The Relationship between Maternal Personality Disorder and Early Birth Outcomes: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
1.1. Preterm Birth
1.2. APGAR
1.3. Low Birth Weight
2. Objectives
- To conduct a systematic review to identify the impact of personality disorder of the mother on risk of preterm birth <37 weeks gestation, birth weight <2500 g, and APGAR score <7;
- To assess the quality of the research literature identified through the systematic search strategy;
- To conduct a meta-analysis to synthesis the data on preterm birth, birth weights, and APGAR score where data is available.
3. Methods
3.1. Eligibility Criteria
- Studies which include mothers with a diagnosis of personality disorder or identified through the study as meeting diagnostic criteria of a personality disorder of any type.
- Studies reporting neonatal outcomes (specifically preterm birth <37 weeks gestation, APGAR scores <7, and low birth weight <2500 g).
3.2. Exclusion Criteria
- Studies are excluded if they are review articles, expert opinion commentaries, or single case report.
- Studies are excluded if they are not written in English language.
- Diagnostic criteria for personality disorder were changed significantly with the introduction of the Diagnostic and Statistical Manual version III in 1980; therefore, studies published before this will not be included.
3.3. Information Sources
3.4. Study Records
3.5. Management
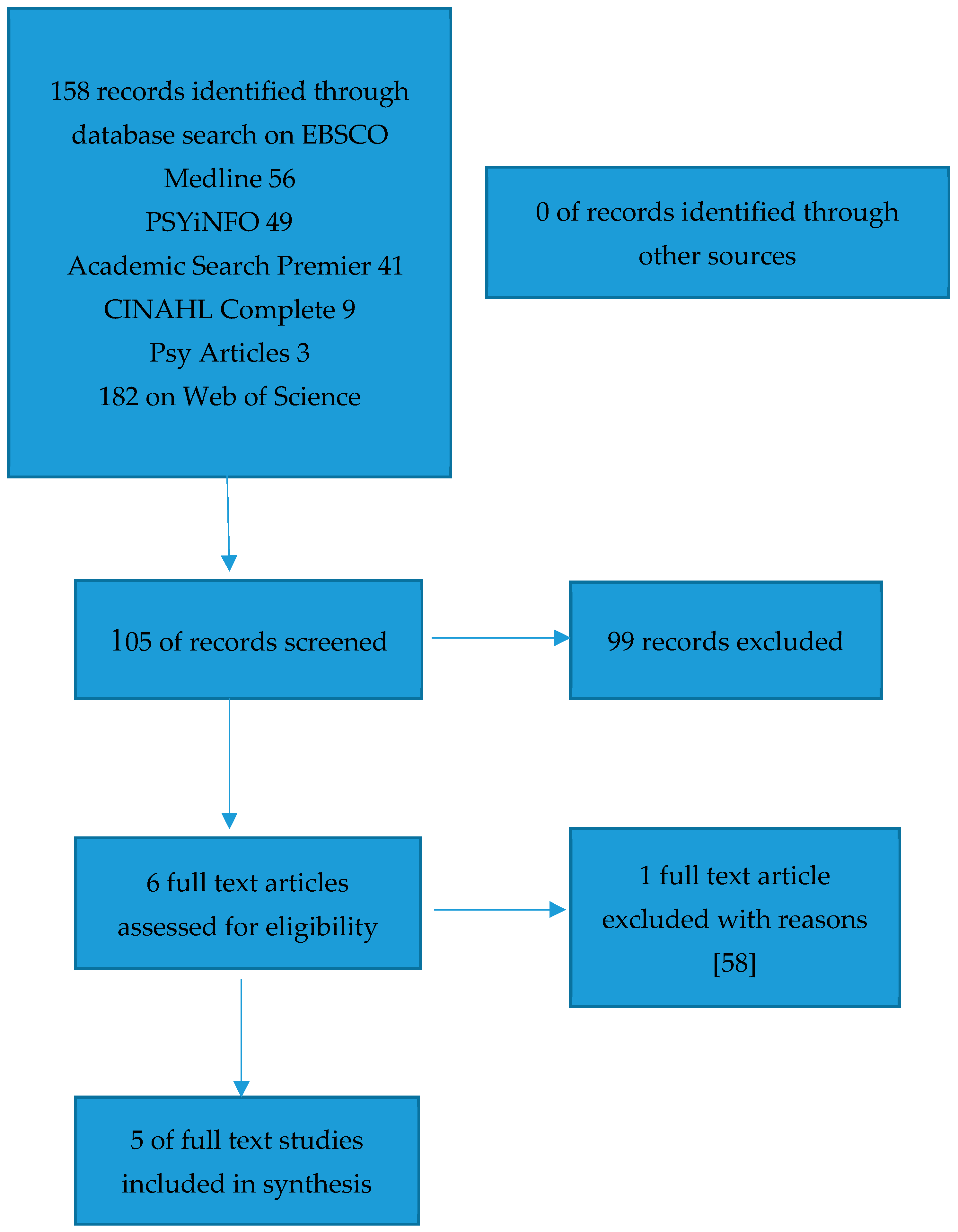
3.6. Selection Process
3.7. Critical Appraisal of Studies
3.8. Meta-Analysis
4. Results
4.1. Study Summary
4.2. Population of Study/Identifying the Population
4.3. Prevalence Rates of Personality Disorder
4.4. Diagnostic Issues and Comorbidity
4.5. Baseline Characteristics of the Participants
4.6. Country of Origin of the Studies
5. Meta-Analysis
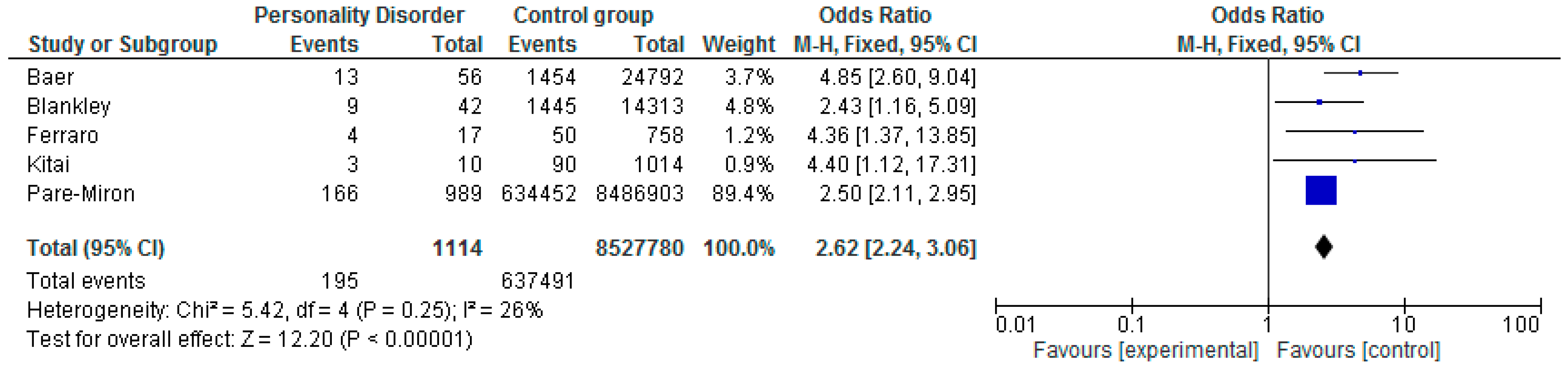
5.1. Preterm Birth < 37 Weeks Gestation
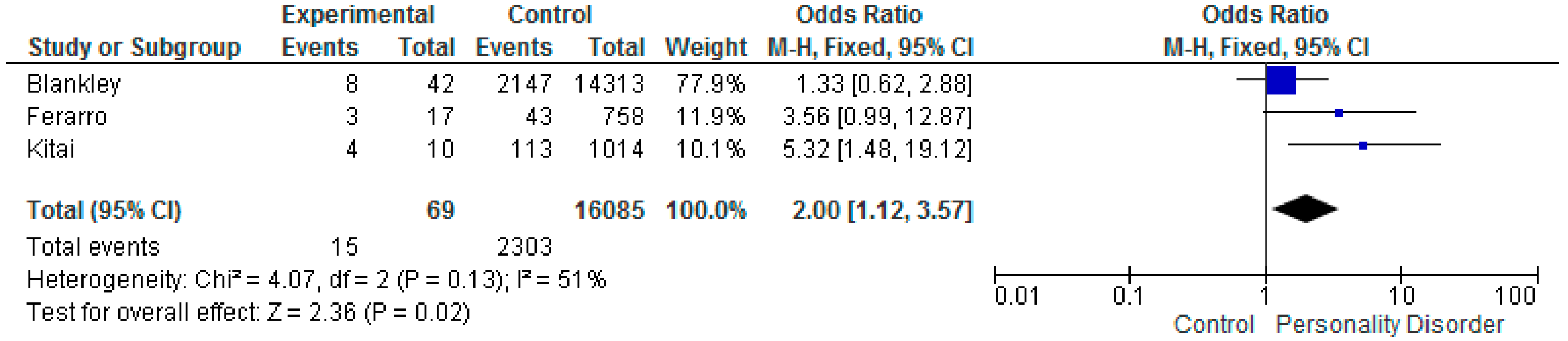
5.2. Low Birth Weight < 2500 g
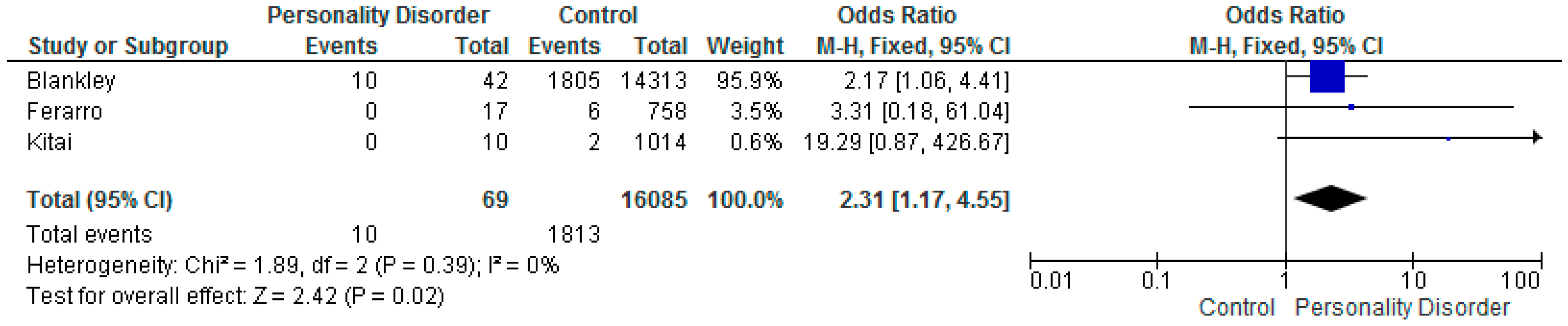
5.3. APGAR Score Less Than 7
6. Discussion
7. Limitations
- Both prospective and retrospective study design have been used across the included studies, which may impact on the overall outcome of the systematic review/meta-analysis.
- Only one study [59] provided detailed information regarding baseline characteristics of the study participants with personality disorder, including the control group.
- Different diagnostic classification systems were employed in the studies which has brought variability to the methods used across the studies.
- There were lower than expected prevalence rates of personality disorder across all the included studies, this may be due to under reporting or misclassification. across all studies.
- There were significant differences in the sample sizes, most were small and although there was a large overall affect across the meta-analysis this is due to 1 study [59].
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Greenmyer, J.R.; Popova, S.; Klug, M.G.; Burd, L. Fetal alcohol spectrum disorder: A systematic review of the cost of and savings from prevention in the United States and Canada. Addiction 2020, 115, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Popova, S.; Dozet, D.; Burd, L. Fetal Alcohol Spectrum Disorder: Can We Change the Future? Alcohol. Clin. Exp. Res. 2020, 44, 815–819. [Google Scholar] [CrossRef] [PubMed]
