Neurological Complications of COVID-19 and Possible Neuroinvasion Pathways: A Systematic Review
Abstract
1. Introduction
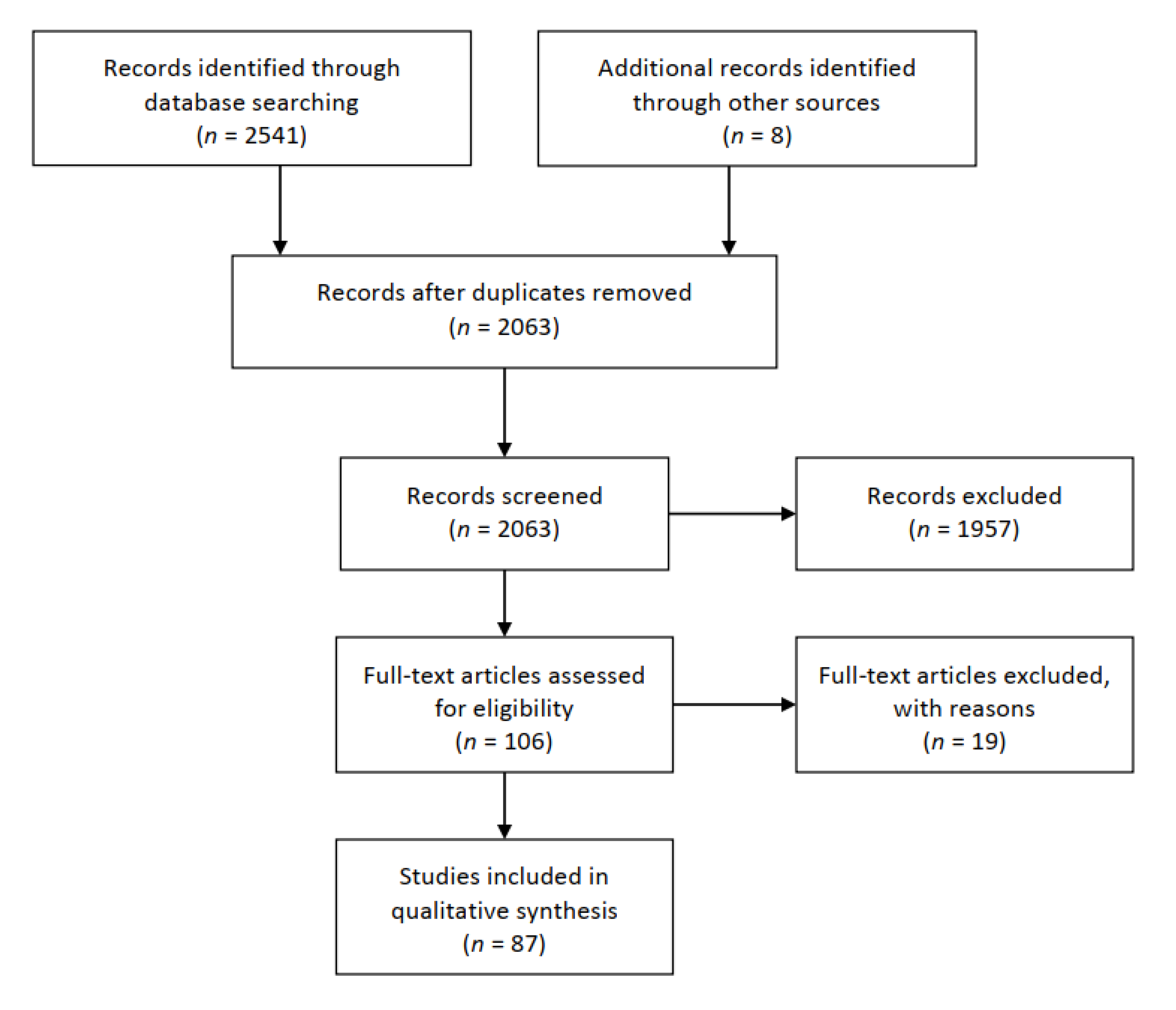
2. Materials and Methods
3. Results and Discussion
3.1. Categorization and Characteristics of the Selected Studies
3.1.1. Effects of SARS-CoV-2 on CNS
3.1.2. Effects of SARS-CoV-2 on PNS
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Paules, C.I.; Marston, H.D.; Fauci, A.S. Coronavirus Infections-More Than Just the Common Cold. JAMA 2020, 323, 707–708. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.Q.; Sun, B.Q.; Fang, Z.F.; Zhao, J.C.; Liu, X.Y.; Li, Y.M.; Sun, X.Z.; Liang, H.F.; Zhong, B.; Huang, Z.F.; et al. Distinct features of SARS-CoV-2-specific IgA response in COVID-19 patients. Eur. Respir. J. 2020, 56, 2001526. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Middle East Respiratory Syndrome Coronavirus (MERS-CoV); World Health Organization: Geneva, Switzerland, 2019. [Google Scholar]
- Lauer, S.A.; Grantz, K.H.; Bi, Q.; Jones, F.K.; Zheng, Q.; Meredith, H.R.; Azman, A.S.; Reich, N.G.; Lessler, J. The incubation period of coronavirus disease 2019 (COVID-19) from publicly reported confirmed cases: Estimation and application. Ann. Intern. Med. 2020, 172, 577–582. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Yuan, Y.; Cao, D.; Zhang, Y.; Ma, J.; Qi, J.; Wang, Q.; Lu, G.; Wu, Y.; Yan, J.; Shi, Y.; et al. Cryo-EM structures of MERS-CoV and SARS-CoV spike glycoproteins reveal the dynamic receptor binding domains. Nat. Commun. 2017, 8, 15092. [Google Scholar] [CrossRef]
- Zhou, P.; Yang, X.L.; Wang, X.G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.R.; Zhu, Y.; Li, B.; Huang, C.L.; et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef] [PubMed]
- Hamming, I.; Timens, W.; Bulthuis, M.L.C.; Lely, A.T.; Navis, G.J.; van Goor, H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004, 203, 631–637. [Google Scholar] [CrossRef]
- Wrapp, D.; Wang, N.; Corbett, K.S.; Goldsmith, J.A.; Hsieh, C.L.; Abiona, O.; Graham, B.S.; McLellan, J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science 2020, 367, 1260–1263. [Google Scholar] [CrossRef]
- Matsuyama, S.; Nao, N.; Shirato, K.; Kawase, M.; Saito, S.; Takayama, I.; Nagata, N.; Sekizuka, T.; Katoh, H.; Kato, F.; et al. Enhanced isolation of SARS-CoV-2 by TMPRSS2-expressing cells. Proc. Natl. Acad. Sci. USA 2020, 117, 7001–7003. [Google Scholar] [CrossRef]
- Sungnak, W.; Huang, N.; Bécavin, C.; Berg, M.; Queen, R.; Litvinukova, M.; Talavera-López, C.; Maatz, H.; Reichart, D.; Sampaziotis, F.; et al. SARS-CoV-2 entry factors are highly expressed in nasal epithelial cells together with innate immune genes. Nat. Med. 2020, 26, 681–687. [Google Scholar] [CrossRef]
- Tortorici, M.A.; Walls, A.C.; Lang, Y.; Wang, C.; Li, Z.; Koerhuis, D.; Boons, G.J.; Bosch, B.J.; Rey, F.A.; de Groot, R.J.; et al. Structural basis for human coronavirus attachment to sialic acid receptors. Nat. Struct. Mol. Biol. 2019, 26, 481–489. [Google Scholar] [CrossRef] [PubMed]
- Hulswit, R.J.G.; Lang, Y.; Bakkers, M.J.G.; Li, W.; Li, Z.; Schouten, A.; Ophorst, B.; van Kuppeveld, F.J.M.; Boons, G.J.; Bosch, B.J.; et al. Human coronaviruses OC43 and HKU1 bind to 9-O-acetylated sialic acids via a conserved receptor-binding site in spike protein domain A. Proc. Natl. Acad. Sci. USA 2019, 116, 2681–2690. [Google Scholar] [CrossRef]
- Chen, Z.; Mi, L.; Xu, J.; Yu, J.; Wang, X.; Jiang, J.; Xing, J.; Shang, P.; Qian, A.; Li, Y.; et al. Function of HAb18G/CD147 in invasion of host cells by severe acute respiratory syndrome coronavirus. J. Infect. Dis. 2005, 191, 755–760. [Google Scholar] [CrossRef] [PubMed]
- Callaway, E. Labs rush to study coronavirus in trans-genic animals-some are in short supply. Nature 2020, 579, 183. [Google Scholar] [CrossRef]
- Moriguchi, T.; Harii, N.; Goto, J.; Harada, D.; Sugawara, H.; Takamino, J.; Ueno, M.; Sakata, H.; Kondo, K.; Myose, N.; et al. A first case of meningitis/encephalitis associated with SARS-Coronavirus-2. Int. J. Infect. Dis. 2020, 94, 55–58. [Google Scholar] [CrossRef]
