Levels and Correlates of Physical Activity in Rural Ingwavuma Community, uMkhanyakude District, KwaZulu-Natal, South Africa
Abstract
1. Introduction
2. Methods
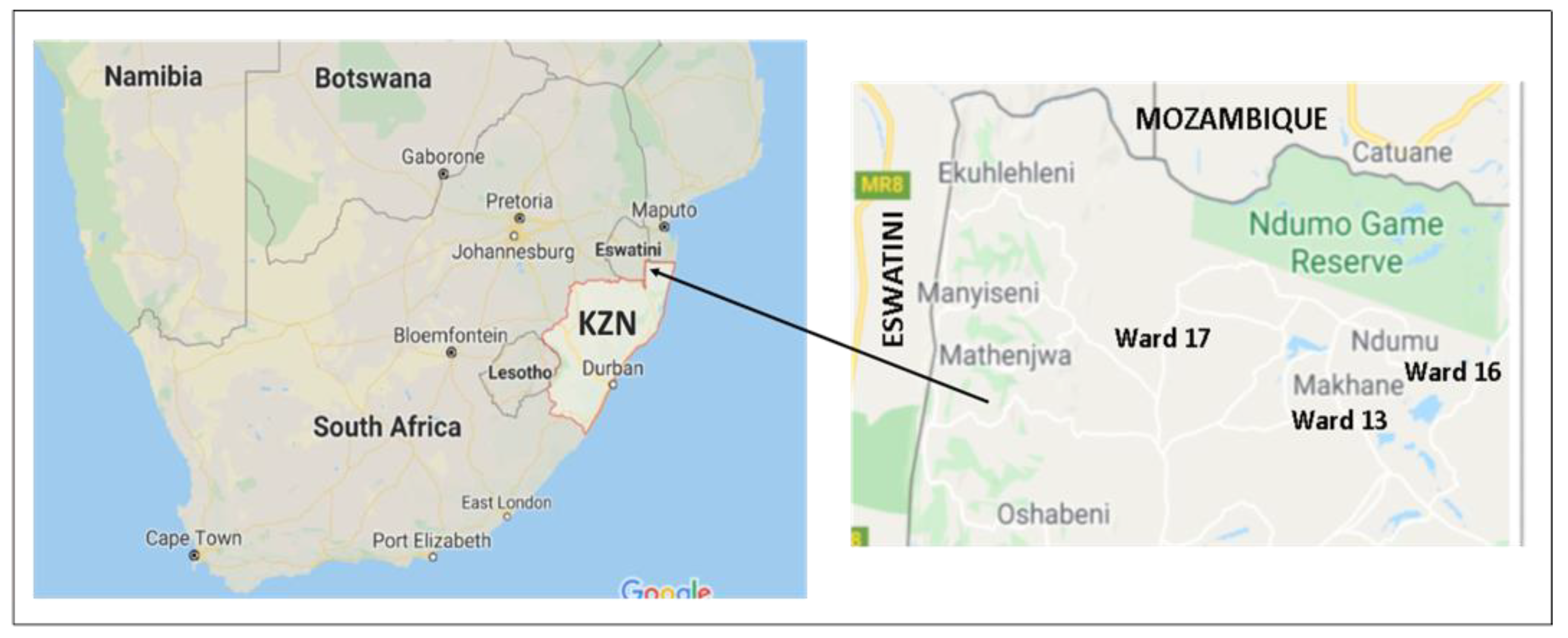
2.1. Study Design and Setting
2.2. Participants and Procedures
2.3. Measures
2.4. Analyses
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. Global Status Report on Noncommunicable Diseases 2014; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- Abegunde, D.O.; Mathers, C.D.; Adam, T.; Ortegon, M.; Strong, K. The burden and costs of chronic diseases in low-income and middle-income countries. Lancet 2007, 370, 1929–1938. [Google Scholar] [CrossRef]
- Pillay-van Wyk, V.; Msemburi, W.; Laubscher, R.; Dorrington, R.E.; Groenewald, P.; Glass, T.; Nojilana, B.; Joubert, J.D.; Matzopoulos, R.; Prinsloo, M.; et al. Mortality trends and differentials in South Africa from 1997 to 2012: Second national burden of disease study. Lancet Glob. Health 2016, 4, e642–e653. [Google Scholar] [CrossRef]
- Maredza, M.; Bertram, M.Y.; Tollman, S.M. Disease burden of stroke in rural South Africa: An estimate of incidence, mortality and disability adjusted life years. BMC Neurol. 2015, 15, 54. [Google Scholar] [CrossRef] [PubMed]
- Barquera, S.; Pedroza-Tobias, A.; Medina, C. Cardiovascular diseases in mega-countries: The challenges of the nutrition, physical activity and epidemiologic transitions, and the double burden of disease. Curr. Opin. Lipidol. 2016, 27, 329–344. [Google Scholar] [CrossRef] [PubMed]
- Mokdad, A.H.; Marks, J.S.; Stroup, D.F.; Gerberding, J.L. Actual causes of death in the United States, 2000. JAMA 2004, 291, 1238–1245. [Google Scholar] [CrossRef]
- Joubert, J.; Norman, R.; Lambert, E.V.; Groenewald, P.; Schneider, M.; Bull, F.; Bradshaw, D.; South African Comparative Risk Assessment Collaborating Group. Estimating the burden of disease attributable to physical inactivity in South Africa in 2000. S. Afr. Med. J. 2007, 97, 725–731. [Google Scholar] [PubMed]
- Kraus, W.E.; Houmard, J.A.; Duscha, B.D.; Knetzger, K.J.; Wharton, M.B.; McCartney, J.S.; Bales, C.W.; Henes, S.; Samsa, G.P.; Otvos, J.D.; et al. Effects of the amount and intensity of exercise on plasma lipoproteins. N. Engl. J. Med. 2002, 347, 1483–1492. [Google Scholar] [CrossRef]
- LeCheminant, J.D.; Tucker, L.A.; Bailey, B.W.; Peterson, T. The relationship between intensity of physical activity and HDL cholesterol in 272 women. J. Phys. Act. Health 2005, 2, 333–344. [Google Scholar] [CrossRef][Green Version]
