A Perspective on Inhabited Urban Space: Land Use and Occupation, Heat Islands, and Precarious Urbanization as Determinants of Territorial Receptivity to Dengue in the City of Rio De Janeiro
Abstract
1. Introduction
2. Methods
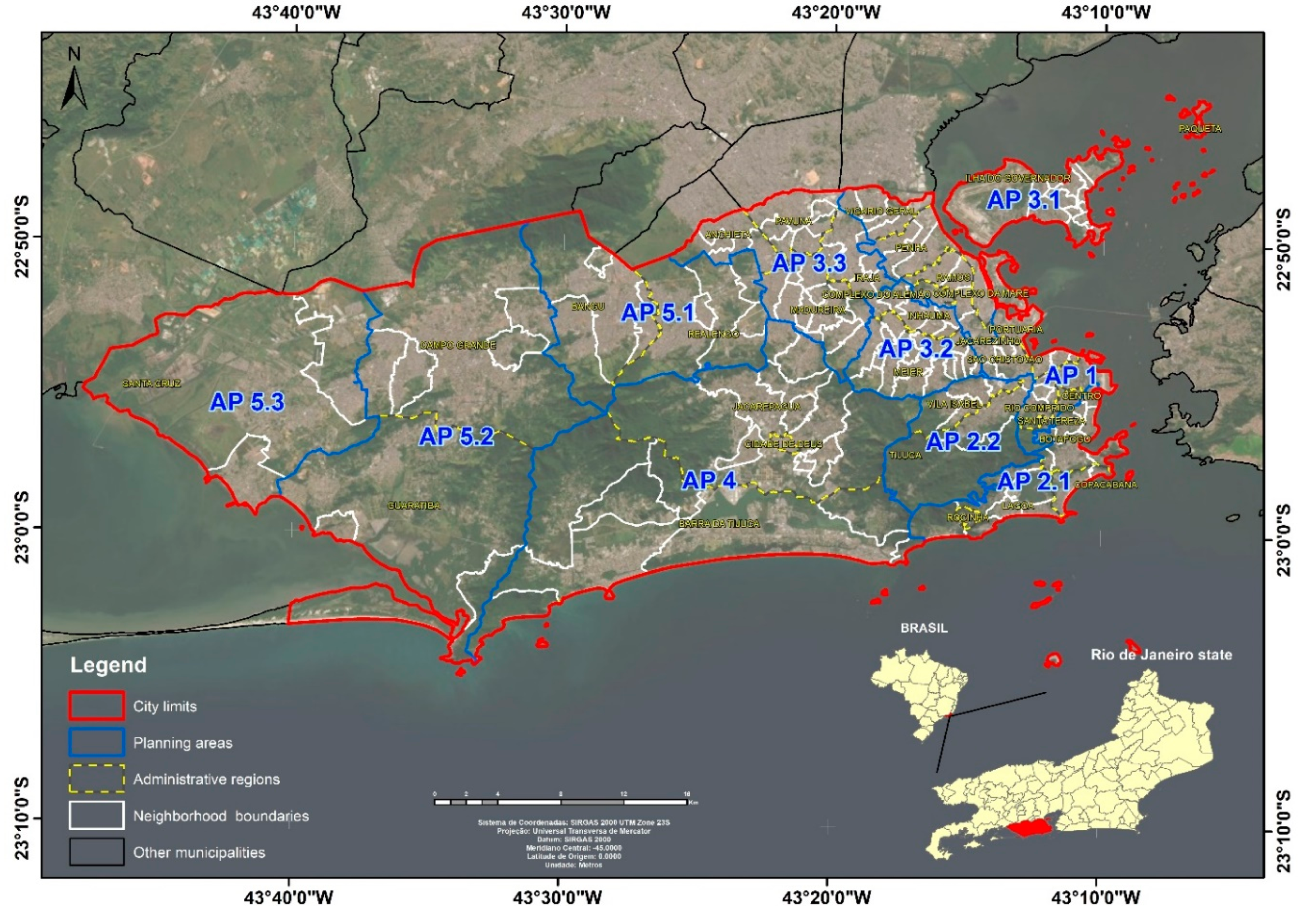
2.1. Study Area
2.2. Study Design
2.3. Construction of Indicators
2.4. Data Analysis
3. Data Sources
3.1. Epidemiological Data
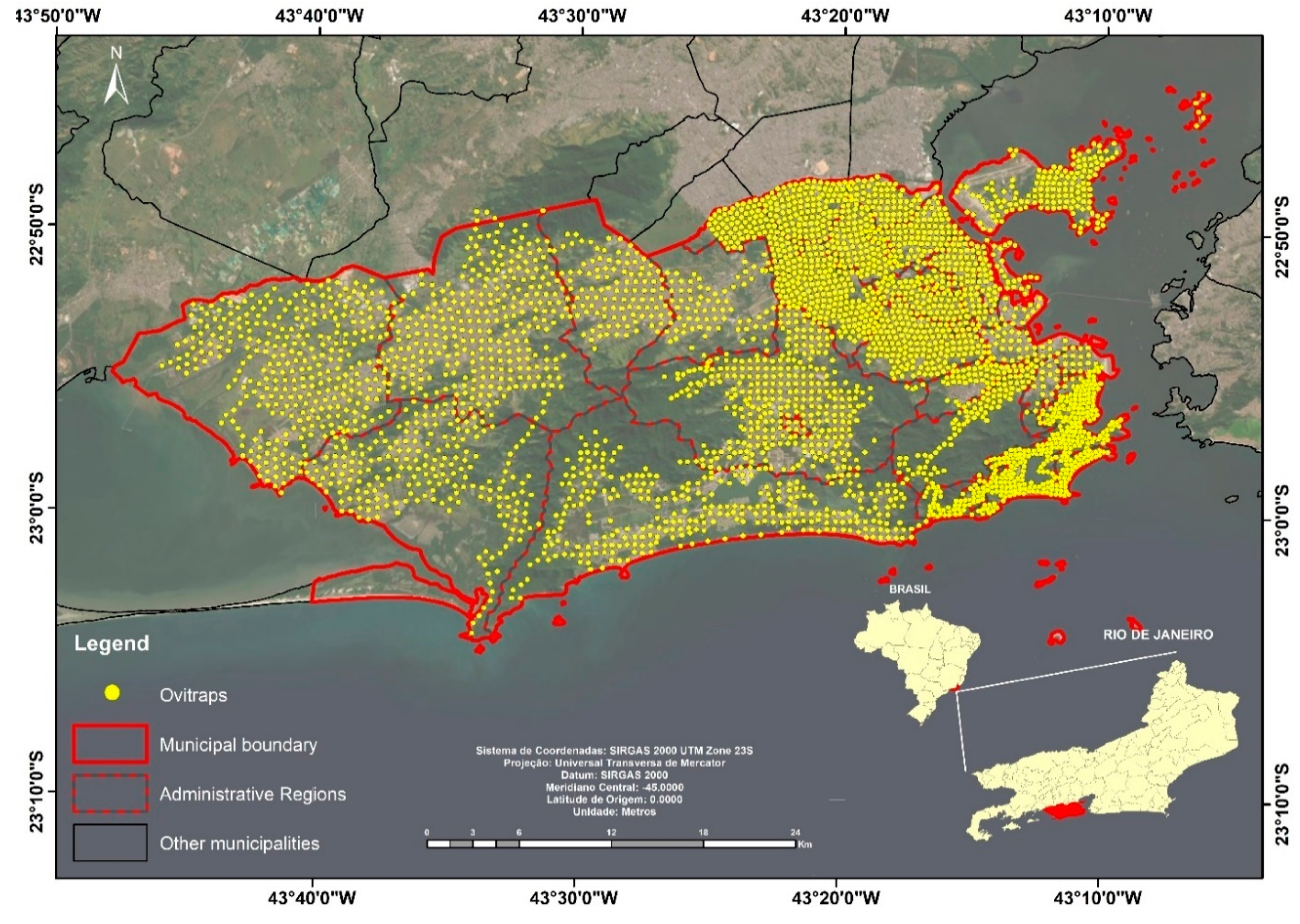
3.2. Entomological Data
3.3. Socioeconomic Data
3.4. Spatial Data
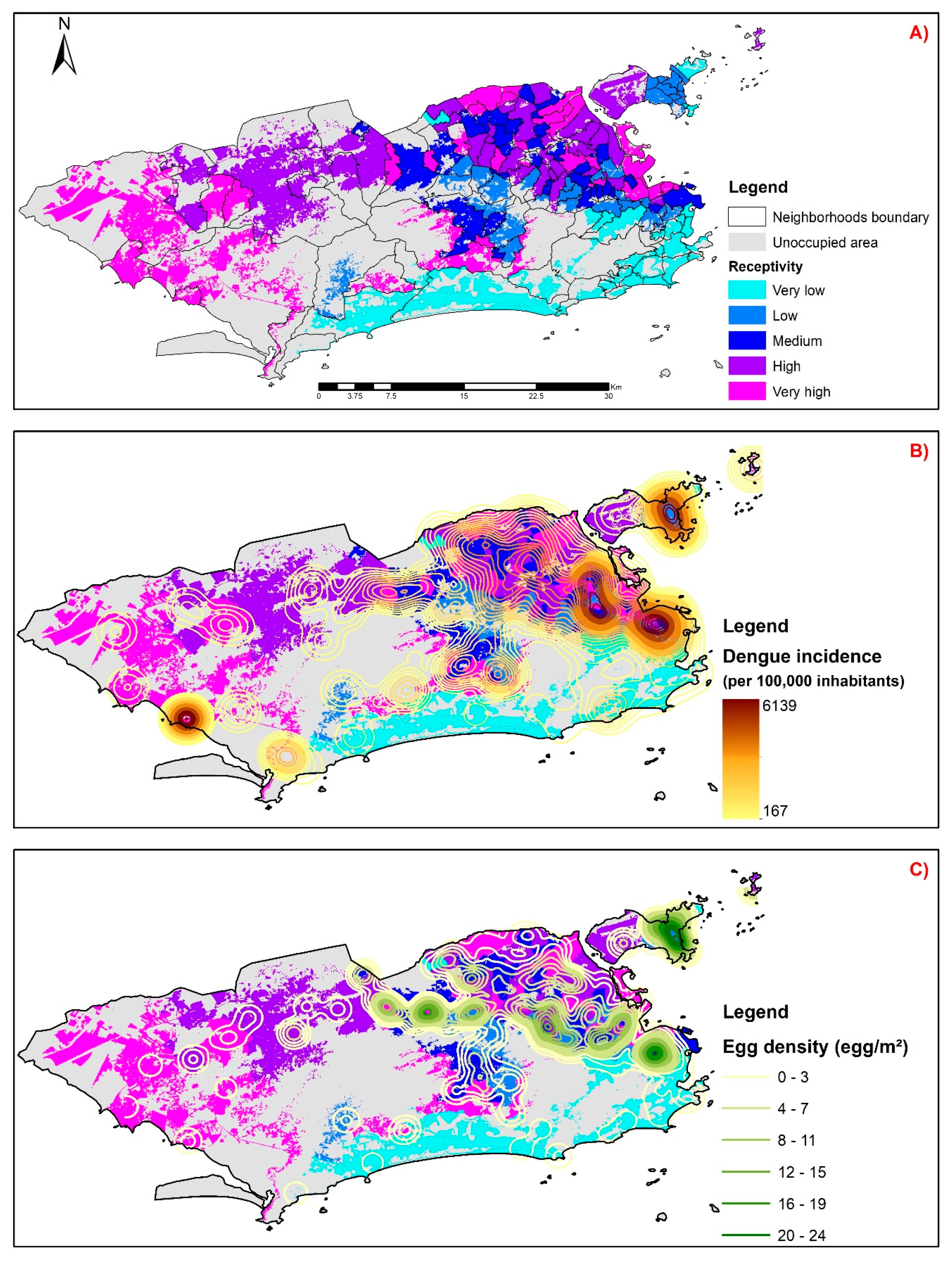
4. Results
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Weaver, S.C.; Reisen, W.K. Present and future arboviral threats. Antivir. Res. 2010, 85, 328–345. [Google Scholar] [CrossRef] [PubMed]
- Honório, N.A.; Câmara, D.C.P.; Calvet, G.A.; Brasil, P. Chikungunya: Uma arbovirose em estabelecimento e expansão no Brasil. Cadernos de Saúde Púb. 2015, 31, 906–908. [Google Scholar] [CrossRef]
- Lima-Camara, T.N. Emerging arboviruses and public health challenges in Brazil. Rev. De Saúde Pública. 2016, 50. Available online: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0034-89102016000100602&lng=en&tlng=en (accessed on 25 March 2018).
- Lowe, R.; Barcellos, C.; Brasil, P.; Cruz, O.; Honório, N.; Kuper, H.; Sá Carvalho, M. The Zika Virus Epidemic in Brazil: From Discovery to Future Implications. Int. J. Environ. Res. Public Health 2018, 15, 96. [Google Scholar] [CrossRef] [PubMed]
- Kuno, G. Review of the Factors Modulating Dengue Transmission. Epidem. Rev. 1995, 17, 321–335. [Google Scholar] [CrossRef] [PubMed]
- Braack, L.; Gouveia de Almeida, A.P.; Cornel, A.J.; Swanepoel, R.; de Jager, C. Mosquito-borne arboviruses of African origin: Review of key viruses and vectors. Parasites Vectors. 2018, 11. Available online: https://parasitesandvectors.biomedcentral.com/articles/10.1186/s13071-017-2559-9 (accessed on 12 June 2018).
- Bhatt, S.; Gething, P.W.; Brady, O.J.; Messina, J.P.; Farlow, A.W.; Moyes, C.L.; Drake, J.M.; Brownstein, J.S.; Hoen, A.G.; Sankoh, O.; et al. The global distribution and burden of dengue. Nature 2013, 496, 504–507. [Google Scholar] [CrossRef]
