Scabies and Pediculosis in Penitentiary Institutions in Poland—A Study of Ectoparasitoses in Confinement Conditions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Data Collection
2.2. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chosidow, O. Scabies and pediculosis. Lancet 2000, 355, 819–826. [Google Scholar] [CrossRef]
- Ciftci, H.; Karaca, S.; Dogru, O.; Cetinkaya, Z.; Kulac, M. Prevalence of pediculosis and scabies in preschool nursery children of Afyon, Turkey. Korean J. Parasitol. 2006, 44, 95–98. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kouotou, E.A.; Nansseu, J.R.N.; Kouawa, M.K.; Zoung-Kanyi Bissek, A.C. Prevalence and drivers of human scabies among children and adolescents living and studying in Cameroonian boarding schools. Parasit. Vectors 2016, 9, 400. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walker, S.L.; Lebas, E.; De Sario, V.; Deyasso, Z.; Doni, S.N.; Marks, M.; Roberts, C.H.; Lambert, S.M. The prevalence and association with health-related quality of life of tungiasis and scabies in schoolchildren in southern Ethiopia. PLoS Negl. Trop. Dis. 2017, 11, e0005808. [Google Scholar] [CrossRef] [PubMed]
- Birkemoe, T.; Lindstedt, H.H.; Ottesen, P.; Soleng, A.; Næss, Ø.; Rukke, B.A. Head lice predictors and infestation dynamics among primary school children in Norway. Fam. Pract. 2016, 33, 23–29. [Google Scholar] [CrossRef] [Green Version]
- Karakus, M.; Arici, A.; Töz, S.Ö.; Özbel, Y. Prevalence of Head Lice in Two Socio-economically Different Schools in the Center of Izmir City, Turkey. Turk. Parazitol. Derg. 2014, 38, 32–36. [Google Scholar] [CrossRef]
- Arlian, L.G.; Morgan, M.S. A review of Sarcoptes scabiei: Past, present and future. Parasit. Vectors 2017, 10, 297. [Google Scholar] [CrossRef]
- Cassell, J.A.; Middleton, J.; Nalabanda, A.; Lanza, S.; Head, M.G.; Bostock, J.; Hewitt, K.; Jones, C.I.; Darley, C.; Karir, S.; et al. Scabies outbreaks in ten care homes for elderly people: A prospective study of clinical features, epidemiology, and treatment outcomes. Lancet Infect. Dis. 2018, 18, 894–902. [Google Scholar] [CrossRef]
- Smith, P.W.; Bennett, G.; Bradley, S.; Drinka, P.; Lautenbach, E.; Marx, J.; Mody, L.; Nicolle, L.; Stevenson, K. SHEA/APIC Guideline: Infection Prevention and Control in the Long-Term Care Facility. Infect. Control Hosp. Epidemiol. 2008, 29, 785–814. [Google Scholar] [CrossRef] [Green Version]
- Falagas, M.E.; Matthaiou, D.K.; Rafailidis, P.I.; Panos, G.; Pappas, G. Worldwide prevalence of head lice. Emerg. Infect. Dis. 2008, 14, 1493–1494. [Google Scholar] [CrossRef]
- Romani, L.; Steer, A.C.; Whitfeld, M.J.; Kaldor, J.M. Prevalence of scabies and impetigo worldwide: A systematic review. Lancet Infect. Dis. 2015, 15, 960–967. [Google Scholar] [CrossRef]
- Mounsey, K.E.; Murray, H.C.; King, M.; Oprescu, F. Retrospective analysis of institutional scabies outbreaks from 1984 to 2013: Lessons learned and moving forward. Epidemiol. Infect. 2016, 144, 2462–2471. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mølle, L.; Gatherer, A.; Jürgens, R.; Stöver, H.; Nikogosian, H. Health in Prisons: A WHO Guide to the Essentials in Prison Health. WHO Regional Office Europe. 2007. Available online: http://www.euro.who.int/__data/assets/pdf_file/0009/99018/E90174.pdf (accessed on 15 May 2020).