- Poyiadji, N.; Shahin, G.; Noujaim, D.; Stone, M.; Patel, S.; Griffith, B. COVID-19–associated Acute Hemorrhagic Necrotizing Encephalopathy: CT and MRI Features. Radiology 2020, 296, E119–E120. [Google Scholar] [CrossRef]
- Paniz-Mondolfi, A.; Bryce, C.; Grimes, Z.; Gordon, R.E.; Reidy, J.; Lednicky, J.; Sordillo, E.M.; Fowkes, M. Central nervous system involvement by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2). J. Med. Virol. 2020, 92, 699–702. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef]
- Al Saiegh, F.; Ghosh, R.; Leibold, A.; Avery, M.B.; Schmidt, R.F.; Theofanis, T.; Mouchtouris, N.; Philipp, L.; Peiper, S.C.; Wang, Z.-X.; et al. Status of SARS-CoV-2 in cerebrospinal fluid of patients with COVID-19 and stroke. Cereb. Dis. 2020, 91, 846–848. [Google Scholar]
- Al-olama, M.; Rashid, A.; Garozzo, D. COVID-19-associated meningoencephalitis complicated with intracranial hemorrhage: A case report. Acta Neurochir. 2020, 162, 1495–1499. [Google Scholar] [CrossRef]
- Barrios-López, J.M.; Rego-García, I.; Martínez, C.M.; Romero-Fábrega, J.C.; Rodríguez, M.R.; Giménez, J.A.R.; Escamilla-Sevilla, F.; Mínguez-Castellanos, A.; Pérez, M.D.F. Ischaemic stroke and SARS-CoV-2 infection: A causal or incidental association? Neurología 2020, 35, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Benger, M.; Williams, O.; Siddiqui, J.; Sztriha, L. Intracerebral haemorrhage and COVID-19: Clinical characteristics from a case series. Brain Behav. Immun. 2020, 88, 940–944. [Google Scholar] [CrossRef] [PubMed]
- Beyrouti, R.; Adams, M.E.; Benjamin, L.; Cohen, H.; Farmer, S.F.; Goh, Y.Y.; Humphries, F.; Jäger, H.R.; Losseff, N.A.; Perry, R.J.; et al. Characteristics of ischaemic stroke associated with COVID-19. J. Neurol. Neurosurg. Psychiatry 2020, 91, 889–891. [Google Scholar] [CrossRef]
- Co, C.O.C.; Yu, J.R.T.; Laxamana, L.C.; David-Ona, D.I.A. Intravenous Thrombolysis for Stroke in a COVID-19 Positive Filipino Patient, a Case Report. J. Clin. Neurosci. 2020, 77, 234–236. [Google Scholar] [CrossRef] [PubMed]
- Craen, A.; Logan, G.; Ganti, L. Novel Coronavirus Disease 2019 and Subarachnoid Hemorrhage: A Case Report. Cureus 2020, 12, e7846. [Google Scholar] [CrossRef]
- Deliwala, S.; Abdulhamid, S.; Abusalih, M.F.; Al-Qasmi, M.M.; Bachuwa, G. Encephalopathy as the Sentinel Sign of a Cortical Stroke in a Patient Infected With Coronavirus Disease-19 (COVID-19). Cureus 2020, 12, e8121. [Google Scholar] [CrossRef]
- Duong, L.; Xu, P.; Liu, A. Meningoencephalitis without respiratory failure in a young female patient with COVID-19 infection in Downtown Los Angeles, early April 2020. Brain Behav. Immun. 2020, 87, 33. [Google Scholar] [CrossRef]
- Fasano, A.; Cavallieri, F.; Canali, E.; Valzania, F. First motor seizure as presenting symptom of SARS-CoV-2 infection. Neurol. Sci. 2020, 41, 1651–1653. [Google Scholar] [CrossRef]
- Filatov, A.; Sharma, P.; Hindi, F.; Espinosa, P.S. Neurological Complications of Coronavirus Disease (COVID-19). Encephalopathy. Cureus 2020, 12, e7352. [Google Scholar] [CrossRef]
- Flamand, M.; Perron, A.; Buron, Y.; Szurhaj, W. Pay more attention to EEG in COVID-19 pandemic. Clin. Neurophysiol. 2020, 131, 2062–2064. [Google Scholar] [CrossRef]
- Franceschi, A.M.; Ahmed, O.; Giliberto, L.; Castillo, M. Hemorrhagic Posterior Reversible Encephalopathy Syndrome as a Manifestation of COVID-19 Infection. Am. J. Neuroradiol. 2020, 41, 1173–1176. [Google Scholar] [CrossRef] [PubMed]
- Frisullo, G.; Bellavia, S.; Scala, I.; Piano, C.; Morosetti, R.; Brunetti, V.; Calabresi, P.; Marca, G.D. Stroke and COVID19: Not only a large-vessel disease. J. Stroke Cereb. Dis. 2020, 29, 105074. [Google Scholar] [CrossRef] [PubMed]
- Gane, S.B.; Kelly, C.; Hopkins, C. Isolated sudden onset anosmia in COVID-19 infection. A novel syndrome? Rhinology 2020, 58, 299–301. [Google Scholar] [CrossRef] [PubMed]