- Randolph, S.E.; Rogers, D.J. The arrival, establishment and spread of exotic diseases: Patterns and predictions. Nat. Rev. Microb. 2010, 8, 361–371. [Google Scholar] [CrossRef]
- Santos, M. O Espaco Dividido: Os dois Circuitos da Economia Urbana dos Paises Subdesenvolvidos, 2nd ed.; USP: São Paulo, Brazil, 2008. [Google Scholar]
- Sabroza, P.C.; Leal, M.C. Saúde, ambiente e desenvolvimento: Alguns conceitos fundamentais. In Saúde, Ambiente e Desenvolvimento; HUCITEC-ABRASCO: Rio de Janeiro, Brazil, 1992; pp. 45–94. [Google Scholar]
- Carvalho, M.S.; Honório, N.A.; Garcia, L.M.T.; Carvalho, L.C.S. Aedes aegypti control in urban areas: A systemic approach to a complex dynamic. PLoS Neg. Trop. Dis. 2017, 11, e0005632. [Google Scholar] [CrossRef]
- Barcellos, C.C.; Sabroza, P.C.; Peiter, P.; Rojas, L.I. Organização espacial, saúde e qualidade de vida: Análise espacial e uso de indicadores na avaliação de situações de saúde. In Informe Epidemiológico do Sus; 2002; 11. Available online: http://scielo.iec.pa.gov.br/scielo.php?script=sci_arttext&pid=S0104-16732002000300003&lng=pt&nrm=iso&tlng=pt (accessed on 12 June 2018). [CrossRef]
- Le Menach, A.; Tatem, A.J.; Cohen, J.M.; Hay, S.I.; Randell, H.; Patil, A.P.; Smith, D.L. Travel risk, malaria importation and malaria transmission in Zanzibar. Sci. Rep. 2011, 1. Available online: http://www.nature.com/articles/srep00093 (accessed on 3 August 2018). [CrossRef]
- Tauil, P.L. The prospect of eliminating malaria transmission in some regions of Brazil. Mem. Inst. Oswaldo Cruz 2011, 106, 105–106. [Google Scholar] [CrossRef][Green Version]
- Harvey, D. Justiça Social e a Cidade; Edward Arnold: London, UK, 1973. [Google Scholar]
- Vasconcelos, P.A. A cidade, o urbano, o lugar. Revista GEOUSP 1999, 6, 11–15. [Google Scholar]
