Winter Is Coming: A Southern Hemisphere Perspective of the Environmental Drivers of SARS-CoV-2 and the Potential Seasonality of COVID-19
Abstract
1. Introduction
2. Why the Southern Hemisphere Is Different
3. Monitoring and Modelling the Spread of COVID-19
3.1. Data Issues
3.2. Epidemiological Models of COVID-19
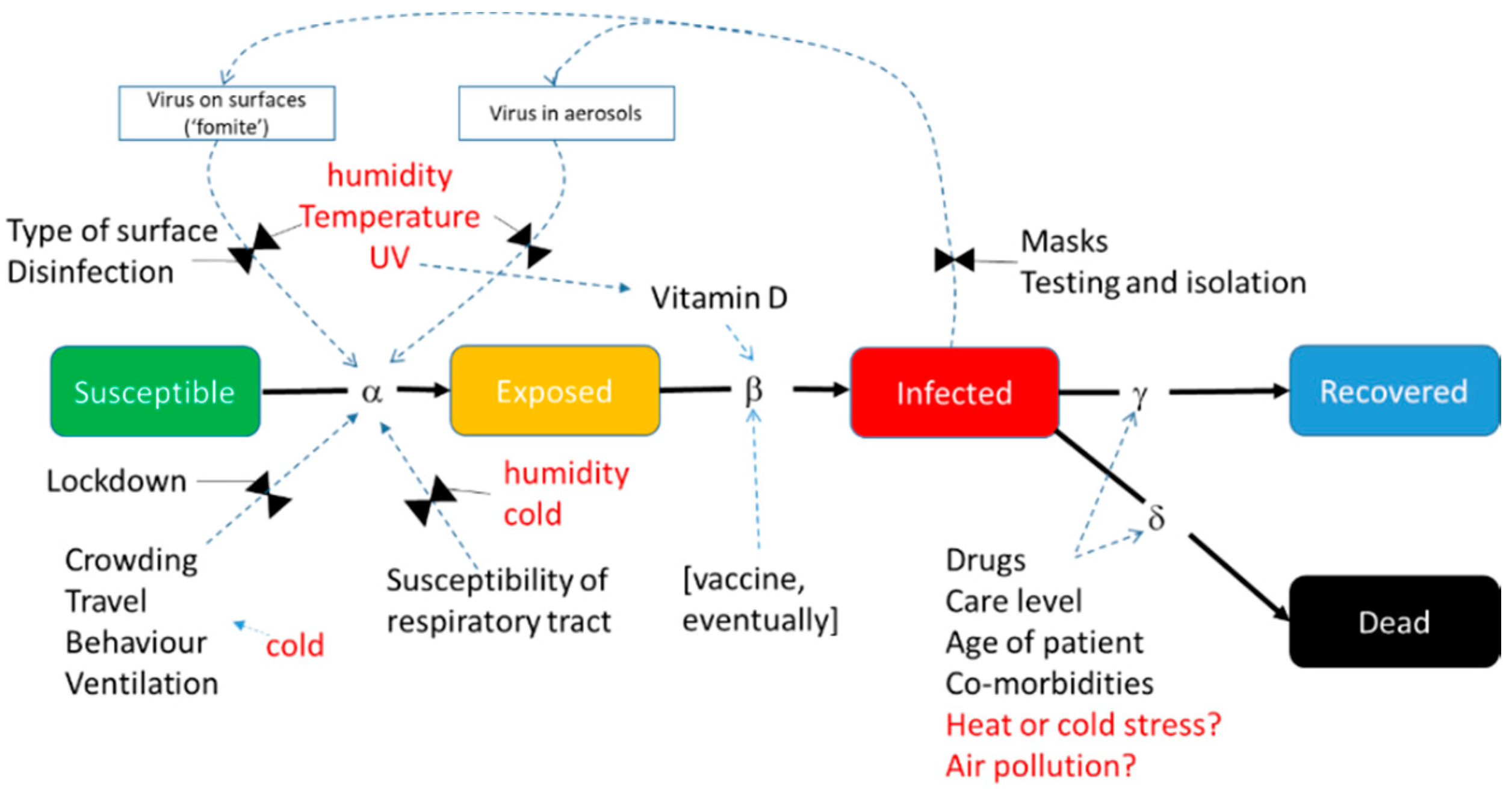
3.3. Incorporating Environmental Drivers into Epidemiological Models
4. Implications for COVID-19 of Environmental Sensitivity in Other Viral Respiratory Diseases
5. Critical Assessment of Studies of COVID-19 Climate Susceptibility
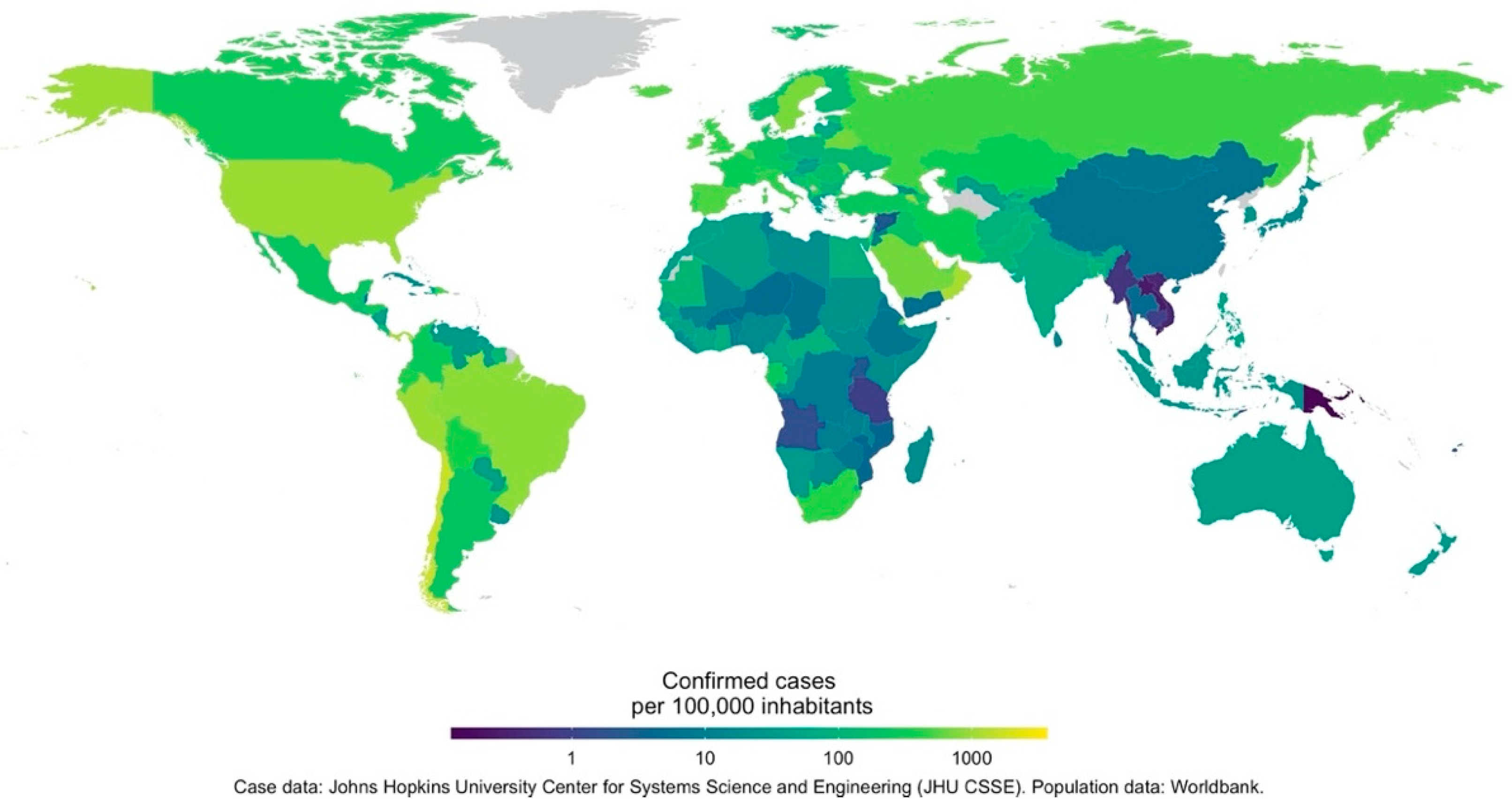
5.1. Geographical Coverage of Studies
5.2. Influential Variables
5.3. Dependent Variables
5.4. Modelling Approaches
5.5. Findings
6. Discussion
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, C.; Horby, P.W.; Hayden, F.G.; Gao, G.F. A novel coronavirus outbreak of global health concern. Lancet 2020, 395, 470–473. [Google Scholar] [CrossRef]
- Nicola, M.; Alsafi, Z.; Sohrabi, C.; Kerwan, A.; Al-Jabir, A.; Iosifidis, C.; Agha, M.; Agha, R. The socio-economic implications of the coronavirus pandemic (COVID-19): A review. Int. J. Surg. 2020, 78, 185–193. [Google Scholar] [CrossRef]
- Kickbusch, I.; Leung, G.M.; Bhutta, Z.A.; Matsoso, M.P.; Ihekweazu, C.; Abbasi, K. Covid-19: How a virus is turning the world upside down. Br. Med. J. 2020, 369. [Google Scholar] [CrossRef]
- Mahler, D.G.; Lakner, C.; Aguilar, R.A.C.; Wu, H. The Impact of COVID-19 (Coronavirus) on Global Poverty: Why Sub-Saharan Africa Might be the Region Hardest Hit; World Bank: Washington, DC, USA, 2020. [Google Scholar]
- Tanne, J.H.; Hayasaki, E.; Zastrow, M.; Pulla, P.; Smith, P.; Rada, A.G. Covid-19: How doctors and healthcare systems are tackling coronavirus worldwide. Br. Med. J. 2020, 368. [Google Scholar] [CrossRef]
- Coronavirus Government Response Tracker. Available online: https://www.bsg.ox.ac.uk/research/research-projects/coronavirus-government-response-tracker (accessed on 15 May 2020).