- Wannamethee, S.G.; Shaper, A.G.; Walker, M. Physical activity and mortality in older men with diagnosed coronary heart disease. Circulation 2000, 102, 1358–1363. [Google Scholar] [CrossRef]
- Kikuchi, H.; Inoue, S.; Lee, I.M.; Odagiri, Y.; Sawada, N.; Inoue, M.; Tsugane, S. Impact of moderate-intensity and vigorous-intensity physical activity on mortality. Med. Sci. Sports Exerc. 2018, 50, 715–721. [Google Scholar] [CrossRef]
- Strong, W.B.; Malina, R.M.; Blimkie, C.J.; Daniels, S.R.; Dishman, R.K.; Gutin, B.; Hergenroeder, A.C.; Must, A.; Nixon, P.A.; Pivarnik, J.M.; et al. Evidence based physical activity for school-age youth. J. Pediatr. 2005, 146, 732–737. [Google Scholar] [CrossRef] [PubMed]
- Hallal, P.C.; Victora, C.G.; Azevedo, M.R.; Wells, J.C. Adolescent physical activity and health: A systematic review. Sports Med. 2006, 36, 1019–1030. [Google Scholar] [CrossRef] [PubMed]
- Van Langendonck, L.; Lefevre, J.; Claessens, A.L.; Thomis, M.; Philippaerts, R.; Delvaux, K.; Lysens, R.; Renson, R.; Vanreusel, B.; Vanden Eynde, B.; et al. Influence of participation in high-impact sports during adolescence and adulthood on bone mineral density in middle-aged men: A 27-year follow-up study. Am. J. Epidemiol. 2003, 158, 525–533. [Google Scholar] [CrossRef]
- WHO. Global Recommendations on Physical Activity for Health; World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- US Department of Health and Human Services. 2008 Physical Activity Guidelines for Americans; US Department of Health and Human Services: Washington, DC, USA, 2008. [Google Scholar]
- Manson, J.E.; Greenland, P.; LaCroix, A.Z.; Stefanick, M.L.; Mouton, C.P.; Oberman, A.; Perri, M.G.; Sheps, D.S.; Pettinger, M.B.; Siscovick, D.S. Walking compared with vigorous exercise for the prevention of cardiovascular events in women. N. Engl. J. Med. 2002, 347, 716–725. [Google Scholar] [CrossRef]
- Oyeyemi, A.L.; Moss, S.J.; Monyeki, M.A.; Kruger, H.S. Measurement of physical activity in urban and rural South African adults: A comparison of two self-report methods. BMC Public Health 2016, 16, 1004. [Google Scholar] [CrossRef] [PubMed]
- Cook, I.; Alberts, M.; Brits, J.S.; Choma, S.R.; Mkhonto, S.S. Descriptive epidemiology of ambulatory activity in rural, black South Africans. Med. Sci. Sports Exerc. 2010, 42, 1261–1268. [Google Scholar] [CrossRef]
- Kruger, H.S.; Venter, C.S.; Vorster, H.H.; Margetts, B.M. Physical inactivity is the major determinant of obesity in black women in the North West Province, South Africa: The THUSA study. Nutrition 2002, 18, 422–427. [Google Scholar] [CrossRef]