- Castilho, C.J.M. O Ambiente Urbano Numa Perspectiva Interdisciplinar: Discussão de Conceitos que Tratam das Inter-Relações Sociedade-Natureza, a partir da Geografia do Recife. Rev. Brasileira de Geografia Física 2011, 5, 872–896. [Google Scholar]
- Honório, N.A.; Codeço, C.T.; Alves, F.C.; Magalhães, M.A.F.M.; Lourenço-de-Oliveira, R. Temporal Distribution of Aedes aegypti in Different Districts of Rio De Janeiro, Brazil, Measured by Two Types of Traps. J. Med. Entom. 2009, 46, 1001–1014. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, R.M.R.; Araújo, J.M.G.; Schatzmayr, H.G. Dengue viruses in Brazil, 1986–2006. Rev. Panamericana de Salud Púb. 2007, 22, 358–363. [Google Scholar] [CrossRef]
- Fares, R.C.G.; Souza, K.P.R.; Añez, G.; Rios, M. Epidemiological Scenario of Dengue in Brazil. BioMed Res. Int. 2015, 2015, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Monken, M.; Barcellos, C. Vigilância em saúde e território utilizado: Possibilidades teóricas e metodológicas. Cadernos de Saúde Púb. 2005, 21, 898–906. [Google Scholar] [CrossRef] [PubMed]
- Flauzino, R.F.; Souza-Santos, R.; Oliveira, R.M. Dengue, geoprocessamento e indicadores socioeconômicos e ambientais: Um estudo de revisão. Rev. Panam. Salud Pub. 2009, 25, 456–461. [Google Scholar] [CrossRef][Green Version]
- Gondim, G.M.M. Territórios da Atenção Básica: Múltiplos, Singulares ou Inexistentes? Ph.D. Thesis, Escola Nacional de Saúde Pública, Rio de Janeiro, Brazil, 2011. [Google Scholar]
- Instituto Brasileiro de Geografia e Estatística. Censo Demográfico 2010: Resultados do Universo por Setor Censitário; 2011. Available online: https://censo2010.ibge.gov.br/ (accessed on 10 June 2018).
- Lessa, C. O Rio De Todos Os Brasis—Uma Reflexão Em Busca Da Auto-Estima, 1st ed.; Record: Rio de Janeiro, Brazil, 2000; 478p. [Google Scholar]
- Rosa-Freitas, M.G.; Tsouris, P.; Reis, I.C.; Magalhães, M.d.A.F.M.; do Nascimento, T.F.S.; Honório, N.A. Dengue and Land Cover Heterogeneity in Rio de Janeiro. Oecologia Aust. 2010, 14, 641–667. [Google Scholar] [CrossRef]