- Garaci, F.; Giuliano, F.D.; Picchi, E.; Ros, V.D.; Floris, R. Venous cerebral thrombosis in COVID-19 patient. J. Neurol. Sci. 2020, 414, 116871. [Google Scholar] [CrossRef] [PubMed]
- Gilani, S.; Roditi, R.; Naraghi, M. COVID-19 and anosmia in Tehran, Iran. Med. Hypotheses 2020, 141, 109757. [Google Scholar] [CrossRef]
- Goldberg, M.F.; Goldberg, M.F.; Cerejo, R.; Tayal, A.H. Cerebrovascular Disease in COVID-19. Am. J. Neuroradiol. 2020, 41, 1170–1172. [Google Scholar] [CrossRef]
- Haddadi, K.; Ghasemian, R.; Shafizad, M. Basal Ganglia Involvement and Altered Mental Status: A Unique Neurological Manifestation of Coronavirus Disease 2019. Cureus 2020, 12, e7869. [Google Scholar] [CrossRef]
- Hayashi, M.; Sahashi, Y.; Baba, Y.; Okura, H.; Shimohata, T. COVID-19 associated mild encephalitis/encephalopathy with a reversible splenial lesion. J. Neurol. Sci. 2020, 415, 116941. [Google Scholar] [CrossRef]
- Hjelmesæth, J.; Skaare, D. Loss of smell or taste as the only symptom of COVID-19. Tidsskr Nor Legeforen 2020, 140. [Google Scholar] [CrossRef]
- Kadono, Y.; Nakamura, Y.; Ogawa, Y.; Yamamoto, S.; Kajikawa, R.; Nakajima, Y.; Matsumoto, M.; Kishima, H. A case of COVID-19 infection presenting with a seizure following severe brain edema. Seizure Eur. J. Epilepsy 2020, 80, 53–55. [Google Scholar] [CrossRef]
- Kaya, Y.; Karab, S.; Akincic, C.; Kocamana, A.S. Transient cortical blindness in COVID-19 pneumonia; a PRES-like syndrome: Case report. J. Neurol. Sci. 2020, 413, 116858. [Google Scholar] [CrossRef]
- Kishfy, L.; Casasola, M.; Banankhah, P.; Parvez, A.; Jan, Y.J.; Shenoy, A.M.; Thomson, C.; AbdelRazek, M.A. Posterior reversible encephalopathy syndrome (PRES) as a neurological association in severe Covid-19. J. Neurol. Sci. 2020, 414, 116943. [Google Scholar] [CrossRef] [PubMed]
- Manganelli, F.; Vargas, M.; Iovino, A.; Iacovazzo, C.; Santoro, L.; Servillo, G. Brainstem involvement and respiratory failure in COVID-19. Neurol. Sci. 2020, 41, 1663–1665. [Google Scholar] [CrossRef] [PubMed]
- Mawhinney, J.A.; Wilcock, C.; Haboubi, H.; Roshanzamir, S. Neurotropism of SARS-CoV-2: COVID-19 presenting with an acute manic episode. BMJ Case Rep. 2020, 13, e236123. [Google Scholar] [CrossRef] [PubMed]
- Melley, L.E.; Bress, E.; Polan, E. Hypogeusia as the initial presenting symptom of COVID-19. BMJ Case Rep. 2020, 13, 236080. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, S.; Petridis, A.; Cornelius, J.F.; Hänggi, D. Letter to editor: Severe brain haemorrhage and concomitant COVID-19 Infection: A neurovascular complication of COVID-19. Brain Behav. Immun. 2020, 87, 150–151. [Google Scholar] [CrossRef] [PubMed]
- Noro, F.; de Mendonça Cardoso, F.; Marchiori, E. COVID-19 and benign intracranial hypertension: A case report. J. Braz. Soc. Trop. Med. 2020, 53, e20200325. [Google Scholar] [CrossRef]
- Ollarves-Carrero, M.F.; Rodriguez-Morales, A.G.; Bonilla-Aldana, D.K.; Rodriguez-Morales, A.J. Anosmia in a healthcare worker with COVID-19 in Madrid, Spain. Travel Med. Infect. Dis. 2020, 35, 101666. [Google Scholar] [CrossRef]
- Ottaviano, G.; Carecchio, M.; Scarpa, B.; Marchese-Ragona, R. Olfactory and rhinological evaluations in SARS-CoV-2 patients complaining of olfactory loss. Rhinology 2020, 58, 400–401. [Google Scholar] [CrossRef]
- Oxley, T.J.; Mocco, J.; Majidi, S.; Kellner, C.P.; Shoirah, H.; Singh, I.P.; Leacy, R.A.D.; Shigematsu, T.; Ladner, T.R.; Yaeger, K.A.; et al. Large-Vessel Stroke as a Presenting Feature of Covid-19 in the Young. N. Engl. J. Med. 2020, 382, e60. [Google Scholar] [CrossRef]
- Reichard, R.R.; Kashani, K.B.; Boire, N.A.; Constantopoulos, E.; Guo, Y.; Lucchinetti, C.F. Neuropathology of COVID-19: A spectrum of vascular and acute disseminated encephalomyelitis (ADEM)-like pathology. Acta Neuropathol. 2020, 140, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Rogg, J.; Baker, A.; Tung, G. Posterior reversible encephalopathy syndrome (PRES): Another imaging manifestation of COVID-19. Interdiscip. Neurosurg. 2020, 22, 100808. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Razavi, A.; Karimi, N.; Rouhani, N. COVID-19 and intracerebral haemorrhage: Causative or coincidental? New Microbes New Infect. 2020, 35, 100669. [Google Scholar] [CrossRef] [PubMed]