- Maimela, E.; Alberts, M.; Modjadji, S.E.; Choma, S.S.; Dikotope, S.A.; Ntuli, T.S.; Van Geertruyden, J.P. The prevalence and determinants of chronic non-communicable disease risk factors amongst adults in the dikgale Health Demographic and Surveillance System (HDSS) site, Limpopo province of South Africa. PLoS ONE 2016, 11, e0147926. [Google Scholar] [CrossRef]
- Jones, J.L. Transboundary conservation: Development implications for communities in KwaZulu-Natal, South Africa. Int. J. Sustain. Dev. World Ecol. 2009, 12, 266–278. [Google Scholar] [CrossRef]
- Schoeman, S.; Faber, M.; Adams, V.; Smuts, C.; Ford-Ngomane, N.; Laubscher, J.; Dhansay, M. Adverse social, nutrition and health conditions in rural districts of the KwaZulu-Natal and Eastern Cape provinces, South Africa. S. Afr. J. Clin. Nutr. 2016, 23, 140–147. [Google Scholar] [CrossRef]
- Nell, W.; de Crom, E.; Coetzee, H.; van Eeden, E. The psychosocial well-being of a “forgotten” South African community: The case of Ndumo, KwaZulu-Natal. J. Psychol. Afr. 2015, 25, 171–181. [Google Scholar] [CrossRef]
- Markides, J.; Forsythe, L. Looking back and Living forward: Indigenous Research Rising Up; Brill Sense: Leiden, The Netherlands, 2018; Volume 125. [Google Scholar]
- Morgenthal, T.L.; Kellner, K.; van Rensburg, L.; Newby, T.S.; van der Merwe, J.P.A. Vegetation and habitat types of the Umkhanyakude Node. S. Afr. J. Bot. 2006, 72, 1–10. [Google Scholar] [CrossRef]
- Hendriks, S.L.; Viljoen, A.; Marais, D.; Wenhold, F.; McIntyre, A.; Ngidi, M.; Van der Merwe, C.; Annandale, J.; Kalaba, M.; Stewart, D. The Current Rain-fed and Irrigated Production of Food Crops and Its Potential to Meet the Year-round Nutritional Requirements of Rural Poor People in North West, Limpopo, Kwazulu-Natal and the Eastern Cape: Report to the Water Research Commission and Department of Agriculture, Forestry & Fisheries; Water Research Commission: Pretoria, Soutth Africa, 2016. [Google Scholar]
- Initiative, H.H. Data Collection Tools for Challenging Environments; KoBoToolbox: Cambridge, MA, USA, 2018. [Google Scholar]
- Riley, L.; Guthold, R.; Cowan, M.; Savin, S.; Bhatti, L.; Armstrong, T.; Bonita, R. The World Health Organization STEPwise approach to noncommunicable disease risk-factor surveillance: Methods, challenges, and opportunities. Am. J. Public Health 2016, 106, 74–78. [Google Scholar] [CrossRef]
- Bahr, R.; Holme, I. Risk factors for sports injuries–A methodological approach. Br. J. Sports Med. 2003, 37, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.F.; Finan, C. Linear regression and the normality assumption. J. Clin. Epidemiol. 2018, 98, 146–151. [Google Scholar] [CrossRef]
