knee Surg. Sports Traumatol. Arth. Off. J. ESSKA 2015, 23, 562–566. [Google Scholar] [CrossRef]
- Liu, J.; Fan, L.; Zhu, Y.; Yu, H.; Xu, T.; Li, G. Comparison of clinical outcomes in all-arthroscopic versus mini-open repair of rotator cuff tears: A randomized clinical trial. Medicine 2017, 96, e6322. [Google Scholar] [CrossRef]
- Liu, X.N.; Noh, Y.M.; Yang, C.J.; Kim, J.U.; Chung, M.H.; Noh, K.C. Effects of a Single-Dose Interscalene Block on Pain and Stress Biomarkers in Patients Undergoing Arthroscopic Rotator Cuff Repair: A Randomized Controlled Trial. Arthroscopy 2017, 33, 918–926. [Google Scholar] [CrossRef]
- Ma, H.L.; Chiang, E.R.; Wu, H.T.; Hung, S.C.; Wang, S.T.; Liu, C.L.; Chen, T.H. Clinical outcome and imaging of arthroscopic single-row and double-row rotator cuff repair: A prospective randomized trial. Arthroscopy 2012, 28, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Mahure, S.A.; Rokito, A.S.; Kwon, Y.W. Transcutaneous electrical nerve stimulation for postoperative pain relief after arthroscopic rotator cuff repair: A prospective double-blinded randomized trial. J. Shoulder Elb. Surg. 2017, 26, 1508–1513. [Google Scholar] [CrossRef] [PubMed]
- Malavolta, E.A.; Gracitelli, M.E.; Ferreira Neto, A.A.; Assunção, J.H.; Bordalo-Rodrigues, M.; de Camargo, O.P. Platelet-rich plasma in rotator cuff repair: A prospective randomized study. Am. J. Sports Med. 2014, 42, 2446–2454. [Google Scholar] [CrossRef] [PubMed]
- Malavolta, E.A.; Gracitelli, M.E.C.; Assunção, J.H.; Ferreira Neto, A.A.; Bordalo-Rodrigues, M.; de Camargo, O.P. Clinical and Structural Evaluations of Rotator Cuff Repair With and Without Added Platelet-Rich Plasma at 5-Year Follow-up: A Prospective Randomized Study. Am. J. Sports Med. 2018, 46, 3134–3141. [Google Scholar] [CrossRef]
- Malik, T.; Mass, D.; Cohn, S. Postoperative Analgesia in a Prolonged Continuous Interscalene Block Versus Single-Shot Block in Outpatient Arthroscopic Rotator Cuff Repair: A Prospective Randomized Study. Arthroscopy 2016, 32, 1544–1550.e1541. [Google Scholar] [CrossRef]
- Mardani-Kivi, M.; Keyhani, S.; Ebrahim-Zadeh, M.H.; Hashemi-Motlagh, K.; Saheb-Ekhtiari, K. Rotator cuff tear with concomitant long head of biceps tendon (LHBT) degeneration: What is the preferred choice? Open subpectoral versus arthroscopic intraarticular tenodesis. J. Orthop. Traumatol. 2019, 20, 26. [Google Scholar] [CrossRef]
- Mazzocca, A.D.; Arciero, R.A.; Shea, K.P.; Apostolakos, J.M.; Solovyova, O.; Gomlinski, G.; Wojcik, K.E.; Tafuto, V.; Stock, H.; Cote, M.P. The Effect of Early Range of Motion on Quality of Life, Clinical Outcome, and Repair Integrity After Arthroscopic Rotator Cuff Repair. Arthroscopy 2017, 33, 1138–1148. [Google Scholar] [CrossRef]
- Menek, B.; Tarakci, D.; Algun, Z.C. The effect of Mulligan mobilization on pain and life quality of patients with Rotator cuff syndrome: A randomized controlled trial. J. Back Musculoskelet Rehabil. 2019, 32, 171–178. [Google Scholar] [CrossRef]
- Merivirta, R.; Äärimaa, V.; Aantaa, R.; Koivisto, M.; Leino, K.; Liukas, A.; Kuusniemi, K. Postoperative fentanyl patch versus subacromial bupivacaine infusion in arthroscopic shoulder surgery. Arthroscopy 2013, 29, 1129–1134. [Google Scholar] [CrossRef]