- Shaman, J.; Kohn, M. Absolute humidity modulates influenza survival, transmission, and seasonality. Proc. Natl. Acad. Sci. USA 2009, 106, 3243–3248. [Google Scholar] [CrossRef]
- Shaman, J.; Pitzer, V.E.; Viboud, C.; Grenfell, B.T.; Lipsitch, M. Absolute humidity and the seasonal onset of influenza in the continental United States. PLoS Boil. 2010, 8, e1000316. [Google Scholar] [CrossRef]
- Xu, S.; Li, Y. Beware of the second wave of COVID-19. Lancet 2020, 395, 1321–1322. [Google Scholar] [CrossRef]
- Nassar, M.S.; Bakhrebah, M.A.; Meo, S.A.; Alsuabeyl, M.S.; Zaher, W.A. Global seasonal occurrence of middle east respiratory syndrome coronavirus (MERS-CoV) infection. Eur. Rev. Med Pharmacol. Sci. 2018, 22, 3913–3918. [Google Scholar] [PubMed]
- Carleton, T.; Meng, K.C. Causal Empirical Estimates Suggest COVID-19 Transmission Rates Are Highly Seasonal. Available online: https://github.com/emlan-ucsb/COVID-seasonality (accessed on 2 April 2020).
- Sajadi, M.M.; Habibzadeh, P.; Vintzileos, A.; Shokouhi, S.; Miralles-Wilhelm, F.; Amoroso, A. Temperature, humidity, and latitude analysis to estimate potential spread and seasonality of coronavirus disease 2019 (COVID-19). JAMA Netw. Open 2020, 3, e2011834. [Google Scholar] [CrossRef] [PubMed]
- Healy, J. Analysing health care systems performance: The story behind the statistics. Aust. N. Z. J. Public Health 2003, 27, 642–644. [Google Scholar] [CrossRef] [PubMed]
- Hirve, S.; Newman, L.P.; Paget, J.; Azziz-Baumgartner, E.; Fitzner, J.; Bhat, N.; Vandemaele, K.; Zhang, W. Influenza seasonality in the tropics and subtropics—When to vaccinate? PLoS ONE 2016, 11, e0153003. [Google Scholar] [CrossRef] [PubMed]
- Fieno, J.; Lloyd-Sherlock, P. Healthcare Reform and Poverty in Latin America; University of London: London, UK, 2002. [Google Scholar]
- Naicker, S.; Plange-Rhule, J.; Tutt, R.C.; Eastwood, J.B. Shortage of healthcare workers in developing countries--Africa. Ethn. Dis. 2009, 19, 60. [Google Scholar]
- Atun, R.; De Andrade, L.O.M.; Almeida, G.; Cotlear, D.; Dmytraczenko, T.; Frenz, P.; García, P.; Gómez-Dantés, O.; Knaul, F.M.; Muntaner, C.; et al. Health-system reform and universal health coverage in Latin America. Lancet 2015, 385, 1230–1247. [Google Scholar] [CrossRef]
- Hampshire, K.; Porter, G.; Owusu, S.A.; Mariwah, S.; Abane, A.; Robson, E.; Munthali, A.; Delannoy, A.; Bango, A.; Gunguluza, N.; et al. Informal m-health: How are young people using mobile phones to bridge healthcare gaps in Sub-Saharan Africa? Soc. Sci. Med. 2015, 142, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Kapata, N.; Ihekweazu, C.; Ntoumi, F.; Raji, T.; Chanda-Kapata, P.; Mwaba, P.; Mukonka, V.; Bates, M.; Tembo, J.; Corman, V.M.; et al. Is Africa prepared for tackling the COVID-19 (SARS-CoV-2) epidemic. Lessons from past outbreaks, ongoing pan-African public health efforts, and implications for the future. Int. J. Infect. Dis. 2020, 93, 233–236. [Google Scholar] [CrossRef]
- Gilbert, M.; Pullano, G.; Pinotti, F.; Valdano, E.; Poletto, C.; Boëlle, P.-Y.; D’Ortenzio, E.; Yazdanpanah, Y.; Eholie, S.P.; Altmann, M.; et al. Preparedness and vulnerability of African countries against importations of COVID-19: A modelling study. Lancet 2020, 395, 871–877. [Google Scholar] [CrossRef]
- Velavan, T.P.; Meyer, C.G. The COVID-19 epidemic. Trop. Med. Int. Health 2020, 25, 278. [Google Scholar] [CrossRef]
- Ji, Y.; Ma, Z.; Peppelenbosch, M.P.; Pan, Q. Potential association between COVID-19 mortality and health-care resource availability. Lancet Glob. Health 2020, 8, e480. [Google Scholar] [CrossRef]
- Talisuna, A.; Okiro, E.A.; Yahaya, A.A.; Stephen, M.; Bonkoungou, B.; Musa, E.O.; Minkoulou, E.M.; Okeibunor, J.; Impouma, B.; Djingarey, H.M.; et al. Spatial and temporal distribution of infectious disease epidemics, disasters and other potential public health emergencies in the World Health Organisation Africa region, 2016–2018. Glob. Health 2020, 16, 9–12. [Google Scholar] [CrossRef]
- Rodriguez-Morales, A.J.; Gallego, V.; Escalera-Antezana, J.P.; Mendez, C.A.; Zambrano, L.I.; Franco-Paredes, C.; Suárez, J.A.; Rodriguez-Enciso, H.D.; Balbin-Ramon, G.J.; Savio-Larriera, E.; et al. COVID-19 in Latin America: The implications of the first confirmed case in Brazil. Travel Med. Infect. Dis. 2020, 35, 101613. [Google Scholar] [CrossRef]
- Steingart, K.R.; Sohn, H.; Schiller, I.A.; Kloda, L.; Boehme, C.C.; Pai, M.; Dendukuri, N. Xpert® MTB/RIF assay for pulmonary tuberculosis and rifampicin resistance in adults. Cochrane Database Syst. Rev. 2013, 1. [Google Scholar] [CrossRef]
- Gelaw, Y.A.; Williams, G.M.; Magalhães, R.J.S.; Gilks, C.F.; Assefa, Y. HIV prevalence among tuberculosis patients in sub-Saharan Africa: A systematic review and meta-analysis. AIDS Behav. 2019, 23, 1561–1575. [Google Scholar] [CrossRef] [PubMed]
- Grant, A.; Charalambous, S.; Fielding, K.; Day, J.H.; Corbett, E.L.; Chaisson, R.E.; De Cock, K.M.; Hayes, R.; Churchyard, G. Effect of routine isoniazid preventive therapy on tuberculosis incidence among HIV-Infected men in South Africa. J. Am. Med. Assoc. 2005, 293, 2719–2725. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Corbett, E.L.; Marston, B.; Churchyard, G.J.; De Cock, K.M. Tuberculosis in sub-Saharan Africa: Opportunities, challenges, and change in the era of antiretroviral treatment. Lancet 2006, 367, 926–937. [Google Scholar] [CrossRef]
- Adenis, A.; Valdes, A.; Cropet, C.; McCotter, O.Z.; Derado, G.; Couppié, P.; Chiller, T.; Nacher, M. Burden of HIV-associated histoplasmosis compared with tuberculosis in Latin America: A modelling study. Lancet Infect. Dis. 2018, 18, 1150–1159. [Google Scholar] [CrossRef]
- Blanco, J.L.; Ambrosioni, J.; Garcia, F.; Martínez, E.; Soriano, A.; Mallolas, J.; Miró, J.M. COVID-19 in HIV Investigators COVID-19 in patients with HIV: Clinical case series. Lancet HIV 2020, 7, e314–e316. [Google Scholar] [CrossRef]
- Jiang, H.; Zhou, Y.; Tang, W. Maintaining HIV care during the COVID-19 pandemic. Lancet HIV 2020, 7, e308–e309. [Google Scholar] [CrossRef]
- Pang, Y.; Liu, Y.; Du, J.; Gao, J.; Li, L. Impact of COVID-19 on tuberculosis control in China. Int. J. Tuberc. Lung Dis. 2020, 24, 545–547. [Google Scholar] [CrossRef]
- Preiser, W.; Van Zyl, G.; Dramowski, A. COVID-19: Getting ahead of the epidemic curve by early implementation of social distancing. S. Afr. Med. J. 2020, 110, 258. [Google Scholar] [CrossRef]
