Resistance Training in Breast Cancer Survivors: A Systematic Review of Exercise Programs
Abstract
1. Introduction
2. Methods
2.1. Search Strategy
2.2. Study Selection
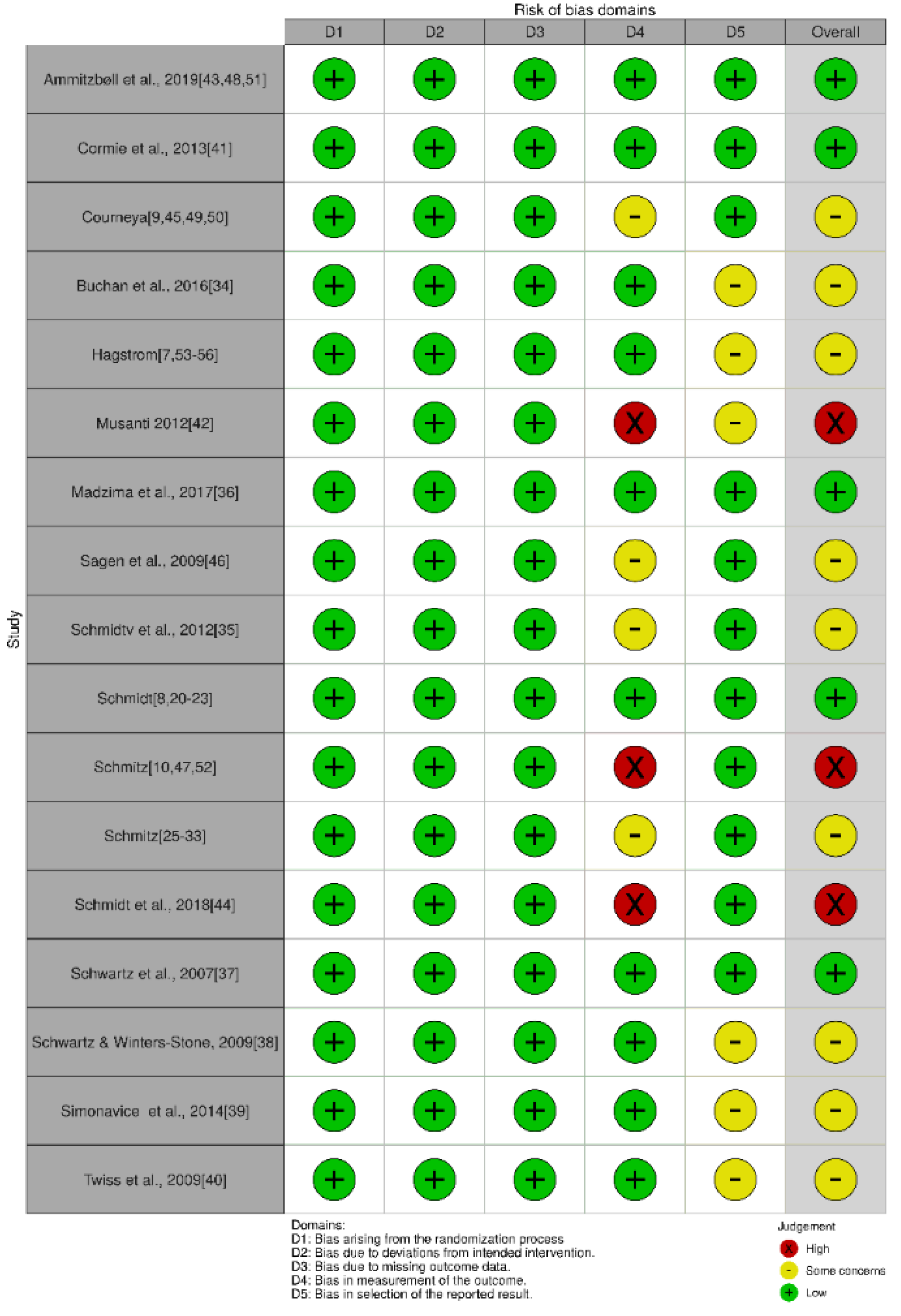
2.3. Data Extraction and Quality Assessment
3. Results
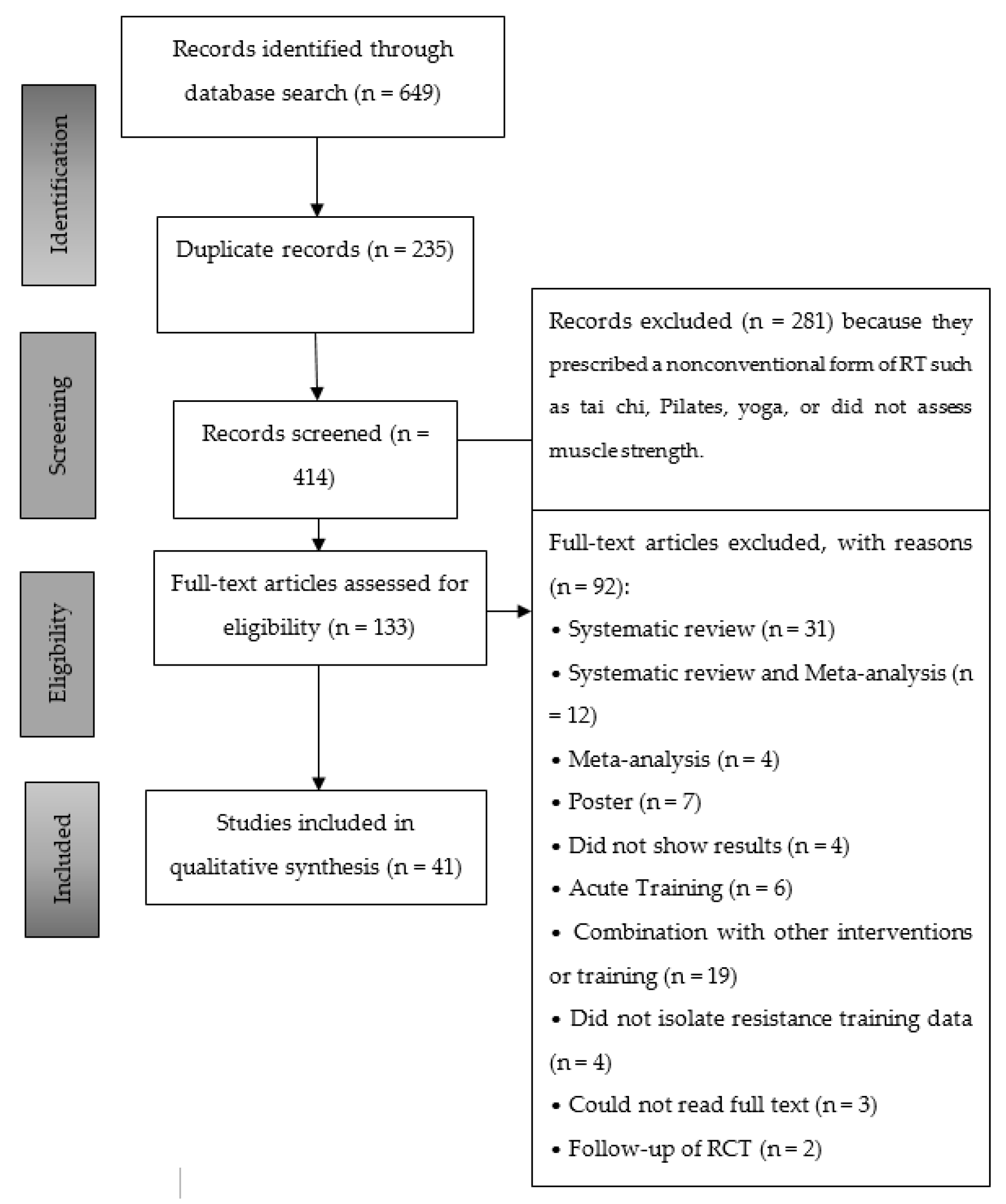
3.1. Study Selection
3.2. Characteristics of the Selected Studies
3.3. Manifestations of Muscle Strength and Evaluation Methods
3.4. Exercise Prescription
3.5. Results and Safety of Resistance Training
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Warburton, D.E.R.; Bredin, S.S.D. Health Benefits of Physical Activity: A Strengths-Based Approach. J. Clin. Med. 2019, 8, 2044. [Google Scholar] [CrossRef]
- Battaglini, C.L.; Mills, R.C.; Phillips, B.L.; Lee, J.T. Twenty-five years of research on the effects of exercise training in breast cancer survivors: A systematic review of the literature. World J. Clin. Oncol. 2014, 5, 177–190. [Google Scholar] [CrossRef]
- Kim, J.; Choi, W.J.; Jeong, S.H. The effects of physical activity on breast cancer survivors after diagnosis. J. Cancer Prev. 2013, 18, 193–200. [Google Scholar] [CrossRef]
- Baumann, F.T.; Bloch, W.; Weissen, A.; Brockhaus, M.; Beulertz, J.; Zimmer, P.; Streckmann, F.; Zopf, E.M. Physical Activity in Breast Cancer Patients during Medical Treatment and in the Aftercare—A Review. Breast Care 2013, 8, 330–334. [Google Scholar] [CrossRef]
- Mishra, S.I.; Scherer, R.W.; Snyder, C.; Geigle, P.M.; Berlanstein, D.R.; Topaloglu, O. Exercise interventions on health–related quality of life for people with cancer during active treatment. Cochrane Database Syst. Rev. 2012, 8. [Google Scholar] [CrossRef]
- Levangie, P.K.; Drouin, J. Magnitude of late effects of breast cancer treatments on shoulder function: A systematic review. Breast Cancer Res. Treat. 2009, 116, 1–15. [Google Scholar] [CrossRef]
- Hagstrom, A.D.; Marshall, P.W.M.; Lonsdale, C.; Cheema, B.S.; Fiatarone, M.A.; Green, S. Resistance training improves fatigue and quality of life in previously sedentary breast cancer survivors: A randomised controlled trial. Eur. J. Cancer Care. 2015. [Google Scholar] [CrossRef]
- Steindorf, K.; Schmidt, M.E.; Klassen, O.; Ulrich, C.M.; Oelmann, J.; Habermann, N.; Beckhove, P.; Owen, R.; Debus, J.; Wiskemann, J.; et al. Randomized, controlled trial of resistance training in breast cancer patients receiving adjuvant radiotherapy: Results on cancer–related fatigue and quality of life. Ann. Oncol. 2014, 25, 2237–2243. [Google Scholar] [CrossRef]
- Courneya, K.S.; Segal, R.J.; McKenzie, D.C.; Dong, H.; Gelmon, K.; Friedenreich, C.M.; Yasui, Y.; Reid, R.D.; Crawford, J.J.; Mackey, J.R. Effects of exercise during adjuvant chemotherapy on breast cancer outcomes. Med. Sci. Sports Exerc. 2014, 46, 1744–1751. [Google Scholar] [CrossRef]
- Ahmed, R.L.; Thomas, W.; Yee, D.; Schmitz, K.H. Randomized controlled trial of weight training and lymphedema in breast cancer survivors. J. Clin. Oncol. 2006, 24, 2765–2772. [Google Scholar] [CrossRef]
- Cheema, B.; Gaul, C.A.; Lane, K.; Fiatarone Singh, M.A. Progressive resistance training in breast cancer: A systematic review of clinical trials. Breast Cancer Res. Treat. 2008, 109, 9–26. [Google Scholar] [CrossRef]
- Courneya, K.S.; McKenzie, D.C.; Mackey, J.R.; Gelmon, K.; Friedenreich, C.M.; Yasui, Y.; Reid, R.D.; Vallerand, J.R.; Adams, S.C.; Proulx, C.; et al. Subgroup effects in a randomised trial of different types and doses of exercise during breast cancer chemotherapy. Br. J. Cancer. 2014, 111, 1718–1725. [Google Scholar] [CrossRef]
- Keilani, M.; Hasenoehrl, T.; Neubauer, M.; Crevenna, R. Resistance exercise and secondary lymphedema in breast cancer survivors–a systematic review. Supportive Care Cancer 2016, 24, 1907–1916. [Google Scholar] [CrossRef]
- Cheema, B.S.; Kilbreath, S.L.; Fahey, P.P.; Delaney, G.P.; Atlantis, E. Safety and efficacy of progressive resistance training in breast cancer: A systematic review and meta–analysis. Breast Cancer Res. Treat. 2014, 148, 249–268. [Google Scholar] [CrossRef] [PubMed]