- Romeo-Aznar, V.; Paul, R.; Telle, O.; Pascual, M. Mosquito-borne transmission in urban landscapes: The missing link between vector abundance and human density. Proc. Biol. Sci. 2018, 285. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6111166/ (accessed on 19 July 2020). [CrossRef]
- IPP IPP. Rio em Síntese. 2017. Available online: http://www.data.rio/pages/rio-em-sntese-2 (accessed on 9 June 2019).
- SIURB IPP. Sistema Municipal de Informações Urbanas. 2017. Available online: http://www.rio.rj.gov.br/web/ipp/siurb (accessed on 26 June 2018).
- Barbosa, G.L.; Lage, M.D.O.; Andrade, V.R.; Gomes, A.H.A.; Quintanilha, J.A.; Chiaravalloti-Neto, F. Influence of strategic points in the dispersion of Aedes aegypti in infested areas. Revista de Saúde Pública 2019, 53, 29. [Google Scholar] [CrossRef]
- Ab Hamid, N.; Mohd Noor, S.N.; Isa, N.R.; Md Rodzay, R.; Bachtiar Effendi, A.M.; Hafisool, A.A.; Azman, F.A.; Abdullah, S.F.; Kamarul Zaman, M.K.; Mohd Norsham, M.I.; et al. Vertical Infestation Profile of Aedes in Selected Urban High-Rise Residences in Malaysia. Trop. Med. Infect. Dis. 2020, 5, 114. [Google Scholar] [CrossRef] [PubMed]
- Lins, T.M.P. Análise de Locais com Potencial Risco de Transmissão de Arboviroses Usando Técnicas de Sensoriamento Remoto e Geoprocessamento. Master’s Thesis, Universidade Federal de Pernambuco, Recife, Brazil, 2019. [Google Scholar]
- Xavier, D.R.; Magalhães, M.d.A.F.M.; Gracie, R.; dos Reis, I.C.; de Matos, V.P.; Barcellos, C. Difusão espaço-tempo do dengue no Município do Rio de Janeiro, Brasil, no período de 2000–2013. Cadernos de Saúde Pública 2017, 33. Available online: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0102-311X2017000205006&lng=pt&tlng=pt (accessed on 16 December 2017).
- IRI IRI for C and, S. IRI/LDEO Climate Data Library. 2017. Available online: http://iridl.ldeo.columbia.edu/ (accessed on 19 June 2018).
- Sistema Alerta Rio da Prefeitura do Rio de Janeiro. 2017. Available online: http://www.sistema-alerta-rio.com.br/dados-meteorologicos/download/dados-pluviometricos/ (accessed on 18 March 2018).