- Liu, Y.; Yan, L.-M.; Wan, L.; Xiang, T.-X.; Le, A.; Liu, J.-M.; Peiris, M.; Poon, L.L.; Zhang, W. Viral dynamics in mild and severe cases of COVID-19. Lancet Infect. Dis. 2020, 20, 656–657. [Google Scholar] [CrossRef]
- Patel, R.; Babady, N.E.; Theel, E.S.; Storch, G.A.; Pinsky, B.A.; George, K.S.; Smith, T.C.; Bertuzzi, S. Report from the American Society for Microbiology COVID-19 International Summit, 23 March 2020: Value of Diagnostic Testing for SARS–CoV-2/COVID-19. mBio 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Ye, F.; Xu, S.; Rong, Z.; Xu, R.; Liu, X.; Deng, P.; Liu, H.; Xu, X. Delivery of infection from asymptomatic carriers of COVID-19 in a familial cluster. Int. J. Infect. Dis. 2020, 94, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Grifoni, A.; Weiskopf, D.; Ramirez, S.I.; Mateus, J.; Dan, J.M.; Moderbacher, C.R.; Rawlings, S.A.; Sutherland, A.; Premkumar, L.; Jadi, R.S.; et al. Targets of T cell responses to SARS-CoV-2 coronavirus in humans with COVID-19 disease and unexposed individuals. Cell 2020, 181, 1489–1501.e15. [Google Scholar] [CrossRef] [PubMed]
- Leon, D.A.; Shkolnikov, V.M.; Smeeth, L.; Magnus, P.; Pechholdová, M.; Jarvis, C.I. COVID-19: A need for real-time monitoring of weekly excess deaths. Lancet 2020, 395, e81. [Google Scholar] [CrossRef]
- Yao, Y.; Pan, J.; Liu, Z.; Meng, X.; Wang, W.; Kan, H.; Wang, W. No association of COVID-19 transmission with temperature or UV radiation in Chinese cities. Eur. Respir. J. 2020, 55, 2000517. [Google Scholar] [CrossRef] [PubMed]
- Şahin, M. Impact of weather on COVID-19 pandemic in Turkey. Sci. Total Environ. 2020, 728, 138810. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Zhu, Y. Association between ambient temperature and COVID-19 infection in 122 cities from China. Sci. Total Environ. 2020, 724, 138201. [Google Scholar] [CrossRef]
- Heesterbeek, J.A.P.; Anderson, R.; Andreasen, V.; Bansal, S.; De Angelis, D.; Dye, C.; Eames, K.T.D.; Edmunds, W.J.; Frost, S.D.; Funk, S.; et al. Modeling infectious disease dynamics in the complex landscape of global health. Science 2015, 347, aaa4339. [Google Scholar] [CrossRef]
- Systrom, K.; Vladeck, T. Rt COVID-19. Available online: https://rt.live/ (accessed on 8 July 2020).
- Della Morte, M.; Orlando, D.; Sannino, F. Renormalization group approach to pandemics: The COVID-19 case. Front. Phys. 2020, 8. [Google Scholar] [CrossRef]
- Roosa, K.; Lee, Y.; Luo, R.; Kirpich, A.; Rothenberg, R.; Hyman, J.; Yan, P.; Chowell, G. Real-time forecasts of the COVID-19 epidemic in China from February 5th to February 24th, 2020. Infect. Dis. Model. 2020, 5, 256–263. [Google Scholar] [CrossRef]
- Anastassopoulou, C.; Russo, L.; Tsakris, A.; Siettos, C. Data-based analysis, modelling and forecasting of the COVID-19 outbreak. PLoS ONE 2020, 15, e0230405. [Google Scholar] [CrossRef] [PubMed]
- Ardabili, S.F.; Mosavi, A.; Ghamisi, P.; Ferdinand, F.; Varkonyi-Koczy, A.R.; Reuter, U.; Rabczuk, T.; Atkinson, P.M. COVID-19 outbreak prediction with machine learning. medRxiv 2020. [Google Scholar] [CrossRef]
- Pinter, G.; Felde, I.; Mosavi, A.; Ghamisi, P.; Gloaguen, R. COVID-19 pandemic prediction for Hungary; a hybrid machine learning approach. Mathematics 2020, 8, 890. [Google Scholar] [CrossRef]
- Cuevas, E. An agent-based model to evaluate the COVID-19 transmission risks in facilities. Comput. Boil. Med. 2020, 121, 103827. [Google Scholar] [CrossRef] [PubMed]
- Shi, P.; Dong, Y.; Yan, H.; Li, X.; Zhao, C.; Liu, W.; He, M.; Tang, S.; Xi, S. The impact of temperature and absolute humidity on the coronavirus disease 2019 (COVID-19) outbreak-evidence from China. medRxiv 2020. [Google Scholar] [CrossRef]
- Neher, R.A.; Dyrdak, R.; Druelle, V.; Hodcroft, E.B.; Albert, J. Potential impact of seasonal forcing on a SARS-CoV-2 pandemic. Swiss Med. Wkly. 2020, 150, w20224. [Google Scholar] [CrossRef]
- Flaxman, S.; Mishra, S.; Gandy, A.; Unwin, H.; Coupland, H.; Mellan, T.; Zhu, H.; Berah, T.; Eaton, J.; Perez Guzman, P.; et al. Estimating the Number of Infections and the Impact of Nonpharmaceutical Interventions on COVID-19 in 11 European Countries; Imperial College London: London, UK, 2020. [Google Scholar] [CrossRef]
- Lofgren, E.T.; Fefferman, N.H.; Naumov, Y.N.; Górski, J.; Naumova, E.N. Influenza seasonality: Underlying causes and modeling theories. J. Virol. 2006, 81, 5429–5436. [Google Scholar] [CrossRef]
- Du Prel, J.-B.; Puppe, W.; Gröndahl, B.; Knuf, M.; Weigl, J.A.I.; Schaaff, F.; Schmitt, H.-J. Are meteorological parameters associated with acute respiratory tract Infections? Clin. Infect. Dis. 2009, 49, 861–868. [Google Scholar] [CrossRef]
- Stewart, P.D.S. Seasonality and selective trends in viral acute respiratory tract infections. Med. Hypotheses 2016, 86, 104–119. [Google Scholar] [CrossRef]
- Drexler, J.F.; Corman, V.M.; Drosten, C. Ecology, evolution and classification of bat coronaviruses in the aftermath of SARS. Antivir. Res. 2014, 101, 45–56. [Google Scholar] [CrossRef]
- Killerby, M.E.; Biggs, H.M.; Haynes, A.; Dahl, R.M.; Mustaquim, D.; Gerber, S.I.; Watson, J.T. Human coronavirus circulation in the United States 2014–2017. J. Clin. Virol. 2018, 101, 52–56. [Google Scholar] [CrossRef] [PubMed]
- Price, R.H.M.; Graham, C.; Ramalingam, S. Association between viral seasonality and meteorological factors. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Sick time. Science 2020, 367, 1294–1297. [Google Scholar] [CrossRef] [PubMed]
- Moriyama, M.; Hugentobler, W.J.; Iwasaki, A. Seasonality of respiratory viral infections. Annu. Rev. Virol. 2020, 7. [Google Scholar] [CrossRef]
- Martinez, M.E. The calendar of epidemics: Seasonal cycles of infectious diseases. PLoS Pathog. 2018, 14, e1007327. [Google Scholar] [CrossRef] [PubMed]
- Fisman, D.N. Seasonality of viral infections: Mechanisms and unknowns. Clin. Microbiol. Infect. 2012, 18, 946–954. [Google Scholar] [CrossRef]
- Viboud, C.; Alonso, W.J.; Simonsen, L. Influenza in tropical regions. PLoS Med. 2006, 3, e89. [Google Scholar] [CrossRef]
- Saunders-Hastings, P.; Krewski, D. Reviewing the history of pandemic influenza: Understanding patterns of emergence and transmission. Pathogens 2016, 5, 66. [Google Scholar] [CrossRef]
- Al-Tawfiq, J.A.; Memish, Z.A. Drivers of MERS-CoV transmission: What do we know? Expert Rev. Respir. Med. 2016, 10, 331–338. [Google Scholar] [CrossRef]