- Vorgias, D.; Bernstein, B. Fetal Alcohol Syndrome; Stat Pearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Gentile, S. Pharmacological management of borderline personality disorder in a pregnant woman with a previous history of alcohol addiction: A case report. Clin. Drug Investig. 2015, 35, 761–763. [Google Scholar] [CrossRef]
- Magnusson, A.; Goransson, M.; Heilig, M. Hazardous alcohol users during pregnancy: Psychiatric health and personality traits. Drug Alcohol. Depend. 2007, 89, 275–281. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Volkert, J.; Gablonski, T.C.; Rabung, S. Prevalence of personality disorders in the general adult population in Western countries: Systematic review and meta-analysis. Br. J. Psychiatry 2018, 213, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Beckwith, H.; Moran, P.F.; Reilly, J. Personality disorder prevalence in psychiatric outpatients: A systematic literature review. Personal. Ment. Health 2014, 8, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Hopwood, C.J.; Kotov, R.; Krueger, R.F.; Watson, D.; Widiger, T.A.; Althoff, R.R.; Ansell, E.B.; Bach, B.; Michael Bagby, R.; Blais, M.A.; et al. The time has come for dimensional personality disorder diagnosis. Personal. Ment. Health 2018, 12, 82–86. [Google Scholar] [CrossRef]
- Grant, B.F.; Chou, S.P.; Goldstein, R.B.; Huang, B.; Stinson, F.S.; Saha, T.D.; Smith, S.M.; Dawson, D.A.; Pulay, A.J.; Pickering, R.P.; et al. Prevalence, correlates, disability, and comorbidity of DSM-IV borderline personality disorder: Results from the Wave 2 National Epidemiologic Survey on Alcohol and Related Conditions. J. Clin. Psychiatry 2008, 69, 533–545. [Google Scholar] [CrossRef]
- Oldham, J.M.; Skodol, A.E.; Kellman, H.D.; Hyler, S.E.; Doidge, N.; Rosnick, L.; Gallaher, P.E. Comorbidity of axis I and axis II disorders. Am. J. Psychiatry 1995, 152, 571–578. [Google Scholar] [CrossRef]
- World Health Organization. International Classification of Diseases; World Health Organization: Geneva, Switzerland, 1978. [Google Scholar]
- Bach, B.; First, M.B. Application of the ICD-11 classification of personality disorders. BMC Psychiatry 2018, 18, 351. [Google Scholar] [CrossRef]
- Tyrer, P.; Mulder, R.; Kim, Y.R.; Crawford, M.J. The Development of the ICD-11 Classification of Personality Disorders: An Amalgam of Science, Pragmatism, and Politics. Annu. Rev. Clin. Psychol. 2019, 15, 481–502. [Google Scholar] [CrossRef] [PubMed]
- Giourou, E.; Skokou, M.; Andrew, S.P.; Alexopoulou, K.; Gourzis, P.; Jelastopulu, E. Complex posttraumatic stress disorder: The need to consolidate a distinct clinical syndrome or to reevaluate features of psychiatric disorders following interpersonal trauma? World J. Psychiatry 2018, 8, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Karatzias, T.; Bisson, J.; Roberts, N.; Shevlin, M.; Hyland, P.; Maercker, A.; Ben-Ezra, M.; Coventry, P.; Murphy, P.; Cloitre, M.; et al. Psychological interventions for ICD-11 complex PTSD symptoms: Systematic review and meta-analysis. Psychol. Med. 2019, 49, 1761–1775. [Google Scholar] [CrossRef] [PubMed]