- Milano, G.; Grasso, A.; Salvatore, M.; Saccomanno, M.F.; Deriu, L.; Fabbriciani, C. Arthroscopic rotator cuff repair with metal and biodegradable suture anchors: A prospective randomized study. Arthroscopy 2010, 26, S112–S119. [Google Scholar] [CrossRef]
- Milano, G.; Grasso, A.; Salvatore, M.; Zarelli, D.; Deriu, L.; Fabbriciani, C. Arthroscopic rotator cuff repair with and without subacromial decompression: A prospective randomized study. Arthroscopy 2007, 23, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Milano, G.; Saccomanno, M.F.; Careri, S.; Taccardo, G.; De Vitis, R.; Fabbriciani, C. Efficacy of marrow-stimulating technique in arthroscopic rotator cuff repair: A prospective randomized study. Arthroscopy 2013, 29, 802–810. [Google Scholar] [CrossRef] [PubMed]
- Moosmayer, S.; Lund, G.; Seljom, U.; Svege, I.; Hennig, T.; Tariq, R.; Smith, H.J. Comparison between surgery and physiotherapy in the treatment of small and medium-sized tears of the rotator cuff: A randomised controlled study of 103 patients with one-year follow-up. J. Bone Joint Surg. Br. 2010, 92, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Moosmayer, S.; Lund, G.; Seljom, U.S.; Haldorsen, B.; Svege, I.C.; Hennig, T.; Pripp, A.H.; Smith, H.J. At a 10-Year Follow-up, Tendon Repair Is Superior to Physiotherapy in the Treatment of Small and Medium-Sized Rotator Cuff Tears. J. Bone Joint Surg. Am. Vol. 2019, 101, 1050–1060. [Google Scholar] [CrossRef] [PubMed]
- Nam, J.H.; Park, S.; Lee, H.R.; Kim, S.H. Outcomes After Limited or Extensive Bursectomy During Rotator Cuff Repair: Randomized Controlled Trial. Arthroscopy 2018. [Google Scholar] [CrossRef]
- Nicholas, S.J.; Lee, S.J.; Mullaney, M.J.; Tyler, T.F.; Fukunaga, T.; Johnson, C.D.; McHugh, M.P. Functional Outcomes After Double-Row Versus Single-Row Rotator Cuff Repair: A Prospective Randomized Trial. Orthop. J. Sports Med. 2016, 4, 2325967116667398. [Google Scholar] [CrossRef]
- Nicholson, J.A.; Searle, H.K.C.; MacDonald, D.; McBirnie, J. Cost-effectiveness and satisfaction following arthroscopic rotator cuff repair: Does age matter? Bone Joint J. 2019, 101-B, 860–866. [Google Scholar] [CrossRef]
- Oh, C.H.; Oh, J.H.; Kim, S.H.; Cho, J.H.; Yoon, J.P.; Kim, J.Y. Effectiveness of subacromial anti-adhesive agent injection after arthroscopic rotator cuff repair: Prospective randomized comparison study. Clin. Orthop. Surg. 2011, 3, 55–61. [Google Scholar] [CrossRef]
- Oh, J.H.; Kim, J.Y.; Choi, J.H.; Park, S.M. Is arthroscopic distal clavicle resection necessary for patients with radiological acromioclavicular joint arthritis and rotator cuff tears? A prospective randomized comparative study. Am. J. Sports Med. 2014, 42, 2567–2573. [Google Scholar] [CrossRef]
- Oh, J.H.; Kim, J.Y.; Chung, S.W.; Park, J.S.; Kim, D.H.; Kim, S.H.; Yun, M.J. Warmed irrigation fluid does not decrease perioperative hypothermia during arthroscopic shoulder surgery. Arthroscopy 2014, 30, 159–164. [Google Scholar] [CrossRef]
- Oh, J.H.; Lee, Y.H.; Kim, S.H.; Park, J.S.; Seo, H.J.; Kim, W.; Park, H.B. Comparison of Treatments for Superior Labrum-Biceps Complex Lesions With Concomitant Rotator Cuff Repair: A Prospective, Randomized, Comparative Analysis of Debridement, Biceps Tenotomy, and Biceps Tenodesis. Arthroscopy 2016, 32, 958–967. [Google Scholar] [CrossRef] [PubMed]