- Lumley, T.; Diehr, P.; Emerson, S.; Chen, L. The importance of the normality assumption in large public health data sets. Annu. Rev. Public Health 2002, 23, 151–169. [Google Scholar] [CrossRef] [PubMed]
- Field, A. Discovering Statistics Using SPSS: (and Sex and Drugs and rock’n’roll); Sage: London, UK, 2009. [Google Scholar]
- Kruger, H.S.; Venter, C.S.; Vorster, H.H. Physical inactivity as a risk factor for cardiovascular disease in communities undergoing rural to urban transition: The THUSA study: Cardiovascular topics. Cardiovasc. J. S. Afr. 2003, 14, 16–23. [Google Scholar]
- Guthold, R.; Louazani, S.A.; Riley, L.M.; Cowan, M.J.; Bovet, P.; Damasceno, A.; Sambo, B.H.; Tesfaye, F.; Armstrong, T.P. Physical activity in 22 African countries: Results from the World Health Organization STEPwise approach to chronic disease risk factor surveillance. Am. J. Prev. Med. 2011, 41, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Kohrt, W.M.; Bloomfield, S.A.; Little, K.D.; Nelson, M.E.; Yingling, V.R.; American College of Sports Medicine. American College of Sports Medicine position stand: Physical activity and bone health. Med. Sci. Sports Exerc. 2004, 36, 1985–1996. [Google Scholar] [CrossRef]
- Fajemiroye, J.O.; Cunha, L.C.d.; Saavedra-Rodríguez, R.; Rodrigues, K.L.; Naves, L.M.; Mourão, A.A.; Silva, E.F.d.; Williams, N.E.E.; Martins, J.L.R.; Sousa, R.B.; et al. Aging-induced biological changes and cardiovascular diseases. BioMed Res. Int. 2018, 2018, 7156435. [Google Scholar] [CrossRef]
- Schuch, F.B.; Vancampfort, D.; Rosenbaum, S.; Richards, J.; Ward, P.B.; Veronese, N.; Solmi, M.; Cadore, E.L.; Stubbs, B. Exercise for depression in older adults: A meta-analysis of randomized controlled trials adjusting for publication bias. Braz. J. Psychiatry 2016, 38, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Usenbo, A.; Kramer, V.; Young, T.; Musekiwa, A. Prevalence of Arthritis in Africa: A Systematic Review and Meta-Analysis. PLoS ONE 2015, 10, e0133858. [Google Scholar] [CrossRef] [PubMed]
- Geere, J.A.; Hunter, P.R.; Jagals, P. Domestic water carrying and its implications for health: A review and mixed methods pilot study in Limpopo Province, South Africa. Environ. Health 2010, 9, 52. [Google Scholar] [CrossRef] [PubMed]
- Chimen, M.; Kennedy, A.; Nirantharakumar, K.; Pang, T.; Andrews, R.; Narendran, P. What are the health benefits of physical activity in type 1 diabetes mellitus? A literature review. Diabetologia 2012, 55, 542–551. [Google Scholar] [CrossRef]