- Paramanandam, V.S.; Roberts, D. Weight training is not harmful for women with breast cancer–related lymphoedema: A systematic review. J. Physiother. 2014, 60, 136–143. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta–analyses of studies that evaluate healthcare interventions: Explanation and elaboration. J. Clin. Epidemiol. 2009, 62. [Google Scholar] [CrossRef]
- Maher, C.G.; Sherrington, C.; Herbert, R.D.; Moseley, A.M.; Elkins, M. Reliability of the PEDro scale for rating quality of randomized controlled trials. Phys. Ther. 2003, 83, 713–721. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Savović, J.; Page, M.; Elbers, R.; Blencowe, N.S.; Boutron, L.; Cates, C.J.; Cheng, H.-Y.; Sorbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366. [Google Scholar] [CrossRef]
- McGuinness, L.A.; Higgins, J.P.T. Risk-of-bias VISualization (robvis): An R package and Shiny web app for visualizing risk-of-bias assessments. Res. Synth. Methods 2020. [Google Scholar] [CrossRef]
- Courneya, K.S.; Segal, R.J.; Mackey, J.R.; Gelmon, K.; Reid, R.D. Effects of aerobic and resistance exercise in breast cancer patients receiving adjuvant chemotherapy: A multicenter randomized controlled trial. J. Clin. Oncol. 2007, 25, 4396–4404. [Google Scholar] [CrossRef]
- Courneya, K.S.; Segal, R.J.; Gelmon, K.; Reid, R.D.; Mackey, J.; Friedenreich, C.M.; Proulx, C.; Lane, K.; Ladha, A.B.; Vallance, J.K.; et al. Six-month follow-up of patient-rated outcomes in a randomized controlled trial of exercise training during breast cancer chemotherapy. Cancer Epidemiol. Biomark. Prev. 2007, 16, 2572–2578. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.C.; Segal, R.J.; McKenzie, D.C.; Vallerand, J.R.; Morielli, A.R.; Machey, J.R.; Gelmon, K.; Friedenreich, C.M.; Reid, R.D.; Courneya, K.S. Impact of resistance and aerobic exercise on sarcopenia and dynapenia in breast cancer patients receiving adjuvant chemotherapy: A multicenter randomized controlled trial. Breast Cancer Res. Treat. 2016, 158, 497–507. [Google Scholar] [CrossRef]
- Schwartz, A.L.; Winters-Stone, K.; Gallucci, B. Exercise effects on bone mineral density in women with breast cancer receiving adjuvant chemotherapy. Oncol. Nurs. Forum 2007, 34, 627–633. [Google Scholar] [CrossRef]
- Schwartz, A.L.; Winters-Stone, K. Effects of a 12-month randomized controlled trial of aerobic or resistance exercise during and following cancer treatment in women. Phys. Sportsmed. 2009, 37, 62–67. [Google Scholar] [CrossRef]
- Sagen, A.; Karesen, R.; Risberg, M.A. Physical activity for the affected limb and arm lymphedema after breast cancer surgery. A prospective, randomized controlled trial with two years follow-up. Acta Oncol. 2009, 48, 1102–1110. [Google Scholar] [CrossRef]
- Schmidt, M.E.; Wiskemann, J.; Krakowski-Roosen, H.; Knicker, A.J.; Habermann, N.; Schneeweiss, A.; Ulrich, C.M.; Steindorf, K. Progressive resistance versus relaxation training for breast cancer patients during adjuvant chemotherapy: Design and rationale of a randomized controlled trial (BEATE study). Contemp. Clin. Trials 2013, 34, 117–125. [Google Scholar] [CrossRef]
- Schmidt, M.E.; Wiskemann, J.; Armbrust, P.; Schneeweiss, A.; Ulrich, C.M.; Steindorf, K. Effects of resistance exercise on fatigue and quality of life in breast cancer patients undergoing adjuvant chemotherapy: A randomized controlled trial. Int. J. Cancer 2015, 137, 471–480. [Google Scholar] [CrossRef]
- Potthoff, K.; Schmidt, M.E.; Wiskemann, J.; Hof, H.; Klassen, O.; Habermann, N.; Beckhove, P.; Debus, J.; Ulrich, C.M.; Steindorf, K. Randomized controlled trial to evaluate the effects of progressive resistance training compared to progressive muscle relaxation in breast cancer patients undergoing adjuvant radiotherapy: The BEST study. BMC Cancer 2013, 13, 162. [Google Scholar] [CrossRef]