- Osbahr, D.C.; Cawley, P.W.; Speer, K.P. The effect of continuous cryotherapy on glenohumeral joint and subacromial space temperatures in the postoperative shoulder. Arthroscopy 2002, 18, 748–754. [Google Scholar] [CrossRef] [PubMed]
- Osti, L.; Buono, A.D.; Maffulli, N. Pulsed electromagnetic fields after rotator cuff repair: A randomized, controlled study. Orthopedics 2015, 38, e223–e228. [Google Scholar] [CrossRef] [PubMed]
- Osti, L.; Del Buono, A.; Maffulli, N. Microfractures at the rotator cuff footprint: A randomised controlled study. Int. Orthop. 2013, 37, 2165–2171. [Google Scholar] [CrossRef] [PubMed]
- Pandey, V.; Bandi, A.; Madi, S.; Agarwal, L.; Acharya, K.K.; Maddukuri, S.; Sambhaji, C.; Willems, W.J. Does application of moderately concentrated platelet-rich plasma improve clinical and structural outcome after arthroscopic repair of medium-sized to large rotator cuff tear? A randomized controlled trial. J. Shoulder Elb. Surg. 2016, 25, 1312–1322. [Google Scholar] [CrossRef] [PubMed]
- Park, J.Y.; Bang, J.Y.; Oh, K.S. Blind suprascapular and axillary nerve block for post-operative pain in arthroscopic rotator cuff surgery. Knee Surg. Sports Traumatol. Arth. Off. J. ESSKA 2016, 24, 3877–3883. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.B.; Koh, K.H.; Shon, M.S.; Park, Y.E.; Yoo, J.C. Arthroscopic distal clavicle resection in symptomatic acromioclavicular joint arthritis combined with rotator cuff tear: A prospective randomized trial. Am. J. Sports Med. 2015, 43, 985–990. [Google Scholar] [CrossRef]
- Perdreau, A.; Joudet, T. Efficacy of multimodal analgesia injection combined with corticosteroids after arthroscopic rotator cuff repair. Orthop. Traumatol. Surg. Res. 2015, 101, S337–S345. [Google Scholar] [CrossRef]
- Piitulainen, K.; Häkkinen, A.; Salo, P.; Kautiainen, H.; Ylinen, J. Does adding a 12-month exercise programme to usual care after a rotator cuff repair effect disability and quality of life at 12 months? A randomized controlled trial. Clin. Rehabil. 2015, 29, 447–456. [Google Scholar] [CrossRef]
- Raab, M.G.; Rzeszutko, D.; O'Connor, W.; Greatting, M.D. Early results of continuous passive motion after rotator cuff repair: A prospective, randomized, blinded, controlled study. Am. J. Orthop. 1996, 25, 214–220. [Google Scholar]
- Randelli, P.; Arrigoni, P.; Aliprandi, A.; Sdao, S.; Ragone, V.; D'Ambrosi, R.; Randelli, F.; Cabitza, P.; Banfi, G. Repair versus shaving of partial-thickness articular-sided tears of the upper subscapularis tendon. A prospective randomized controlled trial. Joints 2015, 3, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Randelli, P.; Arrigoni, P.; Ragone, V.; Aliprandi, A.; Cabitza, P. Platelet rich plasma in arthroscopic rotator cuff repair: A prospective RCT study, 2-year follow-up. J. Shoulder Elb. Surg. 2011, 20, 518–528. [Google Scholar] [CrossRef] [PubMed]
- Randelli, P.; Stoppani, C.A.; Zaolino, C.; Menon, A.; Randelli, F.; Cabitza, P. Advantages of Arthroscopic Rotator Cuff Repair With a Transosseous Suture Technique: A Prospective Randomized Controlled Trial. Am. J. Sports Med. 2017, 45, 2000–2009. [Google Scholar] [CrossRef] [PubMed]