- Stingeni, L.; Tramontana, M.; Principato, M.; Moretta, I.; Principato, S.; Bianchi, L.; Hansel, K. Nosocomial outbreak of crusted scabies in immunosuppressed patients caused by Sarcoptes scabiei var. canis. Br. J. Dermatol. 2020, 182, 498–500. [Google Scholar] [CrossRef] [PubMed]
- Hart, G. Factors associated with pediculosis pubis and scabies. Genitourin. Med. 1992, 68, 294–295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bartosik, K.; Buczek, A.; Zajac, Z.; Kulisz, J. Head pediculosis in schoolchildren in the eastern region of the European Union. Ann. Agric. Environ. Med. 2015, 22, 599–603. [Google Scholar] [CrossRef]
- Czarkowski, M.P.; Cielebąk, E.; Kondej, B.; Staszewska, E. Infectious Diseases and Poisonings in Poland in 2008. Annual Report of National Institute of Public Health—National Institute of Hygiene—Department of Epidemiology. Warsaw 2009. Available online: http://wwwold.pzh.gov.pl/oldpage/epimeld/2008/Ch_2008.pdf (accessed on 3 May 2020).
- Hygiene in Prisons in Individual European Countries—The Report. Available online: http://www.hfhr.pl/wp-content/uploads/2013/08/HFPC_raport_Raport_Higiena_w_zakladach_karnych.pdf (accessed on 5 May 2020).
- Sanders, K.M.; Akiyama, T. The vicious cycle of itch and anxiety. Neurosci. Biobehav. Rev. 2018, 87, 17–26. [Google Scholar] [CrossRef]
- Rogala, D.; Banach, A.; Jachimowicz-Gaweł, D.; Skinder, Ż.; Leźnicka, M. Health care system for persons detained in prisons in Poland. Hygeia Public Health 2013, 48, 441–448. [Google Scholar]
- Mellanby, K.; Johnson, C.G.; Bartley, W.C.; Brown, P. Experiments on the Survival and Behaviour of the Itch Mite, Sarcoptes scabiei DeG. var. hominis. Bull. Entomol. Res. 1942, 33, 267–271. [Google Scholar] [CrossRef]
- Arlian, L.G.; Runyan, R.A.; Achar, S.; Estes, S.A. Survival and infectivity of Sarcoptes scabiei var. canis and var. hominis. J. Am. Acad. Dermatol. 1984, 11, 210–215. [Google Scholar] [CrossRef]