- Schmidt, M.E.; Meynkohn, A.; Habermann, N.; Wiskemann, J.; Oelmann, J.; Hof, H.; Wessels, S.; Klassen, O.; Debus, J.; Potthoff, K.; et al. Resistance Exercise and Inflammation in Breast Cancer Patients Undergoing Adjuvant Radiation Therapy: Mediation Analysis From a Randomized, Controlled Intervention Trial. Int. J. Radiat. Oncol. Biol. Phys. 2016, 94, 329–337. [Google Scholar] [CrossRef]
- Wiskemann, J.; Schmidt, M.E.; Klassen, O.; Debus, J.; Ulrich, C.M.; Potthoff, K. Effects of 12-week resistance training during radiotherapy in breast cancer patients. Scand. J. Med. Sci. Sports 2017, 27, 1500–1510. [Google Scholar] [CrossRef]
- Schmidt, T.; Jonat, W.; Wesch, D.; Oberg, H.-H.; Adam-Klages, S.; Keller, L.; Röcken, C.; Mundhenke, C. Influence of physical activity on the immune system in breast cancer patients during chemotherapy. J. Cancer Res. Clin. Oncol. 2018, 144, 579–586. [Google Scholar] [CrossRef]
- Ammitzboll, G.; Lanng, C.; Kroman, N.; Zerghn, B.; Hyldegaard, O.; Andersen, G.A.; Johansen, C.; Dalton, S.O. Progressive strength training to prevent LYmphoedema in the first year after breast CAncer—The LYCA feasibility study. Acta Oncol. 2017, 56, 360–366. [Google Scholar] [CrossRef]
- Ammitzboll, G.; Kristina Kjaer, T.; Johansen, C.; Lanng, C.; Andersen, E.W.; Kroman, N.; Zerahn, B.; Hyldegaard, O.; Bidstrup, P.E.; Dalton, S.O. Effect of progressive resistance training on health–related quality of life in the first year after breast cancer surgery—Results from a randomized controlled trial. Acta Oncol. 2019, 58, 665–672. [Google Scholar] [CrossRef]
- Ammitzboll, G.; Johansen, C.; Lanng, C.; Andersen, E.W.; Kroman, N.; Zerhn, B.; Hyldegarrd, O.; Wittenkamp, M.C. Progressive resistance training to prevent arm lymphedema in the first year after breast cancer surgery: Results of a randomized controlled trial. Cancer 2019, 125, 1683–1692. [Google Scholar] [CrossRef]
- Schmitz, K.H.; Ahmed, R.L.; Hannan, P.J.; Yee, D. Safety and efficacy of weight training in recent breast cancer survivors to alter body composition, insulin, and insulin-like growth factor axis proteins. Cancer Epidemiol. Biomark. Prev. 2005, 14, 1672–1680. [Google Scholar] [CrossRef]
- Ohira, T.; Schmitz, K.H.; Ahmed, R.L.; Yee, D. Effects of weight training on quality of life in recent breast cancer survivors: The Weight Training for Breast Cancer Survivors (WTBS) study. Cancer 2006, 106, 2076–2083. [Google Scholar] [CrossRef]
- Twiss, J.J.; Waltman, N.L.; Berg, K.; Ott, C.d.; Gross, G.J.; Lindsey, A.M. An exercise intervention for breast cancer survivors with bone loss. J. Nurs. Scholarsh. 2009, 41, 20–27. [Google Scholar] [CrossRef]
- Musanti, R. A study of exercise modality and physical self-esteem in breast cancer survivors. Med. Sci. Sports Exerc. 2012, 44, 352–361. [Google Scholar] [CrossRef]
- Schmidt, T.; Weisser, B.; Jonat, W.; Baumann, F.T.; Mundhenke, H. Gentle strength training in rehabilitation of breast cancer patients compared to conventional therapy. Anticancer Res. 2012, 32, 3229–3233. [Google Scholar]
- Simonavice, E.; Liu, P.Y.; Ilich, J.Z.; Kim, J.-S.; Arjmandi, B.; Panton, L.B. The effects of a 6-month resistance training and dried plum consumption intervention on strength; body composition; blood markers of bone turnover; and inflammation in breast cancer survivors. Appl. Physiol. Nutr. Metab. 2014, 39, 730–739. [Google Scholar] [CrossRef]
- Hagstrom, A.D.; Marshall, P.W.; Lonsdale, C.; Papalia, S.; Cheema, B.; Toben, C.; Baune, B.T.; Fiatarone Singh, M.A.; Green, S. The effect of resistance training on markers of immune function and inflammation in previously sedentary women recovering from breast cancer: A randomized controlled trial. Breast Cancer Res. Treat. 2016, 155, 471–482. [Google Scholar] [CrossRef] [PubMed]