- Shoskes, A.; Migdady, I.; Fernandez, A.; Ruggieri, P.; Rae-Grant, A. Cerebral Microhemorrhage and Purpuric Rash in COVID-19: The Case for a Secondary Microangiopathy. J. Stroke Cereb. Dis. 2020, 29, 105111. [Google Scholar] [CrossRef]
- Somani, S.; Pati, S.; Gaston, T.; Chitlangia, A.; Agnihotri, S. De Novo Status Epilepticus in patients with COVID-19. Ann. Clin. Transl. Neurol. 2020, 7, 1240–1244. [Google Scholar] [CrossRef]
- Tunç, A.; Ünlübaş, Y.; Alemdar, M.; Akyüz, E. Coexistence of COVID-19 and acute ischemic stroke report of four cases. J. Clin. Neurosci. 2020, 77, 227–229. [Google Scholar] [CrossRef]
- Viguier, A.; Delamarre, L.; Duplantier, J.; Olivot, J.M.; Bonneville, F. Acute ischemic stroke complicating common carotid artery thrombosis during a severe COVID-19 infection. J. Neuroradiol. 2020, 47, 393–394. [Google Scholar] [CrossRef]
- Wong, P.F.; Craik, S.; Newman, P.; Makan, A.; Srinivasan, K.; Crawford, E.; Dev, D.; Moudgil, H.; Ahmad, N. Lessons of the month 1: A case of rhombencephalitis as a rare complication of acute COVID-19 infection. Clin. Med. 2020, 20, 293–294. [Google Scholar] [CrossRef]
- Ye, M.; Ren, Y.; Lv, T. Encephalitis as a clinical manifestation of COVID-19. Brain Behav. Immun. 2020, 88, 945–946. [Google Scholar] [CrossRef]
- Zanin, L.; Saraceno, G.; Panciani, P.P.; Renisi, G.; Signorini, L.; Migliorati, K.; Fontanella, M.M. SARS-CoV-2 can induce brain and spine demyelinating lesions. Acta Neurochir. 2020, 162, 1491–1494. [Google Scholar] [CrossRef]
- Zhai, P.; Ding, Y.; Li, Y. The impact of COVID-19 on ischemic stroke. Diagn. Pathol. 2020, 15, 78. [Google Scholar] [CrossRef]
- Zoghi, A.; Ramezani, M.; Roozbeh, M.; Darazam, I.A.; Sahraian, M.A. A case of possible atypical demyelinating event of the central nervous system following COVID-19. Mult. Scler. Relat. Disord. 2020, 44, 102324. [Google Scholar] [CrossRef] [PubMed]
- Avula, A.; Nalleballe, K.; Narula, N.; Sapozhnikov, S.; Dandu, V.; Toom, S.; Glaser, A.; Elsayegh, D. COVID-19 presenting as stroke. Brain Behav. Immun. 2020, 87, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Kandemirli, S.G.; Dogan, L.; Sarikaya, Z.T.; Kara, S.; Akinci, C.; Kaya, D.; Kaya, Y.; Yildirim, D.; Tuzuner, F.; Yildirim, M.S.; et al. Brain MRI Findings in Patients in the Intensive Care Unit with COVID-19 Infection. Radiology 2020. [Google Scholar] [CrossRef] [PubMed]
- Lechien, J.R.; Chiesa-Estomba, C.M.; Siati, D.R.D.; Horoi, M.; Bon, S.D.L.; Rodriguez, A.; Dequanter, D.; Blecic, S.; Afia, F.E.; Distinguin, L.; et al. Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): A multicenter European study. Rhinology 2020, 277, 2251–2261. [Google Scholar]
- Lu, L.; Xiong, W.; Liu, D.; Liu, J.; Yang, D.; Li, N.; Mu, J.; Guo, J.; Li, W.; Wang, G.; et al. New onset acute symptomatic seizure and risk factors in coronavirus disease 2019: A retrospective multicenter study. Epilepsia 2020, 61, e49–e53. [Google Scholar] [CrossRef]
- Mao, L.; Jin, H.; Wang, M.; Hu, Y.; Chen, S.; He, Q.; Chang, J.; Hong, C.; Zhou, Y.; Wang, D.; et al. Neurologic Manifestations of Hospitalized Patients With Coronavirus Disease. JAMA Neurol. 2020, 77, 683–690. [Google Scholar] [CrossRef]
- Morassi, M.; Bagatto, D.; Cobelli, M.; D’Agostini, S.; Gigli, G.L.; Bnà, C.; Vogrig, A. Stroke in patients with SARS-CoV-2 infection: Case series. J. Neurol. 2020, 267, 2185–2192. [Google Scholar] [CrossRef]
- Petrescu, A.M.; Taussig, D.; Bouilleret, V. Electroencephalogram (EEG) in COVID-19: A systematic retrospective study. Clin. Neurophysiol. 2020, 50, 155–165. [Google Scholar] [CrossRef]