- Gallardo, A.; Mougabure Cueto, G.; Picollo, M.I. Pediculus humanus capitis (head lice) and Pediculus humanus humanus (body lice): Response to laboratory temperature and humidity and susceptibility to monoterpenoids. Parasitol. Res. 2009, 105, 163–167. [Google Scholar] [CrossRef]
- Liu, J.M.; Wang, H.W.; Chang, F.W.; Liu, Y.P.; Chiu, F.H.; Lin, Y.C.; Cheng, K.C.; Hsu, R.J. The effects of climate factors on scabies. A 14-year population-based study in Taiwan. Parasite 2016, 23, 54. [Google Scholar] [CrossRef] [PubMed]
- Korycińska, J.; Dzika, E.; Kloch, M. Epidemiology of scabies in relation to socio-economic and selected climatic factors in north-east Poland. Ann. Agric. Environ. Med. 2019. [Google Scholar] [CrossRef]
- Nazari, M.; Goudarztalejerdi, R.; Moradi, A. Investigation of pediculosis prevalence and associated factors in the central prison of Hamadan in 2013. Pajouhan Sci. J. 2015, 13, 13–20. [Google Scholar]
- Shamsaddini, S.; Nasiri Kashani, M.; Sharifi, I.; Khajeh, K.A.M.; Pourlashkari, M. Prevalence of infectious skin diseases in the central prison of Kerman. Iran. J. Dermatol. 2000, 4, 19–25. [Google Scholar]
- Roodsari, M.R.; Malekzad, F.; Roodsari, S.R. Prevalence of scabies and pediculosis in Ghenzel Hesar prison. Iran. J. Clin. Infect. Dis. 2007, 2, 87–90. [Google Scholar]
- Kuruvila, M.; Shaikh, M.I.; Kumar, P. Pattern of dermatoses among inmates of district prison-Mangalore. Indian J. Dermatol. Venereol. Leprol. 2002, 68, 16–18. [Google Scholar]
- Leppard, B.; Naburi, A.E. The use of Ivermectin in controlling an outbreak of scabies in a prison. Br. J. Dermatol. 2000, 143, 520–523. [Google Scholar] [CrossRef]
- Singh, S.; Prasad, R.; Mohanty, A. High prevalence of sexually transmitted and blood-borne infections amongst the prisoners of a district jail in Northern India. Int. J. STD AIDS 1999, 10, 475–478. [Google Scholar] [CrossRef]