- Hagstrom, A.D.; Shorter, K.A.; Marshall, P.W.M. Changes in Unilateral Upper Limb Muscular Strength and Electromyographic Activity After a 16-Week Strength Training Intervention in Survivors of Breast Cancer. J. Strength Cond. Res. 2019, 33, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Hagstrom, A.; Denham, J. The Effect of Resistance Training on Telomere Length in Women Recovering from Breast Cancer. J. Funct. Morphol. Kinesiol. 2018, 3, 9. [Google Scholar] [CrossRef]
- Hagstrom, A.D.; Denham, J. microRNAs in High and Low Responders to Resistance Training in Breast Cancer Survivors. Int. J. Sports Med. 2018, 39, 482–489. [Google Scholar] [CrossRef]
- Madzima, T.A.; Ormsbee, M.J.; Schleicher, E.A.; Moffatt, R.J.; Panton, L.B. Effects of Resistance Training and Protein Supplementation in Breast Cancer Survivors. Med. Sci. Sports Exerc. 2017, 49, 1283–1292. [Google Scholar] [CrossRef]
- Schmitz, K.H.; Troxel, A.B.; Cheville, A.; Grant, L.L.; Bryan, C.J.; Gross, C.R.; Lytle, L.A.; Ahmed, R.L. Physical Activity and Lymphedema (the PAL trial): Assessing the safety of progressive strength training in breast cancer survivors. Contemp. Clin. Trials 2009, 30, 233–245. [Google Scholar] [CrossRef]
- Schmitz, K.H.; Ahmed, R.L.; Troxel, A.; Checile, A. Weight lifting in women with breast-cancer-related lymphedema. N. Engl. J. Med 2009, 361, 664–773. [Google Scholar] [CrossRef]
- Speck, R.M.; Gross, C.R.; Hormes, J.M.; Ahmed, R.L.; Lytle, L.A.; Hwang, W.-T.; Schmitz, K.H. Changes in the Body Image and Relationship Scale following a one-year strength training trial for breast cancer survivors with or at risk for lymphedema. Breast Cancer Res. Treat. 2010, 121, 421–430. [Google Scholar] [CrossRef]
- Schmitz, K.H.; Ahmed, R.L.; Troxel, A.B.; Cheville, A.; Lewis-Grant, L.; Smith, R.; Bryan, C.J.; Williams-Smith, C.T.; Chittams, J. Weight lifting for women at risk for breast cancer-related lymphedema: A randomized trial. JAMA 2010, 304, 2699–2705. [Google Scholar] [CrossRef]
- Hayes, S.C.; Speck, R.M.; Reimet, E.; Stark, A.; Schmitz, K.H. Does the effect of weight lifting on lymphedema following breast cancer differ by diagnostic method: Results from a randomized controlled trial. Breast Cancer Res. Treat. 2011, 130, 227–234. [Google Scholar] [CrossRef]
- Brown, J.C.; Troxel, A.B.; Schmitz, K.H. Safety of weightlifting among women with or at risk for breast cancer-related lymphedema: Musculoskeletal injuries and health care use in a weightlifting rehabilitation trial. Oncologist 2012, 17, 1120–1128. [Google Scholar] [CrossRef]
- Winters-Stone, K.M.; Laudermilk, M.; Woo, K.; Brown, J.C.; Schmitz, K.H. Influence of weight training on skeletal health of breast cancer survivors with or at risk for breast cancer-related lymphedema. J. Cancer Surviv. 2014, 8, 260–268. [Google Scholar] [CrossRef]
- Brown, J.C.; Schmitz, K.H. Weight lifting and appendicular skeletal muscle mass among breast cancer survivors: A randomized controlled trial. Breast Cancer Res. Treat. 2015, 151, 385–392. [Google Scholar] [CrossRef]
- Brown, J.C.; Schmitz, K.H. Weight Lifting and Physical Function Among Survivors of Breast Cancer: A Post Hoc Analysis of a Randomized Controlled Trial. J. Clin. Oncol. 2015, 33, 2184–2189. [Google Scholar] [CrossRef]
- Buchan, J.; Janda, M.; Box, R.; Schmitz, K.; Hayes, S. A Randomized Trial on the Effect of Exercise Mode on Breast Cancer-Related Lymphedema. Med. Sci. Sports Exerc. 2016, 48, 1866–1874. [Google Scholar] [CrossRef]
- Cormie, P.; Pumpa, K.; Galvao, D.A.; Turner, E.; Spry, N.; Saunders, C.; Zissiadis, Y.; Newton, R.U. Is it safe and efficacious for women with lymphedema secondary to breast cancer to lift heavy weights during exercise: A randomised controlled trial. J. Cancer Surviv. 2013, 7, 413–424. [Google Scholar] [CrossRef]
- Hasenoehrl, T.; Keilani, M.; Palma, S.; Crevenna, R. Resistance exercise and breast cancer related lymphedema—A systematic review update. Disabil. Rehabil. 2020, 42, 26–35. [Google Scholar] [CrossRef]
| Terms | Definitions |
|---|---|
| Maximum dynamic force | The maximum load you are able to mobilize once (one repetition maximum, 1RM). |