- Raschhofer, R.; Poulios, N.; Schimetta, W.; Kisling, R.; Mittermaier, C. Early active rehabilitation after arthroscopic rotator cuff repair: A prospective randomized pilot study. Clin. Rehabil. 2017, 31, 1332–1339. [Google Scholar] [CrossRef]
- Rha, D.W.; Park, G.Y.; Kim, Y.K.; Kim, M.T.; Lee, S.C. Comparison of the therapeutic effects of ultrasound-guided platelet-rich plasma injection and dry needling in rotator cuff disease: A randomized controlled trial. Clin. Rehabil. 2013, 27, 113–122. [Google Scholar] [CrossRef]
- Roddey, T.S.; Olson, S.L.; Gartsman, G.M.; Hanten, W.P.; Cook, K.F. A randomized controlled trial comparing 2 instructional approaches to home exercise instruction following arthroscopic full-thickness rotator cuff repair surgery. J. Orthop. Sports Phys. Ther. 2002, 32, 548–559. [Google Scholar] [CrossRef]
- Rodeo, S.A.; Delos, D.; Williams, R.J.; Adler, R.S.; Pearle, A.; Warren, R.F. The effect of platelet-rich fibrin matrix on rotator cuff tendon healing: A prospective, randomized clinical study. Am. J. Sports Med. 2012, 40, 1234–1241. [Google Scholar] [CrossRef]
- Ruiz-Moneo, P.; Molano-Muñoz, J.; Prieto, E.; Algorta, J. Plasma rich in growth factors in arthroscopic rotator cuff repair: A randomized, double-blind, controlled clinical trial. Arthroscopy 2013, 29, 2–9. [Google Scholar] [CrossRef]
- Salviz, E.A.; Xu, D.; Frulla, A.; Kwofie, K.; Shastri, U.; Chen, J.; Shariat, A.N.; Littwin, S.; Lin, E.; Choi, J.; et al. Continuous interscalene block in patients having outpatient rotator cuff repair surgery: A prospective randomized trial. Anesth. Analg. 2013, 117, 1485–1492. [Google Scholar] [CrossRef]
- Schwartzberg, R.S.; Reuss, B.L.; Rust, R. Efficacy of continuous subacromial bupivacaine infusion for pain control after arthroscopic rotator cuff repair. J. Shoulder Elb. Surg. 2013, 22, 1320–1324. [Google Scholar] [CrossRef]
- Senekovic, V.; Poberaj, B.; Kovacic, L.; Mikek, M.; Adar, E.; Markovitz, E.; Maman, E.; Dekel, A. The biodegradable spacer as a novel treatment modality for massive rotator cuff tears: A prospective study with 5-year follow-up. Arch. Orthop. Trauma Surg. 2017, 137, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Seven, M.M.; Ersen, O.; Akpancar, S.; Ozkan, H.; Turkkan, S.; Yıldız, Y.; Koca, K. Effectiveness of prolotherapy in the treatment of chronic rotator cuff lesions. Orthop. Traumatol. Surg. Res. 2017, 103, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Shams, A.; El-Sayed, M.; Gamal, O.; Ewes, W. Subacromial injection of autologous platelet-rich plasma versus corticosteroid for the treatment of symptomatic partial rotator cuff tears. Eur. J. Orthop. Surg. Traumatol. 2016, 26, 837–842. [Google Scholar] [CrossRef] [PubMed]
- Sheps, D.M.; Bouliane, M.; Styles-Tripp, F.; Beaupre, L.A.; Saraswat, M.K.; Luciak-Corea, C.; Silveira, A.; Glasgow, R.; Balyk, R. Early mobilisation following mini-open rotator cuff repair: A randomised control trial. Bone Joint J. 2015, 97-B, 1257–1263. [Google Scholar] [CrossRef] [PubMed]