- Sharmin, S.; Glass, K.; Viennet, E.; Harley, D. Interaction of Mean Temperature and Daily Fluctuation Influences Dengue Incidence in Dhaka, Bangladesh. PLoS Negl. Trop. Dis. 2015, 9, e0003901. [Google Scholar] [CrossRef] [PubMed]
- Tauil, P.L. Urbanização e ecologia do dengue. Cadernos de Saúde Pública 2001, 17, S99–S102. [Google Scholar] [CrossRef]
- Resendes, A.P.d.C.; da Silveira, N.A.P.R.; Sabroza, P.C.; Souza-Santos, R. Determinação de áreas prioritárias para ações de controle da dengue. Revista de Saúde Pública 2010, 44, 274–282. [Google Scholar] [CrossRef]
- Ferreira, D.A.P. Marco Aurélio Pereira Horta. Ph.D. Thesis, National School of Public Health (ENSP-FioCruz), Rio de Janeiro, Brazil, 2013. [Google Scholar]
- Jolliffe, I. Principal Component Analysis, 2nd ed.; Springer International Publishing: Berlin/Heidelberg, Germany, 2002; 518p, Available online: http://cda.psych.uiuc.edu/statistical_learning_course/Jolliffe%20I.%20Principal%20Component%20Analysis%20(2ed.,%20Springer,%202002)(518s)_MVsa_.pdf (accessed on 12 June 2018).
- Jiang, H.; Eastman, J.R. Application of fuzzy measures in multi-criteria evaluation in GIS. Int. J. Geogr. Inf. Sci. 2000, 14, 173–184. [Google Scholar] [CrossRef]
- Spearman, C. The Proof and Measurement of Association between Two Things. Am. J. Psychol. 1904, 15, 72. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: https://www.R-project.org/ (accessed on 8 March 2018).
- ESRI. ArcGis 10.5. 2013. Available online: http://www.esri.com/software/arcgis/index.html (accessed on 10 February 2018).
- Alexandrino, F. Ilhas de Calor e Casos de Dengue na Área Urbana de Taubaté-sp. Master’s Thesis, Paulista State University-UNESP, Guaratinguetá, Brazil, 2017. [Google Scholar]
- Perkins, T.A.; Paz-Soldan, V.A.; Stoddard, S.T.; Morrison, A.C.; Forshey, B.M.; Long, K.C.; Halsey, E.S.; Kochel, T.J.; Elder, J.P.; Kitron, U.; et al. Calling in sick: Impacts of fever on intra-urban human mobility. Proc. R. Soc. B Biol. Sci. 2016, 283, 20160390. [Google Scholar] [CrossRef]
- Castro, R. Habitação, Saneamento Básico e a Proliferação de Dengue, zika e Chikungunya nas Favelas. Rede Dengue, Zika e Chikungunya da Fundação Oswaldo Cruz (FIOCRUZ). 2016. Available online: https://rededengue.fiocruz.br/noticias/524-habitacao-saneamento-basico-e-a-proliferacao-de-dengue-zika-e-chikungunya-nas-favelas (accessed on 19 July 2020).
- Fialho, E.S. Ilha de calor: Reflexões acerca de um conceito. Revista Acta Geográfica 2012, 6, 61–76. [Google Scholar] [CrossRef]
- Filho, O.C.R. A Evolução Da Ilha De Calor Na Região Metropolitana Do Rio De Janeiro. Rev. Geonorte 2012, 3, 8–21. [Google Scholar]
- SMS SM de S do R de J. Levantamento de Índice Rápido para Aedes aegypti (LIRAa). 2016. Available online: http://www.rio.rj.gov.br/web/sms/exibeconteudo?id=2815394 (accessed on 19 March 2018).
- Pereira Horta, M.A.; Pacheco Ferreira, A.; Bruniera de Oliveira, R.; Dias Wermelinger, E.; de Oliveira Ker, F.T.; Navarro Ferreira, A.C.; Catita, C.M.S. Os efeitos do crescimento urbano sobre a dengue. Revista Brasileira em Promoção da Saúde 2013, 26, 539–547. [Google Scholar] [CrossRef]
- Fernandes, N.d.N. O conceito carioca de subúrbio: Um rapto ideológico. Rev. da FAU UFRJ 2010, 2, 8–15. [Google Scholar]
- Abreu, M.d.A. Evolução Urbana no Rio de Janeiro; IPLANRIO: Rio de Janeiro, Brazil, 1987. [Google Scholar]
- de Freitas Lenzi, M.; Coura, L.C. Prevenção da dengue: A informação em foco. Rev. da Soc. Bras. de Med. Trop. 2004, 37, 343–350. [Google Scholar] [CrossRef] [PubMed]
- da Souza Pereira, B.; Porto, G.M.; Soares, H.C. O papel da vigilância epidemiológica no combate a dengue. C&D Rev. El. da Fainor Vitória da Conquista 2011, 4, 87–101. [Google Scholar]
- Nogueira, R.M.; Eppinghaus, A.L. Dengue virus type 4 arrives in the state of Rio de Janeiro: A challenge for epidemiological surveillance and control. Memórias do Instituto Oswaldo Cruz 2011, 106, 255–256. [Google Scholar] [CrossRef]
- dos Santos, J.P.C.; Honório, N.A.; Nobre, A.A. Definition of persistent areas with increased dengue risk by detecting clusters in populations with differing mobility and immunity in Rio de Janeiro, Brazil. Cadernos de Saúde Pública. 2019, 35. Available online: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0102-311X2019001405006&tlng=en (accessed on 23 December 2019).