- Skowronski, D.M.; Astell, C.; Brunham, R.C.; Low, D.E.; Petric, M.; Roper, R.L.; Talbot, P.J.; Tam, T.; Babiuk, L. Severe Acute Respiratory Syndrome (SARS): A Year in Review. Annu. Rev. Med. 2005, 56, 357–381. [Google Scholar] [CrossRef]
- Dowell, S.F.; Ho, M.S. Seasonality of infectious diseases and severe acute respiratory syndrome–what we don’t know can hurt us. Lancet Infect. Dis. 2004, 4, 704–708. [Google Scholar] [CrossRef]
- Kissler, S.; Tedijanto, C.; Goldstein, E.; Grad, Y.H.; Lipsitch, M. Projecting the transmission dynamics of SARS-CoV-2 through the postpandemic period. Science 2020, 368, 860–868. [Google Scholar] [CrossRef] [PubMed]
- Moore, K.A.; Lipsitch, M.; Barry, J.M.; Osterholm, M.T. Part 1: The Future of the COVID-19 Pandemic: Lessons Learned from Pandemic Influenza. COVID-19; The CIDRAP Viewpoint Working Group, University of Minnesota: Minneapolis, MN, USA, 2020. [Google Scholar]
- Chan, K.H.; Peiris, J.S.M.; Lam, S.Y.; Poon, L.L.; Yuen, K.-Y.; Seto, W.H. The Effects of temperature and Relative humidity on the viability of the SARS coronavirus. Adv. Virol. 2011, 2011, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Tellier, R. Aerosol transmission of influenza a virus: A review of new studies. J. R. Soc. Interface 2009, 6. [Google Scholar] [CrossRef]
- Lowen, A.C.; Mubareka, S.; Steel, J.; Palese, P. Influenza virus transmission is dependent on relative humidity and temperature. PLOS Pathog. 2007, 3, e151. [Google Scholar] [CrossRef]
- Lowen, A.; Palese, P. Transmission of influenza virus in temperate zones is predominantly by aerosol, in the tropics by contact: A hypothesis. PLoS Curr. 2009. [Google Scholar] [CrossRef]
- Tamerius, J.D.; Shaman, J.; Alonso, W.J.; Bloom-Feshbach, K.; Uejio, C.K.; Comrie, A.; Viboud, C. Environmental predictors of seasonal influenza epidemics across temperate and tropical climates. PLoS Pathog. 2013, 9, e1003194. [Google Scholar] [CrossRef]
- Baker, R.E.; Mahmud, A.S.; Metcalf, C.J.E. Dynamic response of airborne infections to climate change: Predictions for varicella. Clim. Chang. 2018, 148, 547–560. [Google Scholar] [CrossRef]
- Bourouiba, L. A Sneeze. N. Engl. J. Med. 2016, 375, e15. [Google Scholar] [CrossRef]
- Yang, W.; Elankumaran, S.; Marr, L.C. Relationship between humidity and influenza a viability in droplets and implications for influenza’s seasonality. PLOS ONE 2012, 7, e46789. [Google Scholar] [CrossRef]
- Cole, E.C.; Cook, C.E. Characterization of infectious aerosols in health care facilities: An aid to effective engineering controls and preventive strategies. Am. J. Infect. Control. 1998, 26, 453–464. [Google Scholar] [CrossRef]
- WHO. WHO Guidelines Approved by the Guidelines Review Committee. In Natural Ventilation for Infection Control in Health-Care Settings; Atkinson, J., Chartier, Y., Pessoa-Silva, C.L., Jensen, P., Li, Y., Seto, W.H., Eds.; World Health Organization: Geneva, Switzerland, 2009. [Google Scholar]
- Liu, L.; Wei, J.; Li, Y.; Ooi, A. Evaporation and dispersion of respiratory droplets from coughing. Indoor Air 2016, 27, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Tellier, R.; Li, Y.; Cowling, B.J.; Tang, J.W. Recognition of aerosol transmission of infectious agents: A commentary. BMC Infect. Dis. 2019, 19, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Lowen, A.C.; Steel, J. Roles of humidity and temperature in shaping influenza seasonality. J. Virol. 2014, 88, 7692–7695. [Google Scholar] [CrossRef]
- Zhang, Y.; Bambrick, H.; Mengersen, K.; Tong, S.; Hu, W. Using google trends and ambient temperature to predict seasonal influenza outbreaks. Environ. Int. 2018, 117, 284–291. [Google Scholar] [CrossRef]
- Nielsen, J.; Mazick, A.; Glismann, S.; Mølbak, K. Excess mortality related to seasonal influenza and extreme temperatures in Denmark, 1994-2010. BMC Infect. Dis. 2011, 11, 350. [Google Scholar] [CrossRef]
- Jaakkola, K.; Saukkoriipi, A.; Jokelainen, J.; Juvonen, R.; Kauppila, J.; Vainio, O.; Ziegler, T.; Rönkkö, E.; Jaakkola, J.J.K. Decline in temperature and humidity increases the occurrence of influenza in cold climate. Environ. Health 2014, 13, 22. [Google Scholar] [CrossRef]
- Rabenau, H.; Cinatl, J.; Morgenstern, B.; Bauer, G.; Preiser, W.; Doerr, H.W. Stability and inactivation of SARS coronavirus. Med. Microbiol. Immunol. 2004, 194, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Sauerbrei, A.; Wutzler, P. Testing thermal resistance of viruses. Arch. Virol. 2008, 154, 115–119. [Google Scholar] [CrossRef]
- Bean, B.; Moore, B.M.; Sterner, B.; Peterson, L.R.; Gerding, D.N.; Balfour, H.H. Survival of influenza viruses on environmental surfaces. J. Infect. Dis. 1982, 146, 47–51. [Google Scholar] [CrossRef]
- Boone, S.A.; Gerba, C.P. Significance of fomites in the spread of respiratory and enteric viral disease. Appl. Environ. Microbiol. 2007, 73, 1687–1696. [Google Scholar] [CrossRef]
- Casanova, L.M.; Jeon, S.; Rutala, W.A.; Weber, D.J.; Sobsey, M.D. Effects of air temperature and relative humidity on coronavirus survival on surfaces. Appl. Environ. Microbiol. 2010, 76, 2712–2717. [Google Scholar] [CrossRef]
- Kampf, G.; Todt, D.; Pfaender, S.; Steinmann, E. Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents. J. Hosp. Infect. 2020, 104, 246–251. [Google Scholar] [CrossRef]
- McDevitt, J.; Rudnick, S.; First, M.; Spengler, J. Role of absolute humidity in the inactivation of influenza viruses on stainless steel surfaces at elevated temperatures. Appl. Environ. Microbiol. 2010, 76, 3943–3947. [Google Scholar] [CrossRef]
- Geller, C.; Varbanov, M.; Duval, R.E. Human coronaviruses: Insights into environmental resistance and its influence on the development of new antiseptic strategies. Viruses 2012, 4, 3044–3068. [Google Scholar] [CrossRef] [PubMed]
- Sizun, J.; Yu, M.; Talbot, P. Survival of human coronaviruses 229E and OC43 in suspension and after drying onsurfaces: A possible source of hospital-acquired infections. J. Hosp. Infect. 2000, 46, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Otter, J.; Donskey, C.; Yezli, S.; Douthwaite, S.; Goldenberg, S.D.; Weber, D. Transmission of SARS and MERS coronaviruses and influenza virus in healthcare settings: The possible role of dry surface contamination. J. Hosp. Infect. 2016, 92, 235–250. [Google Scholar] [CrossRef] [PubMed]