- Sheps, D.M.; Silveira, A.; Beaupre, L.; Styles-Tripp, F.; Balyk, R.; Lalani, A.; Glasgow, R.; Bergman, J.; Bouliane, M.; Shoulder and Upper Extremity Research Group of Edmonton (SURGE). Early Active Motion Versus Sling Immobilization After Arthroscopic Rotator Cuff Repair: A Randomized Controlled Trial. Arthroscopy 2019, 35, 749–760.e742. [Google Scholar] [CrossRef] [PubMed]
- Shibata, Y.; Midorikawa, K.; Emoto, G.; Naito, M. Clinical evaluation of sodium hyaluronate for the treatment of patients with rotator cuff tear. J. Shoulder Elb. Surg. 2001, 10, 209–216. [Google Scholar] [CrossRef]
- Shin, S.J. A comparison of 2 repair techniques for partial-thickness articular-sided rotator cuff tears. Arthroscopy 2012, 28, 25–33. [Google Scholar] [CrossRef]
- Syed, U.A.M.; Aleem, A.W.; Wowkanech, C.; Weekes, D.; Freedman, M.; Tjoumakaris, F.; Abboud, J.A.; Austin, L.S. Neer Award 2018: The effect of preoperative education on opioid consumption in patients undergoing arthroscopic rotator cuff repair: A prospective, randomized clinical trial. J. Shoulder Elb. Surg. 2018, 27, 962–967. [Google Scholar] [CrossRef]
- Takada, M.; Fukusaki, M.; Terao, Y.; Yamashita, K.; Ando, Y.; Sumikawa, K. Postoperative analgesic effect of preoperative intravenous flurbiprofen in arthroscopic rotator cuff repair. J. Anesth. 2009, 23, 500–503. [Google Scholar] [CrossRef]
- Tetzlaff, J.E.; Brems, J.; Dilger, J. Intraarticular morphine and bupivacaine reduces postoperative pain after rotator cuff repair. Reg. Anesth. Pain Med. 2000, 25, 611–614. [Google Scholar] [CrossRef]
- Thackeray, E.M.; Swenson, J.D.; Gertsch, M.C.; Phillips, K.M.; Steele, J.W.; Burks, R.T.; Tashjian, R.Z.; Greis, P.E. Diaphragm function after interscalene brachial plexus block: A double-blind, randomized comparison of 0.25% and 0.125% bupivacaine. J. Shoulder Elb. Surg. 2013, 22, 381–386. [Google Scholar] [CrossRef] [PubMed]
- Tirefort, J.; Schwitzguebel, A.J.; Collin, P.; Nowak, A.; Plomb-Holmes, C.; Ladermann, A. Postoperative Mobilization After Superior Rotator Cuff Repair: Sling Versus No Sling: A Randomized Prospective Study. J. Bone Joint Surg. Am. Vol. 2019, 101, 494–503. [Google Scholar] [CrossRef] [PubMed]
- Torrens, C.; Miquel, J.; Santana, F. Do we really allow patient decision-making in rotator cuff surgery? A prospective randomized study. J. Orthop. Surg. Res. 2019, 14, 116. [Google Scholar] [CrossRef] [PubMed]
- van der Zwaal, P.; Thomassen, B.J.; Nieuwenhuijse, M.J.; Lindenburg, R.; Swen, J.W.; van Arkel, E.R. Clinical outcome in all-arthroscopic versus mini-open rotator cuff repair in small to medium-sized tears: A randomized controlled trial in 100 patients with 1-year follow-up. Arthroscopy 2013, 29, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Walsh, M.R.; Nelson, B.J.; Braman, J.P.; Yonke, B.; Obermeier, M.; Raja, A.; Reams, M. Platelet-rich plasma in fibrin matrix to augment rotator cuff repair: A prospective, single-blinded, randomized study with 2-year follow-up. J. Shoulder Elb. Surg. 2018, 27, 1553–1563. [Google Scholar] [CrossRef]
- Wang, A.; McCann, P.; Colliver, J.; Koh, E.; Ackland, T.; Joss, B.; Zheng, M.; Breidahl, B. Do postoperative platelet-rich plasma injections accelerate early tendon healing and functional recovery after arthroscopic supraspinatus repair? A randomized controlled trial. Am. J. Sports Med. 2015, 43, 1430–1437. [Google Scholar] [CrossRef]