- Bateman, A.; Tyrer, P. Services for personality disorder: Organisation for inclusion. Adv. Psychiatr. Treat. 2004, 10, 425–433. [Google Scholar] [CrossRef]
- Ferguson, A. Borderline Personality Disorder and Access to Services: A Crucial Social Justice Issue. Aust. Soc. Work 2016, 69, 206–214. [Google Scholar] [CrossRef]
- Tyrer, P.; Reed, G.M.; Crawford, M.J. Classification, assessment, prevalence, and effect of personality disorder. Lancet 2015, 385, 717–726. [Google Scholar] [CrossRef]
- Sheehan, L.; Nieweglowski, K.; Corrigan, P. The Stigma of Personality Disorders. Curr. Psychiatry Rep. 2016, 18, 11. [Google Scholar] [CrossRef]
- Watts, J. Problems with the ICD-11 classification of personality disorder. Lancet Psychiatry 2019, 6, 461–463. [Google Scholar] [CrossRef]
- Gescher, D.M.; Kahl, K.G.; Hillemacher, T.; Frieling, H.; Kuhn, J.; Frodl, T. Epigenetics in Personality Disorders: Today’s Insights. Front. Psychiatry 2018, 9, 579. [Google Scholar] [CrossRef]
- Amad, A.; Ramoz, N.; Thomas, P.; Jardri, R.; Gorwood, P. Genetics of borderline personality disorder: Systematic review and proposal of an integrative model. Neurosci. Biobehav. Rev. 2014, 40, 6–19. [Google Scholar] [CrossRef]
- Ruocco, A.C.; Carcone, D. A Neurobiological Model of Borderline Personality Disorder: Systematic and Integrative Review. Harv. Rev. Psychiatry 2016, 24, 311–329. [Google Scholar] [CrossRef] [PubMed]
- De Genna, N.M.; Feske, U.; Larkby, C.; Angiolieri, T.; Gold, M.A. Pregnancies, abortions, and births among women with and without borderline personality disorder. Womens Health Issues 2012, 22, e371–e377. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, A.A.; Rohde, L.A.; Polanczyk, G.V.; Argeu, A.; Miguel, E.C.; Grisi, S.; Fleitlich-Bilyk, B. The specific and combined role of domestic violence and mental health disorders during pregnancy on new-born health. BMC Pregnancy Childbirth 2017, 17, 257. [Google Scholar] [CrossRef] [PubMed]
- Murphy, C.C.; Schei, B.; Myhr, T.L.; Du Mont, J. Abuse: A risk factor for low birth weight? A systematic review and meta-analysis. CMAJ 2001, 164, 1567–1572. [Google Scholar] [PubMed]
- Florange, J.G.; Herpertz, S.C. Parenting in Patients with Borderline Personality Disorder, Sequelae for the Offspring and Approaches to Treatment and Prevention. Curr. Psychiatry Rep. 2019, 21, 9. [Google Scholar] [CrossRef]
- Judd, F.; Komiti, A.; Sheehan, P.; Newman, L.; Castle, D.; Everall, I. Adverse obstetric and neonatal outcomes in women with severe mental illness: To what extent can they be prevented? Schizophr. Res. 2014, 157, 305–309. [Google Scholar] [CrossRef]
- Guy, N.; Newton-Howes, G.; Ford, H.; Williman, J.; Foulds, J. The prevalence of comorbid alcohol use disorder in the presence of personality disorder: Systematic review and explanatory modelling. Personal. Ment. Health 2018, 12, 216–228. [Google Scholar] [CrossRef]
- Nesari, M.; Olson, J.K.; Vandermeer, B.; Slater, L.; Olson, D.M. Does a maternal history of abuse before pregnancy affect pregnancy outcomes? A systematic review with meta-analysis. BMC Pregnancy Childbirth 2018, 18, 404. [Google Scholar] [CrossRef]