- Sabroza, P.C. Estudos epidemiológicos na perspectiva do aumento da vulnerabilidade dos sistemas sócio-ambientais brasileiros. Epidemiologia e Serviços de Saúde 2007, 16, 229–232. [Google Scholar] [CrossRef]
- Haesbaert, R. O Mito da Desterritorialização: Do “Fim dos Territórios” à Multiterritorialidade; Bertrand Brasil: Rio de Janeiro, Brazil, 2009. [Google Scholar]
- Mattos Almeida, M.C.; Caiaffa, W.T.; Assunção, R.M.; Proietti, F.A. Spatial Vulnerability to Dengue in a Brazilian Urban Area during a 7-Year Surveillance. J. Urban Health 2007, 84, 334–345. [Google Scholar] [CrossRef]
- Vanlerberghe, V.; Gómez-Dantés, H.; Vazquez-Prokopec, G.; Alexander, N.; Manrique-Saide, P.; Coelho, G.; Toledo, M.E.; Ocampo, C.B.; Van der Stuyft, P. Changing paradigms in Aedes control: Considering the spatial heterogeneity of dengue transmission. Rev. Pan Am. J. Public Health 2017, 41, 1–6. [Google Scholar]
| Code | Indicator | Construction | Unit | Min–Max | Mean (SD) | Period | Source |
|---|---|---|---|---|---|---|---|
| Ind 01 | Percentage of occupied area | Occupied Area/Total Area | % | 3.38–100 | 76.013 (24.524) | 2014 | Municipality |
| Ind 02 | Percentage of residential area | Residential Area/Total Area | % | 0.74–100 | 64.878 (24.117) | 2014 | Municipality |
| Ind 03 | Net demographic density | Total population/Residential area | inhab/km2 | 1372.74–81,171.23 | 16,778.461 (10,518.778) | 2014 | Municipality |
| Ind 04 | Percentage of area of substandard clusters | Substandard Cluster Area/Residential Area | % | 0–98.38 | 11.187 (14.7) | 2014 | Municipality |
| Ind 05 | Percentage of are with strategic points | Strategic Points Area/Occupied Area | % | 0–14.81 | 1.154 (2.547) | 2014 | Municipality |
| Ind 06 | Mean verticalization | Total building heights/Total buildings | m | 2.45–36.69 | 6.37 (4.778) | 2014 | Municipality |
| Ind 07 | Percentage of vegetation | Vegetation Area/Total Area | % | 0–94.34 | 25.108 (24.029) | 2014 | Municipality |
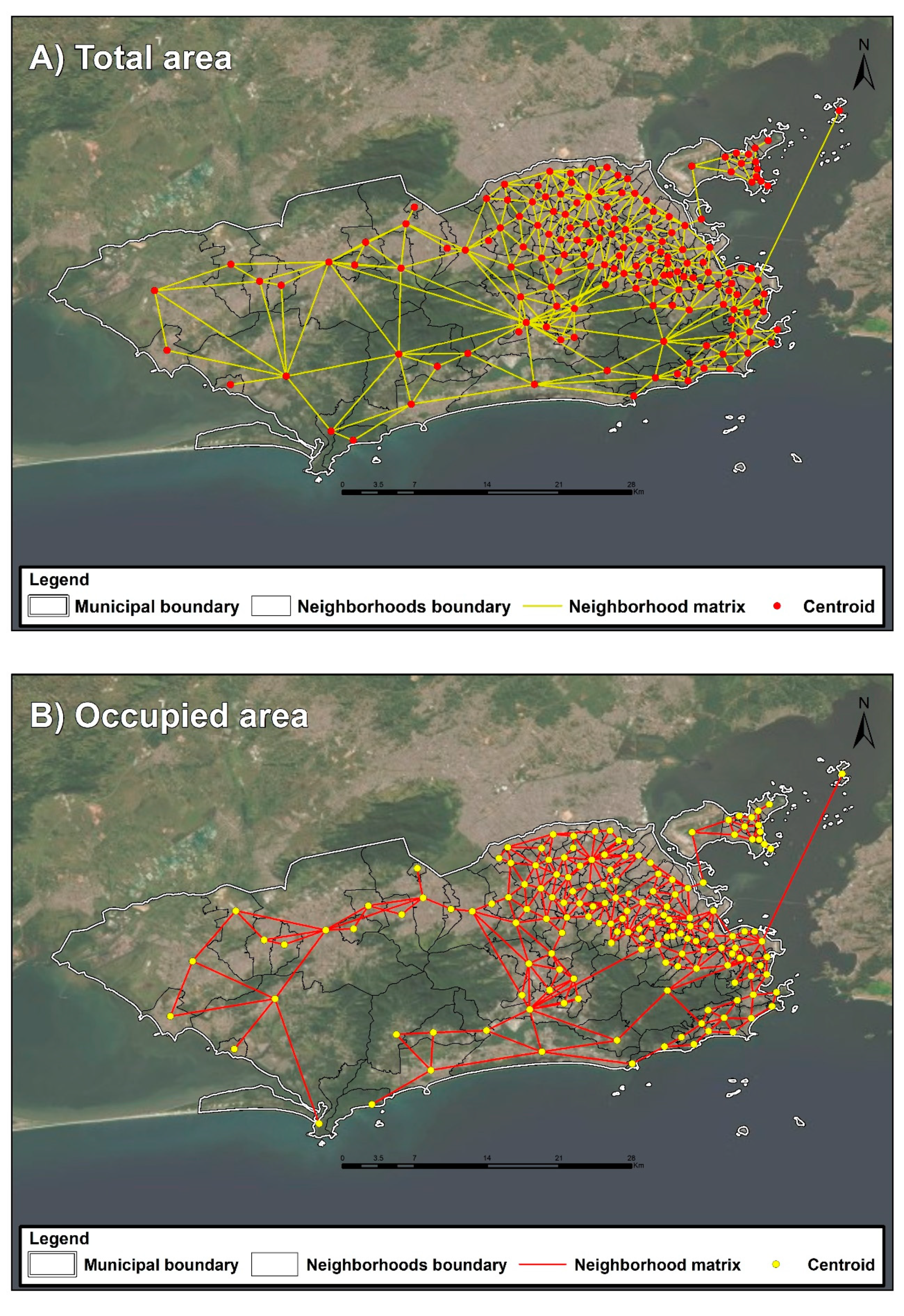
| Ind 08 | Number of neighboring districts | Number of neighbors through neighborhood matrix | districts | 1–13 | 4.375 (1.955) | 2014 | Own |
| Ind 09 | Border perimeter with neighboring districts | Total length of boundary line | m | 12.33–32048.3 | 7095.125 (4658.223) | 2014 | Own |