- Watanabe, K.; Tokumine, J.; Yorozu, T.; Moriyama, K.; Sakamoto, H.; Inoue, T. Particulate-steroid betamethasone added to ropivacaine in interscalene brachial plexus block for arthroscopic rotator cuff repair improves postoperative analgesia. BMC Anesthesiol. 2016, 16, 84. [Google Scholar] [CrossRef]
- Weber, S.C.; Kauffman, J.I.; Parise, C.; Weber, S.J.; Katz, S.D. Platelet-rich fibrin matrix in the management of arthroscopic repair of the rotator cuff: A prospective, randomized, double-blinded study. Am. J. Sports Med. 2013, 41, 263–270. [Google Scholar] [CrossRef]
- Yamakado, K. Efficacy of arthroscopically placed pain catheter adjacent to the suprascapular nerve (continuous arthroscopically assisted suprascapular nerve block) following arthroscopic rotator-cuff repair. Open. Access J. Sports Med. 2014, 5, 129–136. [Google Scholar] [CrossRef]
- Yamamoto, S.; Yamaguchi, H.; Sakaguchi, M.; Yamashita, S.; Satsumae, T. Preoperative droperidol improved postoperative pain relief in patients undergoing rotator-cuff repair during general anesthesia using intravenous morphine. J. Clin. Anesth. 2003, 15, 525–529. [Google Scholar] [CrossRef]
- Yun, M.J.; Oh, J.H.; Yoon, J.P.; Park, S.H.; Hwang, J.W.; Kil, H.Y. Subacromial patient-controlled analgesia with ropivacaine provides effective pain control after arthroscopic rotator cuff repair. Knee Surg. Sports Traumatol. Arth. Off. J. ESSKA 2012, 20, 1971–1977. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhou, J.; Ge, H.; Cheng, B. Tenotomy or tenodesis for long head biceps lesions in shoulders with reparable rotator cuff tears: A prospective randomised trial. Knee Surg. Sports Traumatol. Arth. Off. J. ESSKA 2015, 23, 464–469. [Google Scholar] [CrossRef] [PubMed]
- Zumstein, M.A.; Rumian, A.; Thélu, C.; Lesbats, V.; O'Shea, K.; Schaer, M.; Boileau, P. SECEC Research Grant 2008 II: Use of platelet- and leucocyte-rich fibrin (L-PRF) does not affect late rotator cuff tendon healing: A prospective randomized controlled study. J. Shoulder Elb. Surg. 2016, 25, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Urwin, M.; Symmons, D.; Allison, T.; Brammah, T.; Busby, H.; Roxby, M.; Simmons, A.; Williams, G. Estimating the burden of musculoskeletal disorders in the community: The comparative prevalence of symptoms at different anatomical sites, and the relation to social deprivation. Ann. Rheum. Dis. 1998, 57, 649–655. [Google Scholar] [CrossRef] [PubMed]
- Roquelaure, Y.; Ha, C.; Leclerc, A.; Touranchet, A.; Sauteron, M.; Melchior, M.; Imbernon, E.; Goldberg, M. Epidemiologic surveillance of upper-extremity musculoskeletal disorders in the working population. Arth. Rheum. 2006, 55, 765–778. [Google Scholar] [CrossRef] [PubMed]
- Meislin, R.J.; Sperling, J.W.; Stitik, T.P. Persistent shoulder pain: Epidemiology, pathophysiology, and diagnosis. Am. J. Orthop. 2005, 34, 5–9. [Google Scholar]
- Tashjian, R.Z. AAOS clinical practice guideline: Optimizing the management of rotator cuff problems. J. Am. Acad. Orthop. Surg. 2011, 19, 380–383. [Google Scholar] [CrossRef]
- Beard, D.J.; Rees, J.L.; Cook, J.A.; Rombach, I.; Cooper, C.; Merritt, N.; Shirkey, B.A.; Donovan, J.L.; Gwilym, S.; Savulescu, J.; et al. Arthroscopic subacromial decompression for subacromial shoulder pain (CSAW): A multicentre, pragmatic, parallel group, placebo-controlled, three-group, randomised surgical trial. Lancet 2018, 391, 329–338. [Google Scholar] [CrossRef]