- Newton-Howes, G.; Clark, L.A.; Chanen, A. Personality disorder across the life course. Lancet 2015, 385, 727–734. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence. Antenatal and Postnatal Mental Health: Clinical Management and Service Guidance; NICE Guideline (CG192); NICE: London, UK, 2014. [Google Scholar]
- Royal College of Psychiatrists. Perinatal Mental Health Services. In Recommendations for the Provision of Services for Childbearing Women; College Report; The Royal College of Psychiatrists: London, UK, 2015. [Google Scholar]
- Staneva, A.; Bogossian, F.; Pritchard, M.; Wittkowski, A. The effects of maternal depression, anxiety, and perceived stress during pregnancy on preterm birth: A systematic review. Women Birth 2015, 28, 179–193. [Google Scholar] [CrossRef]
- Lee, H.C.; Lin, H.C. Maternal bipolar disorder increased low birthweight and preterm births: A nationwide population-based study. J. Affect. Disord. 2010, 121, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Mei-Dan, E.; Ray, J.G.; Vigod, S.N. Perinatal outcomes among women with bipolar disorder: A population-based cohort study. Am. J. Obstet. Gynecol. 2015, 212, 367.e1–367.e8. [Google Scholar] [CrossRef] [PubMed]
- Witt, S.H.; Streit, F.; Jungkunz, M.; Frank, J.; Awasthi, S.; Reinbold, C.S.; Treutlein, J.; Degenhardt, F.; Forstner, A.J.; Heilmann-Heimbach, S.; et al. Genome-wide association study of borderline personality disorder reveals genetic overlap with bipolar disorder, major depression and schizophrenia. Transl. Psychiatry 2017, 7, e1155. [Google Scholar] [CrossRef] [PubMed]
- Jablensky, A.V.; Morgan, V.; Zubrick, S.R.; Bower, C.; Yellachich, L.A. Pregnancy, delivery, and neonatal complications in a population cohort of women with schizophrenia and major affective disorders. Am. J. Psychiatry 2005, 162, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Howard, L.M.; Bekele, D.; Rowe, M.; Demilew, J.; Bewley, S.; Marteau, T.M. Smoking cessation in pregnant women with mental disorders: A cohort and nested qualitative study. BJOG 2013, 120, 362–370. [Google Scholar] [CrossRef]
- Blankley, G.; Galbally, M.; Snellen, M.; Power, J.; Lewis, A.J. Borderline Personality Disorder in the perinatal period: Early infant and maternal outcomes. Australas. Psychiatry 2015, 23, 688–692. [Google Scholar] [CrossRef]
- Chawanpaiboon, S.; Vogel, J.P.; Moller, A.B.; Lumbiganon, P.; Petzold, M.; Hogan, D.; Landoulsi, S.; Jampathong, N.; Kongwattanakul, K.; Laopaiboon, M.; et al. Global, regional, and national estimates of levels of preterm birth in 2014: A systematic review and modelling analysis. Lancet Glob. Health 2019, 7, e37–e46. [Google Scholar] [CrossRef]
- Quinn, J.A.; Munoz, F.M.; Gonik, B.; Frau, L.; Cutland, C.; Mallett-Moore, T.; Kissou, A.; Wittke, F.; Das, M.; Nunes, T.; et al. Preterm birth: Case definition & guidelines for data collection, analysis, and presentation of immunisation safety data. Vaccine 2016, 34, 6047–6056. [Google Scholar] [CrossRef]
- Steer, P. The epidemiology of preterm labour. BJOG 2005, 112, 1–3. [Google Scholar] [CrossRef]