- Elsawy, B.; Higgins, K.E. Physical activity guidelines for older adults. Am. Fam. Phys. 2010, 81, 55–59. [Google Scholar]
- Janssen, I.; LeBlanc, A.G. Systematic review of the health benefits of physical activity and fitness in school-aged children and youth. Int. J. Behav. Nutr. Phys. Act. 2010, 7, 40. [Google Scholar] [CrossRef]
- Dergance, J.M.; Calmbach, W.L.; Dhanda, R.; Miles, T.P.; Hazuda, H.P.; Mouton, C.P. Barriers to and benefits of leisure time physical activity in the elderly: Differences across cultures. J. Am. Geriatr. Soc. 2003, 51, 863–868. [Google Scholar] [CrossRef]
- Crombie, I.K.; Irvine, L.; Williams, B.; McGinnis, A.R.; Slane, P.W.; Alder, E.M.; McMurdo, M.E. Why older people do not participate in leisure time physical activity: A survey of activity levels, beliefs and deterrents. Age Ageing 2004, 33, 287–292. [Google Scholar] [CrossRef]
- Peel, N.M.; McClure, R.J.; Bartlett, H.P. Behavioral determinants of healthy aging. Am. J. Prev. Med. 2005, 28, 298–304. [Google Scholar] [CrossRef]
- Prince, S.A.; Adamo, K.B.; Hamel, M.E.; Hardt, J.; Connor Gorber, S.; Tremblay, M. A comparison of direct versus self-report measures for assessing physical activity in adults: A systematic review. Int. J. Behav. Nutr. Phys. Act. 2008, 5, 56. [Google Scholar] [CrossRef]
- Oyeyemi, A.L.; Kolo, S.M.; Rufai, A.A.; Oyeyemi, A.Y.; Omotara, B.A.; Sallis, J.F. Associations of neighborhood walkability with sedentary time in nigerian older adults. Int. J. Environ. Res. Public Health 2019, 16, 1879. [Google Scholar] [CrossRef] [PubMed]
| Variables | Female | Male | Total | ||||
|---|---|---|---|---|---|---|---|
| n | % (SD) | n | % (SD) | n | % (SD) | ||
| Age | 42.1 | 17.5 | 43.9 | 17.2 | 42.7 | 17.4 | |
| Age group | 18–29 | 82 | 30.9 | 34 | 26.8 | 116 | 29.6 |
| 30–39 | 56 | 21.1 | 25 | 19.7 | 81 | 20.7 | |
| 40–49 | 41 | 15.5 | 18 | 14.2 | 59 | 15.1 | |
| 50–59 | 40 | 15.1 | 24 | 18.9 | 64 | 16.3 | |
| 60+ | 46 | 17.4 | 26 | 20.5 | 72 | 18.4 | |
| Marital status | Single | 166 | 62.6 | 78 | 61.4 | 244 | 62.2 |
| Married | 42 | 15.9 | 20 | 15.8 | 62 | 15.8 | |
| Cohabiting | 36 | 13.6 | 25 | 19.7 | 61 | 15.6 | |
| Widowed/divorced | 21 | 7.9 | 4 | 3.2 | 25 | 6.4 | |
| Education level | None | 88 | 33.2 | 35 | 27.6 | 123 | 31.4 |
| Primary school | 67 | 25.3 | 38 | 29.9 | 105 | 26.8 | |
| Secondary school | 106 | 40.0 | 46 | 36.2 | 152 | 38.8 | |
| Post-secondary | 4 | 1.51 | 8 | 6.3 | 12 | 3.1 | |
| Occupational status | Unemployed | 229 | 86.4 | 98 | 77.2 | 327 | 83.4 |
| Self employed | 19 | 7.2 | 9 | 7.1 | 28 | 7.1 | |
| Employed | 4 | 1.5 | 10 | 7.9 | 14 | 3.6 | |