- Radmanesh, A.; Derman, A.; Lui, Y.W.; Raz, E.; Loh, J.P.; Hagiwara, M.; Borja, M.J.; Zan, E.; Fatterpekar, G.M. COVID-19 associated Diffuse Leukoencephalopathy and Microhemorrhages. Radiology 2020. [Google Scholar] [CrossRef]
- Scullen, T.; Keen, J.; Mathkour, M.; Dumont, A.S.; Kahn, L. Coronavirus 2019 (COVID-19)-Associated Encephalopathies and Cerebrovascular Disease: The New Orleans Experience. World Neurosurg. 2020, 141, e437–e446. [Google Scholar] [CrossRef] [PubMed]
- Spinato, G.; Fabbris, C.; Polesel, J.; Cazzador, D.; Borsetto, D.; Hopkins, C.; Boscolo-Rizzo, P. Alterations in Smell or Taste in Mildly Symptomatic Outpatients With SARS-CoV-2 Infection. JAMA 2020, 323, 2089–2090. [Google Scholar] [CrossRef] [PubMed]
- Helms, J.; Kremer, S.; Merdji, H.; Clere-Jehl, R.; Schenck, M.; Kummerlen, C.; Collange, O.; Boulay, C.; Fafi-Kremer, S.; Ohana, M.; et al. Neurologic Features in Severe SARS-CoV-2 Infection. N. Engl. J. Med. 2020, 382, 2268–2270. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, C.; Surda, P.; Whitehead, E.; Kumar, B.N. Early recovery following new onset anosmia during the COVID-19 pandemic—An observational cohort study. J. Otolaryngol.-Head Neck Surg. 2020, 49, 26. [Google Scholar] [CrossRef] [PubMed]
- Vaira, L.A.; Deiana, G.; Fois, A.G.; Pirina, P.; Madeddu, G.; Vito, A.D.; Babudieri, S.; Petrocelli, M.; Serra, A.; Bussu, F.; et al. Objective evaluation of anosmia and ageusia in COVID-19 patients: Single-center experience on 72 cases. Head Neck 2020, 42, 1252–1258. [Google Scholar] [CrossRef] [PubMed]
- Vaira, L.A.; Hopkins, C.; Petrocelli, M.; Lechien, J.R.; Chiesa-Estomba, C.M.; Salzano, G.; Cucurullo, M.; Salzano, F.A.; Saussez, S.; Boscolo-Rizzo, P.; et al. Smell and taste recovery in coronavirus disease 2019 patients: A 60-day objective and prospective study. J. Laryngol. Otol. 2020, 12, 1–7. [Google Scholar] [CrossRef]
- Karadaş, Ö.; Öztürk, B.; Sonkaya, A.R. A prospective clinical study of detailed neurological manifestations in patients with COVID-19. Neurol. Sci. 2020, 41, 1991–1995. [Google Scholar] [CrossRef]
- Lee, Y.; Min, P.; Lee, S.; Kim, S.W. Prevalence and Duration of Acute Loss of Smell or Taste in Covid-19 Patients. J. Korean Med. Sci. 2020, 35, e174. [Google Scholar] [CrossRef]
- Speth, M.M.; Singer-Cornelius, T.; Oberle, M.; Gengler, I.; Brockmeier, S.J.; Sedaghat, A.R. Olfactory Dysfunction and Sinonasal Symptomatology in COVID-19: Prevalence, Severity, Timing, and Associated Characteristics. Otolaryngol.–Head Neck Surg. 2020, 163, 114–120. [Google Scholar] [CrossRef]
- Beltrán-Corbellini, A.; Chico-García, J.L.; Martínez-Poles, J.; Rodríguez-Jorge, F.; Natera-Villalba, E.; Gómez-Corral, J.; Gómez-López, A.; Monreal, E.; Parra-Díaz, P.; Cortés-Cuevas, J.L.; et al. Acute-onset smell and taste disorders in the context of COVID-19: A pilot multicentre polymerase chain reaction based case–control study. Eur. J. Neurol. 2020. [Google Scholar] [CrossRef]
- Carignan, A.; Valiquette, L.; Grenier, C.; Musonera, J.B.; Nkengurutse, D.; Marcil-Héguy, A.; Vettese, K.; Marcoux, D.; Valiquette, C.; Xiong, W.T.; et al. Anosmia and dysgeusia associated with SARS-CoV-2 infection: An age-matched case–control study. Can. Med. Assoc. J. 2020, 192, E702–E707. [Google Scholar] [CrossRef] [PubMed]
- Hornuss, D.; Lange, B.; Schröter, N.; Rieg, S.; Kern, W.V.; Wagner, D. Anosmia in COVID-19 patients. Clin. Microbiol. Infect. 2020. [Google Scholar] [CrossRef] [PubMed]
- Dell’Era, V.; Farri, F.; Garzaro, G.; Gatto, M.; Aluffi Valletti, P.; Garzaro, M. Smell and taste disorders during COVID-19 outbreak: A cross-sectional study on 355 patients. Head Neck 2020, 42, 1591–1596. [Google Scholar] [CrossRef] [PubMed]
- Giacomelli, A.; Pezzati, L.; Conti, F.; Bernacchia, D.; Siano, M.; Oreni, L.; Rusconi, S.; Gervasoni, C.; Ridolfo, A.L.; Rizzardini, G.; et al. Self-reported Olfactory and Taste Disorders in Patients With Severe Acute Respiratory Coronavirus 2 Infection: A Cross-sectional Study. Clin. Infect. Dis. 2020, 71, 889–890. [Google Scholar] [CrossRef]