| Ind 10 | Mean daytime surface temperature | Mean daytime temperature in the occupied area | °C | 23.69–33.48 | 30.22 (2.623) | 2008–2014 | MODIS/IRI |
| Ind 11 | Mean nighttime surface temperature | Mean nighttime temperature in the occupied area | °C | 19.24–22.13 | 21.244 (0.733) | 2008–2014 | MODIS/IRI |
| Ind 12 | Monthly cumulative rainfall | Mean cumulative rainfall in occupied area | mm3 | 85.13–153.31 | 100.058 (10.844) | 2008–2014 | Municipality |
| Ind 13 | Percentage of households with unpaved streets | (v015 + v017 + v019/v001)—Spreadsheet surrounding01 | % | 0–63.63 | 4.831 (10.452) | 2010 | 2010 Census |
| Ind 14 | Percentage of households with no tree-lined streets | (v045 + v047 + v049/v001)—Spreadsheet surrounding01 | % | 0–77.28 | 19.869 (17.862) | 2010 | 2010 Census |
| Ind 15 | Percentage of households with streets without manholes | (v033 + v035 + v037/v001)—Spreadsheet surrounding01 | % | 0–91.96 | 12.342 (15.126) | 2010 | 2010 Census |
| Ind 16 | Percentage of households with streets with exposed trash | (v056 + v058 + v060/v001)—Spreadsheet surrounding01 | % | 0–40.34 | 4.038 (5.584) | 2010 | 2010 Census |
| Ind 17 | Percentage of households with streets with open sewage | (v050 + v052 + v054/v001)—Spreadsheet surrounding01 | % | 0–31.44 | 4.123 (5.534) | 2010 | 2010 Census 2010 |
| Indicators | Factor Loadings | |||
|---|---|---|---|---|
| Comp 1 | Comp 2 | Comp 3 | Comp 4 | |
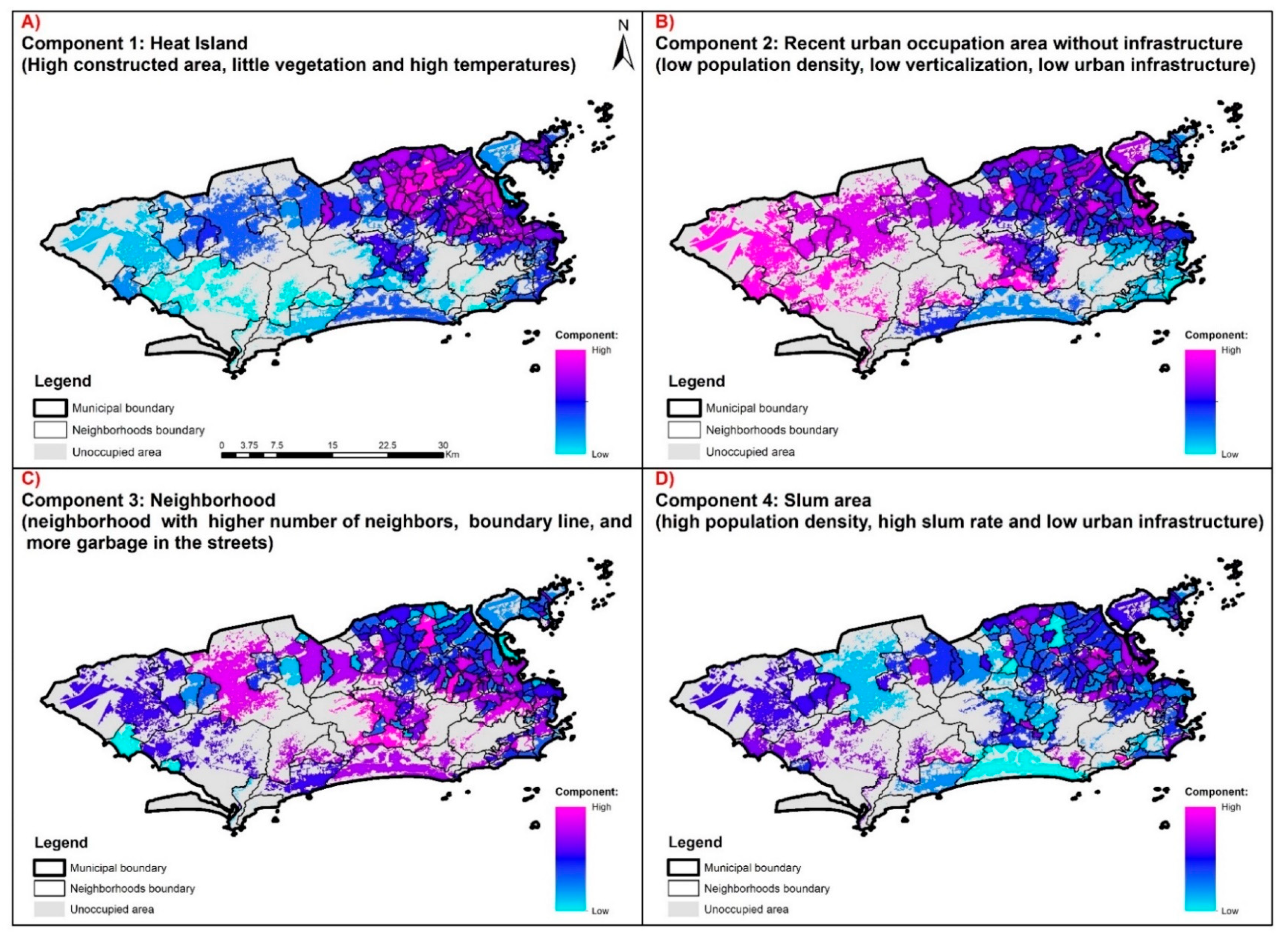
| Ind 01 | 0.91 * | 0.03 | −0.25 | 0.05 |
| Ind 02 | 0.86 * | 0.05 | −0.18 | 0.06 |
| Ind 03 | 0.26 | −0.67 * | −0.03 | 0.52 * |
| Ind 04 | 0.05 | -0.28 | 0.03 | 0.78 * |
| Ind 05 | 0.14 | 0.1 | 0.35 | 0.02 |
| Ind 06 | 0 | −0.67 * | 0 | −0.01 |
| Ind 07 | −0.88 * | 0.02 | 0.25 | −0.03 |
| Ind 08 | 0.45 | 0.14 | 0.64 * | −0.16 |
| Ind 09 | 0.28 | 0.41 | 0.50 * | −0.14 |
| Ind 10 | 0.78 * | 0.44 | 0.06 | 0.08 |
| Ind 11 | 0.87 * | 0.19 | −0.03 | 0.05 |
| Ind 12 | −0.52 | −0.52 | 0.31 | 0.03 |
| Ind 13 | −0.58 | 0.48 | −0.34 | 0.09 |
| Ind 14 | −0.08 | 0.59 * | −0.22 | 0.25 |
| Ind 15 | −0.57 | 0.58 * | −0.33 | 0.1 |
| Ind 16 | −0.09 | 0.36 | 0.44 * | 0.43 * |
| Ind 17 | −0.29 | 0.52 | 0.28 | 0.39 * |
| Eigenvalue | 5.11 | 2.99 | 1.6 | 1.36 |
| % of variance | 30.06 | 17.6 | 9.44 | 7.99 |
| Incidence | EDI | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Period | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 | 2008–2018 | 2013–2014 |