- Kramer, A.; Schwebke, I.; Kampf, G. How long do nosocomial pathogens persist on inanimate surfaces? A systematic review. BMC Infect. Dis. 2006, 6, 130. [Google Scholar] [CrossRef] [PubMed]
- Lowen, A.C.; Steel, J.; Mubareka, S.; Palese, P. High temperature (30 °C) blocks aerosol but not contact transmission of influenza virus. J. Virol. 2008, 82, 5650–5652. [Google Scholar] [CrossRef] [PubMed]
- Ellwanger, J.H.; Chies, J.A.B. Wind: A neglected factor in the spread of infectious diseases. Lancet Planet. Health 2018, 2, e475. [Google Scholar] [CrossRef]
- Peci, A.; Winter, A.-L.; Li, Y.; Gnaneshan, S.; Liu, J.; Mubareka, S.; Gubbay, J.B. Effects of absolute humidity, relative humidity, temperature, and wind speed on influenza activity in Toronto, Ontario, Canada. Appl. Environ. Microbiol. 2019, 85, e02426-18. [Google Scholar] [CrossRef] [PubMed]
- Firestone, S.M.; Cogger, N.; Ward, M.P.; Toribio, J.-A.L.M.L.; Moloney, B.; Dhand, N.K. The influence of meteorology on the spread of influenza: Survival analysis of an equine influenza (A/H3N8) Outbreak. PLoS ONE 2012, 7, e35284. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, S.; Piedimonte, G.; Auais, A.; Demmler, G.; Krishnan, S.; Van Caeseele, P.; Singleton, R.; Broor, S.; Parveen, S.; Avendaño, L.; et al. The relationship of meteorological conditions to the epidemic activity of respiratory syncytial virus. Epidemiol. Infect. 2007, 135, 1077–1090. [Google Scholar] [CrossRef] [PubMed]
- Hervás, D.; Reina, J.; Hervás, J.A. Meteorologic conditions and respiratory syncytial virus activity. Pediatr. Infect. Dis. J. 2012, 31. [Google Scholar] [CrossRef]
- Guo, Q.; Dong, Z.; Zeng, W.; Ma, W.; Zhao, D.; Sun, X.; Gong, S.; Xiao, J.; Li, T.; Hu, W. The effects of meteorological factors on influenza among children in Guangzhou, China. Influenza Other Respir. Viruses 2018, 13, 166–175. [Google Scholar] [CrossRef]
- Adler, R.F.; Gu, G.; Sapiano, M.; Wang, J.J.; Huffman, G.J. Global precipitation: Means, variations and trends during the satellite era (1979–2014). Surv. Geophys. 2017, 38, 679–699. [Google Scholar] [CrossRef]
- Pica, N.; Bouvier, N.M. Environmental factors affecting the transmission of respiratory viruses. Curr. Opin. Virol. 2012, 2, 90–95. [Google Scholar] [CrossRef]
- Murray, E.L.; Klein, M.; Brondi, L.; McGowan, J.E.; Van Mels, C.; Brooks, W.A.; Kleinbaum, D.; Goswami, D.; Ryan, P.B.; Bridges, C.B. Rainfall, household crowding, and acute respiratory infections in the tropics. Epidemiol. Infect. 2011, 140, 78–86. [Google Scholar] [CrossRef]
- Paynter, S.; Weinstein, P.; Ware, R.S.; Lucero, M.G.; Tallo, V.; Nohynek, H.; Barnett, A.; Skelly, C.; Simoes, E.A.F.; Sly, P.D.; et al. Sunshine, rainfall, humidity and child pneumonia in the tropics: Time-series analyses. Epidemiol. Infect. 2012, 141, 1328–1336. [Google Scholar] [CrossRef]
- Chew, F.T.; Doraisingham, S.; Ling, A.E.; Kumarasinghe, G.; Lee, B.W. Seasonal trends of viral respiratory tract infections in the tropics. Epidemiol. Infect. 1998, 121, 121–128. [Google Scholar] [CrossRef]
- Idani, E.; Dastoorpoor, M.; Goudarzi, G.; Khanjani, N. Severe outbreaks of respiratory syndromes following autumn rainfall in Khuzestan, Iran. Arch. Iran. Med. 2016, 19, 884–885. [Google Scholar] [PubMed]
- Duan, S.-M.; Zhao, X.-S.; Wen, R.-F.; Huang, J.-J.; Pi, G.-H.; Zhang, S.-X.; Han, J.; Bi, S.-L.; Ruan, L.; Dong, X.-P. Stability of SARS coronavirus in human specimens and environment and its sensitivity to heating and UV irradiation. Biomed. Environ. Sci. 2003, 16, 246–255. [Google Scholar] [PubMed]
- Darnell, M.E.; Subbarao, K.; Feinstone, S.M.; Taylor, D.R. Inactivation of the coronavirus that induces severe acute respiratory syndrome, SARS-CoV. J. Virol. Methods 2004, 121, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Bedell, K.; Buchaklian, A.H.; Perlman, S. Efficacy of an automated multiple emitter whole-room ultraviolet-C disinfection system against coronaviruses MHV and MERS-CoV. Infect. Control. Hosp. Epidemiol. 2016, 37, 598–599. [Google Scholar] [CrossRef]
- Sagripanti, J.-L.; Lytle, C.D. Inactivation of influenza virus by solar radiation. Photochem. Photobiol. 2007, 83, 1278–1282. [Google Scholar] [CrossRef]
- Weber, T.P.; Stilianakis, N.I. Inactivation of influenza a viruses in the environment and modes of transmission: A critical review. J. Infect. 2008, 57, 361–373. [Google Scholar] [CrossRef]
- Eccles, R. An Explanation for the seasonality of acute upper respiratory tract viral infections. Acta Oto-Laryngol. 2002, 122, 183–191. [Google Scholar] [CrossRef]
- Kudo, E.; Song, E.; Yockey, L.J.; Rakib, T.; Wong, P.W.; Homer, R.J.; Iwasaki, A. Low ambient humidity impairs barrier function and innate resistance against influenza infection. Proc. Natl. Acad. Sci. USA 2019, 116, 10905–10910. [Google Scholar] [CrossRef]
- Cannell, J.; Vieth, R.; Umhau, J.C.; Holick, M.F.; Grant, W.B.; Madronich, S.; Garland, C.F.; Giovannucci, E. Epidemic influenza and vitamin D. Epidemiol. Infect. 2006, 134, 1129–1140. [Google Scholar] [CrossRef]
- Liu, P.T.; Stenger, S.; Li, H.; Wenzel, L.; Tan, B.H.; Krutzik, S.R.; Ochoa, M.T.; Schauber, J.; Wu, K.; Meinken, C.; et al. Toll-Like receptor triggering of a vitamin D-mediated human antimicrobial response. Science 2006, 311, 1770–1773. [Google Scholar] [CrossRef]
- Moan, J.E.; Dahlback, A.; Ma, L.; Juzeniene, A. Influenza, solar radiation and vitamin D. Dermat-Endocrinology 2009, 1, 307–309. [Google Scholar] [CrossRef] [PubMed]
- Yamshchikov, A.V.; Desai, N.S.; Blumberg, H.M.; Ziegler, T.R.; Tangpricha, V. Vitamin D for treatment and prevention of infectious diseases: A systematic review of randomized controlled Trials. Endocr. Pract. 2009, 15, 438–449. [Google Scholar] [CrossRef] [PubMed]
- Shaman, J.; Jeon, C.Y.; Giovannucci, E.; Lipsitch, M. Shortcomings of vitamin D-based model simulations of seasonal influenza. PLoS ONE 2011, 6, e20743. [Google Scholar] [CrossRef]
- Ciencewicki, J.; Jaspers, I. Air pollution and respiratory viral infection, inhalation. Toxicology 2007, 19, 1135–1146. [Google Scholar] [CrossRef]
- Schwartz, J. Air pollution and daily mortality: A review and meta analysis. Environ. Res. 1994, 64, 36–52. [Google Scholar] [CrossRef]
- Clay, K.; Lewis, J.; Severnini, E. Pollution, infectious disease, and mortality: Evidence from the 1918 Spanish influenza pandemic. J. Econ. Hist. 2018, 78, 1179–1209. [Google Scholar] [CrossRef]