| Muscle power | The result of the product of the load imposed by the speed at which it moves in a movement characterized by running in a very short period of time, short and high-intensity. |
| Resistance force (or resistance endurance) | The maximum number of times you are able to mobilize a load. |
| Resistance training | The generic name used to refer to chronic exercise aimed at maintaining or improving neuromuscular performance. |
| Search Mode | Advanced |
|---|---|
| Database coverage date | From 1 January 1990 to 6 December 2019 |
| Search term 1 | OR weight train * OR weight lift * OR resistance train * OR resistance exercise * OR progressive resisted exercise * OR weight-bearing exercise * OR strength exercise * OR strength train * OR strengthening program * OR exercise training * OR exercise program * OR physical activity * OR physical exercise * OR rehabilitation * |
| Search term 2 | breast cancer * OR breast neoplasm * OR breast carcinoma * OR breast tumor * OR mammary neoplasm * OR mammary malignant * OR mammary carcinoma * OR mastectomy * OR lymph node excision * OR lymphedema * |
| Search | Search term 1 AND Search term 2 |
| Design | Randomized Trials |
|---|---|
| Population | Breast cancer survivors |
| Intervention | Resistance training |
| Outcomes | Muscle strength evaluation Resistance training characteristics Resistance training results |
| Comparison | Control group (without the disease, survivors of breast cancer who developed other types of training, breast cancer survivors that remained sedentary) Pre and post-training |
| No. | TRIALS | ARTICLES |
|---|---|---|
| 1 | START: Supervised Trial of Aerobic Versus Resistance Training | Courneya et al., 2007 [20]-Courneya et al., 2007 [21] Courneya et al., 2014 [9]-Adams et al., 2016 [22] |
| 2 | Schwartz et al., 2007 [23] | |
| 3 | Schwartz & Winters-Stone, 2009 [24] | |
| 4 | Sagen et al., 2009 [25] | |
| 5 | BEATE: Exercise and relaxation as therapy against fatigue BEST: Exercise and relaxation for breast cancer patients during radiotherapy | Schmidt et al., 2013 [26]-Schmidt et al., 2015 [27] |
| Potthoff et al., 2013 [28]-Steindorf et al., 2014 [8] Schmidt et al., 2016 [29]-Wiskemann et al., 2017 [30] | ||
| 6 | Schmidt et al., 2018 [31] | |
| 7 | Ammitzbøll and colleagues | Ammitzbøll et al., 2019 [32]-Ammitzbøll et al., 2019 [33] Ammitzbøll et al., 2019 [34] |
| 8 | WTBS: Weight Training for Breast Cancer Survivors | Schmitz et al., 2005 [35]-Ohira et al., 2006 [36] Ahmed et al., 2006 [10] |
| 9 | Twiss et al., 2009 [37] | |
| 10 | Musanti, 2012 [38] | |
| 11 | Schmidt et al., 2012 [39] | |
| 12 | Simonavice et al., 2014 [40] | |
| 13 | Hagstrom and colleagues | Hagstrom et al., 2015 [7]-Hagstrom et al., 2016 [41] Hagstrom, A. D., Shorter, K. A., & Marshall, P. W. 2019 [42]-Hagstrom, A., & Denham, J. 2018 [43] Hagstrom, A. D., & Denham, J. 2018 [44] |
| 14 | Madzima et al., 2017 [45] | |
| 15 | PAL: Physical Activity and Lymphoedema | Schmitz et al., 2009 [46]-Schmitz et al., 2009 [47] Speck et al., 2010 [48]-Schmitz et al., 2010 [49] Hayes et al., 2011 [50]-Brown et al., 2012 [51] Winters-Stone et al., 2014 [52] Brown & Schmitz et al., 2015 [53] Brown & Schmitz et al., 2015 [54] |
| Buchan et al., 2016 [55] | ||
| 16 | Cormie et al., 2013 [56] | |
| Study | Random Allocation | Concealed Allocation | Groups Similar at Baseline | Participant Blinding | Therapist Blinding | Assessor Blinding | <15% Dropouts | Intention-to-Treat Analysis | Between-Group Difference Reported | Point Estimate and Variability Reported | Total (0 to 10) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ammitzbøll et al., 2019 [32,33,34] | Y | Y | Y | N | Y | Y | Y | Y | Y | Y | 9 |
| Cormie et al., 2013 [56] | Y | Y | Y | N | Y | N | Y | Y | Y | Y | 8 |
| Courneya [9,20,21,22] | Y | Y | Y | N | N | Y | N | Y | Y | Y | 7 |
| Buchan et al., 2016 [55] | Y | Y | Y | N | N | N | Y | Y | Y | Y | 7 |