- Raju, T. The “Late Preterm” Birth-Ten Years Later. Pediatrics 2017, 139, e20163331. [Google Scholar] [CrossRef]
- Mangham, L.J.; Petrou, S.; Doyle, L.W.; Draper, E.S.; Marlow, N. The cost of preterm birth throughout childhood in England and Wales. Pediatrics 2009, 123, e312–e327. [Google Scholar] [CrossRef] [PubMed]
- Thorngren-Jerneck, K.; Herbst, A. Low 5-minute Apgar score: A population-based register study of 1 million term births. Obstet. Gynecol. 2001, 98, 65–70. [Google Scholar] [CrossRef]
- Cnattingius, S.; Norman, M.; Granath, F.; Petersson, G.; Stephansson, O.; Frisell, T. Apgar score components at 5 minutes: Risks and prediction of neonatal mortality. Paediatr. Perinat. Epidemiol. 2017, 31, 328–337. [Google Scholar] [CrossRef] [PubMed]
- Casey, B.M.; McIntire, D.D.; Leveno, K.J. The continuing value of the Apgar score for the assessment of newborn infants. N. Engl. J. Med. 2001, 344, 467–471. [Google Scholar] [CrossRef] [PubMed]
- Papile, L.A. The Apgar score in the 21st century. N. Engl. J. Med. 2001, 344, 519–520. [Google Scholar] [CrossRef] [PubMed]
- ACOG. Committee Opinion No. 644: The Apgar Score. Obstet. Gynecol. 2015, 126, e52–e55. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, A.; Cuttini, M.; Wood, R.; Velebil, P.; Delnord, M.; Zile, I.; Barros, H.; Gissler, M.; Hindori-Mohangoo, A.D.; Blondel, B.; et al. Can the Apgar score be used for international comparisons of newborn health? Paediatr. Perinat. Epidemiol. 2017, 31, 338–345. [Google Scholar] [CrossRef]
- UNICEF; WHO. Low Birthweight: Country, Regional and Global Estimates; Wardlaw, T., Blanc, A., Zupan, J., Åhman, E., Eds.; UNICEF: New York, NY, USA; WHO: Geneva, Switzerland, 2004. [Google Scholar]
- Hughes, M.M.; Black, R.E.; Katz, J. 2500-g Low Birth Weight Cutoff: History and Implications for Future Research and Policy. Matern. Child. Health J. 2017, 21, 283–289. [Google Scholar] [CrossRef]
- Risnes, K.R.; Vatten, L.J.; Baker, J.L.; Jameson, K.; Sovio, U.; Kajantie, E.; Osler, M.; Morley, R.; Jokela, M.; Painter, R.C.; et al. Birthweight and mortality in adulthood: A systematic review and meta-analysis. Int. J. Epidemiol. 2011, 40, 647–661. [Google Scholar] [CrossRef]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; Group, P.-P. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef]
- Nadelson, S.; Nadelson, L.S. Evidence-based practice article reviews using CASP tools: A method for teaching EBP. Worldviews Evid. Based Nurs. 2014, 11, 344–346. [Google Scholar] [CrossRef] [PubMed]
- Borjesson, K.; Ruppert, S.; Wager, J.; Bagedahl-Strindlund, M. Personality disorder, psychiatric symptoms and experience of childbirth among childbearing women in Sweden. Midwifery 2007, 23, 260–268. [Google Scholar] [CrossRef] [PubMed]
- Pare-Miron, V.; Czuzoj-Shulman, N.; Oddy, L.; Spence, A.R.; Abenhaim, H.A. Effect of Borderline Personality Disorder on Obstetrical and Neonatal Outcomes. Womens Health Issues 2016, 26, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Baer, R.J.; Chambers, C.D.; Bandoli, G.; Jelliffe-Pawlowski, L.L. Risk of preterm birth by subtype among Medi-Cal participants with mental illness. Am. J. Obstet. Gynecol. 2016, 215, 519.e1–519.e9. [Google Scholar] [CrossRef]