- Clifford, H.D.; Perks, K.L.; Zosky, G.R. Geogenic PM10 exposure exacerbates responses to influenza infection. Sci. Total Environ. 2015, 533, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Ye, Q.; Fu, J.-F.; Mao, J.H.; Shang, S.-Q. Haze is a risk factor contributing to the rapid spread of respiratory syncytial virus in children. Environ. Sci. Pollut. Res. 2016, 23, 20178–20185. [Google Scholar] [CrossRef]
- Peng, R.D.; Dominici, F.; Pastor-Barriuso, R.; Zeger, S.L.; Samet, J.M. Seasonal analyses of air pollution and mortality in 100 US Cities. Am. J. Epidemiol. 2005, 161, 585–594. [Google Scholar] [CrossRef]
- Cichowicz, R.; Wielgosiński, G.; Fetter, W. Dispersion of atmospheric air pollution in summer and winter season. Environ. Monit. Assess. 2017, 189, 605. [Google Scholar] [CrossRef]
- Cheong, K.; Ngiam, J.; Morgan, G.G.; Pek, P.P.; Tan, B.Y.-Q.; Lai, J.W.; Koh, J.M.; Ong, M.; Ho, A.F.W. Acute health impacts of the Southeast Asian transboundary haze problem—A review. Int. J. Environ. Res. Public Health 2019, 16, 3286. [Google Scholar] [CrossRef]
- Auler, A.C.; Cássaro, F.A.M.; da Silva, V.O.; Pires, L.F. Evidence that high temperatures and intermediate relative humidity might favor the spread of COVID-19 in tropical climate: A case study for the most affected Brazilian cities. Sci. Total Environ. 2020, 729, 139090. [Google Scholar] [CrossRef]
- Da Silva, F.L.; Gomes, M.D.A.; Da Silva, A.P.L.; De Sousa, S.C.; De Souza, M.F.S.; Da Silva, G.L.P. Correlation between meteorological factors and COVID-19 infection in the Belem Metropolitan Region. medRxiv 2020. [Google Scholar] [CrossRef]
- Figueiredo, A.M.; Daponte-Codina, A.; Figueiredo, D.C.M.M.; Vianna, R.P.T.; de Lima, K.C.; Gil-García, y.E. Factors associated with the incidence and mortality from COVID-19 in the autonomous communities of Spain. Gac Sanit 2020. [Google Scholar] [CrossRef]
- Neto, R.A.A.; Melo, G.C. Correlation between weather, population size and COVID-19 pandemic: A study of Brazilian capitals. J. Health Boil. Sci. 2020, 8, 1–5. [Google Scholar] [CrossRef]
- Prata, D.N.; Rodrigues, W.; Bermejo, P.H. Temperature significantly changes COVID-19 transmission in (sub) tropical cities of Brazil. Sci. Total Environ. 2020, 729. [Google Scholar] [CrossRef]
- Rodrigues, S.A.; Dal Pai, A.; Grotto, R.M.T.; Sarnighausen, V.C.R. Meteorological variables associations and the occurrence of COVID-19 in the City of São Paulo, Brazil. Revista Ibero-Americana de Ciências Ambientias 2020, 11. Available online: http://www.sustenere.co/index.php/rica/article/view/4311 (accessed on 15 May 2020).
- Velásquez, R.M.A.; Lara, J.V.M. Gaussian approach for probability and correlation between the number of COVID-19 cases and the air pollution in Lima. Urban Clim. 2020, 100664. [Google Scholar] [CrossRef]
- Ward, M.; Xiao, S.; Zhang, Z. The role of climate during the COVID-19 epidemic in New South Wales, Australia. Transbound. Emerg. Dis. 2020. [Google Scholar] [CrossRef]
- Bashir, M.F.; Ma, B.; Bilal, K.B.; Bashir, M.A.; Tan, D.; Bashir, M. Correlation between climate indicators and COVID-19 pandemic in New York, USA. Sci. Total Environ. 2020, 728. [Google Scholar] [CrossRef]
- Tosepu, R.; Gunawan, J.; Effendy, D.S.; Ahmad, L.O.A.I.; Lestari, H.; Bahar, H.; Asfian, P. Correlation between weather and Covid-19 pandemic in Jakarta, Indonesia. Sci. Total Environ. 2020, 725, 138436. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Raghuwanshi, G.S.; Chanda, A. Effect of weather on COVID-19 spread in the US: A prediction model for India in 2020. Sci. Total Environ. 2020, 728. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhou, J.; Yao, J.; Zhang, X.; Li, L.; Xu, X.; He, X.; Wang, B.; Fu, S.; Niu, T.; et al. Impact of meteorological factors on the COVID-19 transmission: A multi-city study in China. Sci. Total Environ. 2020, 726, 138513. [Google Scholar] [CrossRef] [PubMed]
- Poirier, C.; Luo, W.; Majumder, M.S.; Liu, D.; Mandl, K.; Mooring, T.; Santillana, M. The role of environmental factors on transmission rates of the COVID-19 Outbreak: An initial assessment in two spatial scales. SSRN Electron. J. 2020. [Google Scholar] [CrossRef]
- Dee, D.P.; Uppala, S.M.; Simmons, A.J.; Berrisford, P.; Poli, P.; Kobayashi, S.; Andrae, U.; Balmaseda, M.A.; Balsamo, G.; Bauer, D.P.; et al. The ERA-interim reanalysis: Configuration and performance of the data assimilation system. Q. J. R. Meteorol. Soc. 2011, 137, 553–597. [Google Scholar] [CrossRef]
- Jahangiri, M.; Jahangiri, M.; Najafgholipour, M. The sensitivity and specificity analyses of ambient temperature and population size on the transmission rate of the novel coronavirus (COVID-19) in different provinces in Iran. Sci. Total Environ. 2020, 728. [Google Scholar] [CrossRef]
- Gao, M.; Zhou, Q.; Zhang, S.; Yung, K.K.L.; Guo, Y. Non-linear modulation of COVID-19 transmission by climate conditions. SSRN Electron. J. 2020. [Google Scholar] [CrossRef]
- Guo, X.-J.; Zhang, H.; Zeng, Y.-P. Transmissibility of COVID-19 and its association with temperature and humidity. Eur. PMC 2020. [Google Scholar] [CrossRef]
- Gupta, A.; Gharehgozli, A. Developing a machine learning framework to determine the spread of COVID-19. SSRN Electron. J. 2020. [Google Scholar] [CrossRef]
- Jamil, T.; Alam, I.S.; Gojobori, T.; Duarte, C.M. No evidence for temperature-dependence of the COVID-19 epidemic. medRxiv 2020. [Google Scholar] [CrossRef]
- Jia, J.; Ding, J.; Liu, S.; Liao, G.; Li, J.; Duan, B.; Wang, G.; Zhang, R. No evidence for temperature-dependence of the covid-19 epidemic. medRxiv 2020. [Google Scholar] [CrossRef]
- Jebril, N. Predict the transmission of COVID-19 under the effect of air temperature and relative humidity over the year in Baghdad, Iraq. SSRN Electron. J. 2020. [Google Scholar] [CrossRef]
- Luo, W.; Majumder, M.S.; Liu, D.; Poirier, C.; Mandl, K.D.; Lipsitch, M.; Santillana, M. The role of absolute humidity on transmission rates of the COVID-19 outbreak. medRxiv 2020. [Google Scholar] [CrossRef]
- Wang, M.; Jiang, A.; Gong, L.; Luo, L.; Guo, W.; Li, C.; Zheng, J.; Li, C.; Yang, B.; Zeng, J.; et al. Temperature significant change COVID-19 Transmission in 429 cities. medRxiv 2020. [Google Scholar] [CrossRef]
- Carleton, T.; Cornetet, J.; Huybers, P.; Meng, K.; Proctor, J. Ultraviolet radiation decreases COVID-19 growth rates: Global causal estimates and seasonal implications. SSRN Electron. J. 2020. [Google Scholar] [CrossRef]