| Other | 13 | 4.9 | 10 | 7.9 | 23 | 5.9 | |
| Body mass index | Underweight | 14 | 5.3 | 12 | 9.5 | 26 | 6.6 |
| Normal-weight | 91 | 34.2 | 75 | 59.1 | 166 | 42.2 | |
| Overweight | 87 | 32.7 | 26 | 205 | 113 | 28.8 | |
| Obese | 74 | 27.8 | 14 | 11.0 | 88 | 22.4 | |
| Grow crops in field | No | 86 | 32.5 | 45 | 35.4 | 131 | 33.4 |
| Yes | 179 | 67.6 | 82 | 64.6 | 261 | 66.6 | |
| Inactivity may lead to poor health outcomes | No | 177 | 66.8 | 97 | 76.4 | 274 | 69.9 |
| Yes | 67 | 25.3 | 22 | 17.3 | 89 | 22.7 | |
| Don’t know | 21 | 7.9 | 8 | 6.3 | 29 | 7.4 | |
| Advised by a health worker to be physically active | No | 109 | 41.0 | 57 | 44.9 | 166 | 42.2 |
| Yes | 124 | 46.6 | 56 | 44.1 | 180 | 45.8 | |
| Don’t remember | 32 | 12.4 | 14 | 11.0 | 46 | 12.0 | |
| Total | 265 | 100 | 127 | 100 | 392 | 100 | |
| Variable | Weekly Physical Activity Level (%) | |||
|---|---|---|---|---|
| Low | Sufficient | High | ||
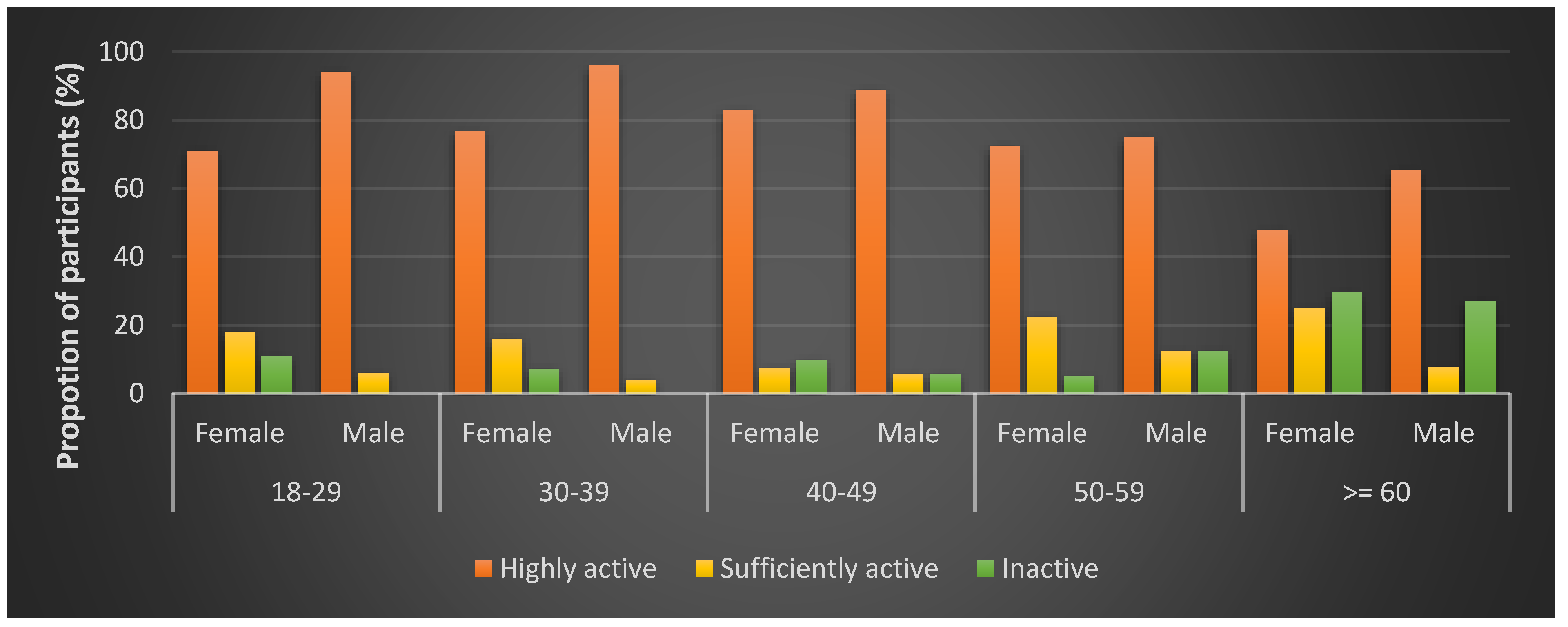
| Overall | 10.9 | 14.3 | 74.8 | |
| Gender | Female | 12.0 | 17.7 | 70.3 |
| Male | 8.7 | 7.1 | 84.3 | |
| Age group | <30 | 7.7 | 14.5 | 77.8 |
| 30–39 | 4.9 | 12.4 | 82.7 | |
| 40–49 | 8.5 | 6.8 | 84.8 | |
| 50–59 | 7.8 | 18.8 | 73.4 | |
| 60+ | 27.8 | 18.1 | 54.2 | |
| Marital status | Single | 6.5 | 12.2 | 81.2 |
| Cohabiting | 16.1 | 19.4 | 64.5 | |
| Married | 14.8 | 16.4 | 68.9 | |
| Other | 32.0 | 16.0 | 52.0 | |
| Education | None | 19.5 | 13.8 | 66.7 |
| Primary | 6.7 | 19.1 | 74.3 | |
| Secondary | 7.2 | 12.4 | 80.4 | |
| Post-secondary | 8.3 | 0.0 | 91.7 | |
| Occupational status | Unemployed | 12.5 | 14.0 | 73.5 |
| Self-employed | 3.6 | 17.9 | 78.6 | |
| Employed | 0.0 | 7.1 | 92.9 | |
| Other | 4.4 | 17.4 | 78.3 | |
| Body mass index | Underweight | 26.9 | 7.7 | 65.4 |
| Normal-weight | 7.8 | 13.3 | 78.9 | |
| Overweight | 8.6 | 12.9 | 78.5 | |
| Obese | 16.1 | 17.3 | 66.7 | |
| Inactivity may lead to poor health outcomes | No | 16.7 | 16.7 | 66.7 |
| Yes | 8.4 | 13.1 | 78.5 | |
| Don’t know | 17.2 | 17.2 | 65.5 | |
| Advised to be physically active | No | 10.8 | 17.5 | 71.7 |
| Yes | 11.7 | 11.1 | 77.2 | |
| Don’t remember | 8.5 | 14.9 | 76.6 | |
| Predictors | Model 1 | Model 2 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| B | β | Sig. | 95% CI | B | β | Sig. | 95% CI | ||
| Constant | 747.6 | 0.000 | 586.5–908.6 | 873.5 | 0.000 | 647.2–1099.7 | |||
| Gender | (Female) Male | 455.4 | 0.25 | 0.000 | 277.9–633.0 | 408.1 | 0.22 | 0.000 | 214.0–602.2 |
| Age-group | (18–29) 30–39 | 164.4 | 0.08 | 0.175 | −73.3–402.0 | 196.9 | 0.09 | 0.126 | −55.8–449.6 |