- Kitai, T.; Komoto, Y.; Kakubari, R.; Konishi, H.; Tanaka, E.; Nakajima, S.; Muraji, M.; Ugaki, H.; Matsunaga, H.; Takemura, M. A comparison of maternal and neonatal outcomes of pregnancy with mental disorders: Results of an analysis using propensity score-based weighting. Arch. Gynecol. Obstet. 2014, 290, 883–889. [Google Scholar] [CrossRef]
- Skodol, A.E. Personality pathology and population health. Lancet Psychiatry 2016, 3, 595–596. [Google Scholar] [CrossRef]
- Paris, J.; Black, D.W. Borderline personality disorder and bipolar disorder: What is the difference and why does it matter? J. Nerv. Ment. Dis. 2015, 203, 3–7. [Google Scholar] [CrossRef]
- Ellard, G.A.; Johnstone, F.D.; Prescott, R.J.; Ji-Xian, W.; Jian-Hua, M. Smoking during pregnancy: The dose dependence of birthweight deficits. Br. J. Obstet. Gynaecol. 1996, 103, 806–813. [Google Scholar] [CrossRef]
- Kelly, R.; Russo, J.; Holt, V.; Danielsen, B.; Zatzick, D.; Walker, E.; Katon, W. Psychiatric and substance use disorders as risk factors for low birth weight and preterm delivery. Obstet. Gynecol. 2002, 100, 297–304. [Google Scholar] [CrossRef]
- Oni, H.; Khan, M.; Abdel-Latif, M.; Buultjens, M.; Islam, M. Short-term health outcomes of newborn infants of substance using mothers in Australia and New Zealand: A systematic review. J. Obstet. Gynaecol. Res. 2019, 45, 1783–1795. [Google Scholar] [CrossRef]
- Manuck, T.A. Racial and ethnic differences in preterm birth: A complex, multifactorial problem. Semin. Perinatol. 2017, 41, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Blumenshine, P.; Egerter, S.; Barclay, C.J.; Cubbin, C.; Braveman, P.A. Socioeconomic disparities in adverse birth outcomes: A systematic review. Am. J. Prev. Med. 2010, 39, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Malacova, E.; Regan, A.; Nassar, N.; Raynes-Greenow, C.; Leonard, H.; Srinivasjois, R.; Shand, A.W.; Lavin, T.; Pereira, G. Risk of stillbirth, preterm delivery, and fetal growth restriction following exposure in a previous birth: Systematic review and meta-analysis. BJOG 2018, 125, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Eke, A.C.; Saccone, G.; Berghella, V. Selective serotonin reuptake inhibitor (SSRI) use during pregnancy and risk of preterm birth: A systematic review and meta-analysis. BJOG 2016, 123, 1900–1907. [Google Scholar] [CrossRef]
- Goldenberg, R.L.; Culhane, J.F.; Iams, J.D.; Romero, R. Epidemiology and causes of preterm birth. Lancet 2008, 371, 75–84. [Google Scholar] [CrossRef]
- Stein, D.J.; Lund, C.; Nesse, R.M. Classification systems in psychiatry, diagnosis and global mental health in the era of DSM-5 and ICD 11. Curr. Opin. Psychiatry 2013, 26, 493. [Google Scholar] [CrossRef]
- Tyrer, P. A comparison of DSM and ICD classifications of mental disorder. Adv. Psychiatr. Treat. 2014, 20, 280–285. [Google Scholar] [CrossRef]
- Morey, L.C.; Benson, K.T. An investigation of adherence to diagnostic criteria, revisited: Clinical diagnosis of the DSM-IV/DSM-5 Section II personality disorders. J. Pers. Disord. 2016, 30, 130–144. [Google Scholar] [CrossRef]