- Caspi, G.; Shalit, U.; Kristensen, S.L.; Aronson, D.; Caspi, L.; Rossenberg, O.; Shina, A.; Caspi, O. Climate effect on COVID-19 spread rate: An online surveillance tool. medRxiv 2020. [Google Scholar] [CrossRef]
- Ficetola, G.F.; Rubolini, D. Climate affects global patterns of COVID-19 early outbreak dynamics. medRxiv 2020. [Google Scholar] [CrossRef]
- Merow, C.; Urban, M.C. Seasonality and uncertainty in COVID-19 growth rates. medRxiv 2020. [Google Scholar] [CrossRef]
- Notari, A. Temperature dependence of COVID-19 transmission. arxiv, 2020; arXiv:2003.12417. [Google Scholar]
- Oliveiros, B.; Caramelo, L.; Ferreira, N.C.; Caramelo, F. Role of temperature and humidity in the modulation of the doubling time of COVID-19 cases. medRxiv 2020. [Google Scholar] [CrossRef]
- Sahafizadeh, E.; Sartoli, S. High temperature has no impact on the reproduction number and new cases of COVID-19 in Bushehr, Iran. medRxiv 2020. [Google Scholar] [CrossRef]
- Skutsch, M.; Dobler, C.; McCall, M.B.B.; Ghilardi, A.; Salinas, M.; McCall, M.K.; Sanchez, F. The association of UV with rates of COVID-19 transmission and deaths in Mexico: The possible mediating role of vitamin D. medRxiv 2020. [Google Scholar] [CrossRef]
- Kotsiou, O.S.; Kotsios, V.S.; Lampropoulos, I.; Zidros, T.; Zarogiannis, S.G.; Gourgoulianis, K.I. High temperature slows coronavirus disease 2019 transmission rate: A within and among country analysis. Res. Sq. 2020. [Google Scholar] [CrossRef]
- Leung, N.Y.; Bulterys, M.A.; Bulterys, P.L. Predictors of COVID-19 incidence, mortality, and epidemic growth rate at the country level. medRxiv 2020. [Google Scholar] [CrossRef]
- Lolli, S.; Chen, Y.-C.; Wang, S.-H.; Vivone, G. Impact of meteorology and air pollution on Covid-19 pandemic transmission in Lombardy region, Northern Italy. Res. Sq. 2020. [Google Scholar] [CrossRef]
- Chen, B.; Liang, H.; Yuan, X.; Hu, Y.; Xu, M.; Zhao, Y.; Zhang, B.; Tian, F.; Zhu, X. Roles of meteorological conditions in COVID-19 transmission on a worldwide scale. medRxiv 2020. [Google Scholar] [CrossRef]
- Lin, J.; Huang, W.; Wen, M.; Li, D.; Ma, S.; Hua, J.; Hu, H.; Yin, S.; Qian, Y.; Chen, P.; et al. Containing the spread of coronavirus disease 2019 (COVID-19): Meteorological factors and control strategies. Sci. Total Environ. 2020, 744, 140935. [Google Scholar] [CrossRef]
- Rahman, A.; Hossain, G.; Singha, A.C.; Islam, S.; Islam, A. A retrospective analysis of influence of environmental/air temperature and relative humidity on SARS-CoV-2 outbreak. PrePrints 2020. [Google Scholar] [CrossRef]
- Wu, Y.; Jing, W.; Liu, J.; Ma, Q.; Yuan, J.; Wang, Y.; Du, M.; Liu, M. Effects of temperature and humidity on the daily new cases and new deaths of COVID-19 in 166 countries. Sci. Total Environ. 2020, 729. [Google Scholar] [CrossRef]
- Bannister-Tyrrell, M.; Meyer, A.; Faverjon, C.; Cameron, A. Preliminary evidence that higher temperatures are associated with lower incidence of COVID-19, for cases reported globally up to 29th February 2020. medRxiv 2020. [Google Scholar] [CrossRef]
- Tobías, A.; Molina, T. Is temperature reducing the transmission of COVID-19? Environ. Res. 2020, 186. [Google Scholar] [CrossRef]
- Wang, J.; Tang, K.; Feng, K.; Lv, W. High Temperature and high humidity reduce the transmission of COVID-19. SSRN Electron. J. 2020. [Google Scholar] [CrossRef]
- Xu, H.; Yan, C.; Fu, Q.; Xiao, K.; Yu, Y.; Han, D.; Wang, W.; Cheng, J. Possible environmental effects on the spread of COVID-19 in China. Sci. Total Environ. 2020, 731. [Google Scholar] [CrossRef]
- Qi, H.; Xiao, S.; Shi, R.; Ward, M.P.; Chen, Y.; Tu, W.; Su, Q.; Wang, W.; Wang, X.; Zhang, Z. COVID-19 transmission in Mainland China is associated with temperature and humidity: A time-series analysis. Sci. Total Environ. 2020, 728, 138778. [Google Scholar] [CrossRef] [PubMed]
- Ujiie, M.; Tsuzuki, S.; Ohmagari, N. Effect of temperature on the infectivity of COVID-19. Int. J. Infect. Dis. 2020, 95, 301–303. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D.J. Weather, Social Distancing, and the Spread of COVID-19; Federal Reserve Bank of San Francisco: San Francisco, CA, USA, 2020; Working Paper 2020-23. [Google Scholar] [CrossRef]
- Ahmadi, M.; Sharifi, A.; Dorosti, S.; Ghoushchi, S.J.; Ghanbari, N. Investigation of effective climatology parameters on COVID-19 outbreak in Iran. Sci. Total Environ. 2020, 729. [Google Scholar] [CrossRef] [PubMed]
- Alipio, M. Do latitude and ozone concentration predict COVID-2019 cases in 34 countries? SSRN Electron. J. 2020. [Google Scholar] [CrossRef]
- Bhattacharjee, S. Statistical investigation of relationship between spread of coronavirus disease (COVID-19) and environmental factors based on study of four mostly affected places of China and five mostly affected places of Italy. arxiv, 2020; arXiv:2003.11277. [Google Scholar]
- Briz-Redón, Á.; Serrano-Aroca, Á. A spatio-temporal analysis for exploring the effect of temperature on COVID-19 early evolution in Spain. Sci. Total Environ. 2020, 728. [Google Scholar] [CrossRef] [PubMed]
- Sobral, M.F.F.; Duarte, G.B.; da Penha Sobral, A.I.G.; Marinho, M.L.M.; de Souza Melo, A. Association between climate variables and global transmission of SARS-CoV-2. Sci. Total Environ. 2020, 729. [Google Scholar] [CrossRef]
- Mollalo, A.; Vahedi, B.; Rivera, K.M. GIS-based spatial modelling of COVID-19 incidence rate in the continental United States. Sci. Total Environ. 2020, 728. [Google Scholar] [CrossRef] [PubMed]
- Pirouz, B.; Haghshenas, S.S.; Pirouz, B.; Haghshenas, S.S.; Piro, P. Development of an assessment method for investigating the impact of climate and urban parameters in confirmed cases of COVID-19: A new challenge in sustainable development. Int. J. Environ. Res. Public Health 2020, 17, 2801. [Google Scholar] [CrossRef]
- Arias-Reyes, C.; Zubieta-DeUrioste, N.; Poma-Machicao, L.; Aliaga-Raudan, F.; Carvajal-Rodriguez, F.; Dutschmann, M.; Schneider-Gasser, E.M.; Zubieta-Calleja, G.; Soliz, J. Does the pathogenesis of SARS-CoV-2 virus decrease at high-altitude? Respir. Physiol. Neurobiol. 2020, 277, 103443. [Google Scholar] [CrossRef] [PubMed]
- Bukhari, Q.; Jameel, Y. Will coronavirus pandemic diminish by summer? SSRN Electron. J. 2020. [Google Scholar] [CrossRef]