| 40–49 | 228.6 | 0.09 | 0.088 | −33.8–491.1 | 293.7 | 0.12 | 0.067 | −20.3–607.7 | |
| 50–59 | −3.3 | −0.00 | 0.980 | −259.2–252.7 | 107.1 | 0.05 | 0.548 | −243.4–457.7 | |
| ≥60 | −276.2 | −0.12 | 0.028 | −522.8–−29.7 | −109.5 | −0.05 | 0.547 | −467.2–248.1 | |
| Education | (Secondary) None | 68.8 | 0.04 | 0.653 | −231.8–369.4 | ||||
| Primary | −37.0 | −0.02 | 0.758 | −280.0–204.1 | |||||
| Post-secondary | −84.5 | −0.02 | 0.744 | −592.2–423.2 | |||||
| Marital status | (Single) Married | −259.1 | −0.12 | 0.037 | −503.0–−15.1 | ||||
| Cohabiting | −322.3 | −0.13 | 0.013 | −577.4–−67.1 | |||||
| Other | −313.2 | −0.09 | 0.102 | −688.6–62.2 | |||||
| Employment status | (Unemployed) Self employed | 213.2 | 0.06 | 0.207 | −118.6–545.1 | ||||
| Employed | 163.3 | 0.04 | 0.493 | −304.7–631.3 | |||||
| Other | −175.3 | −0.05 | 0.362 | −552.6–202.0 | |||||
| BMI | (Normal-weight) Underweight | −68.0 | −0.02 | 0.704 | −419.2–283.2 | ||||
| Overweight | −6.5 | −0.00 | 0.951 | −215.8–202.8 | |||||
| Obese | −203.7 | −0.01 | 0.087 | −437.4–29.9 | |||||
| Inactivity has adverse effects? | (Yes) No | −160.9 | −0.08 | 0.156 | −383.2–61.4 | ||||
| Don’t know | −339.7 | −0.10 | 0.040 | −663.8–−15.5 | |||||
| Advised to be active | (No) Yes | 42.2 | 0.03 | 0.530 | −89.9–174.4 | ||||
| R-squared | 0.92 | 0.000 | 0.14 | 0.118 | |||||
| Adjusted- R-squared | 0.81 | 0.10 | |||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chikafu, H.; Chimbari, M.J. Levels and Correlates of Physical Activity in Rural Ingwavuma Community, uMkhanyakude District, KwaZulu-Natal, South Africa. Int. J. Environ. Res. Public Health 2020, 17, 6739. https://doi.org/10.3390/ijerph17186739
Chikafu H, Chimbari MJ. Levels and Correlates of Physical Activity in Rural Ingwavuma Community, uMkhanyakude District, KwaZulu-Natal, South Africa. International Journal of Environmental Research and Public Health. 2020; 17(18):6739. https://doi.org/10.3390/ijerph17186739
Chicago/Turabian StyleChikafu, Herbert, and Moses J. Chimbari. 2020. "Levels and Correlates of Physical Activity in Rural Ingwavuma Community, uMkhanyakude District, KwaZulu-Natal, South Africa" International Journal of Environmental Research and Public Health 17, no. 18: 6739. https://doi.org/10.3390/ijerph17186739
APA StyleChikafu, H., & Chimbari, M. J. (2020). Levels and Correlates of Physical Activity in Rural Ingwavuma Community, uMkhanyakude District, KwaZulu-Natal, South Africa. International Journal of Environmental Research and Public Health, 17(18), 6739. https://doi.org/10.3390/ijerph17186739