- Stepp, S.D.; Whalen, D.J.; Pilkonis, P.A.; Hipwell, A.E.; Levine, M.D. Children of mothers with borderline personality disorder: Identifying parenting behaviors as potential targets for intervention. Pers. Disord. 2012, 3, 76–91. [Google Scholar] [CrossRef]
- Cannon, M.; Jones, P.B.; Murray, R.M. Obstetric complications and schizophrenia: Historical and meta-analytic review. Am. J. Psychiatry 2002, 159, 1080–1092. [Google Scholar] [CrossRef]
- Byrne, M.; Agerbo, E.; Ewald, H.; Eaton, W.W.; Mortensen, P.B. Parental age and risk of schizophrenia: A case-control study. Arch. Gen. Psychiatry 2003, 60, 673–678. [Google Scholar] [CrossRef] [PubMed]
- Petfield, L.; Startup, H.; Droscher, H.; Cartwright-Hatton, S. Parenting in mothers with borderline personality disorder and impact on child outcomes. Evid. Based Ment. Health 2015, 18, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Grote, N.K.; Bridge, J.A.; Gavin, A.R.; Melville, J.L.; Iyengar, S.; Katon, W.J. A meta-analysis of depression during pregnancy and the risk of preterm birth, low birth weight, and intrauterine growth restriction. Arch. Gen. Psychiatry 2010, 67, 1012–1024. [Google Scholar] [CrossRef] [PubMed]
- Rusner, M.; Berg, M.; Begley, C. Bipolar disorder in pregnancy and childbirth: A systematic review of outcomes. BMC Pregnancy Childbirth 2016, 16, 331. [Google Scholar] [CrossRef] [PubMed]
- Mental Health Taskforce. The Five Year Forward View for Mental Health: A Report from the Independent Mental Health Taskforce to the NHS in England; NHS: London, UK, 2016. [Google Scholar]
| Author Publication Year, Country of Study, Reference | Study Description | Patient Numbers/Participant Details N (%) | PD Identification | <37 Weeks | APGAR < 7 | Low Birth Weight (LBW) < 2500 g | ||
|---|---|---|---|---|---|---|---|---|
| Pare-Miron et al. 2016 US [59] | Retrospective cohort study. All births in study period 2003–2012. Extracting data from the Healthcare Cost and Utilisation project. | Women with PD N = 989 Control Group N = 8,486,903 | Borderline PD code identified on the database. Using ICD 9 diagnostic code to identify women. | PD N = 166 CG N = 634,452 OR 2.50 (CI 2.11–2.95) | Does not report | Does not report | ||
| Maternal age | Women without PD | Women with PD | ||||||
| <18 | 265,826 (3.13) | 21 (2.12) | ||||||
| 18-25 | 2,630,926 (31.00) | 437 (44.19) | ||||||
| 25-34 | 4,338,214 (51.12) | 454 (45.90) | ||||||
| >35 | 1,251,937 (14.75) | 77 (7.79) | ||||||
| Ethnicity | Women without PD | Women with PD | ||||||
| White | 3,584,672 (42.24) | 601 (60.77) | ||||||
| Black | 942,881 (11.11) | 93 (9.40) | ||||||
| Hispanic | 1,626,628 (19.17) | 56 (5.66) | ||||||
| Other | 726,829 (8.56) | 37 (3.74) | ||||||
| Alcohol/Substance Use | Women without PD | Women with PD | ||||||
| Tobacco | 483,895 (5.70) | 388 (39.23) | ||||||
| Alcohol | 8404 (0.10) | 38 (3.84) | ||||||
| Drugs | 234,290 (2.76) | 252 (25.48) | ||||||
| Blankley et al. 2015 Victoria, Australia [41] | Retrospective case review. Patients under the care of a perinatal service in Australia. January 2010 to June 2012. | Women with PD N = 42 Control Group N = 14,313 | Documented evidence in case file that patient met DSM IV diagnostic criteria. Meeting 5 of 9 DSM criteria. | PD N = 9 CG N =1445 OR 2.43 (CI 1.16–5.09) | PD N = 10 CG N = 1,805 OR 2.17 (CI 1.06–4.41) | PD N = 8 CG N = 2,147 OR 1.33 (CI 0.62–2.88) | ||