| Hagstrom [7,41,42,43,44] | Y | Y | Y | Y | N | Y | Y | Y | Y | Y | 9 |
| Musanti 2012 [38] | Y | Y | Y | N | Y | Y | N | N | Y | Y | 7 |
| Madzima et al., 2017 [45] | Y | N | Y | N | N | N | Y | Y | Y | Y | 6 |
| Sagen et al., 2009 [25] | Y | Y | Y | N | N | Y | N | Y | Y | Y | 7 |
| Schmidtv et al., 2012 [39] | Y | Y | N | N | N | N | Y | N | Y | Y | 5 |
| Schmidt [8,26,27,28,29] | Y | Y | Y | Y | Y | N | Y | Y | Y | Y | 9 |
| Schmitz [10,35,36] | Y | Y | Y | N | N | Y | N | N | Y | Y | 6 |
| Schmitz [46,47,48,49,50,51,52,53,54] | Y | Y | Y | N | N | Y | N | Y | Y | Y | 7 |
| Schmidt et al., 2018 [31] | Y | Y | Y | N | Y | Y | N | N | Y | Y | 7 |
| Schwartz et al., 2007 [23] | Y | N | Y | N | N | N | Y | Y | Y | Y | 6 |
| Schwartz & Winters-Stone, 2009 [24] | Y | N | Y | N | N | N | Y | Y | Y | Y | 6 |
| Simonavice et al., 2014 [40] | Y | N | Y | Y | N | N | Y | Y | Y | Y | 7 |
| Twiss et al., 2009 [37] | Y | N | Y | N | N | N | Y | Y | Y | Y | 6 |
| Trial/Author | N | Age | Stage of Cancer | Treatment | Control Groups |
|---|---|---|---|---|---|
| During treatment | |||||
| START [9,20,21,22] | 242 | 49.2 | I–IIIA | M-C | AT-UC |
| Schwartz et al., 2007 [23] | 66 | 50.1 ± 8.7 | I–III | C-R | AT-UC |
| Schwartz & Winters-Stone, 2009 [24] | 101 | 47 ± 9.4 | I–III | C | AT-UC |
| Sagen et al., 2009 [25] | 204 | 55 ± 10 | I–III | M/AND-C-R-HT | UC |
| BEATE [26,27] | 95 | 52.7 ± 10 | I–IV | M/L-C | PMR |
| BEST [8,28,29,30] | 155 | 55.8 ± 9.1 | 0–III | NC-M/L-R-HT | PMR |
| Buchan et al., 2016 [55] | 40 | 56 | NR | S-C-R-HT | AT |
| Schmidt et al., 2018 [31] | 67 | 54 | NR | S-C | AT-UC |
| Ammitzbøll et al., 2019 [32,33,34] | 158 | 52 | I–III | S/AND-C-HT | UC |
| Post-treatment | |||||
| WTBS [10,35,36] | 79 | 53.3 ± 8.7 | I–III | AND-C-R | UC |
| Twiss et al., 2009 [37] | 110 | 58.7 ± 7.5 | 0–II | S-C-R | UC |
| Musanti, 2012 [38] | 42 | 50.5 | I–IIIB | C-R | AT-CT-F |
| Schmidt et al., 2012 [39] | 33 | 58 ± 8.4 | I–III | M/L-C-R | CGE |
| Simonavice et al., 2014 [40] | 23 | 64 ± 5 | 0–III | S-C-R-HT | RT+DP |
| Hagstrom and colleagues [7,41,42,43,44] | 39 | 51.9 ± 8.8 | I–IIA | S-C-R-HT | UC |
| Cormie et al., 2013 [56] | 62 | 57 ± 10 | 0–III | S/AND-C-R-HT | UC |
| PAL [46,47,48,49,50,51,52,53,54] | 295 | 55.3 ± 8.5 | I–III | S/AND-C.HT | UC |
| Madzima et al., 2017 [45] | 33 | 59 ± 9 | 0–III | S-C-R-HT | RT + PRO |
| Manifestation of Muscle Strength | Evaluation Method | Movements/Muscular Groups | ||
|---|---|---|---|---|
| UB | LB | UB | LB | |
| During Treatment | ||||
| Maximal strength | NR [25] | Chest press [9,20,21,31,55] | Leg extension [9,20,21,23,24,31] | |
| 1RM [23,24] | Seated row [23,24] | Knee extensors [8,26,27,28,29] | ||
| MRT (4-8 RM) [9,20,21,22,32,33,34,55] | Shoulder press [23,24,31] | Knee flexors [8,26,27,28,29] | ||
| h1RM - Brzycki-Method [8,26,27,28,29,30,31] | Shoulder rotators [8,26,27,28,29] | Leg curl [31] | ||
| Maximal grip strength [32,33,34] | - | Upper arm curl and extensors [31] | Squat [31] | |
| Isometric strength protocol [32,33,34] | - | Latissimus pull down - Sit-ups [31] | Rowing [31] | |
| Shoulder abductors, adductors, | Leg press [32,33,34] | |||
| flexors, and extensors [32,33,34] | ||||
| Elbow flexion and extension [32,33,34] | ||||
| Post-treatment | ||||
| Maximal strength | 1RM [10,35,36,40,45,46,47,48,49,50,51,52,53,54,56] | 1RM [7,10,35,36,40,41,45,46,47,48,49,50,51,52,53,54,56] | Chest press [7,10,35,36,38,39,40,41,45,46,47,48,49,50,51,52,53,54,56] | Leg extension [10,35,36,38,39,40,45] |
| Maximal grip strength [56] | ||||
| MRT (6 RM) [38] | Seated row [38,56] | Leg press [7,41,46,47,48,49,50,51,52,53,54,56] | ||
| BVSE [37] | Shoulder press [39] | Leg curl [39] | ||
| h1RM [39] | Latissimus pull down [39] | Squat [39] | ||
| Unilateral isometric strength protocol [7,41] | - | Sit ups [38,39] | Rowing [39] | |
| Strength-Endurance | Curl-up test [38] | - | Upper arm curl and extensors [39] | Hip [37] |