- Karjalainen, T.V.; Jain, N.B.; Page, C.M.; Lahdeoja, T.A.; Johnston, R.V.; Salamh, P.; Kavaja, L.; Ardern, C.L.; Agarwal, A.; Vandvik, P.O.; et al. Subacromial decompression surgery for rotator cuff disease. Cochrane Database Syst. Rev. 2019, 1, CD005619. [Google Scholar] [CrossRef]
| Years | Mean Coleman Score | Number of Studies | Standard Deviation |
|---|---|---|---|
| 1996–2010 | 68.54 | 37 | 8927 |
| 2011–2019 | 72.12 | 146 | 7869 |
| Total | 71.40 | 183 | 8196 |
| t | df | Significance (2-tailed) | Mean Difference | Std. Error Difference | 95% Confidence Interval of the Difference Lower Upper | |
|---|---|---|---|---|---|---|
| −2231 | 51,079 | 0.030 | −3583 | 1606 | −6806 | −0.359 |
| CONSORT Flow Diagram | Mean Coleman Score | Number of Studies | Standard Deviation |
|---|---|---|---|
| NO | 70.46 | 136 | 8409 |
| YES | 74.11 | 47 | 6941 |
| Total | 71.40 | 183 | 8196 |
| t | df | Significance (2-tailed) | Mean Difference | Std. Error Difference | 95% Confidence Interval of the Difference Lower Upper | |
|---|---|---|---|---|---|---|
| 2931 | 96,069 | 0.004 | 3643 | 1243 | 1176 | 6111 |
| Years | Mean Number of Checklist Items | Number of Studies | Standard Deviation |
|---|---|---|---|
| 1996–2010 | 19.97 | 37 | 3594 |
| 2011–2019 | 22.10 | 146 | 2822 |
| Total | 21.67 | 183 | 3103 |
| t | df | Significance (2-tailed) | Mean Difference | Std. Error Difference | 95% Confidence Interval of the Difference Lower Upper | |
|---|---|---|---|---|---|---|
| −3342 | 47,839 | 0.002 | −2123 | 0.635 | −3400 | −0.845 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Candela, V.; Longo, U.G.; Di Naro, C.; Facchinetti, G.; Marchetti, A.; Sciotti, G.; Santamaria, G.; Piergentili, I.; De Marinis, M.G.; Nazarian, A.; et al. A Historical Analysis of Randomized Controlled Trials in Rotator Cuff Tears. Int. J. Environ. Res. Public Health 2020, 17, 6863. https://doi.org/10.3390/ijerph17186863
Candela V, Longo UG, Di Naro C, Facchinetti G, Marchetti A, Sciotti G, Santamaria G, Piergentili I, De Marinis MG, Nazarian A, et al. A Historical Analysis of Randomized Controlled Trials in Rotator Cuff Tears. International Journal of Environmental Research and Public Health. 2020; 17(18):6863. https://doi.org/10.3390/ijerph17186863
Chicago/Turabian StyleCandela, Vincenzo, Umile Giuseppe Longo, Calogero Di Naro, Gabriella Facchinetti, Anna Marchetti, Gaia Sciotti, Giulia Santamaria, Ilaria Piergentili, Maria Grazia De Marinis, Ara Nazarian, and et al. 2020. "A Historical Analysis of Randomized Controlled Trials in Rotator Cuff Tears" International Journal of Environmental Research and Public Health 17, no. 18: 6863. https://doi.org/10.3390/ijerph17186863
APA StyleCandela, V., Longo, U. G., Di Naro, C., Facchinetti, G., Marchetti, A., Sciotti, G., Santamaria, G., Piergentili, I., De Marinis, M. G., Nazarian, A., & Denaro, V. (2020). A Historical Analysis of Randomized Controlled Trials in Rotator Cuff Tears. International Journal of Environmental Research and Public Health, 17(18), 6863. https://doi.org/10.3390/ijerph17186863