| Serotype | Denv 2 | Denv 2 | Denv 2 | Denv 1 | Denv 4 | Denv 4 | Denv 4 | Denv 1 | Denv 1 | Denv 2 | Denv 2 | ||
| Component 1 | 0.11 | 0.02 | −0.15 * | −0.19 * | 0.31 * | −0.13 | 0.03 | 0.27 * | 0.20 * | 0.12 | 0.12 | 0.12 | 0.07 |
| Component 2 | 0.29 * | 0.08 | −0.21 * | 0.24 * | 0.41 * | −0.16 * | 0.08 | 0.17 * | 0.46 * | 0.22 * | 0.36 * | 0.37 * | 0.28 * |
| Component 3 | 0.04 | −0.01 | −0.01 | −0.02 | −0.02 | −0.04 | 0.02 | 0.03 | 0.01 | −0.16 * | −0.01 | −0.05 | −0.05 |
| Component 4 | 0.14 * | 0.02 | −0.09 | 0.00 | 0.01 | −0.12 | −0.05 | 0.06 | 0.19 * | 0.17 * | 0.19 * | 0.09 | −0.06 |
| Receptivity | 0.28 * | 0.08 | −0.24 * | 0.14 | 0.44 * | −0.22 * | 0.05 | 0.23 * | 0.47 * | 0.30 * | 0.39 * | 0.35 | 0.21 * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santos, J.P.C.; Honório, N.A.; Barcellos, C.; Nobre, A.A. A Perspective on Inhabited Urban Space: Land Use and Occupation, Heat Islands, and Precarious Urbanization as Determinants of Territorial Receptivity to Dengue in the City of Rio De Janeiro. Int. J. Environ. Res. Public Health 2020, 17, 6537. https://doi.org/10.3390/ijerph17186537
Santos JPC, Honório NA, Barcellos C, Nobre AA. A Perspective on Inhabited Urban Space: Land Use and Occupation, Heat Islands, and Precarious Urbanization as Determinants of Territorial Receptivity to Dengue in the City of Rio De Janeiro. International Journal of Environmental Research and Public Health. 2020; 17(18):6537. https://doi.org/10.3390/ijerph17186537
Chicago/Turabian StyleSantos, Jefferson Pereira Caldas, Nildimar Alves Honório, Christovam Barcellos, and Aline Araújo Nobre. 2020. "A Perspective on Inhabited Urban Space: Land Use and Occupation, Heat Islands, and Precarious Urbanization as Determinants of Territorial Receptivity to Dengue in the City of Rio De Janeiro" International Journal of Environmental Research and Public Health 17, no. 18: 6537. https://doi.org/10.3390/ijerph17186537
APA StyleSantos, J. P. C., Honório, N. A., Barcellos, C., & Nobre, A. A. (2020). A Perspective on Inhabited Urban Space: Land Use and Occupation, Heat Islands, and Precarious Urbanization as Determinants of Territorial Receptivity to Dengue in the City of Rio De Janeiro. International Journal of Environmental Research and Public Health, 17(18), 6537. https://doi.org/10.3390/ijerph17186537