| YMCA Bench Press Endurance Test [38] | - | Wrist [37] | Knee extensors [37] | |
| Repetition maximum test [56] | - | Handgrip Strength [56] | Knee flexor [37] | |
| Trial Duration (Wk.) | Exercise Intensity | Increment | Sets | Repetitions | Sessions/Wk. | Session Duration/Min |
|---|---|---|---|---|---|---|
| 12 [7,8,26,27,28,29,31,38,41,45,55,56] | NR [10,23,24,35,36,37] | NR [7,8,24,26,27,28,29,41] | NR [25] | 8–10 [10,23,35,36] | 1 [39] | NR [9,20,21,23,38,39,40,45] |
| 17 [9,20,21] | <50% 1RM [32,33,34] UB: 1-pound weights [46,47,48,49,50,51,52,53,54] 0.5 kg. [25] | 10% → >12 reps/set [9,20,21,40] 5%–10% → 2 Sess. [56] | 1 [10,31,35,36,38,56] | 8–12 [7,8,9,20,21,26,27,28,29,37,39,40,41] | 2 [8,10,26,27,28,29,31,32,33,34,35,36,37,40,45,46,47,48,49,50,51,52,53,54,55,56] | 20–30 [24] |
| 20–30 [10,23,25,32,33,34,35,36,39,40] | 50%–80% 1RM [7,8,9,20,21,26,27,28,29,31,39,40,41,45,56] | Modifying starting grip position [23] Decreasing 5RM x module [32,33,34] | 2 [9,10,20,21,23,24,35,36,37,39,40,46,47,48,49,50,51,52,53,54,55,56] | 10–12 [10,35,36,38,45,46,47,48,49,50,51,52,53,54,55] | 3 [7,9,20,21,38,41] | >30–45 [37] |
| 48 [10,24,35,36,46,47,48,49,50,51,52,53,54] | >85% 1RM [10,35,36,46,47,48,49,50,51,52,53,54,55] | Tolerance [10,25,35,36,37,39] RPE = ≤3 [31,38] | 3 [7,8,10,24,26,27,28,29,32,33,34,35,36,41,45,46,47,48,49,50,51,52,53,54,55,56] | 11–18/20 [24,25,31,32,33,34] | 4 [23,24] | 50–60 [8,10,26,27,28,29,35,36] |
| 96 [37] | RPE of 3–5 [38] | UB = 1/2 pound → 2 Sess. [46,47,48,49,50,51,52,53,54,55] | 4 [56] | 20–15 RM 10-6 RM [56] | >60–90 [46,47,48,49,50,51,52,53,54] | |
| - | - | LB = Smallest possible increment [46,47,48,49,50,51,52,53,54,55] 1.81 kg→ >10 reps/third set [45] | - | - | - | - |
| Supervised Training | Resistance | Movements/Muscular Groups | |
|---|---|---|---|
| UB | LB | ||
| YES [7,8,9,20,21,25,26,27,28,29,31,39,40,41,45,56] | NR [9,20,21,22,25,56] | Chest press [7,9,10,20,21,31,35,36,38,39,40,41,45,46,47,48,49,50,51,52,53,54,55,56] | Leg extension [7,8,9,20,21,23,26,27,28,29,31,37,40,41,45,46,47,48,49,50,51,52,53,54,55,56] |
| NO [23,24,38] | Bands [23,24,38] | Seated row [7,8,9,20,21,24,26,27,28,29,38,39,40,41,45,46,47,48,49,50,51,52,53,54,55,56] | Leg press [7,8,9,20,21,24,26,27,28,29,37,38,39,40,41,45,46,47,48,49,50,51,52,53,54,55,56] |
| PP [10,32,33,34,35,36,37,46,47,48,49,50,51,52,53,54,55] | BW–Dumbbells [7,10,24,32,33,34,35,36,37,41,46,47,48,49,50,51,52,53,54,55] | Shoulder press [23,24,31,38,39,45,56] | Leg curl [7,8,9,20,21,26,27,28,29,31,37,40,41,45,46,47,48,49,50,51,52,53,54,55] |
| - | Machines [7,8,10,26,27,28,29,30,31,35,36,37,39,40,41,45,46,47,48,49,50,51,52,53,54,55] | Lateral, front, and up raise shoulder [46,47,48,49,50,51,52,53,54,55,56] | Squat [31,38,39,56] |
| - | - | Latissimus pull down [7,8,26,27,28,29,31,37,38,39,41,56] | Calf raises [9,20,21,37] |
| - | - | Triceps extension [9,20,21,31,37,38,39,40,56] | Lunge [37,56] |
| - | - | Triceps pushdown [40,45,46,47,48,49,50,51,52,53,54] | Hip flexion [37,38,39] |
| - | - | Bicep curl [9,20,21,31,37,38,39,40,45,46,47,48,49,50,51,52,53,54,55,56] | Hip extension [37,38,39] |
| - | - | Wrist curl [37,56] | Lower back hyperextension [7,40,41,45] |
| - | - | One-arm row–barbell bent [7,41,46,47,48,49,50,51,52,53,54,55] | Buttocks, thighs, and legs [10,35,36] |
| - | - | Butterfly and butterfly reverse [8,26,27,28,29] | Rowing [31] |
| - | - | Upward row–Push-ups/push-ups on knees–Side hip raise [37] | NR [25,32,33,34] |
| - | - | Sit-ups [7,31,37,38,39,40,41,45] | - |
| - | - | Prone hold [7,41] Seated [23] | - |
| - | - | Ball-gripping–Wrist extension [37] | - |
| - | - | Shoulder flexion; shoulder extension [38] | - |
| - | - | Back extension [7,37,41,46,47,48,49,50,51,52,53,54,55] back [10,35,36] | - |
| - | - | Shoulder rotators [8,26,27,28,29] shoulders [10,35,36] | - |
| - | - | NR [25,32,33,34] | - |
| - | - | - | - |
| Result | Training Safety |