- Gunthe, S.S.; Swain, B.; Patra, S.S.; Amte, A. On the global trends and spread of the COVID-19 outbreak: Preliminary assessment of the potential relation between location-specific temperature and UV index. J. Public Health 2020, 24, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, N.; Fareed, Z.; Shahzad, F.; He, X.; Shahzad, U.; Lina, M. Nexus between COVID-19, temperature and exchange rate in Wuhan city: New findings from partial and multiple wavelet coherence. Sci. Total Environ. 2020, 729. [Google Scholar] [CrossRef] [PubMed]
- Araujo, M.B.; Naimi, B. Spread of SARS-CoV-2 Coronavirus likely to be constrained by climate. medRxiv 2020. [Google Scholar] [CrossRef]
- Coro, G. A global-scale ecological niche model to predict SARS-CoV-2 coronavirus infection rate. Ecol. Model. 2020, 143, 109187. [Google Scholar] [CrossRef]
- Carlson, C.J.; Chipperfield, J.; Benito, B.M.; Telford, R.J.; O’Hara, R.B. Species distribution models are inappropriate for COVID-19. Nat. Ecol. Evol. 2020, 4, 770–771. [Google Scholar] [CrossRef]
- Baker, R.E.; Yang, W.; Vecchi, G.A.; Metcalf, C.J.E.; Grenfell, B.T. Susceptible supply limits the role of climate in the early SARS-CoV-2 pandemic. Science 2020, 369, 315–319. [Google Scholar] [CrossRef]
- Wan, X.; Cheng, C.; Zhang, Z. Early transmission of COVID-19 has an optimal temperature but late transmission decreases in warm climate. medRxiv 2020. [Google Scholar] [CrossRef]
- Kwon, D. How swamped preprint servers are blocking bad coronavirus research. Nature 2020, 581, 130–131. [Google Scholar] [CrossRef] [PubMed]
- Beran, D.; Byass, P.; Gbakima, A.; Kahn, K.; Sankoh, O.; Tollman, S.; Witham, M.; Davies, J.I. Research capacity building—Obligations for global health partners. Lancet Glob. Health 2017, 5, e567–e568. [Google Scholar] [CrossRef]
- Morawska, L.; Cao, J. Airborne transmission of SARS-CoV-2: The world should face the reality. Environ. Int. 2020, 139, 105730. [Google Scholar] [CrossRef] [PubMed]
- COVID-19 Health System Response Monitor. Available online: https://www.covid19healthsystem.org (accessed on 15 May 2020).
- Escobar, L.E.; Molina-Cruz, A.; Barillas-Mury, C. BCG vaccine protection from severe coronavirus disease 2019 (COVID-19). Proc. Natl. Acad. Sci. USA 2020, 117, 202008410. [Google Scholar] [CrossRef]
- Pei, S.; Kandula, S.; Shaman, J. Differential Effects of Intervention Timing on COVID-19 Spread in the United States. medRxiv 2020. [Google Scholar] [CrossRef]
- Gasparrini, A.; Guo, Y.; Hashizume, M.; Lavigne, E.; Zanobetti, A.; Schwartz, J.; Tobías, A.; Tong, S.; Rocklöv, J.; Forsberg, B.; et al. Mortality risk attributable to high and low ambient temperature: A multicountry observational study. Lancet 2015, 386, 369–375. [Google Scholar] [CrossRef]
- Liu, C.; Chen, R.; Sera, F.; Vicedo-Cabrera, A.M.; Guo, Y.; Tong, S.; Coelho, M.S.; Saldiva, P.H.; Lavigne, E.; Matus, P.; et al. Ambient particulate air pollution and daily mortality in 652 cities. N. Engl. J. Med. 2019, 381, 705–715. [Google Scholar] [CrossRef]
- Gardiner, J.C.; Luo, Z.; Roman, L.A. Fixed effects, random effects and GEE: What are the differences? Stat. Med. 2009, 28, 221–239. [Google Scholar] [CrossRef]
- WHO—A Coordinated Global Research Roadmap: 2019 Novel Coronavirus. Available online: https://www.who.int/blueprint/priority-diseases/key-action/Coronavirus_Roadmap_V9.pdf (accessed on 15 May 2020).
- Shumake-Guillemot, J. (WHO/WMO Climate and Health Joint Office, Geneva, Switzerland). Personal communication, May 2020.
| Theoretical Basis | Advantages | Disadvantages | Examples |
|---|---|---|---|
| (a) Simple extrapolation of recent trends—linear or exponential | Few assumptions, nearly theory-free, easily updated as new data come in | Sensitive to data quality; unrealistic for projections more than a few timesteps into the future | Systrom and Vladeck [43] |
| (b) Phenomenological or parameterised models—fit a curve of predetermined form to cumulative case data | Few assumptions, good for explaining large-scale, multi-month patterns like ‘flattening the curve’ | Inflexible and unresponsive to changes in circumstances, such as social distancing policy | Della Morte et al. [44], Roosa et al. [45] |
| (c) Compartment models (e.g., SIR, SEIR) | Classical epidemiological approach, semi-mechanistic | Relatively many parameters that are highly uncertain initially; needs lots of good data | Anastassopoulou et al. [46] |
| (d) Machine learning | Few assumptions other than data homogeneity and stationarity | Requires very large case datasets to be effective; no explicit mechanism | Ardabili et al. [47], Pinter et al. [48] |
| (e) Agent-based models—every person in a population is modelled | Allows a rich set of interpersonal interactions despite simple rules | Data and computationally intensive | Cuevas [49] |
| Relative Humidity | 20 °C | 6 °C | ||||
|---|---|---|---|---|---|---|
| 15 min | 24 h | 72 h | 6 days | 15 min | 24 h | |
| 30% | 87% | 65% | >50% | n.d. | 91% | 65% |
| 50% | 90.9% | 75% | >50% | 20% | 96.5% | 80% |
| 80% | 55% | 3% | 0% | n.d. | 104.8% | 86% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smit, A.J.; Fitchett, J.M.; Engelbrecht, F.A.; Scholes, R.J.; Dzhivhuho, G.; Sweijd, N.A. Winter Is Coming: A Southern Hemisphere Perspective of the Environmental Drivers of SARS-CoV-2 and the Potential Seasonality of COVID-19. Int. J. Environ. Res. Public Health 2020, 17, 5634. https://doi.org/10.3390/ijerph17165634
Smit AJ, Fitchett JM, Engelbrecht FA, Scholes RJ, Dzhivhuho G, Sweijd NA. Winter Is Coming: A Southern Hemisphere Perspective of the Environmental Drivers of SARS-CoV-2 and the Potential Seasonality of COVID-19. International Journal of Environmental Research and Public Health. 2020; 17(16):5634. https://doi.org/10.3390/ijerph17165634
Chicago/Turabian StyleSmit, Albertus J., Jennifer M. Fitchett, Francois A. Engelbrecht, Robert J. Scholes, Godfrey Dzhivhuho, and Neville A. Sweijd. 2020. "Winter Is Coming: A Southern Hemisphere Perspective of the Environmental Drivers of SARS-CoV-2 and the Potential Seasonality of COVID-19" International Journal of Environmental Research and Public Health 17, no. 16: 5634. https://doi.org/10.3390/ijerph17165634
APA StyleSmit, A. J., Fitchett, J. M., Engelbrecht, F. A., Scholes, R. J., Dzhivhuho, G., & Sweijd, N. A. (2020). Winter Is Coming: A Southern Hemisphere Perspective of the Environmental Drivers of SARS-CoV-2 and the Potential Seasonality of COVID-19. International Journal of Environmental Research and Public Health, 17(16), 5634. https://doi.org/10.3390/ijerph17165634