- Abdelnour, L.; Abdalla, M.E.; Babiker, S. COVID 19 infection presenting as motor peripheral neuropathy. J. Formos. Med. Assoc. 2020, 119, 1119–1120. [Google Scholar] [CrossRef]
- Alberti, P.; Beretta, S.; Piatti, M.; Karantzoulis, A.; Piatti, M.L.; Santoro, P.; Viganò, M.; Giovannelli, G.; Pirro, F.; Montisano, D.A.; et al. Guillain-Barré syndrome related to COVID-19 infection. Neurol. Neuroimmunol. Neuroinflammation 2020, 7, e741. [Google Scholar] [CrossRef] [PubMed]
- Coen, M.; Jeanson, G.; Almeida, L.A.C.; Hübers, A.; Stierlin, F.; Najjar, I.; Ongaro, M.; Moulin, K.; Makrygianni, M.; Leemann, B.; et al. Guillain-Barré syndrome as a complication of SARS-CoV-2 infection. Brain Behav. Immun. 2020, 87, 111–112. [Google Scholar] [CrossRef]
- Dinkin, M.; Gao, V.; Kahan, J.; Bobker, S.; Simonetto, M.; Wechsler, P.; Harpe, J.; Greer, C.; Mints, G.; Salama, G.; et al. COVID-19 presenting with ophthalmoparesis from cranial nerve palsy. Neurology 2020, 95, 221–223. [Google Scholar] [CrossRef]
- Farzi, M.A.; Ayromlou, H.; Jahanbakhsh, N.; Bavil, P.H.; Janzadeh, A.; Shayan, F.K. Guillain-Barré syndrome in a patient infected with SARS-CoV-2, a case report. J. Neuroimmunol. 2020, 346, 577294. [Google Scholar] [CrossRef]
- Gutiérrez-Ortiz, C.; Méndez, A.; Rodrigo-Rey, S.; Pedro-Murillo, E.S.; Bermejo-Guerrero, L.; Gordo-Mañas, R.; Aragón-Gómez, F.D.; Benito-León, J. Miller Fisher Syndrome and polyneuritis cranialis in COVID-19. Neurology 2020, 95, e601–e605. [Google Scholar] [CrossRef]
- Homma, Y.; Watanabe, M.; Inoue, K.; Moritaka, T. Coronavirus Disease-19 Pneumonia with Facial Nerve Palsy and Olfactory Disturbance. Intern. Med. 2020, 59, 1773–1775. [Google Scholar] [CrossRef] [PubMed]
- Hutchins, K.L.; Jansen, J.H.; Comer, A.D.; Scheer, R.V.; Zahn, G.S.; Capps, A.E.; Weaver, L.M.; Koontz, N.A. COVID-19 Associated Bifacial Weakness with Paresthesia Subtype of Guillain-Barré Syndrome. Am. J. Neuroradiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Lantos, J.E.; Strauss, S.B.; Lin, E. COVID-19 Associated Miller Fisher Syndrome: MRI Findings. Am. J. Neuroradiol. 2020, 41, 1184–1186. [Google Scholar] [CrossRef]
- Ottaviani, D.; Boso, F.; Tranquillini, E.; Gapeni, I.; Pedrotti, G.; Cozzio, S.; Guarrera, G.M.; Giometto, B. Early Guillain-Barré syndrome in coronavirus disease 2019 (COVID-19): A case report from an Italian COVID-hospital. Neurol. Sci. 2020, 41, 1351–1354. [Google Scholar] [CrossRef] [PubMed]
- Padroni, M.; Mastrangelo, V.; Asioli, G.M.; Pavolucci, L.; Abu-Rumeileh, S.; Piscaglia, M.G.; Querzani, P.; Callegarini, C.; Foschi, M. Guillain-Barré syndrome following COVID-19: New infection, old complication? J. Neurol. 2020, 267, 1877–1879. [Google Scholar] [CrossRef]
- Rana, S.; Lima, A.A.; Chandra, R.; Valeriano, J.; Desai, T.; Freiberg, W.; Small, G. Novel Coronavirus (COVID-19)-Associated Guillain–Barré Syndrome. J. Clin. Neuromuscul. Dis. 2020, 21, 240–242. [Google Scholar] [CrossRef]
- Sedaghat, Z.; Karimi, N. Guillain Barre syndrome associated with COVID-19 infection: A case report. J. Clin. Neurosci. 2020, 76, 233–235. [Google Scholar] [CrossRef]
- Scheidl, E.; Canseco, D.D.; Hadji-Naumov, A.; Bereznai, B. Guillain-Barré syndrome during SARS-CoV-2 pandemic: A case report and review of recent literature. J. Peripher. Nerv. Syst. 2020, 25, 204–207. [Google Scholar] [CrossRef]
- Toscano, G.; Palmerini, F.; Ravaglia, S.; Ruiz, L.; Invernizzi, P.; Cuzzoni, M.G.; Franciotta, D.; Baldanti, F.; Daturi, R.; Postorino, P.; et al. Guillain–Barré Syndrome Associated with SARS-CoV-2. N. Engl. J. Med. 2020, 382, 2574–2576. [Google Scholar] [CrossRef]
- Virani, A.; Rabold, E.; Hanson, T.; Haag, A.; Elrufay, R.; Cheema, T.; Balaan, M.; Bhanot, N. Guillain-Barré Syndrome associated with SARS-CoV-2 infection. IDCases 2020, 20, e00771. [Google Scholar] [CrossRef]
- Webb, S.; Wallace, V.C.; Martin-Lopez, D.; Yogarajah, M. Guillain-Barré syndrome following COVID-19: A newly emerging post-infectious complication. BMJ Case Rep. 2020, 13, e236182. [Google Scholar] [CrossRef]