| Maternal age Age range 15–43 Mean = 27.43 SD 6.22 | ||||||||
| Ethnicity Information not provided for PD Sample | ||||||||
| Women with PD Substance use 18 (42.8%) | ||||||||
| Kitai et al. 2014 Japan [61] | Retrospective cohort study in Japan. January 2009 to December 2011. | Women with PD N = 10 Control Group N = 1014 | Women diagnosed by psychiatrist using ICD10. Women with personality and behavioral disturbance. | PD N = 3 CG N = 90 OR 4.40 (CI 1.12–17.31) | PD N = 0 CG N = 2 OR 19.29 (CI 0.87–426.67) | PD N = 4 CG N = 113 OR 5.32 (CI 1.48–19.12) | ||
| Maternal age Data not provided | ||||||||
| Ethnicity Data not provided | ||||||||
| Alcohol/Drug Use Data not provided | ||||||||
| Ferarro et al. 2017 Sao Paulo, Brazil [26] | Prospective cohort study. Diagnostic interviews to determine whether any had a mental illness. July 2010 to December 2012. | Women with PD N = 17 Control Group N = 758 | ICD 10 coding specifically for antisocial personality disorder. Trained psychologists used the Mini International Neuropsych Interview (MINI). | PD N = 4 CG N = 50 OR 4.36 (CI 1.37–13.85) | PD N = 0 CG N = 6 OR 3.31 (CI 0.18–61.04) | PD N = 3 CG N = 43 OR 3.56 (CI 0.99–12.87) | ||
| Maternal age Data not provided | ||||||||
| Ethnicity Data not provided | ||||||||
| Alcohol/Drug Use Data not provided | ||||||||
| Baer et al. 2016 California, US [60] | Retrospective cohort study of births from 2007 to 2011 in the birth cohort registry. Restricted to women with medical insurance. | Women with PD N = 56 Control Group N = 24,792 | Women identified with ICD codes 9th edition on their hospital discharge records. | PD N = 13 CG N = 1454 OR 4.85 (CI 2.60–9.04) | Does not report | Does not report | ||
| Maternal age Data not provided | ||||||||
| Ethnicity Data not provided | ||||||||
| Alcohol/Drug Use Data not provided | ||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marshall, C.A.; Jomeen, J.; Huang, C.; Martin, C.R. The Relationship between Maternal Personality Disorder and Early Birth Outcomes: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2020, 17, 5778. https://doi.org/10.3390/ijerph17165778
Marshall CA, Jomeen J, Huang C, Martin CR. The Relationship between Maternal Personality Disorder and Early Birth Outcomes: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health. 2020; 17(16):5778. https://doi.org/10.3390/ijerph17165778
Chicago/Turabian StyleMarshall, Claire A., Julie Jomeen, Chao Huang, and Colin R. Martin. 2020. "The Relationship between Maternal Personality Disorder and Early Birth Outcomes: A Systematic Review and Meta-Analysis" International Journal of Environmental Research and Public Health 17, no. 16: 5778. https://doi.org/10.3390/ijerph17165778
APA StyleMarshall, C. A., Jomeen, J., Huang, C., & Martin, C. R. (2020). The Relationship between Maternal Personality Disorder and Early Birth Outcomes: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health, 17(16), 5778. https://doi.org/10.3390/ijerph17165778