- Demoures, B.; Nkodo-Nkodo, E.; Mbam-Mbam, L. Primary health care in a prison environment, the Cameroon experience. Sante 1998, 8, 212–216. [Google Scholar]
- Willems, S.; Lapeere, H.; Haedens, N.; Pasteels, I.; Naeyaert, J.-M.; De Maeseneer, J. The importance of socio-economic status and individual characteristics on the prevalence of head lice in schoolchildren. Eur. J. Dermatol. 2005, 15, 387–392. [Google Scholar]
- Manjrekar, R.R.; Partridge, S.K.; Korman, A.K.; Barwick, R.S.; Juranek, D.D. Efficacy of 1% permethrin for the treatment of head louse infestations among Kosovar refugees. Mil. Med. 2000, 165, 698–700. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walton, S.F. The immunology of susceptibility and resistance to scabies. Parasite Immunol. 2010, 32, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Nazari, M.; Azizi, A. Epidemiological Pattern of Scabies and Its Social Determinant Factors in West of Iran. Health 2014, 6, 1972–1977. [Google Scholar] [CrossRef] [Green Version]
- Clore, E.R. Dispelling the common myths about pediculosis. J. Pediatr. Health Care 1989, 3, 28–33. [Google Scholar] [CrossRef]
- Ross, M.W. Pedagogy for prisoners: An approach to peer health education for inmates. J. Correct. Health Care. 2011, 17, 6–18. [Google Scholar] [CrossRef]
- MacDonald, M.; Rabiee, F.; Weilandt, C. Health promotion and young prisoners: A European perspective. Int. J. Prison Health 2013, 9, 151–164. [Google Scholar] [CrossRef] [PubMed]
- Donelle, L.; Hall, J. Health Promotion Body Maps of Criminalized Woman. J. Correct. Health Care. 2016, 22, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Maestre-Miquel, C.; Zabala-Baños, C.; García, J.A.; Antolín, J.M. Health education for prevalent problems in prison, Ocaña-I proyect (Spain). Rev. Esp. Sanid. Penit. 2016, 18, 86–94. [Google Scholar] [PubMed]