- Wei, H.; Yin, H.; Huang, M.; Guo, Z. The 2019 novel coronavirus pneumonia with onset of oculomotor nerve palsy: A case study. J. Neurol. 2020, 267, 1550–1553. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Shen, D.; Zhou, H.; Liu, J.; Chen, S. Guillain-Barré syndrome associated with SARS-CoV-2 infection: Causality or coincidence? Lancet 2020, 19, 383–384. [Google Scholar] [CrossRef]
- Netland, J.; Meyerholz, D.K.; Moore, S.; Cassell, M.; Perlman, S. Severe Acute Respiratory Syndrome Coronavirus Infection Causes Neuronal Death in the Absence of Encephalitis in Mice Transgenic for Human ACE2. J. Virol. 2008, 82, 7264–7275. [Google Scholar] [CrossRef] [PubMed]
- Brann, D.H.; Tsukahara, T.; Weinreb, C.; Lipovsek, M.; den Berge, K.V.; Gong, B.; Chance, R.; Macaulay, I.C.; Chou, H.J.; Fletcher, R.B.; et al. Non-neuronal expression of SARS-CoV-2 entry genes in the olfactory system suggests mechanisms underlying COVID-19-associated anosmia. Sci. Adv. 2020, 6, eabc5801. [Google Scholar] [CrossRef]
- Fodoulian, L.; Tuberosa, J.; Rossier, D.; Landis, B.N.; Carleton, A.; Rodriguez, I. SARS-CoV-2 receptor and entry genes are expressed by sustentacular cells in the human olfactory neuroepithelium. BioRxiv 2020. [Google Scholar] [CrossRef]
- Ralli, M. Defining the burden of olfactory dysfunction in COVID-19 patients. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 3440–3441. [Google Scholar] [PubMed]
- Orrù, G.; Ciacchini, R.; Gemignani, A.; Conversano, C. Psychological intervention measures during the COVID-19 pandemic. Clin. Neuropsychiatry 2020, 17, 76–79. [Google Scholar]
- Marazziti, D.; Pozza, A.; Di Giuseppe, M.; Conversano, C. The psychosocial impact of COVID-19 pandemic in Italy: A lesson for mental health prevention in the first severely hit European country. Psychol. Trauma Theory Res. Pract. Policy 2020, 12, 531–533. [Google Scholar] [CrossRef]
- Muratori, P.; Ciacchini, R. Children and the COVID-19 transition: Psychological reflections and suggestions on adapting to the emergency. Clin. Neuropsychiatry 2020, 17, 131–134. [Google Scholar]
- Di Giuseppe, M.; Gemignani, A.; Conversano, C. Psychological resources against the traumatic experience of COVID-19. Clin. Neuropsychiatry 2020, 17, 85–87. [Google Scholar]
- Conversano, C.; Marchi, L.; Miniati, M. Psychological distress among healthcare professionals involved in the covid-19 emergency: Vulnerability and resilience factors. Clin. Neuropsychiatry 2020, 17, 94–96. [Google Scholar]
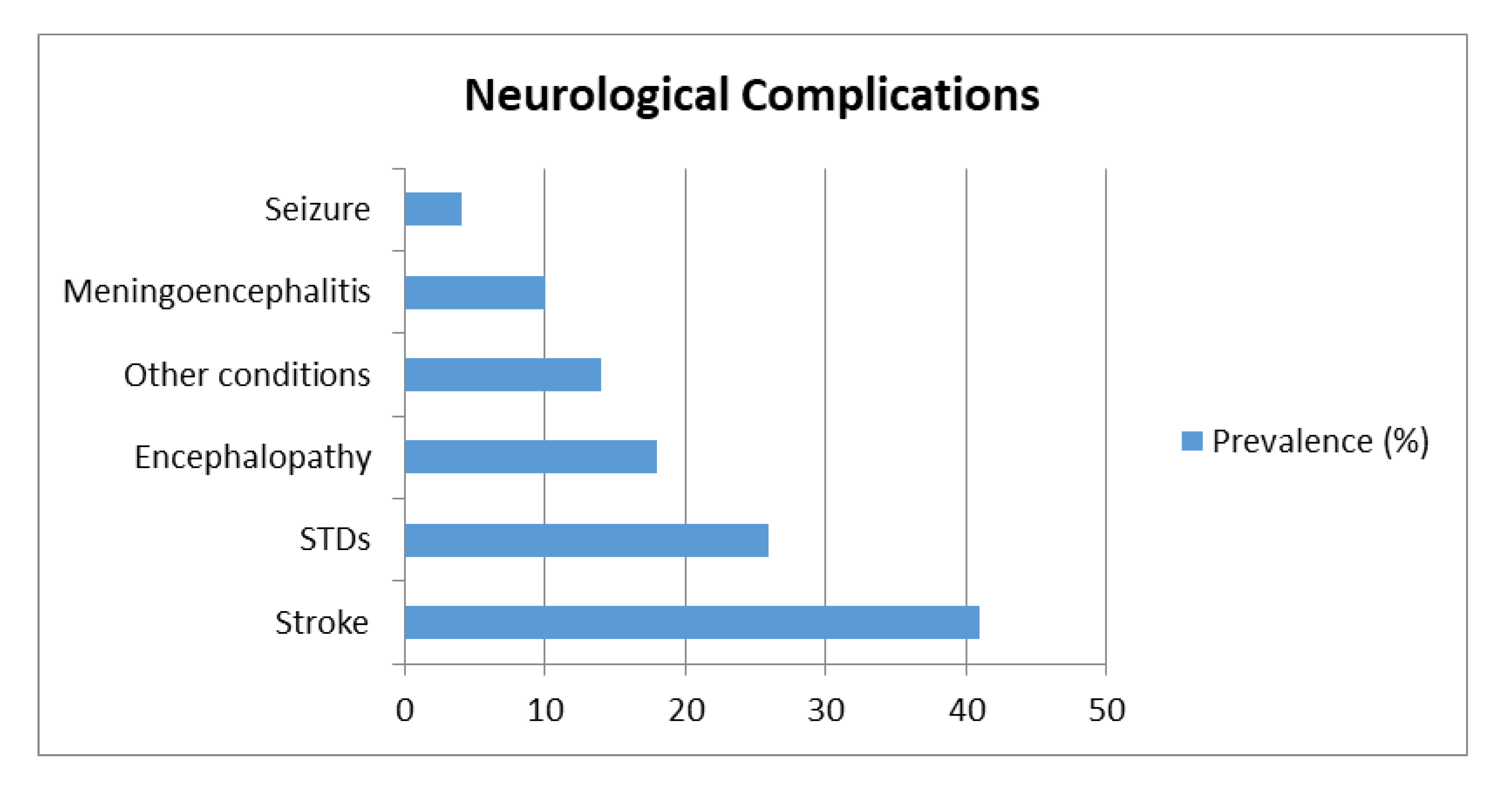
| Article/Sample (n) | p-Value/rho Value/Odd Ratio |
|---|---|
| (a) Stroke | |
| Al Saiegh et al. [20] n = 2 | N.A. |
| Avula et al. [64] n = 4 | N.A. |
| Barrios-Lòpez et al. [22] n = 4 | N.A. |
| Beyrouti et al. [24] n = 6 | N.A. |
| Benger et al. [23] n = 5 | N.A. |
| Co et al. [25] n = 1 | N.A. |
| Craen et al. [26] n = 1 | N.A. |
| Deliwala et al. [27] n = 1 | N.A. |
| Frisullo et al. [33] n = 1 | N.A. |
| Garaci et al. [35] n = 1 | N.A. |
| Goldberg et al. [37] n = 1 | N.A. |
| Haddadi et al. [38] n = 1 | N.A. |
| Helms et al. [82] n = 58 | N.A. |
| Kandemirli et al. [65] n = 23 | N.A. |
| Karadaş et al. [74] n = 239 | N.A. |
| Lu et al. [67] n = 304 | N.A. |
| Manganelli et al. [44] n = 3 | N.A. |
| Mao et al. [68] n = 214 | Stroke prevalence (p = 0.03); Major Complications (p = 0.02) |
| Morassi et al. [69] n = 6 | N.A. |
| Muhammad et al. [47] n = 1 | N.A. |
| Oxley et al. [51] n = 5 | N.A. |
| Radmanesh et al. [71] n = 11 | N.A. |
| Reichard et al. [52] n = 1 | N.A. |
| Sharifi-Razavi et al. [54] n = 1 | N.A. |
| Shoskes et al. [55] n = 1 | N.A. |
| Tunç et al. [57] n = 4 | N.A. |
| Viguier et al. [58] n = 1 | N.A. |
| Zhai et al. [62] n = 1 | N.A. |
| (b) STDs | |
| Beltrán-Corbellini et al. [81] n = 79 | p = 0.003: STDs more frequent among cases |
| Carignan et al. [82] n = 134 | p < 0.001: STDs more frequent among cases as compared to controls |