|---|---|
| During Treatment | |
| ↑Muscle strength [8,9,20,21,23,24,26,27,28,29,30,55] | No adverse events [8,9,20,21,22,24,25,26,27,28,29,30,32,33,34,55] |
| ↑ Aerobic capacity [24,55] | NR [23,31] |
| ↔ Aerobic capacity [8,9,20,21,26,27,28,29] | - |
| (↑) Aerobic capacity [23] | - |
| ↓ Fatigue [8,9,20,21,26,27,28,29] | - |
| ↔ Bodyweight [24] | - |
| ↔ Body composition [24] | - |
| Attenuates the decrease in BMD [23] | - |
| ↑ LBM [9,20,21] | - |
| ↔ Upper Limbs Volume [9,20,21,25,32,33,34,55] | - |
| ↓ Sarcopenia and Dynapenia [22] | - |
| ↑ QoL [8,9,20,21,22,26,27,28,29,33,55] | - |
| ↑ Self-perceptions [9,20,21] | - |
| ↓ Anxiety [9,20,21] | - |
| ↔ Depression [8,26,27,28,29] | - |
| ↓ Pain [8,25,26,27,28,29] | - |
| Stronger effects on DFS, OS, DDFS, RFI [9,20,21] | - |
| (↑) Cognitive performance [8,26,27,28,29] | - |
| ↓ IL-6, IL-6/IL-1ra [8,26,27,28,29] | - |
| Not suppress cellular immunity [31] | - |
| Post Treatment | |
| ↑Muscle strength [7,10,35,36,37,38,40,41,42,45,46,47,48,49,50,51,52,53,54,56] | ↔ Incidence of fractures or falls [37,46,47,48,49,50,51,52,53,54] |
| ↑Muscle endurance [56] | NR [7,38,39,40,41] |
| ↔ EMG [42] | No adverse events [45,46,47,48,49,50,51,52,53,54,56] |
| (↑) Aerobic capacity [39] | ↓ Number and severity of symptoms [46,47,48,49,50,51,52,53,54] |
| ↓ Fatigue [7,38,41] | - |
| ↑ Perceived exertion [39] | - |
| (↑) ROM [38] | - |
| ↑ ROM [56] | - |
| ↔ BMI [10,35,36,40,46,47,48,49,50,51,52,53,54] | - |
| (↓) BMI [39] | - |
| ↔ Body weight [10,35,36,46,47,48,49,50,51,52,53,54] | - |
| ↔ Body composition [7,10,35,36,41,46,47,48,49,50,51,52,53,54] | - |
| ↓ Body fat [45,46,47,48,49,50,51,52,53,54] | - |
| ↔ Bone formation [40,46,47,48,49,50,51,52,53,54] | - |
| ↓ Bone resorption [40] | - |
| ↑ LBM [10,35,36] | - |
| Attenuates the muscle mass decline [46,47,48,49,50,51,52,53,54] | - |
| ↔ Circumference [10,35,36,46,47,48,49,50,51,52,53,54,56] | - |
| ↔ Upper Limbs Volume [46,47,48,49,50,51,52,53,54] | - |
| ↑ Balance [37] | - |
| ↑ QoL [7,10,35,36,39,41,46,47,48,49,50,51,52,53,54,56] | - |
| ↔ Depression [10,35,36,38] | - |
| ↓ Depression [38] | - |
| ↑ Self-perceptions [38,46,47,48,49,50,51,52,53,54] | - |
| ↔ Norman score [46,47,48,49,50,51,52,53,54] | - |
| ↓ Deterioration of physical function [46,47,48,49,50,51,52,53,54] | - |
| ↔ DASH, BPI, FACT-B+4 or QLQ-BR23 [56] | - |
| ↓ IGF-II levels [10,35,36] | - |
| ↑ IGF-1 [45] | - |
| ↓ TNF-α on their NK cells [7,41] | - |
| ↔ miRNA [44] | - |
| Positive correlations between strength improvements and changes to circulating miRNAs [44] | - |
| ↔ Telomere length [43] | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Montaño-Rojas, L.S.; Romero-Pérez, E.M.; Medina-Pérez, C.; Reguera-García, M.M.; de Paz, J.A. Resistance Training in Breast Cancer Survivors: A Systematic Review of Exercise Programs. Int. J. Environ. Res. Public Health 2020, 17, 6511. https://doi.org/10.3390/ijerph17186511
Montaño-Rojas LS, Romero-Pérez EM, Medina-Pérez C, Reguera-García MM, de Paz JA. Resistance Training in Breast Cancer Survivors: A Systematic Review of Exercise Programs. International Journal of Environmental Research and Public Health. 2020; 17(18):6511. https://doi.org/10.3390/ijerph17186511
Chicago/Turabian StyleMontaño-Rojas, Leidy Sofía, Ena Monserrat Romero-Pérez, Carlos Medina-Pérez, María Mercedes Reguera-García, and José Antonio de Paz. 2020. "Resistance Training in Breast Cancer Survivors: A Systematic Review of Exercise Programs" International Journal of Environmental Research and Public Health 17, no. 18: 6511. https://doi.org/10.3390/ijerph17186511
APA StyleMontaño-Rojas, L. S., Romero-Pérez, E. M., Medina-Pérez, C., Reguera-García, M. M., & de Paz, J. A. (2020). Resistance Training in Breast Cancer Survivors: A Systematic Review of Exercise Programs. International Journal of Environmental Research and Public Health, 17(18), 6511. https://doi.org/10.3390/ijerph17186511