- Keogh, B.; McBennett, P.; deVries, J.; Higgins, A.; O’Shea, M.; Doyle, L. Prisoners perceptions of a mental health wellness workshop. Int. J. Prison Health 2017, 13, 81–90. [Google Scholar] [CrossRef]
- Moshki, M.; Zamani-Alavijeh, F.; Mojadam, M. Efficacy of Peer Education for Adopting Preventive Behaviors against Head Lice Infestation in Female Elementary School Students: A Randomised Controlled Trial. PLoS ONE 2017, 12, e0169361. [Google Scholar]
- Yingklang, M.; Sengthong, C.; Haonon, O.; Dangtakot, R.; Pinlaor, P.; Sota, C.; Pinlaor, S. Effect of a health education program on reduction of pediculosis in school girls at Amphoe Muang, Khon Kaen Province, Thailand. PLoS ONE 2018, 13, e0198599. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stingeni, L.; Bianchi, L.; Hansel, K.; Neve, D.; Foti, C.; Corazza, M.; Bini, V.; Moretta, I.; Principato, M. Dermatitis caused by arthropods in domestic environment: An Italian multicentre study. J. Eur. Acad. Dermatol. Venereol. 2017, 31, 1526–1533. [Google Scholar] [CrossRef] [PubMed]
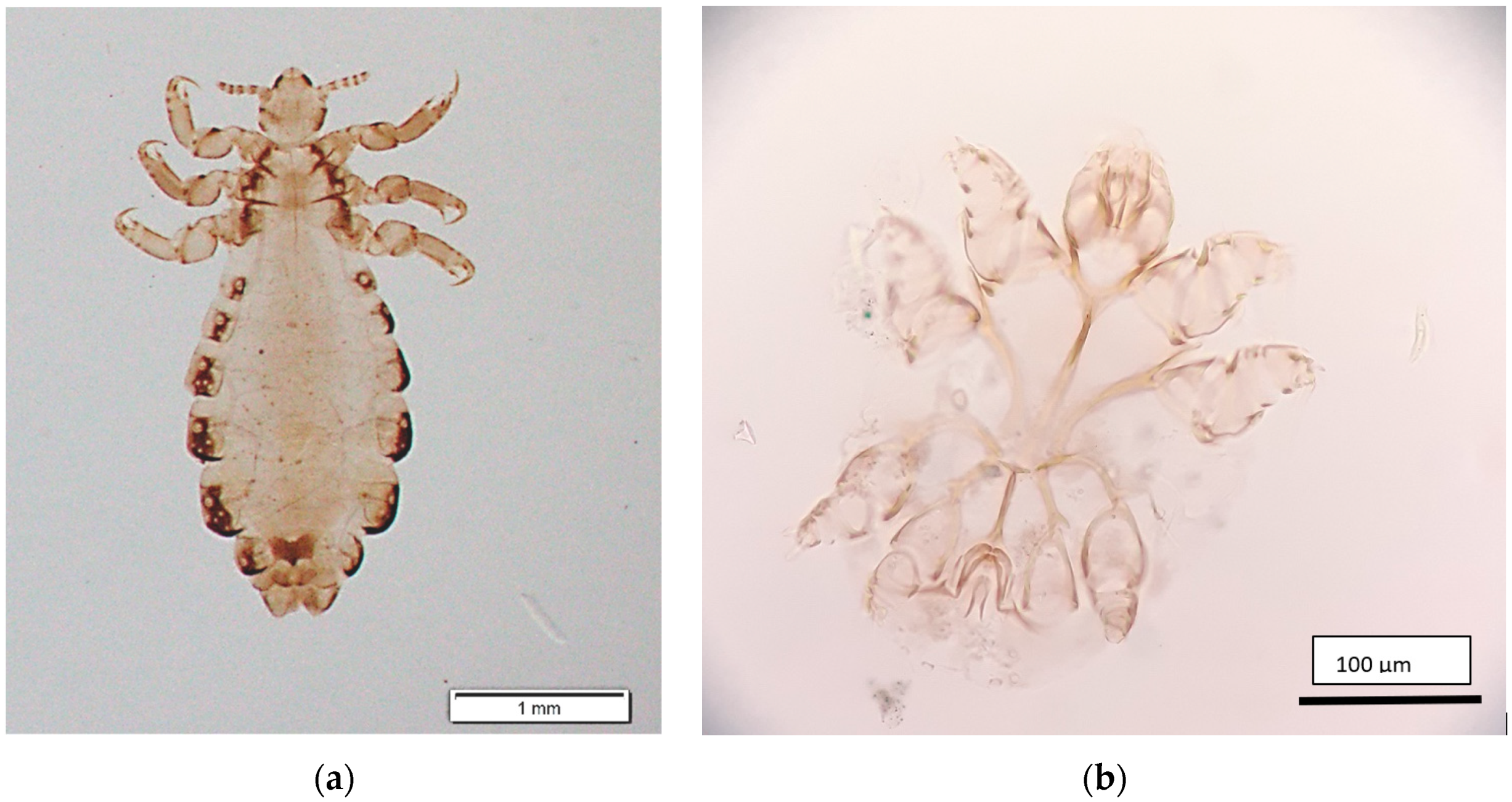
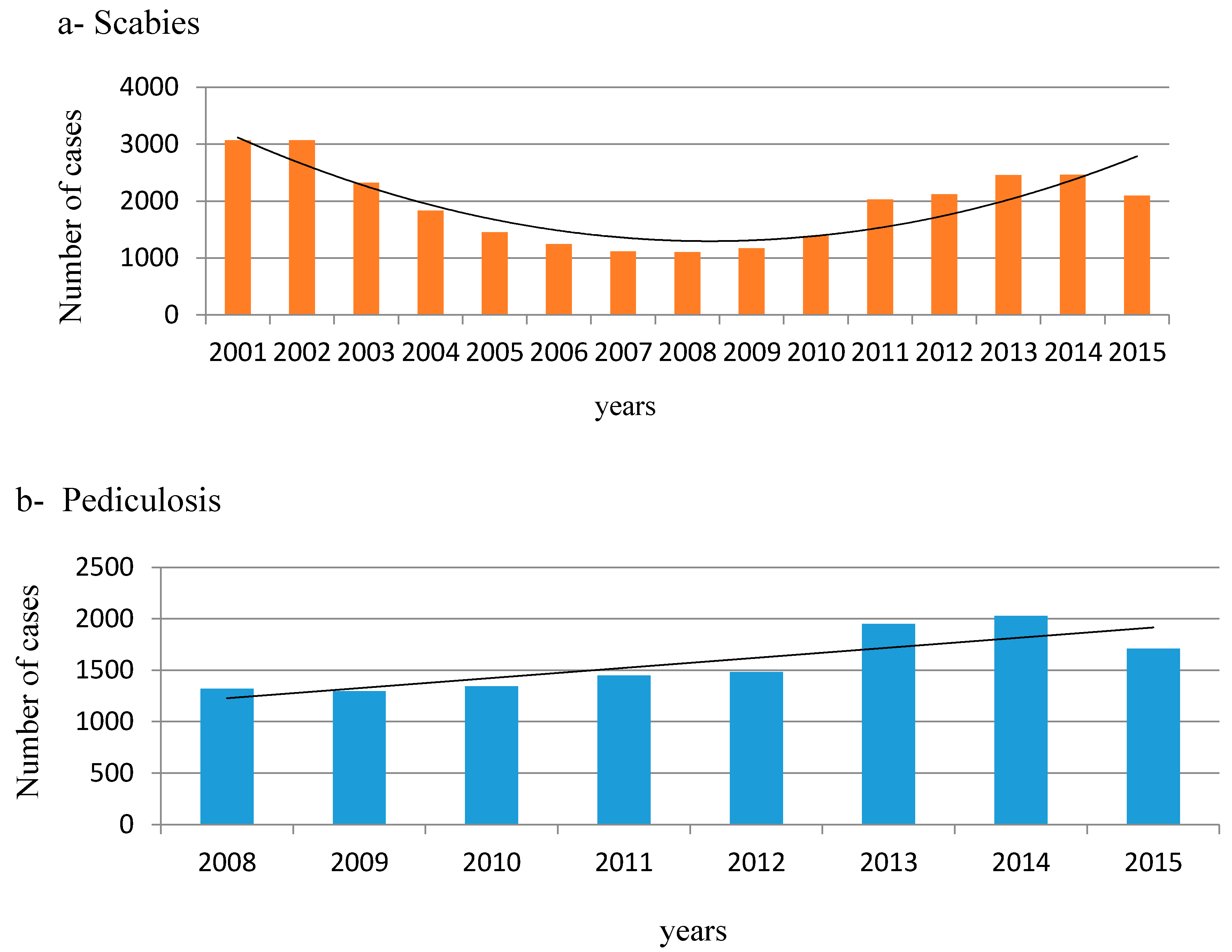
| Years | Number of Prisoners 1 | Scabies | |
|---|---|---|---|
| Number of Cases | Prevalence | ||
| 2001 | 78,716 | 3072 | 3.9% |
| 2002 | 81,391 | 3071 | 3.8% |
| 2003 | 81,321 | 2324 | 2.9% |
| 2004 | 80,239 | 1833 | 2.3% |
| 2005 | 82,761 | 1455 | 1.8% |
| 2006 | 87,370 | 1245 | 1.4% |
| 2007 | 89,995 | 1115 | 1.2% |
| 2008 | 85,920 | 1103 | 1.3% |
| 2009 | 85,384 | 1172 | 1.4% |
| 2010 | 82,863 | 1387 | 1.7% |
| 2011 | 82,558 | 2029 | 2.5% |
| 2012 | 84,399 | 2121 | 2.5% |
| 2013 | 83,898 | 2455 | 2.9% |
| 2014 | 78,987 | 2465 | 3.1% |
| 2015 | 74,814 | 2096 | 2.8% |
| Total 28,943 | M * 2.3% | ||
| Years | Number of Prisoners 1 | Pediculosis | |
|---|---|---|---|
| Number of Cases | Prevalence | ||