| Dell’Era et al. [84] n = 355 | p < 0.001: full recovery after 14 days |
| Gane et al. [34] n = 1 | N.A. |
| Giacomelli et al. [85] n = 59 | p < 0.001: association between anosmia and dysgeusia |
| p = 0.036: STDs more frequent among F | |
| p = 0.035 STDs more frequent among younger patients | |
| Gilani et al. [36] n = 8 | N.A. |
| Hjelmeseth and Skaare [40] n = 60 | N.A. |
| Hopkins et al. [75] n = 382 | p < 0.001: symptoms improvement after 2 weeks |
| Hornuss et al. [83] n = 45 | p < 0.001: STDs more frequent among cases |
| Lechien et al. [66] n = 417 | p < 0.001: anosmia and dysgeusia and more frequent among F |
| Lee et al. [79] n = 3191 | p = 0.01: more common in F |
| p < 0.001 more common in young patients | |
| Melley et al. [46] n = 1 | N.A. |
| Ollarves-Carrero et al. [49] n = 1 | N.A. |
| Ottaviano et al. [50] n = 6 | N.A. |
| Speth et al. [80] n = 103 | p < 0.001: prevalence of STDs |
| OR = 0.96: negative correlation between STDs and older age | |
| OR = 2.46: positive correlation between STDs and F | |
| ρ = 0.87: correlation between severity of taste and olfactory disorders | |
| Spinato et al. [73] n = 202 | p = 0.02: STDs more frequent among women |
| Vaira et al. [76] n = 72 | p = 0.003: taste disorders worse in older patients (age ≥ 50) |
| p = 0.001: STDs improvement after 15 days from the onset | |
| Vaira et al. [77] n = 345 | p = 0.000: symptoms improvement after one or two weeks |
| (c) Encephalopathy | |
| Filatov et al. [30] n = 1 | N.A. |
| Flamand et al. [31] n = 1 | N.A. |
| Franceschi et al. [32] n = 2 | N.A. |
| Hayashi et al. [39] n = 1 | N.A. |
| Kaya et al. [42] n = 1 | N.A. |
| Kishfy et al. [43] n = 2 | N.A. |
| Lu et al. [67] n = 304 | N.A. |
| Poyiadji et al. [17] n = 1 | N.A. |
| Radmanesh et al. [71] n = 11 | N.A. |
| Rogg et al. [53] n = 1 | N.A. |
| Scullen et al. [72] n = 76 | N.A. |
| (d) Meningoencephalitis | |
| Al-olama et al. [21] n = 1 | N.A. |
| Duong et al. [28] n = 1 | N.A. |
| Moriguchi et al. [16] n = 1 | N.A. |
| Reichard et al. [52] n = 1 | N.A. |
| Wong et al. [59] n = 1 | N.A. |
| Ye et al. [60] n = 1 | N.A. |
| Zoghi et al. [63] n = 1 | N.A. |
| (e) Seizures | |
| Fasano et al. [29] n = 1 | N.A. |
| Lu et al. [67] n = 304 | N.A. |
| Somani et al. [56] n = 2 | N.A. |
| (f) Other Neurological Conditions * | |
| Kadono et al. [41] n = 1 | N.A. |
| Karadas et al. [78] n = 239 | N.A. |
| Mawhinney et al. [45] n = 1 | N.A. |
| Noro et al. [48] n = 1 | N.A. |
| Petrescu et al. [70] n = 36 | N.A. |
| Scullen et al. [72] n = 76 | N.A. |
| Zanin et al. [61] n = 1 | N.A. |
| Mao et al. [68] n = 214 | p < 0.001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Orrù, G.; Conversano, C.; Malloggi, E.; Francesconi, F.; Ciacchini, R.; Gemignani, A. Neurological Complications of COVID-19 and Possible Neuroinvasion Pathways: A Systematic Review. Int. J. Environ. Res. Public Health 2020, 17, 6688. https://doi.org/10.3390/ijerph17186688
Orrù G, Conversano C, Malloggi E, Francesconi F, Ciacchini R, Gemignani A. Neurological Complications of COVID-19 and Possible Neuroinvasion Pathways: A Systematic Review. International Journal of Environmental Research and Public Health. 2020; 17(18):6688. https://doi.org/10.3390/ijerph17186688
Chicago/Turabian StyleOrrù, Graziella, Ciro Conversano, Eleonora Malloggi, Francesca Francesconi, Rebecca Ciacchini, and Angelo Gemignani. 2020. "Neurological Complications of COVID-19 and Possible Neuroinvasion Pathways: A Systematic Review" International Journal of Environmental Research and Public Health 17, no. 18: 6688. https://doi.org/10.3390/ijerph17186688
APA StyleOrrù, G., Conversano, C., Malloggi, E., Francesconi, F., Ciacchini, R., & Gemignani, A. (2020). Neurological Complications of COVID-19 and Possible Neuroinvasion Pathways: A Systematic Review. International Journal of Environmental Research and Public Health, 17(18), 6688. https://doi.org/10.3390/ijerph17186688