| 2008 | 85,920 | 1319 | 1.5% |
| 2009 | 85,384 | 1296 | 1.5% |
| 2010 | 82,863 | 1344 | 1.6% |
| 2011 | 82,558 | 1448 | 1.8% |
| 2012 | 84,399 | 1484 | 1.8% |
| 2013 | 83,898 | 1949 | 2.3% |
| 2014 | 78,987 | 2029 | 2.6% |
| 2015 | 74,814 | 1710 | 2.3% |
| Total 12,579 | M * 1.9% | ||
| Years | Number of Prisoners | Scabies | Pediculosis | ||
|---|---|---|---|---|---|
| Number of Cases | Prevalence | Number of Cases | Prevalence | ||
| 2010 | 4301 | 12 | 0.3% | 9 | 0.2% |
| 2011 | 4261 | 39 | 0.9% | 6 | 0.1% |
| 2012 | 4569 | 46 | 1.0% | 32 | 0.7% |
| 2013 | 4397 | 41 | 0.9% | 49 | 1.1% |
| 2014 | 4336 | 33 | 0.8% | 36 | 0.8% |
| 2015 | 4001 | 31 | 0.8% | 32 | 0.8% |
| Total 202 | M * 0.8% | Total 164 | M * 0.6% | ||
| rs = 0.714; p = 0.111 | rs = 0.522; p = 0.288 | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bartosik, K.; Tytuła, A.; Zając, Z.; Buczek, W.; Jasztal-Kniażuk, A.; Błaszkiewicz, P.S.; Borzęcki, A. Scabies and Pediculosis in Penitentiary Institutions in Poland—A Study of Ectoparasitoses in Confinement Conditions. Int. J. Environ. Res. Public Health 2020, 17, 6086. https://doi.org/10.3390/ijerph17176086
Bartosik K, Tytuła A, Zając Z, Buczek W, Jasztal-Kniażuk A, Błaszkiewicz PS, Borzęcki A. Scabies and Pediculosis in Penitentiary Institutions in Poland—A Study of Ectoparasitoses in Confinement Conditions. International Journal of Environmental Research and Public Health. 2020; 17(17):6086. https://doi.org/10.3390/ijerph17176086
Chicago/Turabian StyleBartosik, Katarzyna, Andrzej Tytuła, Zbigniew Zając, Weronika Buczek, Anita Jasztal-Kniażuk, Paweł Szczepan Błaszkiewicz, and Adam Borzęcki. 2020. "Scabies and Pediculosis in Penitentiary Institutions in Poland—A Study of Ectoparasitoses in Confinement Conditions" International Journal of Environmental Research and Public Health 17, no. 17: 6086. https://doi.org/10.3390/ijerph17176086





